Influence of Magnetite Nanoparticles Shape and Spontaneous Surface Oxidation on the Electron Transport Mechanism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Magnetite Nanoparticles in a Hydrophobic–Hydrophilic Environment
2.2. Materials Characterization
3. Results and Discussion
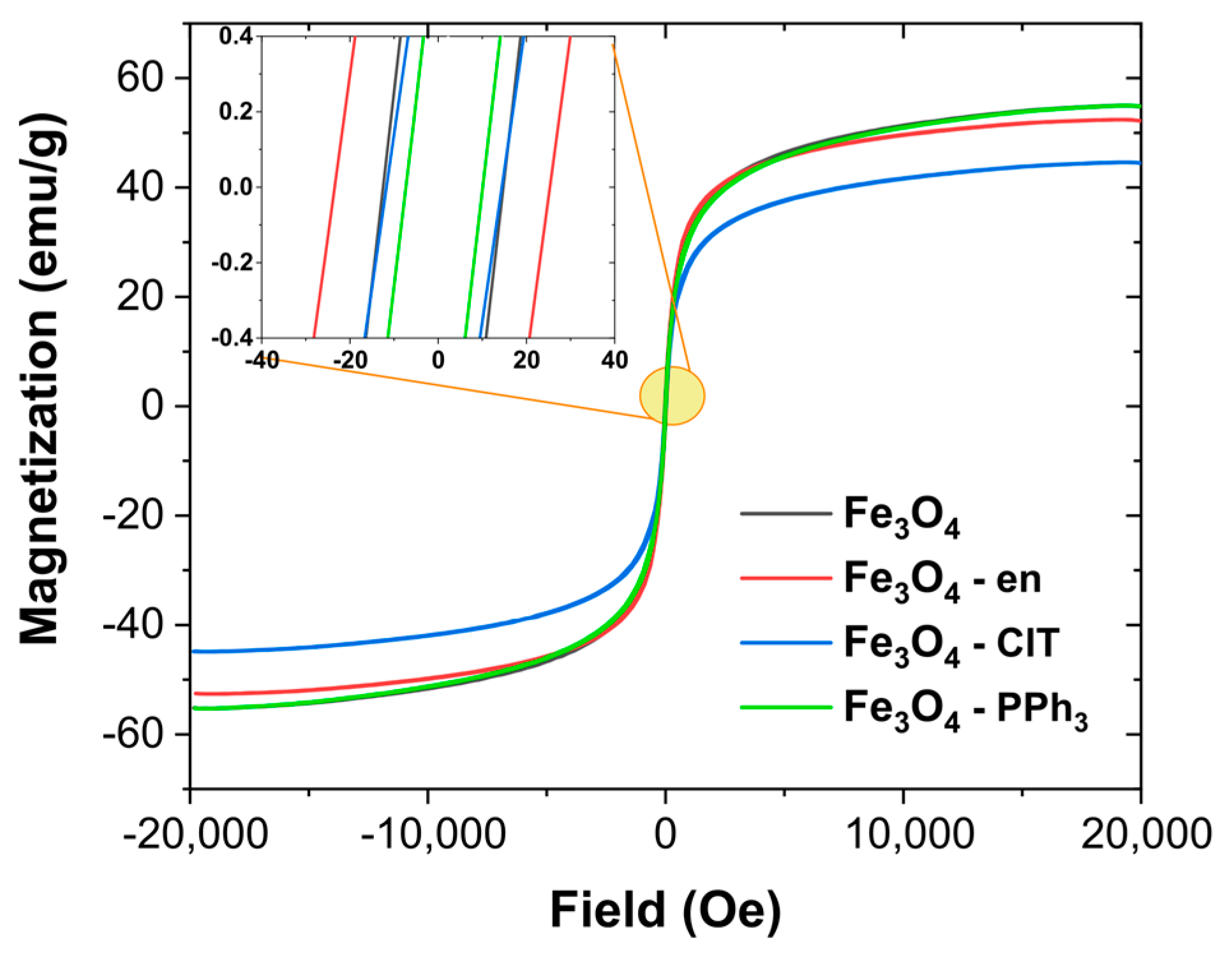
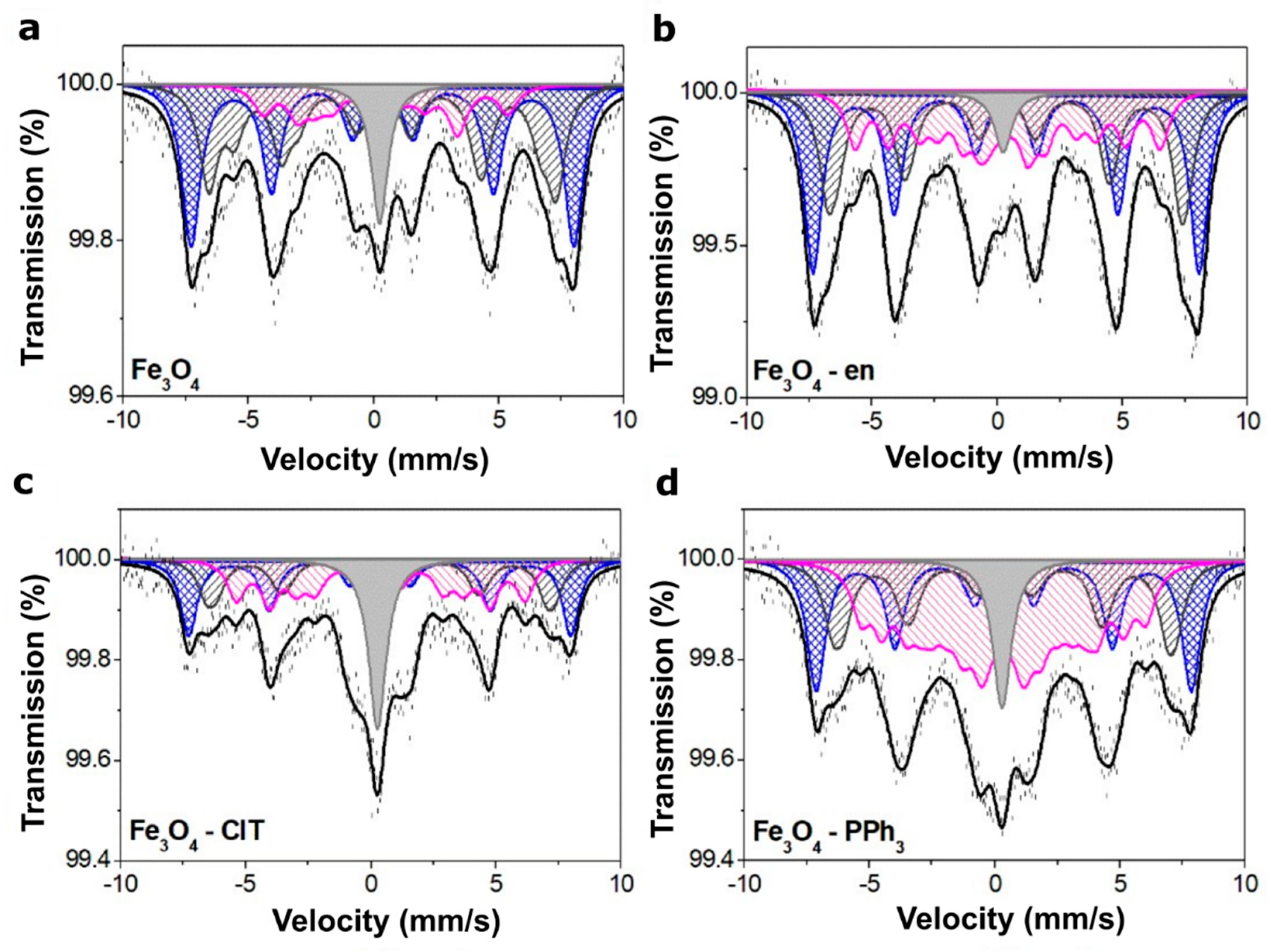
3.1. Structural Analysis and Magnetic Properties of Magnetite Nanoparticles
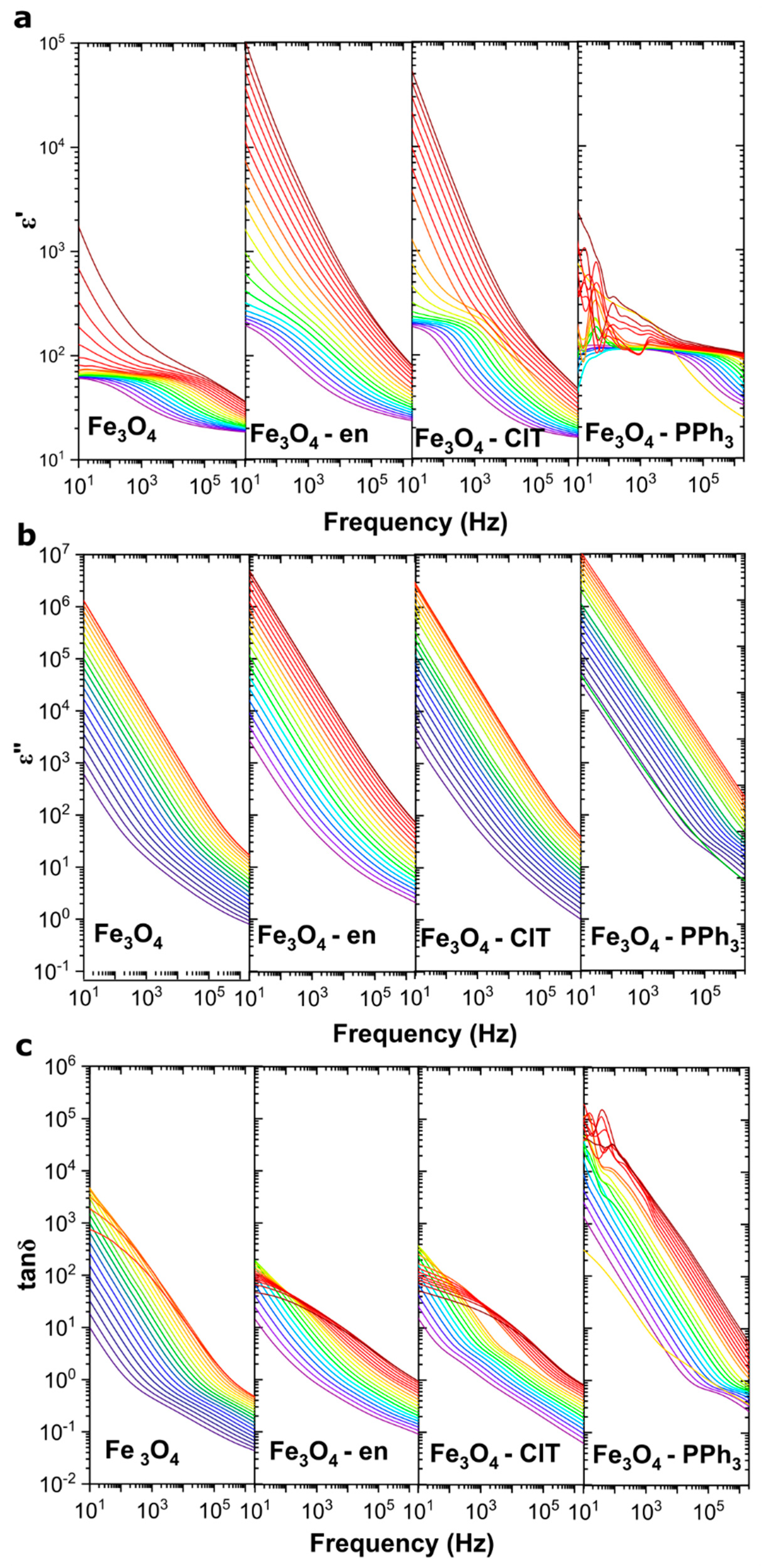
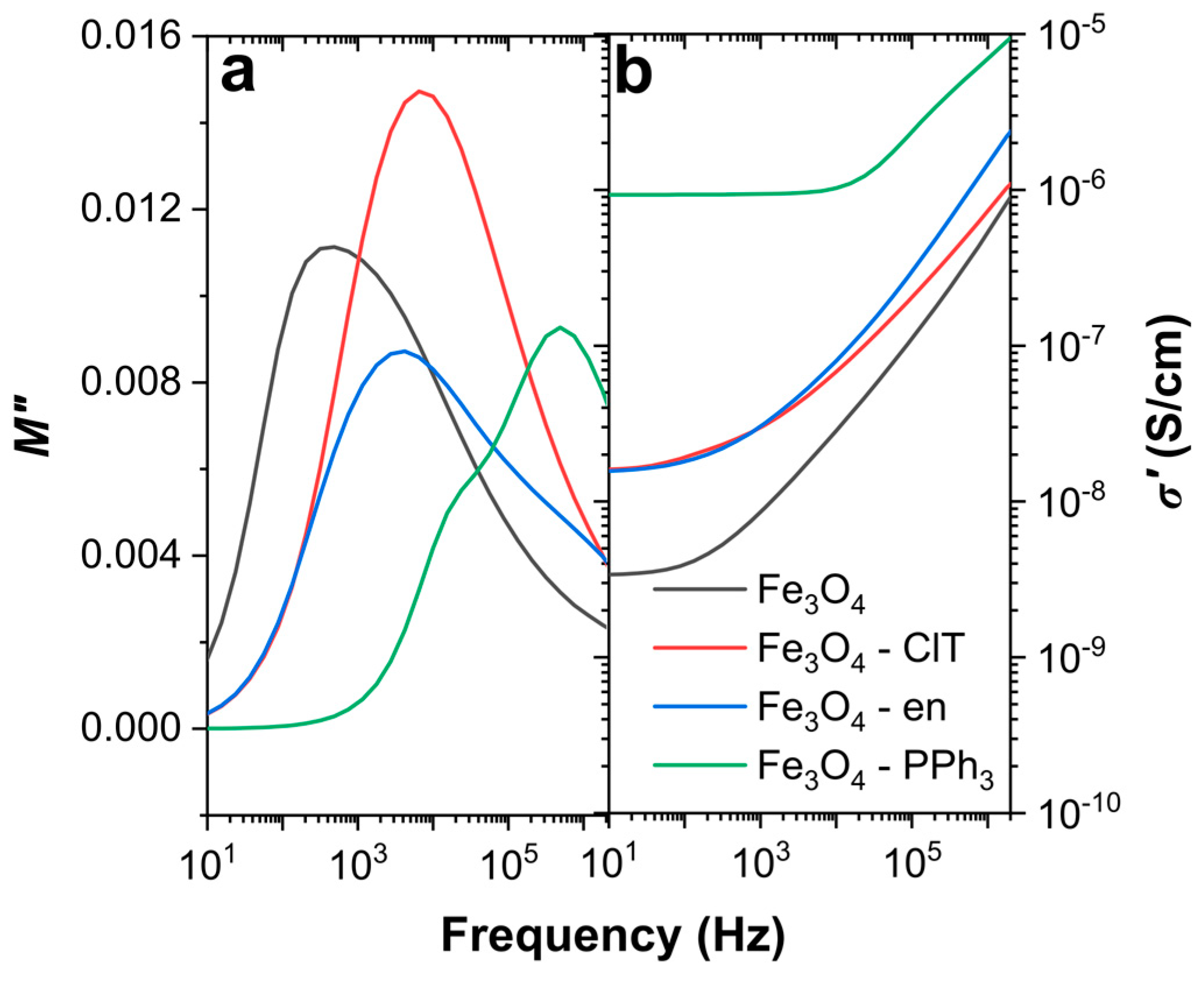
3.2. Dielectric Properties of Magnetite Nanoparticles with Different Sizes, Shapes, and Maghemite Contents
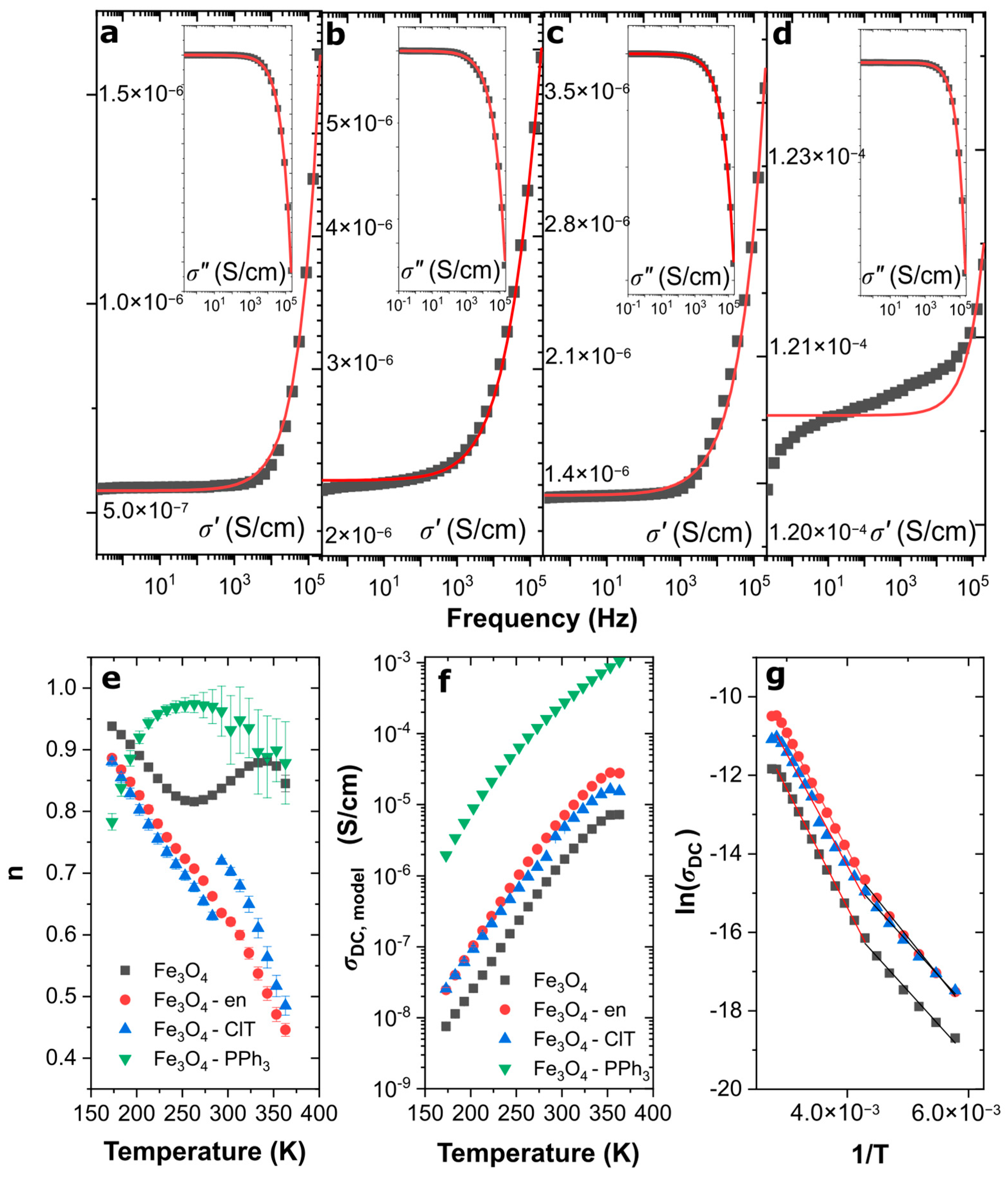
3.3. Electrical Conductivity of Magnetite Nanoparticles
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kumar, R.; Mondal, K.; Panda, P.K.; Kaushik, A.; Abolhassani, R.; Ahuja, R.; Rubahn, H.G.; Mishra, Y.K. Core-shell nanostructures: Perspectives towards drug delivery applications. J. Mater. Chem. B 2020, 8, 8992–9027. [Google Scholar] [CrossRef]
- Tan, L.; Wang, F.; Zhang, P.; Suzuki, Y.; Wu, Y.; Chen, J.; Yang, G.; Tsubaki, N. Design of a core-shell catalyst: An effective strategy for suppressing side reactions in syngas for direct selective conversion to light olefins. Chem. Sci. 2020, 11, 4097–4105. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N.; Sinha Ray, S. Synthesis and functionalization of nanomaterials. In Processing of Polymer-Based Nanocomposites; Springer Series in Materials Science; Sinha Ray, S., Ed.; Springer: Berlin/Heidelberg, Germany, 2018; Volume 277. [Google Scholar]
- Natarajan, S.; Harini, K.; Gajula, G.P.; Sarmento, B.; Neves-Petersen, M.T.; Thiagarajan, V. Multifunctional magnetic iron oxide nanoparticles: Diverse synthetic approaches, surface modifications, cytotoxicity towards biomedical and industrial applications. BMC Mater. 2019, 1, 2. [Google Scholar] [CrossRef]
- Majhi, S.M.; Naik, G.K.; Lee, H.J.; Song, H.G.; Lee, C.R.; Lee, I.H.; Yu, Y.T. Au@NiO core-shell nanoparticles as a p-type gas sensor: Novel synthesis, characterization, and their gas sensing properties with sensing mechanism. Sens. Actuators B Chem. 2018, 268, 223–231. [Google Scholar] [CrossRef]
- Qu, F.; Liu, J.; Wang, Y.; Wen, S.; Chen, Y.; Li, X.; Ruan, S. Hierarchical Fe3O4@Co3O4 core-shell microspheres: Preparation and acetone sensing properties. Sens. Actuators B Chem. 2014, 199, 346–353. [Google Scholar] [CrossRef]
- Sappino, C.; Primitivo, L.; De Angelis, M.; Domenici, M.O.; Mastrodonato, A.; Romdan, I.B.; Tatangelo, C.; Suber, L.; Pilloni, L.; Ricelli, A.; et al. Functionalized Magnetic Nanoparticles as Catalysts for Enantioselective Henry Reaction. ACS Omega 2019, 4, 21809–21817. [Google Scholar] [CrossRef]
- Ingle, A.P.; Philippini, R.R.; de Souza Melo, Y.C.; da Silva, S.S. Acid-functionalized magnetic nanocatalysts mediated pretreatment of sugarcane straw: An eco-friendly and cost-effective approach. Cellulose 2020, 27, 7067–7078. [Google Scholar] [CrossRef]
- Xiong, F.; Hu, K.; Yu, H.; Zhou, L.; Song, L.; Zhang, Y.; Shan, X.; Liu, J.; Gu, N. A Functional Iron Oxide Nanoparticles Modified with PLA-PEG-DG as Tumor-Targeted MRI Contrast Agent. Pharm. Res. 2017, 34, 1683–1692. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.; Xia, Y.; Allan, A.; Rohani, S.; Gillies, E.R. Curcumin-loaded, folic acid-functionalized magnetite particles for targeted drug delivery. RSC Adv. 2015, 5, 37521–37532. [Google Scholar] [CrossRef]
- Radoń, A.; Łoński, S.; Kądziołka-Gaweł, M.; Gębara, P.; Lis, M.; Łukowiec, D.; Babilas, R. Influence of magnetite nanoparticles surface dissolution, stabilization and functionalization by malonic acid on the catalytic activity, magnetic and electrical properties. Colloids Surf. A Physicochem. Eng. Asp. 2020, 607, 125446. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef] [Green Version]
- Schwaminger, S.P.; Bauer, D.; Fraga-García, P.; Wagner, F.E.; Berensmeier, S. Oxidation of magnetite nanoparticles: Impact on surface and crystal properties. CrystEngComm 2017, 19, 246–255. [Google Scholar] [CrossRef] [Green Version]
- Winsett, J.; Moilanen, A.; Paudel, K.; Kamali, S.; Ding, K.; Cribb, W.; Seifu, D.; Neupane, S. Quantitative determination of magnetite and maghemite in iron oxide nanoparticles using Mössbauer spectroscopy. SN Appl. Sci. 2019, 1, 1636. [Google Scholar] [CrossRef] [Green Version]
- Morales, I.; Costo, R.; Mille, N.; da Silva, G.B.; Carrey, J.; Hernando, A.; de la Presa, P. High frequency hysteresis losses on γ-Fe2O3 and Fe3O4: Susceptibility as a magnetic stamp for chain formation. Nanomaterials 2018, 8, 970. [Google Scholar] [CrossRef] [Green Version]
- Radoń, A.; Łukowiec, D.; Kremzer, M.; Mikuła, J.; Włodarczyk, P. Electrical conduction mechanism and dielectric properties of spherical shaped Fe3O4 nanoparticles synthesized by co-precipitation method. Materials (Basel) 2018, 11, 735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radoń, A.; Łukowiec, D.; Włodarczyk, P. Ultraslow electron-phonon scattering and polaron formation in magnetite. J. Mater. 2021, in press. [Google Scholar]
- Truncik, C.J.S.; Huttema, W.A.; Turner, P.J.; Özcan, S.; Murphy, N.C.; Carrière, P.R.; Thewalt, E.; Morse, K.J.; Koenig, A.J.; Sarrao, J.L.; et al. Nodal quasiparticle dynamics in the heavy fermion superconductor CeCoIn5 revealed by precision microwave spectroscopy. Nat. Commun. 2013, 4, 2477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotnala, R.K.; Ahmad, S.; Ahmed, A.S.; Shah, J.; Azam, A. Investigation of structural, dielectric, and magnetic properties of hard and soft mixed ferrite composites. Proc. J. Appl. Phys. 2012, 112, 054323. [Google Scholar] [CrossRef]
- Jotania, R.B.; Nandotaria, R.A.; Chauhan, C.C.; Hashim, M.; Singh Meena, S.; Shirsath, S.E. Structural phases and Maxwell-Wagner relaxation in magnetically soft-ZnFe2O4 and hard-Sr2Cu2Fe12O22 nanocomposites. Ceram. Int. 2016, 42, 2289–2298. [Google Scholar] [CrossRef]
- Siregar, J.; Septiani, N.L.W.; Abrori, S.A.; Sebayang, K.; Irzaman; Fahmi, M.Z.; Humaidi, S.; Sembiring, T.; Sembiring, K.; Yuliarto, B. Review—A Pollutant Gas Sensor Based On Fe3O4 Nanostructures: A Review. J. Electrochem. Soc. 2021, 168, 027510. [Google Scholar] [CrossRef]
- Radoń, A.; Hawełek, Ł.; Łukowiec, D.; Kubacki, J.; Włodarczyk, P. Dielectric and electromagnetic interference shielding properties of high entropy (Zn,Fe,Ni,Mg,Cd)Fe2O4 ferrite. Sci. Rep. 2019, 9, 20078. [Google Scholar] [CrossRef]
- Radoń, A.; Łukowiec, D.; Włodarczyk, P. Microwave absorption by dextrin-magnetite nanocomposite in frequencies below 2.5 GHz: Role of magnetite content, shape and temperature on magneto-dielectric properties. Mater. Des. 2020, 193, 108860. [Google Scholar] [CrossRef]
- Debnath, S.; Das, R. Cobalt doping on nickel ferrite nanocrystals enhances the micro-structural and magnetic properties: Shows a correlation between them. J. Alloys Compd. 2021, 852, 156884. [Google Scholar] [CrossRef]
- Nath, D.; Singh, F.; Das, R. X-ray diffraction analysis by Williamson-Hall, Halder-Wagner and size-strain plot methods of CdSe nanoparticles- a comparative study. Mater. Chem. Phys. 2020, 239, 122021. [Google Scholar] [CrossRef]
- Yuan, K.; Lee, S.S.; Cha, W.; Ulvestad, A.; Kim, H.; Abdilla, B.; Sturchio, N.C.; Fenter, P. Oxidation induced strain and defects in magnetite crystals. Nat. Commun. 2019, 10, 703. [Google Scholar] [CrossRef]
- Sneed, B.T.; Young, A.P.; Tsung, C.K. Building up strain in colloidal metal nanoparticle catalysts. Nanoscale 2015, 7, 12248–12265. [Google Scholar] [CrossRef]
- Dheyab, M.A.; Aziz, A.A.; Jameel, M.S.; Noqta, O.A.; Khaniabadi, P.M.; Mehrdel, B. Simple rapid stabilization method through citric acid modification for magnetite nanoparticles. Sci. Rep. 2020, 10, 10793. [Google Scholar] [CrossRef] [PubMed]
- Radoń, A.; Drygała, A.; Hawełek, Ł.; Łukowiec, D. Structure and optical properties of Fe3O4 nanoparticles synthesized by co-precipitation method with different organic modifiers. Mater. Charact. 2017, 131, 148–156. [Google Scholar] [CrossRef]
- Chernyshova, I.V.; Hochella, M.F.; Madden, A.S. Size-dependent structural transformations of hematite nanoparticles. 1. Phase transition. Phys. Chem. Chem. Phys. 2007, 9, 1736–1750. [Google Scholar] [CrossRef] [PubMed]
- Shipilin, M.A.; Zakharova, I.N.; Shipilin, A.M.; Bachurin, V.I. Mössbauer studies of magnetite nanoparticles. J. Surf. Investig. X-ray Synchrotron Neutron Tech. 2014, 8, 557–561. [Google Scholar] [CrossRef]
- Bonkerud, J.; Zimmermann, C.; Weiser, P.M.; Vines, L.; Monakhov, E.V. On the permittivity of titanium dioxide. Sci. Rep. 2021, 11, 12443. [Google Scholar] [CrossRef]
- Woodward, W.H.H. Broadband Dielectric Spectroscopy—A Practical Guide. In ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2021. [Google Scholar]
- Kooti, M.; Sedeh, A.N.; Gheisari, K.; Figuerola, A. Synthesis, characterization, and performance of nanocomposites containing reduced graphene oxide, polyaniline, and cobalt ferrite. Phys. B Condens. Matter 2021, 612, 412974. [Google Scholar] [CrossRef]
- Boonlakhorn, J.; Manyam, J.; Krongsuk, S.; Thongbai, P.; Srepusharawoot, P. Enhanced dielectric properties with a significantly reduced loss tangent in (Mg2+, Al3+) co-doped CaCu3Ti4O12 ceramics: DFT and experimental investigations. RSC Adv. 2021, 11, 25038–25046. [Google Scholar] [CrossRef]
- Hussain, M.; Khan, R.; Khan, T.Z.; Khan, G.; Khattak, S.; Rahman, M.U.; Ali, S.; Iqbal, Z.; Safeen, K. Dielectric and magnetic properties of cobalt doped γ-Fe2O3 nanoparticles. J. Mater. Sci. Mater. Electron. 2019, 30, 13698–13707. [Google Scholar] [CrossRef]
- Türkay, S.; Tataroğlu, A. Complex dielectric permittivity, electric modulus and electrical conductivity analysis of Au/Si3N4/p-GaAs (MOS) capacitor. J. Mater. Sci. Mater. Electron. 2021, 31, 11418–11425. [Google Scholar] [CrossRef]
- Altındal, Ş.; Ulusoy, M.; Özçelik, S.; Azizian-Kalandaragh, Y. On the frequency-dependent complex-dielectric, complex-electric modulus and conductivity in Au/(NiS:PVP)/n-Si structures. J. Mater. Sci. Mater. Electron. 2021, 32, 20071–20081. [Google Scholar] [CrossRef]
- Megdiche, M.; Perrin-Pellegrino, C.; Gargouri, M. Conduction mechanism study by overlapping large-polaron tunnelling model in SrNiP2O7 ceramic compound. J. Alloys Compd. 2014, 584, 209–215. [Google Scholar] [CrossRef]
- Jebli, M.; Rayssi, C.; Dhahri, J.; Ben Henda, M.; Belmabrouk, H.; Bajahzar, A. Structural and morphological studies, and temperature/frequency dependence of electrical conductivity of Ba0.97La0.02Ti1−xNb4x/5O3perovskite ceramics. RSC Adv. 2021, 11, 23664–23678. [Google Scholar] [CrossRef]
- Liu, H.; Di Valentin, C. Band Gap in Magnetite above Verwey Temperature Induced by Symmetry Breaking. J. Phys. Chem. C 2017, 121, 25736–25742. [Google Scholar] [CrossRef]
- Cuenca, J.A.; Bugler, K.; Taylor, S.; Morgan, D.; Williams, P.; Bauer, J.; Porch, A. Study of the magnetite to maghemite transition using microwave permittivity and permeability measurements. J. Phys. Condens. Matter 2016, 28, 106002. [Google Scholar] [CrossRef] [Green Version]
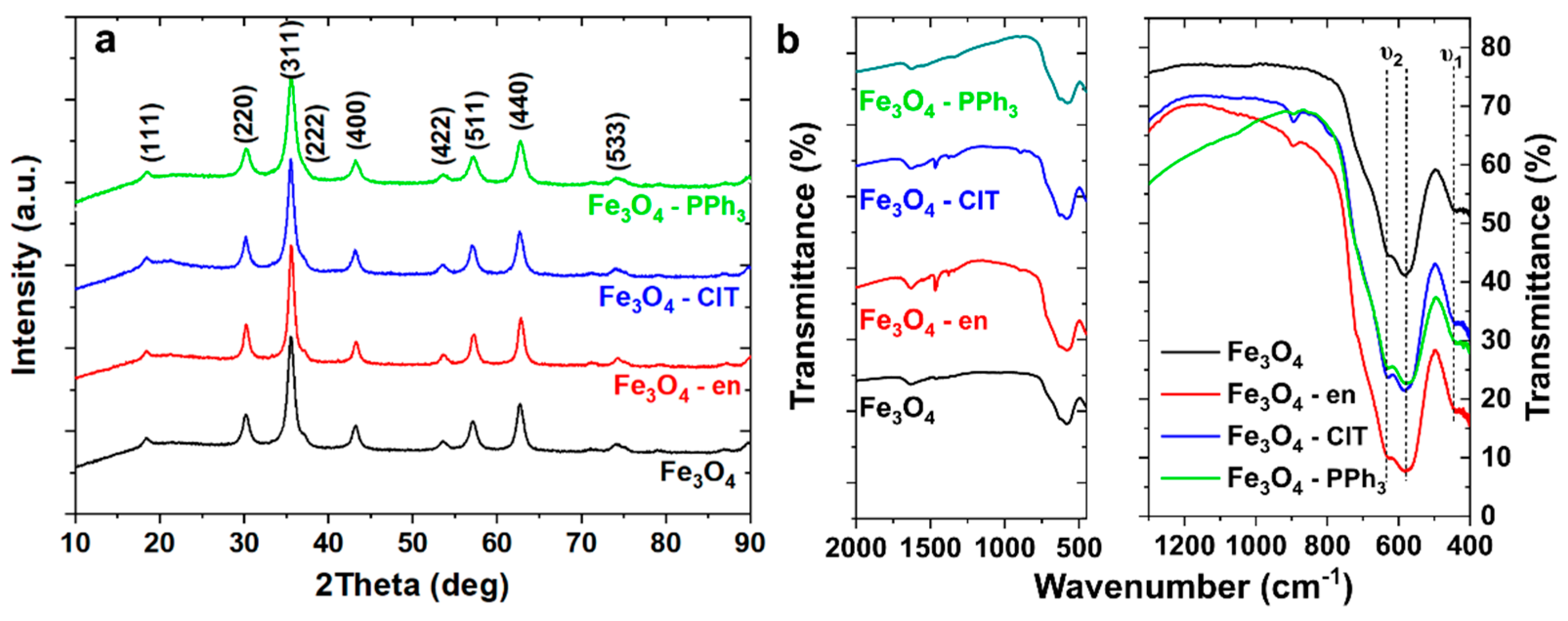

| Sample | DH-W (nm) | ε (10−3) | Dav (nm) | υ1 (cm−1) | υ2 (cm−1) | υ2′ (cm−1) |
|---|---|---|---|---|---|---|
| Fe3O4 | 9.06 | 0 | 11.1 ± 2.2 | 444.5 | 583.6 | 630.0 |
| Fe3O4–en | 11.8 | 14.6 | 10.7 ± 3.8 | 445.5 | 582.4 | 629.6 |
| Fe3O4–ClT | 10 | 13.11 | 11.4 ± 2.6 | 444.5 | 588.0 | 631.6 |
| Fe3O4–PPh3 | 8.04 | 21.7 | 9.6 ± 2.0 | 440.6 | 580.5 | 630.6 |
| Sample | Ms (emu/g) | Mr (emu/g) | Hc (Oe) | Mr/Ms (a.u.) |
|---|---|---|---|---|
| Fe3O4 | 55.1 | 1.4 | 13.6 | 25.2 × 10−3 |
| Fe3O4–en | 52.5 | 2.1 | 24.4 | 39.4 × 10−3 |
| Fe3O4–ClT | 44.7 | 1.1 | 13.1 | 23.7 × 10−3 |
| Fe3O4–PPh3 | 55.2 | 0.9 | 8.7 | 15.8 × 10−3 |
| Sample | Parameters | Sextets | Single Line | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | S5 | S6 | S7 | L | ||
| Fe3O4 | Is (mm/s) | 0.36 | 0.34 | 0.55 | 0.48 | 0.18 | - | - | 0.26 |
| Qs (mm/) | 0.00 | 0.01 | 0.00 | −0.01 | −0.02 | - | |||
| H (T) | 47.6 | 43.2 | 38.2 | 30.3 | 19.9 | - | |||
| A (%) | 41 | 25 | 12 | 7 | 7 | 8 | |||
| Fe3O4–en | Is (mm/s) | 0.32 | 0.26 | 0.56 | 0.36 | 0.34 | 0.18 | - | 0.17 |
| Qs (mm/) | 0.00 | −0.01 | 0.02 | 0.00 | 0.01 | 0.03 | - | ||
| H (T) | 47.9 | 43.9 | 43.0 | 37.9 | 29.5 | 10.4 | - | ||
| A (%) | 38 | 19 | 10 | 12 | 10 | 8 | 3 | ||
| Fe3O4–ClT | Is (mm/s) | 0.37 | 0.25 | 0.60 | 0.39 | 0.36 | 0.32 | - | 0.37 |
| Qs (mm/) | 0.00 | −0.02 | 0.02 | 0.03 | 0.02 | −0.01 | - | ||
| H (T) | 47.2 | 42.8 | 41.8 | 35.8 | 27.3 | 8.6 | - | ||
| A (%) | 23 | 11 | 9 | 13 | 15 | 12 | 15 | ||
| Fe3O4–PPh3 | Is (mm/s) | 0.37 | 0.26 | 0.60 | 0.35 | 0.31 | 0.29 | 0.29 | 0.28 |
| Qs (mm/) | 0.00 | −0.01 | 0.00 | 0.01 | 0.02 | −0.02 | −0.02 | - | |
| H (T) | 46.9 | 41.8 | 41.4 | 35.4 | 29.9 | 23.8 | 9.7 | - | |
| A (%) | 27 | 12 | 10 | 10 | 11 | 10 | 12 | 8 | |
| Sample | Ea,low T (eV) | Ea,highT (eV) |
|---|---|---|
| Fe3O4 | 0.15 | 0.26 |
| Fe3O4–en | 0.17 | 0.25 |
| Fe3O4–ClT | 0.15 | 0.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radoń, A.; Kądziołka-Gaweł, M.; Łukowiec, D.; Gębara, P.; Cesarz-Andraczke, K.; Kolano-Burian, A.; Włodarczyk, P.; Polak, M.; Babilas, R. Influence of Magnetite Nanoparticles Shape and Spontaneous Surface Oxidation on the Electron Transport Mechanism. Materials 2021, 14, 5241. https://doi.org/10.3390/ma14185241
Radoń A, Kądziołka-Gaweł M, Łukowiec D, Gębara P, Cesarz-Andraczke K, Kolano-Burian A, Włodarczyk P, Polak M, Babilas R. Influence of Magnetite Nanoparticles Shape and Spontaneous Surface Oxidation on the Electron Transport Mechanism. Materials. 2021; 14(18):5241. https://doi.org/10.3390/ma14185241
Chicago/Turabian StyleRadoń, Adrian, Mariola Kądziołka-Gaweł, Dariusz Łukowiec, Piotr Gębara, Katarzyna Cesarz-Andraczke, Aleksandra Kolano-Burian, Patryk Włodarczyk, Marcin Polak, and Rafał Babilas. 2021. "Influence of Magnetite Nanoparticles Shape and Spontaneous Surface Oxidation on the Electron Transport Mechanism" Materials 14, no. 18: 5241. https://doi.org/10.3390/ma14185241
APA StyleRadoń, A., Kądziołka-Gaweł, M., Łukowiec, D., Gębara, P., Cesarz-Andraczke, K., Kolano-Burian, A., Włodarczyk, P., Polak, M., & Babilas, R. (2021). Influence of Magnetite Nanoparticles Shape and Spontaneous Surface Oxidation on the Electron Transport Mechanism. Materials, 14(18), 5241. https://doi.org/10.3390/ma14185241







