Evaluation of PC12 Cells’ Proliferation, Adhesion and Migration with the Use of an Extracellular Matrix (CorMatrix) for Application in Neural Tissue Engineering
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material
2.2. Cell Line
2.3. Media and Conditions
2.4. Experimental Model
2.5. Detection of Apoptosis
2.6. Analysis of the Number of Cells on the CorMatrix Material
2.7. Statistical Analysis
3. Results
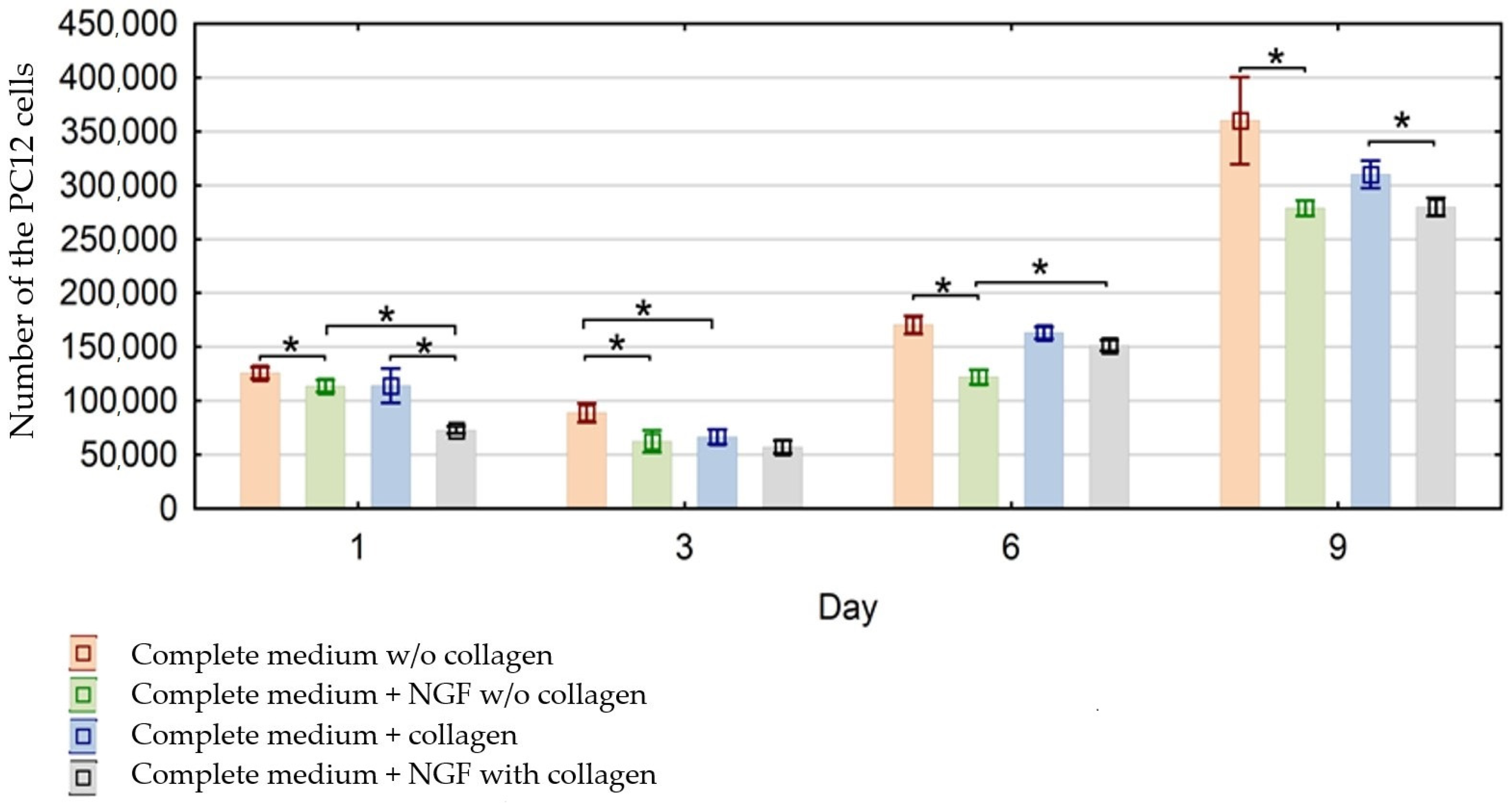
3.1. Evaluation of the Effect of CorMatrix Material on PC12 Cells Proliferation
3.2. Evaluation of the Effect of CorMatrix Material on PC12 Cells Migration
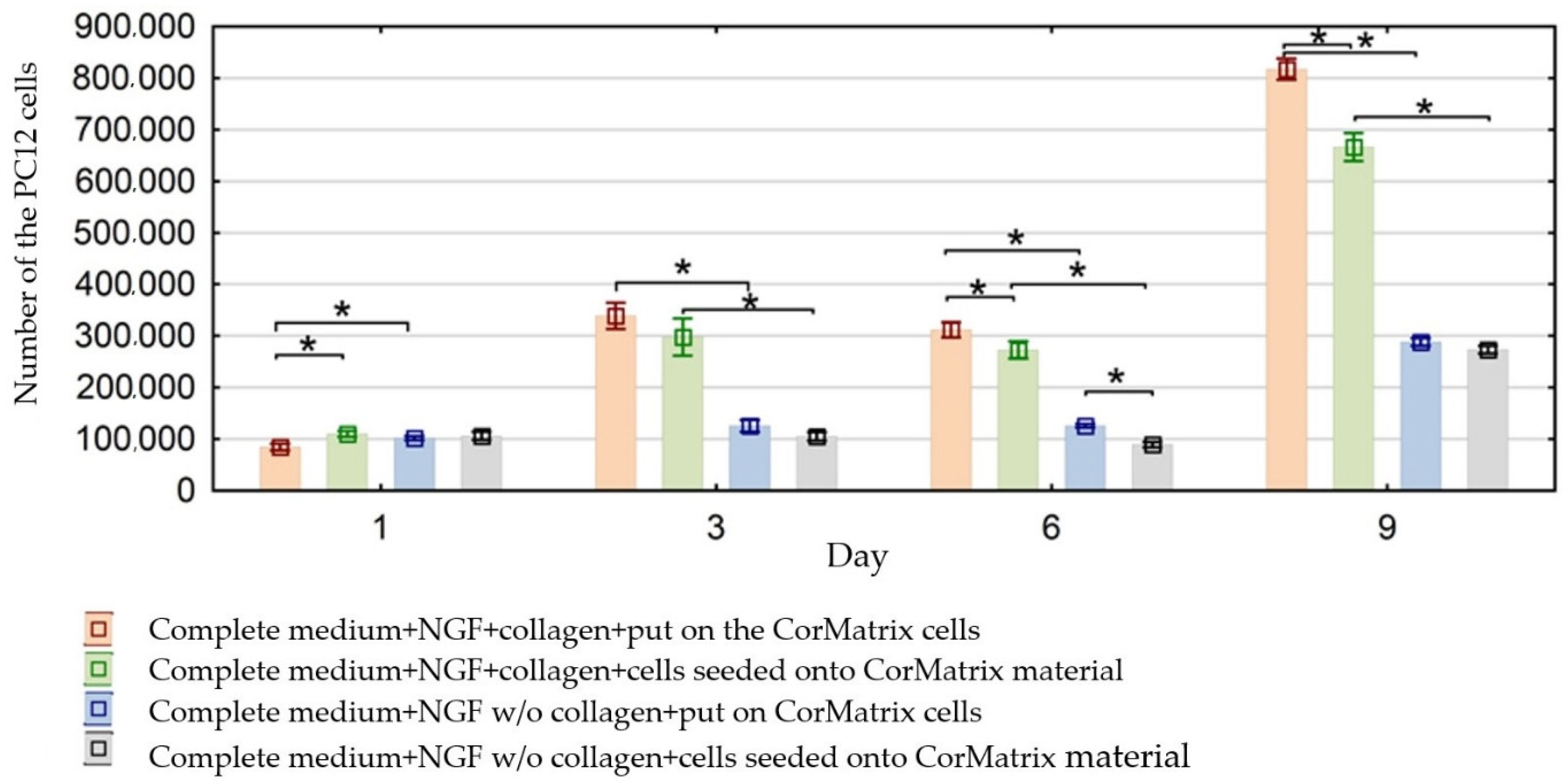
3.3. Evaluation of the Cell Number on the CorMatrix Material
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Crapo, P.M.; Medberry, C.J.; Reing, J.; Tottey, S.; van der Merwe, Y.; Jones, K.; Badylak, S.F. Biologic scaffolds composed of central nervous system extracellular matrix. Biomaterials 2012, 33, 3539–3547. [Google Scholar] [CrossRef]
- Badylak, S.F.; Freytes, D.O.; Gilbert, T. Reprint of: Extracellular matrix as a biological scaffold material: Structure and function. Acta Biomater. 2015, 23, S17–S26. [Google Scholar] [CrossRef]
- Hu, J.; Deng, L.; Wang, X.; Xu, X.-M. Effects of extracellular matrix molecules on the growth properties of oligodendrocyte progenitor cells in vitro. J. Neurosci. Res. 2009, 87, 2854–2862. [Google Scholar] [CrossRef] [PubMed]
- Tavis, M.J.; Thornton, J.; Danet, R.; Bartlett, R.H. Current Status of Skin Substitutes. Surg. Clin. N. Am. 1978, 58, 1233–1248. [Google Scholar] [CrossRef]
- Burke, J.F.; Yannas, I.V.; Quinby, W.C.; Bondoc, C.C.; Jung, W.K. Successful Use of a Physiologically Acceptable Artificial Skin in the Treatment of Extensive Burn Injury. Ann. Surg. 1981, 194, 413–428. [Google Scholar] [CrossRef]
- Wainwright, D. Use of an acellular allograft dermal matrix (AlloDerm) in the management of full-thickness burns. Burn 1995, 21, 243–248. [Google Scholar] [CrossRef]
- Falanga, V.A.; Armstrong, D.G.; Sabolinski, M.L. Graftskin, a Human Skin Equivalent. Diabetes Care 2001, 24, 290–295. [Google Scholar]
- Park, T.H.; Choi, W.Y.; Lee, J.H.; Lee, W.J. Micronized Cross-Linked Human Acellular Dermal Matrices: An Effective Scaffold for Collagen Synthesis and Promising Material for Tissue Augmentation. Tissue Eng. Regen. Med. 2017, 14, 517–523. [Google Scholar] [CrossRef]
- Meng, L.; Liao, W.; Yang, S.; Xiong, Y.; Song, C.; Liu, L. Tissue-engineered tubular substitutions for urinary diversion in a rabbit model. Exp. Biol. Med. 2016, 241, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Rameshbabu, A.P.; Datta, S.; Bankoti, K.; Subramani, E.; Chaudhury, K.; Lalzawmliana, V.; Nandi, S.K.; Dhara, S. Polycaprolactone nanofibers functionalized with placental derived extracellular matrix for stimulating wound healing activity. J. Mater. Chem. B 2018, 6, 6767–6780. [Google Scholar] [CrossRef] [PubMed]
- Klingberg, F.; Chau, G.; Walraven, M.; Boo, S.; Koehler, A.; Chow, M.L.; Hinz, B. The ED-A Domain Enhances the Capacity of Fibronectin to Store Latent TGF-Beta; Binding Pro-tein-1 in the Fibroblast Matrix. J. Cell Sci. 2018, 1–39. [Google Scholar] [CrossRef]
- Amaral, I.; Neiva, I.; Silva, F.; Sousa, S.R.; Piloto, A.; Lopes, C.; Barbosa, M.; Kirkpatrick, C.; Pêgo, A.P. Endothelialization of chitosan porous conduits via immobilization of a recombinant fibronectin fragment (rhFNIII7–10). Acta Biomater. 2013, 9, 5643–5652. [Google Scholar] [CrossRef] [PubMed]
- Herranz-Diez, C.; Mas-Moruno, C.; Neubauer, S.; Kessler, H.; Gil, F.J.; Pegueroles, M.; Manero, J.M.; Guillem-Marti, J. Tuning Mesenchymal Stem Cell Response onto Titanium–Niobium–Hafnium Alloy by Recombinant Fibronectin Fragments. ACS Appl. Mater. Interfaces 2016, 8, 2517–2525. [Google Scholar] [CrossRef]
- Yun, Y.-R.; Pham, L.B.H.; Yoo, Y.-R.; Lee, S.; Kim, H.-W.; Jang, J.-H. Engineering of Self-Assembled Fibronectin Matrix Protein and Its Effects on Mesenchymal Stem Cells. Int. J. Mol. Sci. 2015, 16, 19645–19656. [Google Scholar] [CrossRef]
- Wang, Y.; Zeinali-Davarani, S.; Zhang, Y. Arterial mechanics considering the structural and mechanical contributions of ECM constituents. J. Biomech. 2014, 63, 1–18. [Google Scholar] [CrossRef]
- Stoichevska, V.; Peng, Y.Y.; Vashi, A.V.; Werkmeister, J.A.; Dumsday, G.; Ramshaw, J.A.M. Engineering specific chemical modification sites into a collagen-like protein fromStreptococcus pyogenes. J. Biomed. Mater. Res. Part A 2017, 105, 806–813. [Google Scholar] [CrossRef]
- Martin, S.L.; Vrhovski, B.; Weiss, A.S. Total synthesis and expression in Escherichia coli of a gene encoding human tropoelastin. Gene 1995, 154, 159–166. [Google Scholar] [CrossRef]
- Wang, L.; Ding, L.; Yu, Z.; Zhang, T.; Ma, S.; Liu, J. Intracellular ROS scavenging and antioxidant enzyme regulating capacities of corn gluten meal-derived antioxidant peptides in HepG2 cells. Food Res. Int. 2016, 90, 33–41. [Google Scholar] [CrossRef]
- Kortesmaa, J.; Yurchenco, P.; Tryggvason, K. Recombinant Laminin-8 (α4β1γ1). J. Biol. Chem. 2000, 275, 14853–14859. [Google Scholar] [CrossRef] [PubMed]
- Rodin, S.; Domogatskaya, A.; Ström, S.; Hansson, E.M.; Chien, K.R.; Inzunza, J.; Hovatta, O.; Tryggvason, K. Long-term self-renewal of human pluripotent stem cells on human recombinant laminin-511. Nat. Biotechnol. 2010, 28, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.; Lee, H.; Luo, L.; Kyriakides, T.R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol. Adv. 2020, 42, 107421. [Google Scholar] [CrossRef] [PubMed]
- Lelievre, S.; Weaver, V.M.; Nickerson, J.A.; Larabell, C.A.; Bhaumik, A.; Petersen, O.W.; Bissell, M.J. Tissue phenotype depends on reciprocal interactions between the extracellular matrix and the structural organization of the nucleus. Proc. Natl. Acad. Sci. USA 1998, 95, 14711–14716. Available online: http://www.ncbi.nlm.nih.gov/pubmed/9843954 (accessed on 9 July 2021). [CrossRef] [PubMed]
- Uemura, M.; Refaat, M.M.; Shinoyama, M.; Hayashi, H.; Hashimoto, N.; Takahashi, J. Matrigel supports survival and neuronal differentiation of grafted embryonic stem cell-derived neural precursor cells. J. Neurosci. Res. 2009, 88, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Hynes, R.O. Integrins: Versatility, modulation, and signaling in cell adhesion. Cell 1992, 69, 11–25. [Google Scholar] [CrossRef]
- Smethurst, P.A.; Onley, D.J.; Jarvis, G.; O’Connor, M.N.; Knight, C.G.; Herr, A.; Ouwehand, W.H.; Farndale, R.W. Structural basis for the platelet-collagen interaction: The smallest motif within collagen that recognizes and activates platelet Glycoprotein VI contains two glycine-proline-hydroxyproline triplets. J. Biol. Chem. 2007, 282, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Joshi, J.; Brennan, D.; Beachley, V.; Kothapalli, C.R. Cardiomyogenic differentiation of human bone marrow-derived mesenchymal stem cell spheroids within electrospun collagen nanofiber mats. J. Biomed. Mater. Res. Part A 2018, 106, 3303–3312. [Google Scholar] [CrossRef] [PubMed]
- Altman, A.M.; Matthias, N.; Yan, Y.; Song, Y.-H.; Bai, X.; Chiu, E.S.; Slakey, D.P.; Alt, E.U. Dermal matrix as a carrier for in vivo delivery of human adipose-derived stem cells. Biomaterials 2008, 29, 1431–1442. [Google Scholar] [CrossRef] [PubMed]
- Grier, W.K.; Tiffany, A.S.; Ramsey, M.D.; Harley, B.A. Incorporating β-cyclodextrin into collagen scaffolds to sequester growth factors and modulate mesenchymal stem cell activity. Acta Biomater. 2018, 76, 116–125. [Google Scholar] [CrossRef]
- Hubert, T.; Grimal, S.; Carroll, P.; Fichard-Carroll, A. Collagens in the developing and diseased nervous system. Cell. Mol. Life Sci. 2009, 66, 1223–1238. [Google Scholar] [CrossRef]
- Grauss, R.W.; Hazekamp, M.G.; Oppenhuizen, F.; Van Munsterena, C.J.; Groot, A.C.G.-D.; DeRuiter, M.C. Histological evaluation of decellularised porcine aortic valves: Matrix changes due to different decellularisation methods. Eur. J. Cardio-Thoracic Surg. 2005, 27, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Schenke-Layland, K.; Vasilevski, O.; Opitz, F.; König, K.; Riemann, I.; Halbhuber, K.; Wahlers, T.; Stock, U. Impact of decellularization of xenogeneic tissue on extracellular matrix integrity for tissue engineering of heart valves. J. Struct. Biol. 2003, 143, 201–208. Available online: http://www.ncbi.nlm.nih.gov/pubmed/14572475 (accessed on 9 July 2021). [CrossRef]
- Dahl, S.L.M.; Koh, J.; Prabhakar, V.; Niklason, L.E. Decellularized native and engineered arterial scaffolds for transplantation. Cell Transpl. 2003, 12, 659–666. Available online: http://www.ncbi.nlm.nih.gov/pubmed/14579934 (accessed on 9 July 2021). [CrossRef]
- Uchimura, E.; Sawa, Y.; Taketani, S.; Yamanaka, Y.; Hara, M.; Matsuda, H.; Miyake, J. Novel method of preparing acellular cardiovascular grafts by decellularization with poly(ethylene glycol). J. Biomed. Mater. Res. 2003, 67, 834–837. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.-N.; Ho, H.-O.; Tsai, Y.-T.; Sheu, M.-T. Process development of an acellular dermal matrix (ADM) for biomedical applications. Biomaterials 2004, 25, 2679–2686. Available online: http://www.ncbi.nlm.nih.gov/pubmed/14751754 (accessed on 9 July 2021). [CrossRef] [PubMed]
- Hudson, T.W.; Zawko, S.; Deister, C.; Lundy, S.; Hu, C.Y.; Lee, K.; Schmidt, C.E. Optimized Acellular Nerve Graft Is Immunologically Tolerated and Supports Regeneration. Tissue Eng. 2004, 10, 1641–1651. [Google Scholar] [CrossRef] [PubMed]
- Borschel, G.; Dennis, R.G.; Kuzon, W.M. Contractile Skeletal Muscle Tissue-Engineered on an Acellular Scaffold. Plast. Reconstr. Surg. 2004, 113, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Cartmell, J.S.; Dunn, M.G. Effect of chemical treatments on tendon cellularity and mechanical properties. J. Biomed. Mater. Res. 2000, 49, 134–140. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10559756 (accessed on 9 July 2021). [CrossRef]
- Woods, T.; Gratzer, P.F. Effectiveness of three extraction techniques in the development of a decellularized bone–anterior cruciate ligament–bone graft. Biomaterials 2005, 26, 7339–7349. [Google Scholar] [CrossRef]
- Badylak, S.; Lantz, G.C.; Coffey, A.; Geddes, L.A. Small intestinal submucosa as a large diameter vascular graft in the dog. J. Surg. Res. 1989, 47, 74–80. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2739401 (accessed on 9 July 2021). [CrossRef]
- Badylak, S.; Tullius, R.; Kokini, K.; Shelbourne, K.D.; Klootwyk, T.; Voytik, S.L.; Kraine, M.R.; Simmons, C. The use of xenogeneic small intestinal submucosa as a biomaterial for Achille’s tendon repair in a dog model. J. Biomed. Mater. Res. 1995, 29, 977–985. [Google Scholar] [CrossRef]
- Kropp, B.P.; Eppley, B.L.; Prevel, C.; Rippy, M.; Harruff, R.; Badylak, S.; Adams, M.; Rink, R.; Keating, M. Experimental assessment of small intestinal submucosa as a bladder wall substitute. Urology 1995, 46, 396–400. [Google Scholar] [CrossRef]
- Gilbert, T.; Stolz, D.B.; Biancaniello, F.; Simmons-Byrd, A.; Badylak, S.F. Production and characterization of ECM powder: Implications for tissue engineering applications. Biomaterials 2005, 26, 1431–1435. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Chan, W.C.; Badylak, S.F.; Bhatia, S.N. Assessing Porcine Liver-Derived Biomatrix for Hepatic Tissue Engineering. Tissue Eng. 2004, 10, 1046–1053. [Google Scholar] [CrossRef]
- Gilbert, T.; Sellaro, T.L.; Badylak, S.F. Decellularization of tissues and organs. Biomaterials 2006, 27, 3675–3683. [Google Scholar] [CrossRef] [PubMed]
- Järvinen, T.A.H.; Prince, S. Decorin: A Growth Factor Antagonist for Tumor Growth Inhibition. BioMed Res. Int. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Liang, R.; Yang, G.; Kim, K.E.; D’Amore, A.; Pickering, A.N.; Zhang, C.; Woo, S.L.-Y. Positive effects of an extracellular matrix hydrogel on rat anterior cruciate ligament fibroblast proliferation and collagen mRNA expression. J. Orthop. Transl. 2015, 3, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Mewhort, H.E.; Svystonyuk, D.A.; Turnbull, J.D.; Teng, G.; Belke, D.D.; Guzzardi, D.G.; Park, D.S.; Kang, S.; Hollenberg, M.D.; Fedak, P.W. Bioactive Extracellular Matrix Scaffold Promotes Adaptive Cardiac Remodeling and Repair. JACC Basic Transl. Sci. 2017, 2, 450–464. [Google Scholar] [CrossRef]
- Xu, Q.; Shanti, R.M.; Zhang, Q.; Cannady, S.B.; O’Malley, B.W.; Le, A.D. A Gingiva-Derived Mesenchymal Stem Cell-Laden Porcine Small Intestinal Submucosa Extracellular Matrix Construct Promotes Myomucosal Regeneration of the Tongue. Tissue Eng. Part A 2017, 23, 301–312. [Google Scholar] [CrossRef]
- Keane, T.; Dziki, J.; Sobieski, E.; Smoulder, A.; Castleton, A.; Turner, N.; White, L.J.; Badylak, S.F. Restoring Mucosal Barrier Function and Modifying Macrophage Phenotype with an Extracellular Matrix Hydrogel: Potential Therapy for Ulcerative Colitis. J. Crohn’s Coliti 2016, 11, 360–368. [Google Scholar] [CrossRef]
- Nezhad, Z.M.; Poncelet, A.; De Kerchove, L.; Gianello, P.; Fervaille, C.; El Khoury, G. Small intestinal submucosa extracellular matrix (CorMatrix®) in cardiovascular surgery: A systematic review. Interact. Cardiovasc. Thorac. Surg. 2016, 22, 839–850. [Google Scholar] [CrossRef]
- Slaughter, M.S.; Soucy, K.G.; Matheny, R.G.; Lewis, B.C.; Hennick, M.F.; Choi, Y.; Monreal, G.; Sobieski, M.A.; Giridharan, G.A.; Koenig, S.C. Development of an Extracellular Matrix Delivery System for Effective Intramyocardial Injection in Ischemic Tissue. ASAIO J. 2014, 60, 730–736. [Google Scholar] [CrossRef]
- Soucy, K.G.; Smith, E.F.; Monreal, G.; Rokosh, G.; Keller, B.B.; Yuan, F.; Matheny, R.G.; Fallon, A.M.; Lewis, B.C.; Sherwood, L.C.; et al. Feasibility Study of Particulate Extracellular Matrix (P-ECM) and Left Ventricular Assist Device (HVAD) Therapy in Chronic Ischemic Heart Failure Bovine Model. ASAIO J. 2015, 61, 161–169. [Google Scholar] [CrossRef]
- Quarti, A.; Nardone, S.; Colaneri, M.; Santoro, G.; Pozzi, M. Preliminary experience in the use of an extracellular matrix to repair congenital heart diseases. Interact. Cardiovasc. Thorac. Surg. 2011, 13, 569–572. [Google Scholar] [CrossRef]
- Witt, R.G.; Raff, G.; Van Gundy, J.; Rodgers-Ohlau, M.; Si, M.-S. Short-term experience of porcine small intestinal submucosa patches in paediatric cardiovascular surgery. Eur. J. Cardio-Thoracic Surg. 2013, 44, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Boyd, W.D.; Johnson, W.E.; Sultan, P.K.; Deering, T.F.; Matheny, R.G. Pericardial Reconstruction Using an Extracellular Matrix Implant Correlates with Reduced Risk of Postoperative Atrial Fibrillation in Coronary Artery Bypass Surgery Patients. Hear. Surg. Forum 2010, 13, E311–E316. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, B.; Rao, V.; Yau, T.M.; Cusimano, R.J. Initial Experience with Intraventricular Repair Using CorMatrix Extracellular Matrix. Innov. Technol. Tech. Cardiothorac. Vasc. Surg. 2013, 8, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, A.H.; Nathan, M.; Emani, S.; Baird, C.; del Nido, P.J.; Gauvreau, K.; Harris, M.; Sanders, S.P.; Padera, R.F. Preliminary experience with porcine intestinal submucosa (CorMatrix) for valve reconstruction in congenital heart disease: Histologic evaluation of explanted valves. J. Thorac. Cardiovasc. Surg. 2014, 148, 2216–2225. [Google Scholar] [CrossRef] [PubMed]
- Stelly, M.; Stelly, T.C. Histology of CorMatrix Bioscaffold 5 Years after Pericardial Closure. Ann. Thorac. Surg. 2013, 96, e127–e129. [Google Scholar] [CrossRef] [PubMed]
- Gerdisch, M.W.; Shea, R.J.; Barron, M.D. Clinical experience with CorMatrix extracellular matrix in the surgical treatment of mitral valve disease. J. Thorac. Cardiovasc. Surg. 2014, 148, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Biefer, H.R.C.; Emmert, M.; Falk, V.; Sündermann, S. Use of Extracellular Matrix Materials in Patients with Endocarditis. Thorac. Cardiovasc. Surg. 2012, 62, 076–079. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, B.; Rao, V.; Yau, T.M.; Cusimano, R.J. Potential myocardial regeneration with CorMatrix ECM: A case report. J. Thorac. Cardiovasc. Surg. 2014, 147, e41–e43. [Google Scholar] [CrossRef]
- Drago, J.; Nurcombe, V.; Bartlett, P.F. Laminin through its long arm E8 fragment promotes the proliferation and differentiation of murine neuroepithelial cells in vitro. Exp. Cell Res. 1991, 192, 256–265. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1701728 (accessed on 9 July 2021). [CrossRef]
- Kearns, S.; Laywell, E.; Kukekov, V.; Steindler, D. Extracellular matrix effects on neurosphere cell motility. Exp. Neurol. 2003, 182, 240–244. Available online: http://www.ncbi.nlm.nih.gov/pubmed/12821394 (accessed on 9 July 2021). [CrossRef]
- Londono, R.; Badylak, S.F. Biologic Scaffolds for Regenerative Medicine: Mechanisms of In vivo Remodeling. Ann. Biomed. Eng. 2014, 43, 577–592. [Google Scholar] [CrossRef]
- Krishnan, N.M.; Chatterjee, A.; Rosenkranz, K.M.; Powell, S.G.; Nigriny, J.F.; Vidal, D.C. The cost effectiveness of acellular dermal matrix in expander–implant immediate breast reconstruction. J. Plast. Reconstr. Aesthetic Surg. 2014, 67, 468–476. [Google Scholar] [CrossRef]
- Nafisi, N.; Mahjoub, F.; Mohseni, M.J.; Sabetkish, S.; Khorramirouz, R.; Tehrani, M.; Akbari, M.E.; Kajbafzadeh, A.-M. Application of Human Acellular Breast Dermal Matrix (ABDM) in Implant-Based Breast Reconstruction: An Experimental Study. Aesthetic Plast. Surg. 2017, 41, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.S.; Loh, W. The utility of adipose-derived stem cells and stromal vascular fraction for oncologic soft tissue reconstruction: Is it safe? A matter for debate. Surgeon 2017, 15, 186–189. [Google Scholar] [CrossRef]
- Turner, A.E.; Yu, C.; Bianco, J.; Watkins, J.F.; Flynn, L.E. The performance of decellularized adipose tissue microcarriers as an inductive substrate for human adipose-derived stem cells. Biomaterials 2012, 33, 4490–4499. [Google Scholar] [CrossRef] [PubMed]
- Majeed, A.; Borisuk, M.J.; Padera, R.F.; Baird, C.; Sanders, S.P. Histology of Pericardial Tissue Substitutes Used in Congenital Heart Surgery. Pediatr. Dev. Pathol. 2016, 19, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Neethling, W.M.L.; Puls, K.; Rea, A. Comparison of physical and biological properties of CardioCel® with commonly used bioscaffolds. Interact. Cardiovasc. Thorac. Surg. 2018, 26, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Murugan, R.; Ramakrishna, S.; Wang, X.; Ma, Y.-X.; Wang, S. Fabrication of nano-structured porous PLLA scaffold intended for nerve tissue engineering. Biomaterials 2004, 25, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Rafie, A.H.S.; Abilez, O.J.; Wang, H.; Blundo, J.T.; Pruitt, B.L.; Zarins, C.; Wu, J.C. In vivo imaging and evaluation of different biomatrices for improvement of stem cell survival. J. Tissue Eng. Regen. Med. 2007, 1, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Kurosaka, K.; Takahashi, M.; Watanabe, N.; Kobayashi, Y. Silent Cleanup of Very Early Apoptotic Cells by Macrophages. J. Immunol. 2003, 171, 4672–4679. Available online: http://www.ncbi.nlm.nih.gov/pubmed/14568942 (accessed on 9 July 2021). [CrossRef]
- Greene, L.A.; Tischler, A. Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc. Natl. Acad. Sci. USA 1976, 73, 2424–2428. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1065897 (accessed on 9 July 2021). [CrossRef]
- Bachmann, M.; Conscience, J.-F.; Probstmeier, R.; Carbonetto, S.; Schachner, M. Recognition molecules myelin-associated glycoprotein and tenascin-C inhibit integrin-mediated adhesion of neural cells to collagen. J. Neurosci. Res. 1995, 40, 458–470. [Google Scholar] [CrossRef]
- Ghuman, H.; Mauney, C.; Donnelly, J.; Massensini, A.; Badylak, S.F.; Modo, M. Biodegradation of ECM hydrogel promotes endogenous brain tissue restoration in a rat model of stroke. Acta Biomater. 2018, 80, 66–84. [Google Scholar] [CrossRef]
- Shen, W.; Chen, X.; Hu, Y.; Yin, Z.; Zhu, T.; Hu, J.; Chen, J.; Zheng, Z.; Zhang, W.; Ran, J.; et al. Long-term effects of knitted silk–collagen sponge scaffold on anterior cruciate ligament reconstruction and osteoarthritis prevention. Biomaterials 2014, 35, 8154–8163. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Woo, H.-M.; Kang, B.-J. Impact of collagen-alginate composition from microbead morphological properties to microencapsulated canine adipose tissue-derived mesenchymal stem cell activities. J. Biomater. Sci. Polym. Ed. 2017, 29, 1042–1052. [Google Scholar] [CrossRef]
- Arnaoutova, I.; George, J.; Kleinman, H.K.; Benton, G. Basement Membrane Matrix (BME) has Multiple Uses with Stem Cells. Stem Cell Rev. Rep. 2012, 8, 163–169. [Google Scholar] [CrossRef]
- Anlar, B.; Atilla, P.; Çakar, A.N.; Kose, M.F.; Beksaç, M.S.; Dagdeviren, A.; Akçören, Z. Expression of adhesion and extracellular matrix molecules in the developing human brain. J. Child. Neurol. 2002, 17, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, L.A.; Rebaza, L.M.; Derzic, S.; Schwartz, P.H.; Monuki, E.S. Regulation of human neural precursor cells by laminin and integrins. J. Neurosci. Res. 2006, 83, 845–856. [Google Scholar] [CrossRef] [PubMed]
- Marastoni, S.; Ligresti, G.; Lorenzon, E.; Colombatti, A.; Mongiat, M. Extracellular Matrix: A Matter of Life and Death. Connect. Tissue Res. 2008, 49, 203–206. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gębczak, K.; Wiatrak, B.; Fortuna, W. Evaluation of PC12 Cells’ Proliferation, Adhesion and Migration with the Use of an Extracellular Matrix (CorMatrix) for Application in Neural Tissue Engineering. Materials 2021, 14, 3858. https://doi.org/10.3390/ma14143858
Gębczak K, Wiatrak B, Fortuna W. Evaluation of PC12 Cells’ Proliferation, Adhesion and Migration with the Use of an Extracellular Matrix (CorMatrix) for Application in Neural Tissue Engineering. Materials. 2021; 14(14):3858. https://doi.org/10.3390/ma14143858
Chicago/Turabian StyleGębczak, Katarzyna, Benita Wiatrak, and Wojciech Fortuna. 2021. "Evaluation of PC12 Cells’ Proliferation, Adhesion and Migration with the Use of an Extracellular Matrix (CorMatrix) for Application in Neural Tissue Engineering" Materials 14, no. 14: 3858. https://doi.org/10.3390/ma14143858






