Pearl Powder—An Emerging Material for Biomedical Applications: A Review
Abstract
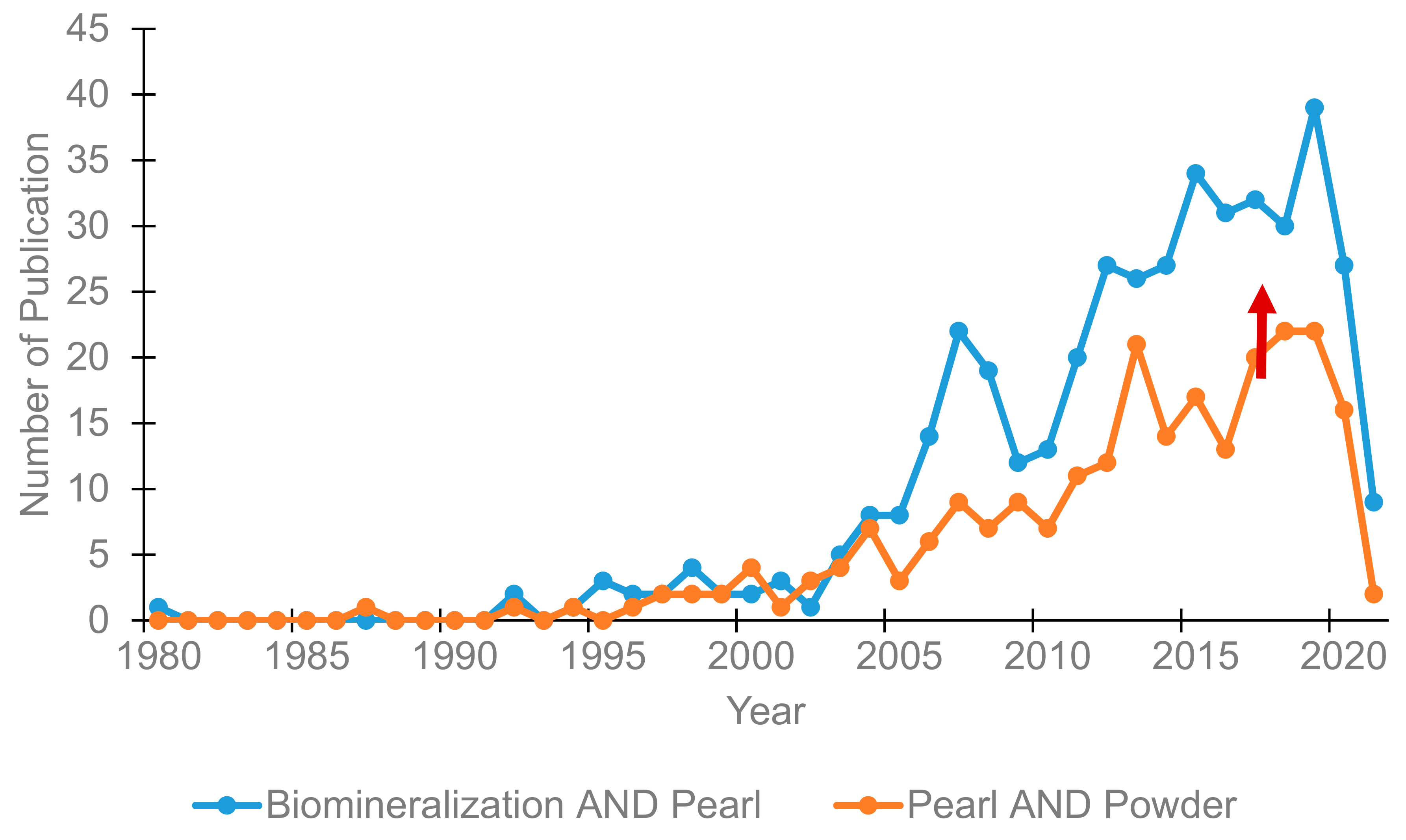
1. Introduction
2. Biomineralization in Pearls
3. Applications
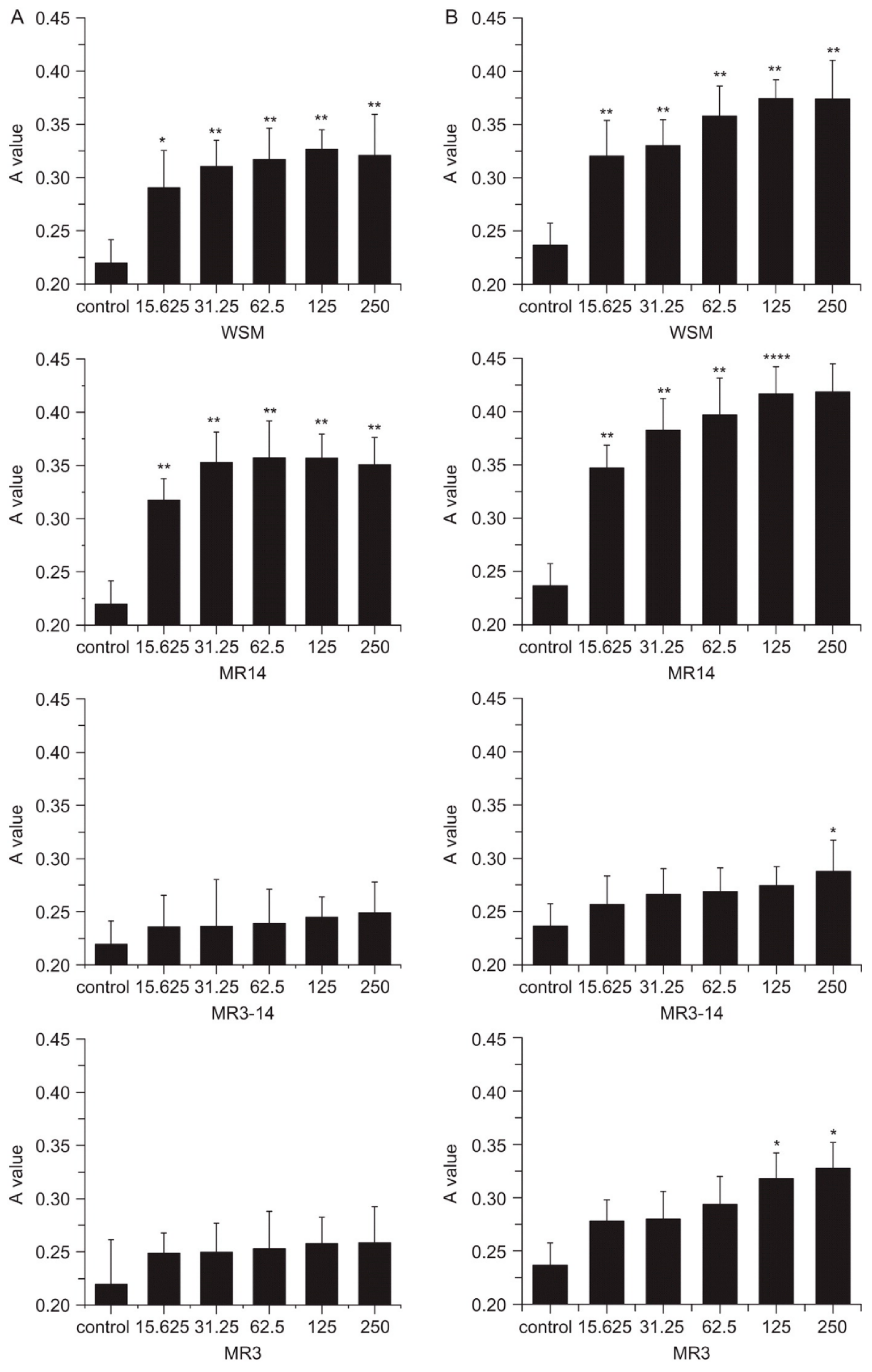
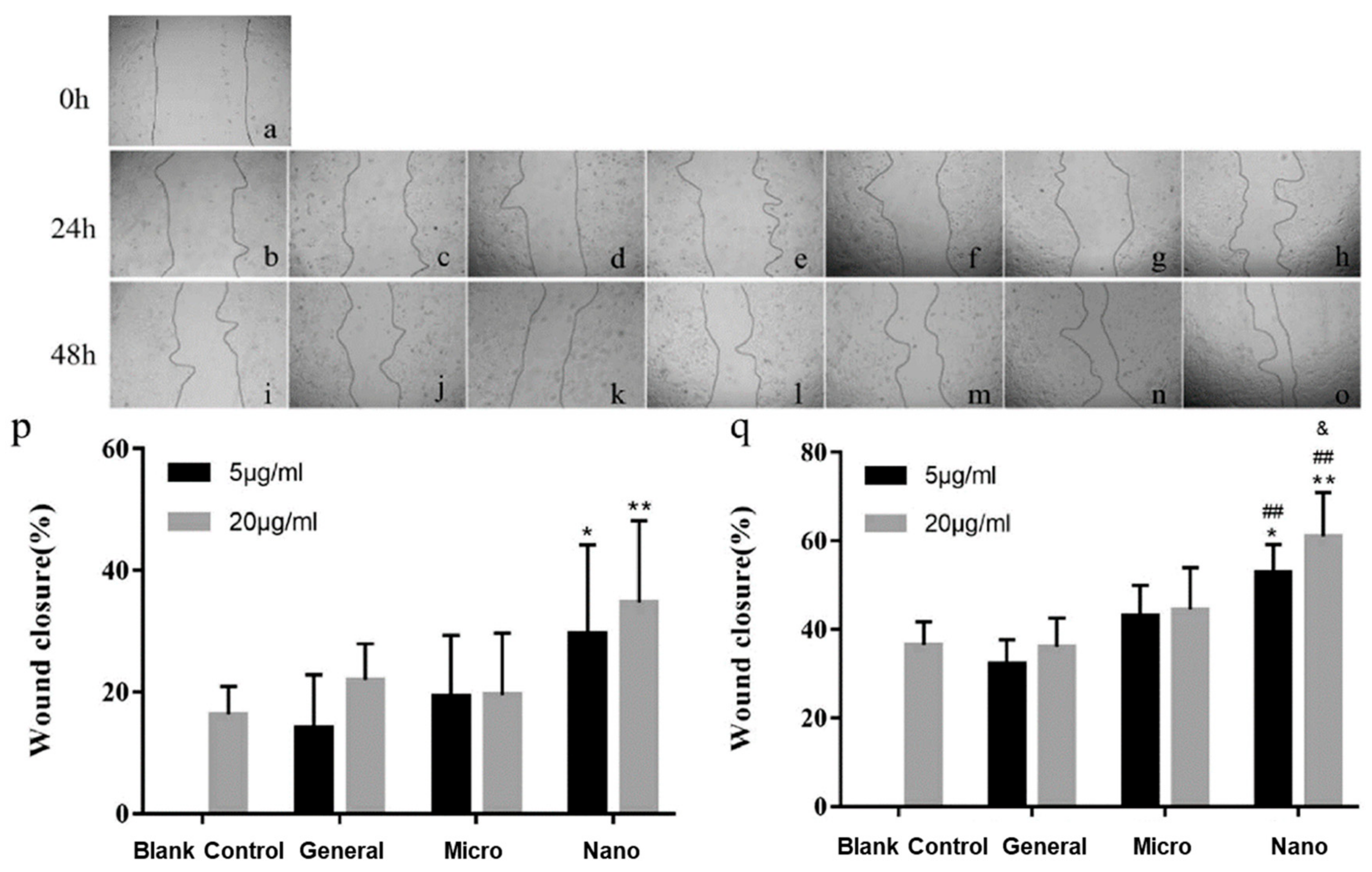
3.1. Wound Healing and Treatment of Skin Conditions
3.2. Anti-Fibrotic and Anti-Inflammatory Action
3.3. Antioxidant and Anti-Aging Applications
3.4. Other Potential Health Effects
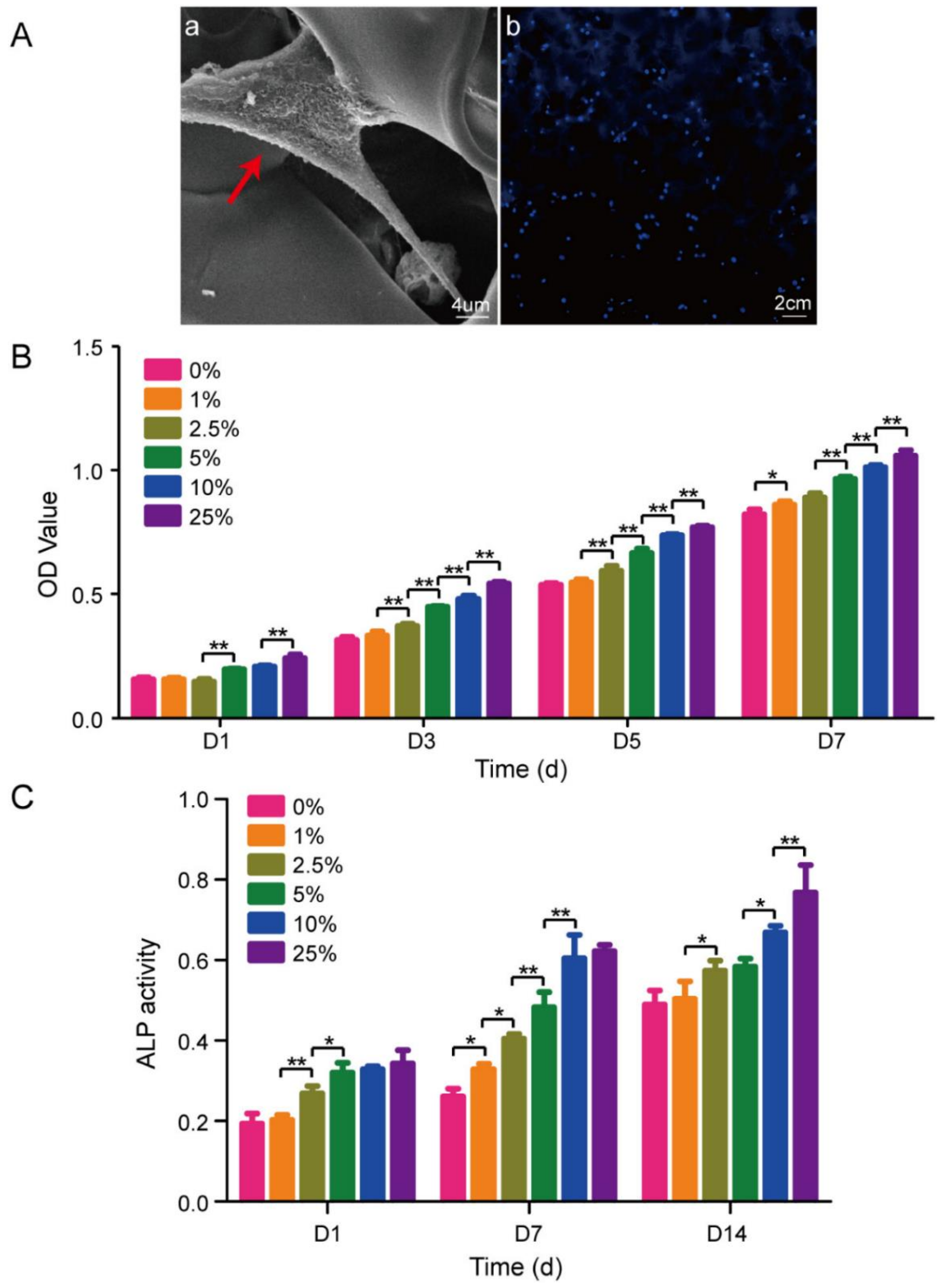
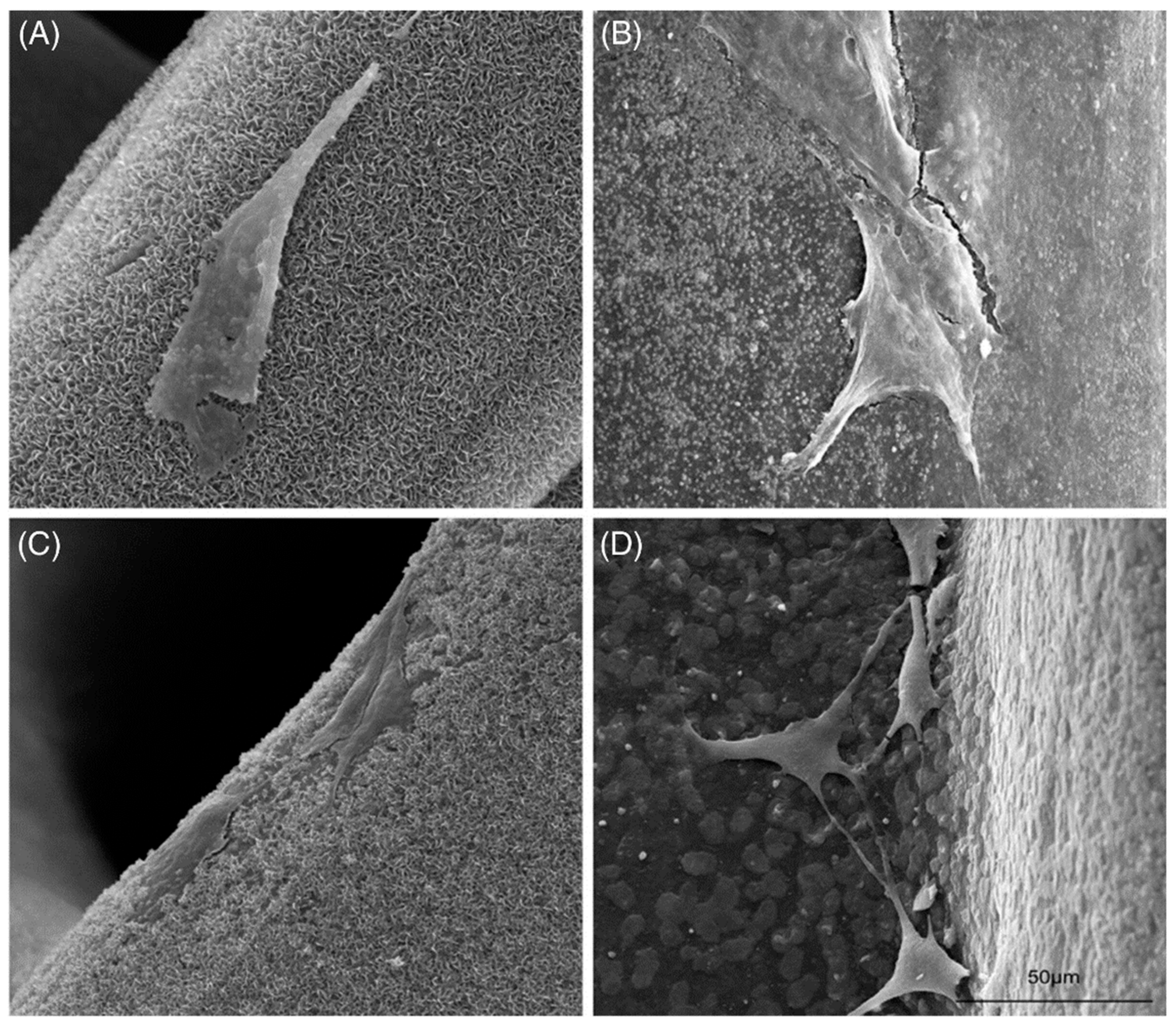
3.5. Scaffolds for Tissue Engineering
4. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALP | alkaline phosphatase |
| Ca3(PO4)2 | calcium phosphate |
| CaCO3 | calcium carbonate |
| CaSO4 | calcium sulphate |
| CCK-8 | cell count kit-8 |
| DOX | doxorubicin hydrochloride |
| ECM | extracellular matrix |
| ERK | extracellular signal-controlled kinase |
| FDA | Food and Drug Administration |
| HA | hydroxyapatite |
| MEK | mitogen-activated protein kinase |
| NPP | nano-pearl powder |
| PAA | polyaminoacid |
| PCL | polycaprolactone |
| PDGF | platelet-derived growth factor |
| PHBV) | poly(3-hydroxybutyrate-co-3-hydroxyvalerate |
| PLA | polylactic acid |
| PLLA | poly-L-lactide |
| PP | polypropylene |
| rBMSC | rat bone marrow-derived mesenchymal stem cells |
| ROS | reactive oxygen species |
| RUNX2 | runt-related transcription factor 2 |
| SBF | simulated body fluid |
| SPP1 | secreted phosphoprotein l |
| WSM | water-soluble matrix |
References
- Zhang, J.; Li, S.; Yao, S.; Si, W.; Cai, L.; Pan, H.; Hou, J.; Yang, W.; Da, J.; Jiang, B.; et al. Ultra-performance liquid chromatography of amino acids for the quality assessment of pearl powder. J. Sep. Sci. 2015, 38, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-L.; Korivi, M.; Lin, M.-K.; Chang, H.C.-W.; Wu, C.-R.; Lee, M.-S.; Chen, W.T.-L.; Hseu, Y.-C. Antihemolytic and antioxidant properties of pearl powder against 2,2′-azobis(2-amidinopropane) dihydrochloride-induced hemolysis and oxidative damage to erythrocyte membrane lipids and proteins. J. Food Drug Anal. 2017, 25, 898–907. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.-F.; Hsiao, S.-C.; Lu, Y.-Y.; Han, Y.-C.; Shen, Y.-C.; Venkatakrishnan, K.; Wang, C.-K. Efficacy of protein rich pearl powder on antioxidant status in a randomized placebo-controlled trial. J. Food Drug Anal. 2018, 26, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Peng, L.-H.; Chee, S.-S.; Shan, Y.-H.; Liang, W.-Q.; Gao, J.-Q. Nanoscaled pearl powder accelerates wound repair and regeneration in vitro and in vivo. Drug Dev. Ind. Pharm. 2019, 45, 1009–1016. [Google Scholar] [CrossRef]
- Nagai, K. A history of the cultured pearl industry. Zool. Sci. 2013, 30, 783–793. [Google Scholar] [CrossRef]
- Yu, Z.R.; Wang, X.D.; Su, B.M.; Zhang, Y. First evidence of the use of freshwater pearls as a cosmetic in ancient China: Analysis of white makeup powder from a Northern Song dynasty Lv tomb (Lantian, Shaanxi province, China). Archaeometry 2017, 59, 762–774. [Google Scholar] [CrossRef]
- Latire, T.; Legendre, F.; Bouyoucef, M.; Marin, F.; Carreiras, F.; Rigot-Jolivet, M.; Lebel, J.-M.; Galéra, P.; Serpentini, A. Shell extracts of the edible mussel and oyster induce an enhancement of the catabolic pathway of human skin fibroblasts, in vitro. Cytotechnology 2017, 69, 815–829. [Google Scholar] [CrossRef]
- Lee, K.; Kim, H.; Kim, J.M.; Chung, Y.H.; Lee, T.Y.; Lim, H.-S.; Lim, J.-H.; Kim, T.; Bae, J.S.; Woo, C.-H.; et al. Nacre-driven water-soluble factors promote wound healing of the deep burn porcine skin by recovering angiogenesis and fibroblast function. Mol. Biol. Rep. 2012, 39, 3211–3218. [Google Scholar] [CrossRef]
- Li, Y.-C.; Chen, C.-R.; Young, T.-H. Pearl extract enhances the migratory ability of fibroblasts in a wound healing model. Pharm. Biol. 2013, 51, 289–297. [Google Scholar] [CrossRef]
- Werner, S.; Krieg, T.; Smola, H. Keratinocyte–fibroblast interactions in wound healing. J. Investig. Dermatol. 2007, 127, 998–1008. [Google Scholar] [CrossRef]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in prevention and therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, D.; Sarashina, I.; Endo, K. Structure and expression of an unusually acidic matrix protein of pearl oyster shells. Biochem. Biophys. Res. Commun. 2004, 320, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xie, L.; Huang, J.; Liu, X.; Zhang, R. A novel matrix protein family participating in the prismatic layer framework formation of pearl oyster, Pinctada fucata. Biochem. Biophys. Res. Commun. 2006, 344, 735–740. [Google Scholar] [CrossRef]
- Park, S.Y.; Ahn, C.-B.; Je, J.-Y. Antioxidant and anti-inflammatory activities of protein hydrolysates from mytilus edulis and ultrafiltration membrane fractions. J. Food Biochem. 2014, 38, 460–468. [Google Scholar] [CrossRef]
- Saiga, A.; Tanabe, S.; Nishimura, T. Antioxidant activity of peptides obtained from porcine myofibrillar proteins by protease treatment. J. Agric. Food Chem. 2003, 51, 3661–3667. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Zhao, M.; Shi, J.; Wang, J.; Jiang, Y.; Cui, C.; Kakuda, Y.; Xue, S.J. Purification and identification of antioxidant peptides from grass carp muscle hydrolysates by consecutive chromatography and electrospray ionization-mass spectrometry. Food Chem. 2008, 108, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Iranzo, O. Manganese complexes displaying superoxide dismutase activity: A balance between different factors. Bioorg. Chem. 2011, 39, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Westbroek, P.; Marin, F. A marriage of bone and nacre. Nature 1998, 392, 861–862. [Google Scholar] [CrossRef]
- Rousseau, M.; Pereira-Mouriès, L.; Almeida, M.-J.; Milet, C.; Lopez, E. The water-soluble matrix fraction from the nacre of Pinctada maxima produces earlier mineralization of MC3T3-E1 mouse pre-osteoblasts. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2003, 135, 1–7. [Google Scholar] [CrossRef]
- Lakshminarayanan, R.; Chi-Jin, E.O.; Loh, X.J.; Kini, R.M.; Valiyaveettil, S. Purification and Characterization of a Vaterite-Inducing Peptide, Pelovaterin, from the Eggshells of Pelodiscus sinensis (Chinese Soft-Shelled Turtle). Biomacromolecules 2005, 6, 1429–1437. [Google Scholar] [CrossRef]
- Lakshminarayanan, R.; Loh, X.J.; Gayathri, S.; Sindhu, S.; Banerjee, Y.; Kini, R.M.; Valiyaveettil, S. Formation of Transient Amorphous Calcium Carbonate Precursor in Quail Eggshell Mineralization: An In Vitro Study. Biomacromolecules 2006, 7, 3202–3209. [Google Scholar] [CrossRef]
- Wang, L.; Sondi, I.; Matijević, E. Preparation of Uniform Needle-Like Aragonite Particles by Homogeneous Precipitation. J. Colloid Interface Sci. 1999, 218, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Nan, Z.; Chen, X.; Yang, Q.; Wang, X.; Shi, Z.; Hou, W. Structure transition from aragonite to vaterite and calcite by the assistance of SDBS. J. Colloid Interface Sci. 2008, 325, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Vecht, A.; Ireland, T.G. The role of vaterite and aragonite in the formation of pseudo-biogenic carbonate structures: Implications for Martian exobiology. Geochim. Cosmochim. Acta 2000, 64, 2719–2725. [Google Scholar] [CrossRef]
- Grasby, S.E. Naturally precipitating vaterite (μ-CaCO3) spheres: Unusual carbonates formed in an extreme environment. Geochim. Cosmochim. Acta 2003, 67, 1659–1666. [Google Scholar] [CrossRef]
- Fan, Y.W.; Wang, R.Z. Submicrometer-Sized Vaterite Tubes Formed through Nanobubble-Templated Crystal Growth. Adv. Mater. 2005, 17, 2384–2388. [Google Scholar] [CrossRef]
- Ma, Y.; Gao, Y.; Feng, Q. Characterization of organic matrix extracted from fresh water pearls. Mater. Sci. Eng. C 2011, 31, 1338–1342. [Google Scholar] [CrossRef]
- Ma, Y.F.; Gao, Y.H.; Feng, Q.L. Effects of pH and temperature on CaCO3 crystallization in aqueous solution with water soluble matrix of pearls. J. Cryst. Growth 2010, 312, 3165–3170. [Google Scholar] [CrossRef]
- Miyamoto, H.; Miyashita, T.; Okushima, M.; Nakano, S.; Morita, T.; Matsushiro, A. A carbonic anhydrase from the nacreous layer in oyster pearls. Proc. Natl. Acad. Sci. USA 1996, 93, 9657–9660. [Google Scholar] [CrossRef]
- Samata, T.; Hayashi, N.; Kono, M.; Hasegawa, K.; Horita, C.; Akera, S. A new matrix protein family related to the nacreous layer formation of Pinctada fucata. FEBS Lett. 1999, 462, 225–229. [Google Scholar] [CrossRef]
- Miyashita, T.; Takagi, R.; Okushima, M.; Nakano, S.; Miyamoto, H.; Nishikawa, E.; Matsushiro, A. Complementary DNA Cloning and Characterization of Pearlin, a New Class of Matrix Protein in the Nacreous Layer of Oyster Pearls. Mar. Biotechnol. 2000, 2, 409–418. [Google Scholar] [CrossRef]
- Michenfelder, M.; Fu, G.; Lawrence, C.; Weaver, J.C.; Wustman, B.A.; Taranto, L.; Evans, J.S.; Morse, D.E. Characterization of two molluscan crystal-modulating biomineralization proteins and identification of putative mineral binding domains. Biopolymers 2003, 70, 522–533. [Google Scholar] [CrossRef]
- Kim, I.W.; Collino, S.; Morse, D.E.; Evans, J.S. A Crystal Modulating Protein from Molluscan Nacre That Limits the Growth of Calcite in Vitro. Cryst. Growth Des. 2006, 6, 1078–1082. [Google Scholar] [CrossRef]
- Gong, N.; Li, Q.; Huang, J.; Fang, Z.; Zhang, G.; Xie, L.; Zhang, R. Culture of outer epithelial cells from mantle tissue to study shell matrix protein secretion for biomineralization. Cell Tissue Res. 2008, 333, 493–501. [Google Scholar] [CrossRef]
- Zhang, C.; Li, S.; Ma, Z.; Xie, L.; Zhang, R. A Novel Matrix Protein p10 from the Nacre of Pearl Oyster (Pinctada fucata) and Its Effects on Both CaCO3 Crystal Formation and Mineralogenic Cells. Mar. Biotechnol. 2006, 8, 624–633. [Google Scholar] [CrossRef]
- Pereira-Mouriès, L.; Almeida, M.-J.; Ribeiro, C.; Peduzzi, J.; Barthélemy, M.; Milet, C.; Lopez, E. Soluble silk-like organic matrix in the nacreous layer of the bivalve Pinctada maxima. Eur. J. Biochem. 2002, 269, 4994–5003. [Google Scholar] [CrossRef]
- Aizenberg, J.; Hanson, J.; Koetzle, T.F.; Weiner, S.; Addadi, L. Control of Macromolecule Distribution within Synthetic and Biogenic Single Calcite Crystals. J. Am. Chem. Soc. 1997, 119, 881–886. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, C.; Yan, Y.; Yang, H.F.; Li, J.F.; Bai, H.; Xi, G.C.; Liao, J. Identification of Pearl Powder Using Microscopic Infrared Reflectance Spectroscopy. Spectrosc. Spectr. Anal. 2014, 34, 2424–2428. [Google Scholar] [CrossRef]
- Liu, S.Q.; Wei, W.; Bai, Z.Y.; Wang, X.C.; Li, X.H.; Wang, C.X.; Liu, X.; Liu, Y.; Xu, C.H. Rapid identification of pearl powder from Hyriopsis cumingii by Tri-step infrared spectroscopy combined with computer vision technology. Spectroc. Acta Part A Mol. Biomol. Spectr. 2018, 189, 265–274. [Google Scholar] [CrossRef]
- Dhand, C.; Venkatesh, M.; Barathi, V.A.; Harini, S.; Bairagi, S.; Goh Tze Leng, E.; Muruganandham, N.; Low, K.Z.W.; Fazil, M.H.U.T.; Loh, X.J.; et al. Bio-inspired crosslinking and matrix-drug interactions for advanced wound dressings with long-term antimicrobial activity. Biomaterials 2017, 138, 153–168. [Google Scholar] [CrossRef] [PubMed]
- Opalenik, S.R.; Davidson, J.M. Fibroblast differentiation of bone marrow-derived cells during wound repair. FASEB J. 2005, 19, 1561–1563. [Google Scholar] [CrossRef] [PubMed]
- Rhett, J.M.; Ghatnekar, G.S.; Palatinus, J.A.; O’Quinn, M.; Yost, M.J.; Gourdie, R.G. Novel therapies for scar reduction and regenerative healing of skin wounds. Trends Biotechnol. 2008, 26, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.J.; Fazil, M.H.U.T.; Dhand, C.; Venkatesh, M.; Goh, E.T.L.; Harini, S.; Eugene, C.; Lim, R.R.; Ramakrishna, S.; Chaurasia, S.S.; et al. Insight into membrane selectivity of linear and branched polyethylenimines and their potential as biocides for advanced wound dressings. Acta Biomater. 2016, 37, 155–164. [Google Scholar] [CrossRef]
- Mayandi, V.; Wen Choong, A.C.; Dhand, C.; Lim, F.P.; Aung, T.T.; Sriram, H.; Dwivedi, N.; Periayah, M.H.; Sridhar, S.; Fazil, M.H.U.T.; et al. Multifunctional Antimicrobial Nanofiber Dressings Containing ε-Polylysine for the Eradication of Bacterial Bioburden and Promotion of Wound Healing in Critically Colonized Wounds. ACS Appl. Mater. Interfaces 2020, 12, 15989–16005. [Google Scholar] [CrossRef]
- Neri, S.; Miyashita, T.; Hashimoto, H.; Suda, Y.; Ishibashi, M.; Kii, H.; Watanabe, H.; Kuwata, T.; Tsuboi, M.; Goto, K.; et al. Fibroblast-led cancer cell invasion is activated by epithelial–mesenchymal transition through platelet-derived growth factor BB secretion of lung adenocarcinoma. Cancer Lett. 2017, 395, 20–30. [Google Scholar] [CrossRef]
- Lopez, E.; Faou, A.L.; Borzeix, S.; Berland, S. Stimulation of rat cutaneous fibroblasts and their synthetic activity by implants of powdered nacre (mother of pearl). Tissue Cell 2000, 32, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, M.; Bédouet, L.; Lati, E.; Gasser, P.; Le Ny, K.; Lopez, E. Restoration of stratum corneum with nacre lipids. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2006, 145, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Hatano, H.; Itasaka, O. Biochemical Studies on Pearl. IX. Amino Acid Composition of Conchiolin in Pearl and Shell. Bull. Chem. Soc. Jpn. 1960, 33, 543–545. [Google Scholar] [CrossRef]
- Jian-Ping, D.; Jun, C.; Yu-Fei, B.; Bang-Xing, H.; Shang-Bin, G.; Li-Li, J. Effects of pearl powder extract and its fractions on fibroblast function relevant to wound repair. Pharm. Biol. 2010, 48, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.A.; Zhang, W.B.; Fan, H.; Xu, P. Water-soluble nano-pearl powder promotes MC3T3-E1 cell differentiation by enhancing autophagy via the MEK/ERK signaling pathway. Mol. Med. Rep. 2018, 18, 993–1000. [Google Scholar] [CrossRef]
- Yang, Y.-L.; Chang, C.-H.; Huang, C.-C.; Liu, H.-W. Anti-inflammation and anti-apoptosis effects of pearl extract gel on UVB irradiation HaCaT cells. Bio-Med. Mater. Eng. 2015, 26, S139–S145. [Google Scholar] [CrossRef]
- Zhao, G.-R.; Xiang, Z.-J.; Ye, T.-X.; Yuan, Y.-J.; Guo, Z.-X. Antioxidant activities of Salvia miltiorrhiza and Panax notoginseng. Food Chem. 2006, 99, 767–774. [Google Scholar] [CrossRef]
- Hsieh, C.-C.; Liao, C.-C.; Liao, Y.-C.; Hwang, L.S.; Wu, L.-Y.; Hsieh, S.-C. Proteomic changes associated with metabolic syndrome in a fructose-fed rat model. J. Food Drug Anal. 2016, 24, 754–761. [Google Scholar] [CrossRef]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Shao, D.-Z.; Wang, C.-K.; Hwang, H.-J.; Hung, C.-H.; Chen, Y.-W. Abstracts: Comparison of hydration, tyrosinase resistance, and antioxidant activation in three kinds of pearl powders. Int. J. Cosmet. Sci. 2010, 32, 396. [Google Scholar] [CrossRef]
- Lee, M.; Park, S.-J.; Jeong, C.; Jang, S.I.; Han, J.; Kim, B.J.; Kim, E. Perception of the blue light effect on Korean women’s faces using the blue pearl pigment. Ski. Res. Technol. 2020, 26, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.S.; Chang, J.H.; Wu, J.S.B. Calcium bioavailability of nanonized pearl powder for adults. J. Food Sci. 2008, 73, H246–H251. [Google Scholar] [CrossRef]
- Xia, M.; Huang, D.L.; Tong, Y.M.; Lin, J. Pearl powder reduces sleep disturbance stress response through regulating proteomics in a rat model of sleep deprivation. J. Cell. Mol. Med. 2020, 24, 4956–4966. [Google Scholar] [CrossRef]
- Jarcho, M. Calcium phosphate ceramics as hard tissue prosthetics. Clin. Orthop. Relat. Res. 1981, 157, 259–278. [Google Scholar] [CrossRef]
- Yu, X.; Ibrahim, M.; Liu, Z.; Yang, H.; Tan, L.; Yang, K. Biofunctional Mg coating on PEEK for improving bioactivity. Bioact. Mater. 2018, 3, 139–143. [Google Scholar] [CrossRef]
- Ning, C.Q.; Zhou, Y. In vitro bioactivity of a biocomposite fabricated from HA and Ti powders by powder metallurgy method. Biomaterials 2002, 23, 2909–2915. [Google Scholar] [CrossRef]
- Shen, Y.; Zhu, J.; Zhang, H.; Zhao, F. In vitro osteogenetic activity of pearl. Biomaterials 2006, 27, 281–287. [Google Scholar] [CrossRef]
- Liu, Y.S.; Huang, Q.L.; Feng, Q.L.; Hu, N.M.; Albert, O. Structural features and mechanical properties of PLLA/pearl powder scaffolds. J. Mech. Med. Biol. 2013, 13, 1350020. [Google Scholar] [CrossRef]
- Shuai, C.; Guo, W.; Gao, C.; Yang, Y.; Xu, Y.; Liu, L.; Qin, T.; Sun, H.; Yang, S.; Feng, P.; et al. Calcium silicate improved bioactivity and mechanical properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) scaffolds. Polymers 2017, 9, 175. [Google Scholar] [CrossRef]
- Pereira Mouriès, L.; Almeida, M.-J.; Milet, C.; Berland, S.; Lopez, E. Bioactivity of nacre water-soluble organic matrix from the bivalve mollusk Pinctada maxima in three mammalian cell types: Fibroblasts, bone marrow stromal cells and osteoblasts. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2002, 132, 217–229. [Google Scholar] [CrossRef]
- Flausse, A.; Henrionnet, C.; Dossot, M.; Dumas, D.; Hupont, S.; Pinzano, A.; Mainard, D.; Galois, L.; Magdalou, J.; Lopez, E.; et al. Osteogenic differentiation of human bone marrow mesenchymal stem cells in hydrogel containing nacre powder. J. Biomed. Mater. Res. Part A 2013, 101, 3211–3218. [Google Scholar] [CrossRef] [PubMed]
- Lamghari, M.; Almeida, M.J.; Berland, S.; Huet, H.; Laurent, A.; Milet, C.; Lopez, E. Stimulation of bone marrow cells and bone formation by nacre: In vivo and in vitro studies. Bone 1999, 25, 91S–94S. [Google Scholar] [CrossRef]
- Asvanund, P.; Chunhabundit, P.; Suddhasthira, T. Potential induction of bone regeneration by nacre: An in vitro study. Implant Dent. 2011, 20, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Gerhard, E.M.; Wang, W.; Li, C.; Guo, J.; Ozbolat, I.T.; Rahn, K.M.; Armstrong, A.D.; Xia, J.; Qian, G.; Yang, J. Design strategies and applications of nacre-based biomaterials. Acta Biomater. 2017, 54, 21–34. [Google Scholar] [CrossRef]
- Li, X.N.; Xu, P.; Cheng, Y.N.; Zhang, W.B.; Zheng, B.; Wang, Q.Z. Nano-pearl powder/chitosan-hyaluronic acid porous composite scaffold and preliminary study of its osteogenesis mechanism. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 111, 9. [Google Scholar] [CrossRef]
- Zhang, X.; Du, X.Y.; Li, D.J.; Ao, R.G.; Yu, B.; Yu, B.Q. Three Dimensionally Printed Pearl Powder/Poly-Caprolactone Composite Scaffolds for Bone Regeneration. J. Biomater. Sci. Polym. Ed. 2018, 29, 1686–1700. [Google Scholar] [CrossRef]
- Dai, J.M.; Bai, J.J.; Jin, J.H.; Yang, S.L.; Li, G. Stimulation by pearl of mineralization and biocompatibility of PLA. Adv. Eng. Mater. 2015, 17, 1691–1697. [Google Scholar] [CrossRef]
- Dai, J.M.; Yang, S.L.; Jin, J.H.; Li, G. Electrospinning of PLA/pearl powder nanofibrous scaffold for bone tissue engineering. RSC Adv. 2016, 6, 106798–106805. [Google Scholar] [CrossRef]
- Liu, Y.S.; Huang, Q.L.; Feng, Q.L. 3D scaffold of PLLA/pearl and PLLA/nacre powder for bone regeneration. Biomed. Mater. 2013, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.S.; Huang, Q.L.; Kienzle, A.; Müller, W.E.G.; Feng, Q.L. In vitro degradation of porous PLLA/pearl powder composite scaffolds. Mater. Sci. Eng. C 2014, 38, 227–234. [Google Scholar] [CrossRef]
- Bai, J.J.; Dai, J.M.; Li, G. Electrospun composites of phbv/pearl powder for bone repairing. Prog. Nat. Sci. 2015, 25, 327–333. [Google Scholar] [CrossRef]
- Wu, Y.N.; Ding, Z.W.; Ren, H.H.; Ji, M.Z.; Yan, Y.G. Preparation, Characterization and In Vitro Biological Evaluation of a Novel Pearl Powder/Poly-Amino Acid Composite as a Potential Substitute for Bone Repair and Reconstruction. Polymers 2019, 11, 831. [Google Scholar] [CrossRef]
- Deng, Y.H.; Li, G.; Song, W.L.; Jiang, J.M. Preparation and properties of pearl powder/polypropylene composites and their biocompatibility. Bio-Med. Mater. Eng. 2015, 26, S27–S34. [Google Scholar] [CrossRef] [PubMed]
- Du, X.Y.; Yu, B.; Pei, P.; Ding, H.F.; Yu, B.Q.; Zhu, Y.F. 3D printing of pearl/CaSO4 composite scaffolds for bone regeneration. J. Mat. Chem. B 2018, 6, 499–509. [Google Scholar] [CrossRef]
- Ding, Y.; Tang, S.; Yu, B.; Yan, Y.; Li, H.; Wei, J.; Su, J. In vitro degradability, bioactivity and primary cell responses to bone cements containing mesoporous magnesium–calcium silicate and calcium sulfate for bone regeneration. J. R. Soc. Interface 2015, 12, 20150779. [Google Scholar] [CrossRef] [PubMed]
- Horie, M.; Nishio, K.; Kato, H.; Endoh, S.; Fujita, K.; Nakamura, A.; Kinugasa, S.; Hagihara, Y.; Yoshida, Y.; Iwahashi, H. Evaluation of cellular influences caused by calcium carbonate nanoparticles. Chem. Biol. Interact. 2014, 210, 64–76. [Google Scholar] [CrossRef]
- Kün-Darbois, J.-D.; Libouban, H.; Camprasse, G.; Camprasse, S.; Chappard, D. In vivo osseointegration and erosion of nacre screws in an animal model. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020. [Google Scholar] [CrossRef]
- Chappard, D.; Kün-Darbois, J.-D.; Pascaretti-Grizon, F.; Camprasse, G.; Camprasse, S. Giant cells and osteoclasts present in bone grafted with nacre differ by nuclear cytometry evaluated by texture analysis. J. Mater. Sci. Mater. Med. 2019, 30, 100. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Loh, X.J. Recent advances of using polyhydroxyalkanoate-based nanovehicles as therapeutic delivery carriers. Wires Nanomed. Nanobiotechnol. 2017, 9, e1429. [Google Scholar] [CrossRef]
- Dai, J.M.; Jin, J.H.; Yang, S.L.; Li, G. Doxorubicin-loaded PLA/pearl electrospun nanofibrous scaffold for drug delivery and tumor cell treatment. Mater. Res. Express 2017, 4, 10. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loh, X.J.; Young, D.J.; Guo, H.; Tang, L.; Wu, Y.; Zhang, G.; Tang, C.; Ruan, H. Pearl Powder—An Emerging Material for Biomedical Applications: A Review. Materials 2021, 14, 2797. https://doi.org/10.3390/ma14112797
Loh XJ, Young DJ, Guo H, Tang L, Wu Y, Zhang G, Tang C, Ruan H. Pearl Powder—An Emerging Material for Biomedical Applications: A Review. Materials. 2021; 14(11):2797. https://doi.org/10.3390/ma14112797
Chicago/Turabian StyleLoh, Xian Jun, David James Young, Hongchen Guo, Liang Tang, Yunlong Wu, Guorui Zhang, Changming Tang, and Huajun Ruan. 2021. "Pearl Powder—An Emerging Material for Biomedical Applications: A Review" Materials 14, no. 11: 2797. https://doi.org/10.3390/ma14112797
APA StyleLoh, X. J., Young, D. J., Guo, H., Tang, L., Wu, Y., Zhang, G., Tang, C., & Ruan, H. (2021). Pearl Powder—An Emerging Material for Biomedical Applications: A Review. Materials, 14(11), 2797. https://doi.org/10.3390/ma14112797






