Kinetics and Thermodynamics Studies for Cadmium (II) Adsorption onto Functionalized Chitosan with Hexa-Decyl-Trimethyl-Ammonium Chloride
Abstract
1. Introduction
2. Experimental Part
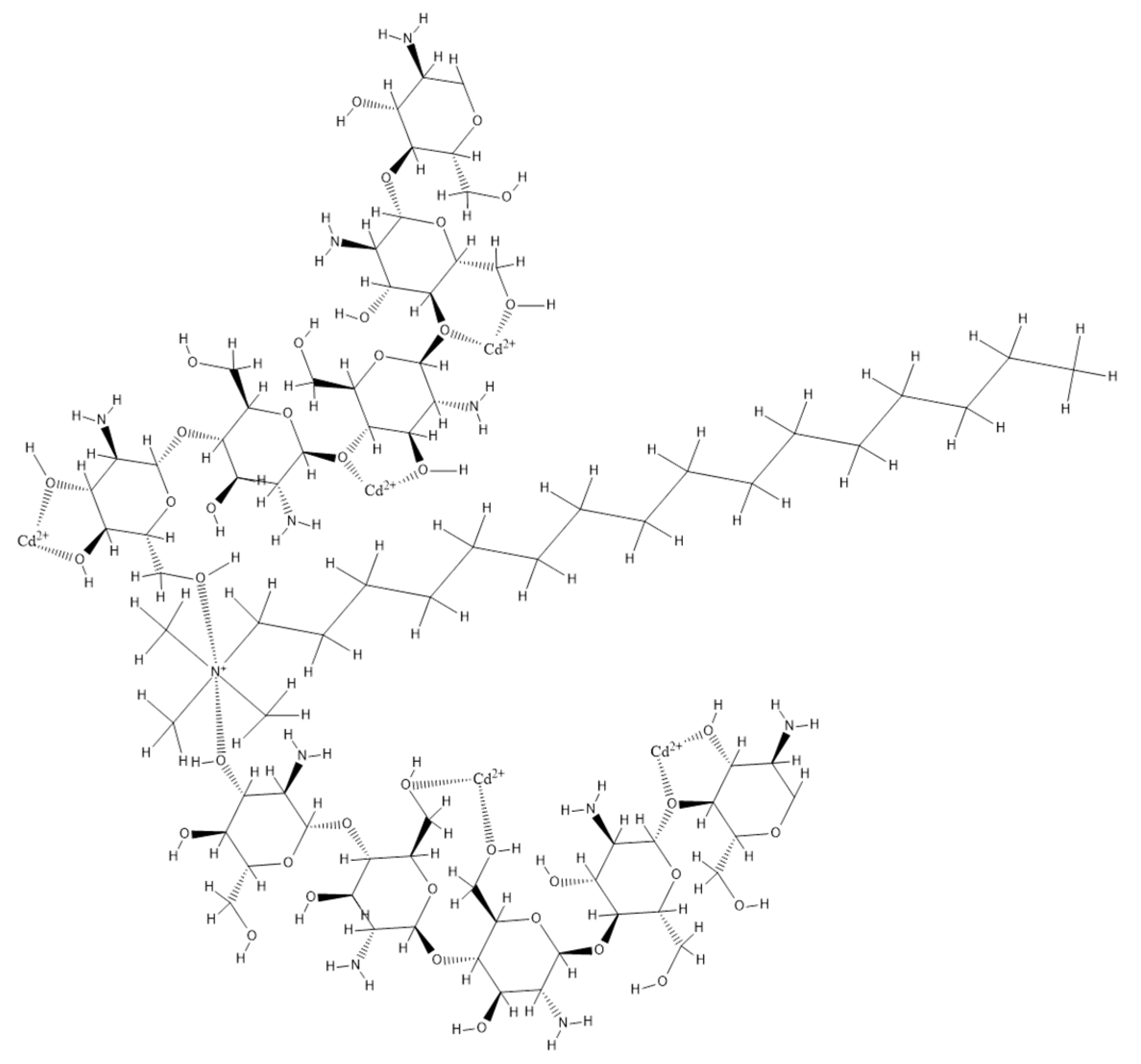
2.1. Obtaining the Biopolymer by Functionalization. Effect of Extractant Dosage
2.2. Characterization of the Functionalized Biopolymer
2.2.1. Batch Studies
- qe—the maximum absorption capacity (mg·g−1).
- C0—initial concentration of cadmium (II) in solution (mg·L−1).
- Ce—the equilibrium concentration of cadmium (II) in solution (mg·L−1).
- V—volume of the aqueous solution with cadmium (II) (L).
- m—mass of the adsorbent (g).
2.2.2. Sorption/Desorption Tests
3. Results and Discussion
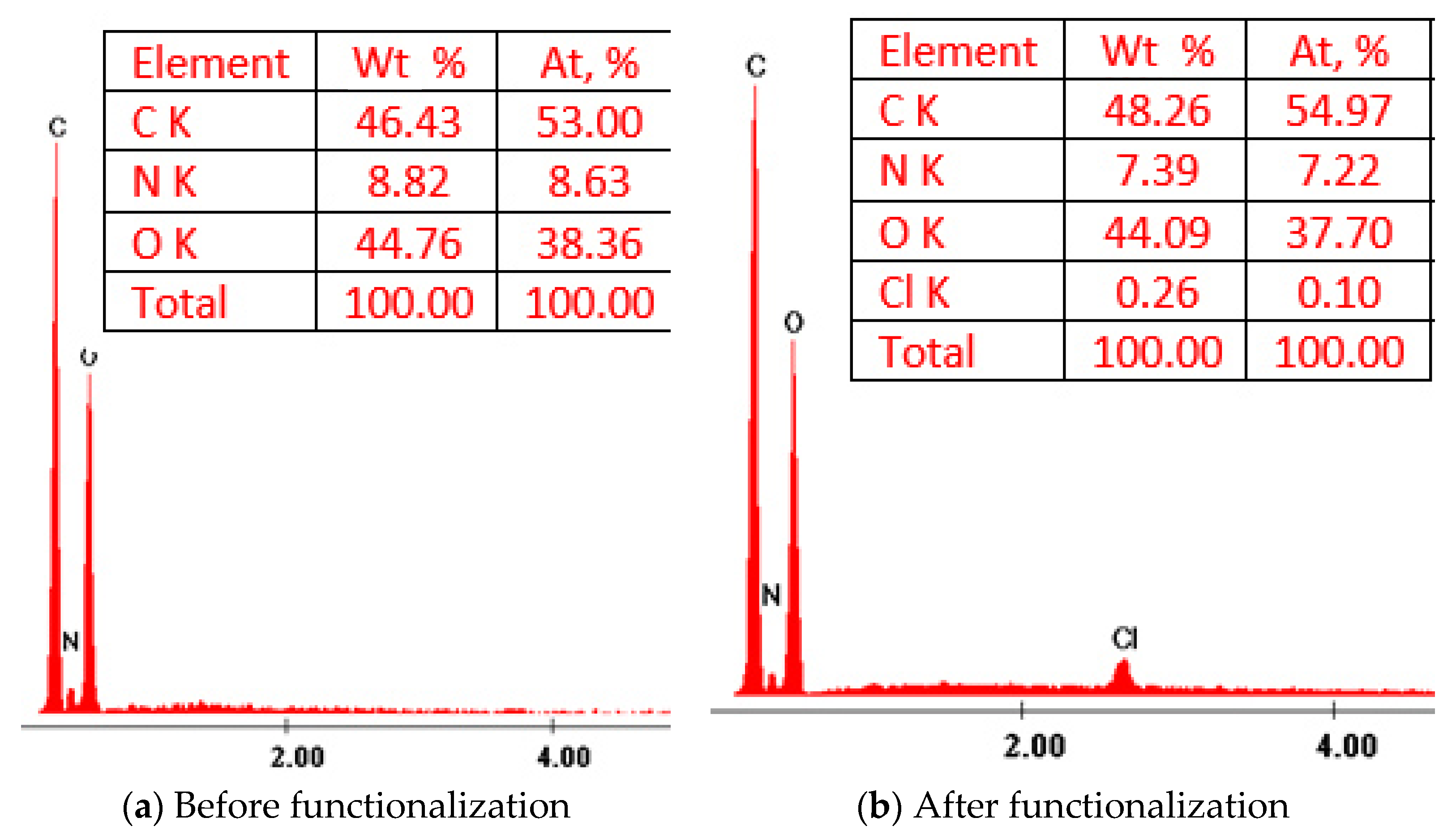
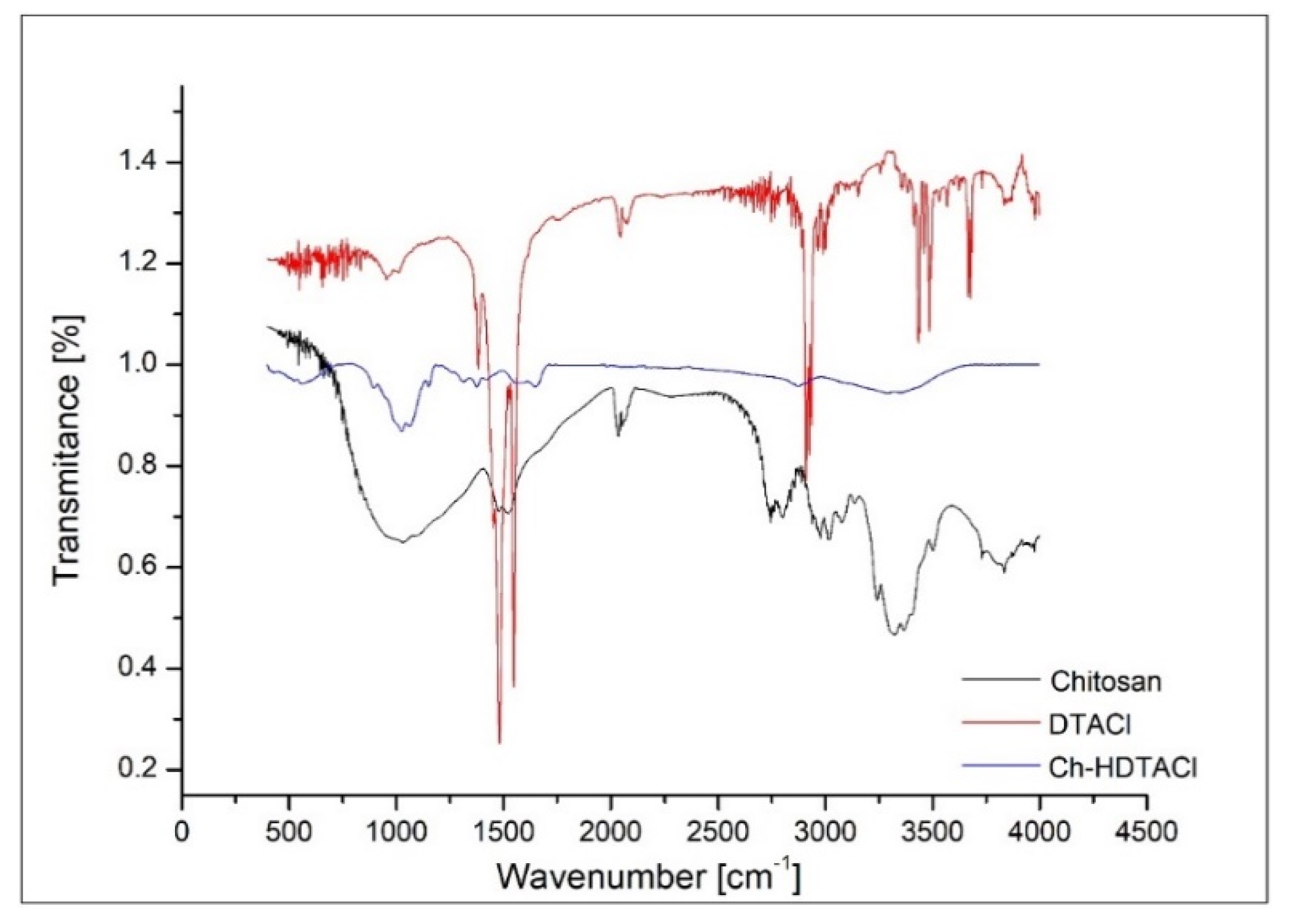
3.1. Characterization of the Functionalized Bio Sorbent
3.1.1. X-ray Energy Dispersive Spectroscopy Analysis
3.1.2. Scanning Electron Microscopic Studies
3.1.3. Fourier Transform Infrared Spectroscopy Analysis
3.2. Sorption Studies
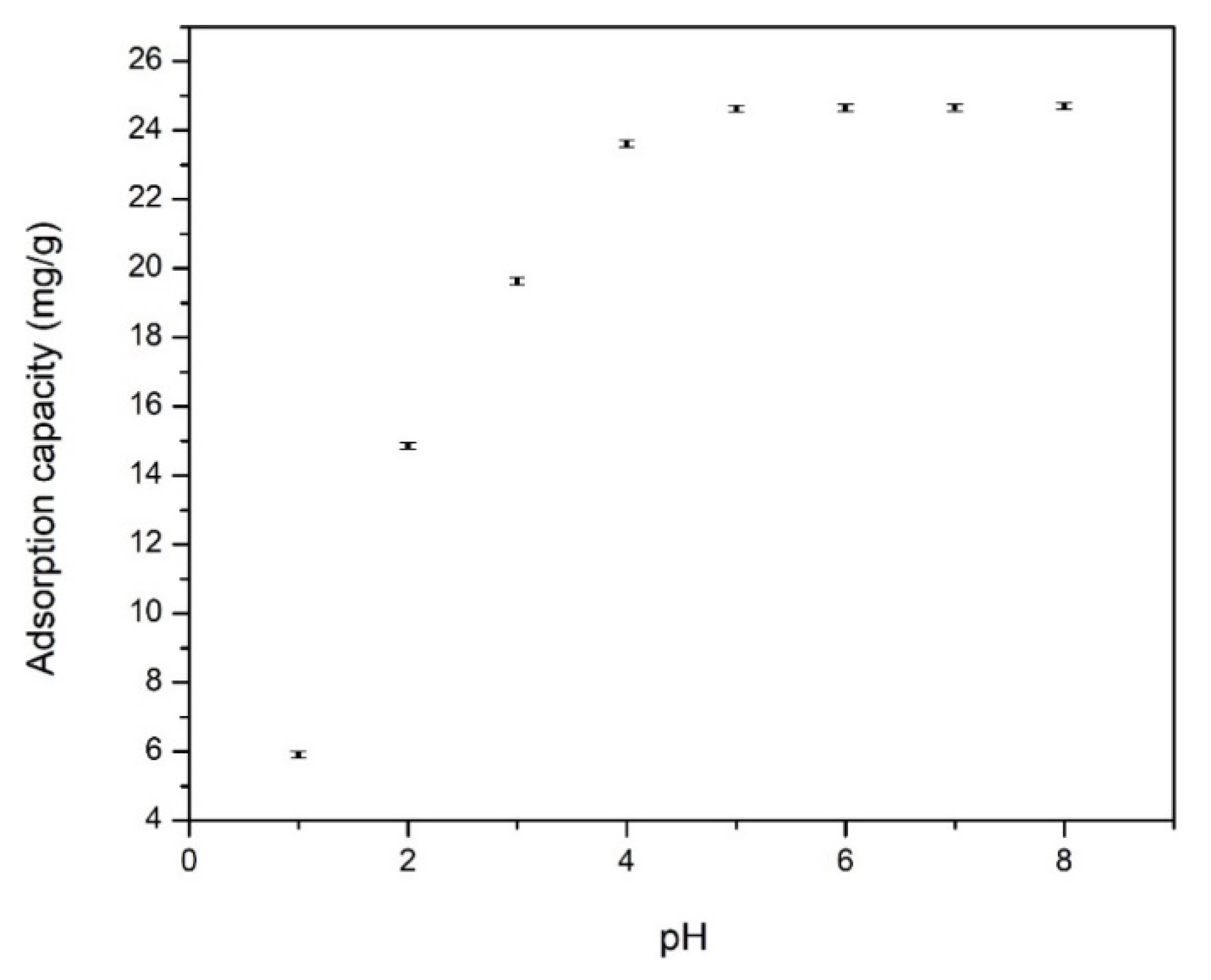
3.2.1. Effect of pH over Cd Adsorption on Functionalized Chitosan
3.2.2. Effect of Bio Adsorbent Dosage
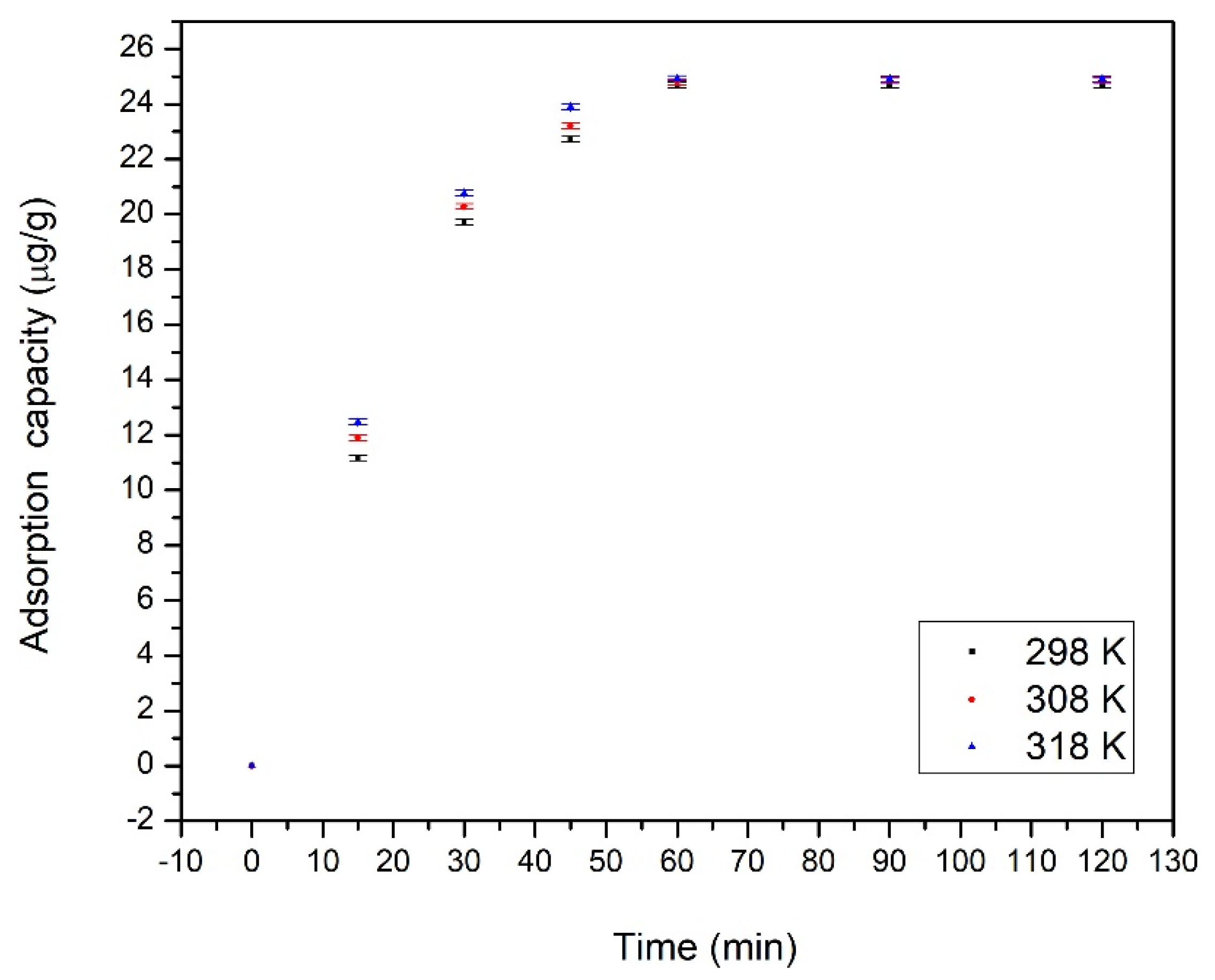
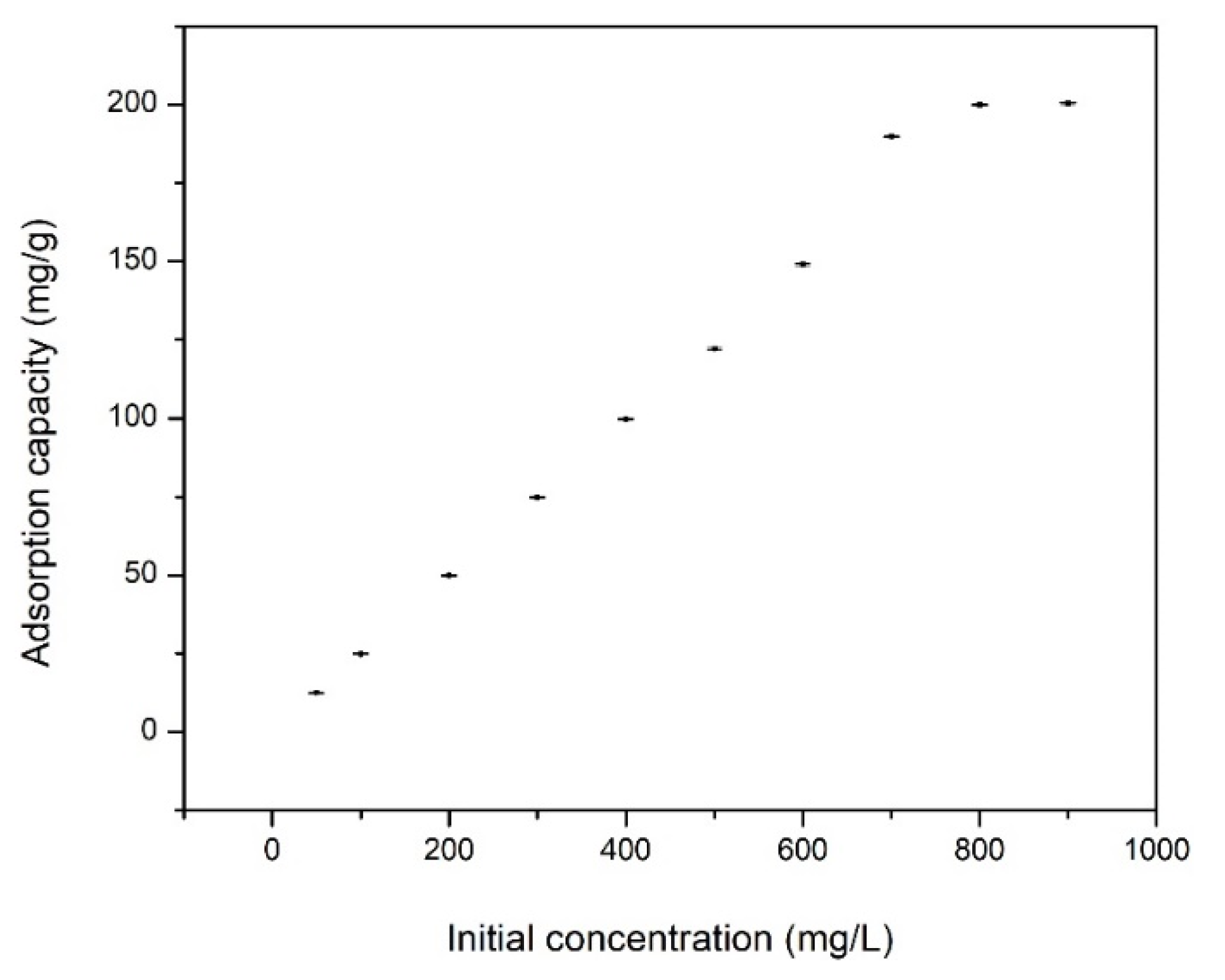
3.2.3. Effect of Contact Time, Temperature and Initial Cadmium Concentration
3.2.4. Adsorptions Kinetics
- qe—the adsorption capacity at equilibrium (mg/g),
- qt—is the adsorption capacity at time t,
- t—the contact time (min) and k1 is the adsorption rate constant (1/min).
- qe—the adsorption capacity at equilibrium (mg/g),
- qt—the adsorption capacity at time t,
- t—the contact time (min) and k2 the adsorption rate constant (g/mg∙min).
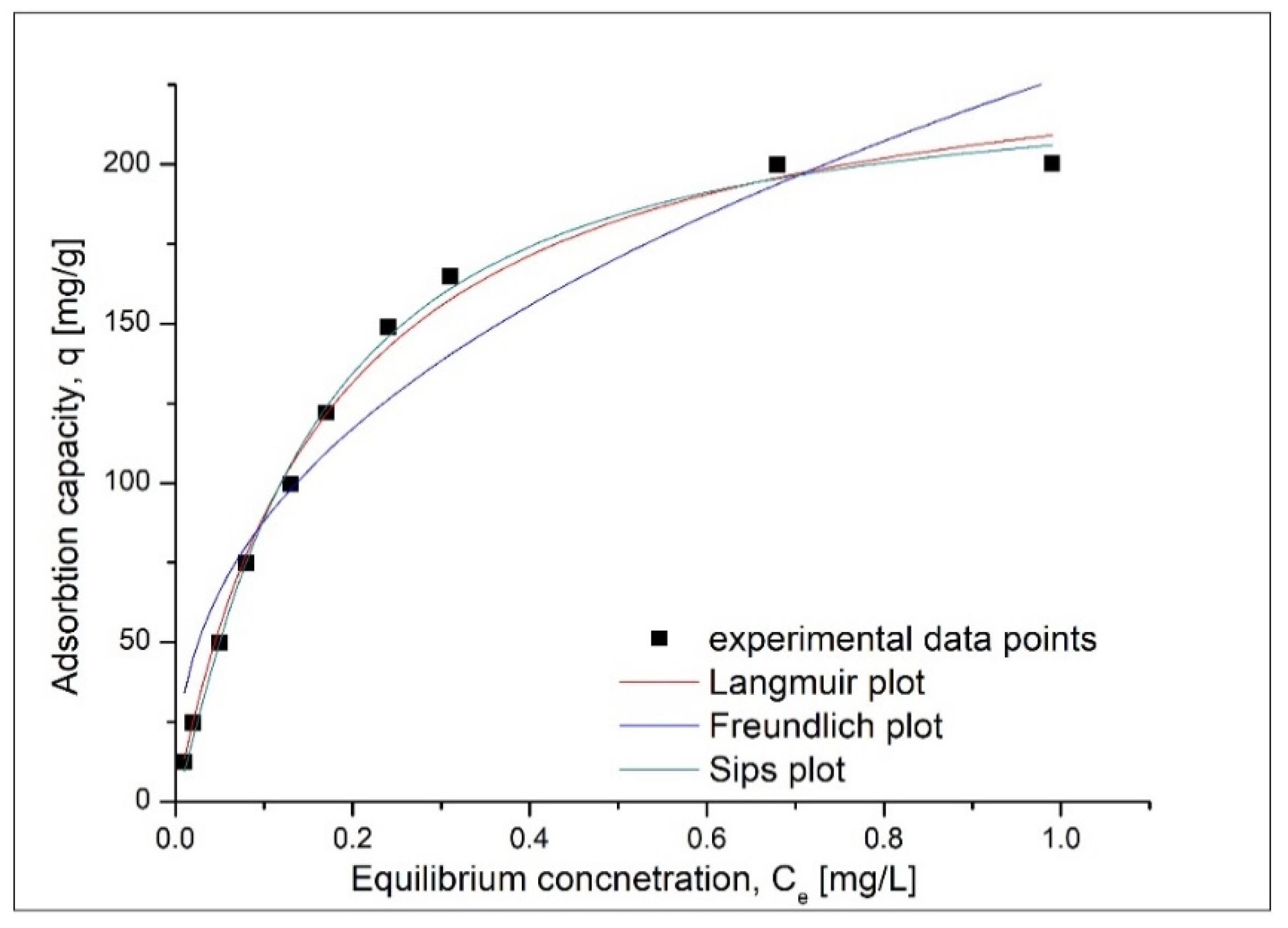
3.2.5. Adsorption Isotherm Modelling
- qe—the maximum adsorption capacity (mg·g−1),
- Ce—the equilibrium concentration of cadmium (II) in solution (mg·L−1),
- qL—Langmuir maximum adsorption capacity (mg·g−1),
- KL—Langmuir constant.
- qe—the equilibrium adsorption capacity (mg·g−1),
- Ce—the equilibrium concentration of adsorbent in the solution (mg·L−1),
- KF and nF—specific constants that are connected to the relative adsorption capacity of the adsorbent material and the intensity of adsorption.
- qS—the maximum adsorption capacity (mg·g−1),
- KS—constant related to the adsorption capacity of the adsorbent,
- nS—the heterogeneity factor.
3.2.6. Thermodynamic Analysis
- ΔG°—Gibbs free energy standard variation (kJ·mol−1),
- ΔH°—enthalpy variation (kJ·mol−1),
- ΔS°—entropy variation (J·mol−1∙K−1),
- T—absolute temperature (K).
- Kd—equilibrium constant,
- ΔH°—enthalpy variation (kJ·mol−1),
- ΔS°—entropy variation (J·mol−1∙K−1),
- T—absolute temperature (K),
- R—ideal gas constant (8.314 J·mol−1∙K−1).
3.2.7. Sorption/Desorption Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rafati-Rahimzadeh, M.; Rafati-Rahimzadeh, M.; Kazemi, S.; Moghadamnia, A. Cadmium toxicity and treatment: An update. Casp. J. Intern. Med. 2017, 8, 135–145. [Google Scholar]
- Goering, P.L.; Waalkes, M.P.; Klaassen, C.D. Toxicology of Cadmium, in Toxicology of Metals: Biochemical Aspects; Goyer, R.A., Cherian, M.G., Eds.; Springer: Berlin/Heidelberg, Berlin, 1995; pp. 189–214. [Google Scholar]
- Godt, J.; Scheidig, F.; Grosse-Siestrup, C.; Esche, V.; Brandenburg, P.; Reich, A.; Groneberg, D.A. The toxicity of cadmium and resulting hazards for human health. J. Occup. Med. Toxicol. 2006, 1, 22. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Du, Y. Cadmium and its neurotoxic effects. Oxidat. Med. Cell. Longev. 2013, 2013, 12. [Google Scholar] [CrossRef] [PubMed]
- Waalkes, M.P. Cadmium carcinogenesis in review. J. Inorg. Biochem. 2000, 79, 241–244. [Google Scholar] [CrossRef]
- Waalkes, M.P. Cadmium carcinogenesis. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2003, 533, 107–120. [Google Scholar] [CrossRef]
- Gupta, B.; Singh, I.; Mahandra, H. Extraction and separation studies on Pt(IV), Ir(III) and Rh(III) using sulphur containing extractant. Separat. Purif. Technol. 2014, 132, 102–109. [Google Scholar] [CrossRef]
- Waalkes, M.P.; Anver, M.; Diwan, B.A. Carcinogenic effects of cadmium in the noble (NBL/Cr) rat: Induction of pituitary, testicular, and injection site tumors and intraepithelial proliferative lesions of the dorsolateral prostate. Toxicolog. Sci. 1999, 52, 154–161. [Google Scholar] [CrossRef]
- Misra, R.R.; Smith, G.T.; Waalkes, M.P. Evaluation of the direct genotoxic potential of cadmium in four different rodent cell lines. Toxicology 1998, 126, 103–114. [Google Scholar] [CrossRef]
- Jha, I.N.; Iyengar, L.; Rao, A.V.S.P. Removal of cadmium using chitosan. J. Environ. Eng. 1988, 114, 962–974. [Google Scholar] [CrossRef]
- Naushad, M.; Al-Othman, Z.A.; Islam, M. Adsorption of cadmium ion using a new composite cation-exchanger polyaniline Sn(IV) silicate: Kinetics, thermodynamic and isotherm studies. Int. J. Environ. Sci. Technol. 2013, 10, 567–578. [Google Scholar] [CrossRef]
- Naushad, M.; Mittal, A.; Rathore, M.; Gupta, V. Ion-exchange kinetic studies for Cd(II), Co(II), Cu(II), and Pb(II) metal ions over a composite cation exchanger. Desalin. Water Treat. 2015, 54, 2883–2890. [Google Scholar] [CrossRef]
- Pyrzynska, K. Removal of cadmium from wastewaters with low-cost adsorbents. J. Environ. Chem. Eng. 2019, 7, 102795. [Google Scholar] [CrossRef]
- Naushad, M.; Ahamadb, T.; Alothmana, Z.A.; Sharc, M.A.; AlHokbanyb, N.S.; Alshehri, S.M. Synthesis, characterization and application of curcumin formaldehyde resin for the removal of Cd2+ from wastewater: Kinetics, isotherms and thermodynamic studies. J. Ind. Eng. Chem. 2015, 29, 78–86. [Google Scholar] [CrossRef]
- Suhas; Gupta, V.K.; Carrott, P.J.M.; Singh, R.; Chaudhary, M.; Kushwaha, S. Cellulose: A review as natural, modified and activated carbon adsorbent. Biores. Technol. 2016, 216, 1066–1076. [Google Scholar] [CrossRef]
- Płotka-Wasylka, J.; Płotka-Wasylka, J.; Szczepańska, N.; de la Guardia, M.; Namieśnik, J. Modern trends in solid phase extraction: New sorbent media. TrAC Trends Anal. Chem. 2016, 77, 23–43. [Google Scholar] [CrossRef]
- Speltini, A.; Scalabrini, A.; Maraschi, F.; Sturini, M.; Profumo, A. Newest applications of molecularly imprinted polymers for extraction of contaminants from environmental and food matrices: A review. Anal. Chim. Acta 2017, 974, 1–26. [Google Scholar] [CrossRef]
- Xiao, J.P.; Wang, J.J.; Fan, H.L.; Zhou, Q.X.; Liu, X.D. Recent advances of adsorbents in solid phase extraction for environmental samples. Int. J. Environ. Anal. Chem. 2016, 96, 407–435. [Google Scholar] [CrossRef]
- Attar, K.; Bouazzaa, D.; Miloudia, H.; Tayeba, A.; Boosb, A.; Sastrec, A.M.; Demey, H. Cadmium removal by a low-cost magadiite-based material: Characterization and sorption applications. J. Environ. Chem. Eng. 2018, 6, 5351–5360. [Google Scholar] [CrossRef]
- Kumar, R.; Chawla, J.; Kaur, I. Removal of cadmium ion from wastewater by carbon-based nanosorbents: A review. J. Water Health 2014, 13, 18–33. [Google Scholar] [CrossRef]
- Sajid, M.; Basheer, C. Layered double hydroxides: Emerging sorbent materials for analytical extractions. TrAC Trends Anal. Chem. 2016, 75, 174–182. [Google Scholar] [CrossRef]
- Dubey, S.; Banerjee, S.; Upadhyay, S.N.; Sharma, Y.C. Application of common nano-materials for removal of selected metallic species from water and wastewaters: A critical review. J. Mol. Liq. 2017, 240, 656–677. [Google Scholar] [CrossRef]
- Ihsanullah; Abbas, A.; Al-Amer, A.M.; Laoui, T.; Al-Marri, M.J.; Nasser, M.S.; Khraisheh, M.; Atieh, M.A. Heavy metal removal from aqueous solution by advanced carbon nanotubes: Critical review of adsorption applications. Separat. Purif. Technol. 2016, 157, 141–161. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Graphene oxide and its application as an adsorbent for wastewater treatment. J. Chem. Technol. Biotechnol. 2014, 89, 196–205. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Kostoglou, M. Green adsorbents for wastewaters: A critical review. Materials 2014, 7, 333–364. [Google Scholar] [CrossRef] [PubMed]
- Pyrzynska, K. Use of nanomaterials in sample preparation. TrAC Trends Anal. Chem. 2013, 43, 100–108. [Google Scholar] [CrossRef]
- Zafar, R.; Zia, K.M.; Tabasum, S.; Jabeen, F.; Noreen, A.; Zuber, M. Polysaccharide based bionanocomposites, properties and applications: A review. Int. J. Biol. Macromol. 2016, 92, 1012–1024. [Google Scholar] [CrossRef]
- Cai, Z.; Li, J.L.; Liew, K.Y.; Hu, J.C. Effect of La2O3-dopping on the Al2O3 supported cobalt catalyst for Fischer-Tropsch synthesis. J. Mol. Catal. A Chem. 2010, 330, 10–17. [Google Scholar] [CrossRef]
- Ezoddin, M.; Shemirani, F.; Abdi, K.; Saghezchi, M.K.; Jamali, M.R. Application of modified nano-alumina as a solid phase extraction sorbent for the preconcentration of Cd and Pb in water and herbal samples prior to flame atomic absorption spectrometry determination. J. Hazard. Mater. 2010, 178, 900–905. [Google Scholar] [CrossRef]
- Gabor, A.; Davidescua, C.M.; Negreaa, A.; Ciopeca, M.; Munteana, C.; Negreaa, P.; Ianasib, C.; Butnariu, M. Magnesium silicate doped with environmentally friendly extractants used for rare earth elements adsorption. Desalin. Water Treat. 2017, 63, 124–134. [Google Scholar]
- Mihailescu, M.; Negrea, A.; Ciopec, M.; Davidescu, C.M.; Negrea, P.; Duţeanu, N.; Rusu, G. Gold (III) adsorption from dilute waste solutions onto Amberlite XAD7 resin modified with L-glutamic acid. Sci. Rep. 2019, 9, 13. [Google Scholar] [CrossRef]
- Akita, S.; Hirano, K.; Ohashi, Y.; Takeuchi, H. Equilibrium distribution of palladium(II) between hydrochloric acid solution and a macromolecular resin containing tri-n-octylamine. Solvent Extr. Ion Exch. 1993, 11, 797–810. [Google Scholar] [CrossRef]
- Villaescusa, I.; Salvadó, V.; de Pablo, J. Liquid-liquid and solid-liquid extraction of gold by trioctylmethylammonium chloride (TOMAC1) dissolved in toluene and impregnated on amberlite XAD-2 resin. Hydrometallurgy 1996, 41, 303–311. [Google Scholar] [CrossRef]
- Horwitz, E.P.; Dietz, M.L.; Chiarizia, R.; Diamond, H.; Maxwel lIII, S.L.; Nelson, M.R. Separation and preconcentration of actinides by extraction chromatography using a supported liquid anion exchanger: Application to the characterization of high-level nuclear waste solutions. Anal. Chim. Acta 1995, 310, 63–78. [Google Scholar] [CrossRef]
- Gabor, A.; Davidescu, C.M.; Negrea, A.; Ciopec, M.; Negrea, P.; Butnariu, M. Separation of La (III) using functionalized material on fixed bed column. Rev. Chim. 2016, 67, 2225–2227. [Google Scholar]
- Ciopec, M.; Gabor, A.; Davidescu, C.M.; Negrea, A.; Negrea, P.; Duteanu, N. Eu (III) removal by tetrabutylammonium di-hydrogen phosphate (TBAH2P) functionalized polymers. Arab. J. Chem. 2020, 13, 3534–3545. [Google Scholar] [CrossRef]
- Madala, S.; Nadavala, S.K.; Vudagandla, S.; Boddu, V.M.; Abburi, K. Equilibrium, kinetics and thermodynamics of Cadmium (II) biosorption on to composite chitosan biosorbent. Arab. J. Chem. 2017, 10, S1883–S1893. [Google Scholar] [CrossRef]
- Lagergren, S. About the theory of so-called adsorption of soluble substabces. Kungl. Svenska Vetenskapsakademiens Handlingar—1898 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–470. [Google Scholar]
- Sips, R. On the structure of a catalyst surface. J. Chem. Phys. 1948, 16, 490–495. [Google Scholar] [CrossRef]
- Hasan, S.; Krishnaiah, A.; Ghosh, T.K.; Viswanath, D.S.; Boddu, V.M.; Smith, E.D. Adsorption of divalent Cadmium (Cd(II)) from aqueous solutions onto chitosan-coated perlite beads. Ind. Eng. Chem. Res. 2006, 45, 5066–5077. [Google Scholar] [CrossRef]
- Hydari, S.; Sharififard, H.; Nabavinia, M.; Parvizi, M.R. A comparative investigation on removal performances of commercial activated carbon, chitosan biosorbent and chitosan/activated carbon composite for cadmium. Chem. Eng. J. 2012, 193-194, 276–282. [Google Scholar] [CrossRef]
- Zhang, G.; Qu, R.J.; Sun, C.M.; Ji, C.N.; Chen, H.; Wang, C.H.; Niu, Y.Z. Adsorption for metal ions of chitosan coated cotton fiber. J. Appl. Polymer Sci. 2008, 110, 2321–2327. [Google Scholar] [CrossRef]
- Monier, M.; Abdel-Latif, D.A. Preparation of cross-linked magnetic chitosan-phenylthiourea resin for adsorption of Hg(II), Cd(II) and Zn(II) ions from aqueous solutions. J. Hazard. Mater. 2012, 209-210, 240–249. [Google Scholar] [CrossRef]
- Atkins, P.; de Paula, J.; Keeler, J. Atkins’ Physical Chemistry; Oxford University Press: Oxford, UK, 2005; p. 1008. [Google Scholar]
- Özcan, A.; Özcan, A.S.; Tunali, S.; Akar, T.; Kiran, I. Determination of the equilibrium, kinetic and thermodynamic parameters of adsorption of copper(II) ions onto seeds of Capsicum annuum. J. Hazard. Mater. 2005, 124, 200–208. [Google Scholar] [CrossRef]

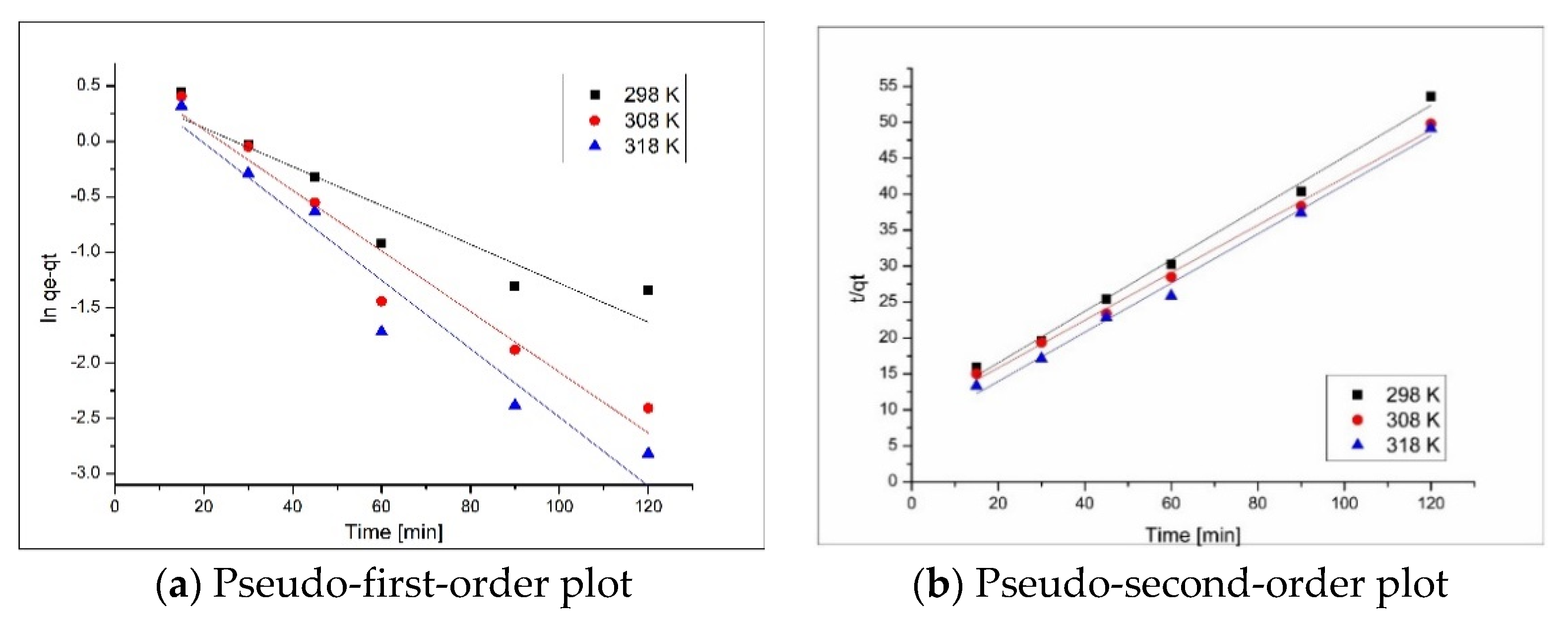

| Pseudo-First Order | ||||
| Temperature (K) | qe,exp (mg·g−1) | k1 (min−1) | qe,calc (mg·g−1) | R2 |
| 298 | 24.70 | 0.0378 | 12.36 | 0.9783 |
| 308 | 24.87 | 0.0465 | 13.80 | 0.9829 |
| 318 | 24.92 | 0.0509 | 13.32 | 0.9834 |
| Pseudo-Second Order | ||||
| Temperature (K) | qe,exp (mg·g−1) | k2 (g·mg−1∙min−1) | qe,calc (mg·g−1) | R2 |
| 298 | 24.70 | 1510.83 | 28.81 | 0.9904 |
| 308 | 24.87 | 1670.06 | 28.57 | 0.9904 |
| 318 | 24.92 | 1838.49 | 28.18 | 0.991 |
| Langmuir Isotherm | |||
| qm,exp (mg·g−1) | KL (L·mg−1) | qL (mg·g−1) | R2 |
| 204.3 | 5.76 | 245.8 | 0.9899 |
| Freundlich Isotherm | |||
| KF (mg·g−1) | 1/nF | R2 | |
| 227.3 | 0.412 | 0.91612 | |
| Sips Isotherm | |||
| KS | qS (mg·g−1) | 1/nS | R2 |
| 0.171 | 228.1 | 9.44 | 0.99551 |
| Adsorbent | Adsorption Capacity, mg·g−1 | Adsorption Conditions | Reference |
|---|---|---|---|
| Chitosan (80% deacetylation) | 6.63 | pH = 6.0, T = 313 K | [10] |
| Chitosan perlite | 179.6 | pH = 6.0, T = 313 K | [43] |
| Chitosan/activated carbon composite | 52.6 | pH = 6.0, T = 313 K | [44] |
| Chitosan coated cotton fiber | 15.74 | pH = 6.5, T = 313 K | [45] |
| Chitosan-phenylthiourea resin | 120 | pH = 5.0, T = 313 K | [46] |
| Ch-HDTACl | 204.3 | pH > 4.0, T = 298 K | Present paper |
| ΔH° (kJ·mol−1) | ΔS° (J·mol−1∙K−1) | ΔG° (kJ·mol−1) | R2 | ||
|---|---|---|---|---|---|
| 298 K | 308 K | 318 K | |||
| 31.1 | 130.4 | −6.4 | −6.9 | −7.3 | 0.9957 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ardean, C.; Ciopec, M.; Davidescu, C.M.; Negrea, P.; Voda, R. Kinetics and Thermodynamics Studies for Cadmium (II) Adsorption onto Functionalized Chitosan with Hexa-Decyl-Trimethyl-Ammonium Chloride. Materials 2020, 13, 5552. https://doi.org/10.3390/ma13235552
Ardean C, Ciopec M, Davidescu CM, Negrea P, Voda R. Kinetics and Thermodynamics Studies for Cadmium (II) Adsorption onto Functionalized Chitosan with Hexa-Decyl-Trimethyl-Ammonium Chloride. Materials. 2020; 13(23):5552. https://doi.org/10.3390/ma13235552
Chicago/Turabian StyleArdean, Cristina, Mihaela Ciopec, Corneliu Mircea Davidescu, Petru Negrea, and Raluca Voda. 2020. "Kinetics and Thermodynamics Studies for Cadmium (II) Adsorption onto Functionalized Chitosan with Hexa-Decyl-Trimethyl-Ammonium Chloride" Materials 13, no. 23: 5552. https://doi.org/10.3390/ma13235552
APA StyleArdean, C., Ciopec, M., Davidescu, C. M., Negrea, P., & Voda, R. (2020). Kinetics and Thermodynamics Studies for Cadmium (II) Adsorption onto Functionalized Chitosan with Hexa-Decyl-Trimethyl-Ammonium Chloride. Materials, 13(23), 5552. https://doi.org/10.3390/ma13235552







