A Low-Cost Open Source Device for Cell Microencapsulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Line
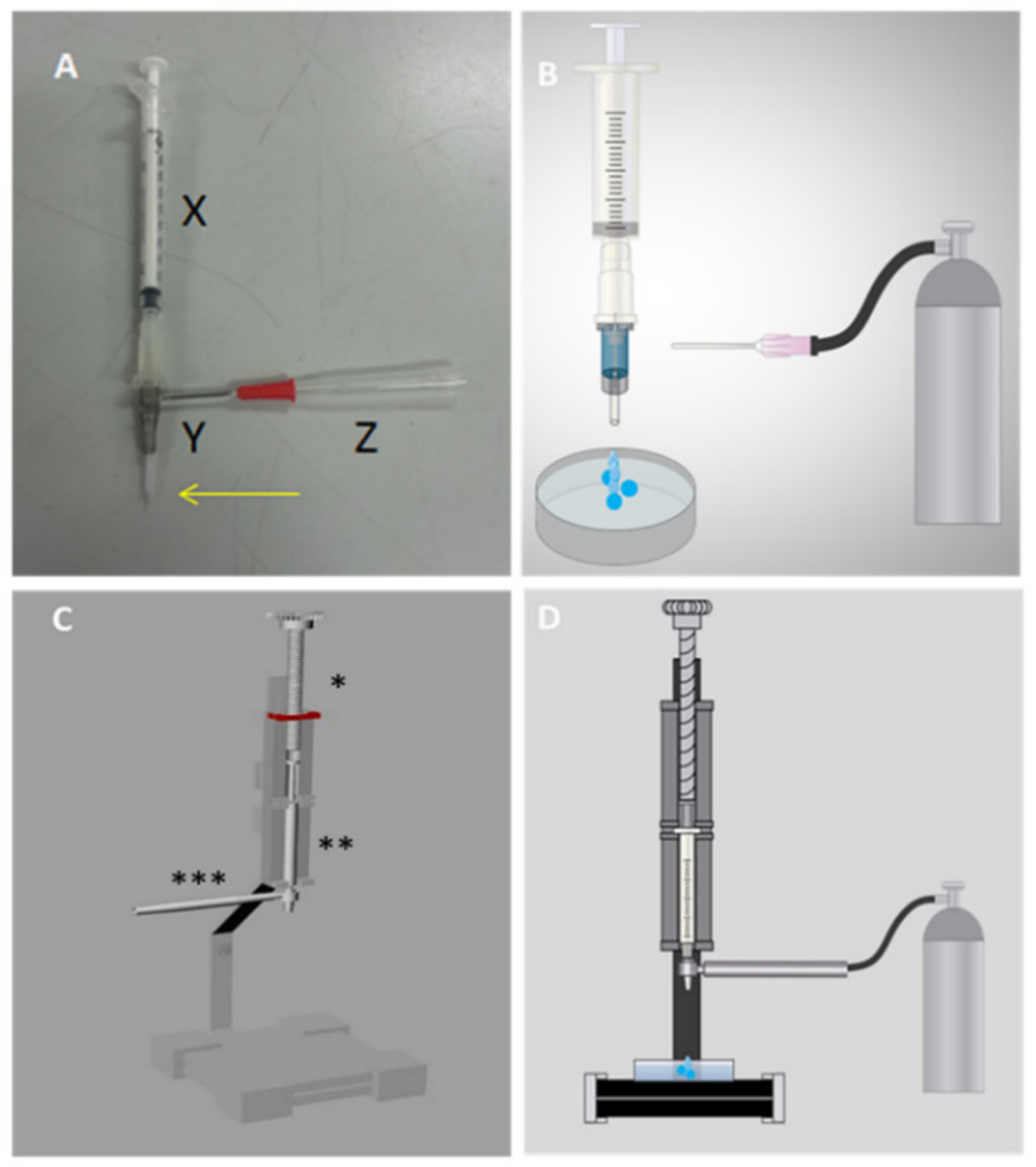
2.2. Device
2.3. Cell Encapsulation
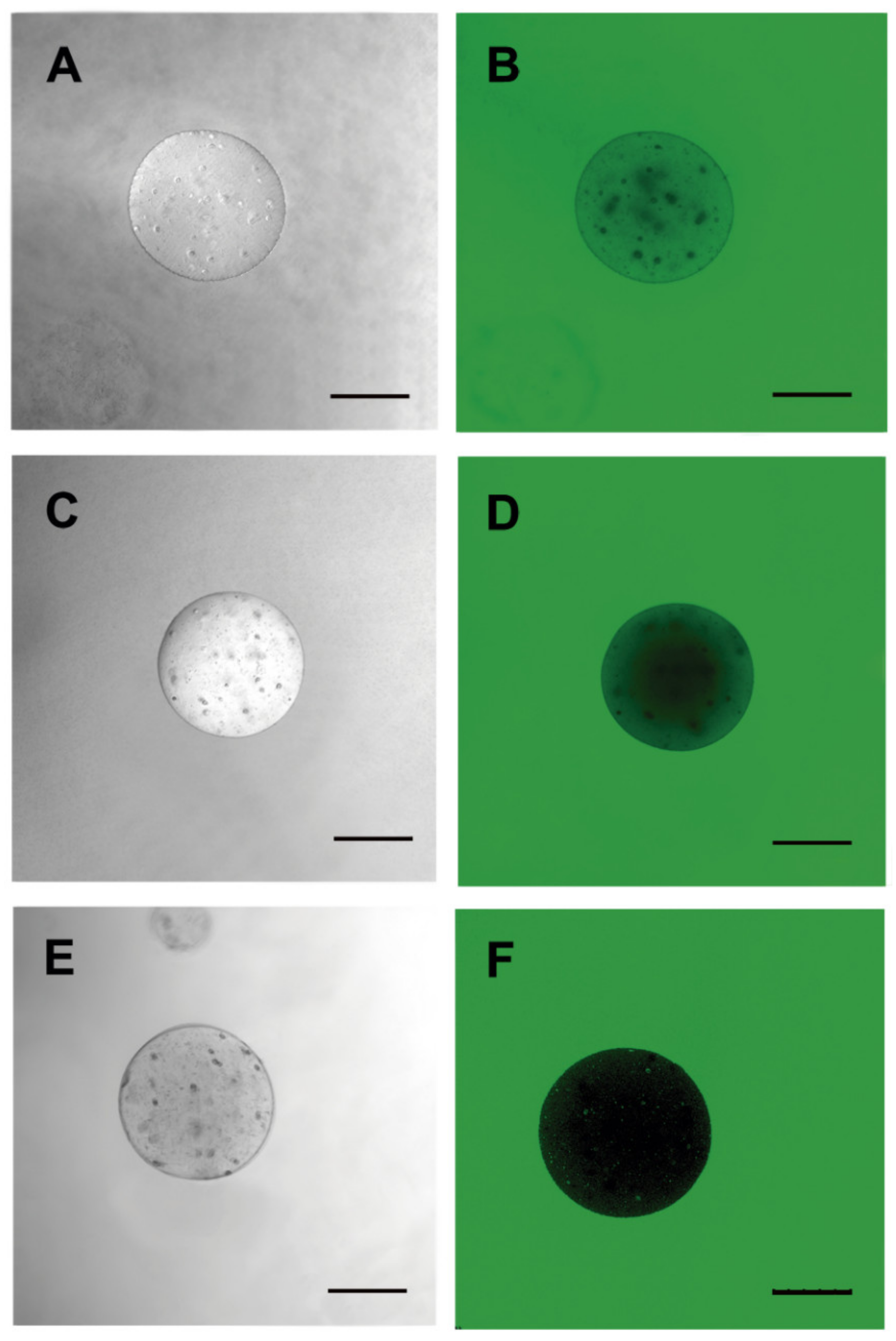
2.4. Sodium Alginate “Membrane” Porosity
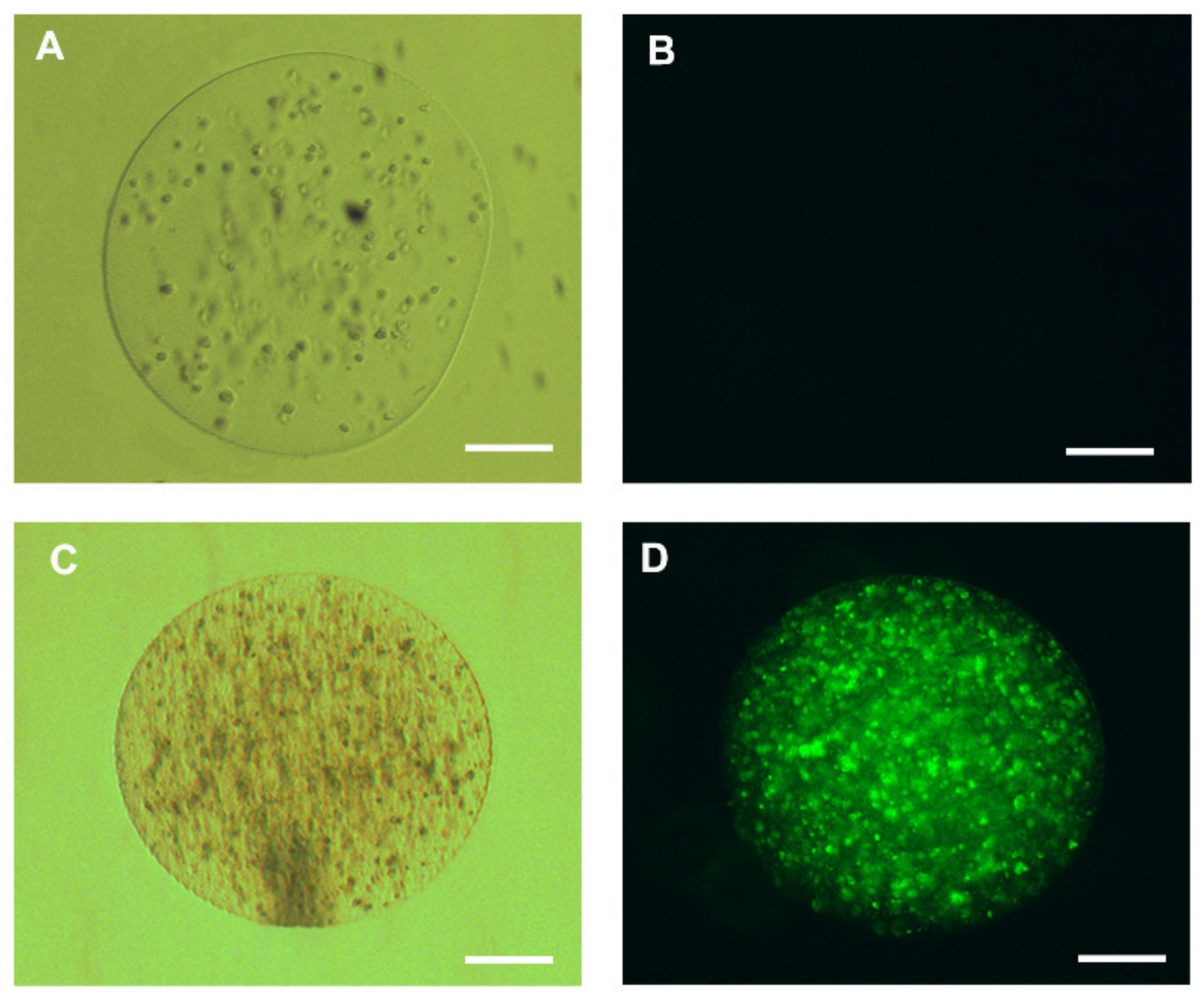
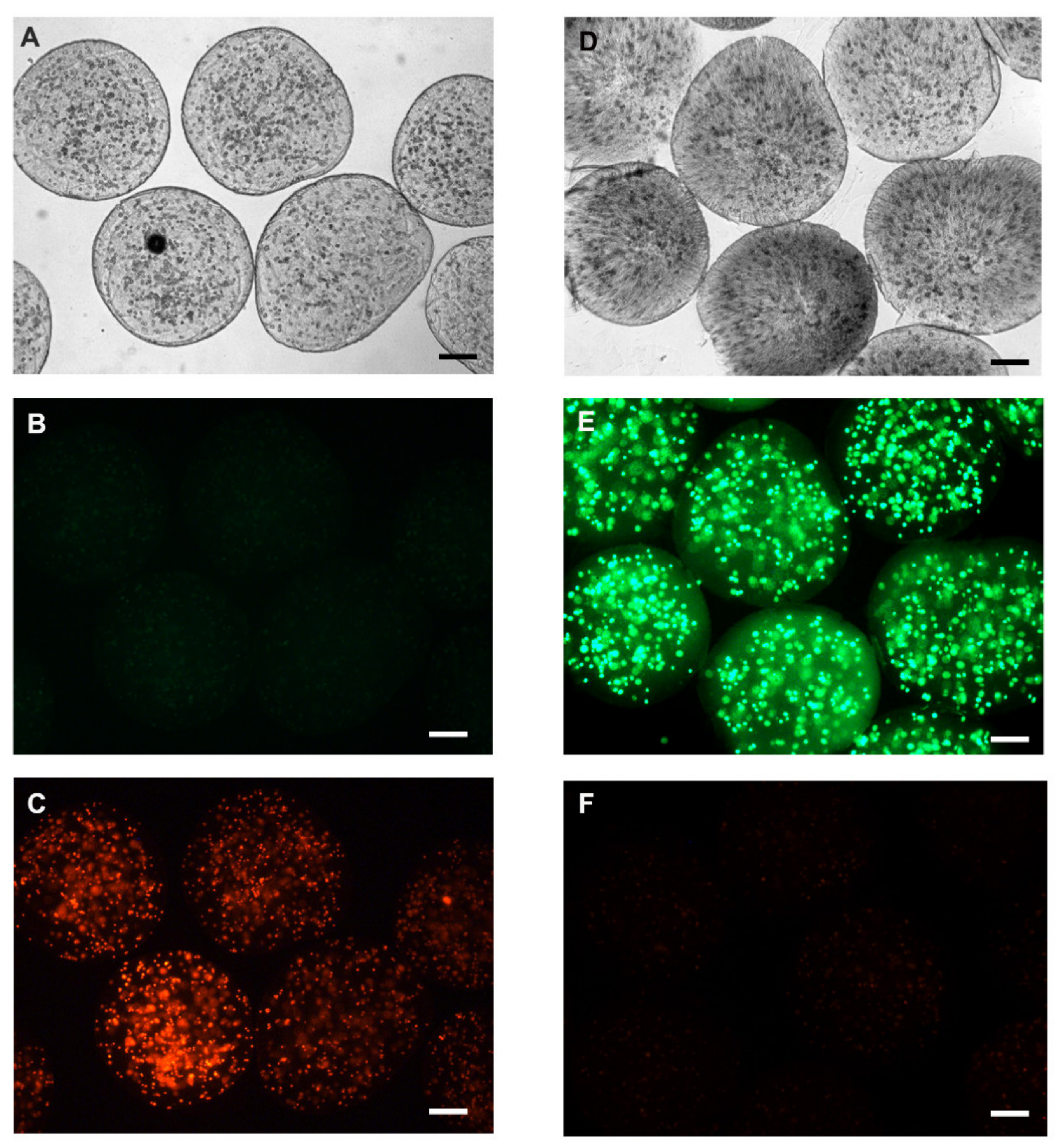
2.5. Viability Assessment of Encapsulated Human Hepatocellular Carcinoma Cells (HepG2)
2.6. Statistical Analysis
3. Results
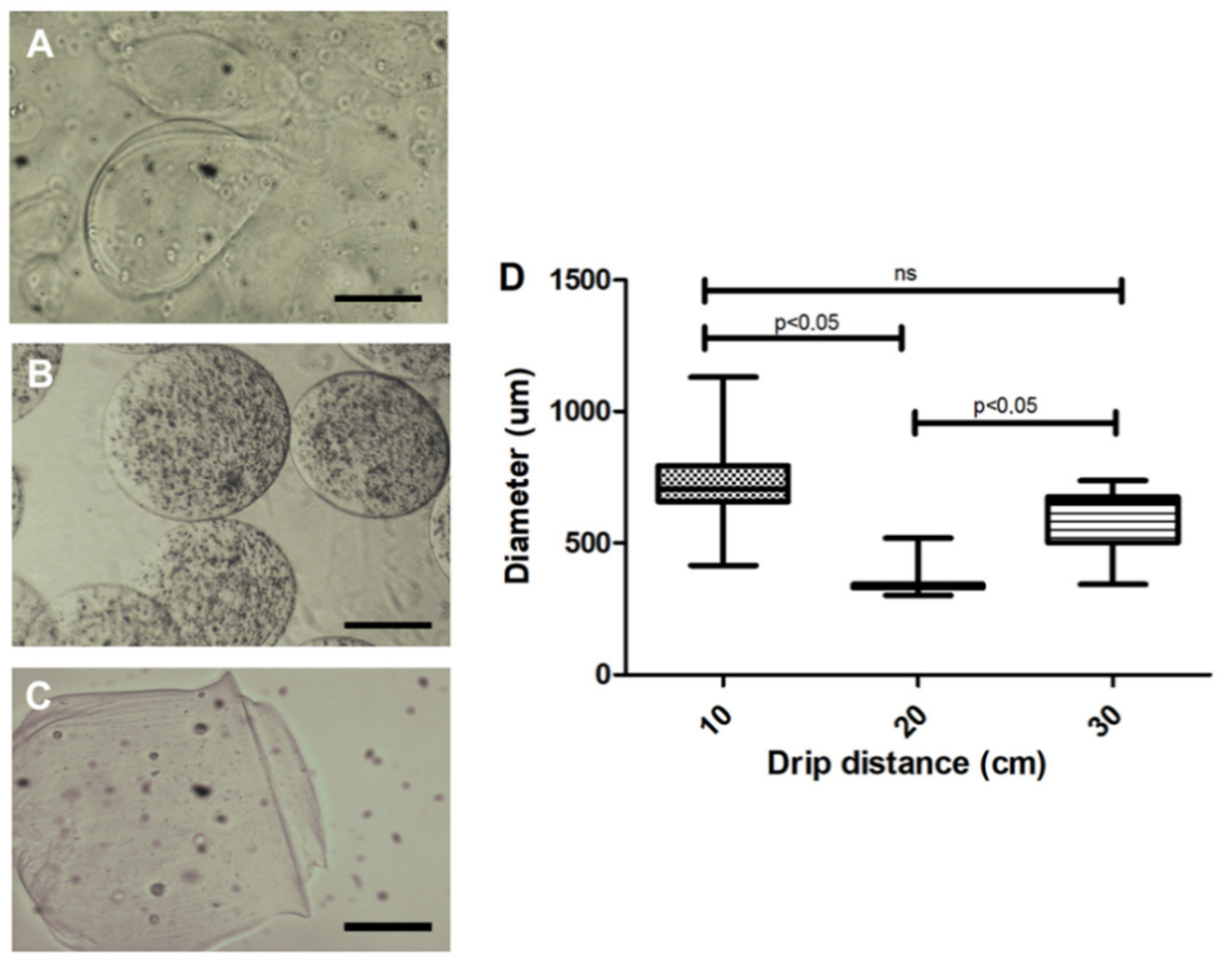
3.1. Cell Encapsulation Prototype Development
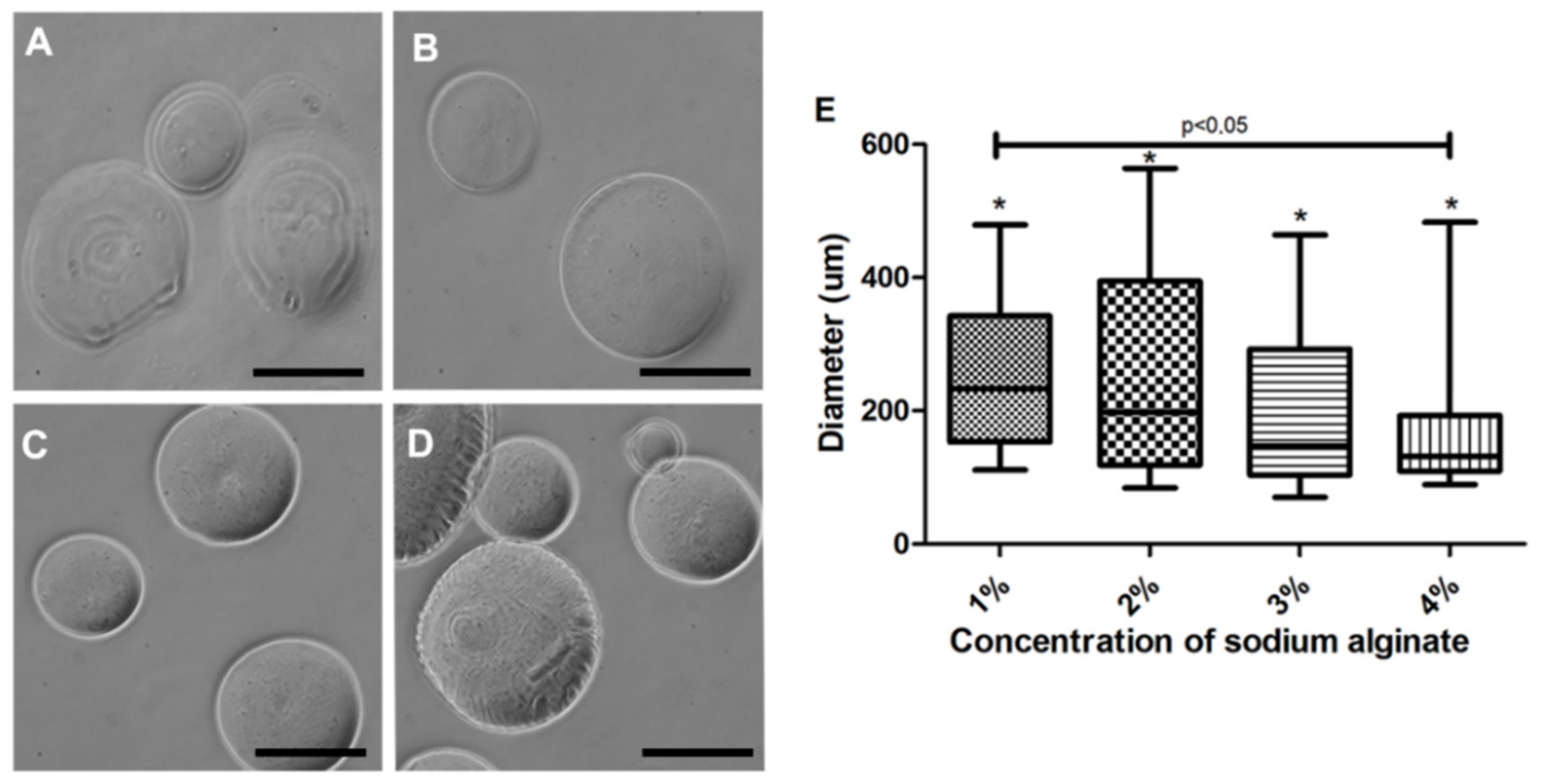
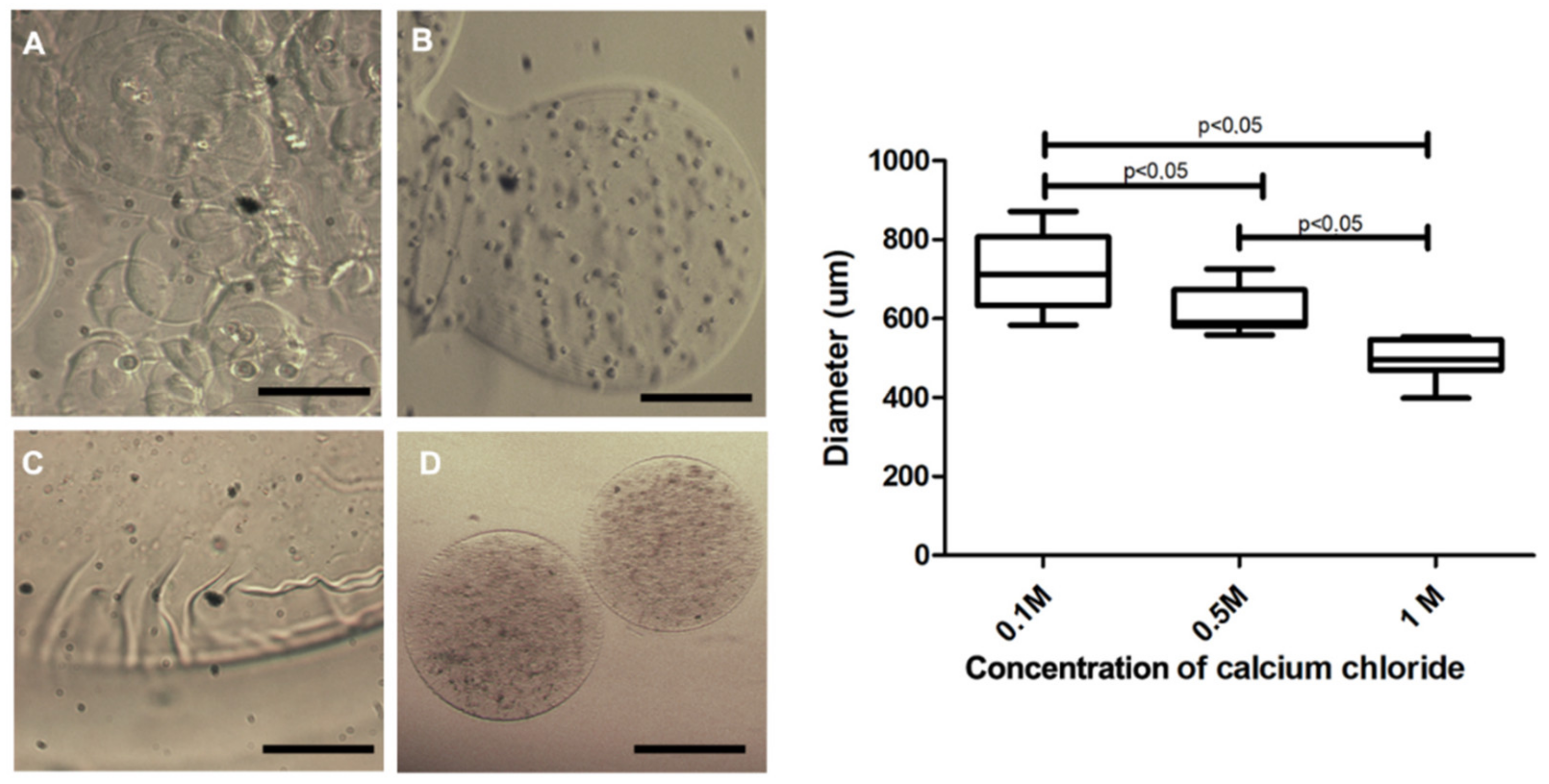
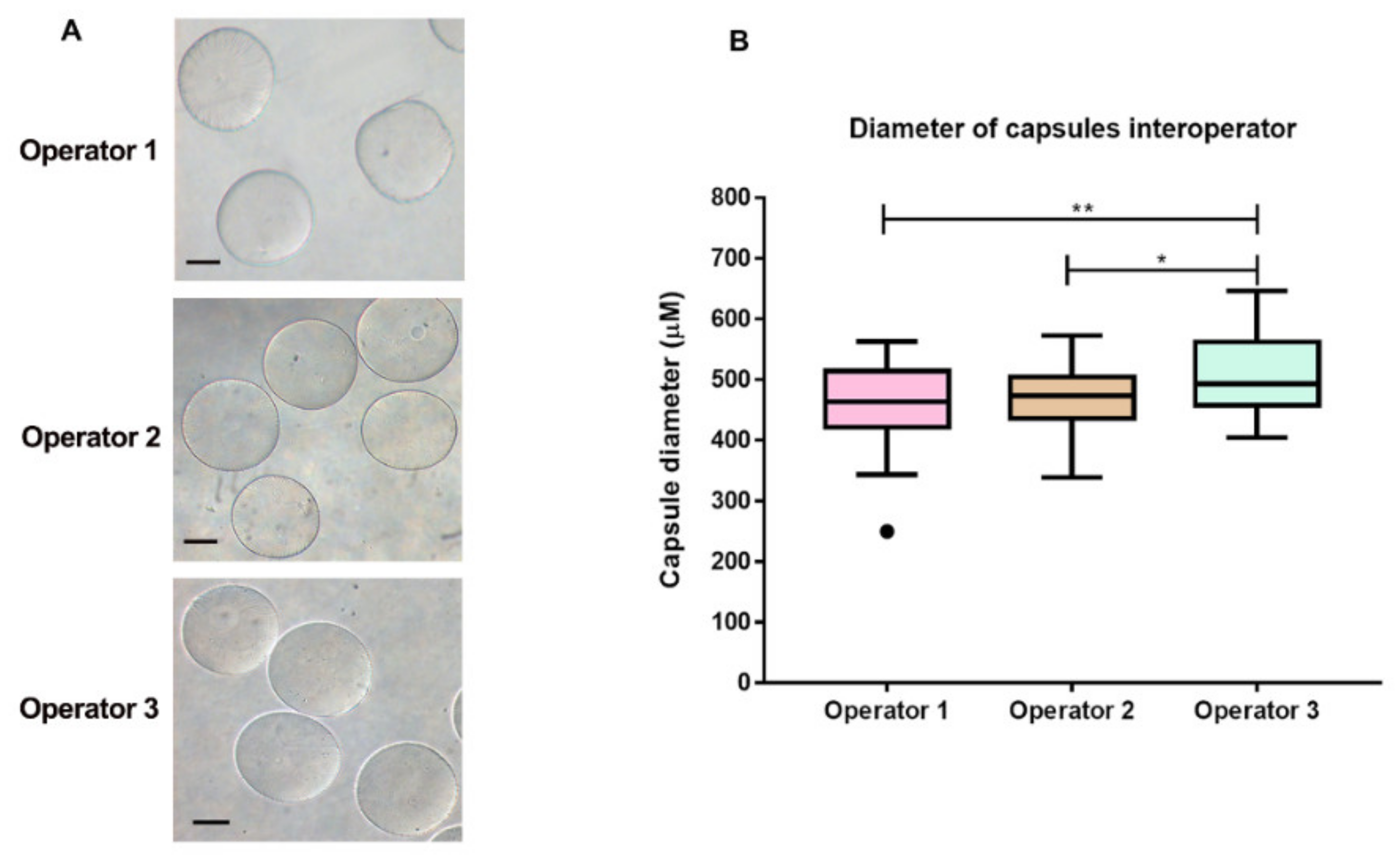
3.2. Microcapsule Characteristics
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Winter, L.; Pellicer-Guridi, R.; Broche, L.; Winkler, S.A.; Reimann, H.M.; Han, H.; Arndt, F.; Hodge, R.; Günyar, S.; Moritz, M.; et al. Open Source Medical Devices for Innovation, Education and Global Health: Case Study of Open Source Magnetic Resonance Imaging. In Co-Creation; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Sinha, S.R.; Barry, M. Health technologies and innovation in the global health arena. N. Engl. J. Med. 2011, 365, 779–782. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Local Production and Technology Transfer to Increase Access to Medical Devices: Addressing the Barriers and Challenges in Low- and Middle-Income Countries. Available online: http://www.who.int/medical_devices/1240EHT_final.pdf (accessed on 24 November 2019).
- Chang, T.M. Semipermeable Microcapsules. Science 1964, 146, 524–525. [Google Scholar] [CrossRef]
- Lim, F.; Sun, A.M. Microencapsulated islets as bioartificial endocrine pancreas. Science 1980, 210, 908–910. [Google Scholar] [CrossRef] [PubMed]
- Olabisi, R.M. Cell microencapsulation with synthetic polymers. J. Biomed. Mater. Res. A 2015, 103, 846–859. [Google Scholar] [CrossRef]
- Aijaz, A.; Perera, D.; Olabisi, R.M. Polymeric Materials for Cell Microencapsulation. Methods Mol. Biol. 2017, 1479, 79–93. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.; Jones, M.L. Artificial Cell Therapy: New Strategies for the Therapeutic Delivery of Live Bacteria. J. Biomed. Biotechnol. 2005, 2005, 44–56. [Google Scholar] [CrossRef]
- Strand, B.L.; Gaserod, O.; Kulseng, B.; Espevik, T.; Skjak-Baek, G. Alginate-polylysine-alginate microcapsules: Effect of size reduction on capsule properties. J. Microencapsul. 2002, 19, 615–630. [Google Scholar] [CrossRef]
- Robitaille, R.; Pariseau, J.F.; Leblond, F.A.; Lamoureux, M.; Lepage, Y.; Halle, J.P. Studies on small (<350 µm) alginate-poly-L-lysine microcapsules. III. Biocompatibility Of smaller versus standard microcapsules. J. Biomed. Mater. Res. 1999, 44, 116–120. [Google Scholar] [CrossRef]
- Risbud, M.V.; Shapiro, I.M.; Vaccaro, A.R.; Albert, T.J. Stem cell regeneration of the nucleus pulposus. Spine J. Off. J. N. Am. Spine Soc. 2004, 4, 348S–353S. [Google Scholar] [CrossRef]
- Yang, Y.; Opara, E.C.; Liu, Y.; Atala, A.; Zhao, W. Microencapsulation of porcine thyroid cell organoids within a polymer microcapsule construct. Exp. Biol. Med. 2017, 242, 286–296. [Google Scholar] [CrossRef]
- Orive, G.; Santos, E.; Pedraz, J.L.; Hernandez, R.M. Application of cell encapsulation for controlled delivery of biological therapeutics. Adv. Drug Deliv. Rev. 2014, 67–68, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Farina, M.; Alexander, J.F.; Thekkedath, U.; Ferrari, M.; Grattoni, A. Cell encapsulation: Overcoming barriers in cell transplantation in diabetes and beyond. Adv. Drug Deliv. Rev. 2019, 139, 92–115. [Google Scholar] [CrossRef]
- Zhang, H.; Zhu, S.J.; Wang, W.; Wei, Y.J.; Hu, S.S. Transplantation of microencapsulated genetically modified xenogeneic cells augments angiogenesis and improves heart function. Gene Ther. 2008, 15, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ouyang, H.; Dass, C.R.; Xu, J. Current research on pharmacologic and regenerative therapies for osteoarthritis. Bone Res. 2016, 4, 15040. [Google Scholar] [CrossRef] [PubMed]
- Fastag, E.; Varon, J.; Sternbach, G. Richard Lower: The origins of blood transfusion. J. Emerg. Med. 2013, 44, 1146–1150. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, A.M.; Pokrywczynska, M.; Ricordi, C. Clinical pancreatic islet transplantation. Nat. Rev. Endocrinol. 2017, 13, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Bryant, S.J.; Anseth, K.S. Hydrogel properties influence ECM production by chondrocytes photoencapsulated in poly(ethylene glycol) hydrogels. J. Biomed. Mater. Res. 2002, 59, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Tibbitt, M.W.; Anseth, K.S. Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnol. Bioeng. 2009, 103, 655–663. [Google Scholar] [CrossRef]
- Luca, G.; Cameron, D.F.; Arato, I.; Mancuso, F.; Linden, E.H.; Calvitti, M.; Falabella, G.; Szekeres, K.; Bodo, M.; Ricci, G.; et al. Xenograft of microencapsulated Sertoli cells for the cell therapy of type 2 diabetes mellitus in spontaneously diabetic nonhuman primates: Preliminary data. Transpl. Proc. 2014, 46, 1999–2001. [Google Scholar] [CrossRef]
- Calafiore, R. Microencapsulation for cell therapy of type 1 diabetes mellitus: The interplay between common beliefs, prejudices and real progress. J. Diabetes Investig. 2018, 9, 231–233. [Google Scholar] [CrossRef]
- Omami, M.; McGarrigle, J.J.; Reedy, M.; Isa, D.; Ghani, S.; Marchese, E.; Bochenek, M.A.; Longi, M.; Xing, Y.; Joshi, I.; et al. Islet Microencapsulation: Strategies and Clinical Status in Diabetes. Curr. Diabetes Rep. 2017, 17, 47. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- De Vos, P.; De Haan, B.; Van Schilfgaarde, R. Effect of the alginate composition on the biocompatibility of alginate-polylysine microcapsules. Biomaterials 1997, 18, 273–278. [Google Scholar] [CrossRef]
- Orive, G.; Ponce, S.; Hernandez, R.M.; Gascon, A.R.; Igartua, M.; Pedraz, J.L. Biocompatibility of microcapsules for cell immobilization elaborated with different type of alginates. Biomaterials 2002, 23, 3825–3831. [Google Scholar] [CrossRef]
- Santos, E.; Zarate, J.; Orive, G.; Hernandez, R.M.; Pedraz, J.L. Biomaterials in cell microencapsulation. Adv. Exp. Med. Biol. 2010, 670, 5–21. [Google Scholar] [PubMed]
- Amsden, B.; Turner, N. Diffusion characteristics of calcium alginate gels. Biotechnol. Bioeng. 1999, 65, 605–610. [Google Scholar] [CrossRef]
- Kong, H.J.; Smith, M.K.; Mooney, D.J. Designing alginate hydrogels to maintain viability of immobilized cells. Biomaterials 2003, 24, 4023–4029. [Google Scholar] [CrossRef]
- Tam, S.K.; Bilodeau, S.; Dusseault, J.; Langlois, G.; Halle, J.P.; Yahia, L.H. Biocompatibility and physicochemical characteristics of alginate-polycation microcapsules. Acta Biomater. 2011, 7, 1683–1692. [Google Scholar] [CrossRef]
- De Vos, P.; Bucko, M.; Gemeiner, P.; Navratil, M.; Svitel, J.; Faas, M.; Strand, B.L.; Skjak-Braek, G.; Morch, Y.A.; Vikartovska, A.; et al. Multiscale requirements for bioencapsulation in medicine and biotechnology. Biomaterials 2009, 30, 2559–2570. [Google Scholar] [CrossRef]
- Vaithilingam, V.; Quayum, N.; Joglekar, M.V.; Jensen, J.; Hardikar, A.A.; Oberholzer, J.; Guillemin, G.J.; Tuch, B.E. Effect of alginate encapsulation on the cellular transcriptome of human islets. Biomaterials 2011, 32, 8416–8425. [Google Scholar] [CrossRef]
- Li, X.; Liu, T.; Song, K.; Yao, L.; Ge, D.; Bao, C.; Ma, X.; Cui, Z. Culture of neural stem cells in calcium alginate beads. Biotechnol. Prog. 2006, 22, 1683–1689. [Google Scholar] [CrossRef] [PubMed]
- Orive, G.; Tam, S.K.; Pedraz, J.L.; Halle, J.P. Biocompatibility of alginate-poly-L-lysine microcapsules for cell therapy. Biomaterials 2006, 27, 3691–3700. [Google Scholar] [CrossRef] [PubMed]
- De Vos, P.; Faas, M.M.; Strand, B.; Calafiore, R. Alginate-based microcapsules for immunoisolation of pancreatic islets. Biomaterials 2006, 27, 5603–5617. [Google Scholar] [CrossRef] [PubMed]
- Dusseault, J.; Tam, S.K.; Menard, M.; Polizu, S.; Jourdan, G.; Yahia, L.; Halle, J.P. Evaluation of alginate purification methods: Effect on polyphenol, endotoxin, and protein contamination. J. Biomed. Mater. Res. A 2006, 76, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Murua, A.; Portero, A.; Orive, G.; Hernandez, R.M.; de Castro, M.; Pedraz, J.L. Cell microencapsulation technology: Towards clinical application. J. Control. Release 2008, 132, 76–83. [Google Scholar] [CrossRef]
- Barkai, U.; Rotem, A.; de Vos, P. Survival of encapsulated islets: More than a membrane story. World J. Transpl. 2016, 6, 69–90. [Google Scholar] [CrossRef]
- Sakata, N.; Sumi, S.; Yoshimatsu, G.; Goto, M.; Egawa, S.; Unno, M. Encapsulated islets transplantation: Past, present and future. World J. Gastrointest. Pathophysiol. 2012, 3, 19–26. [Google Scholar] [CrossRef]
- Aebischer, P.; Buchser, E.; Joseph, J.M.; Favre, J.; de Tribolet, N.; Lysaght, M.; Rudnick, S.; Goddard, M. Transplantation in humans of encapsulated xenogeneic cells without immunosuppression. A preliminary report. Transplantation 1994, 58, 1275–1277. [Google Scholar] [CrossRef]
- Bes, J.C.; Lazorthes, Y.; Sol, J.C.; Tkaczuk, J.; Sallerin, B. Clinical perspectives of xenografts: Encapsulated chromaffin cells and pain. Pathol. Biol. 2000, 48, 365–367. [Google Scholar]
- Etter, J.N.; Karasinski, M.; Ware, J.; Oldinski, R.A. Dual-crosslinked homogeneous alginate microspheres for mesenchymal stem cell encapsulation. J. Mater. Sci. Mater. Med. 2018, 29, 143. [Google Scholar] [CrossRef]
- Farias, C.; Lyman, R.; Hemingway, C.; Chau, H.; Mahacek, A.; Bouzos, E.; Mobed-Miremadi, M. Three-Dimensional (3D) Printed Microneedles for Microencapsulated Cell Extrusion. Bioengineering 2018, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Mobed-Miremadi, M.; Asi, B.; Parasseril, J.; Wong, E.; Tat, M.; Shan, Y. Comparative diffusivity measurements for alginate-based atomized and inkjet-bioprinted artificial cells using fluorescence microscopy. Artif. Cells Nanomed. Biotechnol. 2013, 41, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, S.; Oda, T.; Aoyagi, Y.; Matsuo, R.; Enomoto, T.; Matsumoto, K.; Nakamura, T.; Satake, M.; Ochiai, A.; Ohkohchi, N.; et al. Microfabricated airflow nozzle for microencapsulation of living cells into 150 micrometer microcapsules. Biomed. Microdevices 2007, 9, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Bressel, T.A.; Paz, A.H.; Baldo, G.; Lima, E.O.C.; Matte, U.; Saraiva-Pereira, M.L. An effective device for generating alginate microcapsules. Genet. Mol. Biol. 2008, 31, 136–140. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, M.S.; Cardoso, L.M.d.F.; da Silva, T.B.; Teixeira, A.J.; Mizrahi, S.E.; Ferreira, G.S.M.; Dantas, F.M.L.; Cotta-de-Almeida, V.; Alves, L.A. A Low-Cost Open Source Device for Cell Microencapsulation. Materials 2020, 13, 5090. https://doi.org/10.3390/ma13225090
Pereira MS, Cardoso LMdF, da Silva TB, Teixeira AJ, Mizrahi SE, Ferreira GSM, Dantas FML, Cotta-de-Almeida V, Alves LA. A Low-Cost Open Source Device for Cell Microencapsulation. Materials. 2020; 13(22):5090. https://doi.org/10.3390/ma13225090
Chicago/Turabian StylePereira, Miriam Salles, Liana Monteiro da Fonseca Cardoso, Tatiane Barreto da Silva, Ayla Josma Teixeira, Saul Eliahú Mizrahi, Gabriel Schonwandt Mendes Ferreira, Fabio Moyses Lins Dantas, Vinicius Cotta-de-Almeida, and Luiz Anastacio Alves. 2020. "A Low-Cost Open Source Device for Cell Microencapsulation" Materials 13, no. 22: 5090. https://doi.org/10.3390/ma13225090
APA StylePereira, M. S., Cardoso, L. M. d. F., da Silva, T. B., Teixeira, A. J., Mizrahi, S. E., Ferreira, G. S. M., Dantas, F. M. L., Cotta-de-Almeida, V., & Alves, L. A. (2020). A Low-Cost Open Source Device for Cell Microencapsulation. Materials, 13(22), 5090. https://doi.org/10.3390/ma13225090




