From 1D Coordination Polymers to Metal Organic Frameworks by the Use of 2-Pyridyl Oximes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials, Physical, and Spectroscopic Measurements
2.2. Compound Synthesis
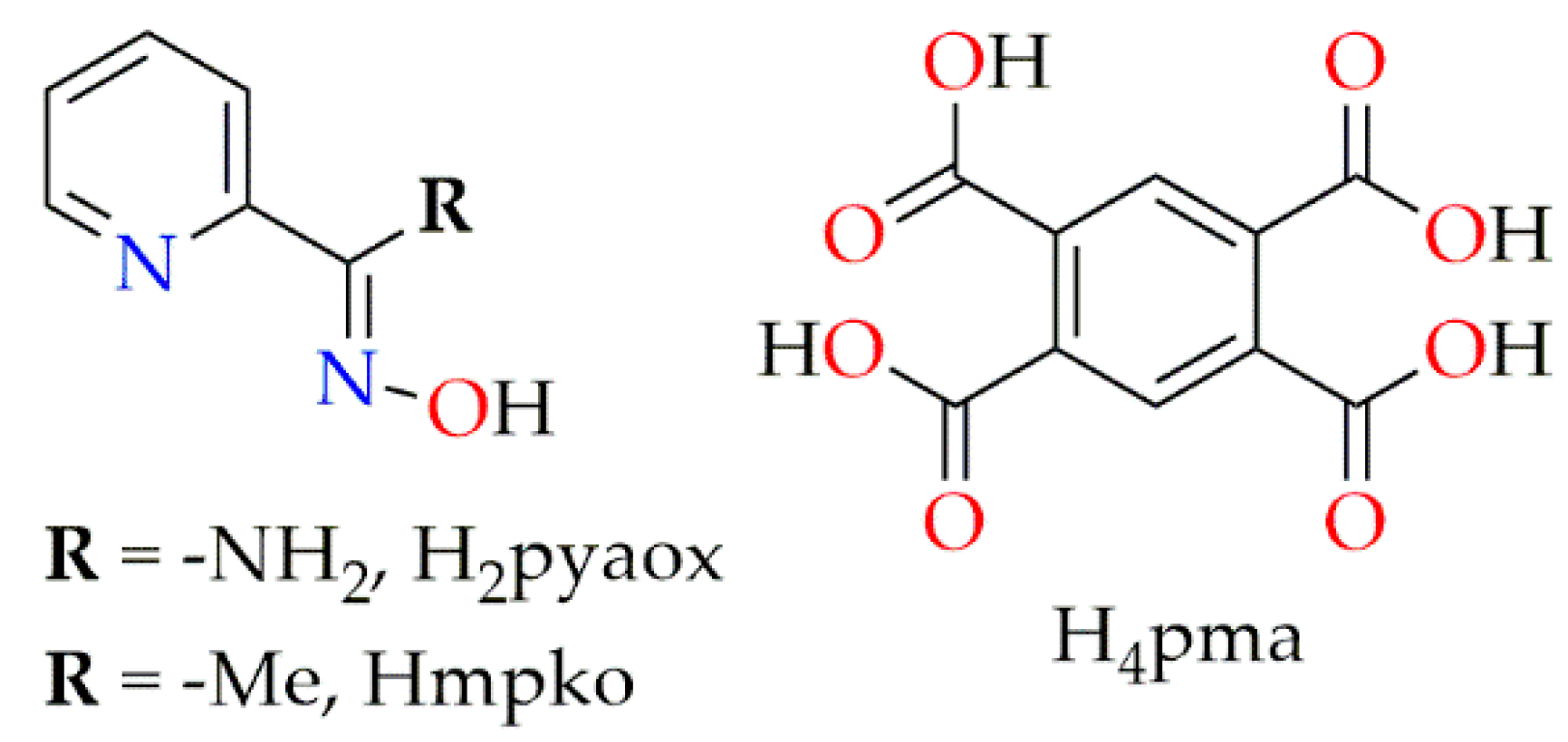
2.2.1. Synthesis of [Zn(H2pma)(H2pyaox)(H2O)2] (1)
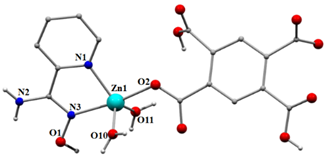
2.2.2. Synthesis of [Zn2(pma)(H2pyaox)2]n (2)
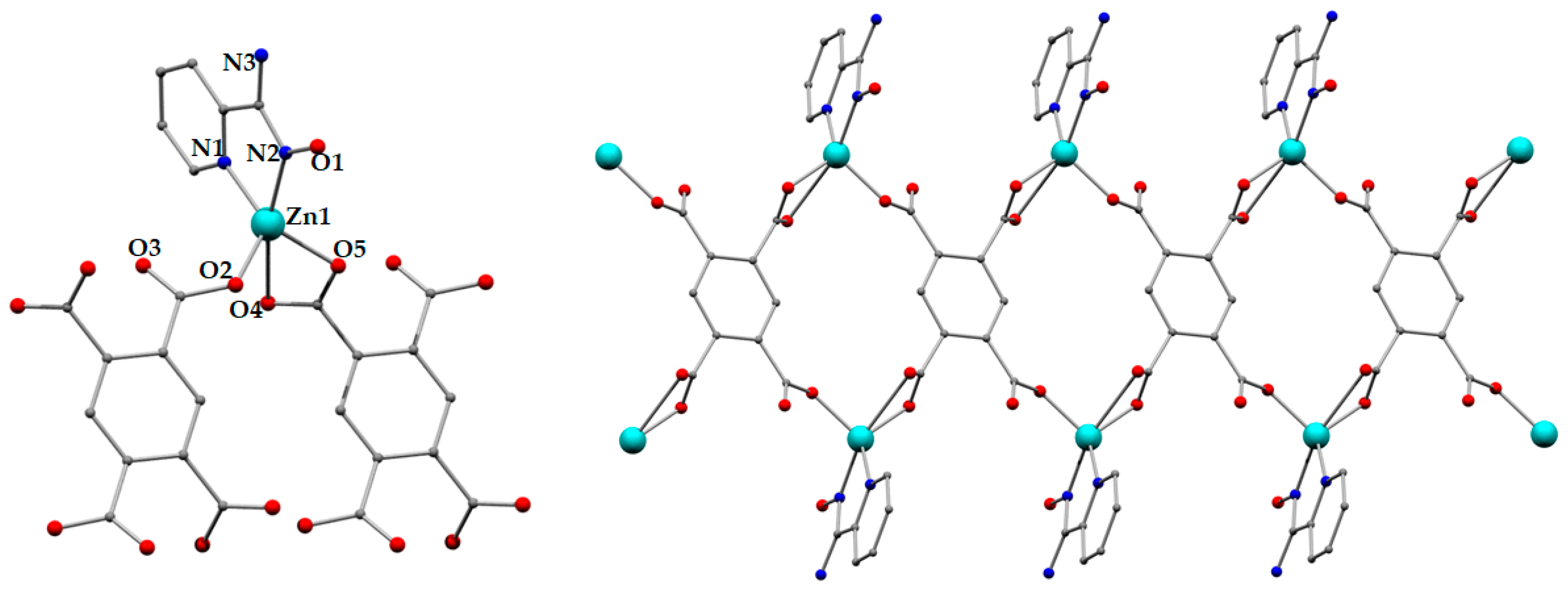
2.2.3. Synthesis of [Zn2(pma)(H2pyaox)2(H2O)2]n (3)
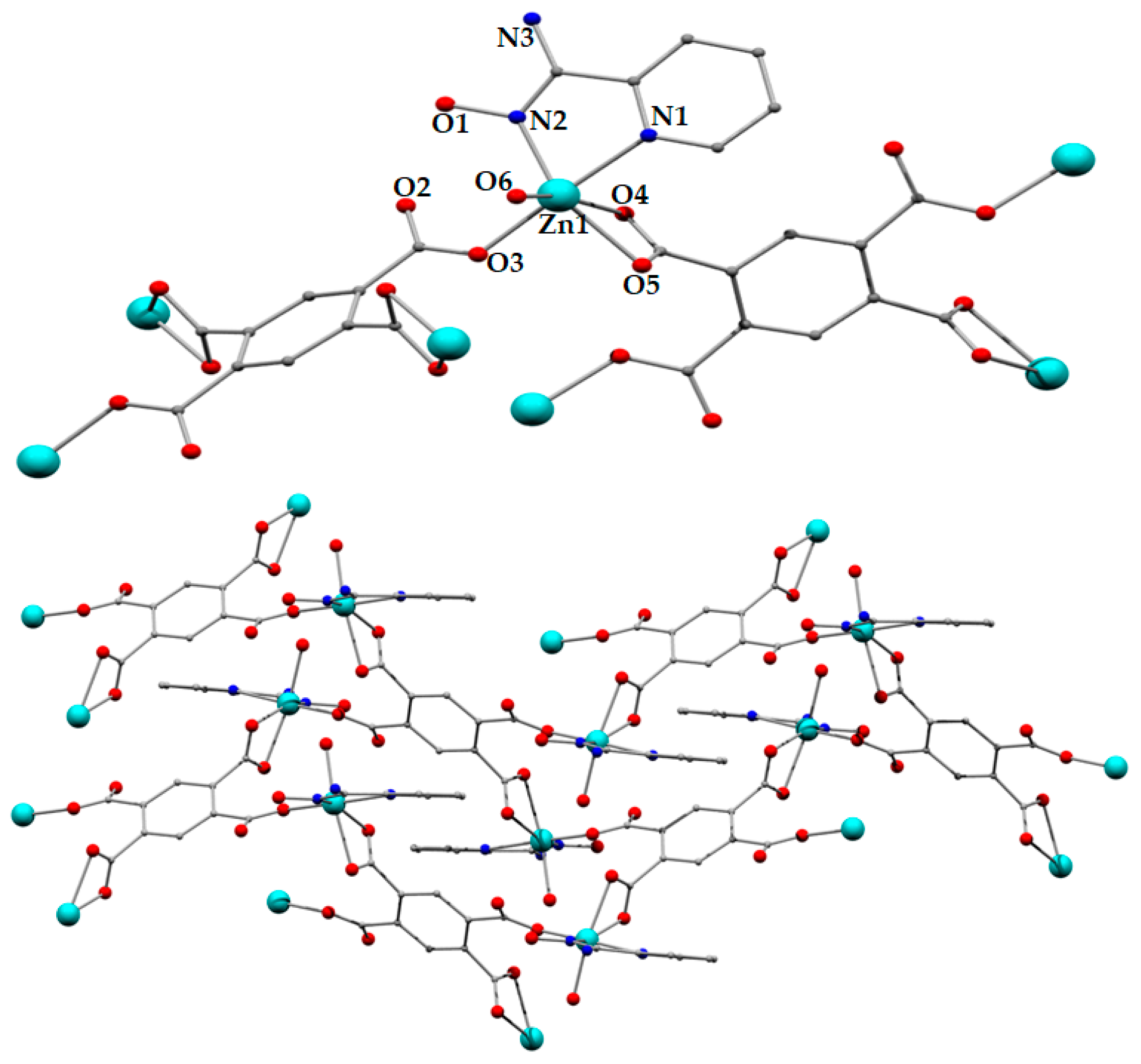
2.2.4. Synthesis of [Co2(pma)(H2pyaox)2(H2O)6] (4)
2.2.5. Synthesis of [Mn2(pma)(H2pyaox)2(H2O)6] (5)
2.2.6. Synthesis of [Cu2(pma)(H2pyaox)2(DMF)2]n (6)
2.2.7. Synthesis of [Zn2(pma)(Hmpko)2(H2O)4]·2H2O (7∙2H2O)
2.2.8. Synthesis of [Cu(pma)0.5(Hmpko)(DMF)]n (8)
2.2.9. Synthesis of [Cu4(OH)2(pma)(mpko)2]n (9)
2.3. Single-Crystal X-ray Crystallography
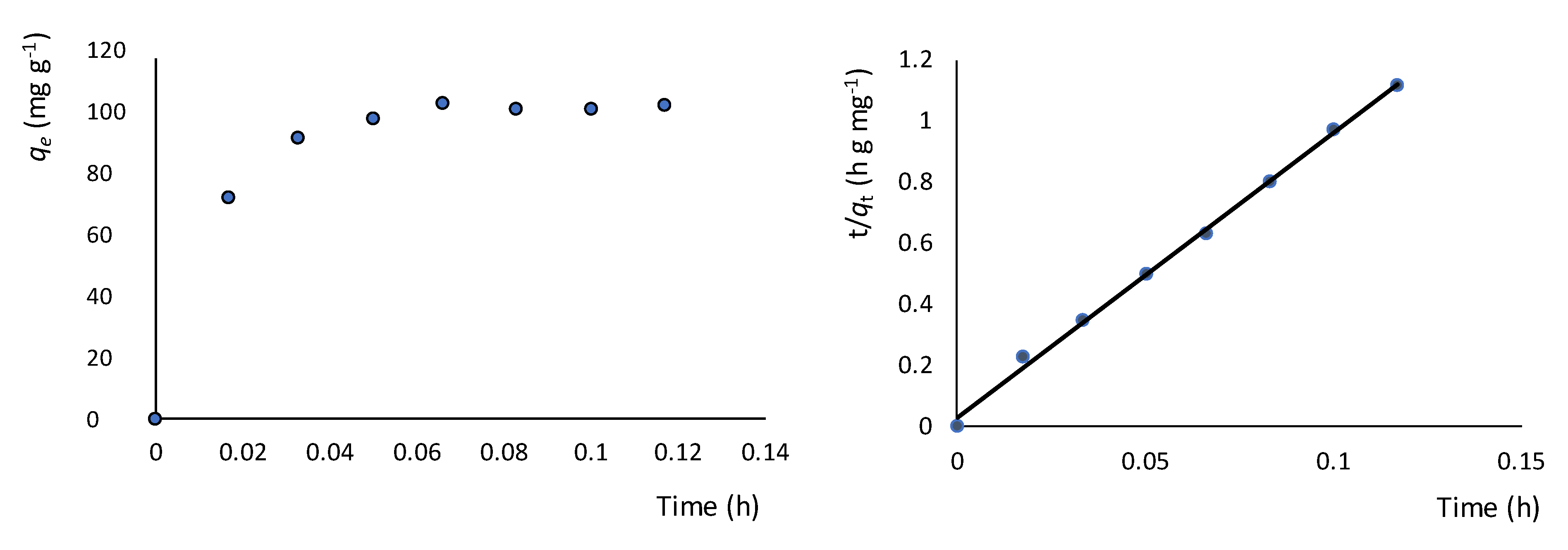
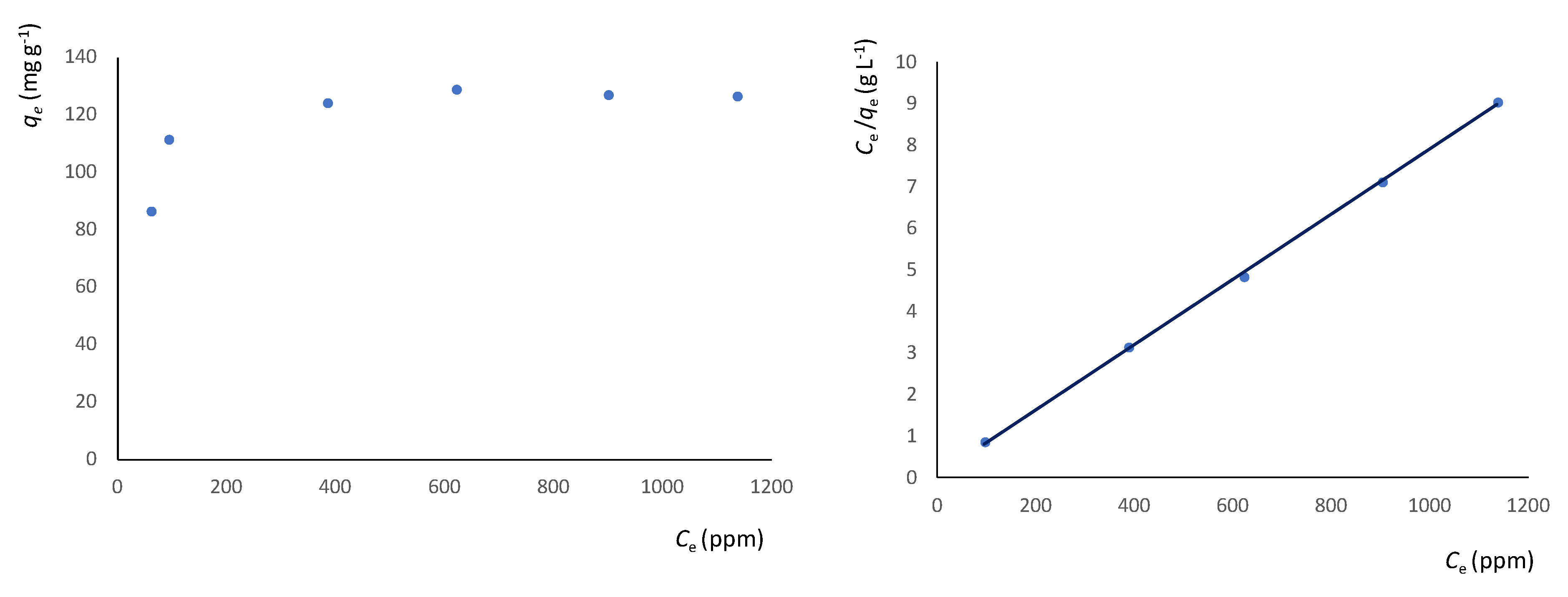
2.4. Metal Ion and 2-methyluracil Adsorption Kinetic and Thermodynamic Studies
3. Results and Discussion
3.1. Synthetic Discussion
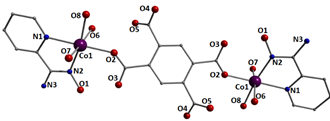
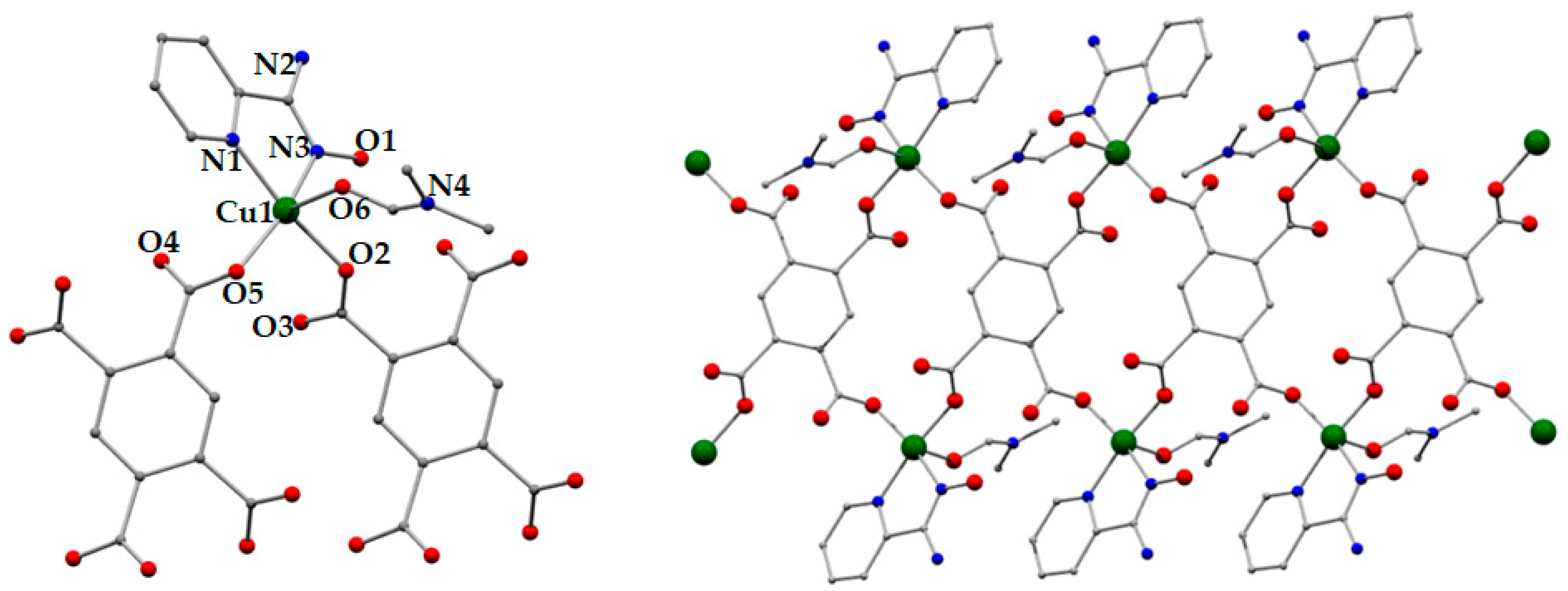
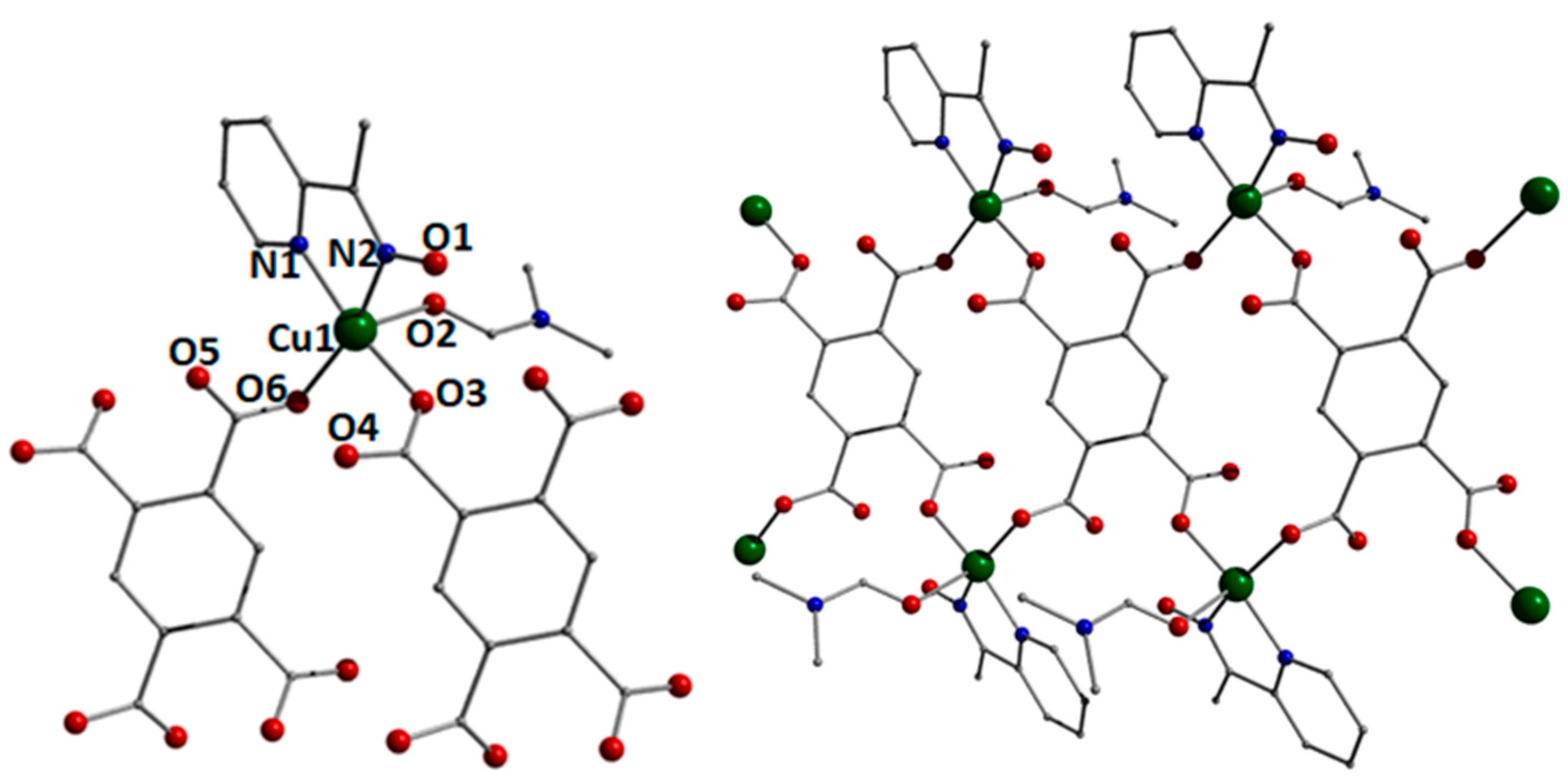
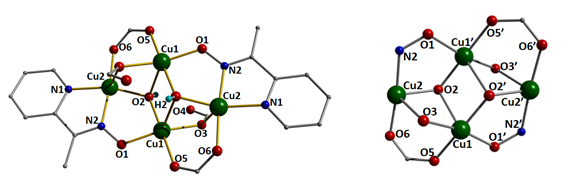
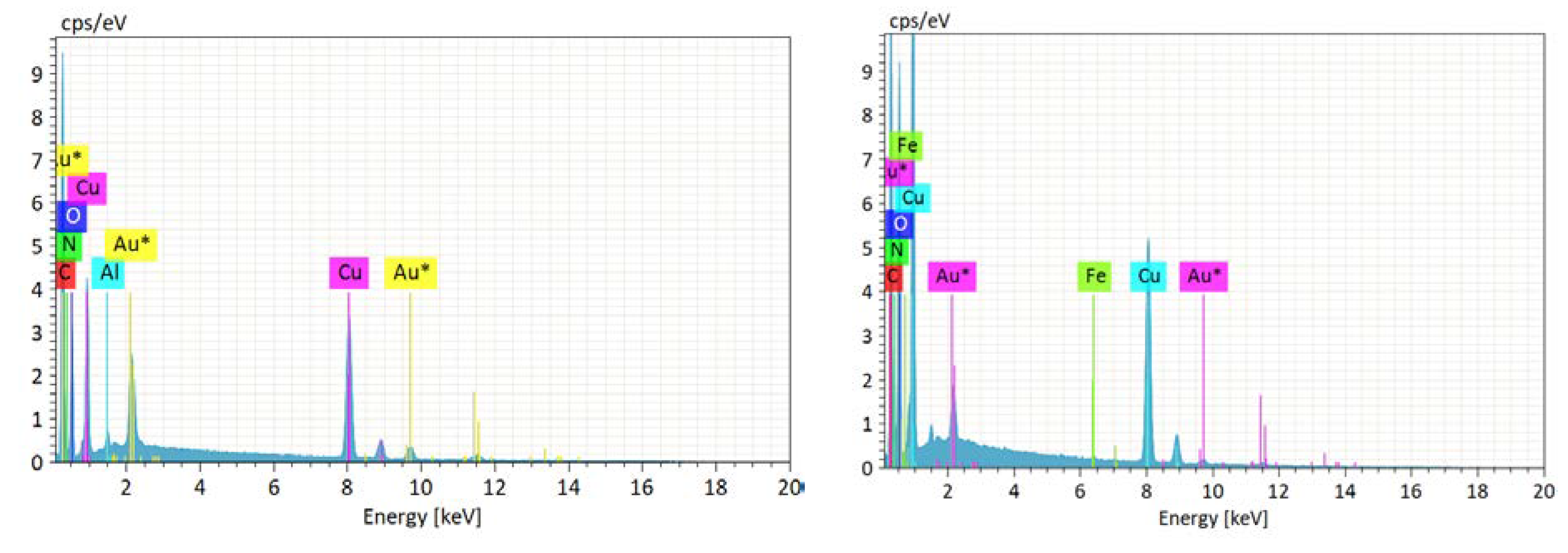
3.2. Description of Structures
3.3. Adsorption Studies
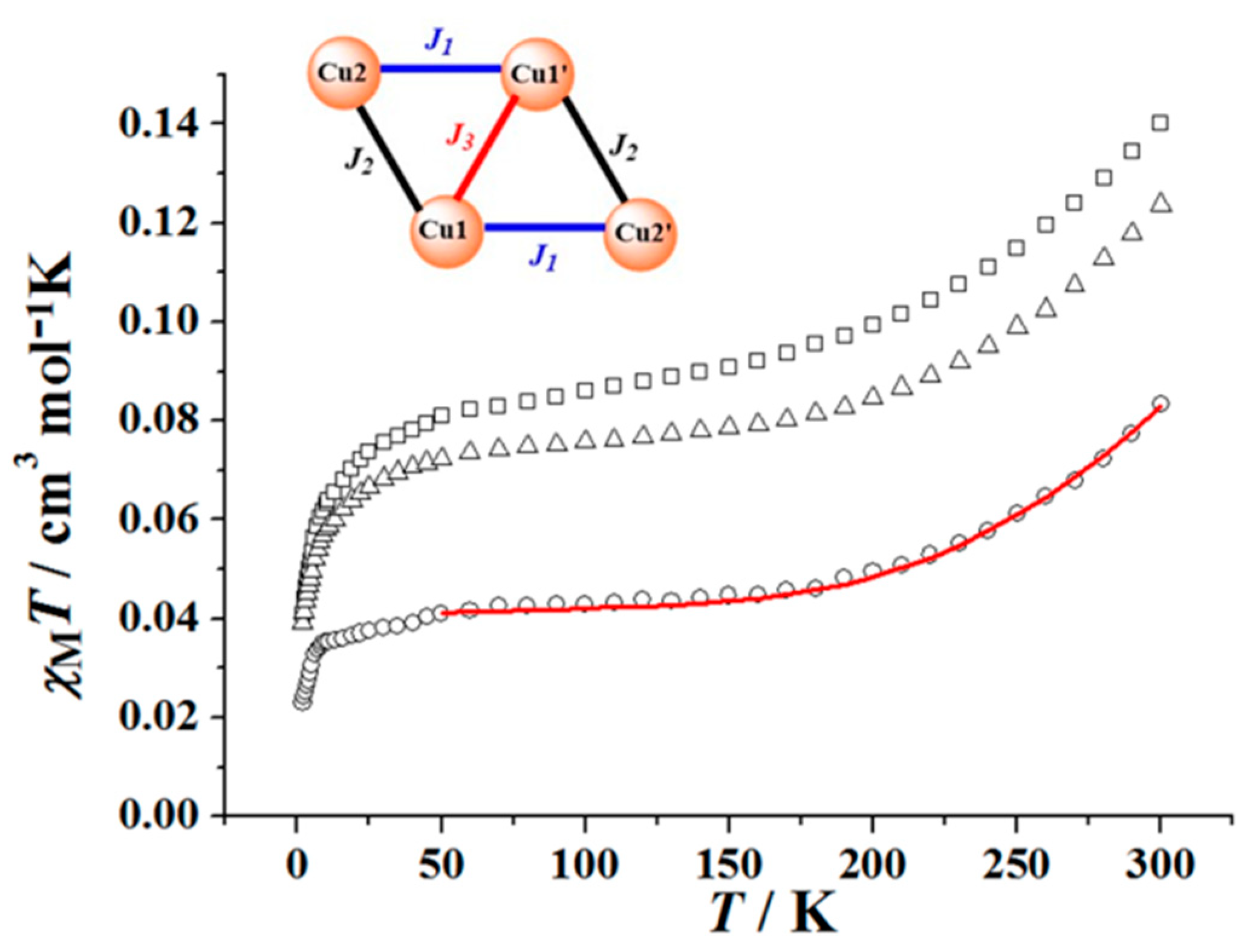
3.4. Magnetism Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Qin, J.-S.; Du, D.-Y.; Guan, W.; Bo, X.-J.; Li, Y.-F.; Guo, L.-P.; Su, Z.-M.; Wang, Y.-Y.; Lan, Y.-Q.; Zhou, H.-C. Ultrastable Polymolybdate-Based Metal–Organic Frameworks as Highly Active Electrocatalysts for Hydrogen Generation from Water. J. Am. Chem. Soc. 2015, 137, 7169–7177. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Falkowski, J.M.; Abney, C.; Lin, W. A series of isoreticular chiral metal–organic frameworks as a tunable platform for asymmetric catalysis. Nat. Chem. 2010, 2, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, A.; Matveevskaya, V.; Pavlov, D.; Yakunenkov, A.; Potapov, A. Coordination Polymers Based on Highly Emissive Ligands: Synthesis and Functional Properties. Materials 2020, 13, 2699. [Google Scholar] [CrossRef]
- Wu, M.-X.; Yang, Y.-W. Metal–Organic Framework (MOF)-Based Drug/Cargo Delivery and Cancer Therapy. Adv. Mater. 2017, 29, 1606134. [Google Scholar] [CrossRef] [PubMed]
- Huxford, R.C.; Rocca, J.D.; Lin, W. Metal-organic frameworks as potential drug carriers. Curr. Opin. Chem. Biol. 2010, 14, 262–268. [Google Scholar] [CrossRef]
- Hu, Z.; Deibert, B.J.; Li, J. Luminescmakeent metal–organic frameworks for chemical sensing and explosive detection. Chem. Soc. Rev. 2014, 43, 5815–5840. [Google Scholar] [CrossRef]
- Douvali, A.; Tsipis, A.C.; Eliseeva, S.V.; Petoud, S.; Papaefstathiou, G.S.; Malliakas, C.D.; Papadas, I.; Armatas, G.S.; Margiolaki, I.; Kanatzidis, M.G.; et al. Turn-on luminescence sensing and real-time detection of traces of water in organic solvents by a flexible metal-organic framework. Angew. Chem. Int. Ed. 2015, 54, 1651. [Google Scholar] [CrossRef]
- Coulon, C.; Miyasaka, H.; Clérac, R. Single-Chain Magnets: Theoretical Approach and Experimental Systems. Struct. Bonding 2006, 122, 163–206. [Google Scholar] [CrossRef]
- Jeon, I.-R.; Clérac, R. Controlled association of single-molecule magnets (SMMs) into coordination networks: Towards a new generation of magnetic materials. Dalton Trans. 2012, 41, 9569–9586. [Google Scholar] [CrossRef]
- Miyasaka, H.; Julve, M.; Yamashita, M.; Clérac, R. Slow Dynamics of the Magnetization in One-Dimensional Coordination Polymers: Single-Chain Magnets. Inorg. Chem. 2009, 48, 3420–3437. [Google Scholar] [CrossRef]
- Bernot, K.; Luzon, J.; Sessoli, R.; Vindigni, A.; Thion, J.; Richeter, S.; Leclercq, D.; Larionova, J.; Van der Lee, A. The Canted Antiferromagnetic Approach to Single-Chain Magnets. J. Am. Chem. Soc. 2008, 130, 1619–1627. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-T.; Ren, M.; Bao, S.-S.; Liu, B.; Pi, L.; Cai, Z.-S.; Zheng, Z.-H.; Xu, Z.-L.; Zheng, L.-M. Effect of Structural Isomerism on Magnetic Dynamics: From Single-Molecule Magnet to Single-Chain Magnet. Inorg. Chem. 2014, 53, 3117–3125. [Google Scholar] [CrossRef] [PubMed]
- Gatteschi, D.; Sessoli, R. Quantum Tunneling of Magnetization and Related Phenomena in Molecular Materials. Angew. Chem. Int. Ed. 2003, 42, 268–297. [Google Scholar] [CrossRef] [PubMed]
- Papatriantafyllopoulou, C.; Zartilas, S.; Manos, M.J.; Pichon, C.; Clérac, R.; Tasiopoulos, A.J. A single-chain magnet based on linear [MnIII2MnII] units. Chem. Commun. 2014, 50, 14873–14876. [Google Scholar] [CrossRef] [PubMed]
- Bogani, L.; Wernsdorfer, W. Molecular spintronics using single-molecule magnets. Nat. Mater. 2008, 7, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Leuenberger, M.N.; Loss, D. Quantum computing in molecular magnets. Nature 2001, 410, 789–793. [Google Scholar] [CrossRef]
- Chen, L.; Ji, Q.; Wang, X.; Pan, Q.; Ccao, X.; Xu, G. Two novel metal–organic coordination polymers based on ligand 1,4-diazabicyclo[2.2.2]octane N,N′-dioxide with phase transition, and ferroelectric and dielectric properties. CrystEngComm 2017, 19, 5907–5914. [Google Scholar] [CrossRef]
- Hui, J.; Kishida, H.; Ishiba, K.; Takemasu, K.; Morikawa, M.; Kimizuka, N. Ferroelectric Coordination Polymers Self-Assembled from Mesogenic Zinc(II) Porphyrin and Dipolar Bridging Ligands. Chem Eur. J. 2016, 22, 14213–14218. [Google Scholar] [CrossRef]
- Wang, H.-N.; Meng, X.; Dong, L.-Z.; Chen, Y.; Li, S.-L.; Lan, Y.-Q. Coordination polymer-based conductive materials: Ionic conductivity vs. electronic conductivity. J. Mater. Chem. A 2019, 7, 24059–24091. [Google Scholar] [CrossRef]
- Givaja, G.; Amo-Ochoa, P.; Gomez-Garcia, C.; Zamora, F. Electrical conductive coordination polymers. Chem. Soc. Rev. 2012, 41, 115–147. [Google Scholar] [CrossRef]
- Yue, Q.; Gao, E.-Q. Azide and carboxylate as simultaneous coupler for magnetic coordination polymers. Coord. Chem. Rev. 2019, 382, 1–31. [Google Scholar] [CrossRef]
- Zhou, H.-C.; Kitagawa, S. Metal-Organic Frameworks (MOFs). Chem. Soc. Rev. 2014, 43, 5415–5418. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Feng, L.; Wang, K.; Pang, J.; Bosch, M.; Lollar, C.; Sun, Y.; Qin, J.; Wang, X.; Zhang, P.; et al. Stable Metal–Organic Frameworks: Design, Synthesis, and Applications. Adv. Mater. 2018, 37, 1704303. [Google Scholar] [CrossRef]
- Eddaoudi, M.; Moler, D.B.; Li, H.; Chen, B.; Rheineke, T.M.; O’Keefe, M.; Yaghi, O.M. Modular Chemistry: Secondary Building Units as a Basis for the Design of Highly Porous and Robust Metal−Organic Carboxylate Frameworks. Acc. Chem. Res. 2001, 34, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Ugale, B.; Singh Dhankhar, S.; Nagaraja, C.M. Interpenetrated Metal–Organic Frameworks of Cobalt(II): Structural Diversity, Selective Capture, and Conversion of CO2. Cryst. Growth Des. 2017, 17, 3295–3305. [Google Scholar] [CrossRef]
- Manos, M.J.; Markoulides, M.S.; Malliakas, C.D.; Papaefstathiou, G.S.; Chronakis, N.; Kanatzidis, M.G.; Trikalitis, P.N.; Tasiopoulos, A.J. A Highly Porous Interpenetrated Metal–Organic Framework from the Use of a Novel Nanosized Organic Linker. Inorg. Chem. 2011, 50, 11297–11299. [Google Scholar] [CrossRef]
- Sezginel, K.B.; Feng, T.; Wilmer, C.E. Discovery of hypothetical hetero-interpenetrated MOFs with arbitrarily dissimilar topologies and unit cell shapes. CrystEngComm 2017, 19, 4497–4504. [Google Scholar] [CrossRef]
- Alhamami, M.; Doan, H.; Cheng, C.-H. A Review on Breathing Behaviors of Metal-Organic-Frameworks (MOFs) for Gas Adsorption. Materials 2014, 7, 3198–3250. [Google Scholar] [CrossRef]
- Nouar, F.; Devic, T.; Chevreau, H.; Guillou, N.; Gibson, E.; Clet, G.; Daturi, M.; Vimont, A.; Grenèche, J.M.; Breeze, M.I.; et al. Tuning the breathing behaviour of MIL-53 by cation mixing. Chem. Commun. 2012, 48, 10237–10239. [Google Scholar] [CrossRef]
- Li, H.; Wang, K.; Sun, Y.; Tollar, C.; Li, J.; Zhou, H.-C. Recent advances in gas storage and separation using metal–organic frameworks. Mater. Today 2018, 21, 108–121. [Google Scholar] [CrossRef]
- Li, H.; Li, L.; Lin, R.-B.; Zhou, W.; Zhang, Z.; Xiang, S.; Chen, B. Porous metal-organic frameworks for gas storage and separation: Status and challenges. EnergyChem 2019, 1, 100006. [Google Scholar] [CrossRef]
- Giménez-Marqués, M.; Hidalgo, T.; Serre, C.; Horcajada, P. Nanostructured metal–organic frameworks and their bio-related applications. Coord. Chem. Rev. 2016, 307, 342–360. [Google Scholar] [CrossRef]
- Xue, T.; Xu, C.; Wang, Y.; Wang, Y.; Tian, H.; Zhang, Y. Doxorubicin-loaded nanoscale metal–organic framework for tumor-targeting combined chemotherapy and chemodynamic therapy. Biomater. Sci. 2019, 7, 4615–4623. [Google Scholar] [CrossRef] [PubMed]
- Mínguez Espallargas, G.; Coronado, E. Magnetic functionalities in MOFs: From the framework to the pore. Coord. Chem. Rev. 2018, 47, 533–557. [Google Scholar] [CrossRef]
- Pascanu, V.; González Miera, G.; Ken Inge, A.; Martín-Matute, B. Metal–Organic Frameworks as Catalysts for Organic Synthesis: A Critical Perspective. J. Am. Chem. Soc. 2019, 141, 7223–7234. [Google Scholar] [CrossRef]
- Ricco, R.; Styles, M.J.; Falcaro, P. Metal-Organic Frameworks (MOFs) for Environmental Applications; Ghosh, S.K., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 383–426. [Google Scholar]
- Molavi, H.; Hakimian, A.; Shojaei, A.; Raeiszadeh, M. Selective dye adsorption by highly water stable metal-organic framework: Long term stability analysis in aqueous media. Appl. Surf. Sci. 2018, 445, 424–436. [Google Scholar] [CrossRef]
- Kobielska, P.A.; Howarth, A.J.; Farha, O.K.; Nayak, S. Metal–organic frameworks for heavy metal removal from water. Coord. Chem. Rev. 2018, 358, 92–107. [Google Scholar] [CrossRef]
- Vellingiri, K.; Szulejko, J.E.; Kumar, P.; Kwon, E.E.; Kim, K.-H.; Deep, A.; Boukhvalov, D.W.; Brown, R.J.C. Metal organic frameworks as sorption media for volatile and semi-volatile organic compounds at ambient conditions. Sci. Rep. 2016, 6, 27813. [Google Scholar] [CrossRef]
- Ahmed, I.; Jhung, S.H. Adsorptive desulfurization and denitrogenation using metal-organic frameworks. J. Hazard. Mater. 2016, 301, 259–276. [Google Scholar] [CrossRef]
- Seo, P.W.; Bhadra, B.N.; Ahmed, I.; Khan, N.A.; Jhung, S.H. Adsorptive Removal of Pharmaceuticals and Personal Care Products from Water with Functionalized Metal-organic Frameworks: Remarkable Adsorbents with Hydrogen-bonding Abilities. Sci. Rep. 2016, 6, 34462. [Google Scholar] [CrossRef]
- Yang, J.; Hou, B.; Wang, J.; Tian, B.; Bi, J.; Wang, N.; Li, X.; Huang, X. Nanomaterials for the removal of heavy metals from wastewater. Nanomaterials 2019, 9, 424. [Google Scholar] [CrossRef]
- Zanin, E.; Scapinello, J.; de Oliveira, M.; Rambo, C.L.; Franscescon, F.; Freitas, L.; de Mello, J.M.M.; Fiori, M.A.; Oliveira, J.V.; Magro, J. Adsorption of heavy metals from wastewater graphic industry using clinoptilolite zeolite as adsorbent. Process Saf. Environ. Prot. 2017, 105, 194–200. [Google Scholar] [CrossRef]
- Dias, E.M.; Petit, C. Towards the use of metal–organic frameworks for water reuse: A review of the recent advances in the field of organic pollutants removal and degradation and the next steps in the field. J. Mater. Chem. A 2015, 3, 22484–22506. [Google Scholar] [CrossRef]
- Zhao, L.; Azhar, M.R.; Li, X.; Duan, X.; Sun, H.; Wang, S.; Fang, X. Adsorption of cerium (III) by HKUST-1 metal-organic framework from aqueous solution. J. Colloid Interface Sci. 2019, 542, 421–428. [Google Scholar] [CrossRef]
- Barreto, J.; Xavier, M.D.G.; Ribeiro, R.P.P.L.; Martins, D.; Esteves, I.A.A.C.; Branco, M.; Tirolien, T.; Mota, J.P.B.; Bonfait, G. Neon Adsorption on HKUST-1 and UiO-66 Metal–Organic Frameworks over Wide Pressure and Temperature Ranges. J. Chem. Eng. Data 2019, 64, 5407–5414. [Google Scholar] [CrossRef]
- Ramanayaka, S.; Vithanage, M.; Sarmah, A.; An, T.; Kim, K.-H.; Sik Ok, Y. Performance of metal–organic frameworks for the adsorptive removal of potentially toxic elements in a water system: A critical review. RSC Adv. 2019, 9, 34359–34376. [Google Scholar] [CrossRef]
- Chen, C.-H.; Wang, X.-S.; Li, L.; Huang, Y.-B.; Cao, R. Highly selective sensing of Fe3+ by an anionic metal–organic framework containing uncoordinated nitrogen and carboxylate oxygen sites. Dalton Trans. 2018, 47, 3452–3458. [Google Scholar] [CrossRef] [PubMed]
- Lustig, W.P.; Mukherjee, S.; Rudd, N.D.; Desai, A.V.; Li, J.; Ghosh, S.K. Metal–organic frameworks: Functional luminescent and photonic materials for sensing applications. Chem. Soc. Rev. 2017, 46, 3242–3285. [Google Scholar] [CrossRef]
- Shum, J.; Kam-Keung Leung, P.; Kam-Wing Lo, K. Luminescent Ruthenium(II) Polypyridine Complexes for a Wide Variety of Biomolecular and Cellular Applications. Inorg. Chem. 2019, 58, 2231–2247. [Google Scholar] [CrossRef] [PubMed]
- Amudhan Senthana, S.; Alexander, V. Synthesis, luminescence, and electrochemical studies of tetra- and octanuclear ruthenium(ii) complexes of tolylterpyridine appended calixarenes. New J. Chem. 2016, 40, 10064–10070. [Google Scholar] [CrossRef]
- Ji, G.; Liu, J.; Gao, X.; Sun, W.; Wang, J.; Zhao, S.; Liu, Z. A luminescent lanthanide MOF for selectively and ultra-high sensitively detecting Pb2+ ions in aqueous solution. J. Mater. Chem. A 2017, 5, 10200–10205. [Google Scholar] [CrossRef]
- Yan, B. Lanthanide-Functionalized Metal–Organic Framework Hybrid Systems To Create Multiple Luminescent Centers for Chemical Sensing. Acc. Chem. Res. 2017, 50, 2789–2798. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Xu, H.; Yue, Y.; Cuo, Z.; Yu, J.; Chen, Z.; Gao, J.; Yang, Y.; Qian, G.; Chen, B. A Luminescent Mixed-Lanthanide Metal-Organic Framework Thermometer. J. Am. Chem. Soc. 2012, 134, 3979–3982. [Google Scholar] [CrossRef] [PubMed]
- Kou, W.-T.; Yang, C.-X.; Yan, X.-P. Post-synthetic modification of metal–organic frameworks for chiral gas chromatography. J. Mater. Chem. A 2018, 6, 17861–17866. [Google Scholar] [CrossRef]
- Ardila-Suárez, C.; Díaz-Lasprilla, A.M.; Díaz-Vaca, L.A.; Balbuena, P.B.; . Baldovino-Medrano, V.G.; Ramírez-Caballero, G.E. Synthesis, characterization, and post-synthetic modification of a micro/mesoporous zirconium–tricarboxylate metal–organic framework: Towards the addition of acid active sites. CrystEngComm 2019, 21, 3014–3330. [Google Scholar] [CrossRef]
- Garibay, S.J.; Cohen, S.M. Isoreticular synthesis and modification of frameworks with the UiO-66 topology. Chem. Commun. 2010, 46, 7700–7702. [Google Scholar] [CrossRef]
- Mai, Z.; Liu, D. Synthesis and Applications of Isoreticular Metal–Organic Frameworks IRMOFs-n (n = 1, 3, 6, 8). Cryst. Growth Des. 2019, 19, 7439–7462. [Google Scholar] [CrossRef]
- Yaghi, O.M.; O’Keeffe, M.; Ockwig, N.W.; Chae, H.K.; Eddaoudi, M.; Kim, J. Reticular synthesis and the design of new materials. Nature 2003, 423, 705–714. [Google Scholar] [CrossRef]
- Klinowski, J.; Almeida Paz, F.A.; Silva, P.; Rocha, J. Microwave-Assisted Synthesis of Metal–Organic Frameworks. Dalton Trans. 2011, 40, 321–330. [Google Scholar] [CrossRef]
- Vinu, M.; Lin, W.-C.; Senthil Raja, D.; Han, J.-L.; Lin, C.-H. Microwave-Assisted Synthesis of Nanoporous Aluminum-Based Coordination Polymers as Catalysts for Selective Sulfoxidation Reaction. Polymers 2017, 9, 498. [Google Scholar] [CrossRef]
- Chen, D.; Zhao, J.; Zhang, P.; Dai, S. Mechanochemical synthesis of metal–organic frameworks. Polyhedron 2019, 162, 59–64. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Z.; Ng, M.; Milner, P.J. Rapid mechanochemical synthesis of metal–organic frameworks using exogenous organic base. Dalton Trans. 2020. Advance Article. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.-L.; Sun, W.-Y. The organic ligands with mixed N-/O-donors used in construction of functional metal–organic frameworks. Cryst. Eng. Comm. 2014, 16, 3247–3258. [Google Scholar] [CrossRef]
- Kourtellaris, A.; Moushi, E.E.; Spanopoulos, I.; Tampaxis, C.; Charalambopoulou, G.; Steriotis, T.A.; Papaefstathiou, G.S.; Trikalitis, P.N.; Tasiopoulos, A.J. A microporous Cu2+ MOF based on a pyridyl isophthalic acid Schiff base ligand with high CO2 uptake. Inorg. Chem. Front. 2016, 3, 1527–1535. [Google Scholar] [CrossRef]
- Moushi, E.E.; Kourtellaris, A.; Spanopoulos, I.; Manos, M.J.; Papaefstathiou, G.S.; Trikalitis, P.N.; Tasiopoulos, A.J. A Microporous Co2+ Metal Organic Framework with Single-Crystal to Single-Crystal Transformation Properties and High CO2 Uptake. Cryst. Growth Des. 2015, 15, 185–193. [Google Scholar] [CrossRef]
- Chen, Z.; Adil, K.; Weselinski, L.J.; Belmabkhout, Y.; Eddaoudi, M.J. A supermolecular building layer approach for gas separation and storage applications: The eea and rtl MOF platforms for CO 2 capture and hydrocarbon separation. Mater. Chem. A 2015, 3, 6276–6281. [Google Scholar] [CrossRef]
- Li, H.; Eddaoudi, M.; O’Keeffe, M.; Yaghi, O.M. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 1999, 402, 276–279. [Google Scholar] [CrossRef]
- Sapchenko, S.A.; Dybtsev, D.N.; Damsonenko, D.G.; Fedin, V.P. Synthesis, crystal structures, luminescent and thermal properties of two new metal–organic coordination polymers based on zinc(II) carboxylates. New J. Chem. 2010, 34, 2445–2450. [Google Scholar] [CrossRef]
- Tranchemontagne, D.J.; Hunt, J.R.; Yaghi, O.M. Room temperature synthesis of metal-organic frameworks: MOF-5, MOF-74, MOF-177, MOF-199, and IRMOF-0. Tetrahedron 2008, 64, 8553–8557. [Google Scholar] [CrossRef]
- Clausen, H.F.; Poulsen, R.D.; Bond, A.D.; Chevallier, M.-A.S.; Iversen, B.B. Solvothermal synthesis of new metal organic framework structures in the zinc terephthalic acid dimethyl formamide system. J. Solid State 2005, 178, 3342–3351. [Google Scholar] [CrossRef]
- Manos, M.J.; Moushi, E.E.; Papaefstathiou, G.S.; Tasiopoulos, A.J. New Zn2+ Metal Organic Frameworks with Unique Network Topologies from the Combination of Trimesic Acid and Amino-Alcohols. Cryst. Growth Des. 2012, 12, 5471–5480. [Google Scholar] [CrossRef]
- Guesh, K.; Caiuby, C.A.D.; Mayoral, Á.; Díaz-García, M.; Díaz, I.; Sanchez-Sanchez, M. Sustainable Preparation of MIL-100(Fe) and Its Photocatalytic Behavior in the Degradation of Methyl Orange in Water. Growth Des. 2017, 17, 1806–1813. [Google Scholar] [CrossRef]
- Horcajada, P.; Surblé, S.; Serre, C.; Hong, D.-Y.; Seo, Y.-K.; Chang, J.-S.; Grenèche, J.-M.; Margiolaki, I.; Férey, G. Synthesis and catalytic properties of MIL-100(Fe), an iron(iii) carboxylate with large pores. Chem. Commun. 2007, 2820–2822. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, G.K.H.; Vaidhyanathan, R.; Taylor, J.M. Phosphonate and sulfonate metal organic frameworks. Chem. Soc. Rev. 2009, 38, 1430–1449. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Singh, U.P.; Butcher, R.J. Luminescent sulfonate coordination polymers: Synthesis, structural analysis and selective sensing of nitroaromatic compounds. CrystEngComm 2017, 19, 7009–7020. [Google Scholar] [CrossRef]
- ZareKarizi, F.; Johariana, M.; Morsali, A. Pillar-layered MOFs: Functionality, interpenetration, flexibility and applications. J. Mater. Chem. A 2018, 6, 19288–19329. [Google Scholar] [CrossRef]
- Yin, Z.; Zhou, Y.-L.; Zeng, M.-H.; Kurmoo, M. The concept of mixed organic ligands in metal–organic frameworks: Design, tuning and functions. Dalton Trans. 2015, 44, 5258–5275. [Google Scholar] [CrossRef]
- Abednatanzi, S.; Gohari Derakhshandeh, P.; Depauw, H.; Coudert, F.-X.; Vrielinck, H.; Van Der Voort, P.; Leus, K. Mixed-metal metal–organic frameworks. Chem. Soc. Rev. 2019, 48, 2535–2565. [Google Scholar] [CrossRef]
- Masoomi, M.Y.; Morsali, A.; Dhakshinamoorthy, A.; Garcia, H. Mixed-Metal MOFs: Unique Opportunities in Metal–Organic Framework (MOF) Functionality and Design. Angew. Chem. Int. Ed. 2019, 58, 15188–15205. [Google Scholar] [CrossRef]
- Zhai, Q.-G.; Bu, X.; Mao, C.; Zhao, X.; Feng, P. Systematic and Dramatic Tuning on Gas Sorption Performance in Heterometallic Metal–Organic Frameworks. J. Am. Chem. Soc. 2016, 138, 2524–2527. [Google Scholar] [CrossRef]
- Milios, C.J.; Stamatatos, T.C.; Perlepes, S.P. The coordination chemistry of pyridyl oximes. Polyhedron 2006, 25, 134–194. [Google Scholar] [CrossRef]
- Papatriantafyllopoulou, C.; Jones, L.F.; Nguyen, T.D.; Matamoros-Salvador, N.; Cunha-Silva, L.; Almeida Paz, F.A.; Rocha, J.; Evangelisti, M.; Brechin, E.K.; Perlepes, S.P. Using pyridine amidoximes in 3d-metal cluster chemistry: A novel ferromagnetic Ni12 complex from the use of pyridine-2-amidoxime. Dalton Trans. 2008, 3153–3155. [Google Scholar] [CrossRef] [PubMed]
- Efthymiou, C.G.; Cunha-Silva, L.; Perlepes, S.P.; Brechin, E.K.; Inglis, R.; Evangelisti, M.; Papatriantafyllopoulou, C. In search of molecules displaying ferromagnetic exchange: Multiple-decker Ni12 and Ni16 complexes from the use of pyridine-2-amidoxime. Dalton Trans. 2016, 17409–17419. [Google Scholar] [CrossRef] [PubMed]
- Papatriantafyllopoulou, C.; Stamatatos, T.C.; Wernsdorfer, W.; Teat, S.J.; Tasiopoulos, A.J.; Escuer, A.; Perlepes, S.P. Combining Azide, Carboxylate, and 2-Pyridyloximate Ligands in Transition-Metal Chemistry: Ferromagnetic NiII5 Clusters with a Bowtie Skeleton. Inorg. Chem. 2010, 49, 10486–10496. [Google Scholar] [CrossRef] [PubMed]
- Polyzou, C.D.; Efthymiou, C.G.; Escuer, A.; Cunha-Silva, L.; Papatriantafyllopoulou, C.; Perlepes, S.P. In search of 3d/4f-metal single-molecule magnets: Nickel(II)/lanthanide(III) coordination clusters. Pure Appl. Chem. 2013, 85, 315–327. [Google Scholar] [CrossRef]
- Papatriantafyllopoulou, C.; . Stamatatos, T.C.; . Efthymiou, C.G.; Cunha-Silva, L.; Almeida Paz, F.A.; Perlepes, S.P.; Christou, G. A High-Nuclearity 3d/4f Metal Oxime Cluster: An Unusual Ni8Dy8 “Core-Shell” Complex from the Use of 2-Pyridinealdoxime. Inorg. Chem. 2010, 49, 9743–9745. [Google Scholar] [CrossRef]
- Papatriantafyllopoulou, C.; Estrader, M.; Efthymiou, C.G.; Dermitzaki, D.; Gkotsis, K.; Terzis, A.; Diaz, C.; Perlepes, S.P. In search for mixed transition metal/lanthanide single-molecule magnets: Synthetic routes to NiII/TbIII and NiII/DyIII clusters featuring a 2-pyridyl oximate ligand. Polyhedron 2009, 28, 1652–1655. [Google Scholar] [CrossRef]
- Efthymiou, C.G.; Mylonas-Margaritis, I.; Das Gupta, S.; Tasiopoulos, A.; Nastopoulos, V.; Christou, G.; Perlepes, S.P.; Papatriantafyllopoulou, C. Synthesis and characterisation of new Ni2Mn, Ni2Mn2 and Mn8 clusters by the use of 2-pyridyl oximes. Polyhedron 2019, 171, 330–337. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Wernsdorfer, W.; Shiddiq, M.; Abboud, K.A.; Hill, S.; Christou, G. Supramolecular aggregates of single-molecule magnets: Exchange-biased quantum tunneling of magnetization in a rectangular [Mn3]4 tetramer. Chem. Sci. 2016, 7, 1156–1173. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Shiddiq, M.; Ghosh, T.; Abboud, K.A.; Hill, S.; Christou, G. Covalently Linked Dimer of Mn3 Single-Molecule Magnets and Retention of Its Structure and Quantum Properties in Solution. J. Am. Chem. Soc. 2015, 137, 7160–7168. [Google Scholar] [CrossRef]
- Ghosh, T.; Abboud, K.A.; Christou, G. New MnIIMnIII8 and MnII2MnIII10MnIV2 clusters from the reaction of methyl 2-pyridyl ketone oxime with [Mn12O12(O2CR)16(H2O)4]. Polyhedron 2019, 173, 114145. [Google Scholar] [CrossRef]
- Stamatatos, T.C.; Foguet-Albiol, D.; Stoumpos, C.C.; Raptopoulou, C.P.; Terzis, A.; Wernsdorfer, W.; Perlepes, S.P.; Christou, G. New Mn3 structural motifs in manganese single-molecule magnetism from the use of 2-pyridyloximate ligands. Polyhedron 2007, 26, 2165–2168. [Google Scholar] [CrossRef]
- Escuer, A.; Vlahopoulou, G.; Mautner, F.A. Assembly of [MnII2MnIII2] S = 9 Clusters via Azido Bridges: A New Single-Chain Magnet. Inorg. Chem. 2011, 50, 2717–2719. [Google Scholar] [CrossRef]
- Mowson, A.M.; Nguyen, T.N.; Abboud, K.A.; Christou, G. Dimeric and tetrameric supramolecular aggregates of single-molecule magnets via carboxylate substitution. Inorg. Chem. 2013, 52, 12320–12322. [Google Scholar] [CrossRef]
- Fabelo, O.; Pasan, J.; Canadillas-Delgado, L.; Delgado, F.S.; Lloret, F.; Julve, M.; Ruiz-Perez, C. Crystal structure and magnetic properties of two isomeric three-dimensional pyromellitate-containing cobalt(II) complexes. Inorg. Chem. 2008, 47, 8053–8061. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.-C.; Guo, H.-B.; Deng, J.-H.; Chen, Q.; Luo, X.-Z. Two coordination polymers of benzene-1, 2, 4, 5-tetracarboxylic acid (H4BTC): In situ ligand syntheses, structures, and luminescent properties. CrystEngComm 2015, 17, 3519–3525. [Google Scholar] [CrossRef]
- Jia, H.-P.; Li, W.; Ju, Z.-F.; Zhang, J. [Co 5 (µ 3-OH) 2 (btec) 2 (bpp)] n: A three-dimensional homometallic molecular metamagnet built from the mixed hydroxide/carboxylate-bridged ferrimagnetic-like chains. Dalton Trans. 2007, 33, 3699–3704. [Google Scholar] [CrossRef]
- Luo, Y.-H.; Tao, C.-Z.; Zhang, D.-E.; Ma, J.-J.; Liu, L.; Tong, Z.-W.; Yu, X. Three new three dimensional Zn (II)-benzenetetracarboxylate coordination polymers: Syntheses, crystal structures and luminescent properties. Polyhedron 2017, 123, 69–74. [Google Scholar] [CrossRef]
- Ji, W.-J.; Hu, M.-C.; Li, S.-N.; Jiang, Y.-C.; Zhai, Q.-C. Ionic liquids used for synthesis of supramolecular isomeric open-frameworks as photocatalysts for visible-light-driven degradation of organic dyes. CrystEngComm 2014, 16, 3474–3477. [Google Scholar] [CrossRef]
- Geng, J.-P.; Wang, Z.-X.; He, Z.; Xiao, H.-P.; Li, M.-X. A novel 2D coordination polymer based on triangular-shaped [Mn3O] units bridged by sodium ions and benzene-1, 2, 4, 5-tetracarboxylate. Inorg. Chem. Commun. 2011, 14, 997–1000. [Google Scholar] [CrossRef]
- Sun, L.-P.; Niu, S.-Y.; Jin, J.; Yang, G.-D.; Ye, L. Synthesis, Structure and Surface Photovoltage of a Series of NiII Coordination Polymers. Eur. J. Inorg. Chem. 2006, 5130–5137. [Google Scholar] [CrossRef]
- Sanselme, M.; Greneche, J.M.; Riou-Cavellec, M.; Ferey, G. [Fe2(C10O8H2)]: An antiferromagnetic 3D iron(ii) carboxylate built from ferromagnetic edge-sharing octahedral chains (MIL-62). Chem. Commun. 2002, 2172–2173. [Google Scholar] [CrossRef] [PubMed]
- Olchowka, J.; Falaise, C.; Volkringer, C.; Henry, N.; Loiseau, T. Structural Observations of Heterometallic Uranyl Copper (II) Carboxylates and Their Solid-State Topotactic Transformation upon Dehydration. Chem.-Eur. J. 2013, 19, 2012–2022. [Google Scholar] [CrossRef] [PubMed]
- Bernasek, E. Pyridineamidoximes. J. Org. Chem. 1957, 22, 1263. [Google Scholar] [CrossRef]
- Orama, M.; Saarinen, H.; Korvenranta, J. Formation of trinuclear copper(II) complexes with three pyridine oxime ligands in aqueous solution. J. Coord. Chem. 1990, 22, 183–190. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT–Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- McArdle, P.; Gilligan, K.; Cunningham, D.; Dark, R.; Mahon, M. A method for the prediction of the crystal structure of ionic organic compounds—The crystal structures of o-toluidinium chloride and bromide and polymorphism of bicifadine hydrochloride. CrystEngComm 2004, 6, 303–309. [Google Scholar] [CrossRef]
- Brandenburg, K. DIAMOND, Version 2003.2001d; Crystal Impact GbR: Bonn, Germany, 2006. [Google Scholar]
- Mylonas-Margaritis, I.; Winterlich, M.; Efthymiou, C.G.; Lazarides, T.; McArdle, P.; Papatriantafyllopoulou, C. New insights into oximic ligands: Synthesis and characterization of 1D chains by the use of pyridine 2-amidoxime and polycarboxylates. Polyhedron 2018, 151, 360–368. [Google Scholar] [CrossRef]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; van Rijn, J.; Verschoor, G.C. Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen–sulphur donor ligands; the crystal and molecular structure of aqua [1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]copper(II) perchlorate. J. Chem. Soc. Dalton Trans 1984, 1346–1356. [Google Scholar] [CrossRef]
- Blatov, V.A.; Shevchenko, A.P.; Proserpio, D.M. Applied topological analysis of crystal structures with the program package ToposPro. Cryst. Growth Des. 2014, 14, 3576–3586. [Google Scholar] [CrossRef]
- O’Keeffe, M.; Peskov, M.A.; Ramsden, S.J.; Yaghi, O.M. The reticular chemistry structure resource (RCSR) database of, and symbols for, crystal nets. Acc. Chem. Res. 2008, 41, 1782–1789. [Google Scholar] [CrossRef] [PubMed]
- Alexandrov, E.V.; Blatov, V.A.; Kochetkov, A.V.; Proserpio, D.M. Underlying nets in three-periodic coordination polymers: Topology, taxonomy and prediction from a computer-aided analysis of the Cambridge Structural Database. CrystEngComm 2011, 13, 3947–3958. [Google Scholar] [CrossRef]
- Lagergren, S. Zur Theorie der Sogenannten Absorption Gelöster Stoffe; PA Norstedt & Söner: Stockholm, Sweden, 1898; pp. 1–39. [Google Scholar]
- Ho, Y.S.; Mckay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Zhang, K.-D.; Tsai, F.-C.; Ma, N.; Xia, Y.; Liu, H.-L.; Zhan, X.-Q.; Yu, X.-Y.; Zeng, X.-Z.; Jiang, T.; Shi, D.; et al. Adsorption behavior of high stable Zr-based MOFs for the removal of acid organic dye from water. Materials 2017, 10, 205. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part I. Solids. J. Am. Chem. Soc. 1915, 38, 2221–2295. [Google Scholar] [CrossRef]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Chilton, N.F.; Anderson, R.P.; Turner, L.D.; Soncini, A.; Murray, K.S. PHI: A powerful new program for the analysis of anisotropic monomeric and exchange-coupled polynuclear d- and f-block complexes. J. Comput. Chem. 2013, 34, 1164–1175. [Google Scholar] [CrossRef]
| Complex | 1 | 2 | 3 |
| Empirical formula | C16H15N3O11Zn | C11H8N3O5Zn | C11H10N3O6 Zn |
| Formula weight | 490.68 | 327.57 | 346.60 |
| Crystal system | Triclinic | Triclinic | Monoclinic |
| Space group | Pī | Pī | P21/n |
| a (Å) | 8.1151(3) | 6.9864(7) | 11.4913(8) |
| b (Å) | 9.8550(4) | 8.9530(8) | 6.6261(5) |
| c (Å) | 11.8506(5) | 10.4524(9) | 16.1381(12) |
| α (o) | 93.283(3) | 83.697(7)° | 90 |
| β (o) | 105.135(3) | 94.887(2) | 104.335(8)° |
| γ (o) | 92.769(3) | 71.582(9)° | 90 |
| V (Å3) | 911.40(6) | 612.98(10) | 1190.54(16) |
| Ζ | 2 | 2 | 4 |
| ρcalc (g cm−3) | 1.788 | 1.775 | 1.934 |
| Radiation, λ (Å) | 0.71073 | 1.54184 | 0.71073 |
| μ (mm−1) | 1.420 | 3.042 | 2.099 |
| Temperature (K) | 297.9(6) | 100(2) | 100(2) |
| Measd/independent reflns (Rint) | 4250/3667 (0.0439) | 3860/2400 (0.0451) | 4714/2088 (0.0371) |
| Parameters refined | 316 | 197 | 197 |
| GoF (on F2) | 1.059 | 1.078 | 1.055 |
| R1a (I > 2σ(Ι)) | 0.0352 | 0.0517 | 0.0353 |
| wR2b (I > 2σ(Ι)) | 0.0834 | 0.1377 | 0.0879 |
| (Δρ)max/(Δρ)min (e Å−3) | 0.572/−0.504 | 2.839/−0.831 | 0.573/−0.632 |
| Complex | 4 | 5 | 6 |
| Empirical formula | C22H28N6O16Co2 | C22H28N6O16Mn2 | C14H15N4O6Cu |
| Formula weight | 750.36 | 742.38 | 398.84 |
| Crystal system | Monoclinic | Monoclinic | Triclinic |
| Space group | P21/n | P21/n | Pī |
| a (Å) | 10.9732(9) | 11.1773(8) | 6.5039(4) |
| b (Å) | 11.7514(8) | 12.2116(8) | 10.0104(9) |
| c (Å) | 11.6014(13) | 11.5293(9) | 12.7604(9) |
| a (o) | 90 | 90 | 96.743(6) |
| β (o) | 107.619(10) | 106.542(7) | 91.660(5) |
| γ (o) | 90 | 90 | 108.375(7) |
| V (Å3) | 1425.8(2) | 1508.5(2) | 781.05(11) |
| Ζ | 2 | 2 | 2 |
| ρcalc (g cm−3) | 1.748 | 1.634 | 1.696 |
| Radiation, λ (Å) | 1.54184 | 0.71073 | 0.71073 |
| μ (mm−1) | 9.917 | 0.921 | 1.440 |
| Temperature (K) | 100(2) | 298.0(2) | 299.0(1) |
| Measd/independent reflns (Rint) | 5285/2830 (0.0324) | 3603/2795 (0.0680) | 3616/2040 (0.1137) |
| Parameters refined | 212 | 244 | 240 |
| GoF (on F2) | 1.112 | 1.047 | 0.873 |
| R1a (I > 2σ(Ι)) | 0.0547 | 0.0479 | 0.0526 |
| wR2b (I > 2σ(Ι)) | 0.1500 | 0.1076 | 0.0868 |
| (Δρ)max/(Δρ)min (e Å−3) | 0.586/−0.602 | 0.825/−0.399 | 0.823/−0.678 |
| Complex | 7∙2H2O | 8 | 9 |
| Empirical formula | C24H34N4O18Zn2 | C30H32N6O12Cu2 | C12H9N2O6Cu2 |
| Formula weight | 797.29 | 795.69 | 404.29 |
| Crystal system | Triclinic | Monoclinic | Monoclinic |
| Space group | Pī | P21/c | I2/a |
| a (Å) | 7.1623(7) | 6.4617(3) | 16.3272(7) |
| b (Å) | 8.3966(8) | 25.5232(9) | 10.4575(4) |
| c (Å) | 13.4476(10) | 10.1083(5) | 15.0472(7) |
| a (o) | 106.056(8) | 90 | 90 |
| β (o) | 104.078(8) | 106.321(5) | 102.188(5) |
| γ (o) | 90.547(8) | 90 | 90 |
| V (Å3) | 751.27(12) | 1599.91(13) | 2511.27(19) |
| Ζ | 1 | 2 | 8 |
| ρcalc (g cm−3) | 1.762 | 1.652 | 2.139 |
| Radiation, λ (Å) | 1.54184 | 0.71073 | 1.54184 |
| μ (mm−1) | 2.785 | 1.404 | 4.522 |
| Temperature (K) | 100(2) | 299.0(2) | 100(2) |
| Measd/independent reflns (Rint) | 4859/2943 (0.0323) | 3877/2762 (0.0754) | 4290/2480 (0.0297) |
| Parameters refined | 225 | 233 | 204 |
| GoF (on F2) | 1.087 | 0.954 | 1.097 |
| R1a (I > 2σ(Ι)) | 0.0638 | 0.0433 | 0.0501 |
| wR2b (I > 2σ(Ι)) | 0.1890 | 0.0978 | 0.1471 |
| (Δρ)max/(Δρ)min (e Å−3) | 0.957/−1.077 | 0.450/−0.496 | 1.587/−1.066 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mylonas-Margaritis, I.; Gérard, A.; Skordi, K.; Mayans, J.; Tasiopoulos, A.; McArdle, P.; Papatriantafyllopoulou, C. From 1D Coordination Polymers to Metal Organic Frameworks by the Use of 2-Pyridyl Oximes. Materials 2020, 13, 4084. https://doi.org/10.3390/ma13184084
Mylonas-Margaritis I, Gérard A, Skordi K, Mayans J, Tasiopoulos A, McArdle P, Papatriantafyllopoulou C. From 1D Coordination Polymers to Metal Organic Frameworks by the Use of 2-Pyridyl Oximes. Materials. 2020; 13(18):4084. https://doi.org/10.3390/ma13184084
Chicago/Turabian StyleMylonas-Margaritis, Ioannis, Auban Gérard, Katerina Skordi, Julia Mayans, Anastasios Tasiopoulos, Patrick McArdle, and Constantina Papatriantafyllopoulou. 2020. "From 1D Coordination Polymers to Metal Organic Frameworks by the Use of 2-Pyridyl Oximes" Materials 13, no. 18: 4084. https://doi.org/10.3390/ma13184084
APA StyleMylonas-Margaritis, I., Gérard, A., Skordi, K., Mayans, J., Tasiopoulos, A., McArdle, P., & Papatriantafyllopoulou, C. (2020). From 1D Coordination Polymers to Metal Organic Frameworks by the Use of 2-Pyridyl Oximes. Materials, 13(18), 4084. https://doi.org/10.3390/ma13184084







