Electron Microscopy Characterization of the High Temperature Degradation of the Aluminide Layer on Turbine Blades Made of a Nickel Superalloy
Abstract
1. Introduction
2. Materials and Methods
3. Results
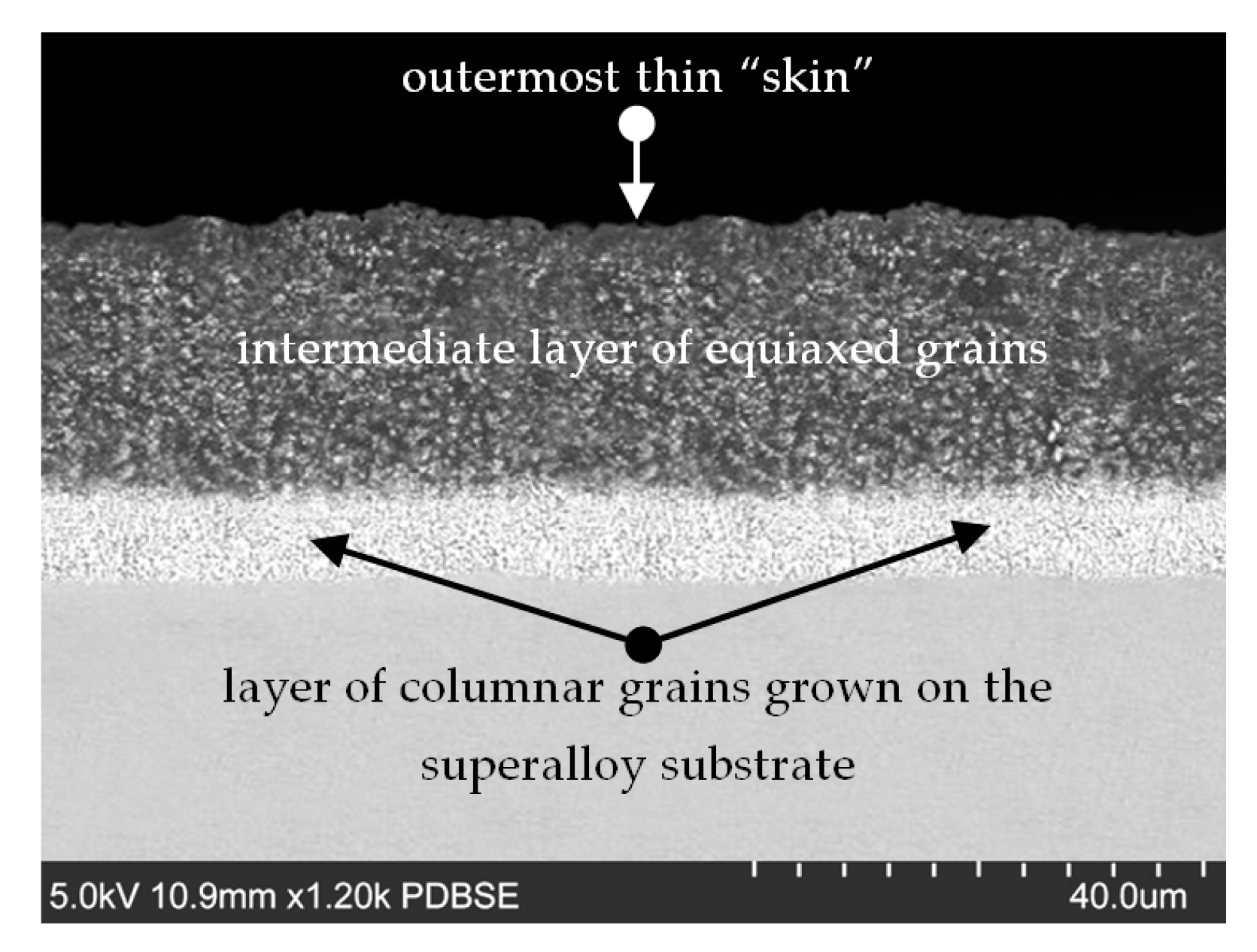
3.1. New Blades (Prior to the Exposure to High Temperatures)
- (a)
- The outermost thin “skin”
- (b)
- An intermediate layer of equiaxed grains
- (c)
- A layer of columnar grains grown on the superalloy substrate.
3.2. Coatings on the Blades Exposed to 1050 °C
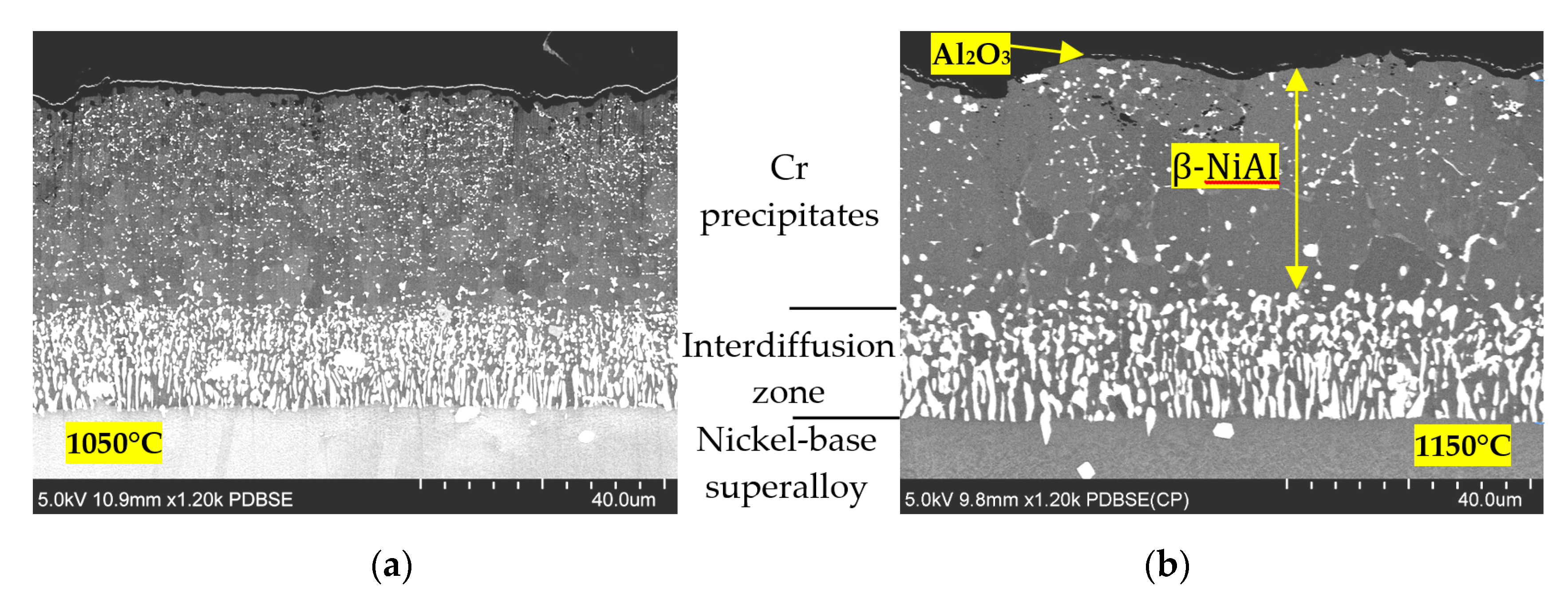
3.3. Comparison of the Coatings Exposed to 1050 and 1150 °C
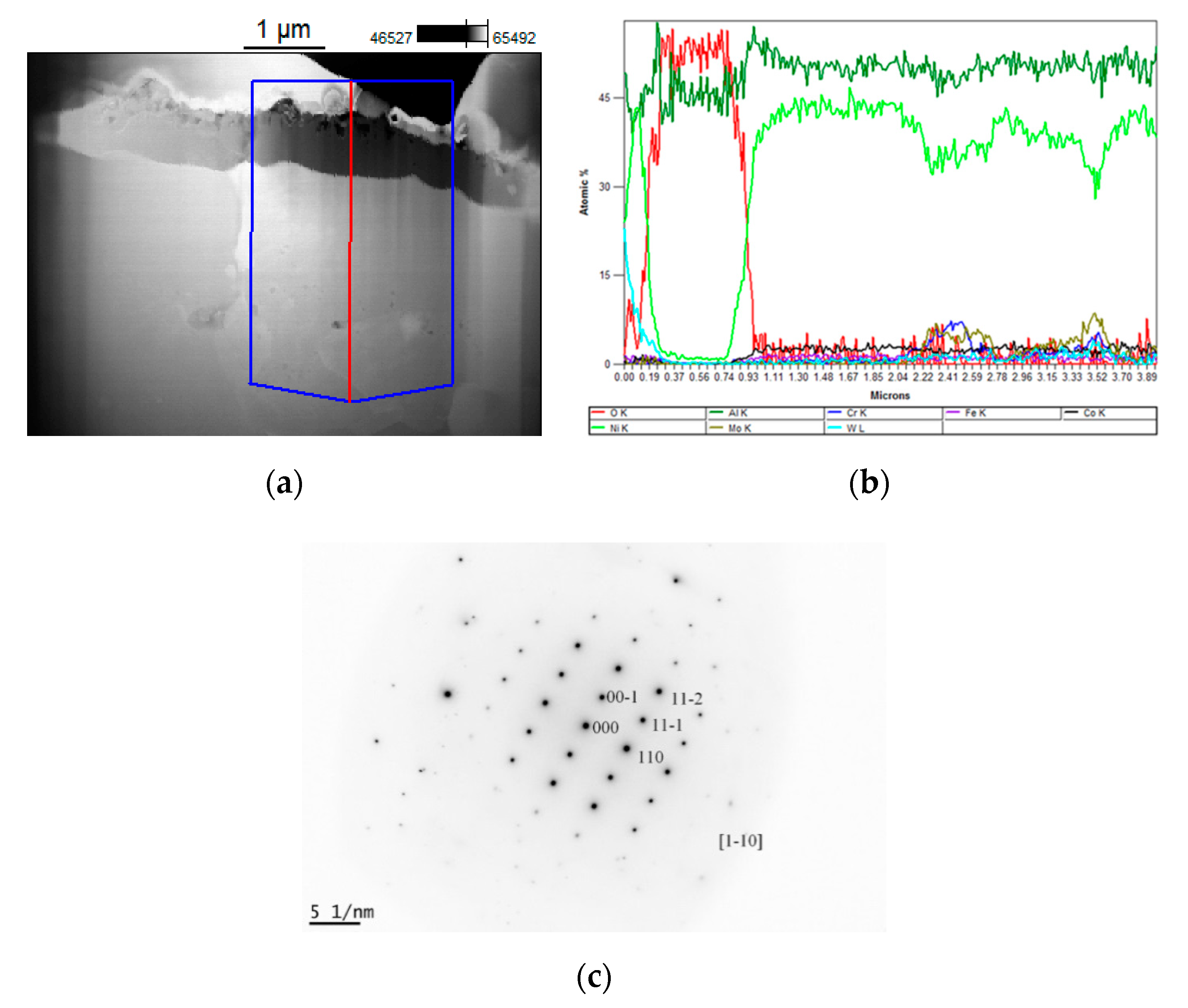
3.4. Surface Morphologies and Microstructure of Near-Surface Zone
- the delamination of the coating layer was not observed in the study;
- the outer most layer of coatings, made of alumina, is approximately 1 μm thick;
- the size of alumina grains is much larger in the sample exposed to 1150 °C, and exceeds the thickness of the layer;
- there are numerous pores present in the coatings, which are much smaller in the case of the samples exposed to 1050 °C and are located underneath alumina layer,
- particles on the outer surface are observed, which, in the case of the exposure to 1050 °C, have an irregular shape, while in the case of 1150 °C, they are spherical.
4. Conclusions
- formation of a thin layer of alumina Al2O3,
- coarsening of the microstructure of the coatings,
- formation of pores,
- outward diffusion of Fe and Cr to the surface,
- formation and subsequent coarsening of Fe-Cr particles on the surface of the alumina layer.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DeMasi-Marcin, J.T.; Gupta, D.K. Protective coatings in the gas turbine engine. Surf. Coat. Technol. 1994, 68, 1–9. [Google Scholar] [CrossRef]
- Smith, A.B.; Kempster, A.; Smith, J. Vapour aluminide coating of internal cooling channels, in turbine blades and vanes. Surf. Coat. Technol. 1999, 120, 112–117. [Google Scholar] [CrossRef]
- Hetmańczyk, M.; Swadźba, L.; Mendala, B. Advanced materials and protective coatings in aero-engines application. JAMME 2007, 24, 372–381. [Google Scholar]
- Han, J.; Dutta, S.; Ekkad, S. Gas Turbine Heat Transfer and Cooling Technology; Taylor and Francis: New York, NY, USA, 2012. [Google Scholar]
- Rajendran, R. Gas turbine coatings—An overview. Eng. Fail. Anal. 2012, 26, 355–369. [Google Scholar] [CrossRef]
- Rannou, B.; Bouchaud, B.; Balmain, J.; Bonnet, G.; Kolarik, V.; Pedraza, F. Comparative isothermal oxidation behaviour of new aluminide coatings from Al particles containing slurries and conventional out-of-pack aluminide coatings. Oxid. Met. 2014, 81, 139–149. [Google Scholar] [CrossRef]
- Warnes, B.M.; Punola, D.C. Clean diffusion coatings by chemical vapor deposition. Surf. Coat. Technol. 1997, 94, 1–6. [Google Scholar] [CrossRef]
- Nicholls, J.R.; Long, K.A.; Simms, N.J. Diffusion coatings. In Shreir’S Corrosion; Richardson, T., Ed.; Elsevier: Oxford, UK, 2010; pp. 2532–2555. [Google Scholar]
- Bozza, F.; Bolelli, G.; Giolli, C.; Giorgetti, A.; Lusvarghi, L.; Sassatelli, P.; Scrivani, A.; Candeli, A.; Thoma, M. Diffusion mechanisms and microstructure development in pack aluminizing of Ni-based alloys. Surf. Coat. Technol. 2014, 239, 147–159. [Google Scholar] [CrossRef]
- Lu, T.; Yao, D.; Zhou, C. Low-temperature formation of aluminide coatings on Ni-base superalloys by pack cementation process. Chin. J. Aeronaut. 2010, 23, 381–385. [Google Scholar]
- Kohlscheen, J.; Stock, H.R. Deposition of silicon enriched nickel aluminide coatings on internally cooled airfoils. Surf. Coat. Technol. 2008, 203, 476–479. [Google Scholar] [CrossRef]
- Swadźba, R.; Wiedermann, J.; Hetmańczyk, M.; Swadźba, L.; Witala, B.; Moskal, G.; Komendera, Ł. Microstructural examination of TGO formed during preoxidation on Pt-aluminizedNi-based superalloy. Mater. Corros. 2014, 65, 319–323. [Google Scholar] [CrossRef]
- Mu, N.; Izumi, T.; Zhang, L.; Gleeson, B. The development and performance of novel Pt + Hf modified γ′-Ni3Al + γ-Ni bond coatings for advanced thermal barrier coatings systems. Miner. Met. Mater. Soc. 2008, 38, 629–637. [Google Scholar]
- Benoist, J.; Badawi, K.F.; Malie, A.; Ramade, C. Microstructure of Pt-modified aluminide coatings on Ni-based superalloys. Surf. Coat. Technol. 2004, 182, 14–23. [Google Scholar] [CrossRef]
- Pint, B.A.; Haynes, J.A.; Besmann, T.M. Effect of Hf and Y additions on aluminide coating performance. Surf. Coat. Technol. 2010, 204, 3287–3293. [Google Scholar] [CrossRef]
- Das, D.K. Microstructure and high temperature oxidation behavior of Pt-modified aluminide bond coats on Ni-base superalloys. Prog. Mater. Sci. 2013, 58, 151–182. [Google Scholar] [CrossRef]
- Fan, Q.; Wang, T.; Tan, X.; Huow, E. High temperature oxidation resistance of Pt/Zr-modified aluminide coating. Corros. Sci. Prot. Technol. 2018, 30, 395–400. [Google Scholar]
- Yang, Y.; Jiang, C.; Yao, H.; Bao, Z.; Zhu, S.; Wang, F. Preparation and enhanced oxidation performance of a Hf-dopedsingle-phase Pt-modified aluminide coating. Corros. Sci. 2016, 113, 17–25. [Google Scholar] [CrossRef]
- Kim, K.; Jun, J.; Lee, J. High temperature corrosion study on yttrium modified aluminide coatings on IN 713C. J. Phys. Colloq. 1993, 03, 521–529. [Google Scholar] [CrossRef]
- Ahmadi, H.; Li, D.Y. Mechanical and tribological properties of aluminide coating modified with yttrium. Surf. Coat. Technol. 2002, 161, 210–217. [Google Scholar] [CrossRef]
- Göbel, M.; Rahmel, A.; Schütze, M. The cyclic-oxidation behavior of several nickel-base single-crystal superalloys without and with coatings. Oxid. Met. 1994, 41, 271–300. [Google Scholar] [CrossRef]
- Smialek, J.L.; Barrett, C.S.; Schaeffer, J.C. Design for oxidation. In ASM Handbook: Design for Properties; ASM International: Materials Park, OH, USA, 1997; Volume 20, pp. 589–602. [Google Scholar]
- Latief, H.; Kakehi, K.; Tashiro, Y. Oxidation behavior characteristics of an aluminized Ni-based single crystal superalloy CM186LC between 900 °C and 1100 °C in air. J. Ind. Eng. Chem. 2013, 19, 1926–1932. [Google Scholar] [CrossRef]
- Błachnio, J.; Bogdan, M.; Zasada, D. Increased temperature impact on durability of gas turbine blades. Eksploat. Niezawodn. 2017, 19, 48–53. [Google Scholar] [CrossRef]
- Bogdan, M.; Błachnio, J.; Spychała, J.; Zasada, D. Assessment of usability of the exploited gas turbine blade heat-resistant coatings. Eng. Fail. Anal. 2019, 105, 337–346. [Google Scholar] [CrossRef]
- Xiaotong, G.; Weiwei, Z.; Chengbo, X.; Longfei, L.; Stoichko, A.; Yunrong, Z.; Qiang, F. Evaluation of microstructural degradation in a failed gas turbine blade due to overheating. Eng. Fail. Anal. 2019, 103, 308–318. [Google Scholar]
- Mishra, R.K.; Thomas, J.; Srinivasan, K.; Nandi, V.; Raghavendra, R. Failure analysis of an un-cooled turbine blade in an aero gas turbine engine. Eng. Fail. Anal. 2017, 79, 836–844. [Google Scholar] [CrossRef]
- Błachnio, J.; Bogdan, M. A non-destructive method to assess condition of gas turbine blades based on the analysis of blade surface image. Russ. J. Nondestruct. 2010, 46, 860–866. [Google Scholar] [CrossRef]
- Bogdan, M.; Derlatka, M.; Błachnio, J. Concept of computer-aided assessment of the technical condition of operated gas turbine vanes. Pol. Marit. Res. 2018, 25, 104–112. [Google Scholar] [CrossRef]
- Bogdan, M.; Błachnio, J.; Kułaszka, A.; Derlatka, M. Assessing the condition of gas turbine rotor blades with the optoelectronic and thermographic methods. Metals 2019, 9, 31. [Google Scholar] [CrossRef]
- Pedraza, F.; Mollard, M.; Rannou, B.; Bouchaud, B.; Balmain, J.; Bonnet, G. Oxidation Resistance of thermal barrier coatings based on hollow alumina particles. Oxid. Met. 2016, 85, 231–244. [Google Scholar] [CrossRef]




| C | Mn | Si | Cr | Fe | Co | Mo | W | Al | B | Ni |
|---|---|---|---|---|---|---|---|---|---|---|
| max | max | max | max | Rest | ||||||
| 0.1 | 0.3 | 0.6 | 9.0 | 4.0 | 14 | 10.3 | 5.0 | 4.5 | 0.02 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogdan, M.; Zieliński, W.; Płociński, T.; Kurzydłowski, K.J. Electron Microscopy Characterization of the High Temperature Degradation of the Aluminide Layer on Turbine Blades Made of a Nickel Superalloy. Materials 2020, 13, 3240. https://doi.org/10.3390/ma13143240
Bogdan M, Zieliński W, Płociński T, Kurzydłowski KJ. Electron Microscopy Characterization of the High Temperature Degradation of the Aluminide Layer on Turbine Blades Made of a Nickel Superalloy. Materials. 2020; 13(14):3240. https://doi.org/10.3390/ma13143240
Chicago/Turabian StyleBogdan, Mariusz, Witold Zieliński, Tomasz Płociński, and Krzysztof Jan Kurzydłowski. 2020. "Electron Microscopy Characterization of the High Temperature Degradation of the Aluminide Layer on Turbine Blades Made of a Nickel Superalloy" Materials 13, no. 14: 3240. https://doi.org/10.3390/ma13143240
APA StyleBogdan, M., Zieliński, W., Płociński, T., & Kurzydłowski, K. J. (2020). Electron Microscopy Characterization of the High Temperature Degradation of the Aluminide Layer on Turbine Blades Made of a Nickel Superalloy. Materials, 13(14), 3240. https://doi.org/10.3390/ma13143240





