Research on the Preparation and Properties of High Belite Sulphoaluminate Cement (HBSAC) Based on Various Industrial Solid Wastes
Abstract
1. Introduction
2. Experimental Details
2.1. Raw Materials
2.2. Mix Design
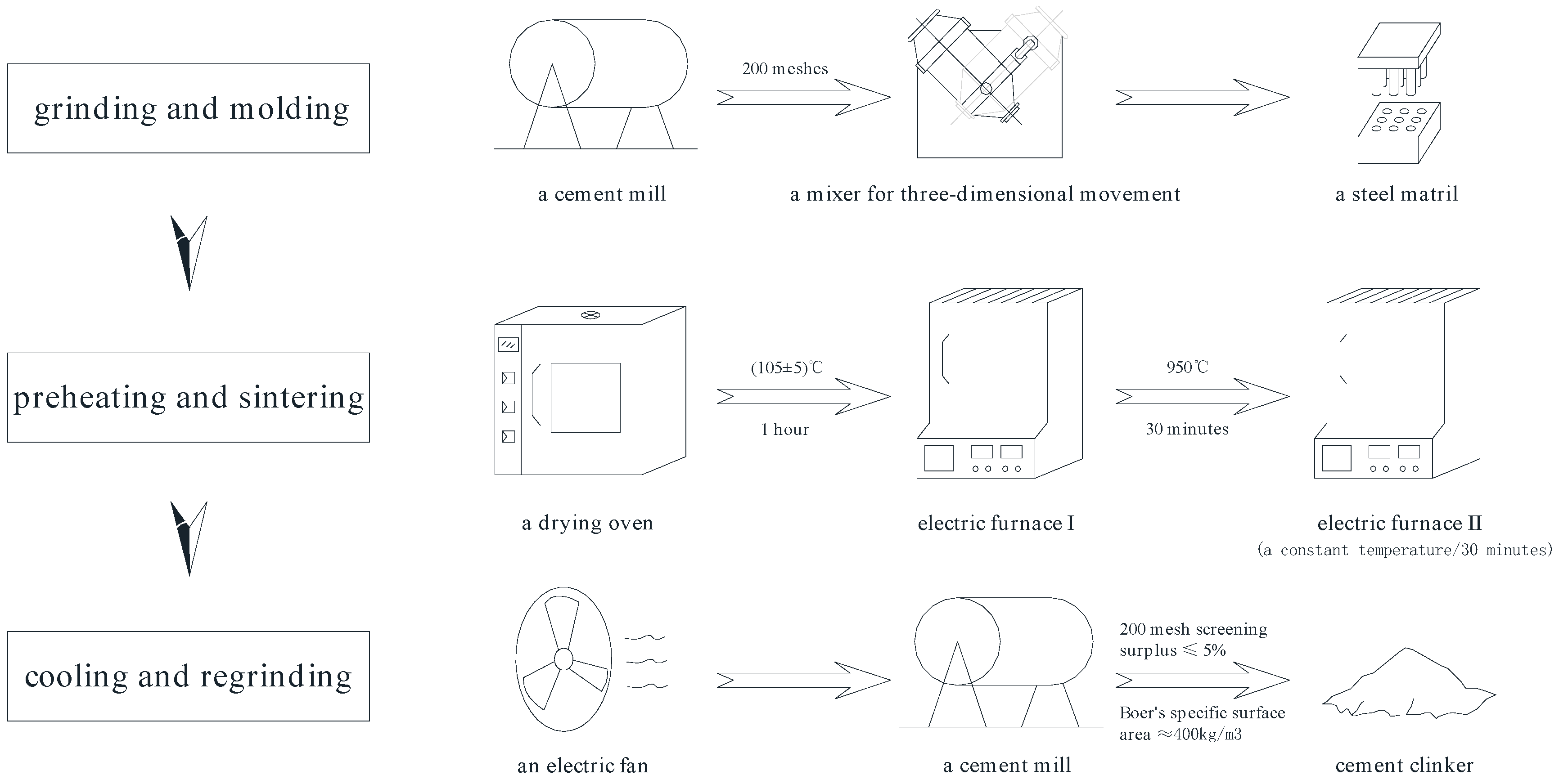
2.3. Preparation of Clinkers
2.4. Test Methods
3. Results and Discussion
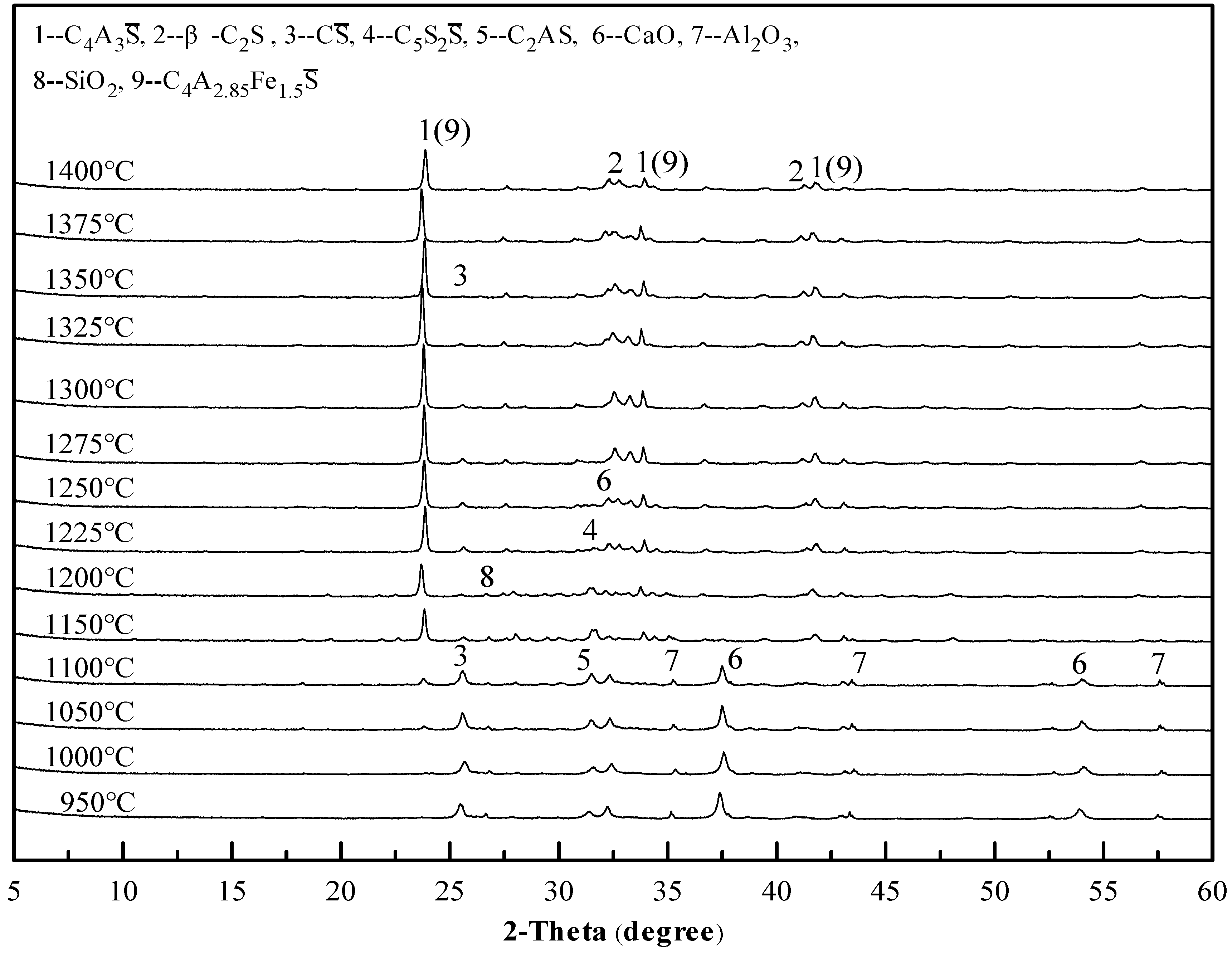
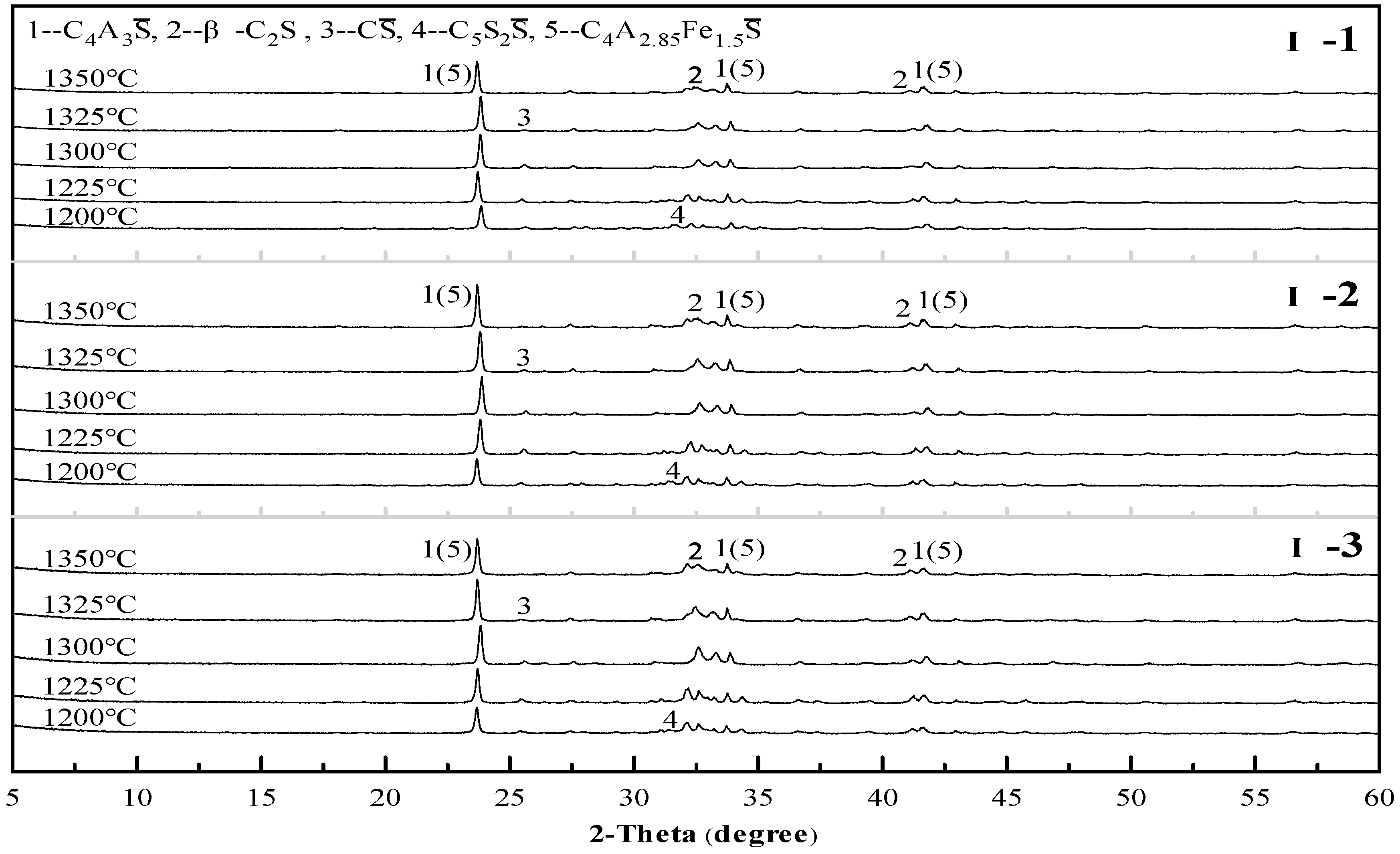
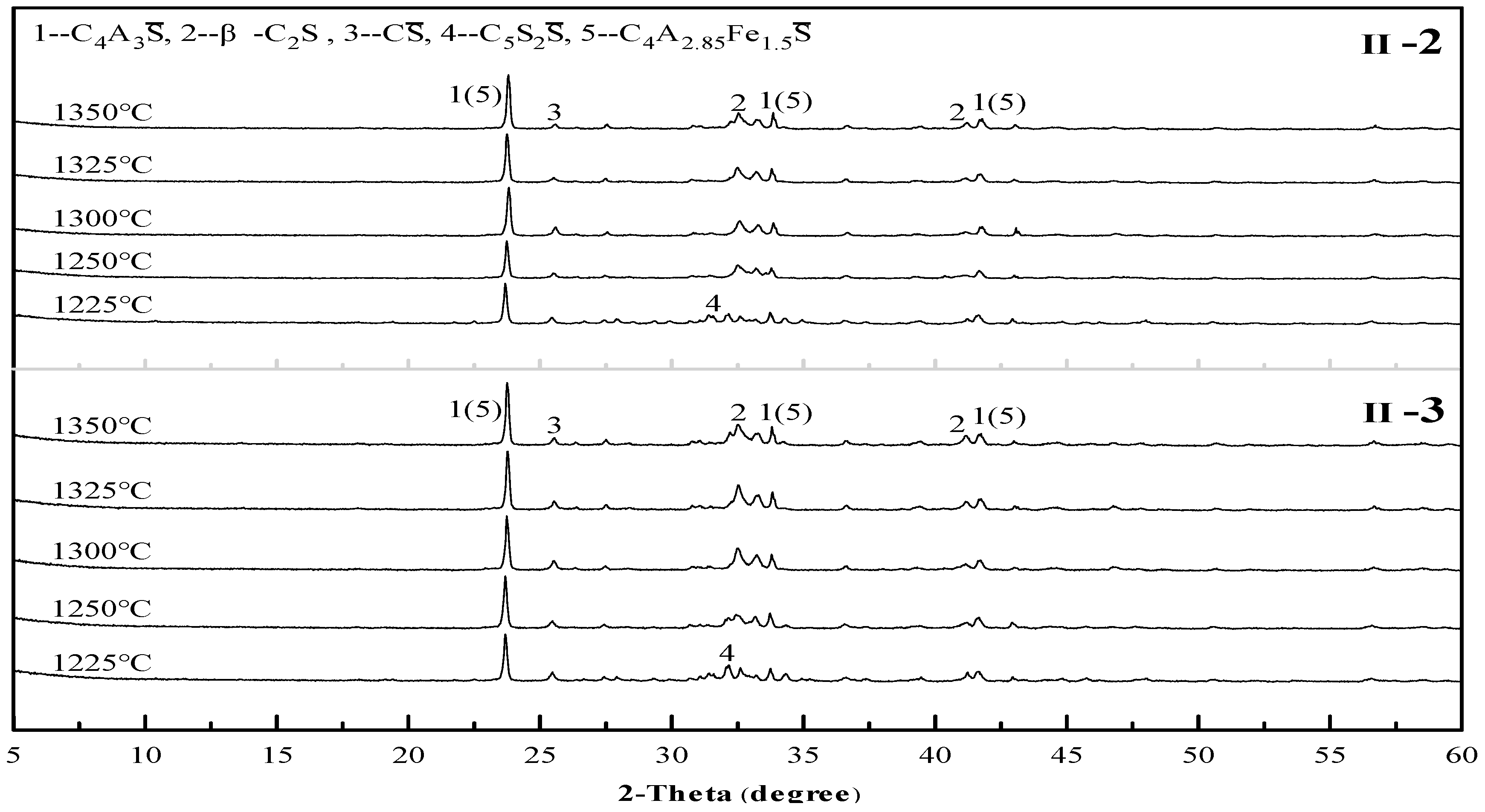
3.1. Sintering Temperature of Clinkers
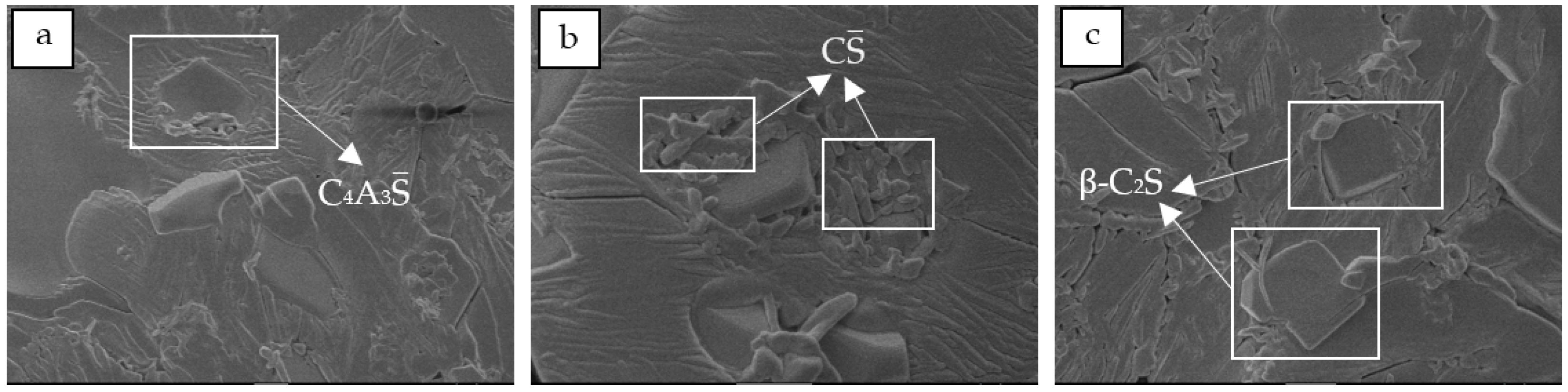
3.2. Mineral Composition of Clinkers
3.3. Physical and Mechanical Properties of the Clinkers
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SAC | sulphoaluminate cement |
| HBSAC | high belite sulphoaluminate cement |
| C4A3S | 3CaO·3Al2O3·CaSO4 |
| β-C2S | 2CaO·SiO2 |
| CS | CaSO4 |
| C2AS | 2CaO·Al2O3·SiO2 |
| C5S2S | 4CaO·2SiO2·CaSO4 |
| C4AF | 4CaO·Al2O3·Fe2O3 |
| C4A2.85Fe1.5S | 3CaO·2.85Al2O3·1.5Fe2O3·CaSO4 |
| AFtAFm | 3CaO·Al2O3·3CaSO4·32H2O3CaO·Al2O3·CaSO4·12H2O |
References
- Wang, Y.M.; Su, M.Z.; Zhang, L. Sulphoaluminate Cement, 1st ed.; Press of Beijing University of Technology: Beijing, China, 1999. (In Chinese) [Google Scholar]
- Coppola, L.; Coffetti, D.; Crotti, E.; Pastore, T. CSA-based Portland-free binders to manufacture sustainable concretes for jointless slabs on ground. Constr. Build. Mater. 2018, 187, 691–698. [Google Scholar] [CrossRef]
- Coppola, L.; Coffetti, D.; Crotti, E. Use of tartaric acid for the production of sustainable Portland-free CSA-based mortars. Constr. Build. Mater. 2018, 171, 243–249. [Google Scholar] [CrossRef]
- Hargis, C.W.; Kirchheim, A.P.; Monteiro, P.J.M.; Gartner, E.M. Early age hydration of calcium sulfoaluminate (synthetic ye’elimite, C4A3S) in the presence of gypsum and varying amounts of calcium hydroxide. Cem. Concr. Res. 2013, 48, 105–115. [Google Scholar] [CrossRef]
- Ali, M.; Gopal, S.; Handoo, S. Studies on the formation kinetics of calcium sulphoaluminate. Cem. Concr. Res. 1994, 24, 715–720. [Google Scholar] [CrossRef]
- Coppola, L.; Coffetti, D.; Crotti, E.; Gazzaniga, G.; Pastore, T. An Empathetic Added Sustainability Index (EASI) for cementitious based construction materials. J. Clean. Prod. 2019, 220, 475–482. [Google Scholar] [CrossRef]
- Ftikos, C.; Philippou, T. Preparation and hydration study of rich C2S cements. Cem. Concr. Res. 1990, 20, 934–940. [Google Scholar] [CrossRef]
- Pelletier-Chaignat, L.; Winnefeld, F.; Lothenbach, B.; Müller, C.J. Beneficial use of limestone filler with calcium sulphoaluminate cement. Constr. Build. Mater. 2012, 26, 619–627. [Google Scholar] [CrossRef]
- Li, J.; Zhou, C.Y.; Yang, Y.J. Optimization of mineral composition of high belite-sulphoaluminate clinker. J. Chin. Ceram. Soc. 2012, 40, 1618–2012. (In Chinese) [Google Scholar]
- El-Alfi, E.; Gado, R.; Gado, R. Preparation of calcium sulfoaluminate-belite cement from marble sludge waste. Constr. Build. Mater. 2016, 113, 764–772. [Google Scholar] [CrossRef]
- Xue, P.; Xu, A.; He, D.; Yang, Q.; Liu, G.; Engström, F.; Björkman, B. Research on the sintering process and characteristics of belite sulphoaluminate cement produced by BOF slag. Constr. Build. Mater. 2016, 122, 567–576. [Google Scholar] [CrossRef]
- Viggh, E.; Menad, N.; Björkman, B.; Adolfsson, D. Steelmaking slags as raw material for sulphoaluminate belite cement. Adv. Cem. Res. 2007, 19, 147–156. [Google Scholar]
- Sahu, S.; Majling, J. Preparation of sulphoaluminate belite cement from fly ash. Cem. Concr. Res. 1994, 24, 1065–1072. [Google Scholar] [CrossRef]
- Xu, G.L. Research of utilizing coal gangue in Shi Zuishan district to produce series of sulphoaluminate cements. Master’s Thesis, Chengdu University of Technology, Chengdu, China, 2009. (In Chinese). [Google Scholar]
- Zhang, J.S.; Sui, Z.T.; Shen, Y.M.; Hui, Z.F.; An, H.Y.; Gao, F. Research of producing high silicon blite sulfur aluminate cement by titanium tailings. Iron Steel Van. Tit. 2004, 25, 41–47. [Google Scholar]
- Huang, Y.B.; Qian, J.S.; Kang, X.J.; Yu, J.C.; Fan, Y.R.; Dang, Y.D. Belite-calcium sulfoaluminate cement prepared with phosphogypsum: Influence of P2O5 and F on the clinker formation and cement performances. Constr. Build. Mater. 2019, 203, 432–442. [Google Scholar] [CrossRef]
- Li, F.W. Research of preparing sulphate aluminium cement using calcium and aluminum residue and low grade bauxite. Master’s Thesis, Nanchang University, Nanchang, China, 2012. (In Chinese). [Google Scholar]
- Wang, X.; Wang, W.; Zhu, J.; Wang, P.; Ma, C. Experimental Investigation and Modeling of Sulfoaluminate Cement Preparation Using Desulfurization Gypsum and Red Mud. Ind. Eng. Chem. 2013, 52, 1261–1266. [Google Scholar] [CrossRef]
- Senff, L.; Castela, A.; Hajjaji, W.; Hotza, D.; Labrincha, J. Formulations of sulfobelite cement through design of experiments. Constr. Mater. 2011, 25, 3410–3416. [Google Scholar] [CrossRef]
- Zhang, Z.Q. “Fast-setting and quick-hardening belite sulphoaluminate cement clinker, its application and production process”. Chinese Patent No. 201410416928.8, 25 August 2015. (In Chinese). [Google Scholar]
- Li, H.; Agrawal, D.K.; Cheng, J.; Silsbee, M.R. Microwave sintering of sulphoaluminate cement with utility wastes. Cem. Concr. Res. 2001, 31, 1257–1261. [Google Scholar] [CrossRef]
- Rungchet, A.; Chindaprasirt, P.; Wansom, S.; Pimraksa, K. Hydrothermal synthesis of calcium sulfoaluminate–belite cement from industrial waste materials. J. Clean. Prod. 2016, 115, 273–283. [Google Scholar] [CrossRef]
- Wang, F.C. The properties and application of petroleum coke desulfuration residues. Master’s Thesis, Qingdao University of Technology, Qingdao, China, 2012. (In Chinese). [Google Scholar]
- Popescu, C.; Muntean, M.; Sharp, J. Industrial trial production of low energy belite cement. Cem. Concr. Compos. 2003, 25, 689–693. [Google Scholar] [CrossRef]
- Bullerjahn, F.; Schmitt, D.; Ben Haha, M. Effect of raw mix design and of clinkering process on the formation and mineralogical composition of (ternesite) belite calcium sulphoaluminate ferrite clinker. Cem. Concr. Res. 2014, 59, 87–95. [Google Scholar] [CrossRef]
- ROY, D.M.; Silsbee, M.R.; Xie, Z.H. Influences of Surplus SO3 in FBC Ash on Formation of Belite-Rich Sulfoaluminate Clinker, 3rd ed.; IAUS: Lexington, KY, USA, 1999. [Google Scholar]
- Bogue, R.H. Calculation of the Compounds in Portland Cement. Ind. Eng. Chem. Anal. Ed. 1929, 1, 192–197. [Google Scholar] [CrossRef]
- Mehta, P.K. Investigations on energy-saving cements. World Cem. Technol. 1980, 11, 166–177. [Google Scholar]
- Majling, J.; Sahu, S.; Vlna, M.; Roy, D.M. Relationship between raw mixture and mineralogical composition of sulfoaluminate belite clinkers in the system CaO-SiO2-Al2O3-Fe2O3-SO3. Cem. Concr. Res. 1993, 23, 1351–1356. [Google Scholar] [CrossRef]
- Sharp, J.H.; Lawrence, C.D.; Yang, R. Calcium sulfoaluminate cements-low energy cements, special cements or what? Adv. Cem. Res. 1999, 11, 3–13. [Google Scholar] [CrossRef]
- Chen, I.A.; Juenger, M.C. Incorporation of coal combustion residuals into calcium sulfoaluminate-belite cement clinkers. Cem. Concr. Compos. 2012, 34, 893–902. [Google Scholar] [CrossRef]
- Glasser, F.; Zhang, L. High-performance cement matrices based on calcium sulfoaluminate–belite compositions. Cem. Concr. Res. 2001, 31, 1881–1886. [Google Scholar] [CrossRef]
- Janotka, I.; Krajci, L.; Mojumdar, S.C. Performance of sulphoaluminate-belite cement with high C4A3S content. Ceram-Silikáty 2007, 51, 74. [Google Scholar]
- Álvarez-Pinazo, G.; Santacruz, I.; Aranda, M.A.G.; De La Torre, A.G.; León-Reina, L. Hydration Reactions and Mechanical Strength Developments of Iron-Rich Sulfobelite Eco-cements. Ind. Eng. Chem. 2013, 52, 16606–16614. [Google Scholar] [CrossRef]
- Berger, S.; Coumes, C.C.D.; Le Bescop, P.; Damidot, D. Influence of a thermal cycle at early age on the hydration of calcium sulphoaluminate cements with variable gypsum contents. Cem. Concr. Res. 2011, 41, 149–160. [Google Scholar] [CrossRef]
- Chen, I.A.; Juenger, M.C.G. Synthesis and hydration of calcium sulfoaluminate belit cements with varied phase compositions. J. Mater. Sci. 2011, 46, 2568–2577. [Google Scholar] [CrossRef]
- Rungchet, A.; Poon, C.; Chindaprasirt, P.; Pimraksa, K. Synthesis of low-temperature calcium sulfoaluminate-belite cements from industrial wastes and their hydration: Comparative studies between lignite fly ash and bottom ash. Cem. Concr. Compos. 2017, 83, 10–19. [Google Scholar] [CrossRef]
- Adolfsson, D.; Menad, N.; Viggh, E.; Björkman, B. Hydraulic properties of sulphoaluminate belite cement based on steelmaking slags. Adv. Cem. Res. 2007, 19, 133–138. [Google Scholar] [CrossRef]
- Sahu, S.; Havlica, J.; Tomkova, V.; Majling, J. Hydration behaviour of sulphoaluminate belite cement in the presence of various calcium sulphates. Thermochim. Acta 1991, 175, 45–52. [Google Scholar] [CrossRef]
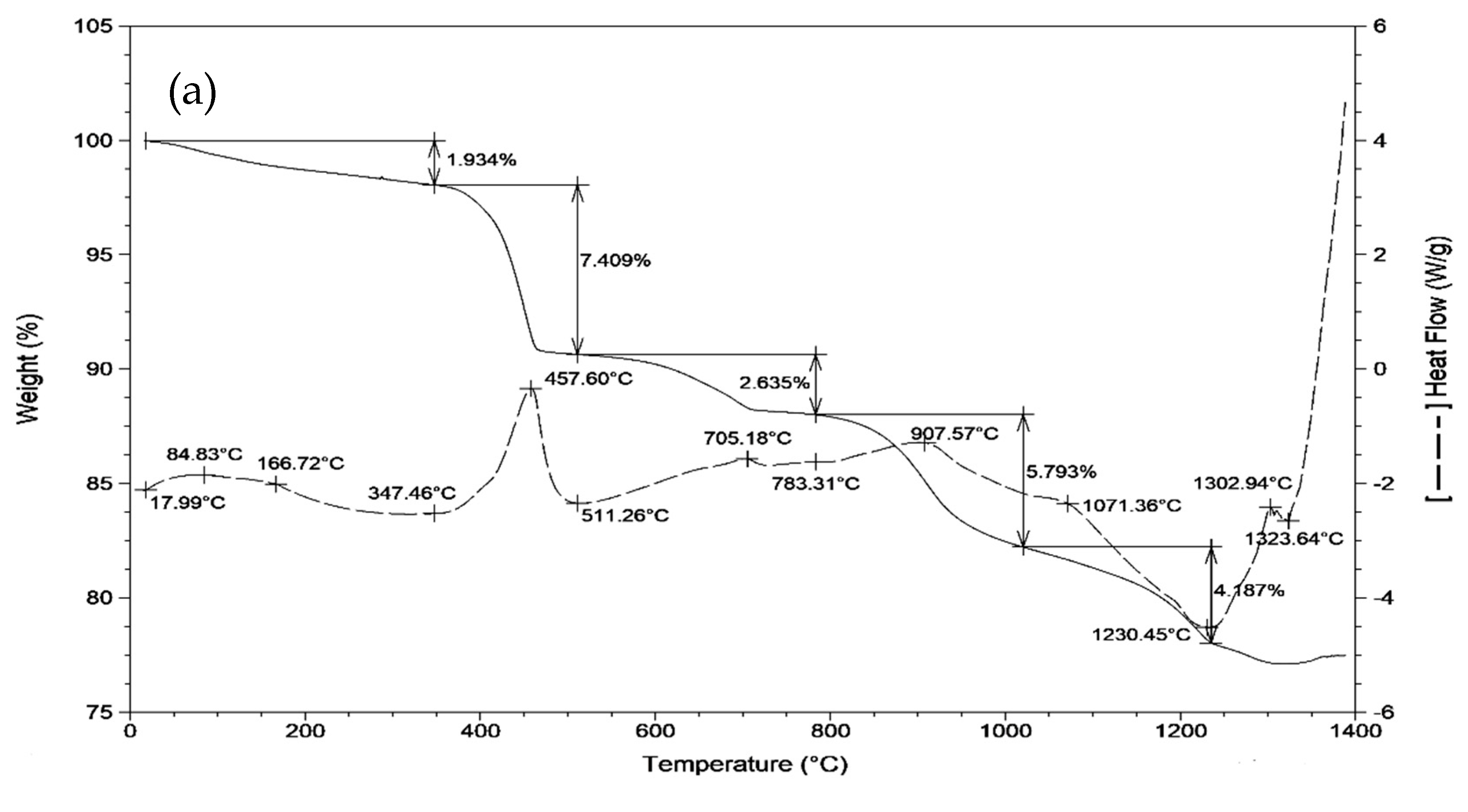

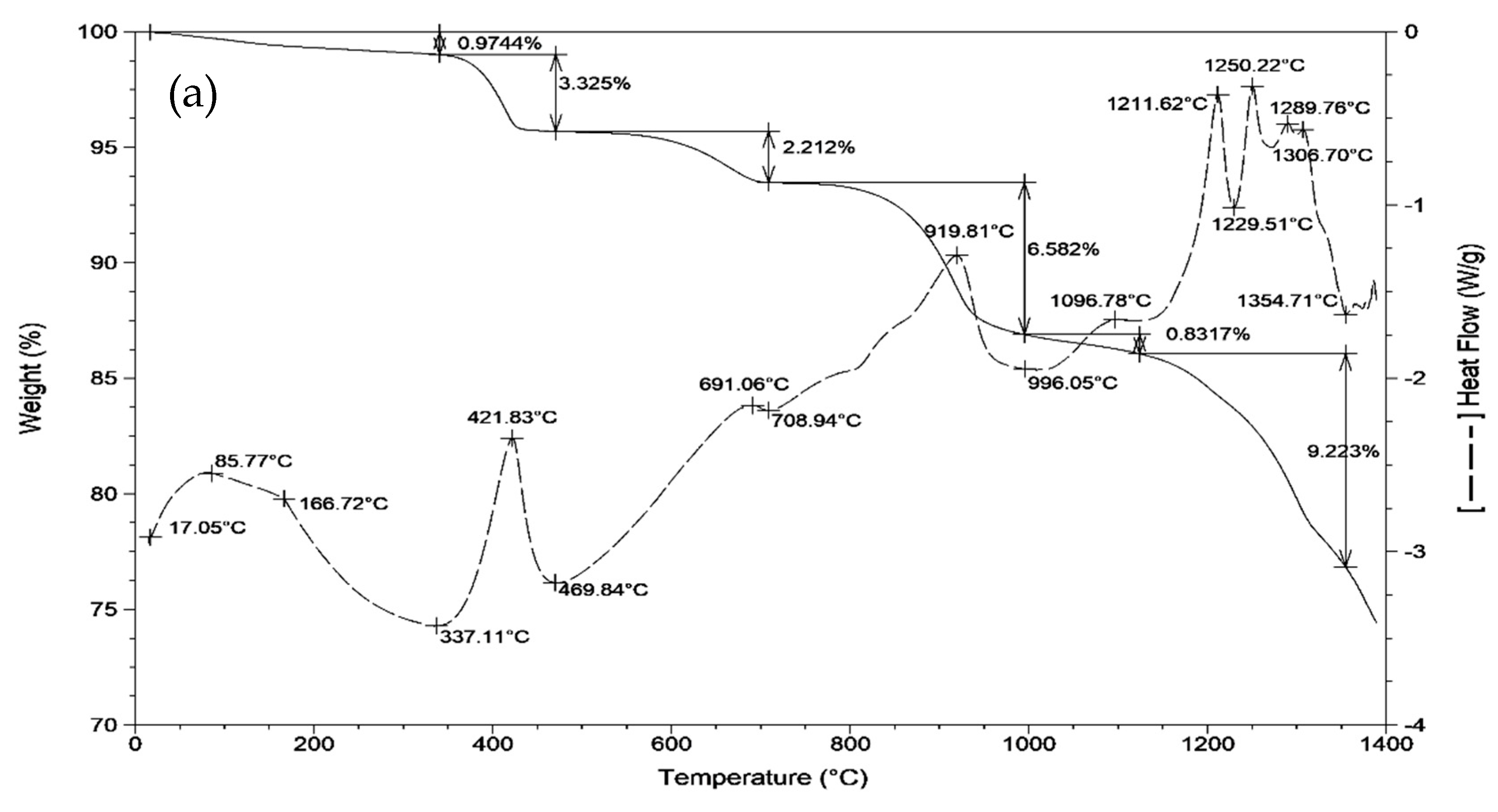
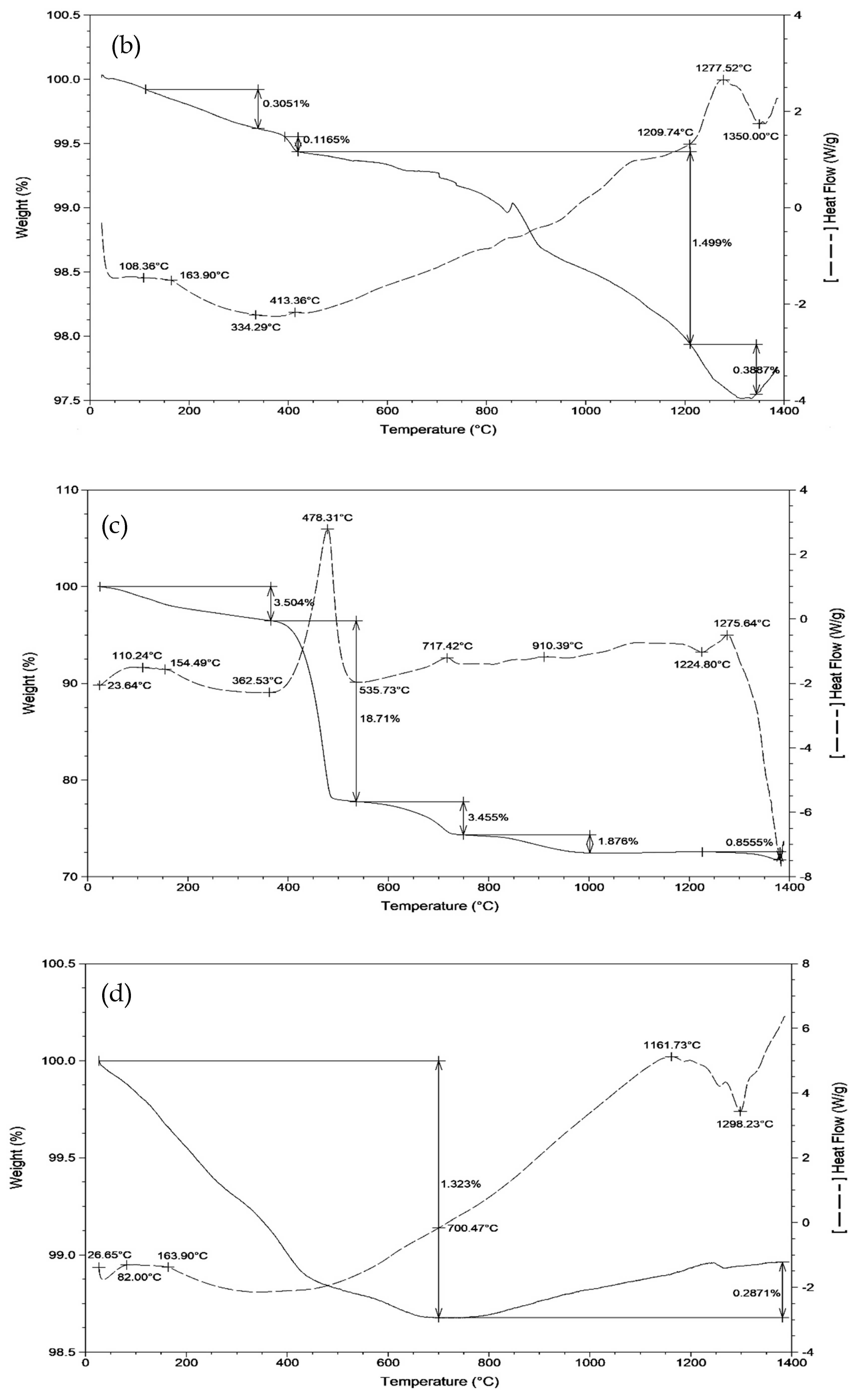



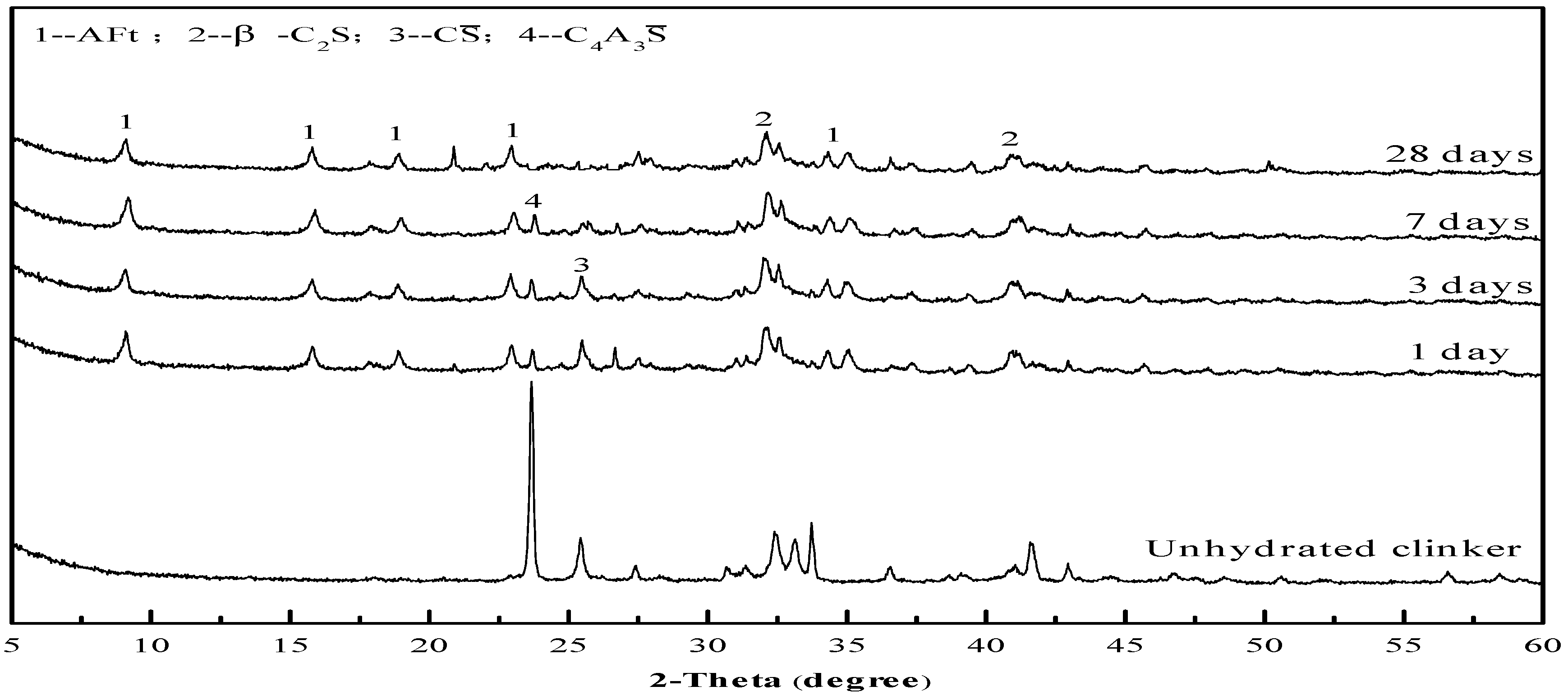
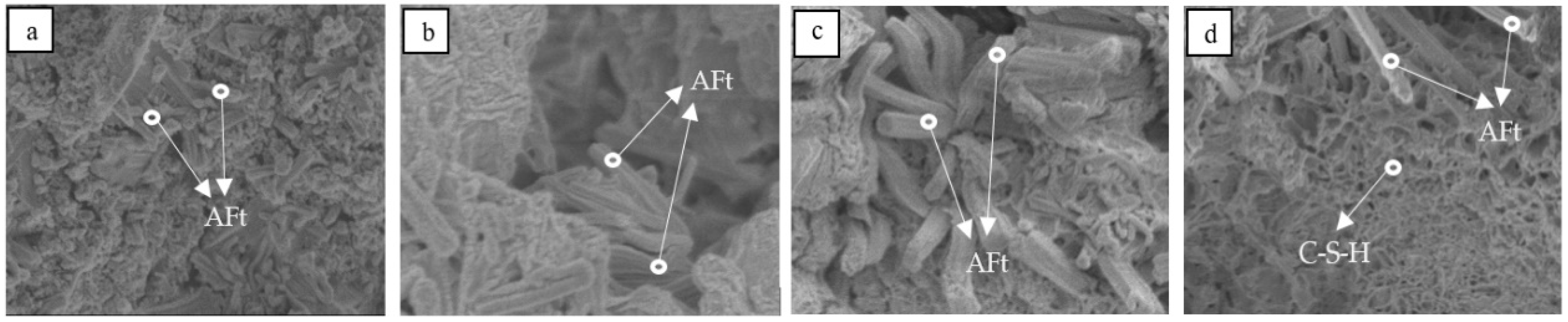
| Raw Material | CaO | Al2O3 | SiO2 | Fe2O3 | SO3 | MgO | TiO2 | LOI | ∑ |
|---|---|---|---|---|---|---|---|---|---|
| Petroleum coke desulfurization slag | 52.93 | 0.96 | 4.56 | 1.13 | 30.12 | 2.03 | 0.00 | 7.48 | 99.21 |
| Fly ash | 7.86 | 27.45 | 52.56 | 4.24 | 1.26 | 1.12 | 0.99 | 1.73 | 97.21 |
| Carbide slag | 66.02 | 1.47 | 4.61 | 0.68 | 1.97 | 0.25 | 0.00 | 24.62 | 99.62 |
| Bauxite | 0.51 | 64.07 | 14.53 | 0.88 | 0.00 | 15.38 | 2.56 | 1.03 | 98.96 |
| Series | Group | Clinker Minerals | Raw Materials | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C4AF | C4A3S | C2S | CS | Petroleum Coke Desulfurization Slag | Fly Ash | Carbide Slag | Bauxite | ||
| I | 1 | 5 | 40 | 45 | 10 | 31.8 | 12.0 | 35.1 | 21.1 |
| 2 | 5 | 35 | 50 | 10 | 29.0 | 16.0 | 38.6 | 16.4 | |
| 3 | 5 | 30 | 55 | 10 | 27.9 | 19.3 | 40.7 | 12.1 | |
| II | 1 | 5 | 40 | 40 | 15 | 39.2 | 10.8 | 29.2 | 20.8 |
| 2 | 5 | 35 | 45 | 15 | 37.5 | 14.3 | 32.0 | 16.2 | |
| 3 | 5 | 30 | 50 | 15 | 35.2 | 18.4 | 35.2 | 11.2 | |
| III | 1 | 5 | 35 | 40 | 20 | 47.2 | 9.9 | 23.2 | 19.7 |
| 2 | 5 | 30 | 45 | 20 | 45.0 | 13.8 | 26.4 | 14.8 | |
| 3 | 5 | 25 | 50 | 20 | 42.5 | 17.9 | 29.8 | 9.8 | |
| Series | Group | CaO | Al2O3 | Fe2O3 | SiO2 | SO3 | MgO | TiO2 | ∑ |
|---|---|---|---|---|---|---|---|---|---|
| I | 1 | 45.76 | 20.36 | 2.36 | 13.95 | 10.55 | 3.20 | 0.73 | 96.91 |
| 2 | 47.23 | 18.09 | 2.44 | 15.46 | 9.86 | 2.42 | 0.64 | 96.14 | |
| 3 | 48.65 | 15.97 | 2.57 | 17.08 | 9.29 | 1.71 | 0.55 | 95.82 | |
| II | 1 | 45.34 | 20.78 | 2.32 | 12.56 | 13.67 | 3.04 | 0.68 | 98.39 |
| 2 | 46.75 | 18.24 | 2.47 | 14.13 | 12.93 | 2.30 | 0.60 | 97.42 | |
| 3 | 48.04 | 16.02 | 2.63 | 16.07 | 12.27 | 1.47 | 0.50 | 97.00 | |
| III | 1 | 45.83 | 18.25 | 2.15 | 12.63 | 15.88 | 3.00 | 0.77 | 98.51 |
| 2 | 47.26 | 16.13 | 2.29 | 14.23 | 15.39 | 2.20 | 0.68 | 98.18 | |
| 3 | 48.75 | 13.88 | 2.45 | 15.64 | 14.65 | 1.37 | 0.58 | 97.32 |
| Series | Group | C4A3S | β-C2S | C4A2.85Fe1.5S | CS | ∑ | Cm |
|---|---|---|---|---|---|---|---|
| I | 1 | 34.83 | 40.04 | 8.21 | 8.93 | 92.01 | 1.01 |
| 2 | 30.12 | 44.37 | 8.49 | 8.75 | 91.73 | 1.02 | |
| 3 | 25.59 | 49.02 | 8.94 | 8.71 | 92.26 | 1.02 | |
| II | 1 | 35.77 | 36.05 | 8.07 | 14.05 | 93.94 | 1.01 |
| 2 | 30.35 | 40.55 | 8.60 | 13.91 | 93.41 | 1.02 | |
| 3 | 25.55 | 46.12 | 9.15 | 13.76 | 94.58 | 1.00 | |
| III | 1 | 31.14 | 36.25 | 7.48 | 18.92 | 93.79 | 1.01 |
| 2 | 26.58 | 40.84 | 7.97 | 19.02 | 94.41 | 1.01 | |
| 3 | 21.72 | 44.89 | 8.53 | 18.75 | 93.89 | 1.03 |
| Series | Group | Water Requirement of Normal Consistency (wt.%) | Initial Setting Time (min) | Final Setting Time (min) |
|---|---|---|---|---|
| I | 1 | 39.5 | 21.80 | 35.95 |
| 2 | 39.0 | 23.17 | 37.75 | |
| 3 | 38.5 | 24.50 | 39.53 | |
| II | 1 | 37.5 | 20.10 | 30.60 |
| 2 | 37.0 | 21.08 | 31.58 | |
| 3 | 36.5 | 22.75 | 32.08 | |
| III | 1 | 37.0 | 17.05 | 23.45 |
| 2 | 36.5 | 18.18 | 26.42 | |
| 3 | 36.0 | 19.38 | 28.20 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, D.; Yue, G.; Li, Q.; Guo, Y.; Gao, S.; Wang, L. Research on the Preparation and Properties of High Belite Sulphoaluminate Cement (HBSAC) Based on Various Industrial Solid Wastes. Materials 2019, 12, 1510. https://doi.org/10.3390/ma12091510
Su D, Yue G, Li Q, Guo Y, Gao S, Wang L. Research on the Preparation and Properties of High Belite Sulphoaluminate Cement (HBSAC) Based on Various Industrial Solid Wastes. Materials. 2019; 12(9):1510. https://doi.org/10.3390/ma12091510
Chicago/Turabian StyleSu, Dunlei, Gongbing Yue, Qiuyi Li, Yuanxin Guo, Song Gao, and Liang Wang. 2019. "Research on the Preparation and Properties of High Belite Sulphoaluminate Cement (HBSAC) Based on Various Industrial Solid Wastes" Materials 12, no. 9: 1510. https://doi.org/10.3390/ma12091510
APA StyleSu, D., Yue, G., Li, Q., Guo, Y., Gao, S., & Wang, L. (2019). Research on the Preparation and Properties of High Belite Sulphoaluminate Cement (HBSAC) Based on Various Industrial Solid Wastes. Materials, 12(9), 1510. https://doi.org/10.3390/ma12091510




