312 MAX Phases: Elastic Properties and Lithiation
Abstract
1. Introduction
2. Computational Methods
3. Results
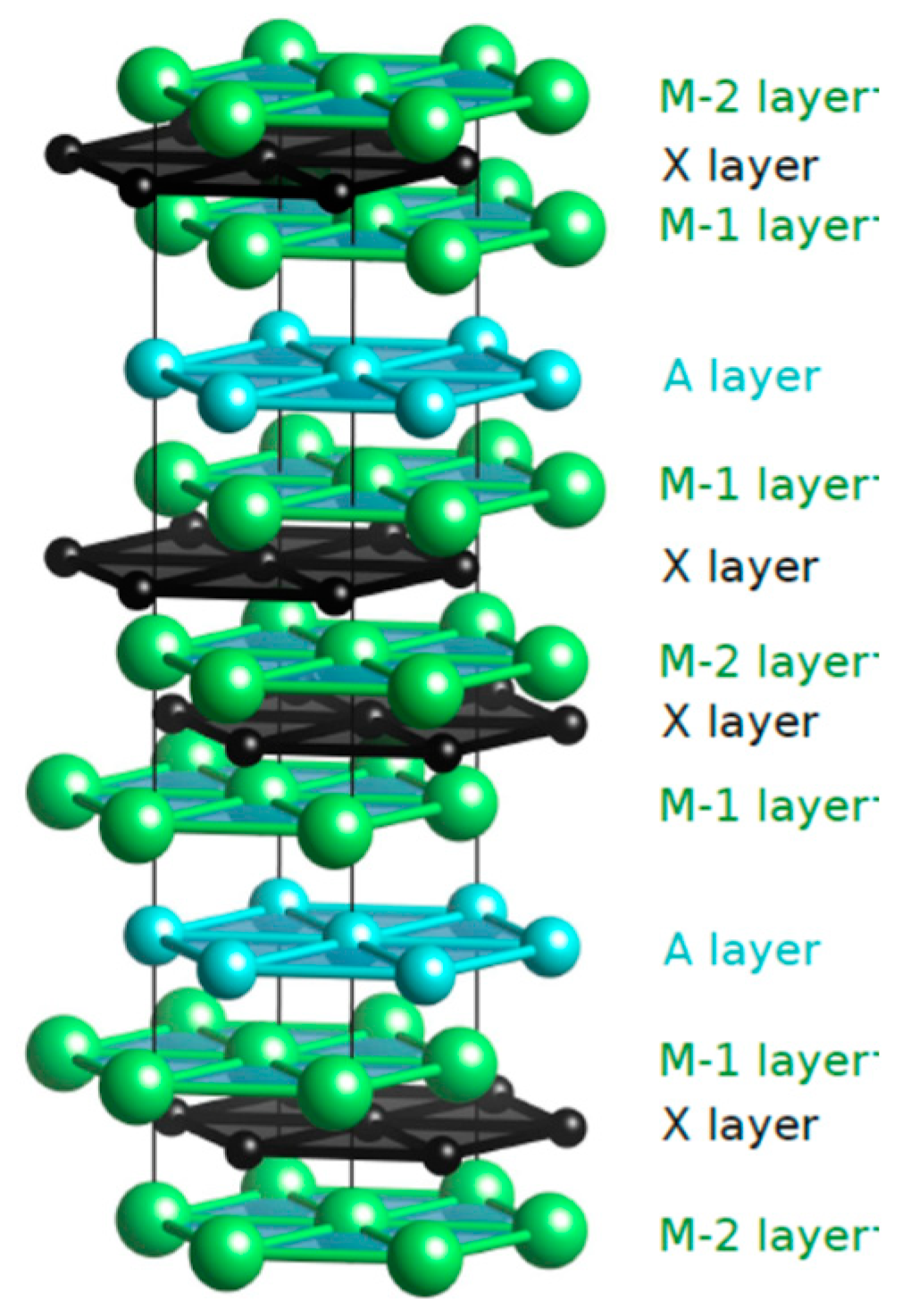
3.1. Elastic Properties
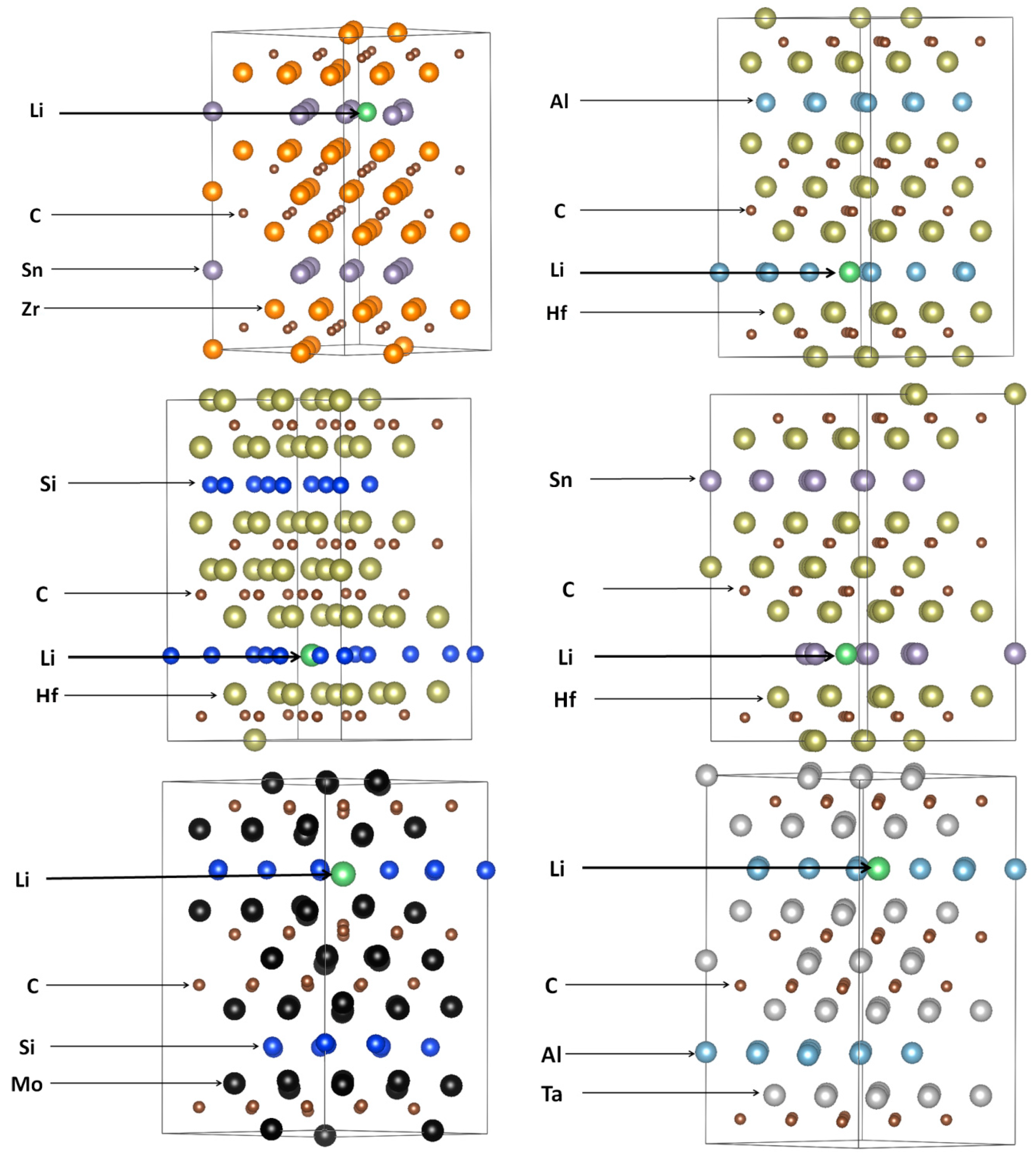
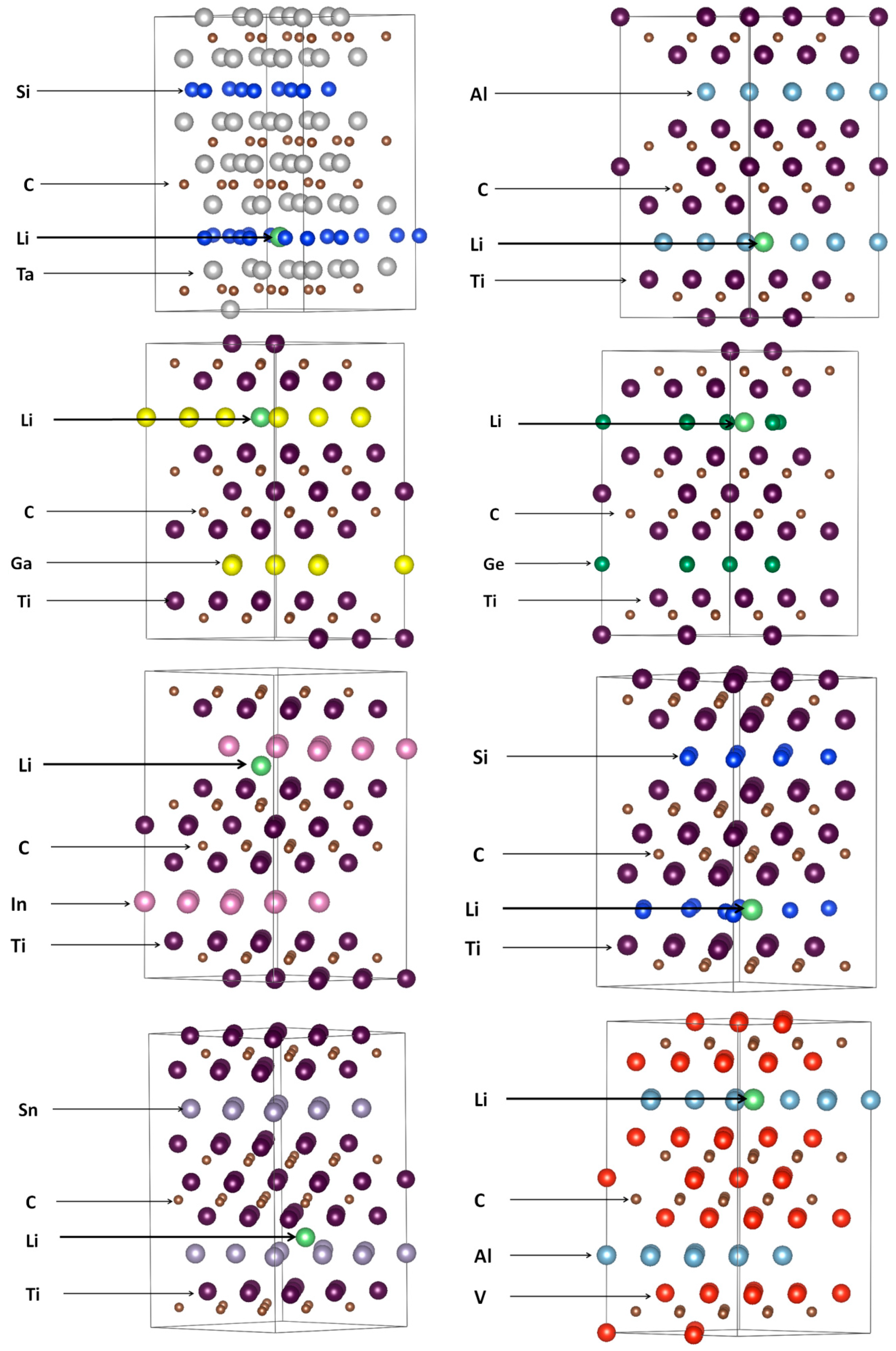
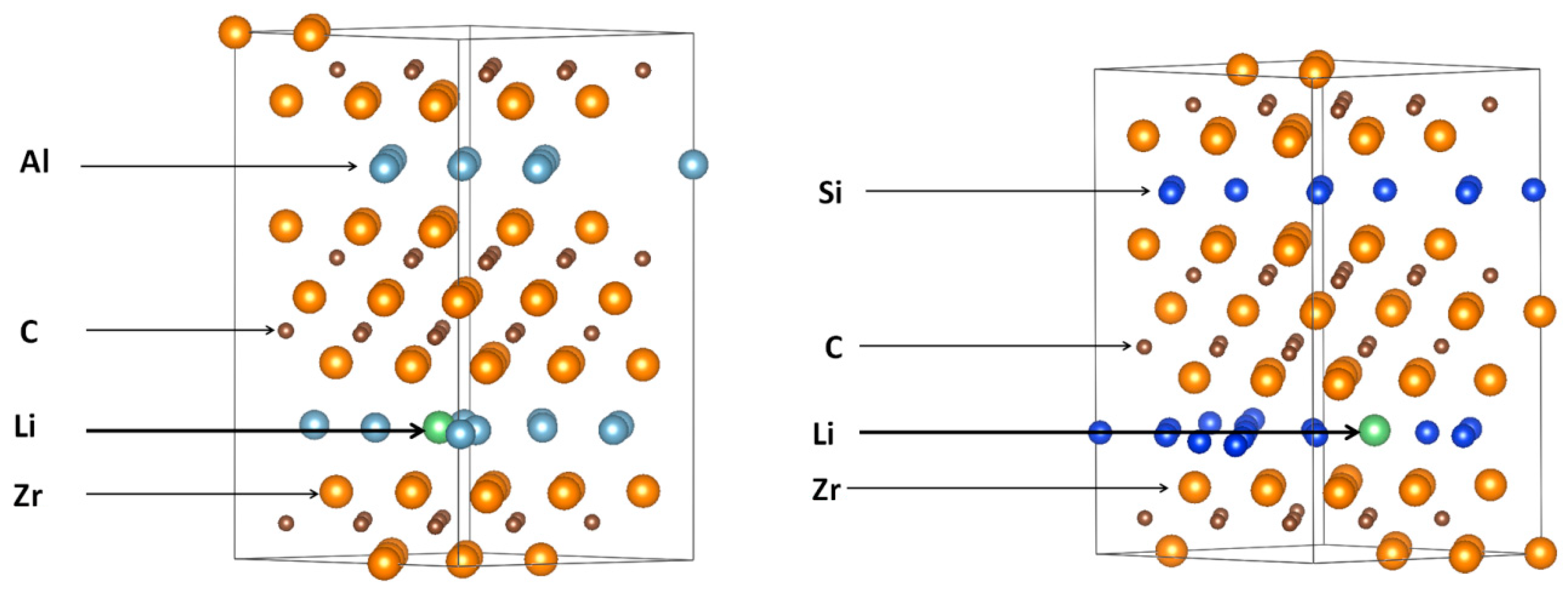
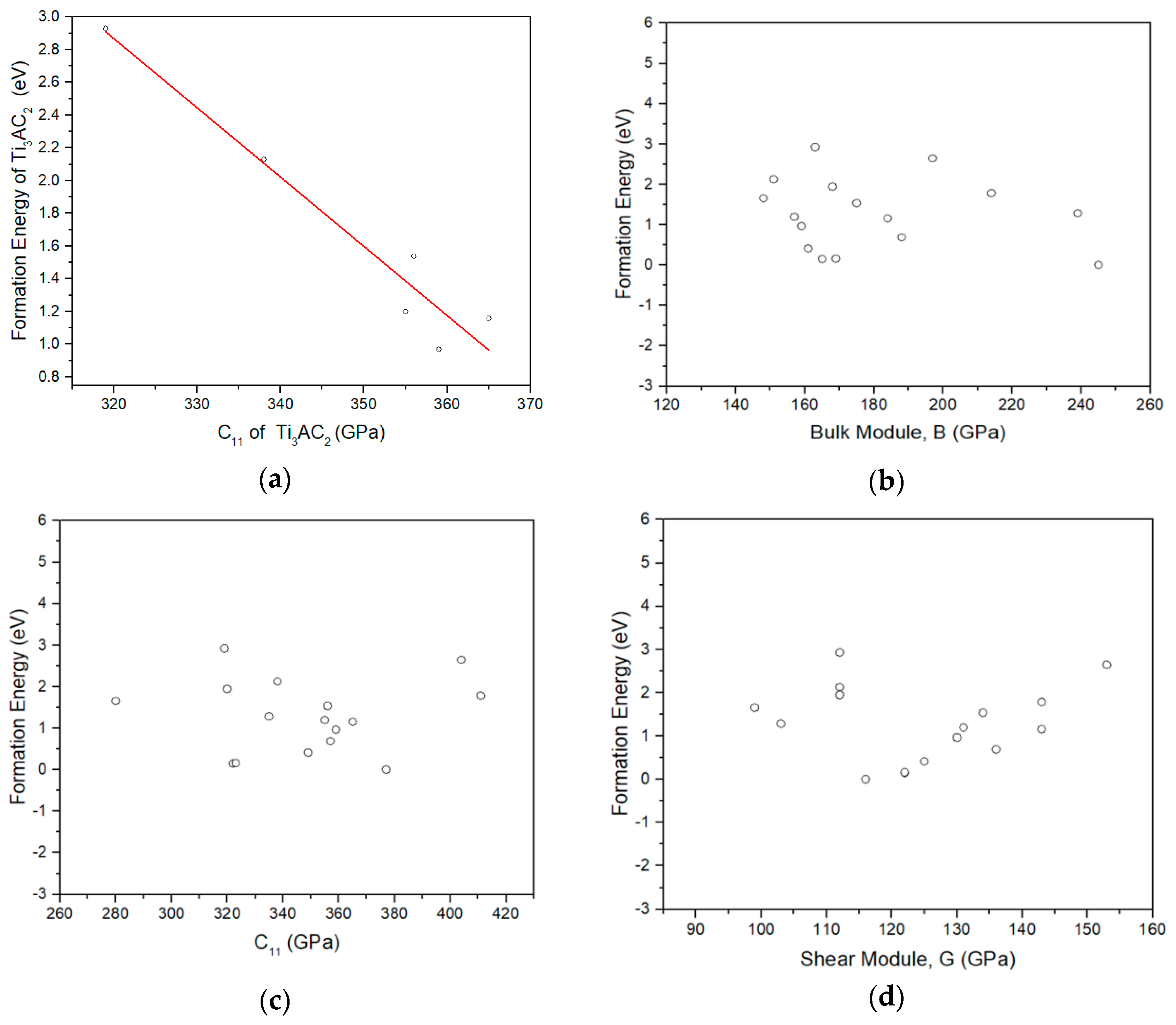
3.2. Lithiation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barsoum, M.W.; El-Raghy, T. The MAX phases: Unique new carbide and nitride materials: Ternary ceramics turn out to be surprisingly soft and machinable, yet also heat-tolerant, strong and lightweight. Am. Sci. 2001, 89, 334–343. [Google Scholar] [CrossRef]
- Barsoum, M.W. The MN+1AXN phases: A new class of solids: Thermodynamically stable nanolaminates. Prog. Solid State Chem. 2000, 28, 201–281. [Google Scholar] [CrossRef]
- Chen, H.; Yang, D.; Zhang, Q.; Jin, S.; Guo, L.; Deng, J.; Chen, X. A series of MAX phases with MA-triangular-prism bilayers and elastic properties. Angew. Chem. 2019, 131, 4624–4628. [Google Scholar] [CrossRef]
- Thore, A.; Rosén, J. An investigation of the in-plane chemically ordered atomic laminates (Mo2/3 Sc 1/3)2 AlC and (Mo2/3 Y1/3)2 AlC from first principles. Phys. Chem. Chem. Phys. 2017, 19, 21595–21603. [Google Scholar] [CrossRef]
- Nowotny, H. Strukturchemieeinigerverbindungen der übergangsmetallemit den elementen C, Si, Ge, Sn. Prog. Solid State Chem. 1971, 5, 27–70. [Google Scholar] [CrossRef]
- Barsoum, M.W.; El-Raghy, T. Synthesis and characterization of a remarkable ceramic. J. Am. Ceram. Soc. 1996, 79, 1953. [Google Scholar] [CrossRef]
- Khazaei, M.; Ranjbar, A.; Esfarjani, K.; Bogdanovski, D.; Dronskowski, R.; Yunoki, S. Insights into exfoliation possibility of MAX phases to MXenes. Phys. Chem. Chem. Phys. 2018, 20, 8579–8592. [Google Scholar] [CrossRef]
- Barsoum, M.W. MAX Phases: Properties of Machinable Ternary Carbides and Nitrides; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Eklund, P.; Rosen, J.; Persson, P.O.Å. Layered ternary Mn+1AXn phases and their 2D derivative MXene: An overview from a thin-film perspective. J. Phys. D Appl. Phys. 2017, 50, 113001. [Google Scholar] [CrossRef]
- Buschow, K.J.; Cahn, R.W.; Flemings, M.C.; Ilschner, B.; Kramer, E.J.; Mahajan, S. Encyclopedia of Materials Science and Technology; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Sun, Z.M. Progress in research and development on MAX phases: A family of layered ternary compounds. Int. Mater. Rev. 2011, 56, 143–166. [Google Scholar] [CrossRef]
- Wang, X.H.; Zhou, Y.C. Layered machinable and electrically conductive Ti2AlC and Ti3AlC2 ceramics: A review. J. Mater. Sci. Tehnol. 2011, 26, 385–416. [Google Scholar] [CrossRef]
- Middleburgh, S.C.; Lumpkin, G.R.; Riley, D. Accommodation, accumulation, and migration of defects in Ti3SiC2 and Ti3AlC2 MAX phases. J. Am. Ceram. Soc. 2013, 96, 3196–3201. [Google Scholar]
- Sokol, M.; Natu, V.; Kota, S.; Barsoum, M.W. On the chemical diversity of the MAX phases. Trends Chem. 2019, 1, 210. [Google Scholar] [CrossRef]
- Horlait, D.; Grasso, S.; Chroneos, A.; Lee, W.E. Attempts to synthesise quaternary MAX phases (Zr,M)2AlC and Zr2(Al,A)C as a way to approach Zr2AlC. Mater. Res. Lett. 2016, 4, 137–144. [Google Scholar] [CrossRef]
- Horlait, D.; Grasso, S.; Al Nasiri, N.; Burr, P.A.; Lee, W.E. Synthesis and high-temperature oxidation of MAX phases in the Cr-Ti-Al-C quaternary system. J. Am. Ceram. Soc. 2016, 99, 682–690. [Google Scholar] [CrossRef]
- Saltas, V.; Horlait, D.; Sgourou, E.N.; Vallianatos, F.; Chroneos, A. Modelling solid solutions with cluster expansion, special quasirandom structures, and thermodynamic approaches. Appl. Phys. Rev. 2017, 4, 041301. [Google Scholar] [CrossRef]
- Horlait, D.; Middleburgh, S.C.; Chroneos, A.; Lee, W.E. Synthesis and DFT investigation of new bismuth-containing MAX phases. Sci. Rep. 2016, 6, 18829. [Google Scholar] [CrossRef]
- Lapauw, T.; Halim, J.; Lu, J.; Cabioc’h, T.; Hultman, L.; Barsoum, M.W.; Lambrinou, K.; Vleugels, J. Synthesis of the novel Zr3AlC2 MAX phase. J. Eur. Ceram. Soc. 2016, 36, 943–947. [Google Scholar] [CrossRef]
- Talapatra, A.; Duong, T.; Son, W.; Gao, H.; Radovic, M.; Arróyave, R. High-throughput combinatorial study of the effect of M site alloying on the solid solution behavior of M2AlC MAX phases. Phys. Rev. B 2016, 94, 104106. [Google Scholar] [CrossRef]
- Tunca, B.; Lapauw, T.; Karakulina, O.M.; Batuk, M.; Cabioc’h, T.; Hadermann, J.; Delville, R.; Lambrinou, K.; Vleugels, J. Synthesis of MAX phases in the Zr-Ti-Al-C system. Inorg. Chem. 2017, 56, 3489–3498. [Google Scholar] [CrossRef]
- Arróyave, R.; Talapatra, A.; Duong, T.; Son, W.; Gao, H.; Radovic, M. Does aluminium play well with others? Intrinsic Al-A alloying behavior in 211/312 MAX phases. Mater. Res. Lett. 2017, 5, 170–178. [Google Scholar]
- Hadi, M.A.; Panayiotatos, Y.; Chroneos, A. Structural and optical properties of the recently synthesized (Zr3-xTix)AlC2 MAX phases. J. Mater. Sci. Mater. Electron. 2017, 28, 3386–3393. [Google Scholar] [CrossRef]
- Lei, J.C.; Zhang, X.; Zhou, Z. Recent advances in MXene: Preparation, properties, and applications. Front. Phys. 2015, 10, 276–286. [Google Scholar] [CrossRef]
- Zapata-Solvas, E.; Hadi, M.A.; Horlait, D.; Parfitt, D.C.; Thibaud, A.; Chroneos, A.; Lee, W.E. Synthesis and physical properties of (Zr1-xTix)AlC2 MAX phases. J. Am. Ceram. Soc. 2017, 100, 3393–3401. [Google Scholar] [CrossRef]
- Kim, H.; Park, K.Y.; Hong, J.; Kang, K. All-graphene-battery: Bridging the gap between supercapacitors and lithium ion batteries. Sci. Rep. 2014, 4, 5278. [Google Scholar] [CrossRef]
- Naguib, M.; Come, J.; Dyatkin, B.; Presser, V.; Taberna, P.L.; Simon, P.; Barsoum, M.W.; Gogotsi, Y. MXene: A promising transition metal carbide anode for lithium-ion batteries. Electrochem. Commun. 2012, 16, 61–64. [Google Scholar] [CrossRef]
- Liang, X.; Garsuch, A.; Nazar, L.F. Sulfur cathodes based on conductive MXene nanosheets for high-performance lithium–sulfur batteries. Angew. Chem. 2015, 54, 3907–3911. [Google Scholar] [CrossRef]
- Khazaei, M.; Arai, M.; Sasaki, T.; Chung, C.Y.; Venkataramanan, N.S.; Estili, M.; Sakka, Y.; Kawazoe, Y. Novel electronic and magnetic properties of two-dimensional transition metal carbides and nitrides. Adv. Funct. Mater. 2013, 23, 2185–2192. [Google Scholar] [CrossRef]
- Peer Mohamed, A. Shear induced micromechanical synthesis of Ti3SiC2 MAXene nanosheets for functional applications. RSC Adv. 2015, 5, 51242–51247. [Google Scholar]
- Zhou, J.; Zha, X.; Chen, F.Y.; Ye, Q.; Eklund, P.; Du, S.; Huang, Q. A two-dimensional zirconium carbide by Selective etching of Al3C3 from nanolaminated Zr3Al3C5. Angew. Chem. 2016, 55, 5008–5013. [Google Scholar] [CrossRef]
- Zapata-Solvas, E.; Christopoulos, S.R.G.; Ni, N.; Parfitt, D.C.; Horlait, D.; Fitzpatrick, M.E.; Chroneos, A.; Lee, W.E. Experimental synthesis and density functional theory investigation of radiation tolerance of Zr3 (Al1-xS ix) C2 MAX phases. J. Am. Ceram. Soc. 2017, 100, 1377–1387. [Google Scholar] [CrossRef]
- Farle, A.M.; Van der Zwaag, S.; Sloof, W.G. A conceptual study into the potential of max-phase ceramics for self-healing of crack damage. In Proceedings of the 4th International Conference on Self-Healing Materials, ICSHM 2013, Ghent, Belgium, 16–20 June 2013. [Google Scholar]
- Lapauw, T.; Tunca, B.; Cabioc’h, T.; Lu, J.; Persson, P.O.A.; Lambrinou, K.; Vleugels, J. Synthesis of MAX phases in the Hf–Al–C system. Inorg. Chem. 2016, 55, 10922–10927. [Google Scholar] [CrossRef]
- Pshyk, A.V.; Coy, E.; Kempiński, M.; Scheibe, B.; Jurga, S. Low-temperature growth of epitaxial Ti2AlC MAX phase thin films by low-rate layer-by-layer PVD. Mater. Res. Let. 2019, 7, 244–250. [Google Scholar] [CrossRef]
- Siebert, J.P.; Bischoff, L.; Lepple, M.; Zintler, A.; Molina-Luna, L.; Wiedwald, U.; Birkel, C.S. Sol–gel based synthesis and enhanced processability of MAX phase Cr 2 GaC. J. Mater. Chem. C 2019, 7, 6034–6040. [Google Scholar] [CrossRef]
- Payne, M.C.; Teter, M.P.; Allan, D.C.; Arias, T.A.; Joannopoulos, J.D. Iterative minimization techniques for ab initio total-energy calculations: Molecular dynamics and conjugate gradients. Rev. Mod. Phys. 1992, 64, 1045. [Google Scholar] [CrossRef]
- Segall, M.D.; Lindan, P.J.; Probert, M.A.; Pickard, C.J.; Hasnip, P.J.; Clark, S.J.; Payne, M.C. First-principles simulation: Ideas, illustrations and the CASTEP code. J. Phys. Condens. Matter. 2002, 14, 2717. [Google Scholar] [CrossRef]
- Perdew, J.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef]
- Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B 1990, 41, 7892. [Google Scholar] [CrossRef]
- Monkhorst, H.J.; Pack, J.D. Special points for Brillouin-zone integrations. Phys. Rev. B 1976, 13, 5188. [Google Scholar] [CrossRef]
- Barsoum, M.W. The Mn+1AXn phases and their properties. In Ceramics Science and Technology; Riedel, R.R., Chen, I.W., Eds.; Wiley-VCH Verlag GmbH & Co: Weinheim, Germany, 2010; Volume 2. [Google Scholar]
- Born, M. On the stability of crystal lattices. In Mathematical Proceedings of the Cambridge Philosophical Society; Cambridge University Press: Cambridge, UK, 1940; Volume 36, pp. 160–172. [Google Scholar]
- Christopoulos, S.R.; Filippatos, P.P.; Hadi, M.A.; Kelaidis, N.; Fitzpatrick, M.E.; Chroneos, A. Intrinsic defect processes and elastic properties of Ti3AC2 (A= Al, Si, Ga, Ge, In, Sn) MAX phases. J. Appl. Phys. 2018, 123, 025103. [Google Scholar] [CrossRef]
- Hadi, M.A.; Christopoulos, S.R.; Naqib, S.H.; Chroneos, A.; Fitzpatrick, M.E.; Islam, A.K.M.A. Physical properties and defect processes of M3SnC2 (M = Ti, Zr, Hf) MAX phases: Effect of M-elements. J. Alloy. Comp. 2018, 748, 804–813. [Google Scholar] [CrossRef]
- Aryal, S.; Sakidja, R.; Barsoum, M.W.; Ching, W.Y. A genomic approach to the stability, elastic, and electronic properties of the MAX phases. Phys. Status Solidi B 2014, 251, 1480–1497. [Google Scholar] [CrossRef]
- Ali, M.S.; Islam, A.K.M.A.; Hossain, M.M.; Parvin, F. Phase stability, elastic, electronic, thermal and optical properties of Ti3Al1 − xSixC2 (0 ≤ × ≤ 1): First principle study. Phys. B 2012, 407, 4221–4228. [Google Scholar] [CrossRef]
- Wang, W.; Sun, L.; Yang, Y.; Dong, J.; Gu, Z.; Jin, H. Pressure effects on electronic, anisotropic elastic and thermal properties of Ti3AC2 (Si, Ge and Sn) by ab initio calculations. Results Phys. 2017, 7, 1055–1065. [Google Scholar] [CrossRef]
- Finkel, P.; Barsoum, M.W.; El-Raghy, T. Low temperature dependencies of the elastic properties of Ti4AlN3, Ti3 Al1.1 C1.8, and Ti3SiC2. J. Appl. Phys. 2000, 87, 1701–1703. [Google Scholar] [CrossRef]
- Roknuzzaman, M.; Hadi, M.A.; Ali, M.A.; Hossain, M.M.; Jahan, N.; Uddin, M.M.; Alarco, J.A.; Ostrikov, K. First hafnium-based MAX phase in the 312 family, Hf3AlC2: A first principles study. J. Alloy Compd. 2017, 727, 616–626. [Google Scholar] [CrossRef]
- He, X.; Bai, Y.; Zhu, C.; Sun, Y.; Li, M.; Barsoum, M.W. General trends in the structural, electronic and elastic properties of the M3AlC2 phases (M¼ transition metal): A first-principle study. Comput. Mater. Sci. 2010, 49, 691–698. [Google Scholar] [CrossRef]
- Radovic, M.; Barsoum, M.W.; Ganguly, A.; Zhen, T.; Finkel, P.; Kalidindi, S.R.; Lara-Curzio, E. On the elastic properties and mechanical damping of Ti3SiC2, Ti3GeC2, Ti3Si0.5Al0.5C2 and Ti2AlC in the 300–1573 K temperature range. Acta Mater. 2006, 54, 2757–2767. [Google Scholar] [CrossRef]
- Pugh, S.F. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals. Philos. Mag. 1954, 45, 823–843. [Google Scholar] [CrossRef]
- Anderson, O.L.; Demarest, H.H., Jr. Elastic constants of the central force model for cubic structures: Polycrystalline aggregates and instabilities. J. Geophy. Res. 1971, 76, 1349–1369. [Google Scholar] [CrossRef]
- Frantsevich, I.N.; Voronov, F.F.; Bokuta, S.A. Elastic Constants and Elastic Moduli of Metals and Insulators; Frantsevich, I.N., Ed.; NaukovaDumka: Kiev, Ukraine, 1983; Volume 60, p. 180. [Google Scholar]
- Vaitheeswaran, G.; Kanchana, V.; Svane, A.; Delin, A. Elastic properties of MgCNi3—A superconducting perovskite. J. Phys. Condens. Matter. 2007, 19, 326214. [Google Scholar] [CrossRef]
- Ge, Y.; Ma, S.; Bao, K.; Tao, Q.; Zhao, X.; Feng, X.; Li, L.; Liu, B.; Zhu, P.; Cui, T. Superconductivity with high hardness in Mo3 C2. Inorg. Chem. Front. 2019, 6, 1282–1288. [Google Scholar] [CrossRef]
- Borysiuk, V.N.; Mochalin, V.N.; Gogotsi, Y. Molecular dynamic study of the mechanical properties of two-dimensional titanium carbides Tin+1Cn (MXenes). Nanotechnology 2015, 26, 265705. [Google Scholar] [CrossRef]
- Bai, Y.; Zhou, K.; Srikanth, N.; Pang, J.H.; He, X.; Wang, R. Dependence of elastic and optical properties on surface terminated groups in two-dimensional MXene monolayers: A first-principles study. RSC Adv. 2016, 6, 35731–35739. [Google Scholar] [CrossRef]
- Xie, C.; Oganov, A.R.; Li, D.; Debela, T.T.; Liu, N.; Dong, D.; Zeng, Q. Effects of carbon vacancies on the structures, mechanical properties, and chemical bonding of zirconium carbides: A first-principles study. Phys. Chem. Chem. Phys. 2016, 18, 12299–12306. [Google Scholar] [CrossRef]
- Zhu, J.J.; Chroneos, A.; Eppinger, J.; Schwingenschlögl, U. S-functionalized MXenes as electrode materials for Li-ion batteries. Appl. Mater. Today 2016, 5, 19–24. [Google Scholar] [CrossRef]
- Zhu, J.; Chroneos, A.; Wang, L.; Rao, F.; Schwingenschloegl, U. Stress-enhanced lithiation in MAX compounds for battery applications. Appl. Mater. Today 2017, 9, 192–195. [Google Scholar] [CrossRef]
- Pang, J.B.; Mendes, R.G.; Bachmatiuk, A.; Zhao, L.; Ta, H.Q.; Gemming, T.; Liu, H.; Liu, Z.F.; Rummeli, M.H. Applications of 2D MXenes in energy conversion and storage systems. Chem. Soc. Rev. 2019, 48, 72–133. [Google Scholar] [CrossRef]
- Luan, S.R.; Zhou, J.S.; Xi, Y.K.; Han, M.Z.; Wang, D.; Gao, J.J.; Hou, L.; Gao, F.M. High lithium-ion storage performance of Ti3SiC2 MAX by oxygen doping. Chem. Sel. 2019, 4, 5319–5321. [Google Scholar]
- Er, D.; Li, J.; Naguib, M.; Gogotsi, Y.; Shenoy, V.B. Ti3C2 MXene as a high capacity electrode material for metal (Li, Na, K., Ca) ion batteries. ACS Appl. Mater. Interfaces 2014, 6, 11173–11179. [Google Scholar] [CrossRef]
- Varotsos, P. Point defect parameters in β-PbF2 revisited. Solid State Ion. 2008, 179, 438–441. [Google Scholar] [CrossRef]
- Chroneos, A.; Vovk, R.V. Modeling self-diffusion in UO2 and ThO2 by connecting point defect parameters with bulk properties. Solid State Ion. 2015, 274, 1–3. [Google Scholar] [CrossRef]
- Cooper, M.W.D.; Grimes, R.W.; Fitzpatrick, M.E.; Chroneos, A. Modeling oxygen self-diffusion in UO2 under pressure. Solid State Ion. 2015, 282, 26–30. [Google Scholar] [CrossRef]
| Phase | c11 | c12 | c13 | c33 | c44 | A | kc/ka | B | G | Y | B/G | v | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ti3AlC2 | 355 | 74 | 66 | 295 | 125 | 0.971 | 1.314 | 157 | 131 | 307 | 1.199 | 0.174 | [44] |
| 358 | 84 | 75 | 293 | 122 | 0.974 | 1.343 | 163 | 127 | 303 | 1.279 | 0.190 | [47] | |
| 361 | 75 | 70 | 299 | 124 | 0.954 | 1.297 | 160 | 131 | 309 | 1.221 | 0.178 | [47] | |
| 368 | 81 | 76 | 313 | 130 | 0.983 | 1.253 | 168 | 135 | 320 | 1.245 | 0.183 | [46] | |
| - | - | - | - | - | - | - | 165 | 124 | 297 | 1.331 | 0.20 | [49] | |
| Zr3AlC2 | 322 | 84 | 97 | 287 | 138 | 1.330 | 1.116 | 165 | 122 | 294 | 1.353 | 0.203 | This |
| 314 | 78 | 79 | 262 | 107 | 1.024 | 1.279 | 151 | 110 | 266 | 1.373 | 0.207 | [46] | |
| V3AlC2 | 404 | 84 | 108 | 361 | 158 | 1.151 | 1.075 | 197 | 153 | 364 | 1.288 | 0.191 | This |
| 390 | 82 | 116 | 358 | 158 | 1.225 | 0.991 | 196 | 147 | 354 | 1.333 | 0.200 | [46] | |
| Hf3AlC2 | 349 | 79 | 79 | 283 | 123 | 0.963 | 1.324 | 161 | 125 | 298 | 1.288 | 0.192 | This |
| 347 | 77 | 80 | 291 | 127 | 0.941 | 1.251 | 162 | 127 | 302 | 1.276 | 0.189 | [50] | |
| 357 | 82 | 83 | 283 | 126 | 0.940 | 1.365 | 166 | 128 | 305 | 1.297 | 0.193 | [51] | |
| 348 | 79 | 82 | 290 | 112 | 1.058 | 1.264 | 163 | 121 | 291 | 1.347 | 0.203 | [52] | |
| Ta3AlC2 | 411 | 113 | 136 | 343 | 156 | 0.772 | 1.217 | 214 | 143 | 351 | 1.497 | 0.227 | This |
| 441 | 132 | 138 | 382 | 175 | 0.781 | 1.217 | 231 | 157 | 384 | 1.471 | 0.223 | [46] | |
| Ti3SiC2 | 365 | 89 | 99 | 352 | 156 | 1.202 | 1.012 | 184 | 143 | 341 | 1.287 | 0.191 | [44] |
| 370 | 99 | 111 | 349 | 151 | 1.209 | 1.038 | 192 | 138 | 334 | 1.392 | 0.210 | [46] | |
| 372 | 88 | 98 | 353 | 167 | 1.267 | 1.036 | 185 | 149 | 352 | 1.245 | 0.183 | [48] | |
| - | - | - | - | - | - | - | 185 | 139 | 333 | 1.331 | 0.20 | [49] | |
| - | - | - | - | - | - | - | 186 | 144 | 343 | 1.291 | 0.192 | [48] | |
| Hf3SiC2 | 357 | 93 | 115 | 334 | 157 | 1.362 | 1.005 | 188 | 136 | 329 | 1.382 | 0.209 | This |
| 348 | 101 | 120 | 335 | 144 | 1.300 | 0.972 | 190 | 127 | 312 | 1.496 | 0.227 | [46] | |
| Ta3SiC2 | 335 | 145 | 221 | 325 | 179 | 3.284 | 0.365 | 239 | 103 | 270 | 2.320 | 0.317 | This |
| 352 | 220 | 210 | 345 | 182 | 2.628 | 1.126 | 256 | 102 | 270 | 2.509 | 0.324 | [46] | |
| Zr3SiC2 | 323 | 85 | 99 | 304 | 135 | 0.794 | 1.024 | 169 | 122 | 295 | 1.385 | 0.209 | This |
| 320 | 100 | 107 | 296 | 125 | 0.804 | 1.090 | 174 | 113 | 279 | - | 0.233 | [46] | |
| Mo3SiC2 | 377 | 175 | 186 | 364 | 151 | 1.637 | 1.011 | 245 | 116 | 301 | 2.112 | 0.300 | This |
| Hf3SnC2 | 320 | 95 | 96 | 300 | 115 | 1.075 | 1.093 | 168 | 112 | 275 | 1.500 | 0.227 | [45] |
| 326 | 96 | 97 | 300 | 107 | 0.991 | 1.123 | 170 | 110 | 272 | 1.550 | 0.234 | [46] | |
| Ti3SnC2 | 319 | 103 | 80 | 304 | 113 | 0.976 | 1.170 | 163 | 112 | 273 | 1.455 | 0.221 | [44] |
| 331 | 96 | 80 | 285 | 108 | 0.943 | 1.302 | 161 | 113 | 274 | 1.436 | 0.217 | [46] | |
| 331 | 91 | 81 | 299 | 129 | 1.103 | 1.193 | 162 | 122 | 285 | 1.328 | 0.208 | [48] | |
| Zr3SnC2 | 280 | 92 | 84 | 257 | 110 | 1.192 | 1.179 | 148 | 99 | 243 | 1.495 | 0.227 | [45] |
| 297 | 90 | 87 | 268 | 95 | 0.972 | 1.177 | 154 | 98 | 244 | 1.571 | 0.237 | [46] | |
| Ti3InC2 | 338 | 80 | 63 | 276 | 92 | 0.754 | 1.371 | 151 | 111 | 267 | 1.360 | 0.205 | [44] |
| 340 | 85 | 67 | 263 | 97 | 0.826 | 1.478 | 152 | 111 | 267 | 1.362 | 0.205 | [46] | |
| Ti3GaC2 | 359 | 78 | 69 | 292 | 123 | 0.959 | 1.341 | 159 | 130 | 306 | 1.223 | 0.179 | [44] |
| 356 | 86 | 75 | 285 | 113 | 0.920 | 1.390 | 162 | 122 | 293 | 1.324 | 0.198 | [46] | |
| Ti3GeC2 | 356 | 88 | 91 | 324 | 140 | 1.125 | 1.125 | 175 | 134 | 320 | 1.306 | 0.195 | [44] |
| 357 | 100 | 97 | 325 | 129 | 1.051 | 1.152 | 180 | 126 | 307 | 1.426 | 0.216 | [46] | |
| 355 | 85 | 94 | 338 | 148 | 1.171 | 1.032 | 177 | 138 | 312 | 1.283 | 0.207 | [48] |
| 312 MAX Phases | Formation Energy/eV |
|---|---|
| Ti3AlC2 | 1.1966 |
| Zr3AlC2 | 0.1499 |
| V3AlC2 | 2.6507 |
| Hf3AlC2 | 0.4172 |
| Ta3AlC2 | 1.7890 |
| Ti3SiC2 | 1.1597 |
| Hf3SiC2 | 0.6902 |
| Ta3SiC2 | 1.2943 |
| Mo3SiC2 | 0.0056 |
| Zr3SiC2 | 0.1608 |
| Hf3SnC2 | 1.9508 |
| Ti3SnC2 | 2.9303 |
| Zr3SnC2 | 1.6645 |
| Ti3InC2 | 2.1320 |
| Ti3GaC2 | 0.9735 |
| Ti3GeC2 | 1.5416 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filippatos, P.P.; Hadi, M.A.; Christopoulos, S.-R.G.; Kordatos, A.; Kelaidis, N.; Fitzpatrick, M.E.; Vasilopoulou, M.; Chroneos, A. 312 MAX Phases: Elastic Properties and Lithiation. Materials 2019, 12, 4098. https://doi.org/10.3390/ma12244098
Filippatos PP, Hadi MA, Christopoulos S-RG, Kordatos A, Kelaidis N, Fitzpatrick ME, Vasilopoulou M, Chroneos A. 312 MAX Phases: Elastic Properties and Lithiation. Materials. 2019; 12(24):4098. https://doi.org/10.3390/ma12244098
Chicago/Turabian StyleFilippatos, P.P., M.A. Hadi, S.-R.G. Christopoulos, A. Kordatos, N. Kelaidis, M.E. Fitzpatrick, M. Vasilopoulou, and A. Chroneos. 2019. "312 MAX Phases: Elastic Properties and Lithiation" Materials 12, no. 24: 4098. https://doi.org/10.3390/ma12244098
APA StyleFilippatos, P. P., Hadi, M. A., Christopoulos, S.-R. G., Kordatos, A., Kelaidis, N., Fitzpatrick, M. E., Vasilopoulou, M., & Chroneos, A. (2019). 312 MAX Phases: Elastic Properties and Lithiation. Materials, 12(24), 4098. https://doi.org/10.3390/ma12244098








