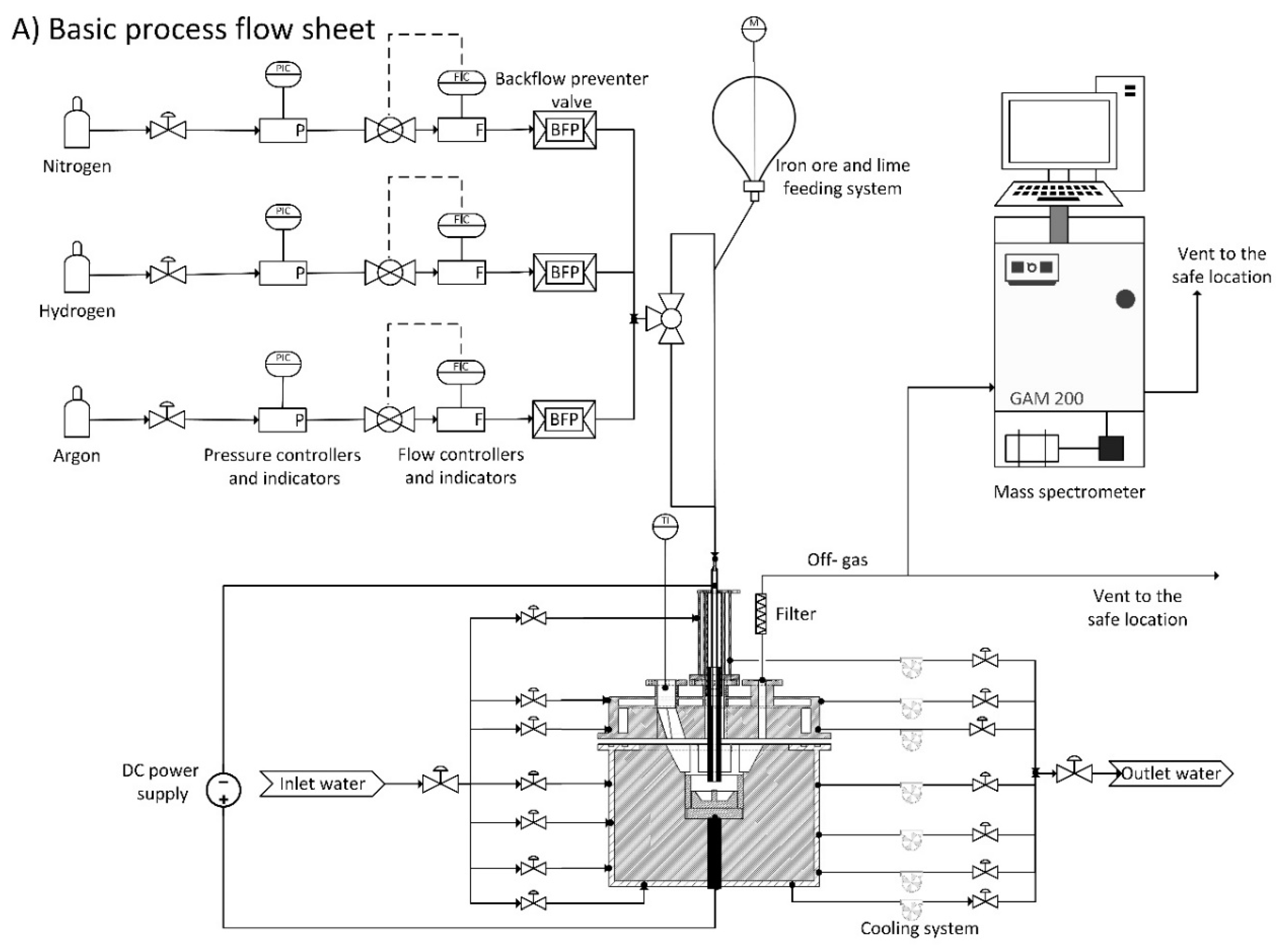
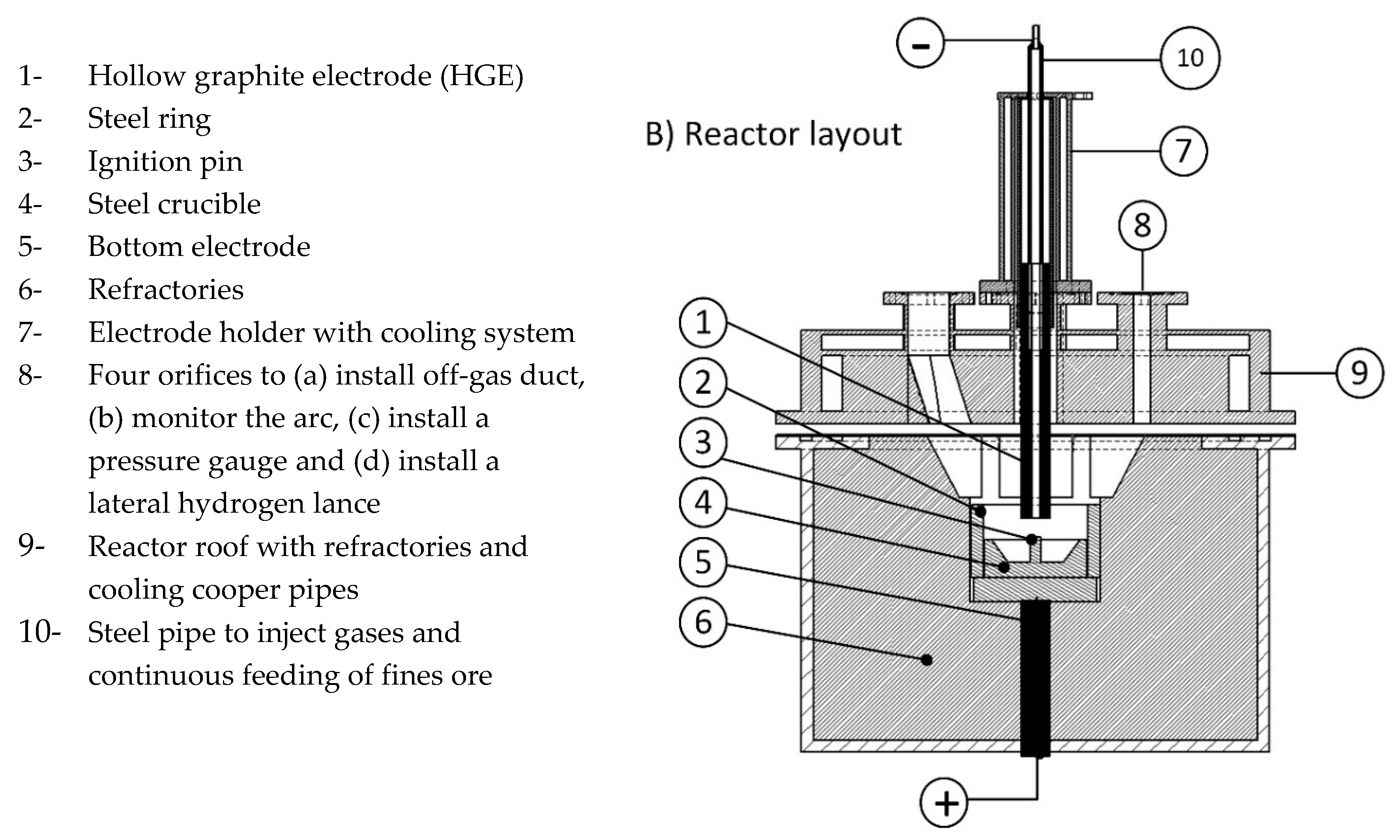
3.1. Materials and Sample Preparation
In this study, the reduction behaviour of haematite using a pre-mixture of H
2–Ar plasma arc was assessed. The experiments were carried out with three different weights of iron ore powder in steel crucibles. The powders were melted and reduced by hydrogen to study the parameters that influence the iron ore reduction behaviour.
Table 2 shows the experimental program with the definition of the experiment parameters.
For the reduction of iron ore, a premixed 50% H2–50% Ar gas with a total flowrate of 5 L/min for all runs was used. The flow controllers could supply a mixture with different hydrogen-to-argon ratios and different flow rates.
Carajas iron ore as a high-quality raw material was selected to run the experiments. The chemical composition of Carajas iron ore is shown in
Table 3.
However, there was a possibility to feed the iron ore continuously with a continuous feeding system, and in this study the total amount of iron ore was charged into the crucible before the test run. The steel crucible was located on a steel disc connected to the 4-pin electrodes. One steel ring with a layer of MgO refractory was located on the outer diameter of the crucible to protect the reactor side-wall from the plasma arc radiation. Then, the roof was assembled and the whole system was completely sealed. To adjust the slag basicity, lime with the same grain size distribution was used. Grain size distribution of lime and iron ore were separately classified, and then mixed.
Table 4 shows the grain size distribution of the Carajas iron ore and lime.
To remove the gases produced due to the lime calcination in the plasma reactor, it was calcined at the temperature of 1100 °C before mixing with iron ore powder. To prepare the sample powder, 3.4 g of calcined lime with 97% of CaO and 2.2% of MgO content was mixed with 100 g of iron ore to reach the basicity of two.
Not only the ignition pin but also the steel crucible were melted during pre-melting and were mixed with the iron oxide liquid. Therefore, the carbon content of those steel parts contributed to the reduction reactions. The crucible was partially melted, so it was essential to assess the melting of the crucible after each test run to determine the amount of carbon contributed to the reduction reactions from the steel crucible. The crucible was cut from the middle and analysed using the spectrometer, and the micro- and macrostructure were evaluated using an optical microscope. The partially melted crucible from each test run was observed and the weight was approximately calculated. The other source of carbon introduced to the melt was from the ignition pin. The weight of the ignition pin was 15 g. The chemical composition of the ignition pin and the steel crucible is shown in
Table 5.
One HGE as a cathode with an outer diameter of 26 mm and inner diameter of 8 mm was used to inject the premixed gas through it. By means of a glass window installed on one of the orifices on the reactor roof, the plasma arc was monitored.
The off-gas contained dust, mainly carbon from graphite electrode. Dust creates problems for the mass spectrometer (MS), so it should be appropriately cleaned before entering the MS. Therefore, a steel grid, glass wool, coolant/water trap, molecular sieve 3A° type 562 C and silica gel were used for the off-gas cleaning. This off-gas cleaning system setup can not only capture the accompanying dust but also remove water vapour from the off-gas. The off-gas was analysed using a mass spectrometer GAM 200 during the operations. The MS was calibrated by a calibration gas which the chemical composition is shown in
Table 6. The calibration gas was produced to meet the requirements of Grade 1. Therefore, the relative uncertainty for the components with a content between 0.1% and 4.9% is ±2% and for those more than 4.9% is ±1%.
3.2. Description of the Operation
Prior to the ignition of the plasma arc, the whole system was purged by argon with a flow rate of 5 L/min for 10 min to withdraw oxygen from the system. While purging, the chemical composition of the outlet gas was monitored in the MS to be certain of decreasing the oxygen percentage to less than 1%. Then, the arc was ignited and pure argon with a flow rate of 5 L/min was used for pre-melting to make a liquid pool of iron ore inside the steel crucible. This step of the operation was run for 3 min, and then the injection gas switched from pure argon to premixed hydrogen/argon with a flow rate of 2.5/2.5 L/min to begin the reduction process.
During the reduction step, the gas flowrate and the composition were kept constant. The voltage was not constant during the reduction step due to the changes of hydrogen concentration. At the beginning of the reduction process, the degree of hydrogen utilization was high, which caused a decrease in the hydrogen concentration. Then the degree of hydrogen utilization decreased continuously during the reduction process, and it led to a higher energy consumption and an increase of voltage. Because hydrogen is a diatomic molecule and needs more energy than argon for the ionization [
34,
35,
36], when the voltage exceeded 100 V, it caused a cut off of the plasma arc. To prevent the operation shutting down, the arc length, the distance from the tip of the electrode and the surface of the molten metal were all readjusted. Hence, corresponding to the voltage, the HGE was manually driven downwards to decrease the arc length.
The reduction step lasted until reached less than 15% which is a low degree of hydrogen utilization. At the lower degrees, the arc mainly runs between the graphite electrode and the edge of the steel crucible or on the already reduced sections, therefore, the operation was stopped. After completing the reduction process, hydrogen injection was stopped, and the reactor was purged by nitrogen with a flow rate of 5 L/min to remove hydrogen from the system. To conduct the mass balance, the crucible, refractory ring, electrode and filters were weighed before and after each experiment. To assess the reduction process and the chemical composition of the steel products, slag was separated from the crucible, and then the crucible was cut from the middle and analysed using a spectrometer.
3.3. Method of Calculation of Hydrogen Utilization Degree and Degree of Reduction
To assess the data and to obtain results with the minimum amount of deviation, the raw chemical composition shown by MS should be corrected in two steps. The first step is to remove the unwanted elements from the chemical composition, namely nitrogen, oxygen and water. After removing water from the off-gas, less than 0.2% water is still shown by MS as remaining in the off-gas. Small amounts of nitrogen (in a range between 0.2% and 0.5%) and oxygen (less than 0.06%) were also shown in MS. The reason was the sucking of air by the MS from the outlet pipe because the off-gas flow rate and pressure were too low. In order to reduce the errors, the off-gas composition was corrected by removing H
2O, N
2 and O
2. The second step is to remove the deviation of the MS. For this reason, after calibration of the MS, the calibration gas was again analysed by MS. Even after this calibration, the composition of the off-gas shown by MS was not exactly the same as its actual composition. This means that there was a deviation in the chemical composition shown by MS. Therefore, to eliminate the MS deviation, the results of the MS were corrected by the deviation factors of each element, which were obtained from the difference between the real calibration gas composition and the measured values.
Table 7 shows the deviation of each element.
The deviation of MS by this method was removed from the results. Therefore, only the relative uncertainty of each component should be considered.
The measurement cycle by MS was set to 8.4 s, which was enough time to deliver reliable results for the gas composition changes during operations.
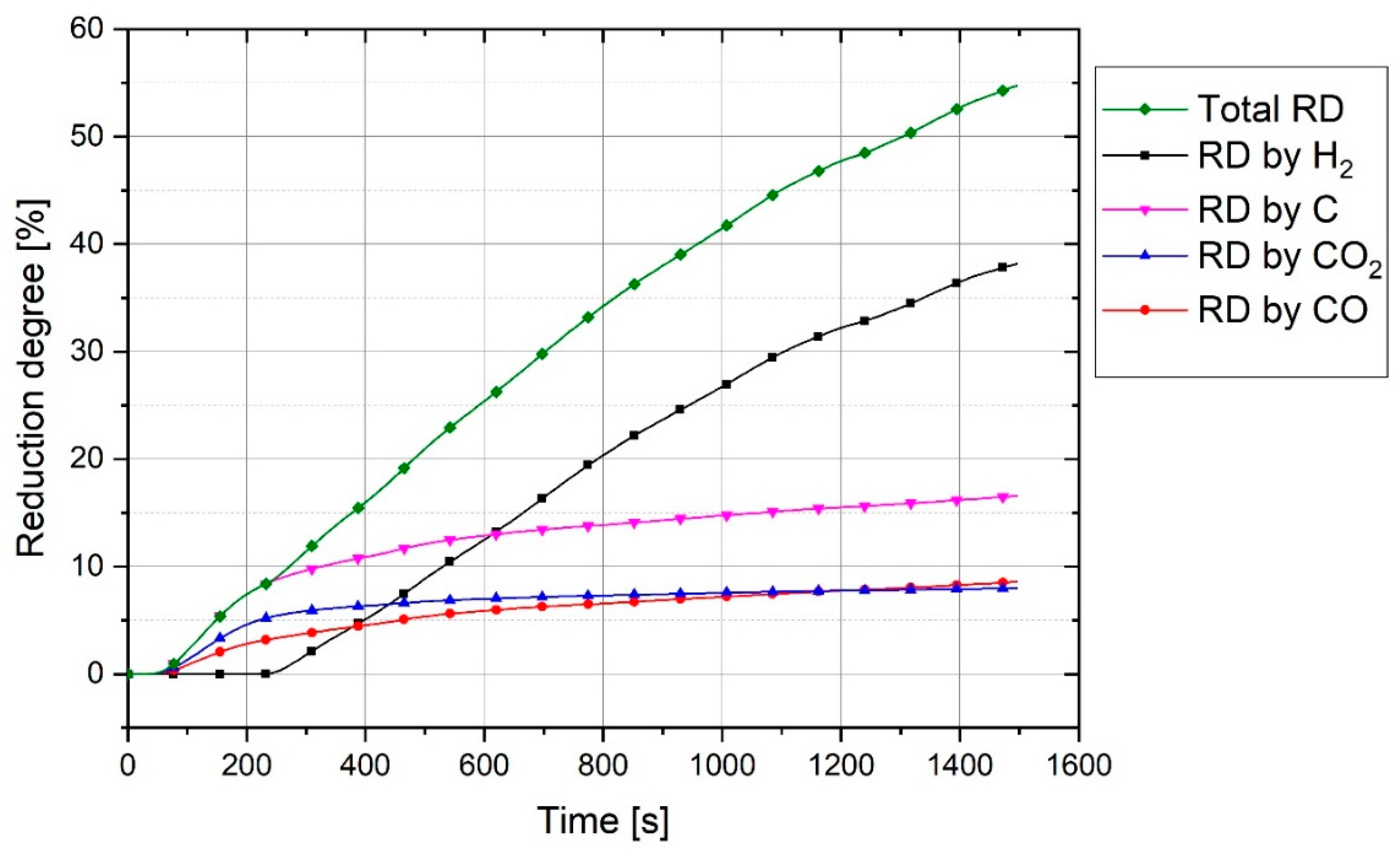
Regarding the premixed argon/hydrogen inlet gas and the reduction reactions, the off-gas released from the reactor comprises:
Ar: argon was the unreacted gas and can therefore leave the reactor without any reaction.
H2: a significant amount of hydrogen left the reactor without any reaction due to the thermodynamic equilibrium and kinetics limitations.
H2O: water vapor was the product of the reduction reactions of metal oxides with hydrogen.
CO and CO2: these gases were the products of the reduction of metal oxides by carbon. The erosion of the graphite electrode and carbon from melting of the ignition pin and steel crucible caused carbon to enter the iron oxide melting pool. The carbon then reduced the liquid iron oxide and CO and CO2 were released.
The off-gas flow rate was not equal to the inlet gas flow rate due to the formation of CO and CO
2 in the reactor and the condensation of H
2O in the off-gas cleaning system. To define the total flow rate and accordingly the flow rate of each gas, the flow rate of argon was used as a reference. Argon was an unreacted element and left the plasma reactor without any reaction. However, the outlet flowrate of argon was not the same as the inlet flowrate. To obtain the reference flow rate, argon and hydrogen were injected into the reactor without arcing to simulate the flow of off-gas.
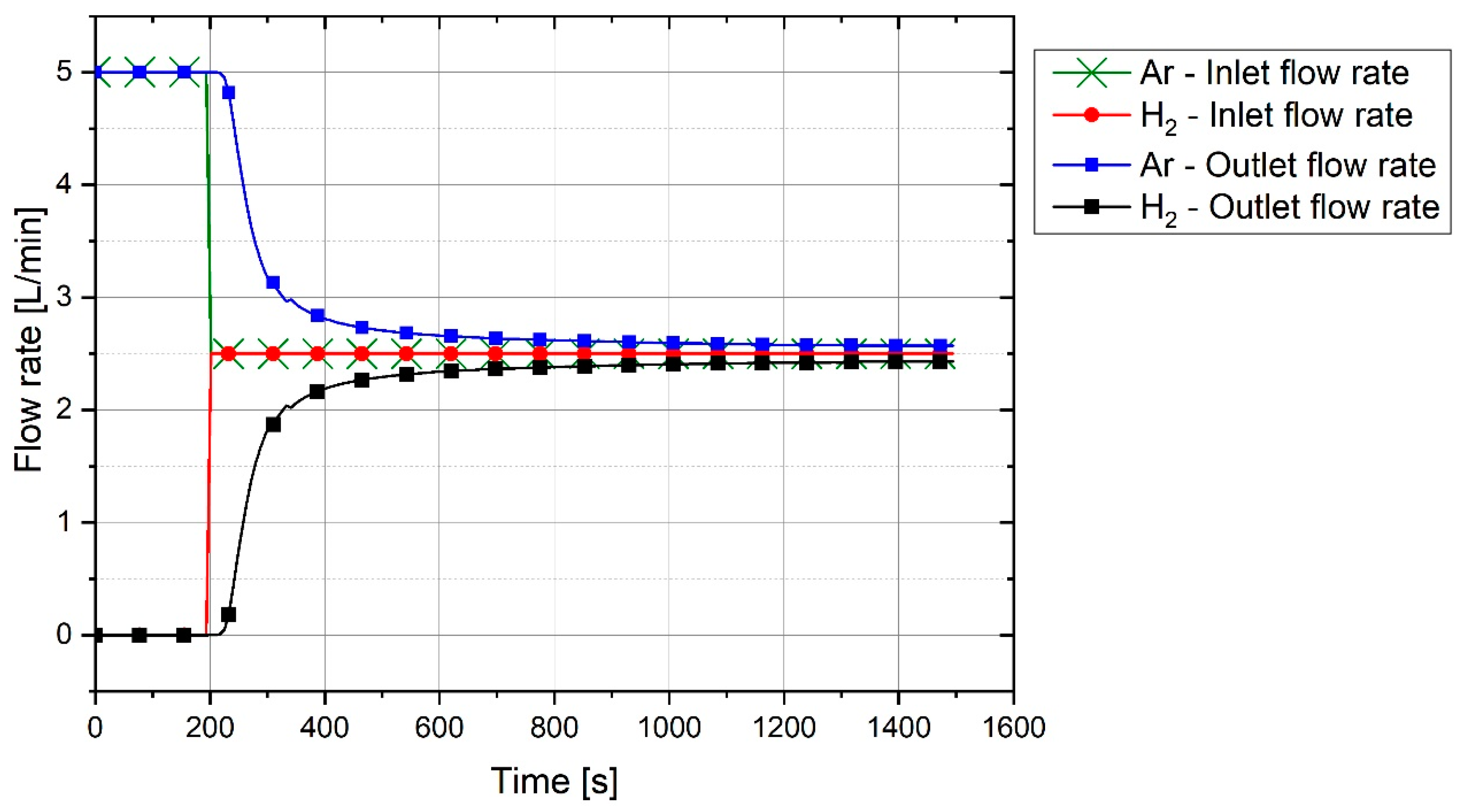
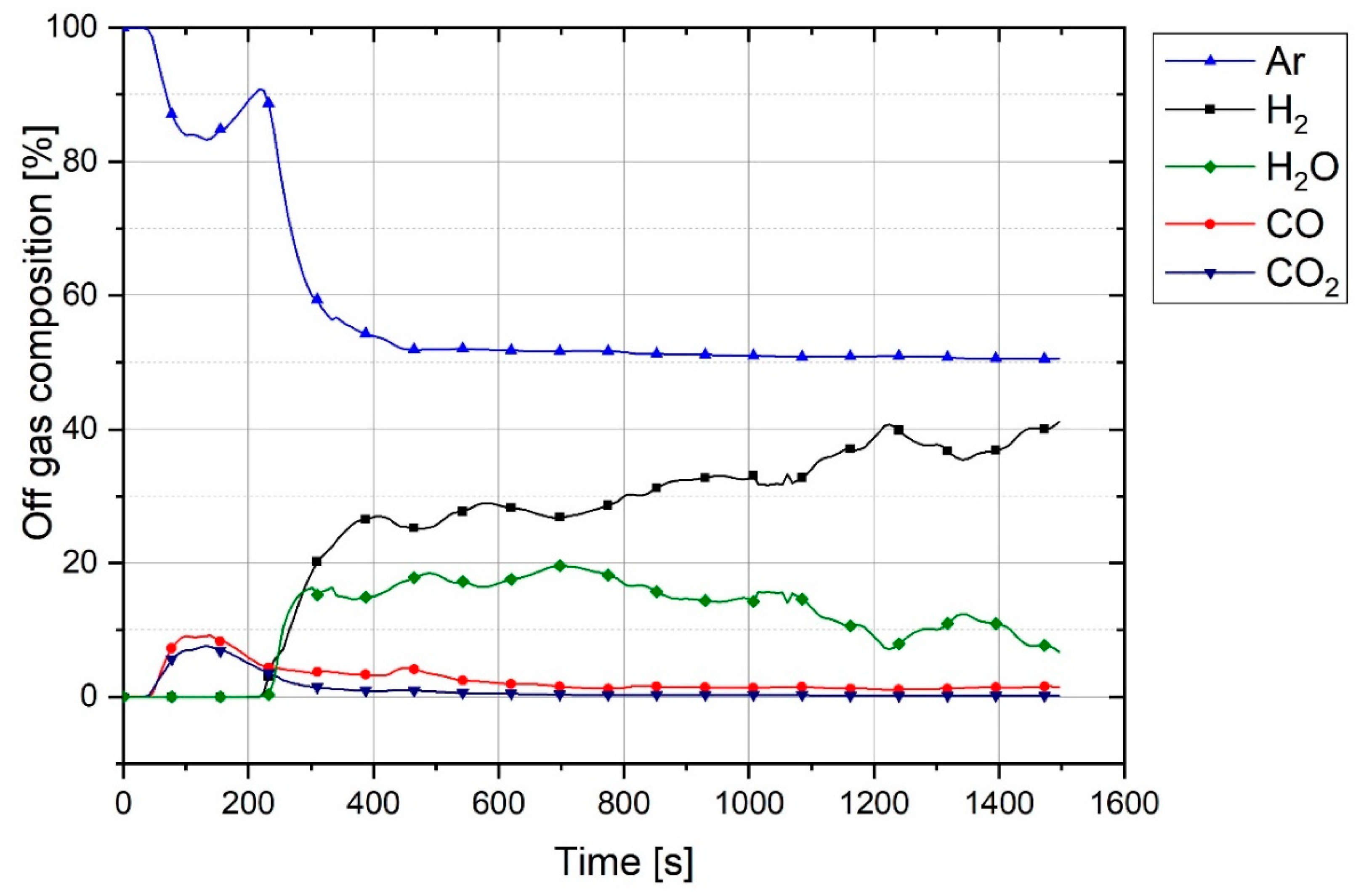
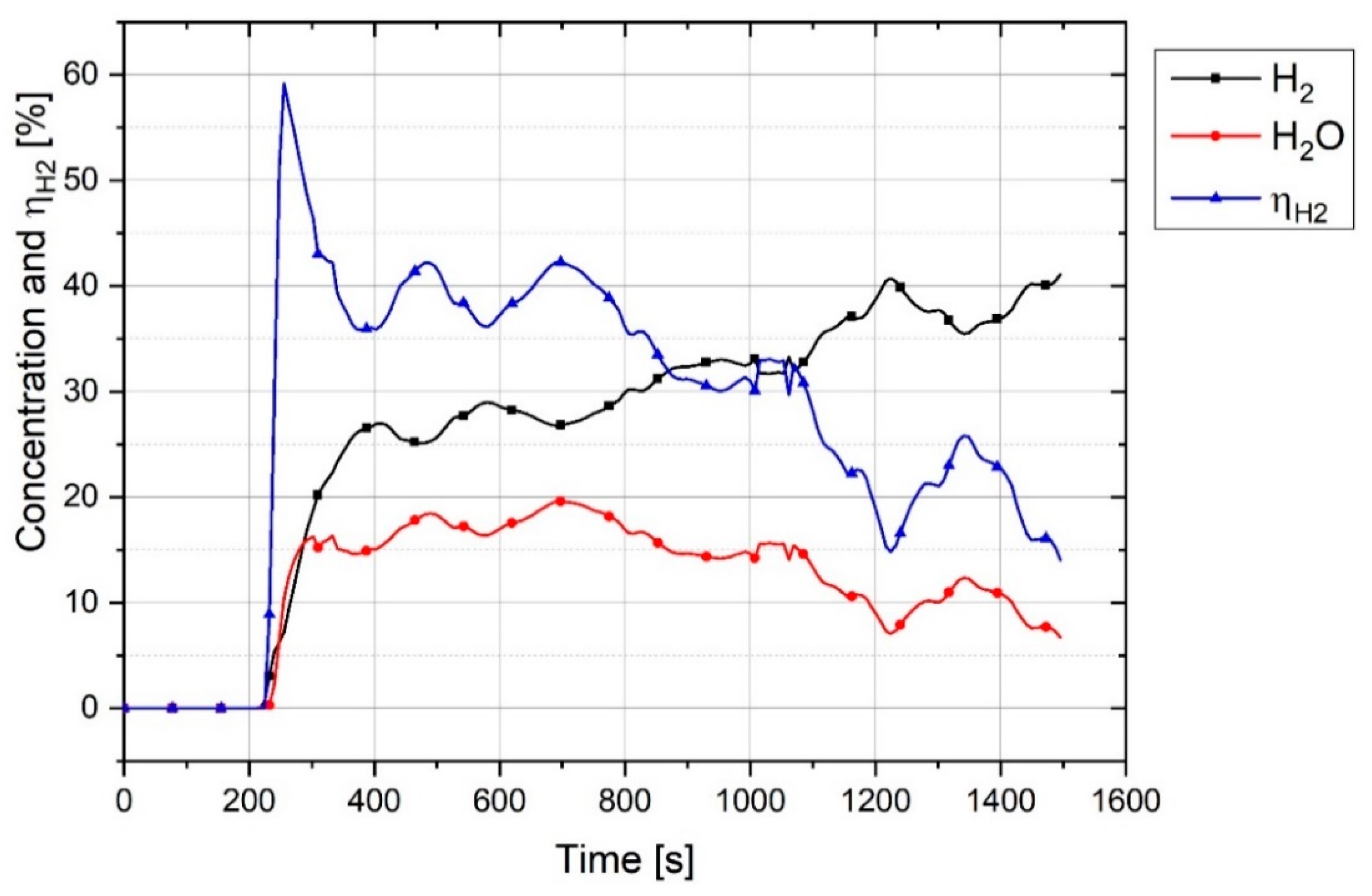
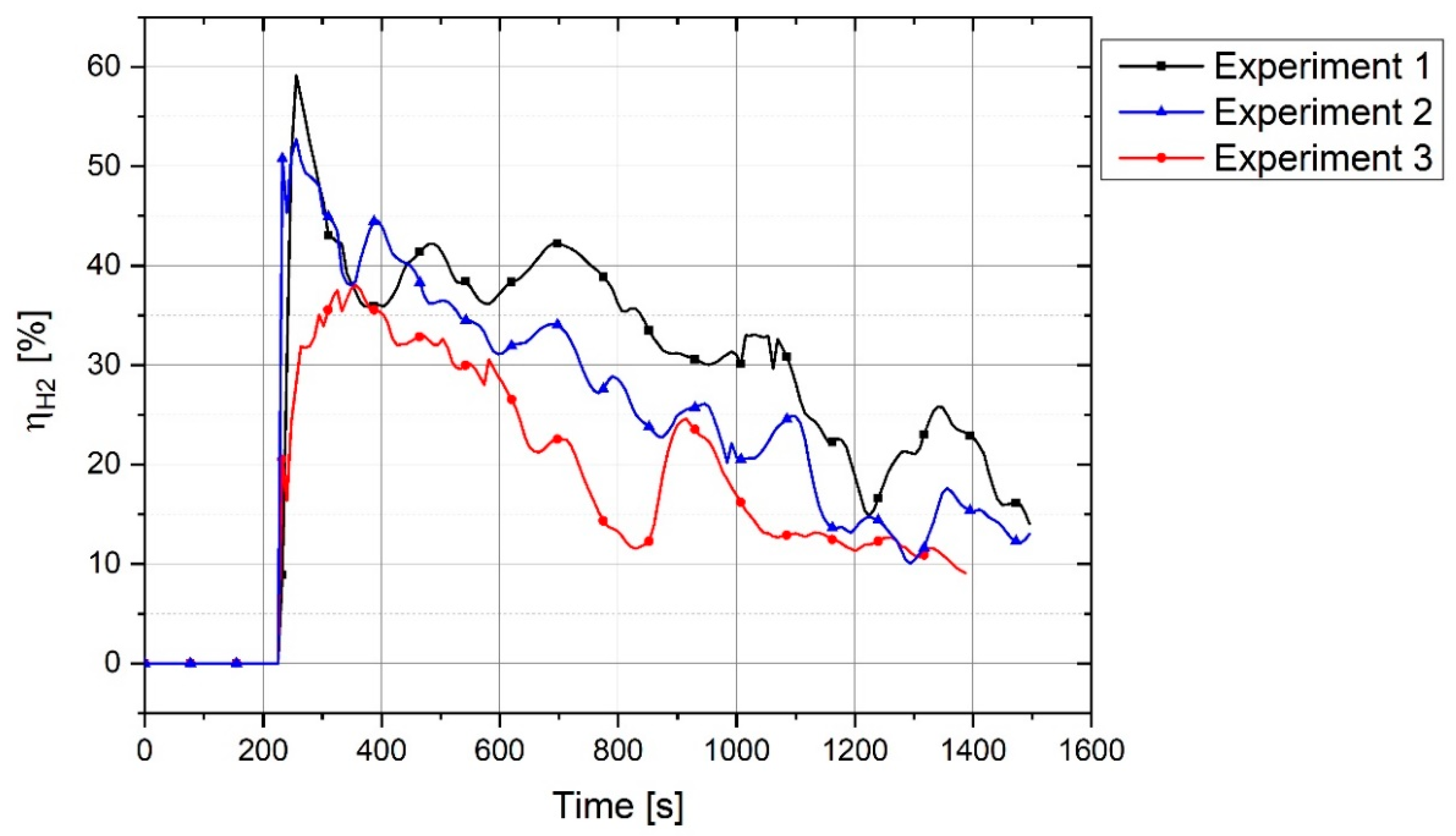
Figure 4 shows the graph of outlet gas composition after switching the gas from pure argon to a mixture of 50% argon and 50% hydrogen with a total flow rate of 5 L/min. This gas composition and the flowrate were used for all three experiments.
The plot shows that it takes 100 s for hydrogen to reach the MS. With the passage of time, argon was replaced by hydrogen. It is seen that, even after 1200 s, the composition of the outlet gas was not the same as that of the inlet gas. Therefore, it took time for the chemical composition of the inlet gas and the outlet gas to become closer. Hence, this graph was used as a reference to compare the results of the experiments and to find the real amount of hydrogen and water vapor produced.
For the experiments, before the start of arcing, the plasma reactor was purged with argon so that only argon was inside the reactor. Pre-melting was done by flowing pure argon, and for the reduction process, the gas was switched to a hydrogen/argon mixture. To calculate the degree of hydrogen utilization, the difference of hydrogen concentration between the reference flow rate and the MS result was considered.
Corresponding to the amount of argon from the reference, the total flow rate was defined by:
From the total flow rate, the flow rate of each gas was calculated. Water vapor was condensed in both the off-gas duct and the cleaning system. Hence, it was not possible to calculate the flow rate of the water vapor directly from the off-gas composition. Therefore, from the difference between the hydrogen flow rate in the off-gas and in the reference flow rate at the same time, the water vapor was calculated.
Finally, the total flow rate regarding water formation was calculated and the chemical composition of the off-gas was defined.
is the hydrogen utilization and was accrued from the H
2 and H
2O amount by
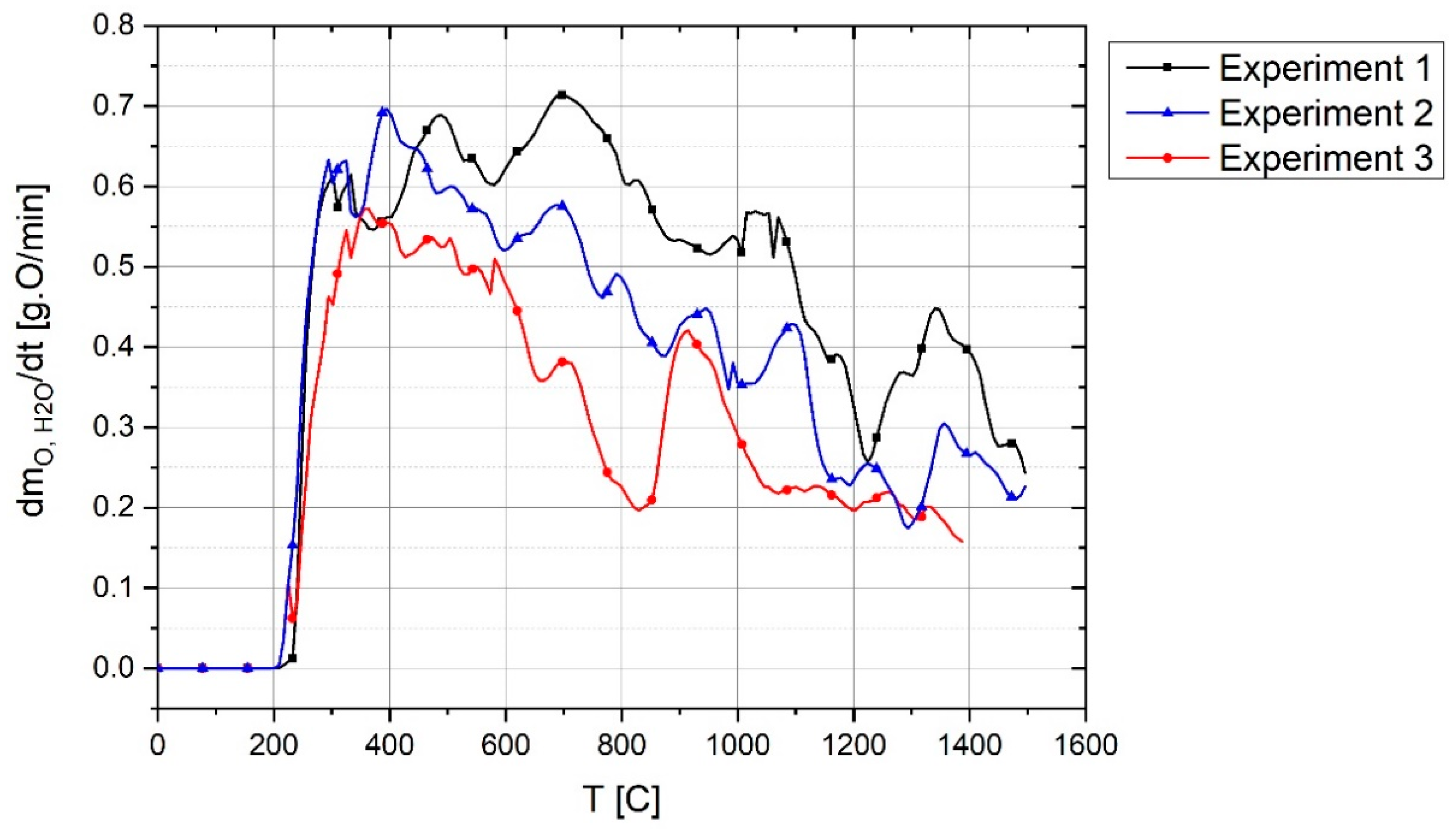
The reduced amount of oxygen was calculated by summing up the amount of oxygen in H
2O, CO and CO
2 by
where m
O,tot is the total mass of oxygen,
,
and
are the mass of oxygen in H
2O, CO and CO
2 espectively, and C
n, C
n−1 (S) are cycles n and n − 1 from MS, respectively. The degree of reduction (R
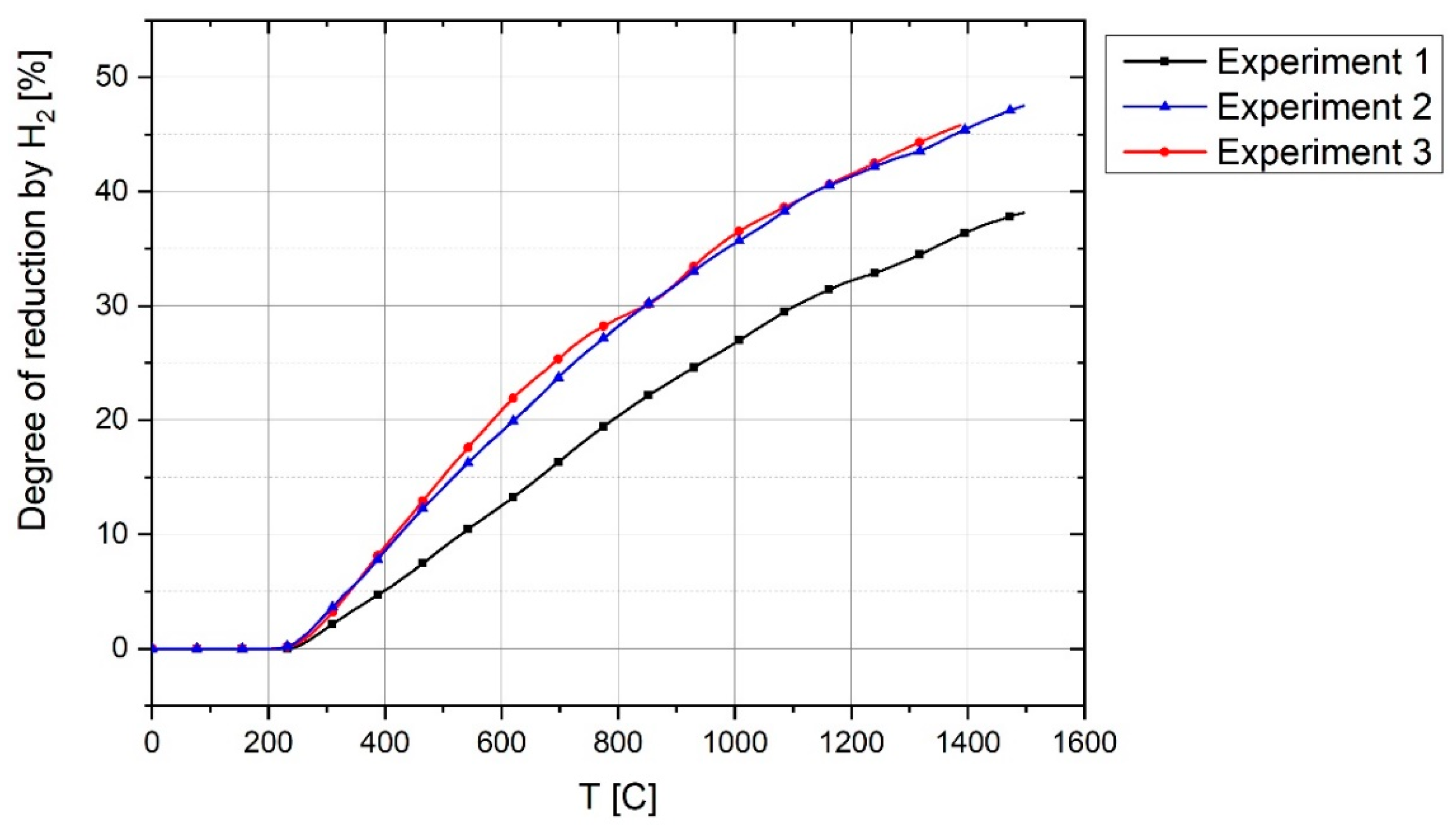
Degree), which is the oxygen reduced by carbon and hydrogen during the experiment, was calculated by
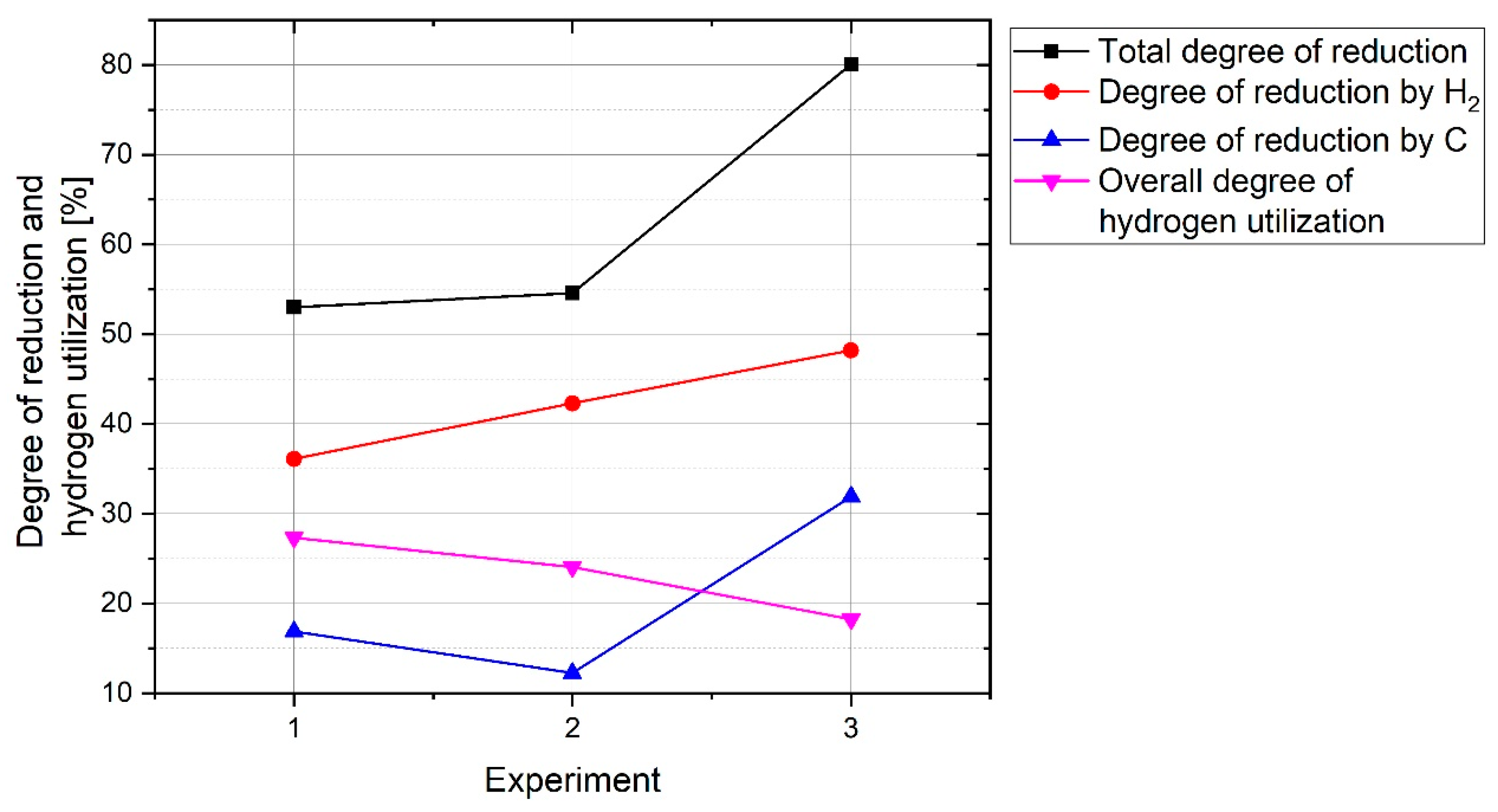
Therefore, the total degree of reduction was
Carbon was introduced into the melt from three different sources, which were the graphite electrode, ignition pin and steel crucible. Therefore, the contribution of the carbon from those sources to the reduction reactions should be taken into account. The ignition pin was completely melted and mixed with the melt. However, the steel crucible was not completely melted. The steel crucible of each experiment was cut in half and different points were analysed by a spectrometer, while the micro- and macrostructure were assessed using optical microscopy to estimate the amount of melted section. No sign of melting was observed in the crucible of Experiment 1.
Figure 5 shows the cross-section of the crucible. However, the crucibles of Experiments 2 and 3 were partially melted.
The total amount of carbon that contributed to the reduction of oxides was calculated in two ways. The first was to calculate from the chemical composition of the off-gas. The second was calculated by the following three steps:
The loss of graphite electrode weight by weighing before and after the experiment
Due to the complete melting of the ignition pin, the carbon from the ignition pin was the total carbon in the ignition pin minus the carbon remaining in the produced iron
Regarding the partial melting of the crucible, the carbon introduced from the crucible was the difference of the carbon content in the crucible and the produced iron multiplied by the estimated weight of the melted crucible.
The total mass of carbon obtained from weighing the parts before and after each experiment should have been equal to the total carbon calculated by the results of MS.