3.1. Influence of Surface Treatment Method on Interface Inclusions
The SEM images of the interface for different surface treatments are shown in
Figure 3. It can be seen that the surface of the stainless steel after brush grinding was not flat and exhibited an irregular shape. The stainless steel contains a high content of Cr, which can form a stable Cr
2O
3 passive film at atmosphere or in the presence of oxidizing medium. During the process of brush grinding, the Cr
2O
3 passive film on the surface of the stainless steel was lifted by the grinding force, and was subsequently covered irregularly on the surface of stainless steel. Three different positions for chemical composition analysis were used, as shown in
Figure 3a. The WDS analysis of these three positions on the stainless steel surface is shown in
Table 2. O element was observed at the third point. Thus, region 3 was stainless steel substrates. O was present at the first and second point, and the content of oxygen in region 1 was greater than region 2. The content of O decreases with increasing depth based on the structural characteristics of stainless steel passive film [
15], which indicated that region 1 was the outer layer of the passive film on the surface of stainless steel, and region 2 was the secondary outer layer. Also, the surface at region 1 was not polished for its lower position.
Figure 3b shows a valley-like surface after pickling, and it can be seen that more granular inclusions were formed at the bottom of valley, as shown in the higher magnification image. Valleys are formed at the dislocations, inclusions, and other defects at the grain boundaries in the steel matrix, and can be easily corroded by the acid solution [
16,
17].
The SEM images of the surface of 304 stainless steel and carbon steel after high-temperature oxidation experiments for the two kinds of surface treatment are shown in
Figure 4. It can be seen that the irregular lamellar shape of the stainless steel surface after brush grinding was basically eliminated in
Figure 4a, but the surface was still not flat and initial joining did not appear on the composite surface. The stainless steel substrate was austenite after cooling. With the growth of grains, the surface of the stainless steel exhibited some ups and downs, but not obviously. Moreover, the physicochemical properties of the two metals were quite different, and they required more pressure to generate bonding. The clamping force was insufficient to form the initial bonding. The inclusions were not detected in the flat area of stainless steel in the enlarged area by WDS analysis. A number of circular inclusions of different sizes were formed in depressions [
18]. The main constituents were O: 48.0 at %, Al: 5.2 at %, Si: 10.0 at %, Mn: 4.3 at %, Cr: 4.2 at %, Ni: 0.9 at %, and the balance was Fe.
When the metal is oxidized, the free energy (ΔG
0) depending on metal oxide is formed, which indicates the affinity between the metal and oxygen. The lower the value, the higher the stability of oxide and easier the metal is oxidized under lower free energy [
19].
Figure 5 shows the ΔG
0–T diagram of the metal oxidation reaction [
20], which is used to determine the possibility of an oxidation reaction. When the temperature is 1200 °C, 304 stainless steel and Q345 carbon steel are expected to form complex oxidation. The ΔG
0 of Al, Si, Mn, Cr, and Fe oxides are different, and the increasing sequence is ΔG
Al2O3 < ΔG
SiO2 < ΔG
MnO < ΔG
Cr2O3 < ΔG
FeO < ΔG
Fe3O4 < ΔG
Fe2O3, that is, the affinities of Al, Si, Mn, Cr, Fe, with O decrease. Thus, Al was preferentially oxidized to Al
2O
3. Meanwhile, Al diffused to the surface due to concentration difference. The content of oxygen in the valley or depressed surface region is high, and Al below the surface did not diffuse to the interface to react. While Al on the surface was consumed, the oxygen continued to react with Si and Mn. However, the oxygen content was limited, which resulted in the formation of composite inclusions dominated by Al
2O
3 and Si-Mn oxides.
Meanwhile, the protruded grain and the depressed grain boundary of the carbon steel can be seen in
Figure 4b. This is because, in the cooling process, the process of grain growth and recrystallization was completed based on sufficient phase transition in carbon steel. When the undercooled austenite in carbon steel changes to ferrite, pearlite and martensite, the volume expansion occurs. However, the expansion is small because of the presence of carbides at the grain boundaries. Martensite was also observed within grains of carbon steel [
18,
21,
22], as shown in
Figure 4b, which indicated that the microstructure of carbon steel near the interface was transformed to martensite on cooling without rolling.
In order to further verify the presence of martensite, TEM specimens were prepared by an ion thinning method. The microstructure of the composite region was clearly observed. A large number of lath martensite structures and diffraction spots of bcc martensite were observed in
Figure 6, which confirmed the existence of martensite. The chemical composition of carbon steel surface inclusions was studied by WDS and was O: 20.5 at %, Al: 10.3 at %, Si: 0.6 at %, Mn: 2.2 at %, Cr: 7.1 at %, Ni: 0.7 at %, and the balance was Fe, and Al
2O
3 and Si-Mn oxides were formed according to selective oxidation.
The surface image of stainless steel after pickling is shown in
Figure 4c. There were three features; holes, oxide inclusions, and a small number of dimples. Residual sulcus after pickling were not eliminated during high temperature oxidation, and circular holes were formed. A certain degree of metallurgical bonding formed in the localized area of the surface during high temperature oxidation stage. An initial bond formed, and dimples were formed after separation at the initial bond point. A certain number of small oxides were formed at the grain boundaries, and the enlarged view is shown in
Figure 7. The chemical composition was detected by WDS, O: 24.8 at %, Mg: 16.5 at %, Al: 6.4 at %, Si: 1.5 at %, Ca: 8.1 at %, Mn: 3.8 at %, the balance was Fe at the core of oxide. O: 20.7 at %, Mg: 1.8 at %, Al: 3.2 at %, Si: 3.6 at %, Ca: 0.6 at %, Mn: 3.8 at %, the balance Fe was detected on the shell of oxide. So, the composite oxides mainly consisted of a core of Ca, Mg, and Al oxides and a shell of Si and Mn oxides.
Considering that the stainless steel billet experienced deoxidation during the smelting process, oxides and silicates were formed. At the same time, a little amount of oxygen, sulfur, aluminum and other impurity elements remained in the steel in the form of compounds or elements [
23], and the complex oxide inclusions such as SiO
2, Al
2O
3, MgO and CaO were formed in stainless steel. After pickling, oxygen accumulated in the valley of the grain boundary, and the concentration was high. During heating, oxygen preferentially reacts with Ca, Mg and Al due to selective oxidation. Then, Si and Mn reacted with oxygen to form oxides, and complex oxides were eventually formed.
The surface of the carbon steel after pickling is shown in
Figure 4d, which also shows the morphology of the protruded grain and the depressed grain boundary, and the phase transformation of martensite as described above. Three characteristic locations were selected for component analysis, as identified in
Figure 4d. The chemical composition of the carbon steel surface was detected by WDS, N: 27.1 at %, Ti: 5.8 at %, Si: 1.0 at %, Mn: 1.2 at %, with the balance Fe at point 1. At the second point, S: 49.9 at %, Mn: 44.8 at %, with the balance Fe. At the third point, only Al and O were detected. The inclusions at the grain boundary were TiN, granular MnS, and Al
2O
3. At the temperature of 1200 °C, the standard free energy of TiN is the lowest among all metal nitrides. Thus, TiN were formed. A large number of small TiN inclusions were formed at the grain boundaries. During smelting, a certain amount of Mn was added to the steel in order to avoid the formation of a hot brittle phase of FeS. Thus, Mn and S combined at high temperature to form MnS, and was stable even at 1200 °C. Al
2O
3 was formed based on the rule of selective oxidation as described above.
The OM images of the composite interface of clad plates with two surface treatments after the hot rolling are shown in
Figure 8. It can be seen that the bonding interface was relatively straight, and excellent bonding was achieved without any unwelded zones after rolling.
The microstructure of the interface after brush grinding is presented in
Figure 8a,b. There were two kinds of inclusions with different morphologies at the composite interface. That is, the sporadic distribution of particles and the short rod-like segregation into the interfacial distribution along the band. Nomura et al. [
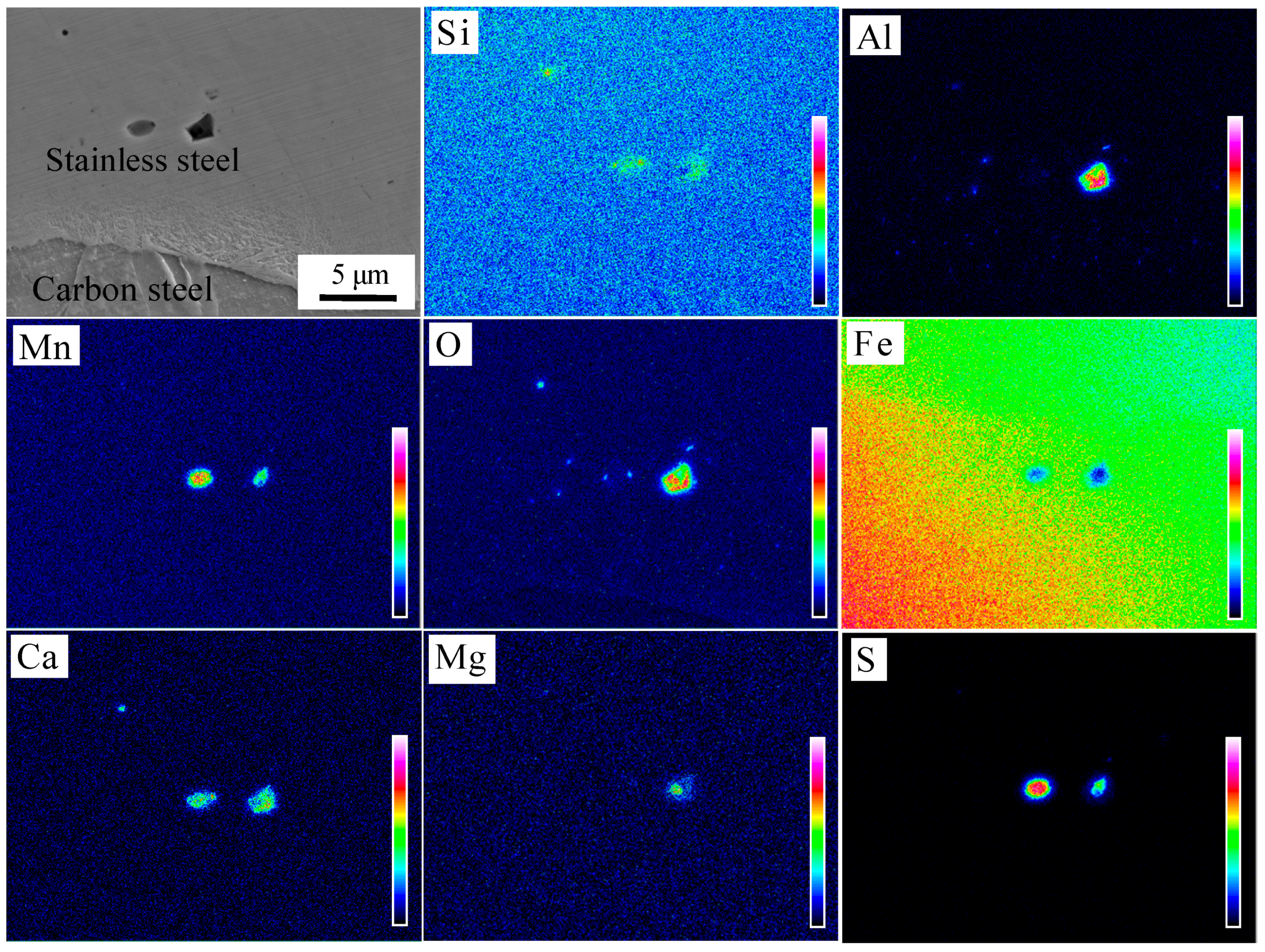
24] found that Si-Mn oxides were easily formed on the surface of steel containing Si and Mn, which are considered to be sensitive to oxidation. The machined carbon steel and stainless steel surface adsorbed O on the surface. These O atoms at the interface reacted preferentially with the easily oxidized elements, which were Si and Mn on both sides of the interface during prolonged heating and formed a thin layer of Si-Mn oxides. The oxidation layer was dispersed at the composite interface after rolling. The EPMA analysis of the composite interface is shown in
Figure 9. Elements Al, Si, Mn, and O were segregated near the interface. The segregation of Al element is less, and Al
2O
3 inclusions were preferentially generated based on the principle of selective oxidation. However, Si and Mn had a higher amount of segregation and reacted with the remaining O; Si-Mn oxides were finally formed.
The microstructure of the interface after pickling is shown in
Figure 8c,d. It can be seen that fewer interface inclusions were present, mainly in the form of granules or strips near the interface compared with the brush grinding. Three typical locations were selected for component analysis, as identified in
Figure 8c,d. The chemical composition of inclusions at the interface was detected by WDS, O: 18.8 at %, Al: 10.4 at %, Si: 0.7 at %, Cr: 8.2 at %, Mn: 1.5 at %, Ni: 1.6 at %, with the balance Fe at point 1; O: 21.2 at %, Al: 1.8 at %, Si: 4.1 at %, S: 17.8 at %, Ca: 1.9 at %, Cr: 7.6 at %, Mn: 18.8 at %, Ni: 2.2 at %, with the balance Fe at point 2; and O: 31.2 at %, Al: 6.0 at %, Si: 7.2 at %, Ca: 3.5 at %, Mg: 1.8 at %, Cr: 6.6 at %, Mn: 3.5 at %, Ni: 0.8 at %, with the balance Fe at point 3. The main types of inclusions were Al
2O
3, MnS, and Ca-Mg-Al-Si mixed oxide. The distribution and types of inclusions at the interface were analyzed by EPMA, as seen in
Figure 10. The mechanism of formation of MnS and Al
2O
3 inclusions has been described before, but the size decreased and the distribution became more diffuse under the effect of larger rolling force. The Ca-Mg-Al-Si composite oxide was derived from the small round oxide, when the 304 stainless steel was oxidized at high temperature. The types of inclusions were consistent with those formed in high temperature oxidation experiments, which indicated that the interfacial inclusions after hot rolling were all formed during the heating stage.
In general, continuous or aggregated Al2O3 and Si-Mn composite oxides formed at the composite interface after brush grinding. A small amount of oxides such as Al2O3, MnS, etc. were generated at the composite interface after pickling. The oxide inclusions after hot rolling were formed during the high-temperature oxidation stage. Therefore, the interface inclusions after pickling were small and less, and interface oxidation was greatly reduced.
3.2. Impact of Vacuum Degree on Interface Inclusions
Through the study for interface inclusions under different surface treatment methods, it was found that the interface inclusions of clad plates were less after pickling. The surface treatment by pickling was used to study the formation of oxidations at the composite interface at high temperature with different degrees of vacuum.
Figure 11 shows the microstructure of a cross section of interface after high-temperature oxidation at different degrees of vacuum.
Figure 11a,d are micrographs at vacuum of 10
−2 Pa. It can be seen that the surface layer of carbon steel was oxidized, and many fine granular inclusions were dispersed on the surface. It was only enriched with Al and O based on WDS analysis (except matrix elements), and the atomic ratio of Al to O was 2:3. Thus, the inclusions were Al
2O
3. The cross section of stainless steel was clean and clear without oxide.
Figure 11b,e are images at a vacuum level of 1 Pa. Carbon steel surface layers also undergo oxidation. As the vacuum level decreased, the content of oxygen increased, and the number, density and the size of oxide inclusions were all increased. Al
2O
3 and Si-Mn composite oxides were detected.
The cross section of stainless steel was relatively clean, but there were a certain number of holes in the localized area, which were formed on the stainless steel surface after pickling and could not be completely eliminated during heating at a high temperature. Thus, a large amount of oxygen is trapped here, which easily reacted with the active metals in the stainless steel to form oxides.
Figure 11c,f are images at vacuum of 10
5 Pa. With the decrease in vacuum, the oxide content clearly increased, and an oxide layer and larger oxides were formed on the carbon steel side near the interface. At high-temperature and high-oxygen content, Fe was oxidized rapidly to form an oxide layer. Because sufficient oxygen was enriched on the surface, Al
2O
3 and Si-Mn composite oxides were sequentially generated based on the preferential selective oxidation mechanism.
Figure 11f shows that oxides were also formed on the side of the stainless steel, which contained high content of Si, Mn, Cr, Fe according to WDS analysis, and eventually the Si-Mn-Cr-Fe composite oxides were formed. When oxidation occurred, Si-Mn composite oxide is first formed based on the principle of selective oxidation, when elements of Si and Mn are depleted, the remaining O reacts with Cr and Fe.
The microstructure of high-temperature oxidation of stainless steel and carbon steel at different vacuum degree are shown in
Figure 12.
Micrographs of carbon steel and stainless steel corresponding to the vacuum of 10
−2 Pa are shown in
Figure 12a,d. The surface inclusions of carbon steel were mainly TiN, MnS and Al
2O
3 after pickling in the high-temperature oxidation experiments. A small amount of Ca-Mg-Al-Si composite oxides were formed on the surface of stainless steel according to high temperature oxidation experiments. The forming mechanism of surface inclusions has been discussed in
Section 3.1.
Morphologies of the samples with a vacuum of 1 Pa are shown in
Figure 12b,e. It can be seen that the surface of carbon steel was similar to that in the high vacuum, there was no large-scale oxidation, and only a small amount of oxide was formed. O: 21.5 at %, Al: 4.7 at %, Si: 5.6 at %, Mn: 4.6 at % and the balance of Fe were detected by WDS. Al
2O
3 and Si-Mn composite oxides were detected and distributed on the surface because of selective oxidation. With the decrease in vacuum, the content of oxides was increased on the surface of the depressions for stainless steel, and larger Al-Si-Mn oxides were formed. When the vacuum was 1 Pa, the interface was in a low-vacuum sealed state. Although the content of oxygen at the lower vacuum was higher than that under the higher vacuum, and the initial oxygen partial pressure was also high, the total oxygen content was limited. With the oxidation reaction, oxygen was gradually consumed until the partial pressure of oxygen was reduced to a level such that oxide was not formed. Thus, the composite interface did not undergo severe oxidation at lower vacuum, but was similar to the degree of reaction at high vacuum and a had a slightly higher degree of oxidation. The result was an increase in oxide production compared to the high vacuum [
25].
Morphologies of samples with the vacuum of 10
5 Pa are shown in
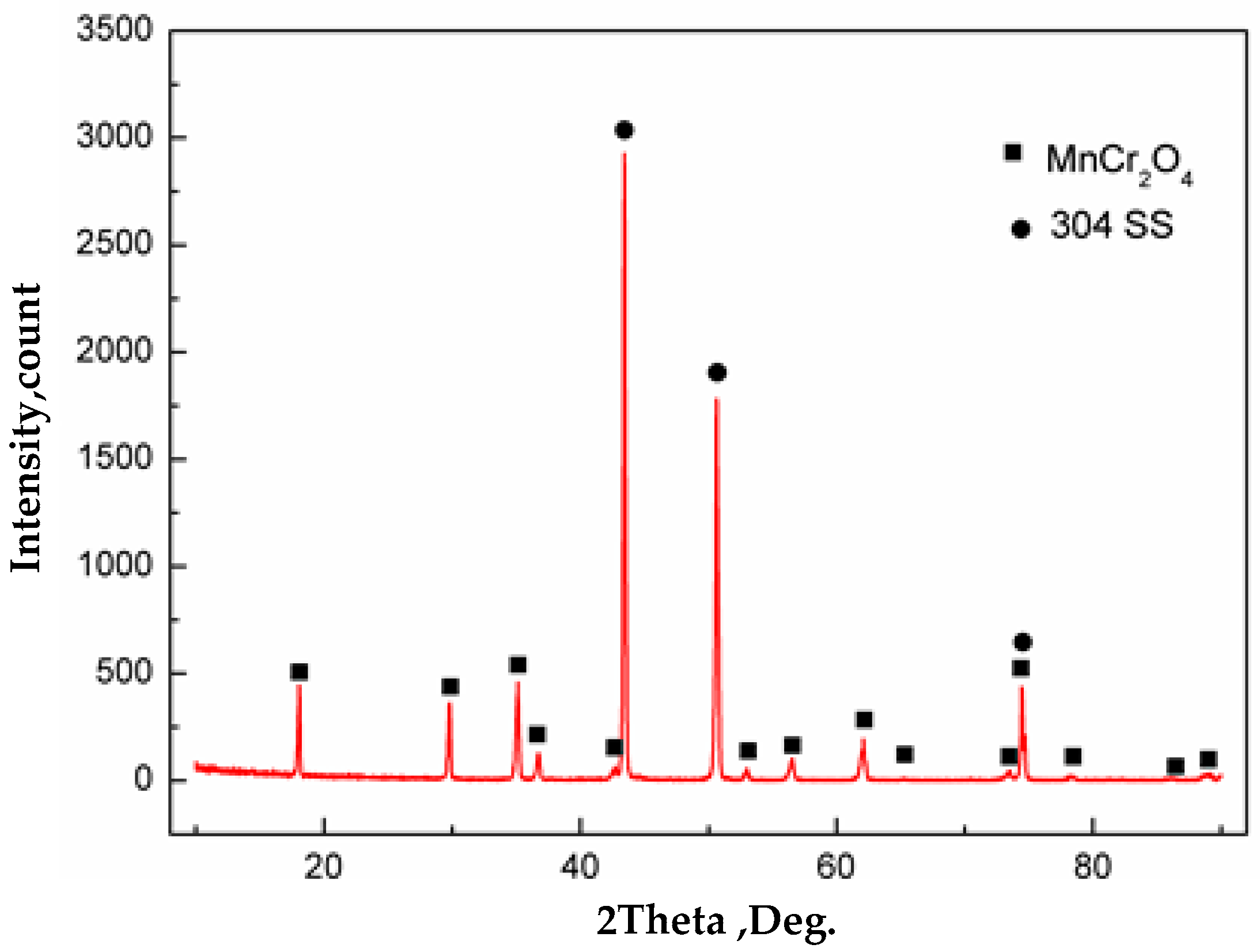
Figure 12c,f. It can be seen that the surface of carbon steel and stainless steel were completely covered with oxides. The chemical composition of carbon steel surface was detected by WDS, which were O: 37.3 at %, Si: 0.8 at %, and the balance was Fe. Thus, oxides were formed on the surface of carbon steel. Severe oxidation of iron and oxygen occurred at 1200 °C. The chemical composition of stainless steel surface was O: 51.5 at %, Cr: 24.0 at %, Mn: 12.6 at % and the balance of Fe, as detected by WDS. Also, based on the XRD analysis of the surface of stainless steel as shown in
Figure 13, almost all the inclusions on the surface of stainless steel were MnCr
2O
4. The composite interface was in contact with the outside atmosphere at vacuum of 10
5 Pa. During subsequent heating, the stainless steel was severely oxidized. Due to the high content of Cr in stainless steel, a layer of Cr
2O
3 protective film was first formed on the surface of stainless steel at high-temperature and high-oxygen conditions according to the selective oxidation mechanism. With the progression of the oxidation, Cr
2O
3 was continuously generated and covered the entire stainless steel surface, which isolated the stainless steel matrix from the atmosphere. If Cr continues to oxidize on the composite surface, alloying elements must diffuse through the Cr
2O
3 layer to the surface and react with oxygen. Meanwhile, the lattice diffusion coefficient of Mn ions in Cr
2O
3 was two orders of magnitude higher than that of Cr ions, and its diffusion rate was faster than Si, Fe and Ni in stainless steel [
26]. Therefore, Mn in the stainless steel matrix is the first to diffuse to the surface of Cr
2O
3 and reacts with oxygen to form MnO. When Cr
2O
3 and MnO are present on the surface of stainless steel, MnCr
2O
4 spinel oxides are formed [
27,
28].
Thus, when the vacuum was reduced, the degree of interfacial oxidation became more serious, which was unfavorable to obtaining the excellent interface of the hot-rolled clad plate. Therefore, the degree of vacuum at the composite interface should be controlled between 1 Pa and10−2 Pa, or less.