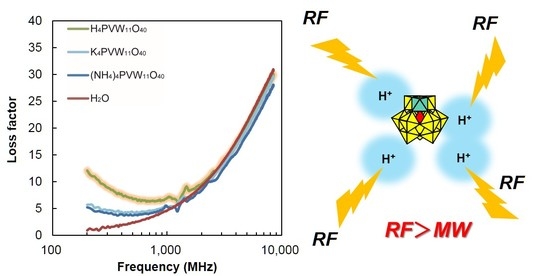
Proton-Enhanced Dielectric Properties of Polyoxometalates in Water under Radio-Frequency Electromagnetic Waves
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Dielectric Relaxation Spectroscopy
3. Results and Discussion
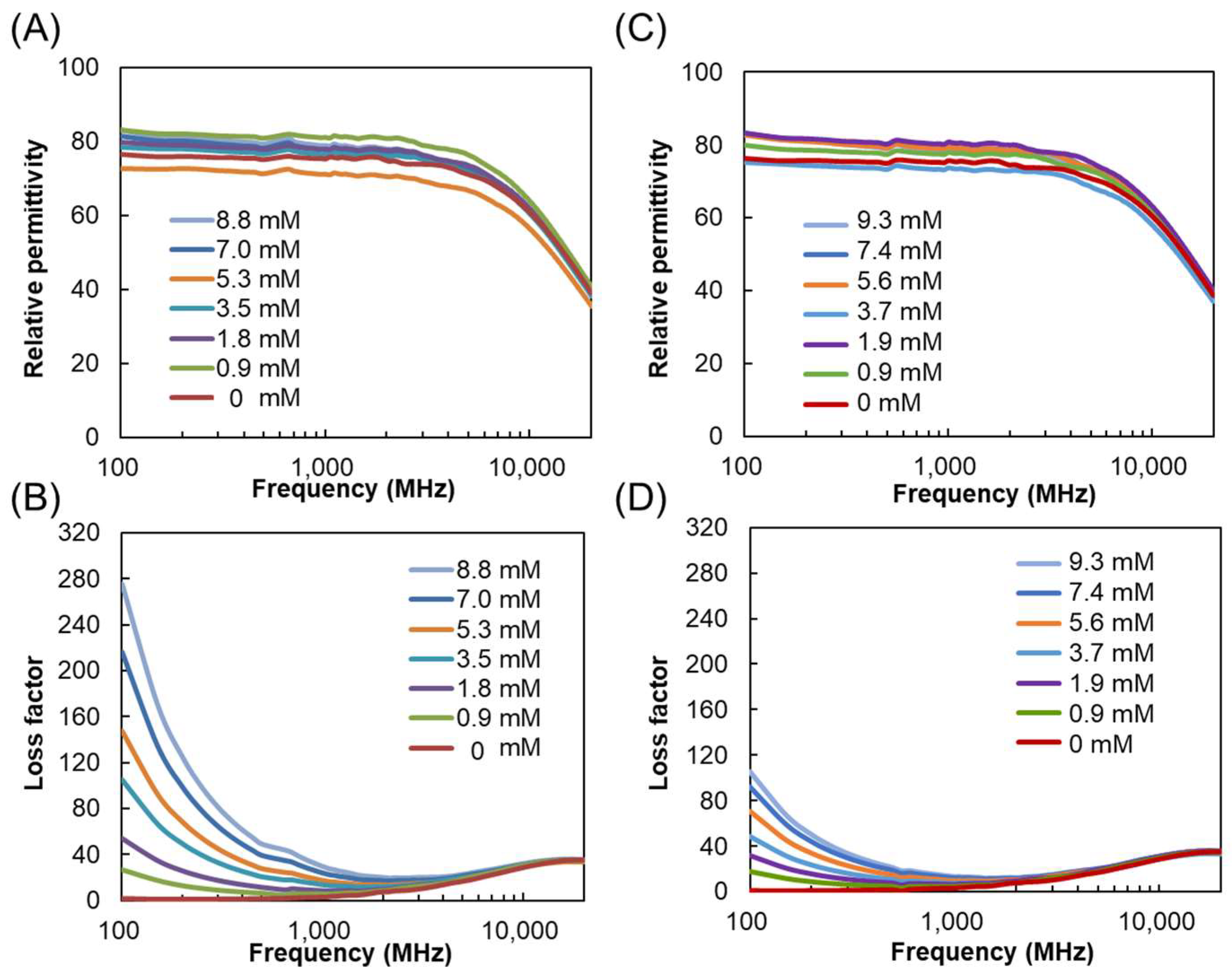
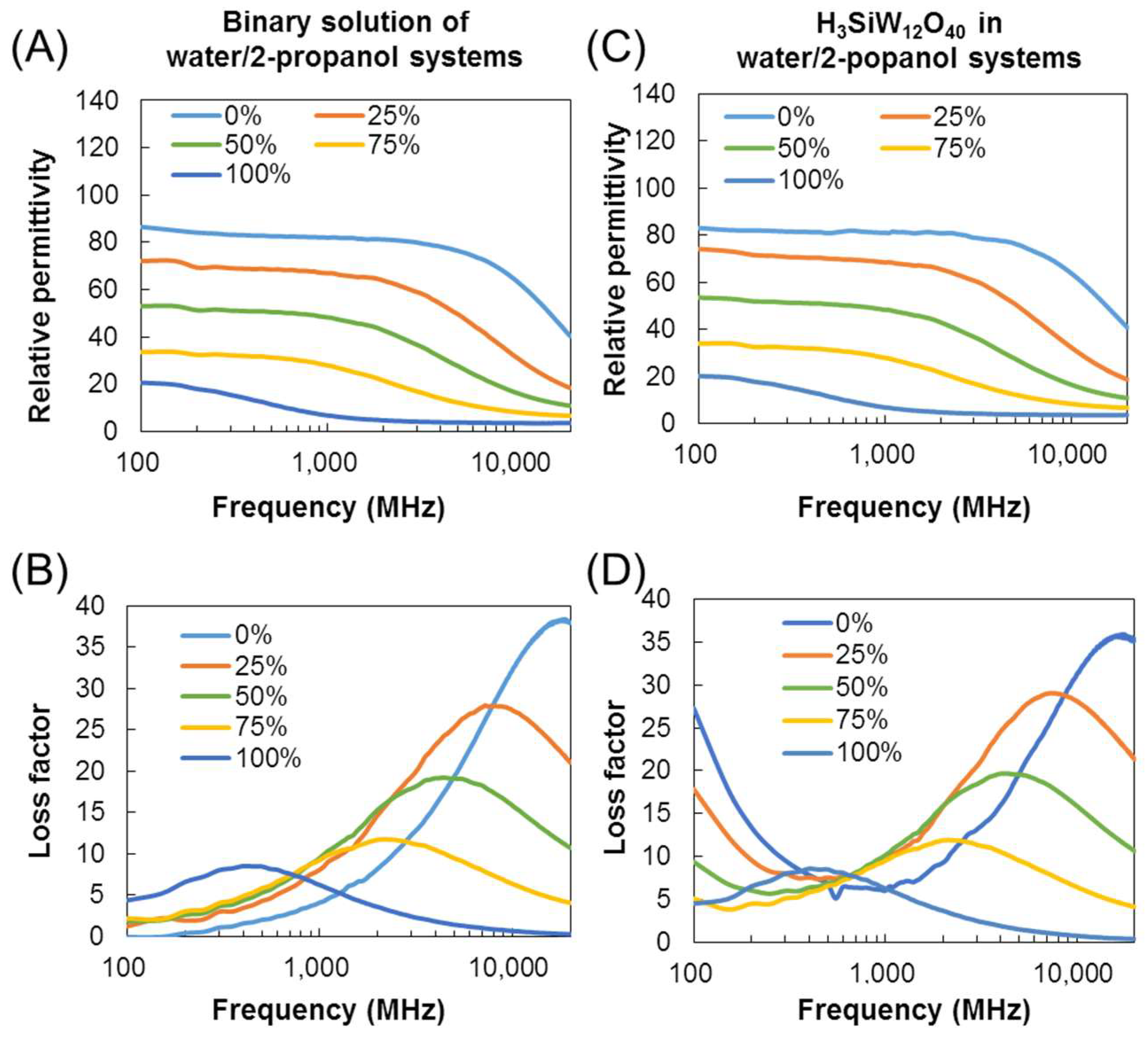
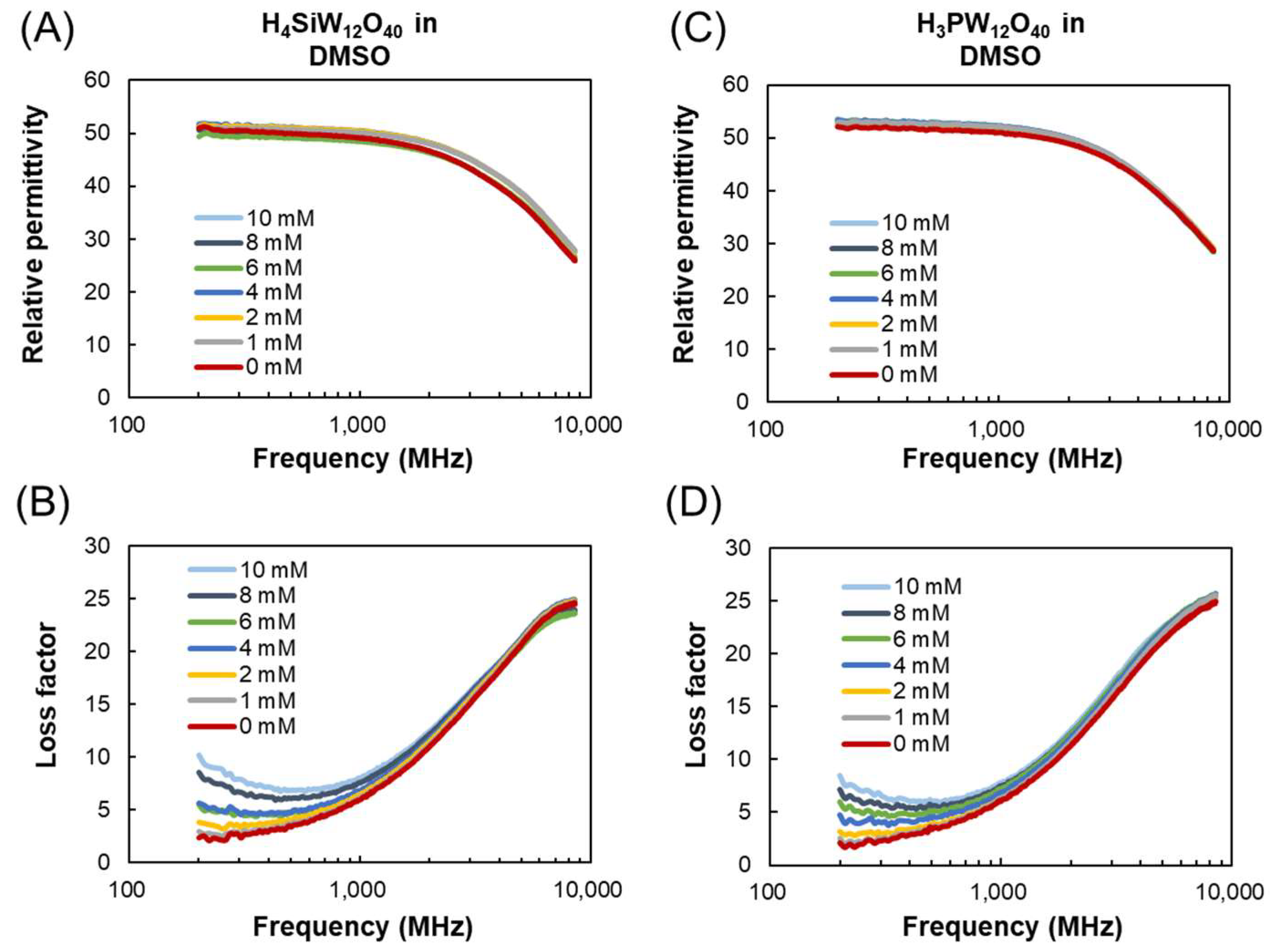
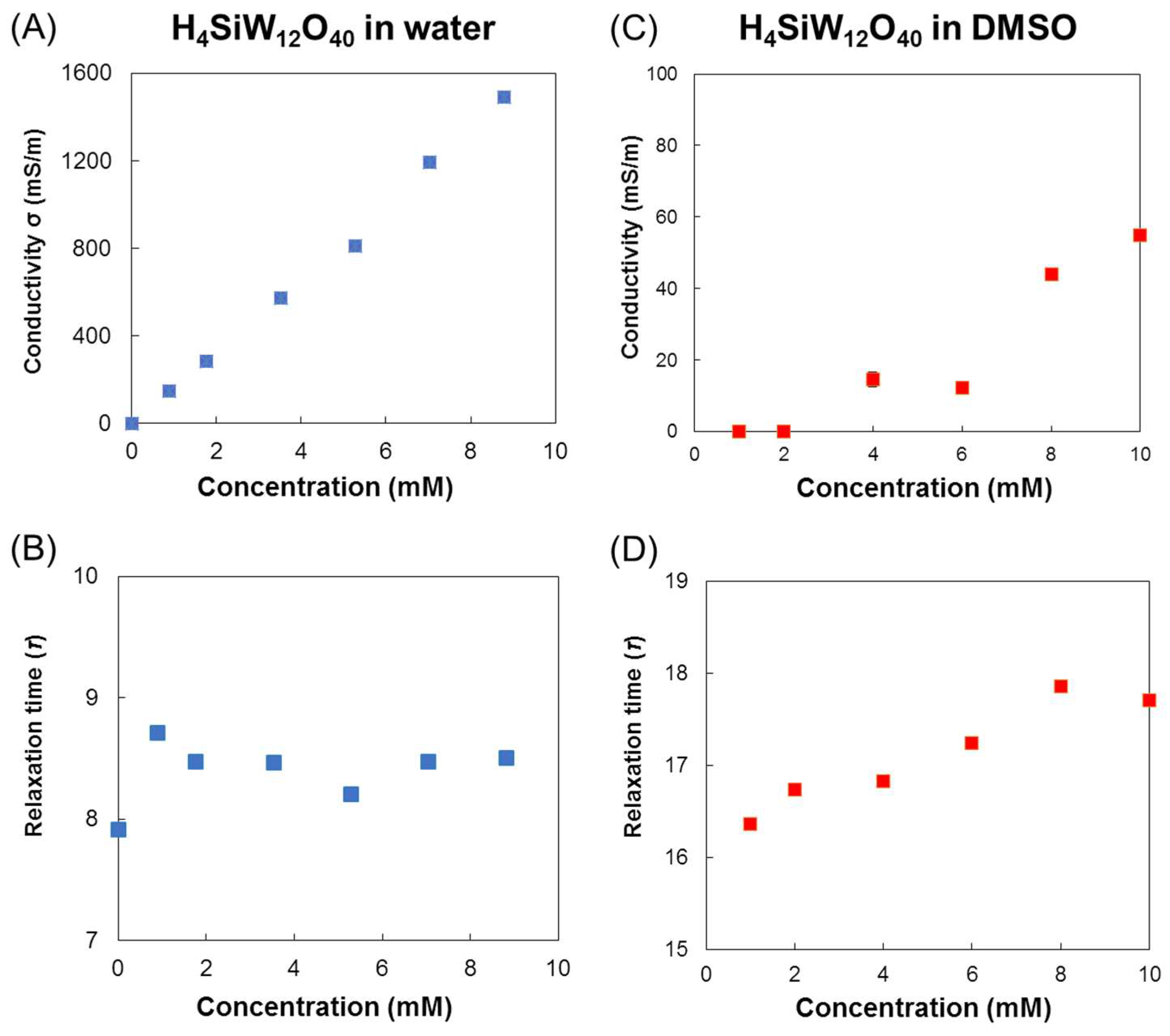
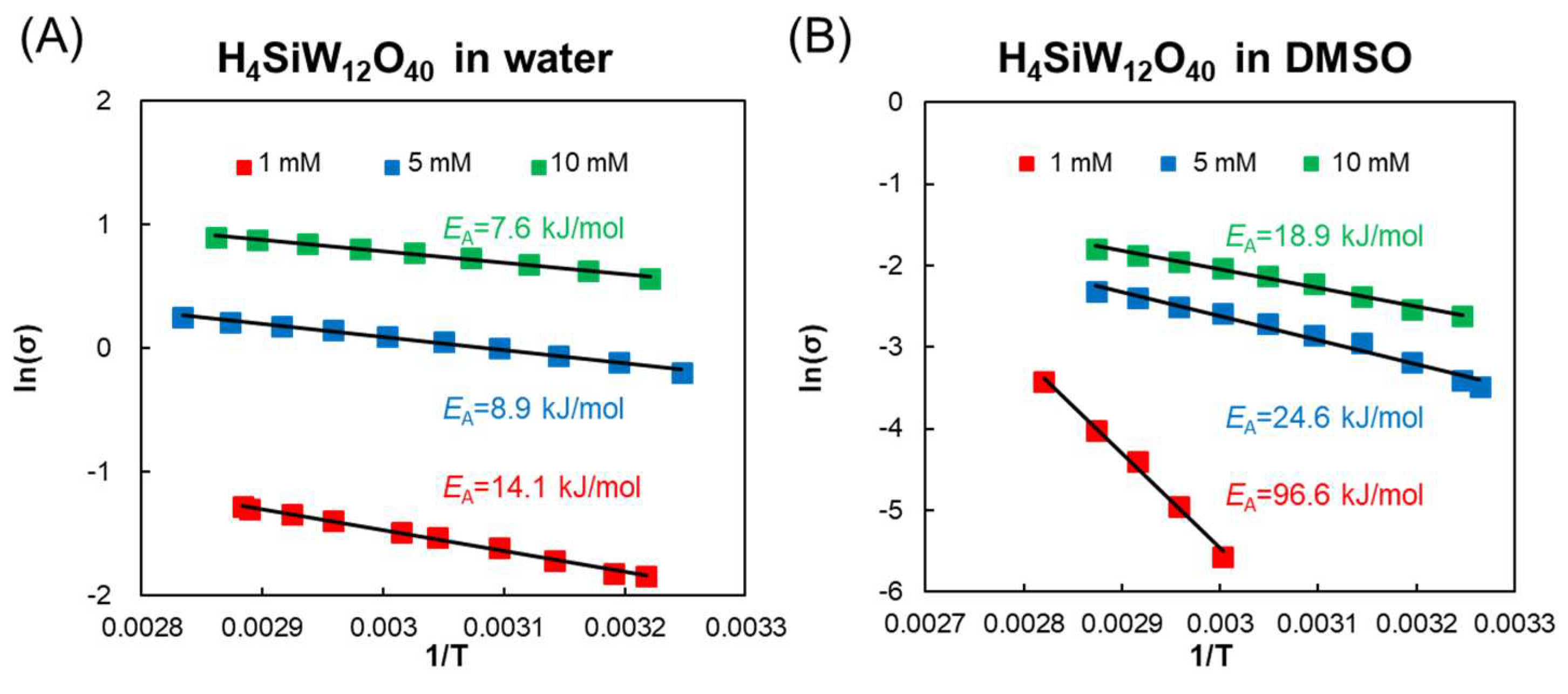
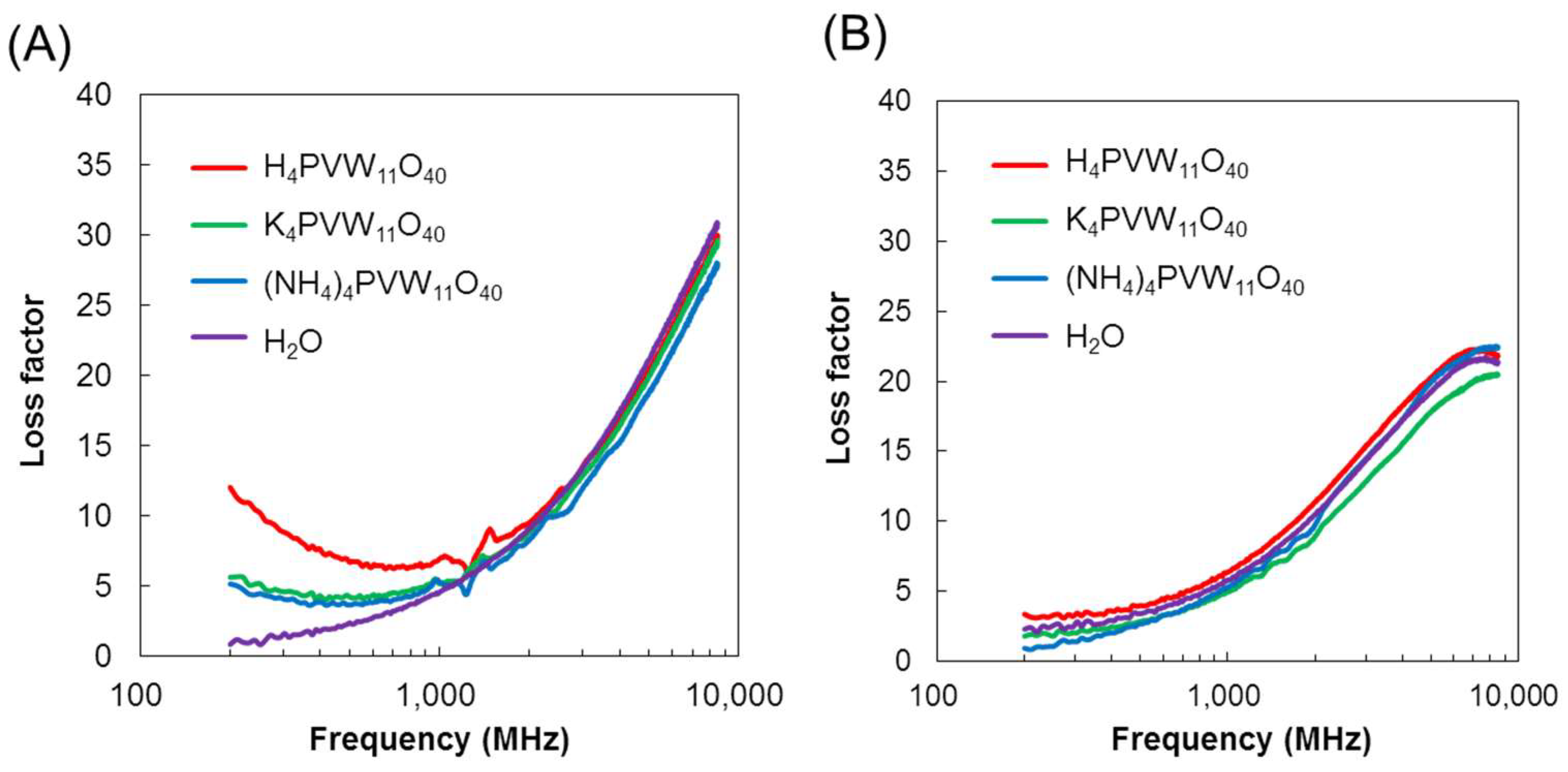
3.1. Dielectric Properties of POMs in Water, 2-Propanol, and DMSO
3.2. Mechanism of Interaction of POMs and Electromagnetic Waves in Water
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kozhevnikov, I.V. Catalysis by heteropoly acids and multicomponent polyoxometalates in liquid-phase reactions. Chem. Rev. 1998, 98, 171–198. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-S.; Yang, G.-Y. Recent advances in polyoxometalate-catalyzed reactions. Chem. Rev. 2015, 115, 4893–4962. [Google Scholar] [CrossRef] [PubMed]
- Shimidzu, K.I.; Furukawa, H.; Kobayashi, N.; Itaya, Y.; Satsuma, A. Effects of Brønsted and Lewis acidities on activity and selectivity of heteropolyacid-based catalysts for hydrolysis of cellobiose and cellulose. Green Chem. 2009, 11, 1627–1632. [Google Scholar] [CrossRef]
- Tian, J.; Wang, J.; Zhap, S.; Jiang, C.; Zhang, X.; Wang, X. Hydrolysis of cellulose by the heteropoly acid H3PW12O40. Cellulose 2010, 17, 587–594. [Google Scholar] [CrossRef]
- Weinstcok, I.A.; Barbuzzi, E.M.G.; Wemple, M.W.; Cowan, J.J.; Reiner, R.S.; Sonnen, D.M.; Heintz, R.A.; Bonda, J.S.; Hill, C.L. Equilibrating metal-oxide cluster ensembles for oxidation reactions using oxygen in water. Nature 2001, 414, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Bikash, B.; Neumann, R. Polyoxometalate-mediated electron transfer–oxygen transfer oxidation of cellulose and hemicellulose to synthesis gas. Nat. Commun. 2014, 5, 4621. [Google Scholar] [CrossRef]
- Reichert, J.; Brunner, B.; Jess, A.; Wasserscheid, P.; Albert, J. Biomass oxidation to formic acid in aqueous media using polyoxometalate catalysts–boosting FA selectivity by in-situ extraction. Energy Environ. Sci. 2015, 8, 2985–2990. [Google Scholar] [CrossRef]
- Liu, W.; Cui, Y.; Du, X.; Zhang, Z.; Chao, Z.; Deng, Y. High efficiency hydrogen evolution from native biomass electrolysis. Energy Environ. Sci. 2016, 9, 467–472. [Google Scholar] [CrossRef]
- Tsubaki, S.; Oono, K.; Ueda, T.; Onda, A.; Yanagisawa, K.; Mitani, T.; Azuma, J. Microwave-assisted hydrolysis of polysaccharides over polyoxometalate clusters. Bioresour. Technol. 2013, 144, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Tsubaki, S.; Oono, K.; Hiraoka, M.; Ueda, T.; Onda, A.; Yanagisawa, K.; Azuma, J. Hydrolysis of green-tide forming Ulva spp. by microwave irradiation with polyoxometalate cluster. Green Chem. 2014, 16, 2227–2233. [Google Scholar] [CrossRef]
- Tsubaki, S.; Oono, K.; Onda, A.; Ueda, T.; Mitani, T.; Hiraoka, M. Microwave-assisted hydrolysis of biomass over activated carbon supported polyoxometalates. RSC Adv. 2017, 7, 12346–12350. [Google Scholar] [CrossRef]
- De la Hoz, A.; Díaz-Ortiz, Á.; Moreno, A. Microwaves in organic synthesis. Thermal and non-thermal microwave effects. Chem. Soc. Rev. 2005, 34, 164–178. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.J.; Vaidhyanathan, B.; Ganguli, M.; Ramakrishnan, P.A. Synthesis of inorganic solids using microwaves. Chem. Mater. 1999, 11, 882–895. [Google Scholar] [CrossRef]
- Bilecka, I.; Niederberger, M. Microwave chemistry for inorganic nanomaterials synthesis. Nanoscale 2010, 2, 1358–1374. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Agrawal, D.; Cheng, J.; Gedevanishvili, S. Full sintering of powdered-metal bodies in a microwave field. Nature 1999, 399, 668–670. [Google Scholar] [CrossRef]
- Chandrasekaran, S.; Ramanatha, S.; Basak, T. Microwave food processing—A review. Food Res. Int. 2013, 52, 243–261. [Google Scholar] [CrossRef]
- Tsubaki, S.; Azuma, J.I.; Yoshimura, T.; Maitani, M.M.; Suzuki, E.; Fujii, S.; Wada, Y. Microwave-Driven Biomass Fractionation. In Biomass Fractionation Technologies for a Lignocellulosic Feedstock Based Biorefinery; Mussatto, S., Ed.; Elsevier, B.V.: Amsterdam, The Netherlands, 2016; pp. 103–126. ISBN 978-0-12-802323-5. [Google Scholar]
- Kumar, V.B.; Pulidindi, I.N.; Gedanken, A. Glucose production from potato peel waste under microwave irradiation. J. Mol. Catal. A Chem. 2016, 417, 163–167. [Google Scholar] [CrossRef]
- Bosco, M.; Rat, S.; Dupré, N.; Hasenknopf, B.; Lacôte, E.; Malacria, M.; Rémy, P.; Kovensky, J.; Thorimbert, S.; Wadouachi, A. Lewis-acidic polyoxometalates as reusable catalysts for the synthesis of glucuronic acid esters under microwave irradiation. ChemSusChem 2010, 3, 1249–1252. [Google Scholar] [CrossRef] [PubMed]
- Bonchio, M.; Carraro, M.; Sartorel, A.; Scorrano, G.; Kortz, U. Bio-inspired oxidations with polyoxometalate catalysts. J. Mol. Catal. A Chem. 2006, 251, 93–99. [Google Scholar] [CrossRef]
- Lu, L.; Li, L.; Hu, T.; Zhang, W.; Huang, X.; Zhang, J.; Liu, X. Preparation, characterization, and photocatalytic activity of three-dimensionally ordered macroporous hybrid monosubstituted polyoxometalate K5[Co(H2O)PW11O39] amine functionalized titanium catalysts. J. Mol. Catal. A Chem. 2014, 394, 283–294. [Google Scholar] [CrossRef]
- Ritchie, C.; Bryant, G. Microwave assisted synthesis of a mono organoimido functionalized Anderson polyoxometalate. Dalton Trans. 2015, 44, 20826–20829. [Google Scholar] [CrossRef] [PubMed]
- Spillane, S.; Sharma, R.; Zavras, A.; Mulder, R.; Ohlin, C.A.; Goerigk, L.; O’Hair, R.A.J.; Ritchie, C. Non-aqueous microwave-assisted syntheses of deca- and hexa-molybdovanadates. Angew. Chem. Int. Ed. 2017, 56, 8568–8572. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.-M.; Liang, S.; Yu, W.-D.; Yuan, H.; Yan, J. Microwave-assisted preparation and characterization of a polyoxometalate-based inorganic 2D framework anode for enhancing lithium-ion battery performance. Chem. Asian J. 2018, 13, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Mingos, D.M.P.; Baghurst, D. Applications of microwave dielectric heating effects to synthetic problems in chemistry. Chem. Soc. Rev. 1991, 20, 1–47. [Google Scholar] [CrossRef]
- Gabriel, C.; Gabriel, S.; Grant, E.H.; Halstead, B.S.J.; Mingos, D.M.P. Dielectric parameters relevant to microwave dielectric heating. Chem. Soc. Rev. 1998, 27, 213–223. [Google Scholar] [CrossRef]
- Buchner, R.; Hefter, G. Interaction and dynamics in electrolyte solutions by dielectric spectroscopy. Phys. Chem. Chem. Phys. 2009, 11, 8984–8999. [Google Scholar] [CrossRef] [PubMed]
- Ueda, T.; Yamasita, K.; Onda, A. New extraction procedure for protonated polyoxometalates prepared in aqueous-organic solution and characterisation of their catalytic ability. Appl. Catal. A Gen. 2014, 485, 181–187. [Google Scholar] [CrossRef]
- Fukasawa, T.; Sato, T.; Watanabe, J.; Hama, Y.; Kunz, W.; Buchner, R. Relation between dielectric and low-frequency Raman spectra of hydrogen-bond liquids. Phys. Rev. Lett. 2005, 95, 197802. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Chiba, A.; Nozaki, R. Dielectric relaxation mechanism and dynamical structures of the alcohol/water mixtures. J. Mol. Liq. 2002, 101, 99–111. [Google Scholar] [CrossRef]
- Sato, T.; Buchner, R. Dielectric relaxation spectroscopy of 2-propanol–water mixtures. J. Chem. Phys. 2003, 118, 4606–4613. [Google Scholar] [CrossRef]
- Kolthoff, I.M. Acid-base equilibria in dipolar aprotic solvents. Anal. Chem. 1974, 46, 1992–2003. [Google Scholar] [CrossRef]
- Khenkin, A.M.; Neumann, R. Oxygen transfer from sulfoxides: Selective oxidation of alcohols catalyzed by polyoxomolybdates. J. Org. Chem. 2002, 67, 7075–7079. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, K.; Wu, L. Electrical transport properties and interaction of heteropolyacid in N,N-dimethylformamide by dielectric spectroscopy. J. Chem. Phys. 2014, 141, 599–602. [Google Scholar] [CrossRef] [PubMed]
- Horky, A.; Kherani, N.P.; Xu, G. Conductivity and transport properties of aqueous phosphotungstic and silicotungstic acid electrolytes for room-temperature fuel cell. J. Electrochem. Soc. 2003, 150, A1219–A1224. [Google Scholar] [CrossRef]
- Tsubaki, S.; Hiraoka, M.; Hadano, S.; Nishimura, H.; Kashimura, K.; Mitani, T. Functional group dependent dielectric properties of sulfated hydrocolloids extracted from green macroalgal biomass. Carbohydr. Polym. 2014, 107, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Tsubaki, S.; Hiraoka, M.; Hadano, S.; Okamura, K.; Ueda, T.; Nishimura, H.; Kashimura, K.; Mitani, T. Effects of acidic functional groups on dielectric properties of sodium alginates and carrageenans in water. Carbohydr. Polym. 2015, 115, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Sato, M. Microwave heating of water, ice, and saline solution: Molecular dynamics study. J. Chem. Phys. 2007, 126, 034509. [Google Scholar] [CrossRef] [PubMed]
- Tsubaki, S.; Oono, K.; Hiraoka, M.; Onda, A.; Mitani, T. Microwave-assisted hydrothermal extraction of sulfated polysaccharides from Ulva spp. and Monostroma latissimum. Food Chem. 2016, 210, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Tsubaki, S.; Oono, K.; Onda, A.; Yanagisawa, K.; Mitani, T.; Azuma, J. Effects of ionic conduction on hydrothermal hydrolysis of corn starch and crystalline cellulose induced by microwave irradiation. Carbohydr. Polym. 2016, 137, 594–599. [Google Scholar] [CrossRef] [PubMed]

| POM | Hydration Number |
|---|---|
| H4SiW12O40 | 28 |
| H3PW12O40 | 25 |
| H3PMo12O40 | 28 |
| H3AsW12O40 | 10 |
| H4PVW11O40 | 10 |
| H4S2W18O62 | 27 |
| (NH4)4PVW11O40 | 9 |
| K4PVW11O40 | 4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsubaki, S.; Hayakawa, S.; Ueda, T.; Mitani, T.; Suzuki, E.-i.; Fujii, S.; Wada, Y. Proton-Enhanced Dielectric Properties of Polyoxometalates in Water under Radio-Frequency Electromagnetic Waves. Materials 2018, 11, 1202. https://doi.org/10.3390/ma11071202
Tsubaki S, Hayakawa S, Ueda T, Mitani T, Suzuki E-i, Fujii S, Wada Y. Proton-Enhanced Dielectric Properties of Polyoxometalates in Water under Radio-Frequency Electromagnetic Waves. Materials. 2018; 11(7):1202. https://doi.org/10.3390/ma11071202
Chicago/Turabian StyleTsubaki, Shuntaro, Shogo Hayakawa, Tadaharu Ueda, Tomohiko Mitani, Ei-ichi Suzuki, Satoshi Fujii, and Yuji Wada. 2018. "Proton-Enhanced Dielectric Properties of Polyoxometalates in Water under Radio-Frequency Electromagnetic Waves" Materials 11, no. 7: 1202. https://doi.org/10.3390/ma11071202
APA StyleTsubaki, S., Hayakawa, S., Ueda, T., Mitani, T., Suzuki, E.-i., Fujii, S., & Wada, Y. (2018). Proton-Enhanced Dielectric Properties of Polyoxometalates in Water under Radio-Frequency Electromagnetic Waves. Materials, 11(7), 1202. https://doi.org/10.3390/ma11071202