Magnetron-Sputtered, Biodegradable FeMn Foils: The Influence of Manganese Content on Microstructure, Mechanical, Corrosion, and Magnetic Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Microstructure
2.2.1. X-ray Diffraction
2.2.2. Scanning Electron Microscopy/Energy-Dispersive X-ray Spectroscopy
2.3. Corrosion
2.3.1. Electrochemical Polarization Measurements
2.3.2. Immersion Tests
2.4. Mechanical Properties
2.5. Magnetic Properties
3. Results
3.1. Microstructure
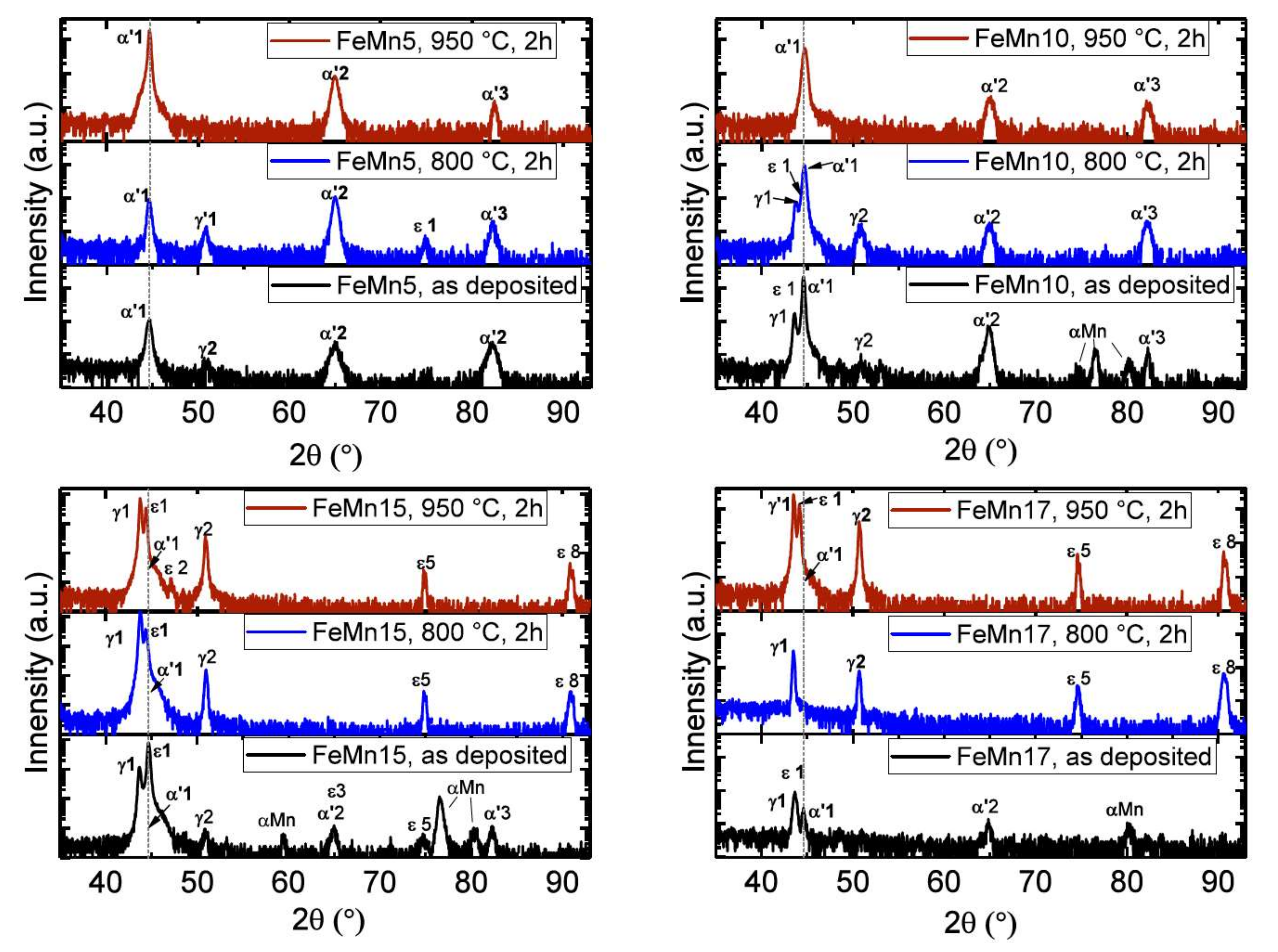
3.1.1. X-ray Diffraction
3.1.2. Scanning Electron Microscopy
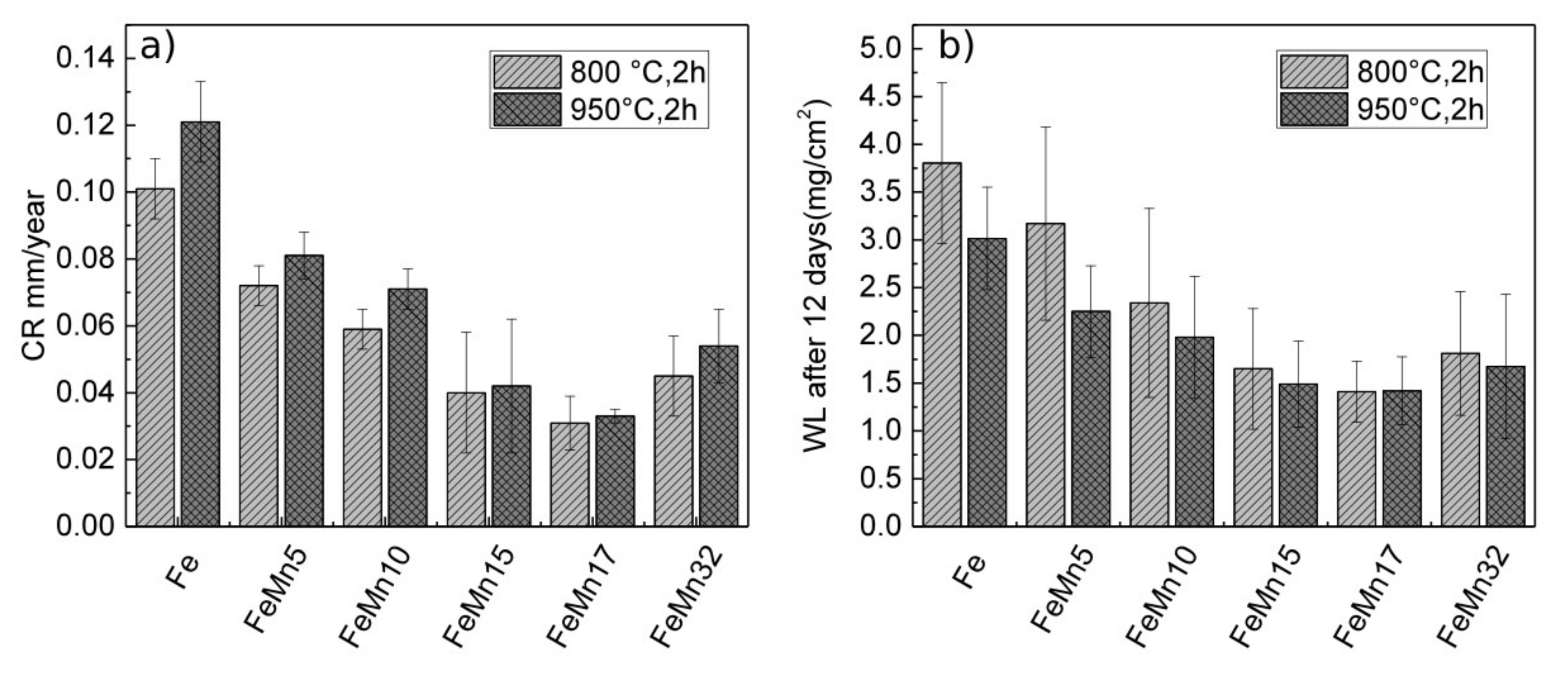
3.2. Corrosion
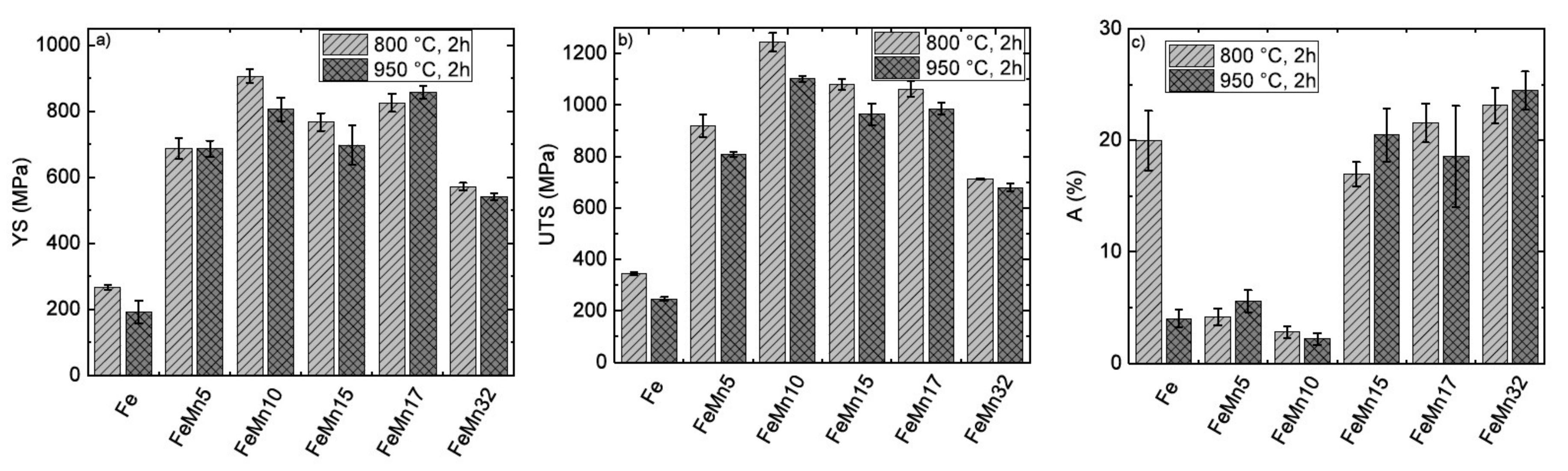
3.3. Mechanical Properties
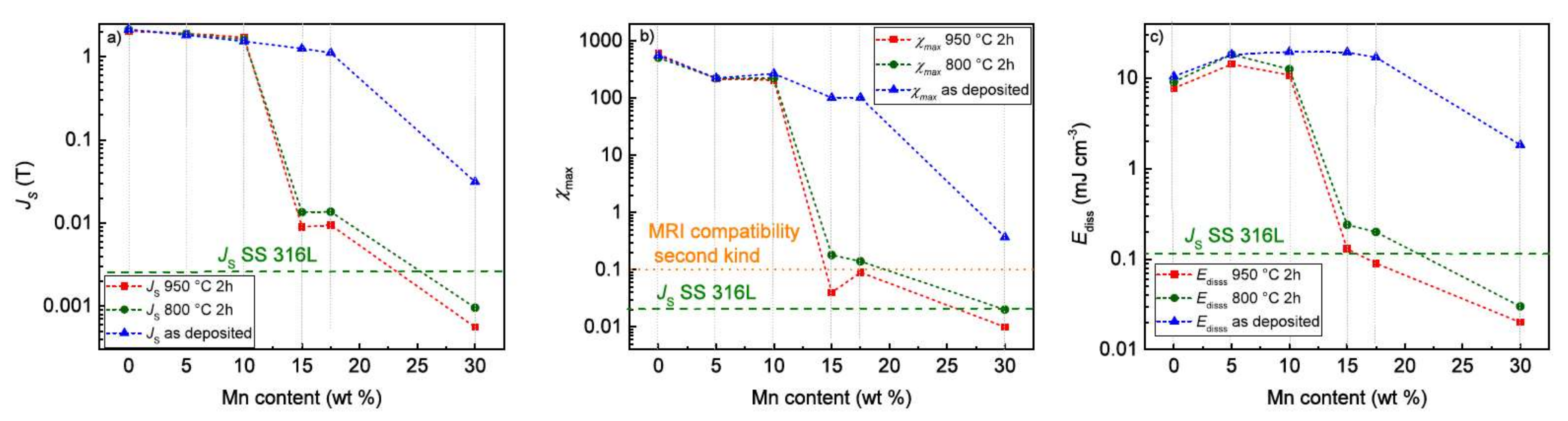
3.4. Magnetic Properties
4. Discussion
4.1. Microstructure
4.2. Mechanical Properties
4.3. Corrosion
4.4. Magnetic Properties
5. Conclusions
- FeMn5: Showed a decrease of the ferromagnetic behavior and a decrease of the CR compared to pure Fe—a high strength, but a rather low ductility
- FeMn10: Further decrease of the ferromagnetic behavior and a further decrease of CR compared to FeMn5—an increase of strength, but lower ductility.
- FeMn15: Marked drop of ferromagnetic characteristics and a slightly decreased CR compared to FeMn10—lower but still high strength, with enhanced ductility.
- FeMn17: Similar magnetic and corrosion behavior to FeMn15, with a higher YS but similar UTS and ductility, compared to FeMn15.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Peuster, M.; Wohlsein, P.; Brügmann, M.; Ehlerding, M.; Seidler, K.; Fink, C.; Brauer, H.; Fischer, A.; Hausdorf, G. A novel approach to temporary stenting: Degradable cardiovascular stents produced from corrodible metal-results 6–18 months afterimplantation into New Zealand white rabbits. Heart 2001, 86, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Peuster, M.; Hesse, C.; Schloo, T.; Fink, C.; Beerbaum, P.; von Schnakenburg, C. Long-term biocompatibility of a corrodible peripheral iron stent in the porcine descending aorta. Biomaterials 2006, 27, 4955–4962. [Google Scholar] [CrossRef] [PubMed]
- Schenck, J.F. The role of magnetic susceptibility in magnetic resonance imaging: MRI magnetic compatibility of the first and second kinds. Med. Phys. 1996, 23, 815–850. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, A.B.; Sulsky, S.I. Risk assessment of an essential element: Manganese. J. Toxicol. Environ. Health A 2010, 73, 128–155. [Google Scholar] [CrossRef] [PubMed]
- Lide, D. Handbook of Chemistry and Physics-crc, 90th ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Grässel, O.; Krüger, L.; Frommeyer, G.; Meyer, L. High strength Fe-Mn-(Al, Si) TRIP/TWIP steelsdevelopment—Properties—Application. Int. J. Plast. 2000, 16, 1391–1409. [Google Scholar] [CrossRef]
- Schumann, H. Die martensitischen Umwandlungen in kohlenstoffarmen Manganstählen. Arch. Eisenhüttenwes 1967, 38, 647–656. [Google Scholar] [CrossRef]
- Endoh, Y.; Ishikawa, Y. Antiferromagnetism of Iron Manganes Alloys. J. Phys. Soc. Jpn. 1971, 30, 1614–1627. [Google Scholar] [CrossRef]
- Rabinkin, A. On magnetic contributions to γ → ε phase transformations in Fe-Mn alloys. Calphad 1979, 3, 77–84. [Google Scholar] [CrossRef]
- Hermawan, H.; Dubé, D.; Mantovani, D. Degradable metallic biomaterials: Design and development of Fe-Mn alloys for stents. J. Biomed. Mater. Res. A 2010, 93, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, H.; Alamdari, H.; Mantovani, D.; Dubé, D. Iron–manganese. Powder Met. 2013, 51, 38–45. [Google Scholar] [CrossRef]
- Jurgeleit, T.; Quandt, E.; Zamponi, C. Magnetron Sputtering a New Fabrication Method of Iron Based Biodegradable Implant Materials. Adv. Mater. Sci. Eng. 2015, 2015, 294686. [Google Scholar] [CrossRef]
- Zamponi, C.; Schürmann, U.; Jurgeleit, T.; Kienle, L.; Quandt, E. Microstructures of magnetron sputtered FeAu thin films. IJMR 2015, 106, 103–107. [Google Scholar] [CrossRef]
- Jurgeleit, T.; Quandt, E.; Zamponi, C. Mechanical Properties and In Vitro Degradation of SputteredBiodegradable Fe-Au Foils. Materials 2016, 9, 928. [Google Scholar] [CrossRef] [PubMed]
- Jurgeleit, T.; Quandt, E.; Zamponi, C. Magnetron Sputtering as a Fabrication Method for a Biodegradable Fe32Mn Alloy. Materials 2017, 10, 1196. [Google Scholar] [CrossRef] [PubMed]
- Zamponi, C.; Rumpf, H.; Schmutz, C.; Quandt, E. Structuring of sputtered superelastic NiTi thin films byphotolithography and etching. Mater. Sci. Eng. A 2008, 481–482, 623–625. [Google Scholar] [CrossRef]
- De Miranda, R.L.; Zamponi, C.; Quandt, E. Fabrication of TiNi thin film stents. Smart Mater. Struct. 2009, 18, 104010. [Google Scholar] [CrossRef]
- DeMiranda, R.L.; Zamponi, C.; Quandt, E. Micropatterned Freestanding Superelastic TiNi Films. Adv. Eng. Mater. 2013, 15, 66–69. [Google Scholar] [CrossRef]
- Siekmeyer, G.; Schüßler, A.; de Miranda, R.L.; Quandt, E. Comparison of the Fatigue Performance of Commercially Produced Nitinol Samples versus Sputter-Deposited Nitinol. J. Mater. Eng. Perform. 2014, 23, 2437–2445. [Google Scholar] [CrossRef]
- Schlüter, K.; Zamponi, C.; Piorra, A.; Quandt, E. Comparison of the corrosion behaviour of bulk and thin film magnesium alloys. Corros. Sci. 2010, 52, 3973–3977. [Google Scholar] [CrossRef]
- Schlüter, K.; Zamponi, C.; Hort, N.; Kainer, K.U.; Quandt, E. Polycrystalline and amorphous MgZnCa thin films. Corros. Sci. 2012, 63, 234–238. [Google Scholar] [CrossRef]
- Schlüter, K.; Shi, Z.; Zamponi, C.; Cao, F.; Quandt, E.; Atrens, A. Corrosion performance and mechanical properties of sputter-deposited MgY and MgGd alloys. Corros. Sci. 2014, 78, 43–54. [Google Scholar] [CrossRef]
- Haffner, D.; Zamponi, C.; de Miranda, R.L.; Quandt, E. Micropatterned freestanding magnetron sputtered Mg-alloy scaffolds. BioNanoMaterials 2015, 16, 19–22. [Google Scholar] [CrossRef]
- Jynge, P.; Brurok, H.; Asplund, A.; Towart, R.; Refsum, H.; Karlsson, J.O.G. Cardiovascular safety of MnDPDP and MnCl2. Acta Radiol. 1997, 38, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Crossgrove, J.; Zheng, W. Manganese toxicity upon overexposure. NMR Biomed. 2004, 17, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-K.; Jun, J.-H.; Choi, C.-S. Damping Capacity in Fe-Mn Binary Alloys. ISIJ Int. 1997, 37, 1023–1030. [Google Scholar] [CrossRef]
- Martínez, J.; Cotes, S.M.; Cabrera, A.F.; Desimoni, J.; Fernández Guillermet, A. On the relative fraction of ε martensite in γ-Fe-Mn alloys. Mater. Sci. Eng. A 2005, 408, 26–32. [Google Scholar] [CrossRef]
- American Society for Testing Materials. Annual Book of ASTM Standards: Metal Test Methods and Analytical Procedures; Metals—Mechanical Testing; Elevated and Low-Temperature Tests; Metallography. Section 3. Volume 03.01; American Society for Testing Materials: West Conshohocken, PA, USA, 2007. [Google Scholar]
- Stern, M.; Geary, A.L. Electrochemical polarization I. A theoretical analysis of the shape of polarization curves. J. Electrochem. Soc. 1957, 104, 56–63. [Google Scholar] [CrossRef]
- Jones, D.A. Principles and Prevention of Corrosion, 2nd ed.; Pearson-Prentice Hall: London, UK, 1996. [Google Scholar]
- Zhu, S.; Huang, N.; Xu, L.; Zhang, Y.; Liu, H.; Sun, H.; Leng, Y. Biocompatibility of pure iron. Mater. Sci. Eng. B 2009, 29, 1589–1592. [Google Scholar] [CrossRef]
- Mouzou, E.; Paternoster, C.; Tolouei, R.; Purnama, A.; Chevallier, P.; Prima, F.; Mantovani, D. In vitro degradation behavior of Fe-20 Mn-1.2C alloy in three different pseudo-physiological solutions. Matter. Sci. Eng. C Mater. Biol. Appl. 2016, 61, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Moravej, M.; Prima, F.; Fiset, M.; Mantovani, D. Electroformed iron as new biomaterial for degradable stents: Development process and structure properties relationship. Acta Biomater. 2010, 6, 1726–1735. [Google Scholar] [CrossRef] [PubMed]
- Heiden, M.; Walker, E.; Stanciu, L. Magnesium, Iron and Zinc Alloys, the Trifecta of Bioresorbable Orthopaedic and Vascular Implantation-A Review. J. Biotechnol. Biomater. 2015. [Google Scholar] [CrossRef]
- Thornton, J.A. High Rate Thick Film Growth. Annu. Rev. Mater. Sci. 1977, 7, 239–260. [Google Scholar] [CrossRef]
- Hall, E.O. The Deformation and Ageing of Mild Steel: III Discussion of Results. Proc. Phys. Soc. Sect. B 1951, 64, 747–752. [Google Scholar] [CrossRef]
- Ding, H.; Tang, Z.Y.; Li, W.; Wang, M.; Song, D. Microstructures and Mechanical Properties of Fe-Mn-(Al, Si) TRIP/TWIP Steels. J. Iron Steel Res. Int. 2006, 13, 66–70. [Google Scholar] [CrossRef]
- Bouaziz, O.; Allain, S.; Scott, C.P.; Cugy, P.; Barbier, D. High manganese austenitic twinning induced plasticity steels. Curr. Opin. Solid State Mater. 2011, 15, 141–168. [Google Scholar] [CrossRef]
- Neu, R.W. Performance and Characterization of TWIP Steels for Automotive Applications. Mater. Perform. Charact. 2013, 2, 244–284. [Google Scholar] [CrossRef]
- Hermawan, H.; Mantovani, D. Process of prototyping coronary stents from biodegradable Fe-Mn alloys. Acta Biomater. 2013, 9, 8585–8592. [Google Scholar] [CrossRef] [PubMed]
- Mouzou, E.; Paternoster, C.; Tolouei, R.; Chevallier, P.; Biffi, C.A.; Tuissi, A.; Mantovani, D. CO2-rich atmosphere strongly affects the degradation of Fe-21Mn-1C for biodegradable metallic implants. Mater. Lett. 2016, 181, 362–366. [Google Scholar] [CrossRef]
- Ivan, N.J.; Schuller, K. Exchange bias. J. Magn. 1999, 192, 203–232. [Google Scholar]
- Paduani, C.; Galvão da Silva, E.; Perez-Alcazar, G.A.; McElfresh, M. Mössbauer effect and magnetization studies of α-FeMn alloys. J. Appl. Phys. 1991, 70, 7524–7531. [Google Scholar] [CrossRef]
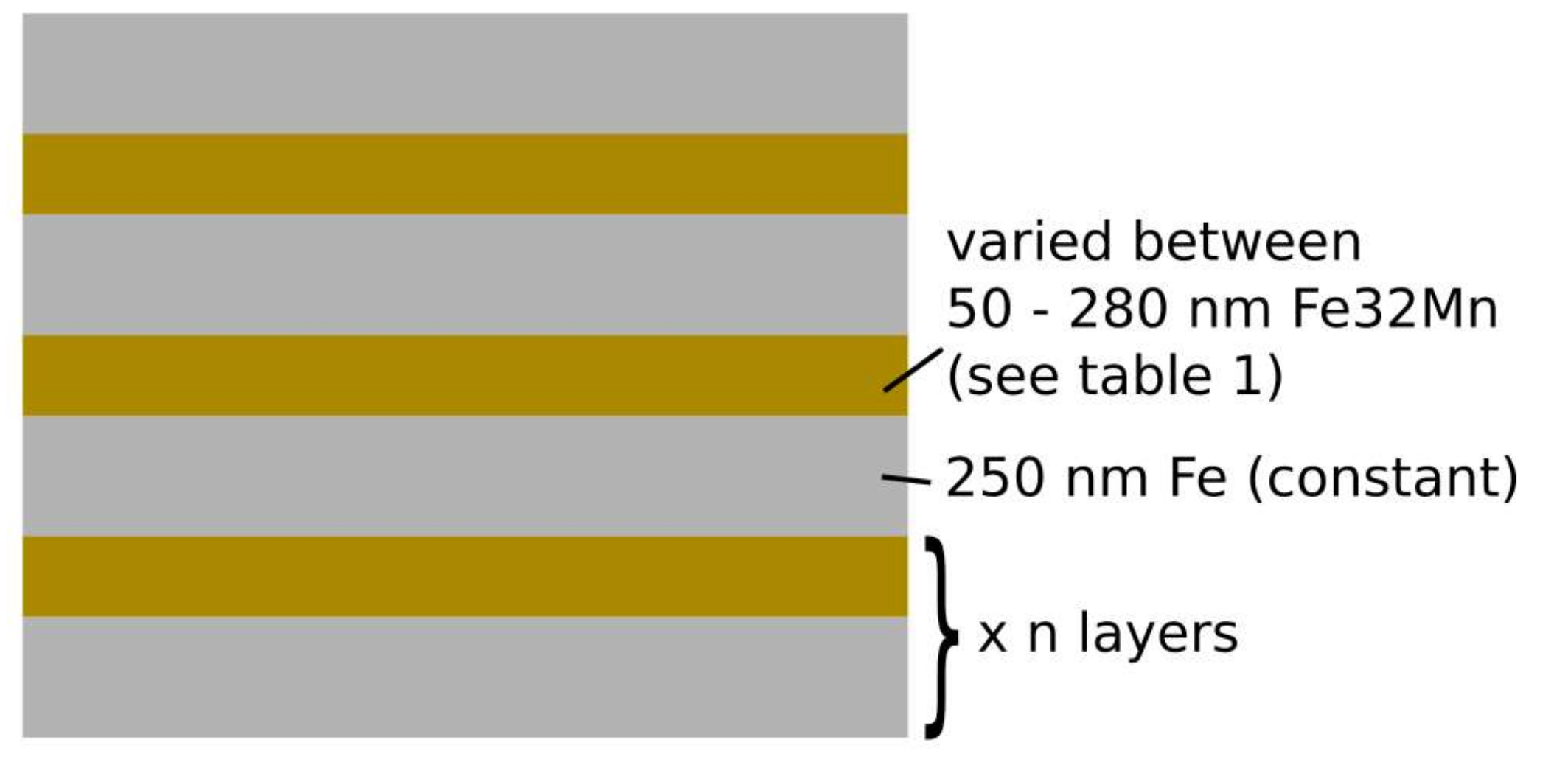

| Sample | Target (s) | FeMn32 Thickness (nm) | Nominal Mn Content (wt %) |
|---|---|---|---|
| Fe [12] | pure Fe | - | 0.0 |
| FeMn5 | Fe-FeMn35 multilayer | 50 | 4.8 |
| FeMn10 | Fe-FeMn35 multilayer | 125 | 10.0 |
| FeMn15 | Fe-FeMn35 multilayer | 250 | 15.5 |
| FeMn17 | Fe-FeMn35 multilayer | 325 | 17.2 |
| FeMn32 [15] | Pre-alloyed FeMn35 | - | 32 |
| Symbol | 2θ (°) | hkl |
|---|---|---|
| α′ 1 | 44.660 | 110 |
| α′ 2 | 65.000 | 200 |
| α′ 3 | 82.300 | 211 |
| γ 1 | 43.120 | 111 |
| γ 2 | 50.220 | 200 |
| γ 3 | 73.760 | 220 |
| γ 4 | 89.460 | 311 |
| ε 1 | 41.160 | 100 |
| ε 2 | 44.380 | 002 |
| ε 3 | 47.040 | 101 |
| ε 4 | 62.120 | 102 |
| ε 5 | 75.020 | 110 |
| ε 6 | 83.640 | 103 |
| ε 7 | 89.360 | 200 |
| ε 8 | 91.540 | 112 |
| Sample | (μm) | dmin (μm) | dmax (μm) |
|---|---|---|---|
| Fe 800 °C, 2 h [12] | 3.01 | - | - |
| FeMn5 800 °C, 2 h | 0.621 | 0.062 | 2.139 |
| FeMn10 800 °C, 2 h | 0.521 | 0.093 | 1.731 |
| FeMn15 800 °C, 2 h | 0.481 | 0.076 | 1.497 |
| FeMn17 800 °C, 2 h | 0.406 | 0.078 | 1.133 |
| FeMn32 800 °C, 2 h [15] | 0.965 | 3.193 | 0.068 |
| FeMn5 950 °C, 2 h | 0.965 | 0.223 | 3.478 |
| FeMn10 950 °C, 2 h | 0.860 | 0.179 | 2.208 |
| FeMn15 950 °C, 2 h | 0.843 | 0.216 | 2.635 |
| FeMn17 950 °C, 2 h | 0.578 | 0.990 | 1.618 |
| FeMn32 950 °C, 2 h [15] | 1.305 | 5.078 | 0.143 |
| Sample | U0 (V) | j0 (A/m²) | CR (mm/year) | WL (mg/cm²) |
|---|---|---|---|---|
| Fe 800 °C, 2 h [12,14] | −0.661 ± 0.019 | 0.092 ± 0.008 | 0.101 ± 0.009 | 3.80 ± 0.84 |
| Fe 950 °C, 2 h | −0.657 ± 0.025 | 0.109 ± 0.025 | 0.120 ± 0.012 | 3.01 ± 0.54 |
| FeMn5 800 °C, 2 h | −0.705 ± 0.007 | 0.651 ± 0.010 | 0.072 ± 0.006 | 3.17 ± 1.01 |
| FeMn5 950 °C, 2 h | −0.704 ± 0.015 | 0.073 ± 0.006 | 0.081 ± 0.007 | 2.25 ± 0.48 |
| FeMn10 800 °C, 2 h | −0.724 ± 0.006 | 0.053 ± 0.005 | 0.059 ± 0.006 | 2.34 ± 0.99 |
| FeMn10 950 °C, 2 h | −0.719 ± 0.004 | 0.064 ± 0.007 | 0.071 ± 0.006 | 1.98 ± 0.64 |
| FeMn15 800 °C, 2 h | −0,735 ± 0,008 | 0.036 ± 0.006 | 0.040 ± 0.018 | 1.65 ± 0.63 |
| FeMn15 950 °C, 2 h | −0.728 ± 0.008 | 0.038 ± 0.019 | 0.042 ± 0.008 | 1.49 ± 0.45 |
| FeMn17 800 °C, 2 h | −0.737 ± 0.018 | 0.034 ± 0.008 | 0.030 ± 0.008 | 1.41 ± 0.32 |
| FeMn17 950 °C, 2 h | −0.737 ± 0,019 | 0.030 ± 0.002 | 0.033 ± 0.002 | 1.42 ± 0.35 |
| FeMn32 800 °C, 2 h [15] | −0.749 ± 0.006 | 0.041 ± 0.011 | 0.045 ± 0.012 | 1.81 ± 0.65 |
| FeMn32 950 °C, 2 h [15] | −0.741 ± 0.010 | 0.048 ± 0.010 | 0.054 ± 0.011 | 1.68 ± 0.75 |
| Sample | YS (MPa) | UTS (MPa) | A (%) |
|---|---|---|---|
| Fe 800 °C, 2 h [12] | 266.9 ± 7.5 | 343.8 ± 4.8 | 20.0 ± 2.7 |
| Fe 950 °C, 2 h | 191.3 ± 34.7 | 246.6 ± 7.3 | 4.0 ± 0.8 |
| FeMn5 800 °C, 2 h | 686.2 ± 32.0 | 918.2 ± 44.6 | 4.2 ± 0.6 |
| FeMn5 950 °C, 2 h | 686.3 ± 24.1 | 809.1 ± 9.7 | 5.6 ± 1.0 |
| FeMn10 800 °C, 2 h | 905.8 ± 21.3 | 1242.9 ± 36.2 | 2.8 ± 0.5 |
| FeMn10 950 °C, 2 h | 805.3 ± 35.3 | 1100.9 ±11.5 | 2.2 ± 0.5 |
| FeMn15 800 °C, 2 h | 766.6 ± 27.1 | 1080.7± 20.9 | 17.0 ± 1.1 |
| FeMn15 950 °C, 2 h | 697.4 ± 59.7 | 963.9 ± 41.8 | 20.5 ± 2.4 |
| FeMn17 800 °C, 2 h | 824.8 ± 27.0 | 1061.4 ± 29.4 | 21.5 ± 1.7 |
| FeMn17 950 °C, 2 h | 857.0 ± 18.8 | 984.7 ± 22.9 | 18.5 ± 4.6 |
| FeMn32 800 °C, 2 h [15] | 571.0 ± 11.5 | 712.5 ± 3.7 | 23.1 ± 1.6 |
| FeMn32 950 °C, 2 h [15] | 541.0 ± 10.9 | 678.9 ± 15.9 | 24.4 ± 1.7 |
| SS 316 L [10,33] | 190 | 490 | 40 |
| Required properties [34] | >200 | >300 | >15–18 |
| Sample | JS (T) | JR (T) | μ0HC (mT) | χmax | Ediss (mJ/cm3) |
|---|---|---|---|---|---|
| Fe as deposited [15] | 2.206 | 0.650 | 1.874 | 560.59 | 10.50 |
| Fe 800 °C, 2 h [15] | 2.141 | 0.545 | 1.695 | 510.26 | 9.10 |
| Fe 950 °C, 2 h [15] | 2.052 | 0.715 | 1.147 | 595.34 | 7.77 |
| FeMn5 as deposited | 1.822 | 0.462 | 2.115 | 225.18 | 18.32 |
| FeMn5 800 °C, 2 h | 1.888 | 0.340 | 1.463 | 223.26 | 18.35 |
| FeMn5 950 °C, 2 h | 1.919 | 0.391 | 1.804 | 218.45 | 14.30 |
| FeMn10 as deposited | 1.542 | 0.682 | 2.62 | 270.22 | 19.59 |
| FeMn10 800 °C, 2 h | 1.619 | 0.486 | 2.530 | 221.57 | 12.59 |
| FeMn10 950 °C, 2 h | 1.726 | 0.395 | 1.876 | 207.46 | 10.83 |
| FeMn15 as deposited | 1.257 | 0.360 | 3.699 | 101.92 | 19.38 |
| FeMn15 800 °C, 2 h | 0.014 | 1.12 × 10−3 | 8.440 | 0.18 | 0.24 |
| FeMn15 950 °C, 2 h | 9.31 × 10−3 | 0.38 × 10−3 | 12.549 | 0.04 | 0.13 |
| FeMn17 as deposited | 1.118 | 0.289 | 2.959 | 102.41 | 17.04 |
| FeMn17 800 °C, 2 h | 0.014 | 0.84 × 10−3 | 8.026 | 0.14 | 0.20 |
| FeMn17 950 °C, 2 h | 9.40 × 10−3 | 0.13 × 10−3 | 7.129 | 0.09 | 0.09 |
| FeMn32 as deposited [15] | 0.032 | 6.02 × 10−3 | 26.32 | 0.37 | 1.83 |
| FeMn32 800 °C, 2 h [15] | 1.09 × 10−3 | 0.13 × 10−3 | 14.752 | 0.02 | 0.03 |
| FeMn32 950 °C, 2 h [15] | 0.64 × 10−3 | 0.04 × 10−3 | 8.586 | 0.01 | 0.02 |
| SS 316L [15] | 2.65 × 10−3 | 0.51 × 10−3 | 30.212 | 0.02 | 0.11 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jurgeleit, T.; Jessen, L.K.; Quandt, E.; Zamponi, C. Magnetron-Sputtered, Biodegradable FeMn Foils: The Influence of Manganese Content on Microstructure, Mechanical, Corrosion, and Magnetic Properties. Materials 2018, 11, 482. https://doi.org/10.3390/ma11040482
Jurgeleit T, Jessen LK, Quandt E, Zamponi C. Magnetron-Sputtered, Biodegradable FeMn Foils: The Influence of Manganese Content on Microstructure, Mechanical, Corrosion, and Magnetic Properties. Materials. 2018; 11(4):482. https://doi.org/10.3390/ma11040482
Chicago/Turabian StyleJurgeleit, Till, Lea Katharina Jessen, Eckhard Quandt, and Christiane Zamponi. 2018. "Magnetron-Sputtered, Biodegradable FeMn Foils: The Influence of Manganese Content on Microstructure, Mechanical, Corrosion, and Magnetic Properties" Materials 11, no. 4: 482. https://doi.org/10.3390/ma11040482
APA StyleJurgeleit, T., Jessen, L. K., Quandt, E., & Zamponi, C. (2018). Magnetron-Sputtered, Biodegradable FeMn Foils: The Influence of Manganese Content on Microstructure, Mechanical, Corrosion, and Magnetic Properties. Materials, 11(4), 482. https://doi.org/10.3390/ma11040482





