Foliage and Grass as Fuel Pellets–Small Scale Combustion of Washed and Mechanically Leached Biomass
Abstract
:1. Introduction
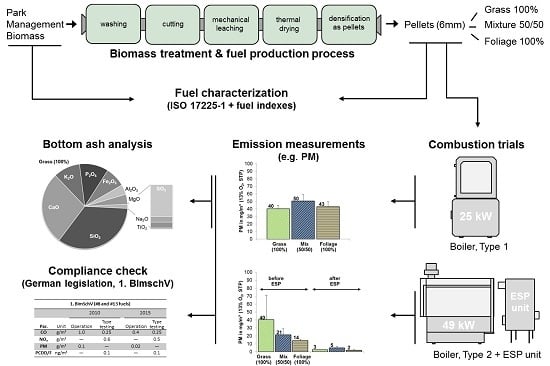
2. Materials and Methods
2.1. Raw Material and Fuel Types
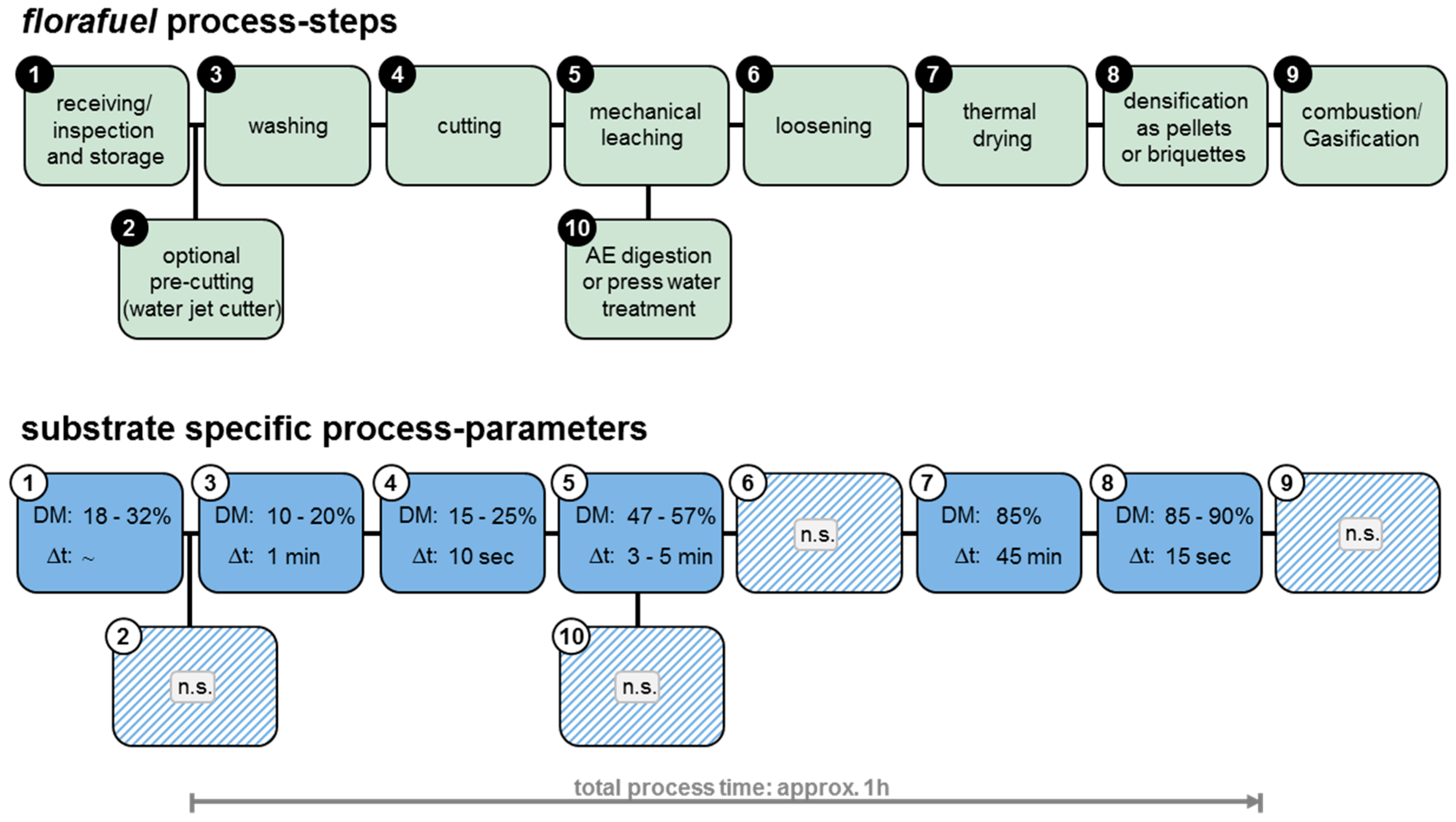
2.2. Mineral Leaching (florafuel Process) and Densification of the Biomass
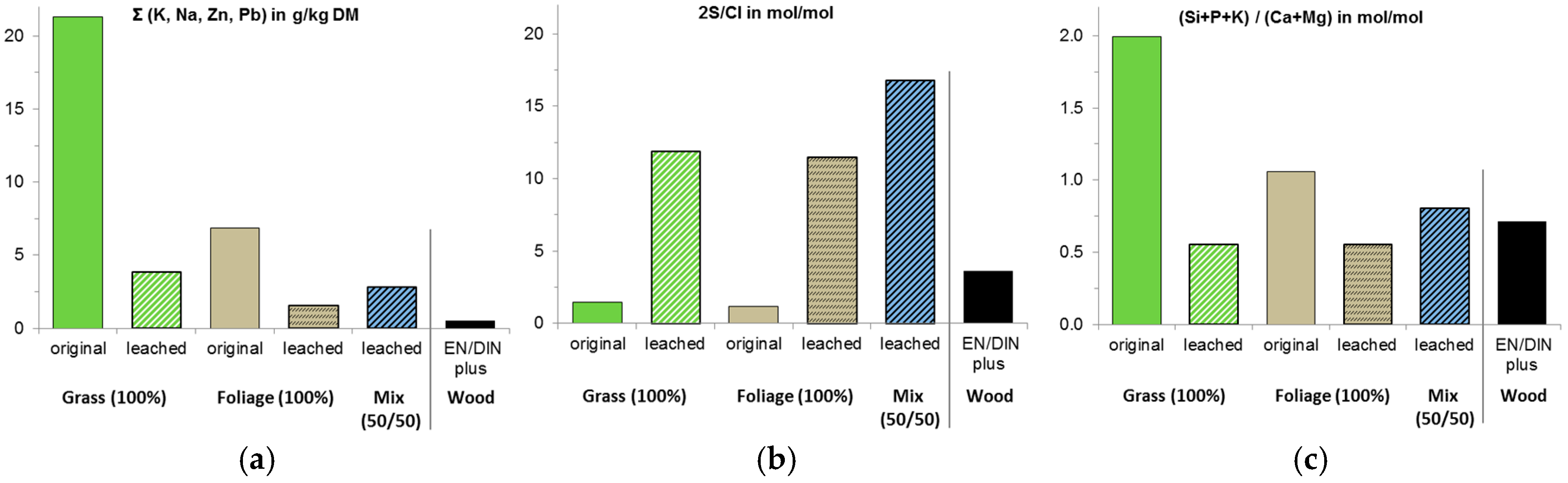
2.3. Raw Material, Fuel, and Ash Characteristics
2.4. Combustion Technology and Emission Measurements
3. Results and Discussion
3.1. Fuel Characteristics
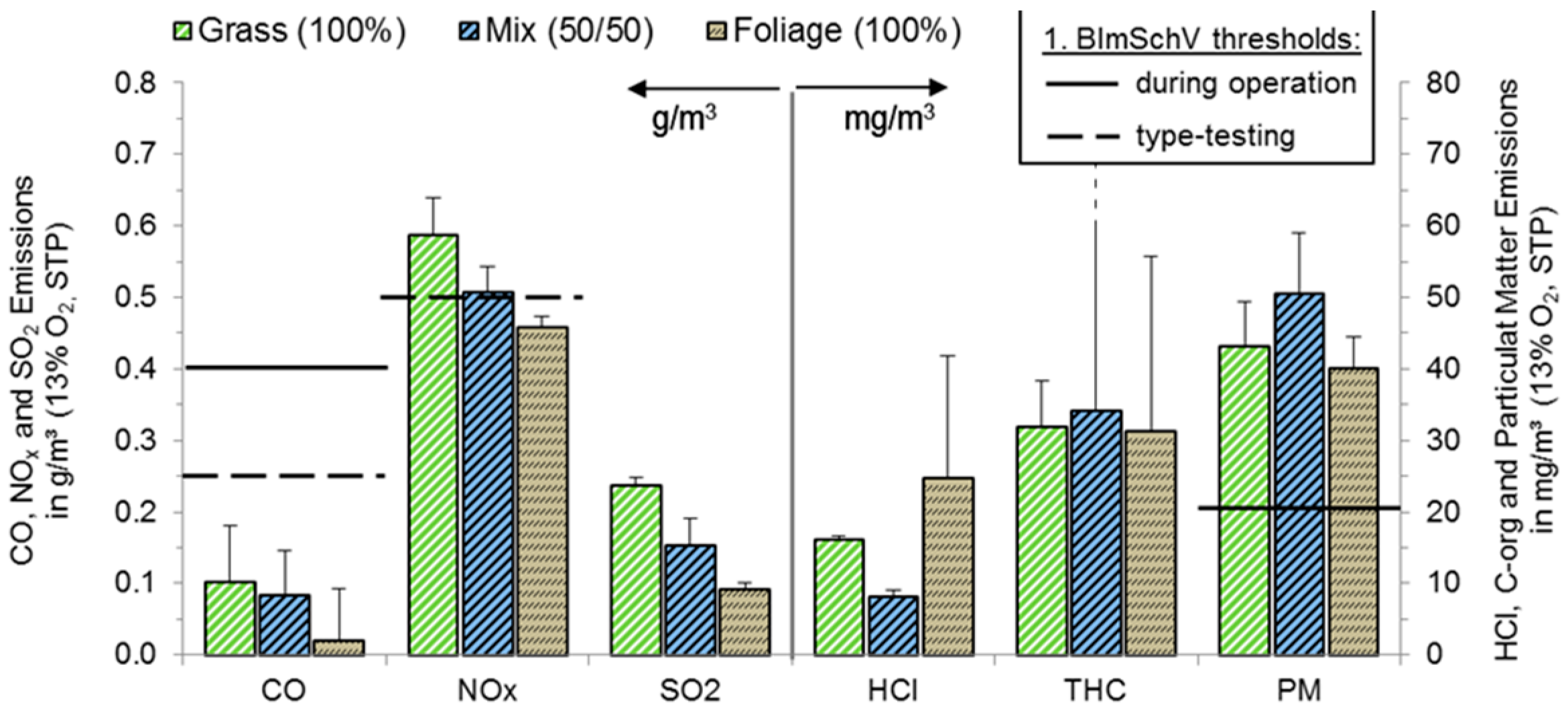
3.2. Combustion
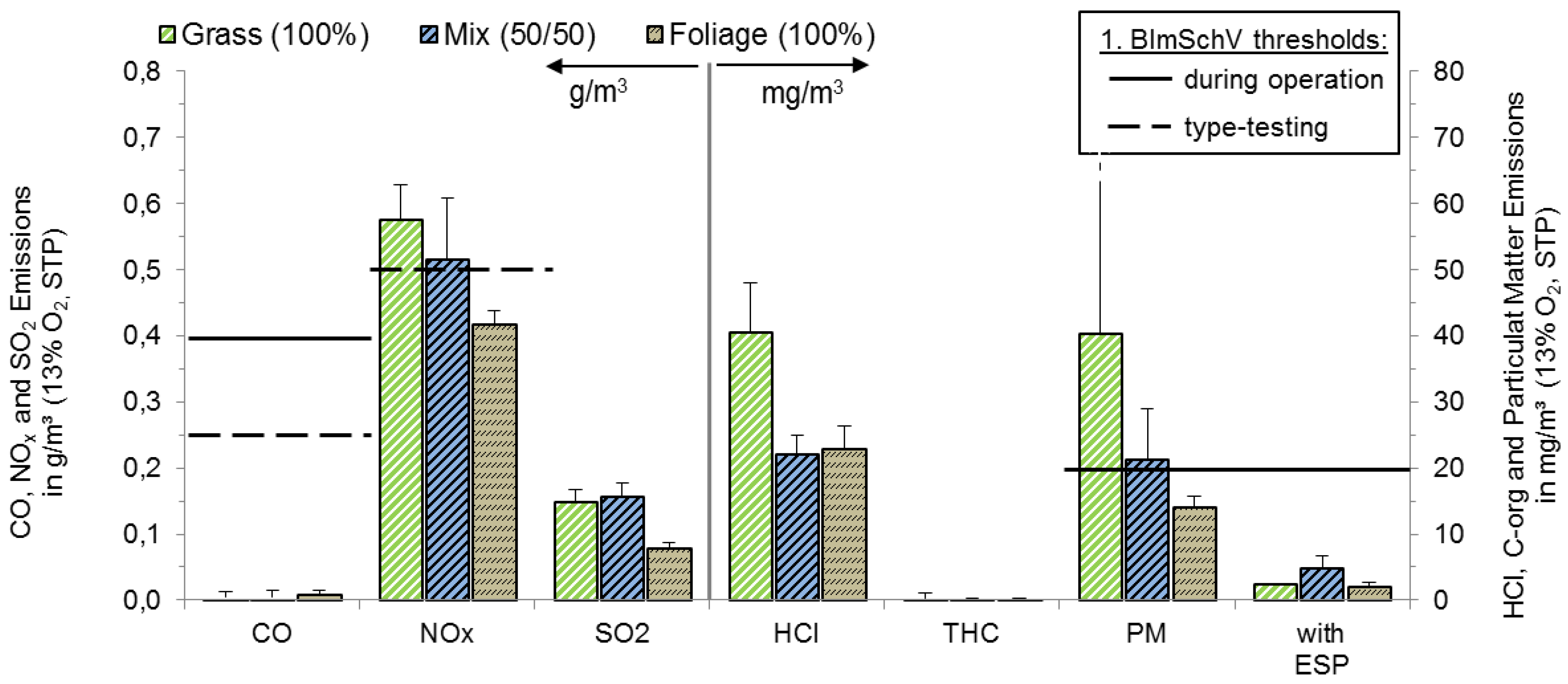
3.2.1. Legislative Background
3.2.2. Boiler (25 kWth) with Moving Grate
3.2.3. Boiler (49 kWth) with Water-Cooled Burner Tube and Electrostatic Precipitator
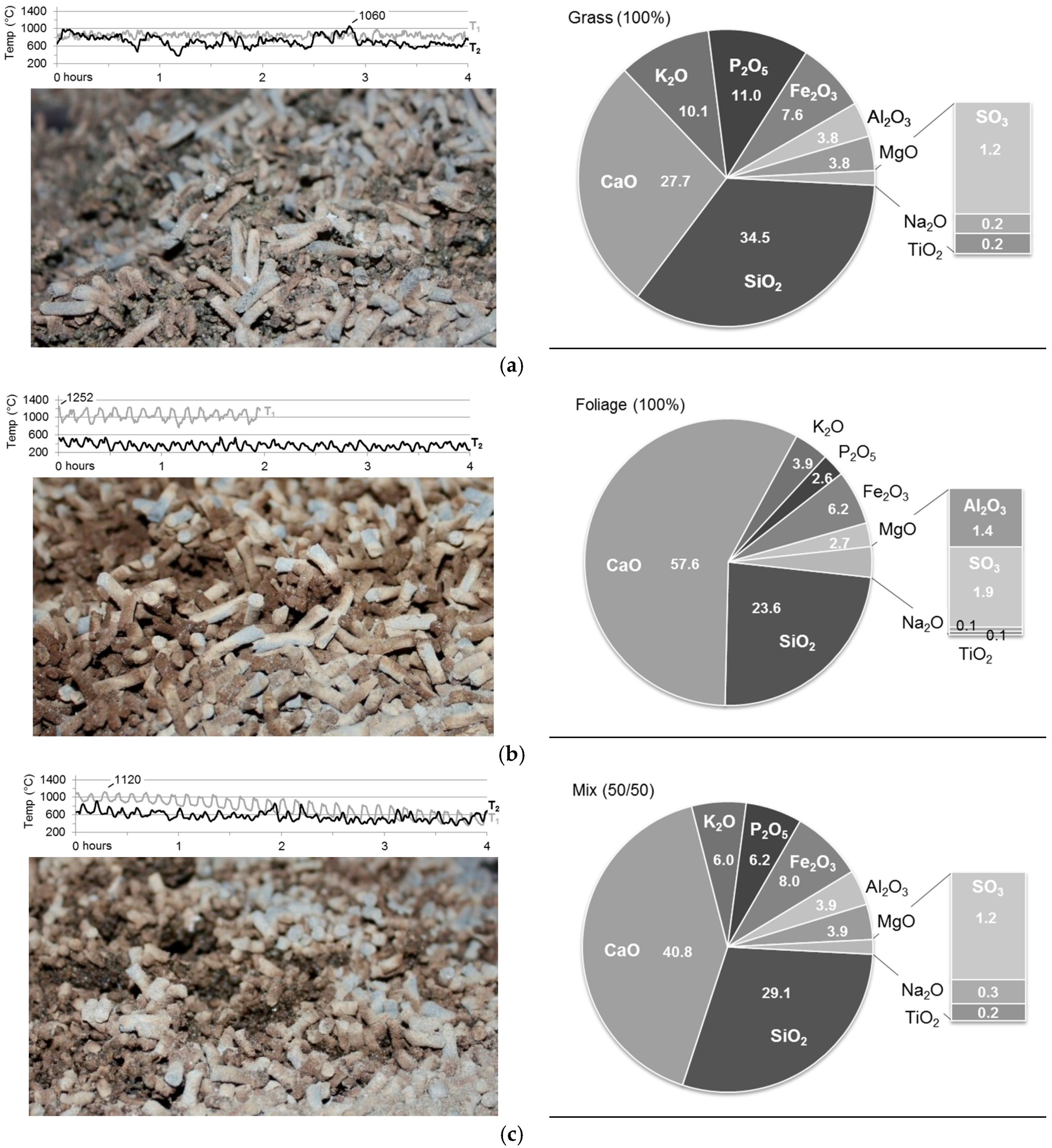
3.3. Bottom Ash and Ash Melting
3.3.1. Grate Temperature Profile
3.3.2. Ash Melting Characteristics
3.3.3. Bottom Ash Composition
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| DM | dry matter |
| DT | deformation temperature |
| ESP | electrostatic precipitator |
| FM | fresh matter |
| FT | flow temperature |
| FTIR | Fourier transformed infrared |
| HT | hemisphere temperature |
| LHV | lower heating value |
| PCDD/F | polychlorinated dioxin/furan |
| PM | particulate matter |
| SST | shrinkage starting temperature |
| STP | standard temperature and pressure |
| THC | total hydrocarbon |
| BImSchV | First Ordinance on the Implementation of the Federal Immission Control Act |
References
- Atanasiu, B. The Role of Bioenergy in the National Renewable Energy Action Plans: A First Identification of Issues and Uncertainties. Publication of the BIOMASS FUTURES project (IEE 08 653 SI2. 529 241), 2010. Available online: http://www.ieep.eu/assets/753/bioenergy_in_NREAPs.pdf (accessed on 1 May 2016).
- Smart Bioenergy; Thrän, D. (Ed.) Springer International Publishing: Cham, Switzerland, 2015.
- Kretzschmar, J.; Thrän, D. Energy recovery from roadside vegetation. Müll Abfall 2009, 11, 577–583. (In German) [Google Scholar]
- Bühle, L.; Hensgen, F.; Donnison, I.; Heinsoo, K.; Wachendorf, M. Life cycle assessment of the integrated generation of solid fuel and biogas from biomass (IFBB) in comparison to different energy recovery, animal-based and non-refining management systems. Bioresour. Technol. 2012, 111, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Prochnow, A.; Heiermann, M.; Plöchl, M.; Amon, T.; Hobbs, P.J. Bioenergy from permanent grassland—A review: 2. Combustion. Bioresour. Technol. 2009, 100, 4945–4954. [Google Scholar] [CrossRef] [PubMed]
- Piepenschneider, M.; Nurmatov, N.; Bühle, L.; Hensgen, F.; Wachendorf, M. Chemical Properties and Ash Slagging Characteristics of Solid Fuels from Urban Leaf Litter. Waste Biomass Valorization 2015, 1–9. [Google Scholar] [CrossRef]
- Döhling, F.; Kirsten, C.; Khalsa, J.; Weller, N. Foliage as Solid Fuel—Fuel Enhancement by Washing and Mechanical Leaching. In Proceedings of the International Conference on Solid Wastes 2015: Knowledge Transfer for Sustainable Resource Management, Hong Kong, China, 19–25 May 2015; Wong, J.W.C., Tyagi, R.D., Nelles, M., Selvam, A., Eds.; Hong Kong Baptist University: Hong Kong, China, 2015; pp. 1002–1005. [Google Scholar]
- Obernberger, I.; Brunner, T.; Bärnthaler, G. Chemical properties of solid biofuels—Significance and impact. Biomass Bioenergy 2006, 30, 973–982. [Google Scholar] [CrossRef]
- Sommersacher, P.; Brunner, T.; Obernberger, I. Fuel indexes: A novel method for the evaluation of relevant combustion properties of new biomass fuels. Energy Fuels 2012, 26, 380–390. [Google Scholar] [CrossRef]
- Arvelakis, S.; Vourliotis, P.; Kakaras, E.; Koukios, E.G. Effect of leaching on the ash behavior of wheat straw and olive residue during fluidized bed combustion. Biomass Bioenergy 2001, 20, 459–470. [Google Scholar] [CrossRef]
- Bühle, L.; Dürl, G.; Hensgen, F.; Urban, A.; Wachendorf, M. Effects of hydrothermal conditioning and mechanical dewatering on ash melting behaviour of solid fuel produced from European semi-natural grasslands. Fuel 2014, 118, 123–129. [Google Scholar] [CrossRef]
- Jenkins, B.M.; Bakker, R.R.; Wei, J.B. On the properties of washed straw. Biomass Bioenergy 1996, 10, 177–200. [Google Scholar] [CrossRef]
- Turn, S.Q.; Kinoshita, C.M.; Ishimura, D.M. Removal of inorganic constituents of biomass feedstocks by mechanical dewatering and leaching. Biomass Bioenergy 1997, 12, 241–252. [Google Scholar] [CrossRef]
- Hensgen, F.; Bühle, L.; Donnison, I.; Frasier, M.; Vale, J.; Corton, J.; Heinsoo, K.; Melts, I.; Wachendorf, M. Mineral concentrations in solid fuels from European semi-natural grasslands after hydrothermal conditioning and subsequent mechanical dehydration. Bioresour. Technol. 2012, 118, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Schlederer, S.; Günther, F.W. Local biomass as a decentral source of energy. Müll Abfall 2013, 3, 141–147. (In German) [Google Scholar]
- Bühle, L.; Reulein, J.; Stülpnagel, R.; Zerr, W.; Wachendorf, M. Methane yields and digestion dynamics of press fluids from mechanically dehydrated maize silages using different types of digesters. Bioenergy Res. 2011, 5, 294–305. [Google Scholar] [CrossRef]
- Andruschkewitsch, M.; Wachendorf, C.; Wachendorf, M. Effects of digestates from different biogas production systems on above and belowground grass growth and the nitrogen status of the plant-soil-system. Grassl. Sci. 2013, 59, 183–195. [Google Scholar] [CrossRef]
- Piepenschneider, M.; Moor, S.D.; Hensgen, F.; Meers, E.; Wachendorf, M. Element concentrations in urban grass cuttings from roadside verges in the face of energy recovery. Environ. Sci. Pollut. Res. 2015, 22, 7808–7820. [Google Scholar] [CrossRef] [PubMed]
- Paulrud, S.; Nilsson, C.; Öhman, M. Reed canary-grass ash composition and its melting behaviour during combustion. Fuel 2001, 80, 1391–1398. [Google Scholar] [CrossRef]
- Garcia-Maraver, A.; Zamorano, M.; Fernandes, U.; Rabaçal, M.; Costa, M. Relationship between fuel quality and gaseous and particulate matter emissions in a domestic pellet-fired boiler. Fuel 2014, 119, 141–152. [Google Scholar] [CrossRef]
- Carvalho, L.; Elisabeth, W.; Christian, P. Performance of a pellet boiler fired with agricultural fuels. Appl. Energy 2012, 104, 286–296. [Google Scholar] [CrossRef]
- Stelte, W.; Sanadi, A.R.; Shang, L.; Holm, J.K.; Ahrenfeldt, J.; Henriksen, U.B. Recent developments in biomass pelletization—A review. Bioresources 2012, 7, 4451–4490. [Google Scholar]
- REN21. Renewables 2015 Global Status Report; REN21 Secretariat: Paris, France, 2015. [Google Scholar]
- Kirsten, C.; Lenz, V.; Schröder, H.-W.; Repke, J.-U. Hay pellets—The influence of particle size reduction on their physical–mechanical quality and energy demand during production. Fuel Process. Technol. 2016, 148, 163–174. [Google Scholar] [CrossRef]
- Pilz, A.; Döhling, F.; Kirsten, C.; Weller, N.; Zeng, T. Pelletizing and energy related use of hay from landscape preservation. Landtechnik 2013, 68, 349–352. (In German) [Google Scholar]
- Bundesgesetzblatt. Erste Verordnung zur Durchführung des Bundes-Immissionsschutzgesetzes (Verordnung über kleine und mittlere Feuerungsanlagen—1. BimschV), 2010. Available online: https://www.gesetze-im-internet.de/bimschv_1_2010/BJNR003800010.html (accessed on 1 May 2016). (In German)
- Villeneuve, J.; Palacios, J.H.; Savoie, P.; Godbout, S. A critical review of emission standards and regulations regarding biomass combustion in small scale units (<3 MW). Bioresour. Technol. 2012, 111, 1–11. [Google Scholar] [PubMed]
- DIN EN ISO 17225-1. Solid Biofuels—Fuel Specifications and Classes—Part 1: General Requirements; Beuth Verlag: Berlin, Germany, 2014. (In German) [Google Scholar]
- DIN CEN/TS 15370-1. Solid Biofuels—Method for the Determination of Ash Melting Behaviour—Part 1: Characteristic Temperatures Method; Beuth Verlag: Berlin, Germany, 2006. (In German) [Google Scholar]
- DIN EN 1948. Stationary Source Emissions—Determination of the Mass Concentration of PCDDs/PCDFs and Dioxin-Like PCBs—Part 1: Sampling of PCDDs/PCDFs; Beuth Verlag: Berlin, Germany, 2006. (In German) [Google Scholar]
- VDI-Richtlinie 2066 Blatt 1. Gravimetrische Bestimmung der Staubbeladung, VDI/DIN-Handbuch Reinhaltung der Luft, Band 4: Analysen-und Messverfahren; Beuth Verlag: Berlin, Germany, 2006. (In German) [Google Scholar]
- Khalsa, J.; Fricke, T.; Weisser, W.W.; Weigelt, A.; Wachendorf, M. Effects of functional groups and species richness on biomass constituents relevant for combustion: results from a grassland diversity experiment. Grass Forage Sci. 2012, 67, 569–588. [Google Scholar] [CrossRef]
- Khalsa, J.; Döhling, F.; Weller, N.; Lenz, V. Prospects and Limitations of Solid Fuel Enhancement Through Torrefaction, Hydrothermal Carbonization or Mechanical Leaching—Conclusions from Combustion Experiments and Emission Measurements. In Proceedings of the 22nd European Biomass Conference and Exhibition, Hamburg, Germany, 23–26 June 2014; pp. 998–1003.
- Carvalho, L.; Lundgren, J.; Wopienka, E. Challenges in small-scale combustion of agricultural biomass fuels. Int. J. Energy Clean Environ. 2008, 9, 127–142. [Google Scholar]
- Knudsen, J.N.; Jensen, P.A.; Dam-Johansen, K. Transformation and Release to the Gas Phase of Cl, K, and S during Combustion of Annual Biomass. Energy Fuels 2004, 18, 1385–1399. [Google Scholar] [CrossRef]
- Paulrud, S.; Nilsson, C. Briquetting and combustion of spring-harvested reed canary-grass: Effect of fuel composition. Biomass Bioenergy 2001, 20, 25–35. [Google Scholar] [CrossRef]
- Skotland, C.H. Measurement of temperature conditions in grate zone of a 1 MW wood-pellets boiler fired with high ash content wood-pellets. Master’s Thesis, Norwegian University of Science and Technology, Trondheim, 2009. Available online: http://daim.idi.ntnu.no/masteroppgaver/004/4715/masteroppgave.pdf (accessed on 1 May 2016). [Google Scholar]
- ASTM D1857-87(2000). In Standard Test Method for Fusibility of Coal and Coke Ash; ASTM International: West Conshohocken, PA, USA, 2000.
- DIN 51730:2007-09. In Testing of Solid Fuels—Determination of Fusibility of Fuel Ash; Beuth Verlag: Berlin, Germany, 2007. (In German)
- Huffman, G.P.; Huggins, F.E.; Dunmyre, G.R. Investigation of the high-temperature behaviour of coal ash in reducing and oxidizing atmospheres. Fuel 1981, 60, 585–597. [Google Scholar] [CrossRef]
- Lloyd, W.G.; Riley, J.T.; Zhou, S.; Risen, M.A.; Tibbitts, R.L. Ash fusion temperatures under oxidizing conditions. Energy Fuels 1993, 7, 490–494. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933. [Google Scholar] [CrossRef]
| Fuel Composition | Method | Grass (100%) | Foliage (100%) | Mixture (50/50) | Wood | |||
|---|---|---|---|---|---|---|---|---|
| Parameter | Unit | (DIN EN ISO) | Leached | ∆leached (in %) | Leached | ∆leached (in %) | Leached | Mean values (n = 4) |
| LHV | DM MJ/kg | 18125 | 18.4 | +10 | 18.9 | +7 | 18.2 | 18.8 |
| Ash | % DM | 18122 | 8.1 | −58 | 10.5 | −40 | 11.0 | 0.32 |
| Water | % FM | 18134-1 | 14.6 | - | 16.2 | - | 13.9 | 6.2 |
| Volatiles | % DM | 18123 | 75.1 | +19 | 72.1 | +8 | 71.8 | 84.9 |
| C | % DM | 16948 | 48.0 | +7 | 51.3 | +7 | 49.1 | 51.4 |
| H | % DM | 16948 | 5.8 | +6 | 6.0 | +19 | 5.3 | 6.3 |
| N | % DM | 16948 | 1.4 | −29 | 0.8 | −7 | 1.3 | 0.2 |
| S | % DM | 16994 | 0.10 | −55 | 0.06 | −42 | 0.13 | 0.007 |
| Cl | % DM | 16994 | 0.02 | −94 | 0.01 | −94 | 0.02 | 0.004 |
| Al | g/kg DM | 16967 | 0.70 | −82 | 0.50 | −76 | 1.32 | 0.05 |
| Pb | g/kg DM | 16968 | 0.004 | −43 | - | - | - | 0.001 |
| Ca | g/kg DM | 16967 | 15.1 | −42 | 31.6 | −16 | 26.4 | 1.0 |
| Fe | g/kg DM | 16967 | 1.1 | −70 | 1.2 | −41 | 1.9 | 0.08 |
| K | g/kg DM | 16967 | 3.26 | −84 | 1.47 | −77 | 2.48 | 0.5 |
| Mg | g/kg DM | 16967 | 2.59 | −62 | 1.70 | −57 | 3.03 | 0.2 |
| Na | g/kg DM | 16967 | 0.45 | −4 | - | - | 0.23 | 0.01 |
| P | g/kg DM | 16967 | 2.03 | −68 | 0.60 | −73 | 1.50 | 0.09 |
| Si | g/kg DM | 16967 | 3.33 | −89 | 11.7 | −55 | 14.6 | 0.64 |
| Ti | g/kg DM | 16967 | 0.05 | −75 | 0.04 | −76 | 0.09 | 0.003 |
| Zn | g/kg DM | 16968 | 0.09 | +30 | 0.05 | +53 | 0.11 | 0.01 |
| Pellet characteristics | - | Grass | - | Foliage | - | Mixture | - | |
| Bulk density | kg/m3 | 17828 | 600 | - | 659 | - | 628 | - |
| Mechanical durability | % | 17831-1 | 98.7 | - | 97.7 | - | 98.7 | - |
| Parameter | Unit | 1. BImSchV (#8 and #13 Fuels) | |||
|---|---|---|---|---|---|
| 2010 | 2015 | ||||
| Operation | Type Testing | Operation | Type Testing | ||
| CO | g/m3 | 1.0 | 0.25 | 0.4 | 0.25 |
| NOx | g/m3 | - | 0.6 | - | 0.5 |
| PM | g/m3 | 0.1 | - | 0.02 | - |
| PCDD/F | ng/m3 | - | 0.1 | - | 0.1 |
| Pellets | Ash Melting Behavior | |||
|---|---|---|---|---|
| SST (°C) | DT (°C) | HT (°C) | FT (°C) | |
| Oxidizing | ||||
| Grass (100%) | 1150 | 1180 | 1210 | 1220 |
| Foliage (100%) | 950 | >1500 | >1500 | >1500 |
| Mix (50/50) | 1160 | 1230 | 1280 | 1290 |
| Inert | ||||
| Grass (100%) | 760 | 1160 | 1200 | 1230 |
| Foliage (100%) | 770 | >1500 | >1500 | >1500 |
| Mix (50/50) | 950 | 1270 | 1290 | 1300 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalsa, J.H.A.; Döhling, F.; Berger, F. Foliage and Grass as Fuel Pellets–Small Scale Combustion of Washed and Mechanically Leached Biomass. Energies 2016, 9, 361. https://doi.org/10.3390/en9050361
Khalsa JHA, Döhling F, Berger F. Foliage and Grass as Fuel Pellets–Small Scale Combustion of Washed and Mechanically Leached Biomass. Energies. 2016; 9(5):361. https://doi.org/10.3390/en9050361
Chicago/Turabian StyleKhalsa, Jan Hari Arti, Frank Döhling, and Florian Berger. 2016. "Foliage and Grass as Fuel Pellets–Small Scale Combustion of Washed and Mechanically Leached Biomass" Energies 9, no. 5: 361. https://doi.org/10.3390/en9050361
APA StyleKhalsa, J. H. A., Döhling, F., & Berger, F. (2016). Foliage and Grass as Fuel Pellets–Small Scale Combustion of Washed and Mechanically Leached Biomass. Energies, 9(5), 361. https://doi.org/10.3390/en9050361