Electrochemical Mechanism for FeS2/C Composite in Lithium Ion Batteries with Enhanced Reversible Capacity
Abstract
:1. Introduction
2. Experiments
2.1. Preparation of FeS2 and Doped FeS2@C
2.2. Materials Characterization and Electrochemical Evaluation
3. Results and Discussion
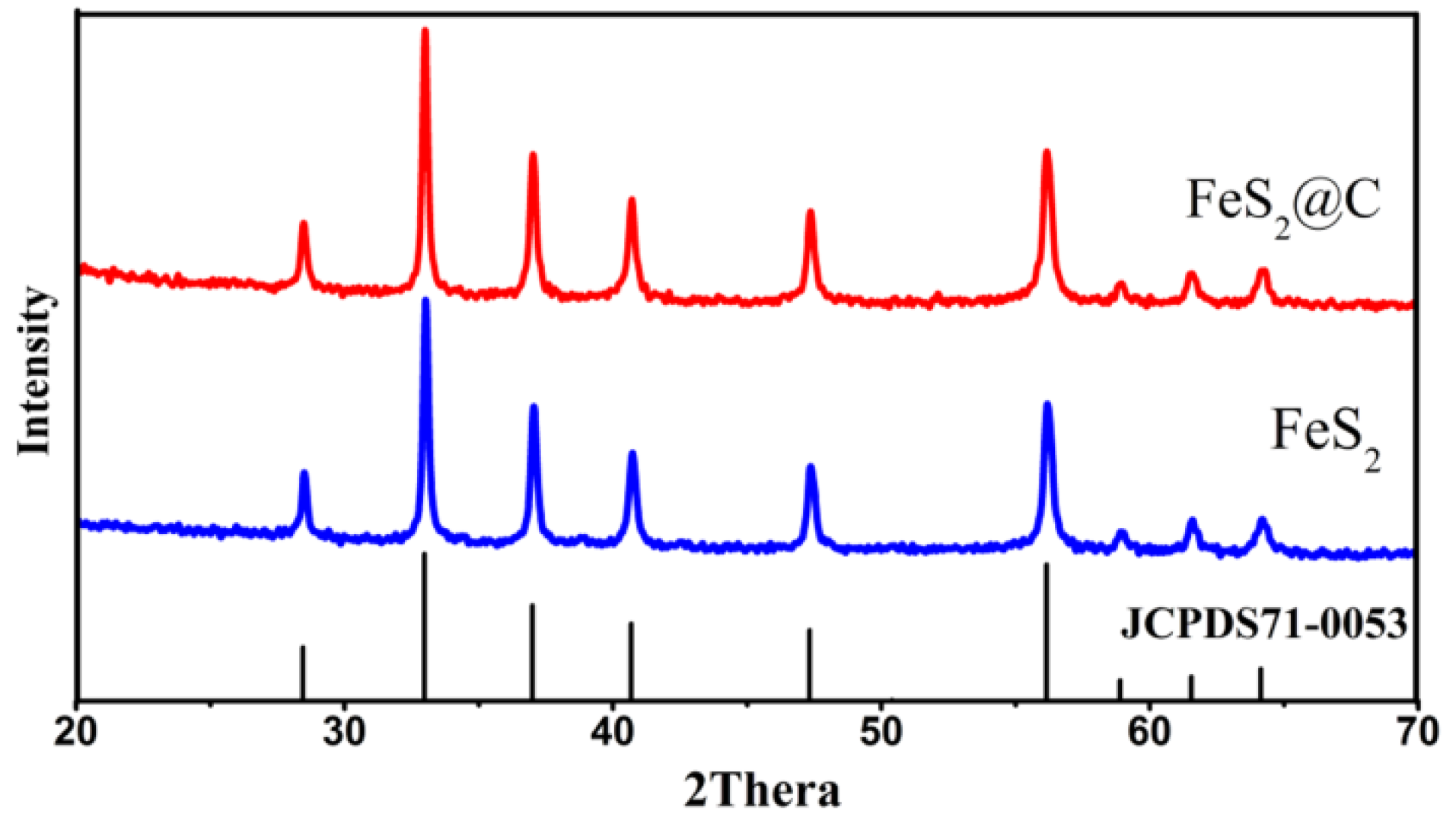
3.1. X-Ray Diffraction (XRD)
3.2. Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM)
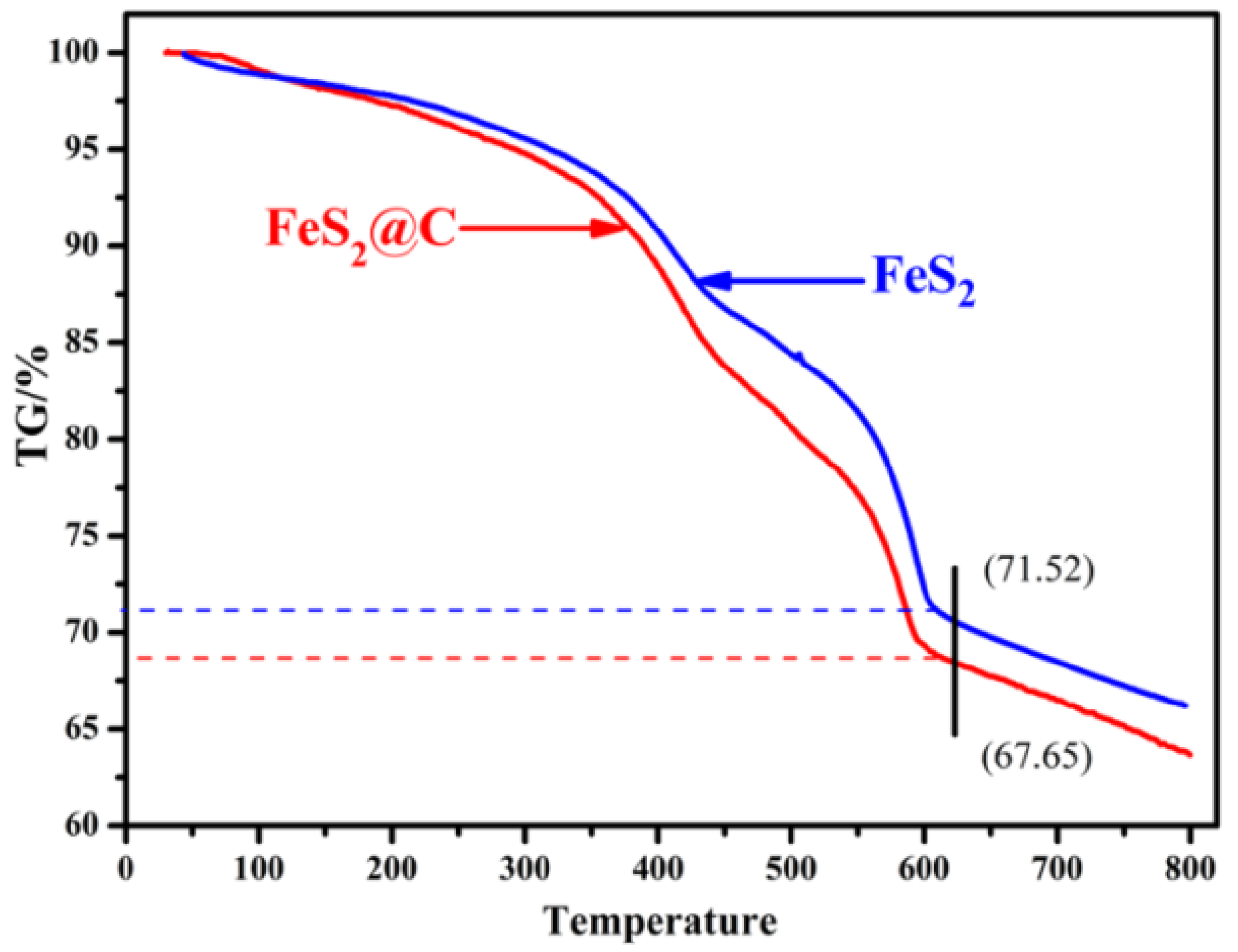
3.3. Thermogravimetric Analysis (TGA)
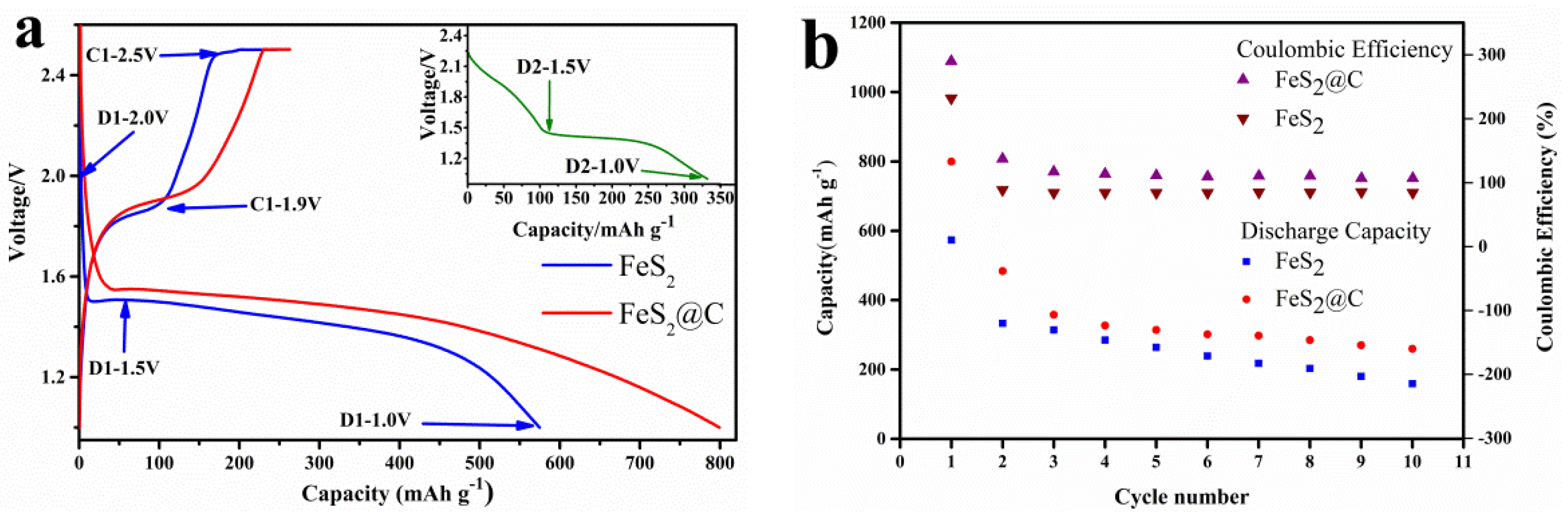
3.4. Galvanostatic Discharge and Charge Test
3.5. Electrochemical Impedance Spectroscopy (EIS)
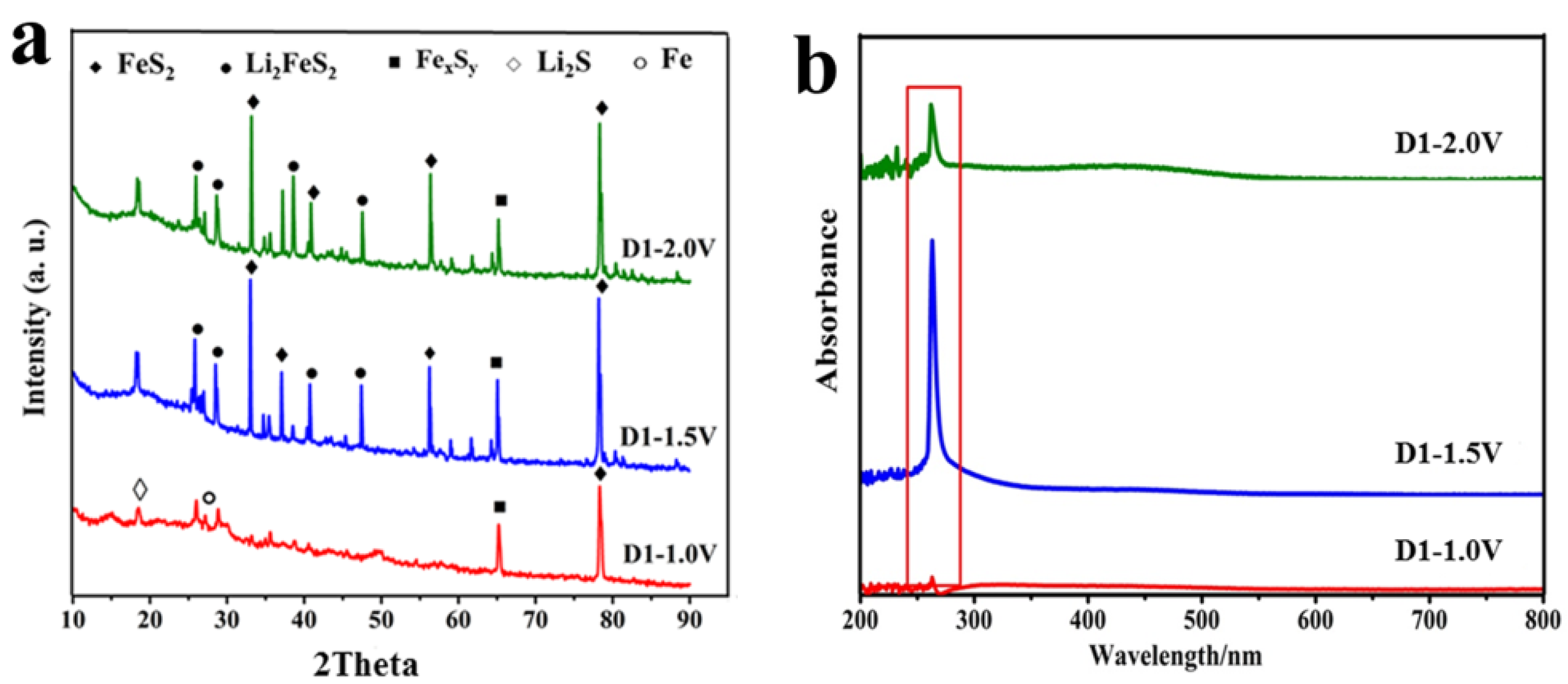
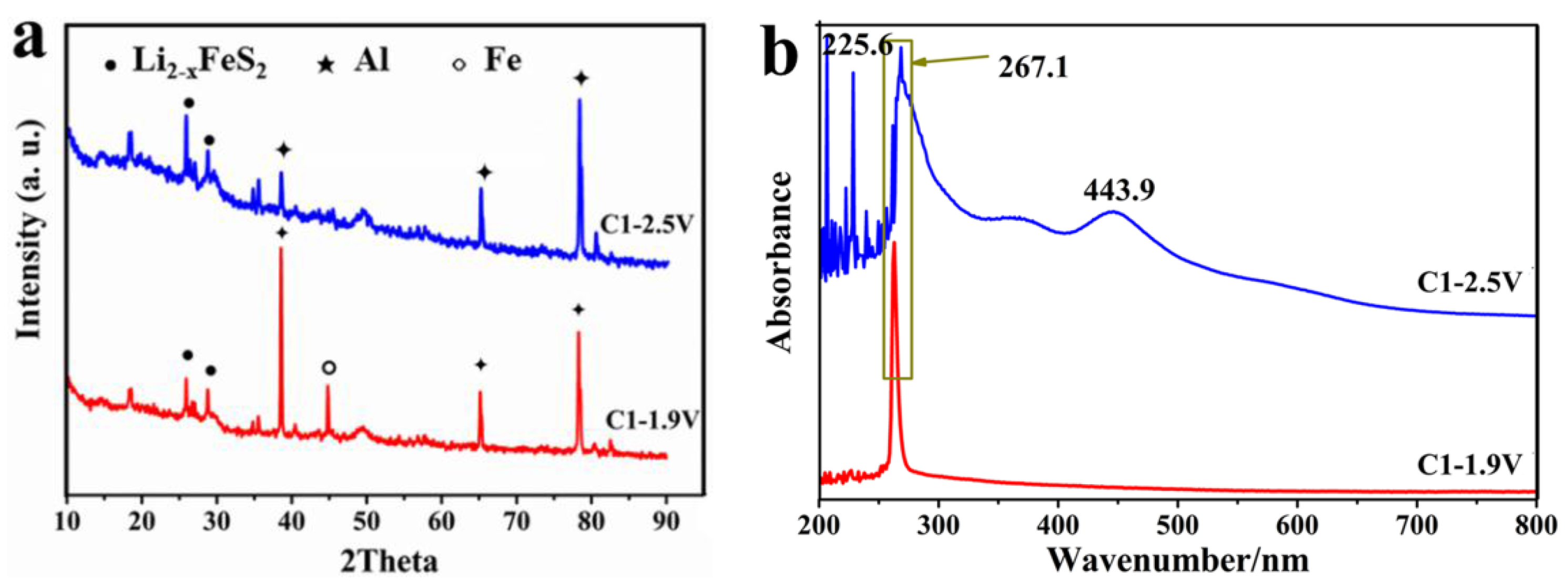
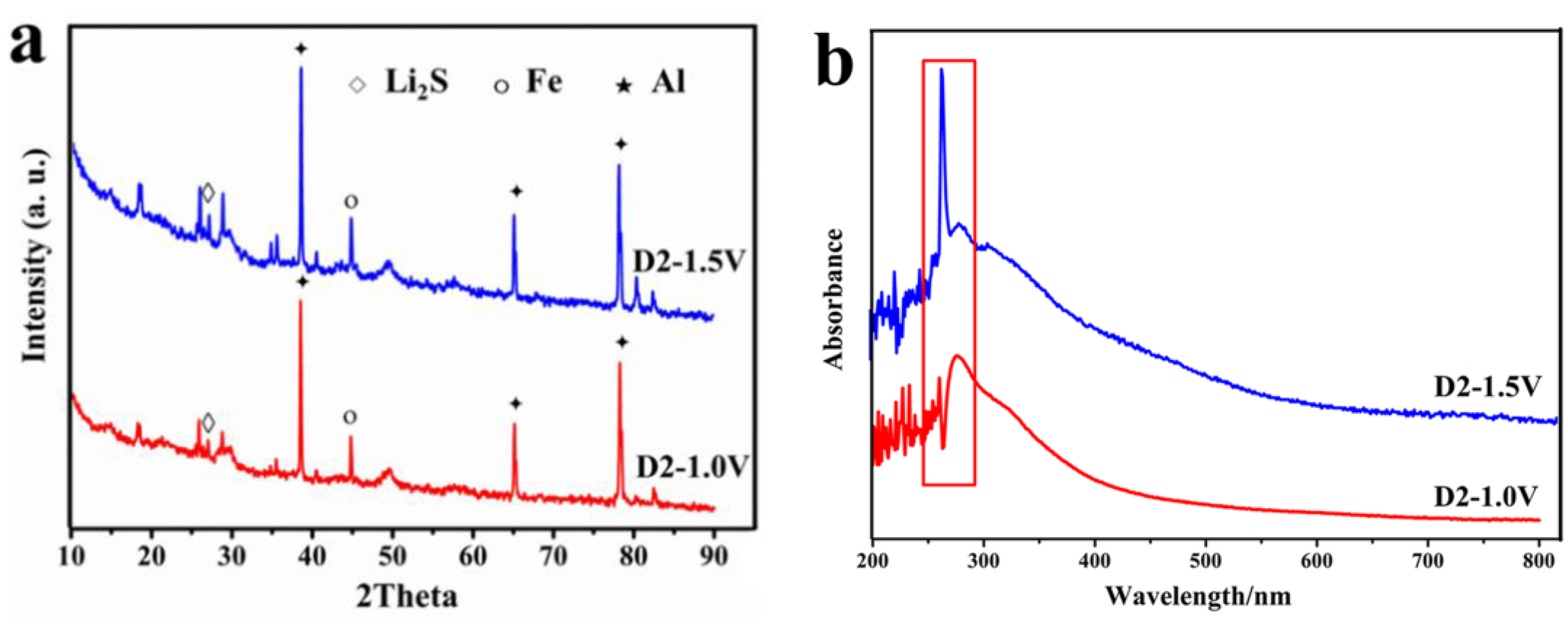
3.6. XRD and UV at Different Depths of Discharge-Charge
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhao, H.G.; Wang, S.P.; Fan, L.R.; Wu, J.P. The modification of natural pyrite and its electrochemical properties in Li/FeS2 batteries. Funct. Mater. Lett. 2014, 7. [Google Scholar] [CrossRef]
- Liu, Y.; Dang, Z.; Wu, P.; Lu, J.; Shu, X.; Zheng, L. Influence of ferric iron on the electrochemical behavior of pyrite. Ionics 2011, 17, 169–176. [Google Scholar] [CrossRef]
- Zhong-Zhi, Y.; Ling, L. Synthesis and Electrochemical Characteristics of the Novel FeS2/VGCF Material for Lithium-Ion Batteries. J. Inorg. Mater. 2013, 28, 1291–1295. [Google Scholar]
- Vu, A.; Stein, A. Lithium iron phosphate spheres as cathode materials for high power lithium ion batteries. J. Power Sources 2014, 245, 48–58. [Google Scholar] [CrossRef]
- da F.L. Amorim, B.; Moura, F.; Brocchi, E.; Vieira, M.; da C. Rupp, M. Synthesis and Characterization of Iron Disulfide for Cathodic Material. Metall. Mater. Trans. B 2012, 43, 781–786. [Google Scholar] [CrossRef]
- Zhang, D.; Mai, Y.J.; Xiang, J.Y.; Xia, X.H.; Qiao, Y.Q.; Tu, J.P. FeS2/C composite as an anode for lithium ion batteries with enhanced reversible capacity. J. Power Sources 2012, 217, 229–235. [Google Scholar] [CrossRef]
- Zhang, D.; Tu, J.P.; Mai, Y.J.; Zhang, J.; Qiao, Y.Q.; Wang, X.L. Preparation and characterization of FeS2/polyaniline composite electrode in lithium-ion battery. J. Aust. Ceram. Soc. 2012, 48, 189–193. [Google Scholar]
- Choi, Y.J.; Kang, W.G.; Ryu, H.S.; Nam, T.H.; Ahn, H.J.; Cho, K.K.; Kim, K.W.; Ryu, K.S. Effect of Fe addition on cycle performance of FeS2 cathode for Li/FeS2 battery. Mater. Technol. 2012, 27, 124–126. [Google Scholar] [CrossRef]
- Huijun, Z.; Yongzhen, Y.; Xuguang, L.; Bingshe, X. Preparation of surface molecularly imprinted matrix materials porous carbon microspheres from glucose by hydrothermal carbonization method. China Sci. Pap. 2013, 7, 898–903. [Google Scholar]
- Lu, X.; Pellechia, P.J.; Flora, J.R.V.; Berge, N.D. Influence of reaction time and temperature on product formation and characteristics associated with the hydrothermal carbonization of cellulose. Bioresour. Technol. 2013, 138, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Zhang, X.; Sun, Z.; Fang, Y.; Li, F.; Chen, K.; Huang, C. Preparation and mechanism of magnetic carbonaceous polysaccharide microspheres by low-temperature hydrothermal method. J. Magn. Magn. Mater. 2011, 323, 2741–2747. [Google Scholar] [CrossRef]
- He, X.; Li, H.; Liu, Y.; Huang, H.; Kang, Z.; Lee, S.-T. Water soluble carbon nanoparticles: Hydrothermal synthesis and excellent photoluminescence properties. Colloid Surf. B 2011, 87, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Li, Y. Colloidal Carbon Spheres and Their Core/Shell Structures with Noble-Metal Nanoparticles. Angew. Chem. Int. Ed. Engl. 2004, 43, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Miyamoto, M. Hydrothermal experiments on alteration of Ca-Al-rich inclusions (CAIs) in carbonaceous chondrites: Implication for aqueous alteration in parent asteroids. Geochim. Cosmochim. Acta 1998, 62, 3575–3588. [Google Scholar] [CrossRef]
- Shembel, E.M.; Polischuk, Y.V.; Chervakov, O.V.; Reisner, D. Reactivity of natural and synthesized FeS2 relative to the components of polymer electrolytes. Ionics 2006, 12, 41–46. [Google Scholar] [CrossRef]
- Li, H.; Zhou, H. Enhancing the performances of Li-ion batteries by carbon-coating: present and future. Chem. Commun. 2012, 48, 1201–1217. [Google Scholar] [CrossRef] [PubMed]
- Moya, A.A. Electrochemical impedance of ion-exchange systems with weakly charged membranes. Ionics 2013, 19, 1271–1283. [Google Scholar] [CrossRef]
- Schmidt, J.P.; Chrobak, T.; Ender, M.; Illig, J.; Klotz, D.; Ivers-Tiffée, E. Studies on LiFePO4 as cathode material using impedance spectroscopy. J. Power Sources 2011, 196, 5342–5348. [Google Scholar] [CrossRef]
- Andre, D.; Meiler, M.; Steiner, K.; Walz, H.; Soczka-Guth, T.; Sauer, D.U. Characterization of high-power lithium-ion batteries by electrochemical impedance spectroscopy. II: Modelling. J. Power Sources 2011, 196, 5349–5356. [Google Scholar] [CrossRef]
- Cui, Y.; Zhao, X.; Guo, R. Improved electrochemical performance of La0.7Sr0.3MnO3 and carbon co-coated LiFePO4 synthesized by freeze-drying process. Electrochimica Acta 2010, 55, 922–926. [Google Scholar] [CrossRef]
- Qu, T.; Tian, Y.-W.; Zhai, Y.-C. Measurement of diffusion coefficient of lithium in LiFePO4 cathode material for Li-ion battery by PITT and EIS. Chin. J. Nonferrous Met. 2007, 17, 1255–1295. [Google Scholar]
- Reddy, C.V.S.; Chen, M.; Jin, W.; Zhu, Q.; Chen, W.; Mho, S.-I. Characterization of (PVDF+LiFePO4) solid polymer electrolyte. J. Appl. Electrochem. 2007, 37, 637–642. [Google Scholar] [CrossRef]
- Takeuchi, T.; Kageyama, H.; Nakanishi, K.; Inada, Y.; Katayama, M.; Ohta, T.; Senoh, H.; Sakaebe, H.; Sakai, T.; Tatsumi, K.; et al. Improvement of Cycle Capability of FeS2 Positive Electrode by Forming Composites with Li2S for Ambient Temperature Lithium Batteries. J. Electrochem. Soc. 2012, 159, A75–A84. [Google Scholar] [CrossRef]
- Iwakura, C.; Isobe, N.; Tamura, H. Preparation of iron disulfide and its use for lithium batteries. Electrochimica Acta 1983, 28, 269–275. [Google Scholar] [CrossRef]
- Brec, R.; Prouzet, E.; Ouvrard, G. Redox processes in the LixFeS2/Li electrochemical system studied through crystal, Mössbauer, and EXAFS analyses. J. Power Sources 1989, 26, 325–332. [Google Scholar] [CrossRef]
- Fong, R.; Dahn, J.R.; Jones, C.H.W. Electrochemistry of pyrite-based cathodes for ambient-ambient lithium batteries. J. Electrochem. Soc. 1989, 136, 3206–3210. [Google Scholar] [CrossRef]
- Fong, R.; Jones, C.H.W.; Dahn, J.R. A study of pyrite-based cathodes for ambient temperature lithium batteries by in situ 57Fe Mossbauer spectroscopy. J. Power Sources 1989, 136, 333–339. [Google Scholar] [CrossRef]
- Zu, C.; Fu, Y.; Manthiram, A. Highly reversible Li/dissolved polysulfide batteries with binder-free carbon nanofiber electrodes. J. Mater. Chem. A 2013, 1, 10362–10367. [Google Scholar] [CrossRef]
- Ardel, G.; Golodnitsky, D.; Freedman, K.; Peled, E.; Appetecchi, G.B.; Romagnoli, P.; Scrosati, B. Rechargeable lithium/hybrid-electrolyte/pyrite battery. J. Power Sources 2002, 110, 152–162. [Google Scholar] [CrossRef]

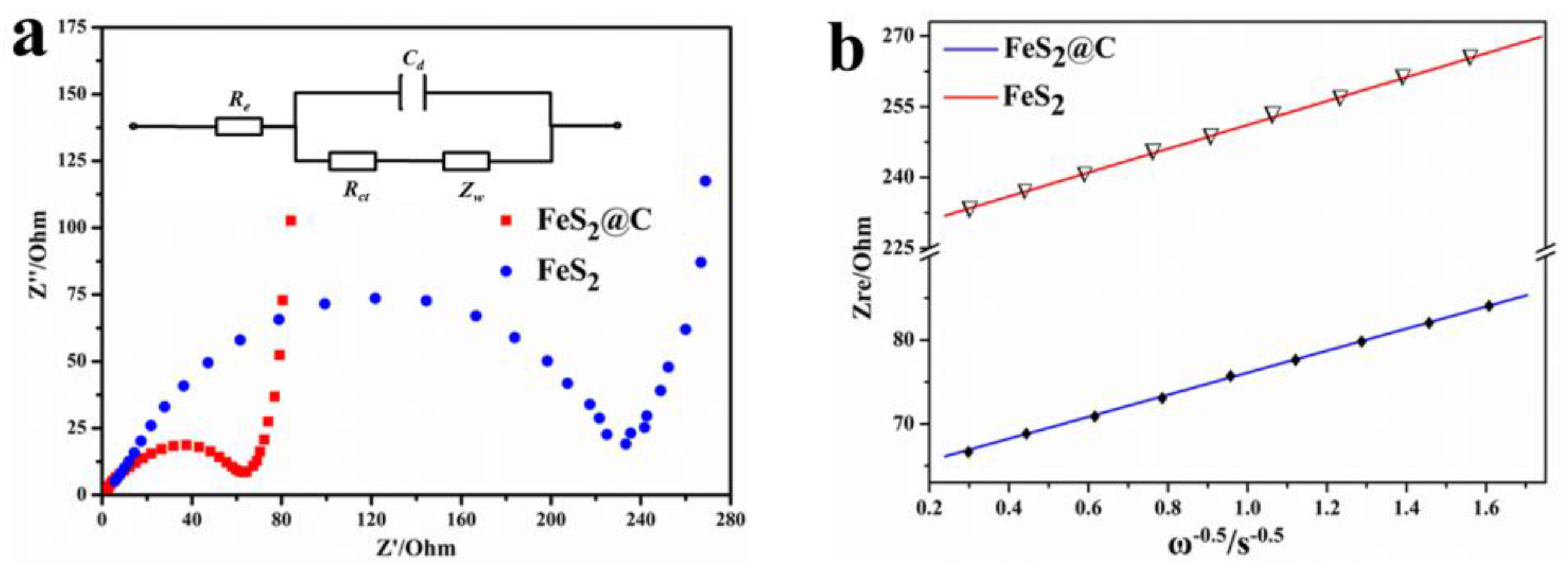
| Sample | Rct (Ω) | σω (Ω·cm2·s−0.5) | D (cm2·s−1) | σ (S·cm−1) |
|---|---|---|---|---|
| FeS2@C | 65.49 | 13.131 | 3.47 × 10−11 | 3.45 × 10−5 |
| FeS2 | 234 | 25.316 | 7.115 × 10−13 | 9.66 × 10−6 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Yu, J. Electrochemical Mechanism for FeS2/C Composite in Lithium Ion Batteries with Enhanced Reversible Capacity. Energies 2016, 9, 225. https://doi.org/10.3390/en9040225
Wang S, Yu J. Electrochemical Mechanism for FeS2/C Composite in Lithium Ion Batteries with Enhanced Reversible Capacity. Energies. 2016; 9(4):225. https://doi.org/10.3390/en9040225
Chicago/Turabian StyleWang, Shengping, and Jingxian Yu. 2016. "Electrochemical Mechanism for FeS2/C Composite in Lithium Ion Batteries with Enhanced Reversible Capacity" Energies 9, no. 4: 225. https://doi.org/10.3390/en9040225
APA StyleWang, S., & Yu, J. (2016). Electrochemical Mechanism for FeS2/C Composite in Lithium Ion Batteries with Enhanced Reversible Capacity. Energies, 9(4), 225. https://doi.org/10.3390/en9040225





