Recent Advances in the Use of Sodium Borohydride as a Solid State Hydrogen Store
Abstract
:1. Introduction
| Borohydride | Cost a/(USD/g) | Hydrogen density/wt% | Td b/°C | Reaction | References |
|---|---|---|---|---|---|
| NaBH4 | 6.47 | 10.6 | 505 | NaBH4 → Na + B + 2H2 | [6,7,8] |
| LiBH4 | 15.65 | 18.5 | 380 | LiBH4 → Li + B + 2H2 | [9,10,11,12] |
| Mg(BH4)2 | 116.5 | 14.9 | 320 | Mg(BH4)2 → MgB2 + 4H2 | [13,14,15] |
| Ca(BH4)2 | 142 | 11.6 | 367 | Ca(BH4)2 → 2/3CaH2 + 1/3CaB6 + 10/3H2 | [16,17,18] |
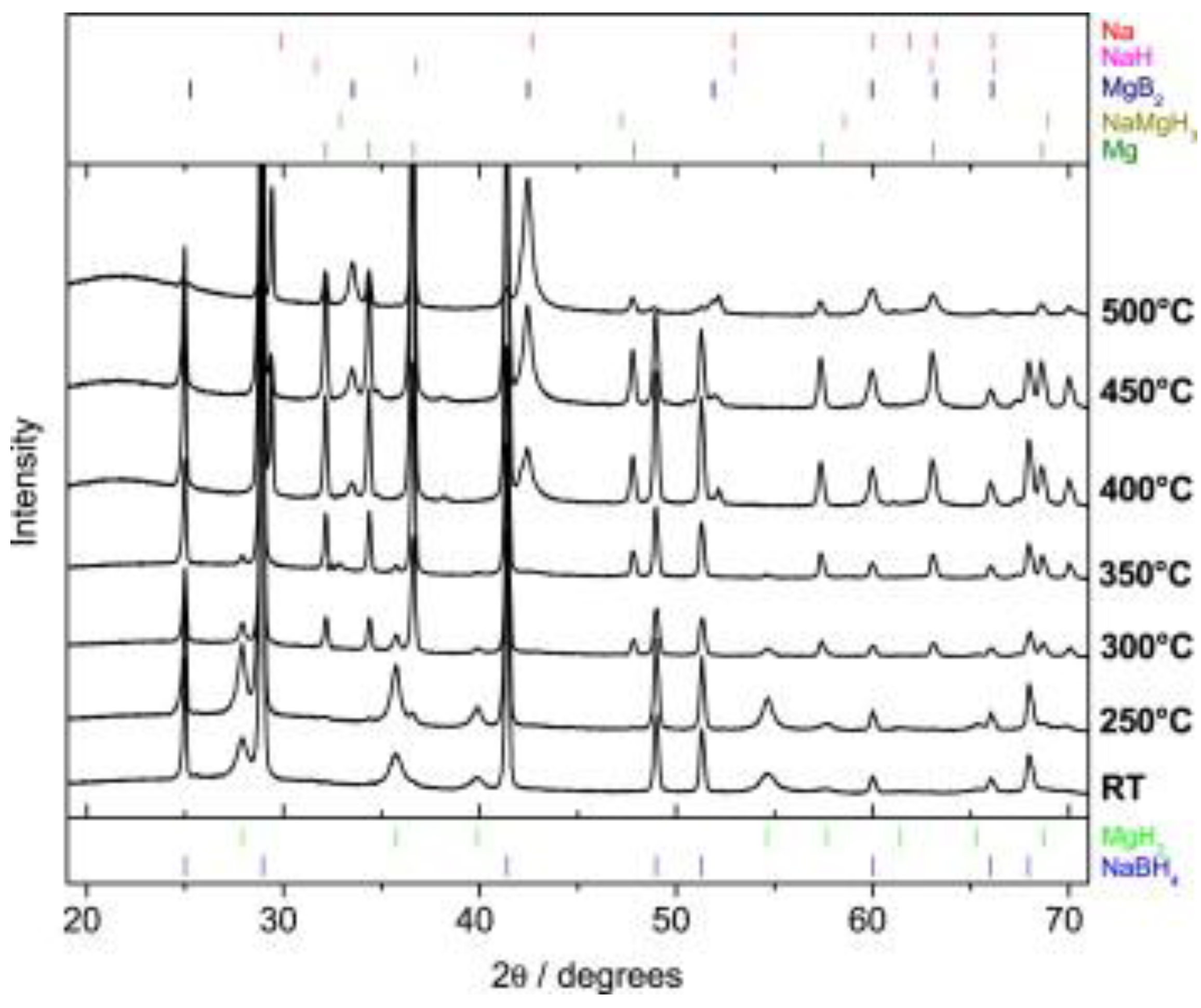
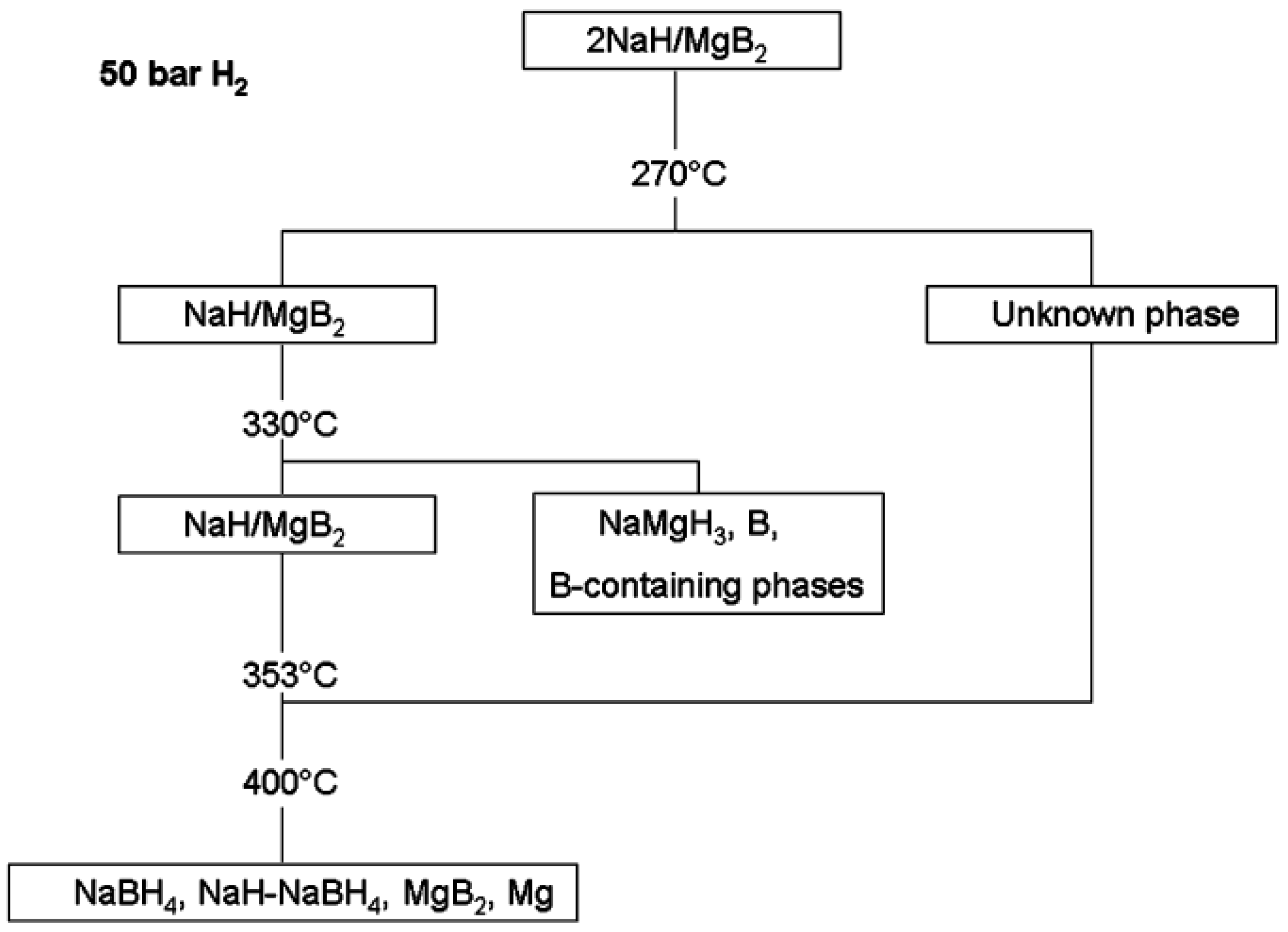
2. Thermal Decomposition
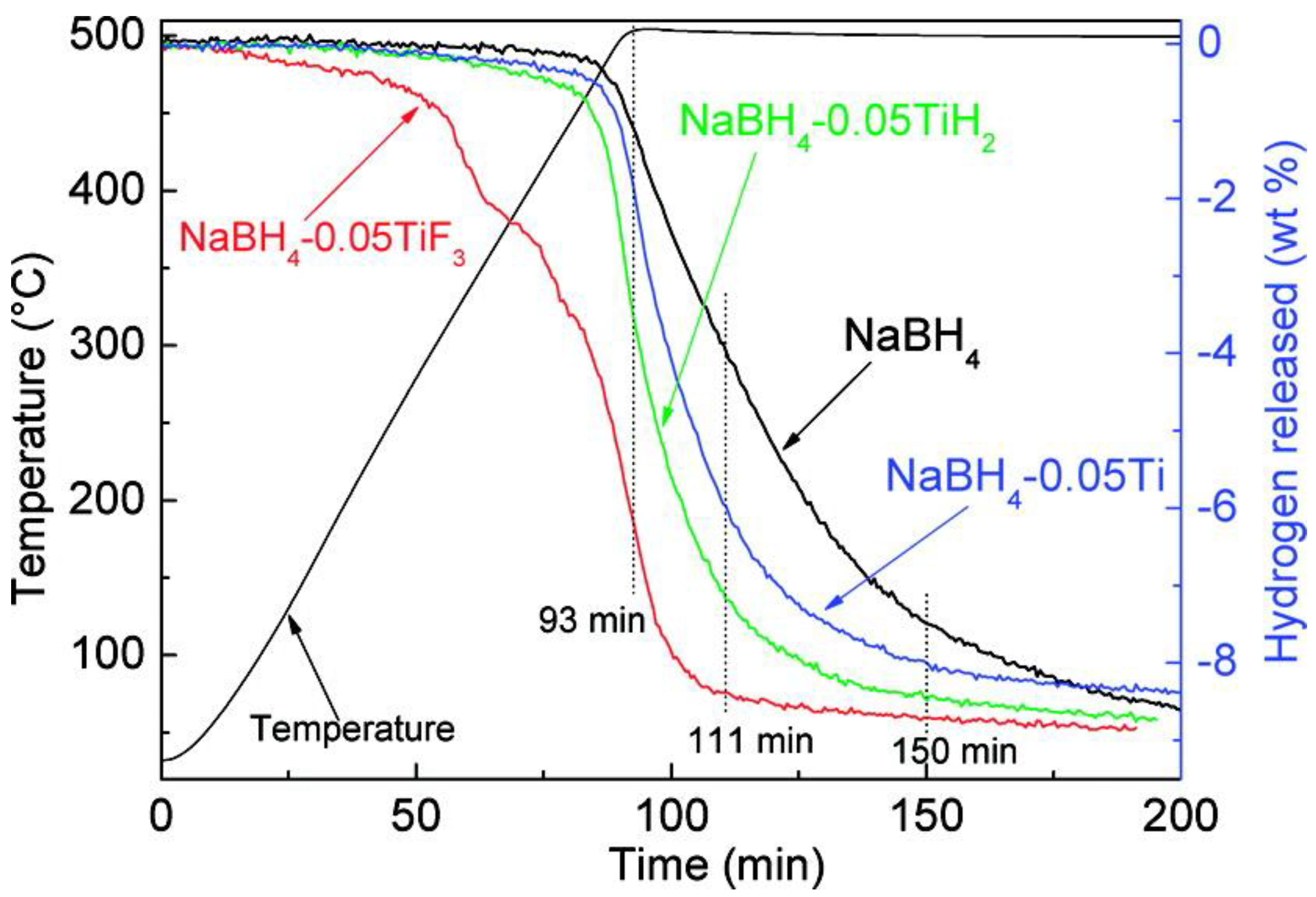
3. Strategies for Promoting H2 Release from Solid-State Thermolysis of NaBH4
3.1. Catalytic Doping
3.2. Nano-Engineering

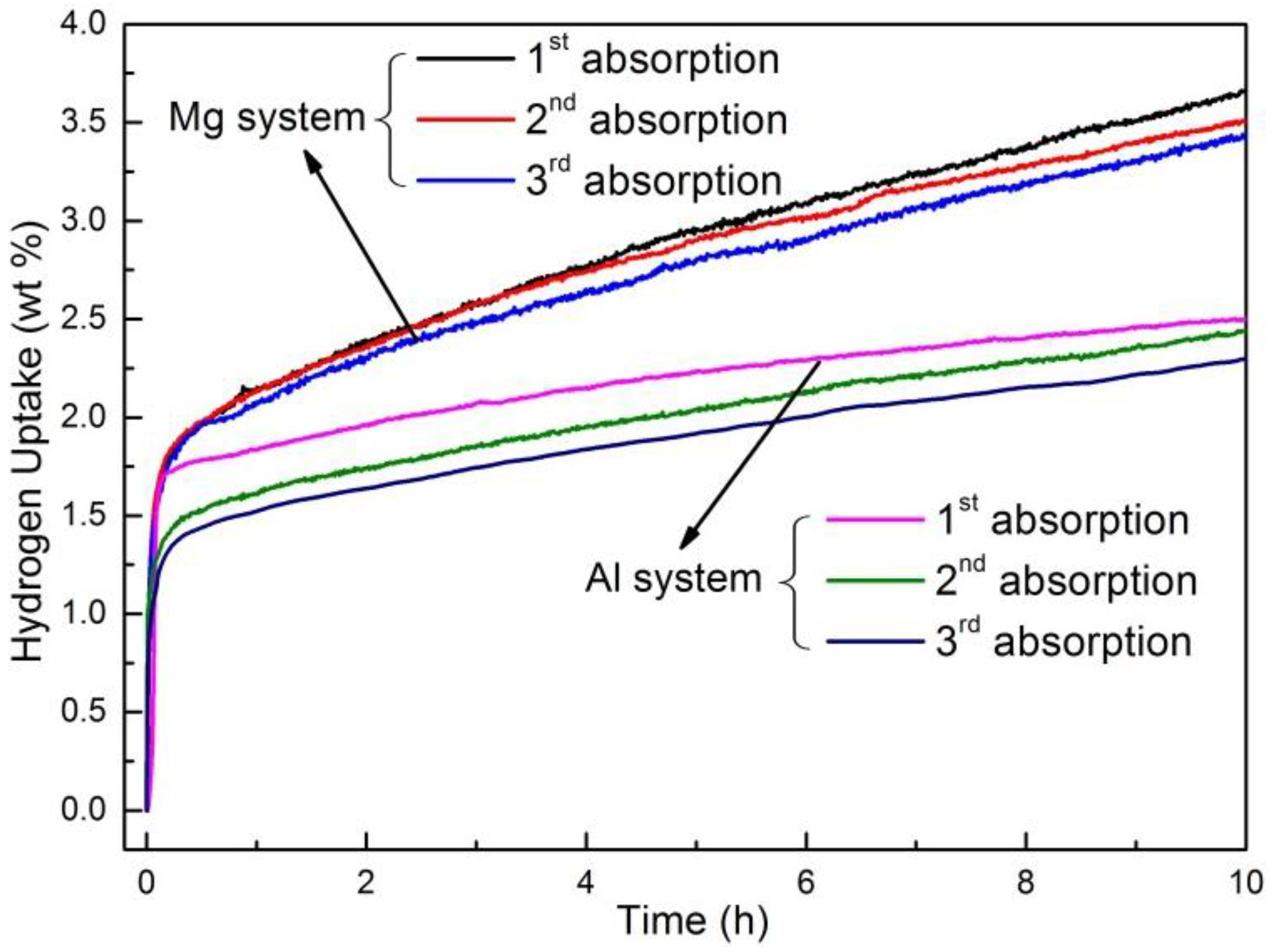
3.3. Destabilization Using Reactive Additives
3.3.1. Hydride Destabilization
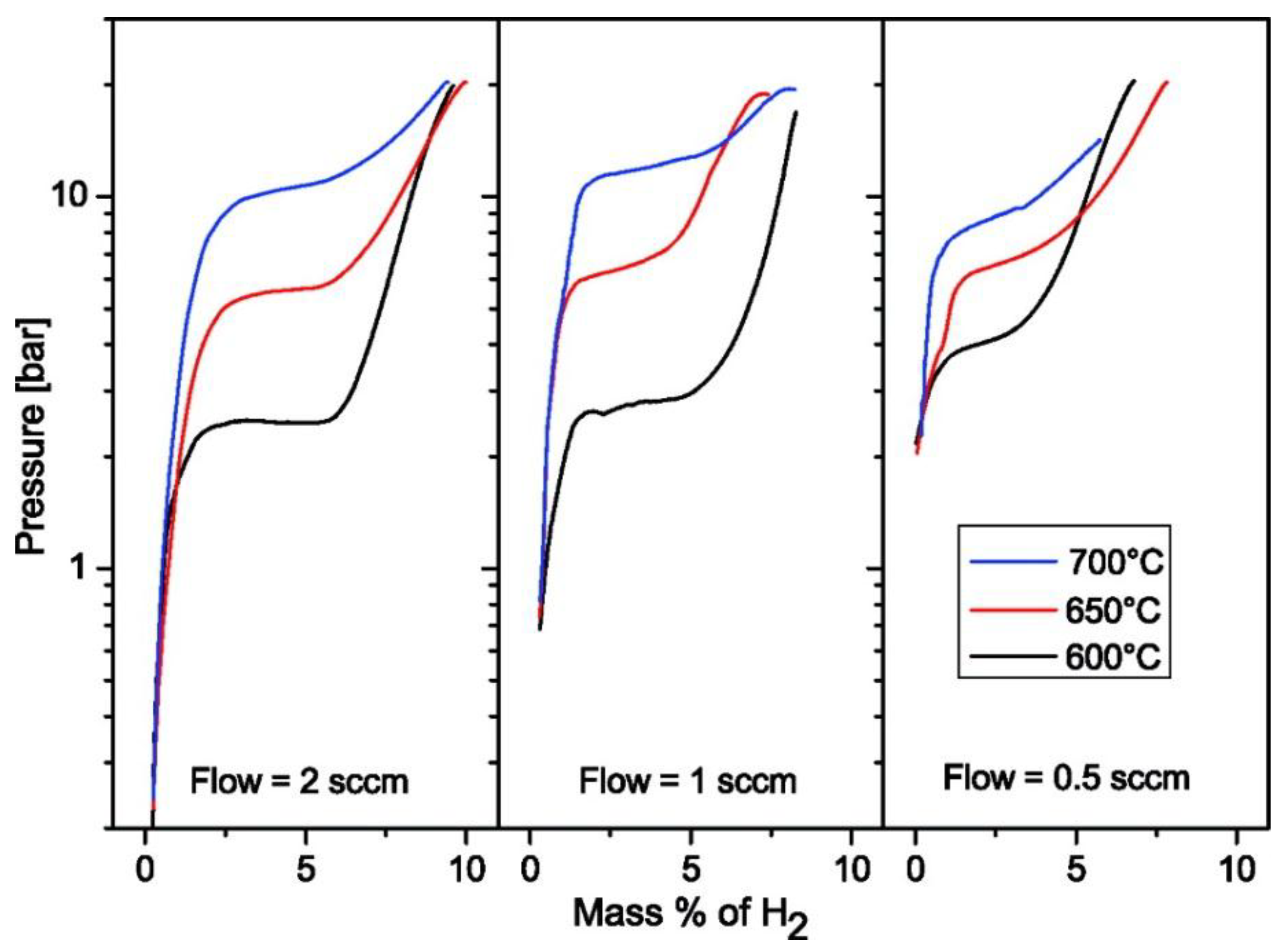
3.3.2. Fluoride Destabilization
3.4. Chemical Modification
3.4.1. Combination of Protic and Hydridic H Atoms
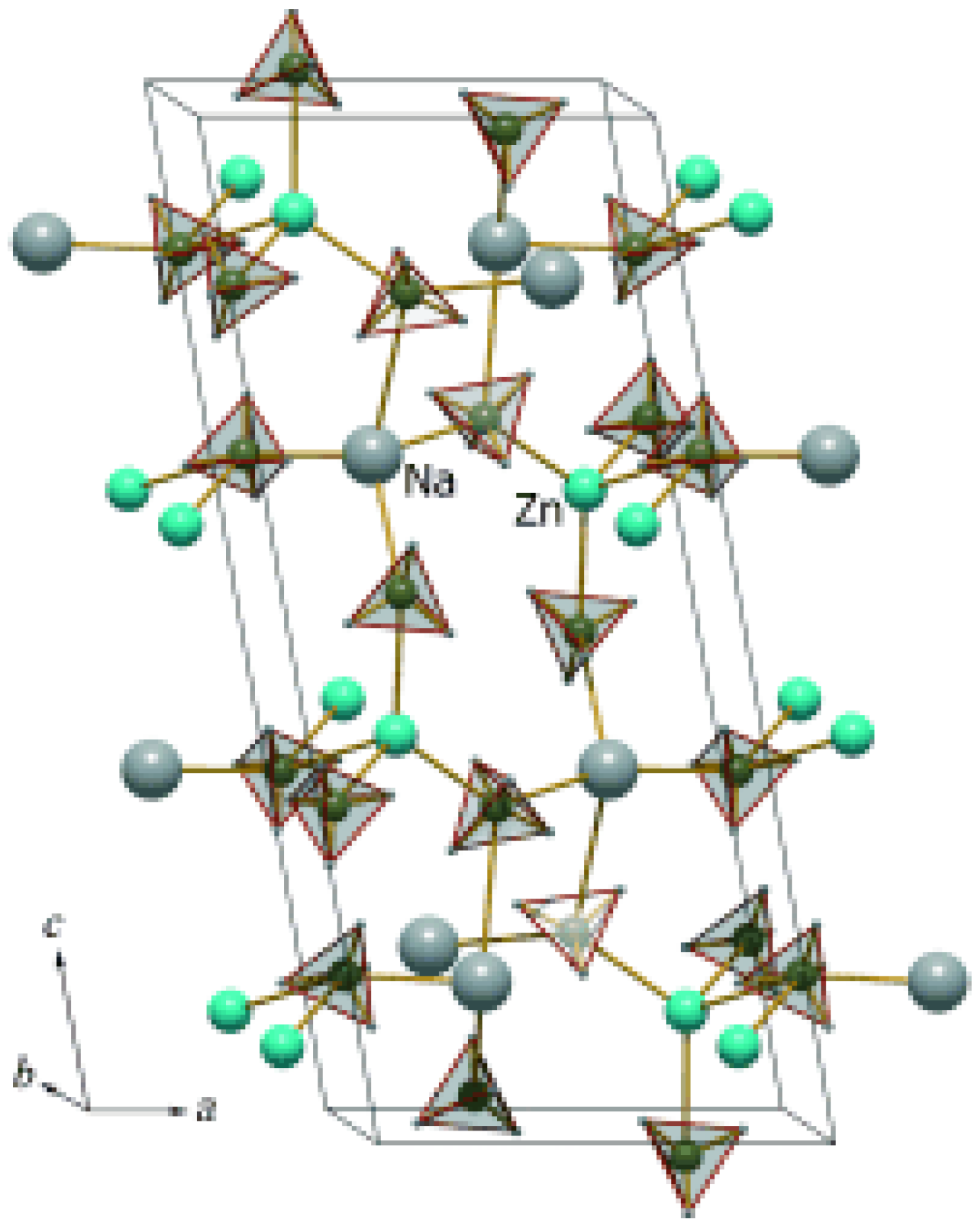
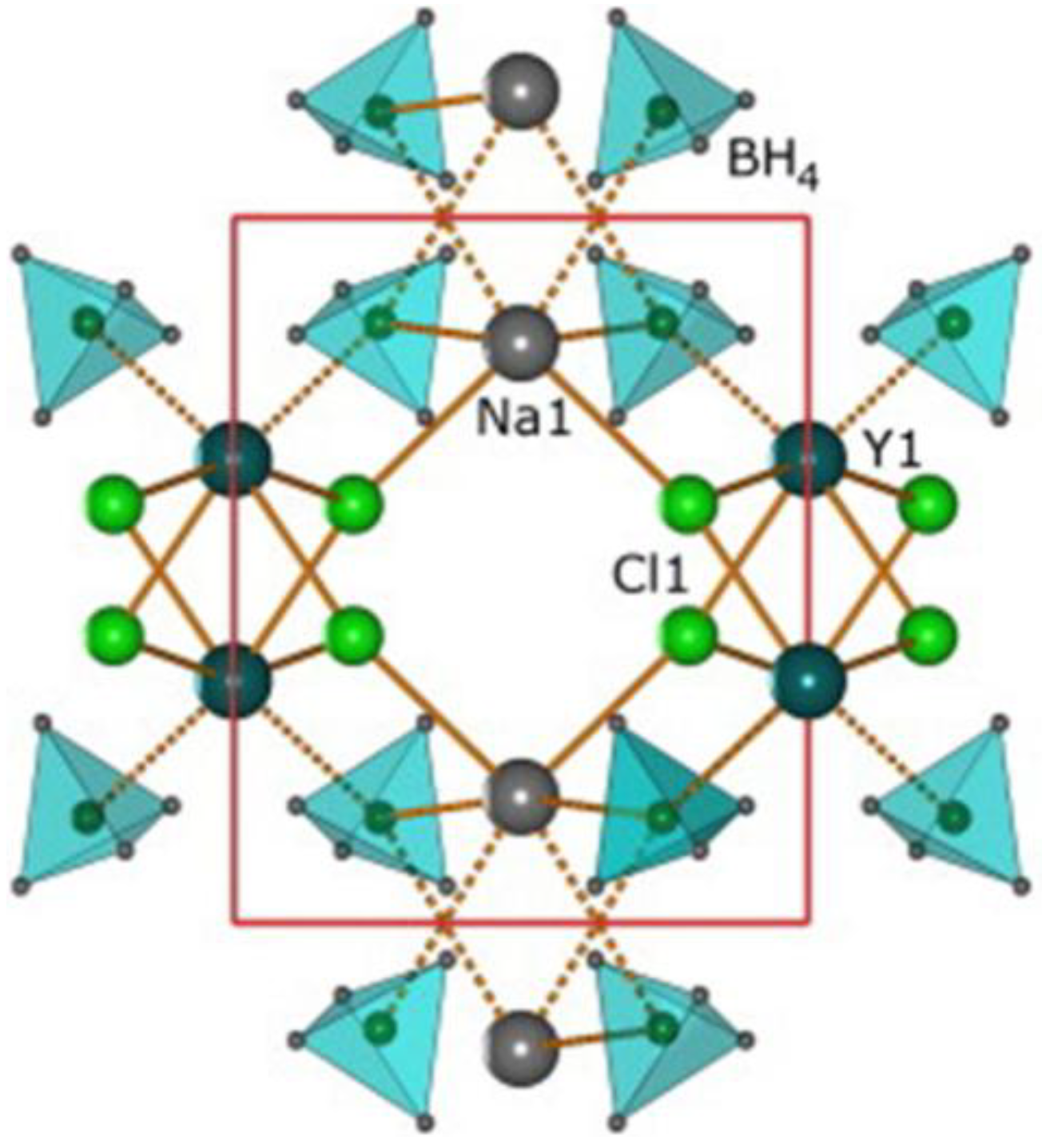
3.4.2. Bimetallic Borohydrides

| Starting reagents | Na(BH4)1−xClx cell parameter, a/Å | Cl content, x | Decomposition T/°C | Mass loss at T ≤ 600 °C/wt% |
|---|---|---|---|---|
| NaCl only | 5.6400(5) | 1 | - | - |
| NaBH4/TiCl2 | 5.7685(3) | 0.71 | 401 | 2.7 |
| NaBH4/VCl3 | 5.7306(4) | 0.79 | 391 | 3.0 |
| NaBH4/CrCl3 | 5.7383(2) | 0.77 | 397 | - |
| NaBH4/MnCl2 | 5.7863(4) | 0.68 | 146 | 5.7 |
| NaBH4/FeCl3 | 5.7407(4) | 0.77 | 397 | 0.2 |
| NaBH4/CoCl2 | 5.8011(3) | 0.65 | 413 | 3.6 |
| NaBH4/NiCl2 | 5.7837(9) | 0.68 | 391 | 4.0 |
| NaBH4/CuCl2 | 5.7801(3) | 0.69 | 343 | 3.7 |
| NaBH4/ZnCl2 | 5.6576(2) | 0.92 | 103 | 21.7 |
| NaBH4/CdCl2 | 5.7572(8) | 0.74 | 521 | - |
| NaBH4 only | 6.13080(10) | 0 | - | - |
4. Closing Remarks
| Strategy | Td a | Kinetics | wt% H2 b | Cyclability | References |
|---|---|---|---|---|---|
| Catalytic doping | + | + | − | + | [32,33] |
| Nano-confinement | + | + | − | + | [29,36,37,38,39] |
| H− destabilisation | + | + | + | + | [27,41,42,43,44,45] |
| F− destabilisation | + | + | − | + | [40,59,60,61,62,63,64,65] |
| Hδ+-Hδ− “composites” | + | + | − | − | [66,67,68,69,70,71] |
| Bimetallic Na borohydrides | + | + | − | − | [74,75,76,77,78,79,80] |
Acknowledgments
Conflicts of Interest
References
- Schlapbach, L.; Züttel, A. Hydrogen-storage materials for mobile applications. Nature 2001, 414, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Eberle, U.; Felderhoff, M.; Schüth, F. Chemical and physical solutions for hydrogen storage. Angew. Chem. Int. Ed. 2009, 48, 6608–6630. [Google Scholar] [CrossRef]
- Yang, J.; Sudik, A.; Wolverton, C.; Siegel, D.J. High capacity hydrogen storage materials: Attributes for automotive applications and techniques for materials discovery. Chem. Soc. Rev. 2010, 39, 656–675. [Google Scholar] [CrossRef] [PubMed]
- Reardon, H.; Hanlon, J.M.; Hughes, R.W.; Godula-Jopek, A.; Mandal, T.K.; Gregory, D.H. Emerging concepts in solid-state hydrogen storage the role of nanomaterials design. Energy Environ. Sci. 2012, 5, 5951–5979. [Google Scholar] [CrossRef]
- U.S. Department of Energy, Office of Energy Efficiency and Renewable Energy and the FreedomCAR and Fuel Partnership. Technical System Targets: Onboard Hydrogen Storage for Light-Duty Fuel Cell Vehicles. Available online: http://energy.gov/sites/prod/files/2014/03/f12/targets_onboard_hydro_storage.pdf (accessed on 23 December 2014).
- Urgnani, J.; Torres, F.J.; Palumbo, M.; Baricco, M. Hydrogen release from solid state NaBH4. Int. J. Hydrog. Energy 2008, 33, 3111–3115. [Google Scholar] [CrossRef]
- Martelli, P.; Caputo, R.; Remhof, A.; Mauron, P.; Borgschulte, A.; Züttel, A. Stability and decomposition of NaBH4. J. Phys. Chem. C 2010, 114, 7173–7177. [Google Scholar] [CrossRef]
- Milanese, C.; Garroni, S.; Girella, A.; Mulas, G.; Berbenni, V.; Bruni, G.; Suriach, S.; Baró, M.D.; Marini, A. Thermodynamic and kinetic investigations on pure and doped NaBH4-MgH2 system. J. Phys. Chem. C 2011, 115, 3151–3162. [Google Scholar] [CrossRef]
- Züttel, A.; Rentsch, S.; Fischer, P.; Wenger, P.; Sudan, P.; Mauron, P.; Emmenegger, C. Hydrogen storage properties of LiBH4. J. Alloys Compd. 2003, 356–357, 515–520. [Google Scholar]
- Mao, J.F.; Guo, Z.P.; Liu, H.K.; Yu, X.B. Reversible hydrogen storage in titanium-catalyzed LiAlH4–LiBH4 system. J. Alloys Compd. 2009, 487, 434–438. [Google Scholar] [CrossRef]
- Mauron, P.; Buchter, F.; Friedrichs, O.; Remhof, A.; Bielmann, M.; Christoph, N.Z.; Züttel, A. Stability and reversibility of LiBH4. J. Phys. Chem. B 2008, 112, 906–910. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.B.; Grant, D.M.; Walker, G.S. Dehydrogenation of LiBH4 destabilized with various oxides. J. Phys. Chem. C 2009, 113, 17945–17949. [Google Scholar] [CrossRef]
- Chłopek, K.; Frommen, C.; Léon, A.; Zabara, O.; Fichtner, M. Synthesis and properties of magnesium tetrahydroborate, Mg(BH4)2. J. Mater. Chem. 2007, 17, 3496–3503. [Google Scholar] [CrossRef]
- Li, H.W.; Kikuchi, K.; Nakamori, Y.; Miwa, K.; Towata, S.; Orimo, S. Effects of ball milling and additives on dehydriding behaviors of well-crystallized Mg(BH4)2. Scr. Mater. 2007, 57, 679–682. [Google Scholar] [CrossRef]
- Newhouse, R.J.; Stavila, V.; Hwang, S.-J.; Klebanoff, L.E.; Zhang, J.Z. Reversibility and improved hydrogen release of magnesium borohydride. J. Phys. Chem. C 2010, 114, 5224–5232. [Google Scholar] [CrossRef]
- Mao, J.F.; Guo, Z.P.; Poh, C.K.; Ranjbar, A.; Guo, Y.H.; Yu, X.B.; Liu, H.K. Study on the dehydrogenation kinetics and thermodynamics of Ca(BH4)2. J. Alloys Compd. 2010, 500, 200–205. [Google Scholar] [CrossRef]
- Kim, Y.; Hwang, S.-J.; Lee, Y.S.; Suh, J.Y.; Han, H.N.; Cho, Y.W. Hydrogen back-pressure effects on the dehydrogenation reactions of Ca(BH4)2. J. Phys. Chem. C 2012, 116, 25715–25720. [Google Scholar] [CrossRef]
- Riktor, M.D.; Sørby, M.H.; Chopek, K.; Fichtner, M.; Haubac, B.C. The identification of a hitherto unknown intermediate phase CaB2HX from decomposition of Ca(BH4)2. J. Mater. Chem. 2009, 19, 2754–2759. [Google Scholar] [CrossRef]
- Orimo, S.; Nakamori, Y.; Eliseo, J.; Züttel, A.; Jensen, C.M. Complex hydrides for hydrogen storage. Chem. Rev. 2007, 107, 4111–4132. [Google Scholar] [CrossRef] [PubMed]
- Li, H.W.; Yan, Y.G.; Orimo, S.; Züttel, A.; Jensen, C.M. Recent progress in metal borohydrides for hydrogen storage. Energies 2011, 4, 185–214. [Google Scholar] [CrossRef]
- Muir, S.S.; Yao, X.D. Progress in sodium borohydride as a hydrogen storage material: Development of hydrolysis catalysts and reaction systems. Int. J. Hydrog. Energy 2011, 36, 5983–5997. [Google Scholar] [CrossRef]
- U.S. Department of Energy Hydrogen Program. Go/No-Go Recommendation for Sodium Borohydride for On-Board Vehicular Hydrogen Storage; NREL/MP-150-42220; National Renewable Energy Laboratory (NREL): Golden, CO, USA, 2007. [Google Scholar]
- Sigma-Aldrich. Available online: http://www.sigmaaldrich.com/united-states (accessed on 19 October 2014).
- Santos, D.M.F.; Sequeira, C.A.C. Sodium borohydride as a fuel for the future. Renew. Sustain. Energy Rev. 2011, 15, 3980–4001. [Google Scholar] [CrossRef]
- Cakır, D.; Wijs, G.A.D.; Brocks, G. Native defects and the dehydrogenation of NaBH4. J. Phys. Chem. C 2011, 115, 24429–24434. [Google Scholar] [CrossRef]
- Grochala, W.; Edwards, P. Thermal decomposition of the non-interstitial hydrides for the storage and production of hydrogen. Chem. Rev. 2004, 104, 1283–1315. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Guo, Z.; Yu, X.; Liu, H. Improved hydrogen storage properties of NaBH4 destabilized by CaH2 and Ca(BH4)2. J. Phys. Chem. C 2011, 115, 9283–9290. [Google Scholar] [CrossRef]
- Garroni, S.; Milanese, C.; Pottmaier, D.; Mulas, G.; Nolis, P.; Girella, A.; Caputo, R.; Olid, D.; Teixdor, F.; Baricco, M.; et al. Experimental evidence of Na2[B12H12] and Na formation in the desorption pathway of the 2NaBH4 + MgH2 System. J. Phys. Chem. C 2011, 115, 16664–16671. [Google Scholar] [CrossRef]
- Ngene, P.; van den Berg, R.; Verkuijlen, M.H.W.; de Jong, K.P.; de Jongh, P.E. Reversibility of the hydrogen desorption from NaBH4 by confinement in nanoporous carbon. Energy Environ. Sci. 2011, 4, 4108–4115. [Google Scholar] [CrossRef]
- Friedrichs, O.; Remhof, A.; Hwang, S.-J.; Züttel, A. Role of Li2B12H12 for the formation and decomposition of LiBH4. Chem. Mater. 2009, 22, 3265–3268. [Google Scholar] [CrossRef]
- Caputo, R.; Garroni, S.; Olid, D.; Teixidor, F.; Suriňach, S.; Dolors Baró, M. Can Na2[B12H12] be a decomposition product of NaBH4? Phys. Chem. Chem. Phys. 2010, 12, 15093–15100. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.F.; Guo, Z.P.; Nevirkovets, I.P.; Liu, H.K.; Dou, S.X. Hydrogen de-/absorption improvement of NaBH4 catalyzed by titanium-based additives. J. Phys. Chem. C 2012, 116, 1596–1604. [Google Scholar] [CrossRef]
- Humphries, T.D.; Kalantzopoulos, G.N.; Llamas-Jansa, I.; Olsen, J.E.; Hauback, B.C. Reversible hydrogenation studies of NaBH4 milled with Ni-containing additives. J. Phys. Chem. C 2013, 117, 6060–6065. [Google Scholar] [CrossRef]
- De Jongh, P.E.; Adelhelm, P. Nanosizing and nanoconfinement: New strategies towards meeting hydrogen storage goals. ChemSusChem 2010, 3, 1332–1348. [Google Scholar] [CrossRef] [PubMed]
- Varin, R.A.; Chiu, C. Structural stability of sodium borohydride (NaBH4) during controlled mechanical milling. J. Alloys Compd. 2005, 397, 276–281. [Google Scholar] [CrossRef]
- Ampoumogli, A.; Steriotis, T.; Trikalitis, P.; Giasafaki, D.; Bardaji, E.G.; Fichtner, M.; Charalambopoulou, G. Nanostructured composites of mesoporous carbons and boranates as hydrogen storage materials. J. Alloys Compd. 2011, 509, S705–S708. [Google Scholar] [CrossRef]
- Peru, F.; Garroni, S.; Campesi, R.; Milanese, C.; Marini, A.; Pellicer, E.; Baró, M.D.; Mulas, G. Ammonia-free infiltration of NaBH4 into highly-ordered mesoporous silica and carbon matrices for hydrogen storage. J. Alloys Compd. 2013, 580, S309–S312. [Google Scholar] [CrossRef]
- Christian, M.L.; Aguey-Zinsou, K.F. Core–shell strategy leading to high reversible hydrogen storage capacity for NaBH4. ACS Nano 2012, 6, 7739–7751. [Google Scholar] [CrossRef] [PubMed]
- Christian, M.L.; Aguey-Zinsou, K.F. Synthesis of core–shell NaBH4@M (M = Co, Cu, Fe, Ni, Sn) nanoparticles leading to various morphologies and hydrogen storage properties. Chem. Commun. 2013, 49, 6794–6796. [Google Scholar] [CrossRef]
- Zhang, L.T.; Xiao, X.Z.; Fan, X.L.; Li, S.Q.; Ge, H.W.; Wang, Q.D.; Chen, L.X. Fast hydrogen release under moderate conditions from NaBH4 destabilized by fluorographite. RSC Adv. 2014, 4, 2550–2556. [Google Scholar] [CrossRef]
- Mao, J.F.; Yu, X.B.; Guo, Z.P.; Liu, H.K.; Wu, Z.; Ni, J. Enhanced hydrogen storage performances of NaBH4–MgH2 system. J. Alloys Compd. 2009, 479, 619–623. [Google Scholar] [CrossRef]
- Garroni, S.; Pistidda, C.; Brunelli, M.; Vaughan, G.B.M.; Suriňach, S.; Baró, M.D. Hydrogen desorption mechanism of 2NaBH4 + MgH2 composite prepared by high-energy ball milling. Scr. Mater. 2009, 60, 1129–1132. [Google Scholar] [CrossRef]
- Dornheim, M. Tailoring Reaction Enthalpies of Hydrides. In Handbook of Hydrogen Storage; Hirscher, M., Ed.; Wiley-VCH: New York, NY, USA, 2010; pp. 187–214. [Google Scholar]
- Mao, J.F.; Yu, X.B.; Guo, Z.P.; Poh, C.K.; Liu, H.K.; Wu, Z.; Ni, J. Improvement of the LiAlH4-NaBH4 system for reversible hydrogen storage. J. Phys. Chem. C 2009, 113, 10813–10818. [Google Scholar] [CrossRef]
- Afonso, G.; Bonakdarpour, A.; Wilkinson, D.P. Hydrogen storage properties of the destabilized 4NaBH4/5Mg2NiH4 composite system. J. Phys. Chem. C 2013, 117, 21105–21111. [Google Scholar] [CrossRef]
- Li, G.Q.; Matsuo, M.; Deledda, S.; Hauback, B.C.; Orimo, S. Dehydriding property of NaBH4 combined with Mg2FeH6. Mater. Trans. 2014, 55, 1141–1143. [Google Scholar] [CrossRef]
- Garroni, S.; Milanese, C.; Girell, A.; Marini, A.; Mulas, G.; Meneňdez, E.; Pistidda, C.; Dornheim, M.; Suriňach, S.; Baró, M.D. Sorption properties of NaBH4/MH2 (M = Mg, Ti) powder systems. Int. J. Hydrog. Energy 2010, 35, 5434–5441. [Google Scholar] [CrossRef]
- Pottmaier, D.; Pistidda, C.; Groppo, E.; Bordiga, S.; Spoto, G.; Dornheim, M.; Baricco, M. Dehydrogenation reactions of 2NaBH4 + MgH2 system. Int. J. Hydrog. Energy 2011, 36, 7891–7896. [Google Scholar] [CrossRef]
- Nwakwuo, C.C.; Pistidda, C.; Dornheim, M.; Hutchison, J.L.; Sykes, J.M. Microstructural analysis of hydrogen absorption in 2NaH + MgB2. Scr. Mater. 2011, 64, 351–354. [Google Scholar] [CrossRef]
- Pistidda, C.; Garroni, S.; Minella, C.; Dolci, F.; Jensen, T.R.; Nolis, P.; Bösenberg, U.; Cerenius, Y.; Lohstroh, W.; Fichtner, M.; et al. Pressure effect on the 2NaH + MgB2 hydrogen absorption reaction. J. Phys. Chem. C 2010, 114, 21816–21823. [Google Scholar] [CrossRef]
- Pistidda, C.; Pottmaier, D.; Karimi, F.; Garroni, S.; Rzeszutek, A.; Tolkiehn, M.; Fichtner, M.; Lohstroh, W.; Baricco, M.; Klassen, T.; et al. Effect of NaH/MgB2 ratio on the hydrogen absorption kinetics of the system NaH + MgB2. Int. J. Hydrog. Energy 2014, 39, 5030–5036. [Google Scholar] [CrossRef]
- Garroni, S.; Minella, C.B.; Pottmaier, D.; Pistidda, C.; Milanese, C.; Marini, A.; Enzo, S.; Mulas, G.; Dornheim, M.; Baricco, M.; et al. Mechanochemical synthesis of NaBH4 starting from NaH-MgB2 reactive hydride composite system. Int. J. Hydrog. Energy 2013, 38, 2363–2369. [Google Scholar] [CrossRef]
- Pistidda, C.; Barkhordarian, G.; Rzeszutek, A.; Garroni, S.; Bonatto Minella, C.; Baró, M.D.; Nolis, P.; Bormann, R.; Klassen, T.; Dornheim, M. Activation of the reactive hydride composite 2NaBH4 + MgH2. Scr. Mater. 2011, 64, 1035–1038. [Google Scholar] [CrossRef]
- Mulas, G.; Campesi, R.; Garroni, S.; Napolitano, E.; Milanese, C.; Dolci, F.; Pellicer, E.; Baró, D.; Marini, A. Hydrogen storage in 2NaBH4 + MgH2 mixtures: Destabilization by additives and nanoconfinement. J. Alloys Compd. 2012, 536, S236–S240. [Google Scholar] [CrossRef]
- Lemke, C.H. Sodium and sodium alloys. In Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons: New York, NY, USA, 1983; pp. 181–204. [Google Scholar]
- Young, J.A. Sodium fluoride. J. Chem. Educ. 2002, 79. [Google Scholar] [CrossRef]
- Yin, L.C.; Wang, P.; Kang, X.D.; Sun, C.H.; Cheng, H.M. Functional anion concept: Effect of fluorine anion on hydrogen storage of sodium alanate. Phys. Chem. Chem. Phys. 2007, 9, 1499–1502. [Google Scholar] [CrossRef] [PubMed]
- Rude, L.H.; Filsø, U.; D’Anna, V.; Spyratou, A.; Richter, B.; Hino, S.; Zavorotynska, O.; Baricco, M.; Sørby, M.H.; Hauback, B.C.; et al. Hydrogen–fluorine exchange in NaBH4–NaBF4. Phys. Chem. Chem. Phys. 2013, 15, 18185–18194. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.F.; Guo, Z.P.; Liu, H.K.; Dou, S.X. Reversible storage of hydrogen in NaF–MB2 (M = Mg, Al) composites. J. Mater. Chem. A 2013, 1, 2806–2811. [Google Scholar] [CrossRef]
- Zhang, Z.G.; Wang, H.; Zhu, M. Hydrogen release from sodium borohydrides at low temperature by the addition of zinc fluoride. Int. J. Hydrog. Energy 2011, 36, 8203–8208. [Google Scholar] [CrossRef]
- Kalantzopoulos, G.N.; Guzik, M.N.; Deledda, S.; Heyn, R.H.; Mullera, J.; Hauback, B.C. Destabilization effect of transition metal fluorides on sodium borohydride. Phys. Chem. Chem. Phys. 2014, 16, 20483–20491. [Google Scholar] [CrossRef] [PubMed]
- Chong, L.N.; Zou, J.X.; Zeng, X.Q.; Ding, W.J. Mechanisms of reversible hydrogen storage in NaBH4 through NdF3 addition. J. Mater. Chem. A 2013, 1, 3983–3991. [Google Scholar]
- Zou, J.X.; Li, L.J.; Zeng, X.Q.; Ding, W.J. Reversible hydrogen storage in a 3NaBH4/YF3 composite. Int. J. Hydrog. Energy 2012, 37, 17118–17125. [Google Scholar] [CrossRef]
- Chong, L.N.; Zou, J.X.; Zeng, X.Q.; Ding, W.J. Effects of La fluoride and La hydride on the reversible hydrogen sorption behaviors of NaBH4: A comparative study. J. Mater. Chem. A 2014, 2, 8557–8570. [Google Scholar] [CrossRef]
- Chong, L.N.; Zou, J.X.; Zeng, X.Q.; Ding, W.J. Study on reversible hydrogen sorption behaviors of a 3NaBH4/HoF3 composite. Int. J. Hydrog. Energy 2014, 39, 14275–14281. [Google Scholar] [CrossRef]
- Lu, J.; Fang, Z.Z.; Sohn, H.Y. A dehydrogenation mechanism of metal hydrides based on interactions between Hδ+ and H−. Inorg. Chem. 2006, 45, 8749–8754. [Google Scholar] [CrossRef] [PubMed]
- Chater, P.A.; Anderson, P.A.; Prendergast, J.W.; Walton, A.; Mann, V.S.J.; Book, D.; David, W.I.F.; Johnson, S.R.; Edwards, P.P. Synthesis and characterization of amide–borohydrides: New complex light hydrides for potential hydrogen storage. J. Alloys Compd. 2007, 446–447, 350–354. [Google Scholar]
- Somer, M.; Acar, S.; Koz, C.; Kokal, I.; Hohn, P.; Cardoso-Gil, R.; Aydemir, U.; Akselrud, L. α- and β-Na2[BH4][NH2]: Two modifications of a complex hydride in the system NaNH2–NaBH4; syntheses, crystal structures, thermal analyses, mass and vibrational spectra. J. Alloys Compd. 2010, 491, 98–105. [Google Scholar] [CrossRef]
- Wu, C.; Bai, Y.; Yang, J.H.; Wu, F.; Long, F. Characterizations of composite NaNH2–NaBH4hydrogen storage materials synthesized via ball milling. Int. J. Hydrog. Energy 2012, 37, 889–893. [Google Scholar] [CrossRef]
- Drozd, V.; Saxena, S.; Garimella, S.V.; Durygin, A. Hydrogen release from a mixture of NaBH4 and Mg(OH)2. Int. J. Hydrog. Energy 2007, 32, 3370–3375. [Google Scholar] [CrossRef]
- Varin, R.A.; Parviz, R. Hydrogen generation from the ball milled composites of sodium and lithium borohydride (NaBH4/LiBH4) and magnesium hydroxide (Mg(OH)2) without and with the nanometric nickel (Ni) additive. Int. J. Hydrog. Energy 2012, 37, 1584–1293. [Google Scholar] [CrossRef]
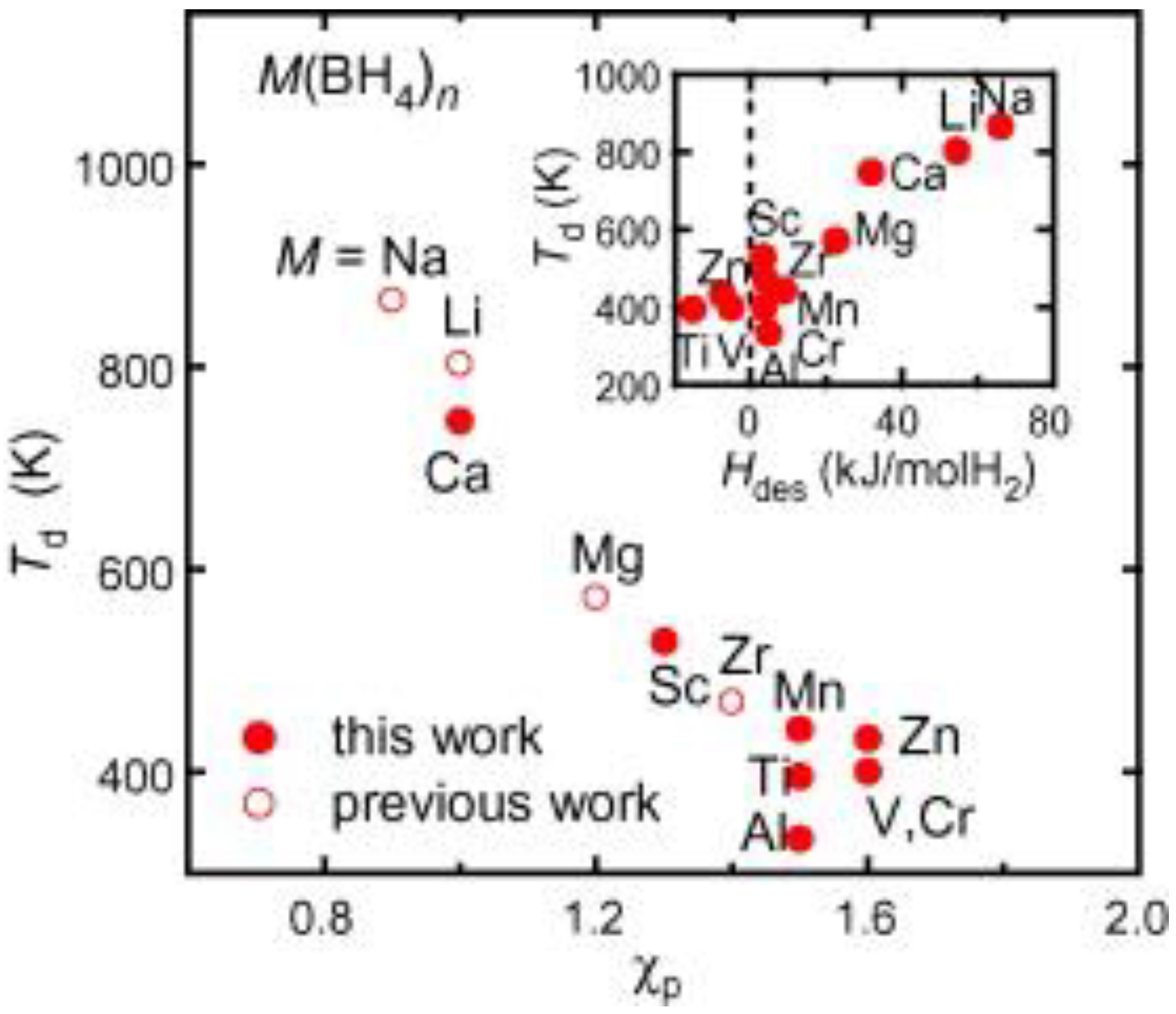
- Nakamori, Y.; Miwa, K.; Ninoyiya, A.; Li, H.; Ohba, N.; Towata, S.; Züttel, A.; Orimo, S. Correlation between thermodynamical stabilities of metal borohydrides and cation electronegativities: First-principles calculations and experiments. Phys. Rev. B 2006, 74. [Google Scholar] [CrossRef]
- Nakamori, Y.; Li, H.W.; Kikuchi, K.; Aoki, M.; Miwa, K.; Towata, S.; Orimo, S. Thermodynamical stabilities of metal-borohydrides. J. Alloys Compd. 2007, 446–447, 296–300. [Google Scholar]
- Seballos, L.; Zhang, J.Z.; Rönnebro, E.; Herbergd, J.L.; Majzoub, E.H. Metastability and crystal structure of the bialkali complex metal borohydride NaK(BH4)2. J. Alloys Compd. 2009, 476, 446–450. [Google Scholar] [CrossRef]
- Llamas-Jansa, I.; Aliouane, N.; Deledda, S.; Fonneløp, J.E.; Frommen, C.; Humphries, T.; Lieutenant, K.; Sartori, S.; Sørby, M.H.; Hauback, B.C. Chloride substitution induced by mechano-chemical reactions between NaBH4 and transition metal chlorides. J. Alloys Compd. 2012, 530, 186–192. [Google Scholar] [CrossRef]
- Černý, R.; Severa, G.; Ravnsbæk, D.B.; Filinchuk, Y.; D’Anna, V.; Hagemann, H.; Haase, D.; Jensen, C.M.; Jensen, T.R. NaSc(BH4)4: A novel scandium-based borohydride. J. Phys. Chem. C 2010, 114, 1357–1364. [Google Scholar] [CrossRef]
- Ravnsbæk, D.; Filinchuk, Y.; Cerenius, Y.; Jakobsen, H.J.; Besenbacher, F.; Skibsted, J.; Jensen, T.R. A series of mixed-metal borohydrides. Angew. Chem. Int. Ed. 2009, 48, 6659–6663. [Google Scholar] [CrossRef]
- Xia, G.L.; Li, L.; Guo, Z.P.; Gu, Q.F.; Guo, Y.H.; Yu, X.B.; Liu, H.K.; Liu, Z.W. Stabilization of NaZn(BH4)3 via nanoconfinement in SBA-15 towards enhanced hydrogen release. J. Mater. Chem. A 2013, 1, 250–257. [Google Scholar] [CrossRef]
- Ravnsbæk, D.B.; Ley, M.B.; Lee, Y.S.; Hagemann, H.; D’Anna, V.; Cho, Y.W.; Filinchuk, Y.; Jensen, T.R. A mixed-cation mixed-anion borohydride NaY(BH4)2Cl2. Int. J. Hydrog. Energy 2012, 37, 8428–8438. [Google Scholar] [CrossRef]
- Hummelshøj, J.S.; Landis, D.D.; Voss, J.; Jiang, T.; Tekin, A.; Bork, N.; Dułak, M.; Mortensen, J.J.; Adamska, L.; Andersin, J.; et al. Density functional theory based screening of ternary alkali-transition metal borohydrides: A computational material design project. J. Chem. Phys. 2009, 131. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mao, J.; Gregory, D.H. Recent Advances in the Use of Sodium Borohydride as a Solid State Hydrogen Store. Energies 2015, 8, 430-453. https://doi.org/10.3390/en8010430
Mao J, Gregory DH. Recent Advances in the Use of Sodium Borohydride as a Solid State Hydrogen Store. Energies. 2015; 8(1):430-453. https://doi.org/10.3390/en8010430
Chicago/Turabian StyleMao, Jianfeng, and Duncan H. Gregory. 2015. "Recent Advances in the Use of Sodium Borohydride as a Solid State Hydrogen Store" Energies 8, no. 1: 430-453. https://doi.org/10.3390/en8010430
APA StyleMao, J., & Gregory, D. H. (2015). Recent Advances in the Use of Sodium Borohydride as a Solid State Hydrogen Store. Energies, 8(1), 430-453. https://doi.org/10.3390/en8010430