Microbial Conversion of Waste Glycerol from Biodiesel Production into Value-Added Products
Abstract
:1. Introduction
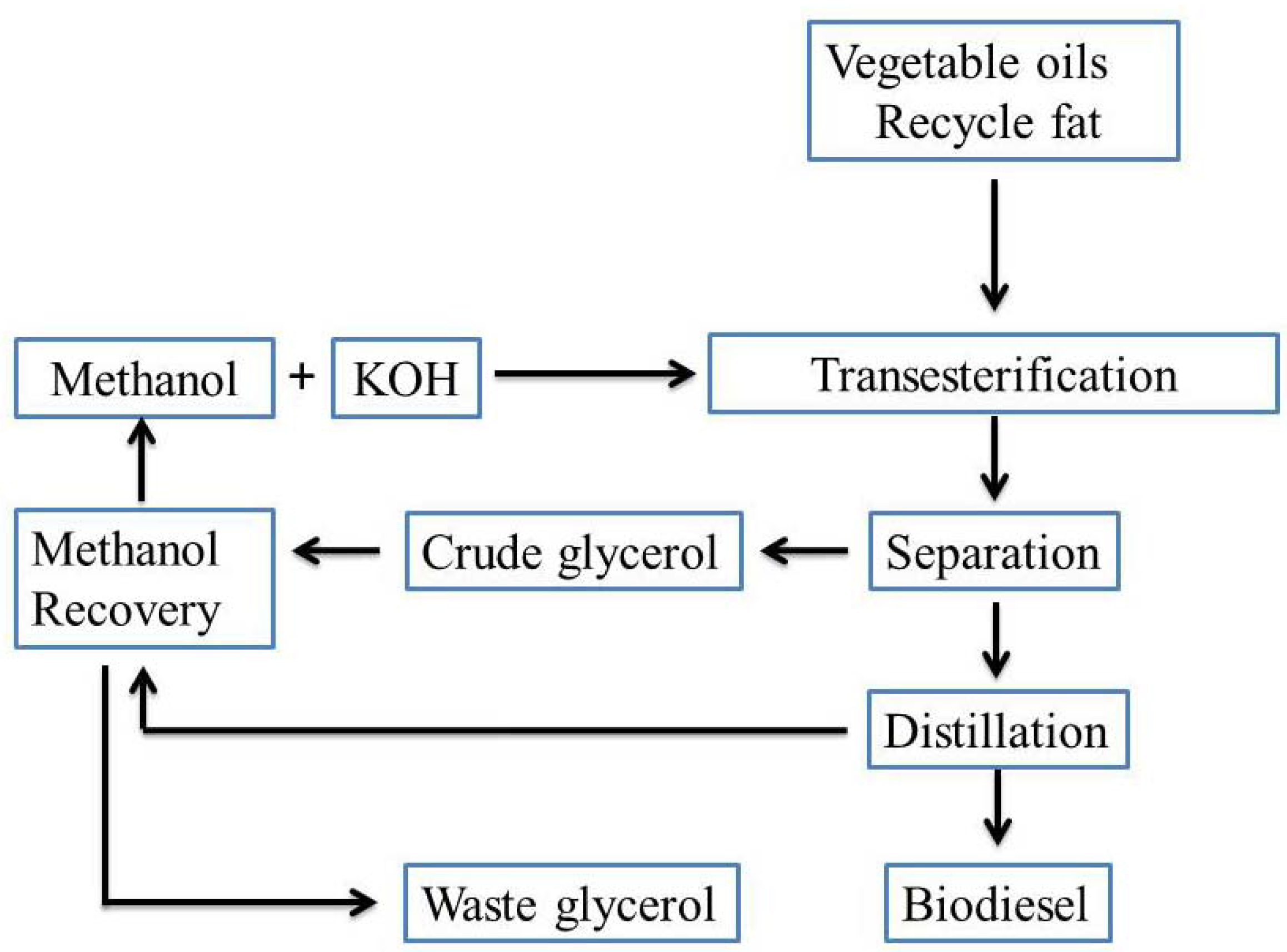
2. Microbial Conversion of Glycerol
2.1. Fermentation of Glycerol by Enterobacteriaceae Family
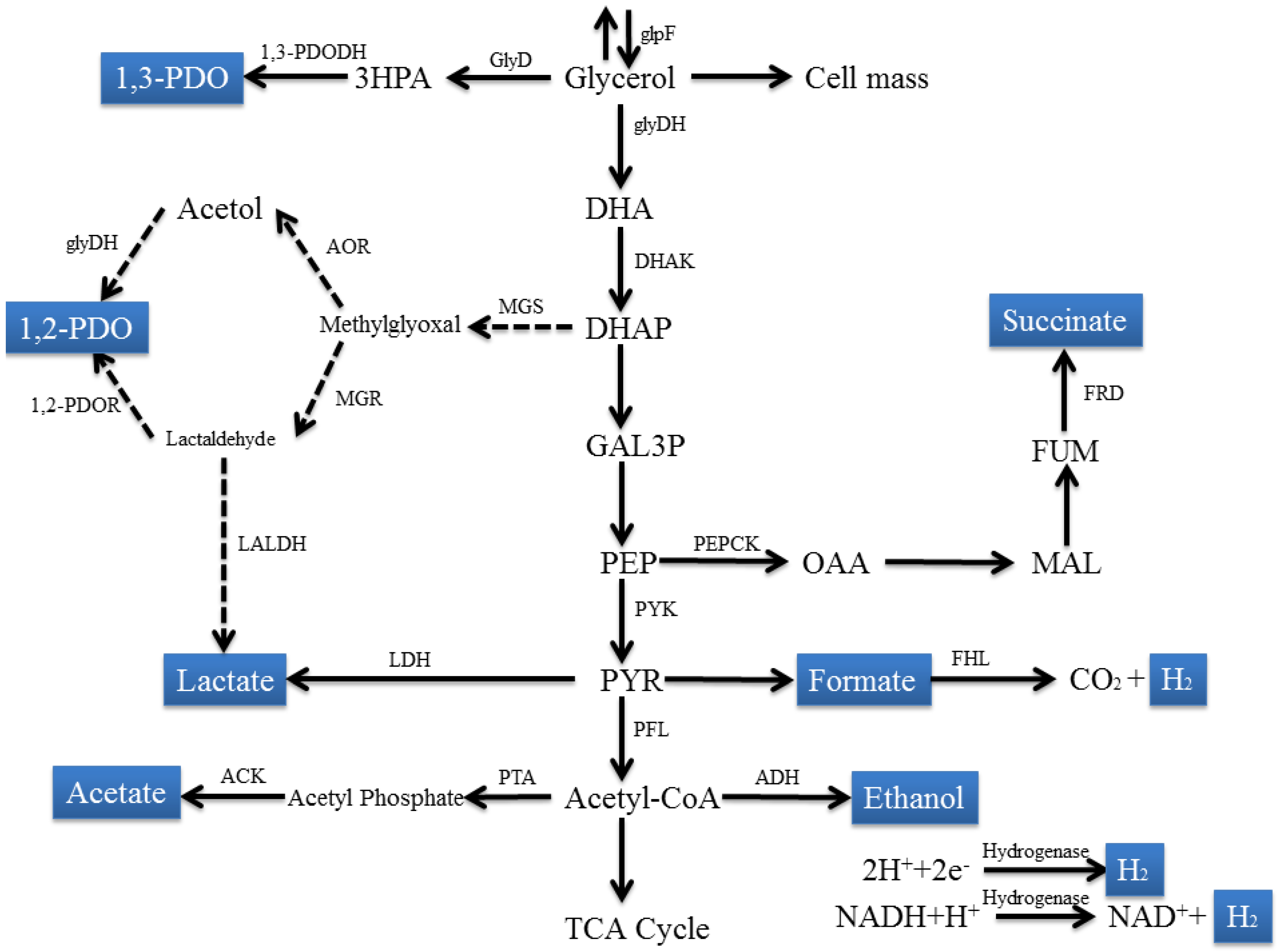
2.1.1. Escherichia Species
| Bacterial species | Strain | Product | Yield (mol/mol glycerol) | Productivity (g/L/h) | Final concentration (g/L) | Ref. |
|---|---|---|---|---|---|---|
| Escherichia coli | Engineered E. coli SY03 | Ethanol | 1 | 0.051 | 5 | [26] |
| E. coli AC521 | Lactic acid | 0.9 | 0.97 | 85.8 | [27] | |
| Engineered E. coli | d-lactic acid | 0.82 (pure glycerol) 0.87 (waste glycerol) | 1.5 (pure glycerol) | 32 (pure glycerol) 34 (waste glycerol) | [28] | |
| Engineered E. coli | l-lactic acid | 0.91 | - | 50 | [29] | |
| Engineered E. coli | 1,2-PDO | 0.26 | - | 5.6 | [30] | |
| Engineered E. coli | 1,3-PDO | 1.09 | 2.61 | 104.4 | [31] | |
| Engineered E. coli | Succinate | 0.8 | - | 12 | [32] | |
| Klebsiella | K. pneumonia (Encapsulated) | 1,3-PDO | 0.65 (batch) 0.43 (continuous) | 4.46 (continuous) | - | [33] |
| K. pneumonia (Pilot scale) | 1,3-PDO | 0.58 | 0.92 | 58.8 | [34] | |
| K. oxytoca (Lactate deficient) | 1,3-PDO | 0.41–0.53 | 0.63–0.83 | - | [35] | |
| K. pneumonia (Inactivated ADH) | 1,3-PDO | 0.70 | 1.07 | - | [36] | |
| K. pneumonia | 2,3-BD | 0.36 | 0.18 | 49.2 | [37] | |
| Engineered K. pneumonia | Ethanol | 0.89 | 1.2 | 31.0 | [38] | |
| Citrobacter | C. freundii FMCC-B294 | 1,3-PDO | 0.48 | 0.79 | 68.1 | [39] |
| C. werkmanii DSM 17579 | 1,3-PDO | 0.62 | 2.84 | - | [40] | |
| C. freundii H3 | H2 | 0.94 | - | - | [41] | |
| Engineered C. freundii | Violacein | - | 82.6 mg/L/h | 4.13 | [42] | |
| Clostridium | C. butyricum VPI 3266 | 1,3-PDO | 0.65 | 10.3 | - | [43] |
| C. butyricum AKR102a | 1,3-PDO | 0.63 (pure glycerol) | 3.3 (pure glycerol) | 93.7 (pure glycerol) | [44] | |
| C. butyricum VPI 1718 | 1,3-PDO | 0.665 | - | 67.9 | [45] | |
| Engineered C. acetobutylicum | 1,3-PDO | 0.66 | 3 | - | [46] | |
| C. pasteurianam (immobilized) | n-butanol | 0.43 | 0.074 | 8.84 | [47] | |
| C. pasteurianam ATCC 6013 | 1,3-PDO and butanol | 0.17 (1,3-PDO) 0.28 (butanol) | 0.42 (1,3-PDO) 2.49 (butanol) | - | [48] | |
| Propionibacterium bacteria | Engineered P. acidipropionici strain | Propionic acid | 0.66 (pure glycerol), 0.88 (waste glycerol) | 0.10 (pure glycerol), 0.085 (waste glycerol) | 106 (pure glycerol) | [49] |
| P. freudenreichi subsp. Shermanii NCIM 5137 | Trehalose | 391 mg/g biomass | - | - | [50] | |
| Other bacteria and mixed culture | R. palustris CGA009 | H2 | 6 | - | - | [51] |
| P. macerans | H2 | 0.801 | - | - | [52] | |
| Thermoanaerobacterium sp. | H2 | 0.30 | - | - | [53] | |
| Mixed culture | H2 | 0.96 | 91 mL/L/h | - | [54] | |
| Mixed culture | H2/formate | 0.80 | - | - | [55] | |
| Mixed culture | H2 | 0.28 (pure glycerol) 0.31 (waste glycerol) | - | - | [23] | |
| L. acidophilus | Probiotic cell mass | 0.37 g/g | - | 2.11 | ||
| L. diolivorans | 1,3-PDO | - | - | 73.7 | ||
| Anaerobic co-digestion | Biogas | - | - | 1210 mL/d | [56] | |
| C. necator DSM 545 | PHAs | - | 1.1 | - | [57] | |
| Z. denitrificans MW1 | PHAs | 0.31 g/g glycerol | - | - | [58] | |
| P. putida GO16 | PHAs | - | 0.11 | - | [59] | |
| Yeasts | Y. lipolytica NCIM 3589 | Citric acid | - | - | 77.4 | [60] |
| Y. lipolytica Wratislavia AWG7 | Citric acid | 0.33 | 1.16 | 139 | [61] | |
| C. bombicola ATCC 22214 | Sophorolipids | - | - | 60 | [62] | |
| P. antarctica JCM 10317 | Mannosylerythritol lipid | - | - | 16.3 | [63] | |
| Cryptococcus curvatus | SCO | 52% lipid content | - | 17.1 | [64] | |
| Rhodotorula glutinis | SCO | 36.5% lipid content | - | 5.4 | [65] | |
| S. ruberrimus CSB 2636 | Carotenoid | 41.9 μg/g glycerol, | 56.9μg/L/h | 3425.9μg/L | [66] | |
| Engineered S. cerevisiae | Ethanol | - | - | 2.4 g/L | [67] | |
| Engineered S. cerevisiae | 1,2-PDO | 0.258 | - | 2.19 | [68] | |
| Fungi | L. edodes strains | SCO | 0.1 g/g biomass | - | 0.52 | [69] |
| A. niger strains | SCO | 0.41 to 0.57 g/g biomass | - | 3.1 to 3.5 | [69] | |
| Galactomyces geotrichum | SCO | 0.44 g/g biomass | - | - | [70] | |
| Thamnidium elegans | SCO | - | - | 11.6 | [71] | |
| Pythium irregulare | EPA | - | 14.9 mg/L/day | 90 mg/L | [72] | |
| Blakeslea trispora | β-carotene | 15 mg/g biomass | - | - | [73] | |
| Microalgae | S. limacinum SR21 | DHA | - | 0.51 | - | [74] |
| Microbial Electrochemical Technology | B. subtilis MFC | Electricity | Maximum power density 600 mW/m2 | [75] | ||
| Single chamber MFC | Electricity | Maximum power density 2110 mW/m2 | [76] | |||
| Single chamber MFC | Electricity | Maximum power density 4579 mW/m3 with pure glycerol, 2324 mW/m3 with waster glycerol | [77] | |||
| E. aerogenes MEC | H2 | 0.74 | - | - | [78] | |
| Mixed culture MEC | H2 | 3.9 | - | - | [79] | |
| MEC with gas phase cathode | H2 | 5.4 | 0.6 L/L/day | - | [80] | |
2.1.2. Klebsiella Species
2.1.3. Citrobacter Species
2.2. Clostridium Species
2.3. Propionibacterium Species
2.4. Other Bacterial Species and Mixed Culture
2.5. Microbial Conversion of Glycerol by Yeast
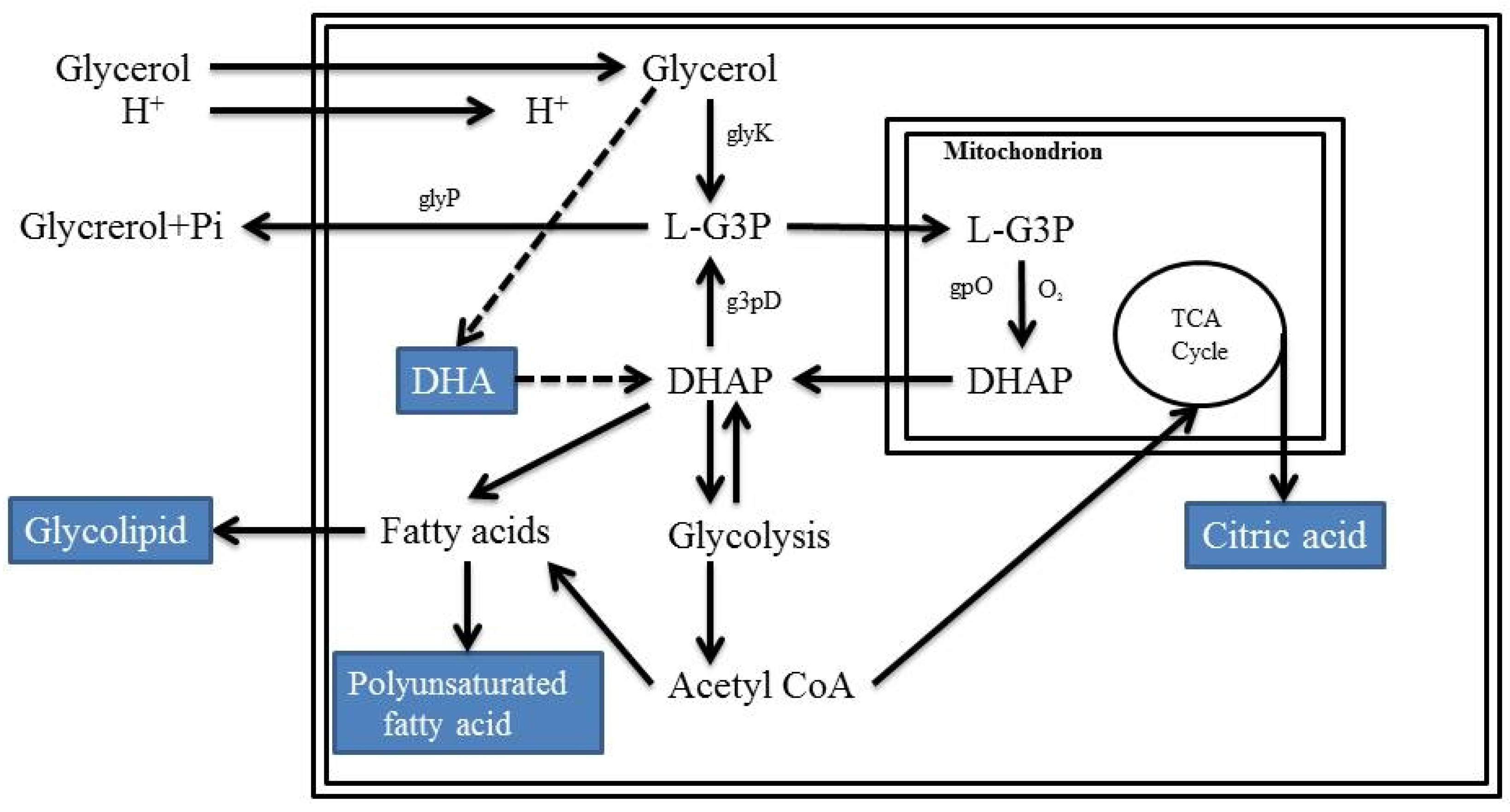
2.6. Microbial Conversion of Glycerol by Fungi
2.7. Glycerol as Carbon Source for Microalgae Species
2.8. Converting Glycerol to Value-Added Products Using Fermentation, Co-Digestion, and Microbial Electrochemical Technologies
2.8.1. Fermentation
2.8.2. Anaerobic Co-Digestion
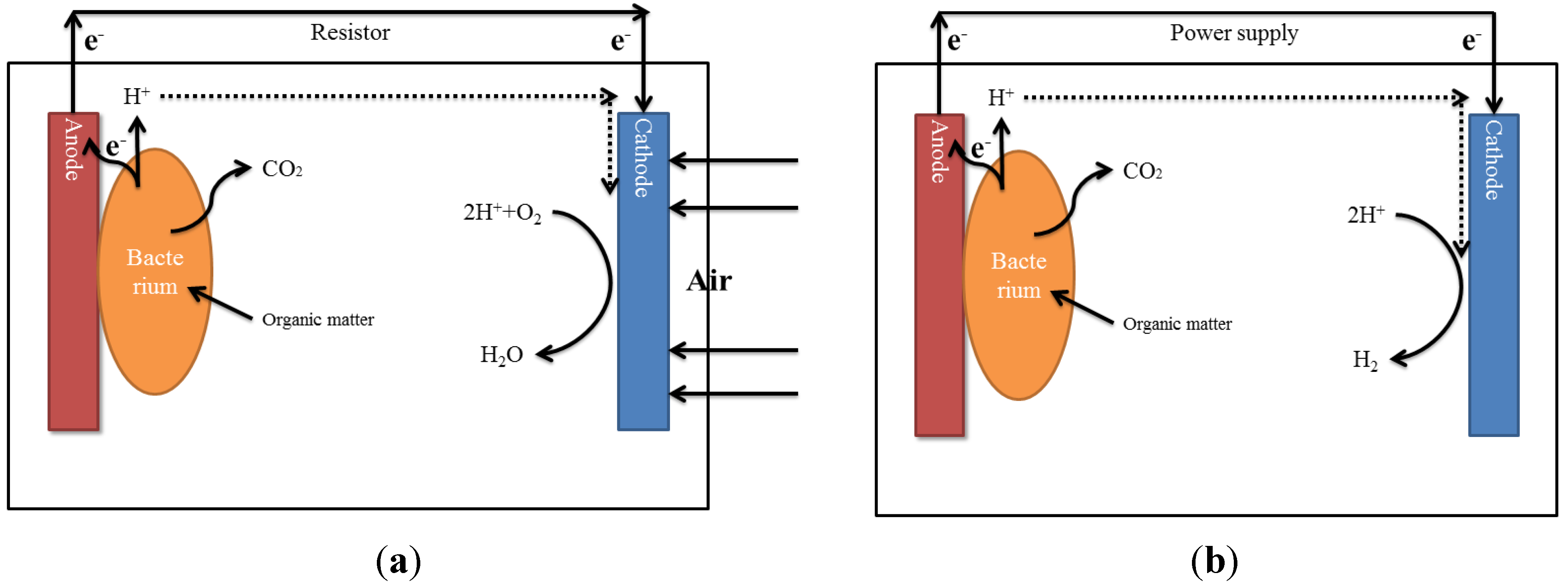
2.8.3. Microbial Electrochemical Conversion
3. Outlook
Acknowledgements
Conflicts of Interest
References
- Yang, F.X.; Hanna, M.A.; Sun, R.C. Value-added uses for crude glycerol—A byproduct of biodiesel production. Biotechnol. Biofuels 2012, 5. [Google Scholar] [CrossRef] [PubMed]
- Monthly Biodiesel Production Report; U.S. Energy Information Administration: Washington, DC, USA, 2013.
- Moser, B.R. Biodiesel Production, Properties, and Feedstocks. In Biofuels; Tomes, D., Lakshmanan, P., Songstad, D., Eds.; Springer: New York, NY, USA, 2011; pp. 285–347. [Google Scholar]
- Hu, S.J.; Luo, X.L.; Wan, C.X.; Li, Y.B. Characterization of crude glycerol from biodiesel plants. J. Agric. Food Chem. 2012, 60, 5915–5921. [Google Scholar] [CrossRef] [PubMed]
- Christoph, R.; Schmidt, B.; Steinberner, U.; Dilla, W.; Karinen, R. Glycerol. In Ullmann’s Encyclopedia of Industrial Chemistry; Morrison, L.R., Ed.; Wiley: New York, NY, USA, 2006. [Google Scholar]
- Yazdani, S.S.; Gonzalez, R. Anaerobic fermentation of glycerol: A path to economic viability for the biofuels industry. Curr. Opin. Biotech. 2007, 18, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Kerr, B.; Dozier, W.; Bregendahl, K. Nutritional Value of Crude Glycerin for Nonruminants. In Proceedings of the 23rd Annual Carolina Swine Nutrition Conference, Raleigh, NC, USA, 13 November 2007; pp. 6–18.
- Bohon, M.D.; Metzger, B.A.; Linak, W.P.; King, C.J.; Roberts, W.L. Glycerol combustion and emissions. Proc. Combust. Inst. 2011, 33, 2717–2724. [Google Scholar] [CrossRef]
- Johnson, D.T.; Taconi, K.A. The glycerin glut: Options for the value-added conversion of crude glycerol resulting from biodiesel production. Environ. Prog. 2007, 26, 338–348. [Google Scholar] [CrossRef]
- Siles, J.A.; Martín, M.A.; Chica, A.F.; Martín, A. Anaerobic co-digestion of glycerol and wastewater derived from biodiesel manufacturing. Bioresour. Technol. 2010, 101, 6315–6321. [Google Scholar] [CrossRef] [PubMed]
- Lammers, P.J.; Kerr, B.J.; Weber, T.E.; Bregendahl, K.; Lonergan, S.M.; Prusa, K.J.; Ahn, D.U.; Stoffregen, W.C.; Dozier, W.A.; Honeyman, M.S. Growth performance, carcass characteristics, meat quality, and tissue histology of growing pigs fed crude glycerin-supplemented diets. J. Anim. Sci. 2008, 86, 2962–2970. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.C.C. Glycerol dissmilation and its regulation in bacteria. Annu. Rev. Microbiol. 1976, 30, 535–578. [Google Scholar] [CrossRef] [PubMed]
- Neijssel, O.M.; Hueting, S.; Crabbendam, K.J.; Tempest, D.W. Dual pathways of glycerol assimilation in Klebsiella aerogenes NCIB 418. Arch. Microbiol. 1975, 104, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Abad, S.; Turon, X. Valorization of biodiesel derived glycerol as a carbon source to obtain added-value metabolites: Focus on polyunsaturated fatty acids. Biotechnol. Adv. 2012, 30, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.H.; Burton, R.; Zhou, Y.C. Glycerol (byproduct of biodiesel production) as a source for fuels and chemicals. Open Fuels Energy J. 2010, 3, 17–22. [Google Scholar] [CrossRef]
- Imhoff, J.F. Enterobacteriales. In Bergey’s Manual® of Systematic Bacteriology; Brenner, D.J., Krieg, N.R., Staley, J.T., Garrity, G.M., Boone, D.R., Vos, P., Goodfellow, M., Rainey, F.A., Schleifer, K.-H., Eds.; Springer: New York, NY, USA, 2005; pp. 587–850. [Google Scholar]
- Da Silva, G.P.; Mack, M.; Contiero, J. Glycerol: A promising and abundant carbon source for industrial microbiology. Biotechnol. Adv. 2009, 27, 30–39. [Google Scholar]
- Murarka, A.; Dharmadi, Y.; Yazdani, S.S.; Gonzalez, R. Fermentative utilization of glycerol by Escherichia coli and its implications for the production of fuels and chemicals. Appl. Environ. Microbiol. 2008, 74, 1124–1135. [Google Scholar] [CrossRef] [PubMed]
- Braun, T.; Philippsen, A.; Wirtz, S.; Borgnia, M.J.; Agre, P.; Kühlbrandt, W.; Engel, A.; Stahlberg, H. The 3.7 Å projection map of the glycerol facilitator glpf: A variant of the aquaporin tetramer. EMBO Rep. 2000, 1, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Trinh, C.T.; Srienc, F. Metabolic engineering of Escherichia coli for efficient conversion of glycerol to ethanol. Appl. Environ. Microbiol. 2009, 75, 6696–6705. [Google Scholar] [CrossRef] [PubMed]
- Bouvet, O.M.M.; Lenormand, P.; Ageron, E.; Grimont, P.A.D. Taxonomic diversity of anaerobic glycerol dissimilation in the Enterobacteriaceae. Res. Microbiol. 1995, 146, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.; Murarka, A.; Dharmadi, Y.; Yazdani, S.S. A new model for the anaerobic fermentation of glycerol in enteric bacteria: Trunk and auxiliary pathways in Escherichia coli. Metab. Eng. 2008, 10, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Selembo, P.A.; Perez, J.M.; Lloyd, W.A.; Logan, B.E. Enhanced hydrogen and 1,3-propanediol production from glycerol by fermentation using mixed cultures. Biotechnol. Bioeng. 2009, 104, 1098–1106. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhou, L.; Tian, K.; Kumar, A.; Singh, S.; Prior, B.A.; Wang, Z. Metabolic engineering of Escherichia coli: A sustainable industrial platform for bio-based chemical production. Biotechnol. Adv. 2013, in press. [Google Scholar]
- Cardona, C.A.; Sánchez, Ó.J. Fuel ethanol production: Process design trends and integration opportunities. Bioresour. Technol. 2007, 98, 2415–2457. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, S.S.; Gonzalez, R. Engineering Escherichia coli for the efficient conversion of glycerol to ethanol and co-products. Metab. Eng. 2008, 10, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Hong, A.-A.; Cheng, K.-K.; Peng, F.; Zhou, S.; Sun, Y.; Liu, C.-M.; Liu, D.-H. Strain isolation and optimization of process parameters for bioconversion of glycerol to lactic acid. J. Chem. Technol. Biotechnol. 2009, 84, 1576–1581. [Google Scholar] [CrossRef]
- Mazumdar, S.; Clomburg, J.M.; Gonzalez, R. Escherichia coli strains engineered for homofermentative production of d-lactic acid from glycerol. Appl. Environ. Microbiol. 2010, 76, 4327–4336. [Google Scholar] [CrossRef] [PubMed]
- Mazumdar, S.; Blankschien, M.D.; Clomburg, J.M.; Gonzalez, R. Efficient synthesis of l-lactic acid from glycerol by metabolically engineered Escherichia coli. Microb. Cell Fact. 2013, 12. [Google Scholar] [CrossRef]
- Clomburg, J.M.; Gonzalez, R. Metabolic engineering of Escherichia coli for the production of 1,2-propanediol from glycerol. Biotechnol. Bioeng. 2011, 108, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Tan, Y.; Zhu, H.; Zhao, K.; Shen, W. Microbial conversion of glycerol to 1,3-propanediol by an engineered strain of Escherichia coli. Appl. Environ. Microbiol. 2009, 75, 1628–1634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shanmugam, K.T.; Ingram, L.O. Fermentation of glycerol to succinate by metabolically engineered strains of Escherichia coli. Appl. Environ. Microbiol. 2010, 76, 2397–2401. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.-N.; Chen, G.; Yao, S.-J. Microbial production of 1,3-propanediol from glycerol by encapsulated Klebsiella pneumoniae. Biochem. Eng. J. 2006, 32, 93–99. [Google Scholar] [CrossRef]
- Cheng, K.-K.; Zhang, J.-A.; Liu, D.-H.; Sun, Y.; Liu, H.-J.; Yang, M.-D.; Xu, J.-M. Pilot-scale production of 1,3-propanediol using Klebsiella pneumoniae. Process Biochem. 2007, 42, 740–744. [Google Scholar] [CrossRef]
- Yang, G.; Tian, J.; Li, J. Fermentation of 1,3-propanediol by a lactate deficient mutant of Klebsiella oxytoca under microaerobic conditions. Appl. Microbiol. Biotechnol. 2007, 73, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Y.; Du, C.; Liu, M.; Cao, Z. Inactivation of aldehyde dehydrogenase: A key factor for engineering 1,3-propanediol production by Klebsiella pneumoniae. Metab. Eng. 2006, 8, 578–586. [Google Scholar] [CrossRef] [PubMed]
- Petrov, K.; Petrova, P. High production of 2,3-butanediol from glycerol by Klebsiella pneumoniae G31. Appl. Microbiol. Biotechnol. 2009, 84, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Oh, B.-R.; Seo, J.-W.; Heo, S.-Y.; Hong, W.-K.; Luo, L.H.; Kim, S.; Kwon, O.; Sohn, J.-H.; Joe, M.-H.; Park, D.-H.; et al. Enhancement of ethanol production from glycerol in a Klebsiella pneumoniae mutant strain by the inactivation of lactate dehydrogenase. Process Biochem. 2012, 47, 156–159. [Google Scholar] [CrossRef]
- Metsoviti, M.; Zeng, A.-P.; Koutinas, A.A.; Papanikolaou, S. Enhanced 1,3-propanediol production by a newly isolated Citrobacter freundii strain cultivated on biodiesel-derived waste glycerol through sterile and non-sterile bioprocesses. J. Biotechnol. 2013, 163, 408–418. [Google Scholar] [CrossRef]
- Maervoet, V.E.T.; Beauprez, J.; de Maeseneire, S.L.; Soetaert, W.K.; de Mey, M. Citrobacter werkmanii, a new candidate for the production of 1,3-propanediol: Strain selection and carbon source optimization. Green Chem. 2012, 14, 2168–2178. [Google Scholar] [CrossRef]
- Maru, B.T.; Constanti, M.; Stchigel, A.M.; Medina, F.; Sueiras, J.E. Biohydrogen production by dark fermentation of glycerol using Enterobacter and Citrobacter sp. Biotechnol. Prog. 2013, 29, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Jiang, P.; Xiao, S.; Zhang, C.; Lou, K.; Xing, X.-H. Fed-batch fermentation of recombinant Citrobacter freundii with expression of a violacein-synthesizing gene cluster for efficient violacein production from glycerol. Biochem. Eng. J. 2011, 57, 55–62. [Google Scholar] [CrossRef]
- González-Pajuelo, M.; Andrade, J.C.; Vasconcelos, I. Production of 1,3-Propanediol by Clostridium butyricum VPI 3266 in continuous cultures with high yield and productivity. J. Ind. Microbiol. Biotechnol. 2005, 32, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Wilkens, E.; Ringel, A.K.; Hortig, D.; Willke, T.; Vorlop, K.-D. High-level production of 1,3-propanediol from crude glycerol by Clostridium butyricum AKR102a. Appl. Microbiol. Biotechnol. 2012, 93, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- Chatzifragkou, A.; Papanikolaou, S.; Dietz, D.; Doulgeraki, A.I.; Nychas, G.-J.E.; Zeng, A.-P. Production of 1,3-propanediol by Clostridium butyricu growing on biodiesel-derived crude glycerol through a non-sterilized fermentation process. Appl. Microbiol. Biotechnol. 2011, 91, 101–112. [Google Scholar] [CrossRef] [PubMed]
- González-Pajuelo, M.; Meynial-Salles, I.; Mendes, F.; Andrade, J.C.; Vasconcelos, I.; Soucaille, P. Metabolic engineering of Clostridium acetobutylicum for the industrial production of 1,3-propanediol from glycerol. Metab. Eng. 2005, 7, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Goyal, A.; Moholkar, V.S. Production of n-butanol from biodiesel derived crude glycerol using Clostridium pasteurianum immobilized on Amberlite. Fuel 2011, 112, 557–561. [Google Scholar] [CrossRef]
- Jensen, T.Ø.; Kvist, T.; Mikkelsen, M.J.; Christensen, P.V.; Westermann, P. Fermentation of crude glycerol from biodiesel production by Clostridium pasteurianum. J. Ind. Microbiol. Biotechnol. 2012, 39, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Yang, S.-T. Propionic acid production from glycerol by metabolically engineered Propionibacterium acidipropionici. Process Biochem. 2009, 44, 1346–1351. [Google Scholar] [CrossRef]
- Ruhal, R.; Choudhury, B. Improved trehalose production from biodiesel waste using parent and osmotically sensitive mutant of Propionibacterium freudenreichii subsp. shermanii under aerobic conditions. J. Ind. Microbiol. Biotechnol. 2012, 39, 1153–1160. [Google Scholar] [CrossRef]
- Sabourin-Provost, G.; Hallenbeck, P.C. High yield conversion of a crude glycerol fraction from biodiesel production to hydrogen by photofermentation. Bioresour. Technol. 2009, 100, 3513–3517. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Murarka, A.; Campbell, P.; Gonzalez, R. Anaerobic fermentation of glycerol in Paenibacillus macerans: Metabolic pathways and environmental determinants. Appl. Environ. Microbiol. 2009, 75, 5871–5883. [Google Scholar] [CrossRef] [PubMed]
- Sittijunda, S.; Reungsang, A. Media optimization for biohydrogen production from waste glycerol by anaerobic thermophilic mixed cultures. Int. J. Hydrog. Energy 2012, 37, 15473–15482. [Google Scholar] [CrossRef]
- Varrone, C.; Giussani, B.; Izzo, G.; Massini, G.; Marone, A.; Signorini, A.; Wang, A. Statistical optimization of biohydrogen and ethanol production from crude glycerol by microbial mixed culture. Int. J. Hydrog. Energy 2012, 37, 16479–16488. [Google Scholar] [CrossRef]
- Temudo, M.F.; Poldermans, R.; Kleerebezem, R.; van Loosdrecht, M.C.M. Glycerol fermentation by (open) mixed cultures: A chemostat study. Biotechnol. Bioeng. 2008, 100, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Fountoulakis, M.S.; Manios, T. Enhanced methane and hydrogen production from municipal solid waste and agro-industrial by-products co-digested with crude glycerol. Bioresour. Technol. 2009, 100, 3043–3047. [Google Scholar] [CrossRef] [PubMed]
- Cavalheiro, J.M.B.T.; de Almeida, M.C.M.D.; Grandfils, C.; da Fonseca, M.M.R. Poly(3-hydroxybutyrate) production by Cupriavidus necator using waste glycerol. Process Biochem. 2009, 44, 509–515. [Google Scholar] [CrossRef]
- Ibrahim, M.H.A.; Steinbüchel, A. Zobellella denitrificans strain MW1, a newly isolated bacterium suitable for poly(3-hydroxybutyrate) production from glycerol. J. Appl. Microbiol. 2010, 108, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Kenny, S.T.; Runic, J.N.; Kaminsky, W.; Woods, T.; Babu, R.P.; O’Connor, K.E. Development of a bioprocess to convert PET derived terephthalic acid and biodiesel derived glycerol to medium chain length polyhydroxyalkanoate. Appl. Microbiol. Biotechnol. 2012, 95, 623–633. [Google Scholar] [CrossRef]
- Imandi, S.B.; Bandaru, V.R.; Somalanka, S.R.; Garapati, H.R. Optimization of medium constituents for the production of citric acid from byproduct glycerol using Doehlert experimental design. Enzym. Microb. Technol. 2007, 40, 1367–1372. [Google Scholar] [CrossRef]
- Rywińska, A.; Rymowicz, W.; Żarowska, B.; Wojtatowicz, M. Biosynthesis of citric acid from glycerol by acetate mutants of Yarrowia lipolytica in fed-batch fermentation. Food Technol. Biotechnol. 2009, 47, 1–6. [Google Scholar]
- Ashby, R.; Nuñez, A.; Solaiman, D.; Foglia, T. Sophorolipid biosynthesis from a biodiesel co-product stream. J. Am. Oil Chem. Soc. 2005, 82, 625–630. [Google Scholar] [CrossRef]
- Morita, T.; Konishi, M.; Fukuoka, T.; Imura, T.; Kitamoto, D. Microbial conversion of glycerol into glycolipid biosurfactants, mannosylerythritol lipids, by a basidiomycete yeast, Pseudozyma antarctica JCM 10317T. J. Biosci. Bioeng. 2007, 104, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Cui, Y.; Trushenski, J.; Blackburn, J.W. Converting crude glycerol derived from yellow grease to lipids through yeast fermentation. Bioresour. Technol. 2010, 101, 7581–7586. [Google Scholar] [CrossRef] [PubMed]
- Yen, H.-W.; Yang, Y.-C.; Yu, Y.-H. Using crude glycerol and thin stillage for the production of microbial lipids through the cultivation of Rhodotorula glutinis. J. Biosci. Bioeng. 2012, 114, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Valduga, E.; Tatsch, P.O.; Tiggemann, L.; Zeni, J.; Colet, R.; Cansian, J.M.; Treichel, H.; Luccio, M. Evaluation of the conditions of carotenoids production in a synthetic medium by Sporidiobolus salmonicolor (CBS 2636) in a bioreactor. Int. J. Food Sci. Technol. 2009, 44, 2445–2451. [Google Scholar] [CrossRef]
- Yu, K.O.; Kim, S.W.; Han, S.O. Engineering of glycerol utilization pathway for ethanol production by Saccharomyces cerevisiae. Bioresour. Technol. 2010, 101, 4157–4161. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Yun, H.S.; Lee, J.W.; Oh, M.K. Production of 1,2-propanediol from glycerol in Saccharomyces cerevisiae. J. Microbiol. Biotechnol. 2011, 21, 846–853. [Google Scholar] [CrossRef] [PubMed]
- André, A.; Diamantopoulou, P.; Philippoussis, A.; Sarris, D.; Komaitis, M.; Papanikolaou, S. Biotechnological conversions of bio-diesel derived waste glycerol into added-value compounds by higher fungi: Production of biomass, single cell oil and oxalic acid. Ind. Crop. Prod. 2010, 31, 407–416. [Google Scholar] [CrossRef]
- Marchand, K.; Lubitz, W.; Nicol, R. Utilization of biodiesel derived crude glycerol by fungal isolates for biomass and single cell oil production. J. Biobased. Mater. Bio. 2013, 7, 415–419. [Google Scholar] [CrossRef]
- Chatzifragkou, A.; Makri, A.; Belka, A.; Bellou, S.; Mavrou, M.; Mastoridou, M.; Mystrioti, P.; Onjaro, G.; Aggelis, G.; Papanikolaou, S. Biotechnological conversions of biodiesel derived waste glycerol by yeast and fungal species. Energy 2011, 36, 1097–1108. [Google Scholar] [CrossRef]
- Athalye, S.K.; Garcia, R.A.; Wen, Z. Use of biodiesel-derived crude glycerol for producing eicosapentaenoic acid (EPA) by the fungus Pythium irregulare. J. Agric. Food Chem. 2009, 57, 2739–2744. [Google Scholar] [CrossRef] [PubMed]
- Mantzouridou, F.; Naziri, E.; Tsimidou, M.Z. Industrial glycerol as a supplementary carbon source in the production of β-carotene by Blakeslea trispora. J. Agric. Food Chem. 2008, 56, 2668–2675. [Google Scholar] [CrossRef] [PubMed]
- Chi, Z.; Pyle, D.; Wen, Z.; Frear, C.; Chen, S. A laboratory study of producing docosahexaenoic acid from biodiesel-waste glycerol by microalgal fermentation. Process Biochem. 2007, 42, 1537–1545. [Google Scholar] [CrossRef]
- Nimje, V.R.; Chen, C.-Y.; Chen, C.-C.; Chen, H.-R.; Tseng, M.-J.; Jean, J.-S.; Chang, Y.-F. Glycerol degradation in single-chamber microbial fuel cells. Bioresour. Technol. 2011, 102, 2629–2634. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Yang, Q.; Wang, X.; Liu, Y.; Lee, H.; Ren, N. Treatment of biodiesel production wastes with simultaneous electricity generation using a single-chamber microbial fuel cell. Bioresour. Technol. 2011, 102, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Sharma, Y.; Parnas, R.; Li, B. Bioenergy production from glycerol in hydrogen producing bioreactors (HPBs) and microbial fuel cells (MFCs). Int. J. Hydrog. Energy 2011, 36, 3853–3861. [Google Scholar] [CrossRef]
- Sakai, S.; Yagishita, T. Microbial production of hydrogen and ethanol from glycerol-containing wastes discharged from a biodiesel fuel production plant in a bioelectrochemical reactor with thionine. Biotechnol. Bioeng. 2007, 98, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Selembo, P.A.; Perez, J.M.; Lloyd, W.A.; Logan, B.E. High hydrogen production from glycerol or glucose by electrohydrogenesis using microbial electrolysis cells. Int. J. Hydrog. Energy 2009, 34, 5373–5381. [Google Scholar] [CrossRef]
- Escapa, A.; Manuel, M.F.; Morán, A.; Gómez, X.; Guiot, S.R.; Tartakovsky, B. Hydrogen production from glycerol in a membraneless microbial electrolysis cell. Energy Fuel. 2009, 23, 4612–4618. [Google Scholar] [CrossRef]
- Hofvendahl, K.; Hahn-Hägerdal, B. Factors affecting the fermentative lactic acid production from renewable resources. Enzym. Microb. Technol. 2000, 26, 87–107. [Google Scholar] [CrossRef]
- Okano, K.; Tanaka, T.; Ogino, C.; Fukuda, H.; Kondo, A. Biotechnological production of enantiomeric pure lactic acid from renewable resources: Recent achievements, perspectives, and limits. Appl. Microbiol. Biotechnol. 2010, 85, 413–423. [Google Scholar] [CrossRef] [PubMed]
- John, R.P.; Anisha, G.S.; Nampoothiri, K.M.; Pandey, A. Direct lactic acid fermentation: Focus on simultaneous saccharification and lactic acid production. Biotechnol. Adv. 2009, 27, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Wee, Y.J.; Yun, J.S.; Park, D.H.; Ryu, H.W. Biotechnological production of L(+)-lactic acid from wood hydrolyzate by batch fermentation of Enterococcus faecalis. Biotechnol. Lett. 2004, 26, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhu, Y.-L.; Zheng, H.-Y.; Li, Y.-W.; Zeng, Z.-Y. Continuous production of 1,2-propanediol by the selective hydrogenolysis of solvent-free glycerol under mild conditions. J. Chem. Technol. Biotechnol. 2008, 83, 1670–1675. [Google Scholar] [CrossRef]
- Shelley, S. A renewable route to propylene glycol. Chem. Eng. Prog. 2007, 103, 6–9. [Google Scholar]
- Werpy, T.; Petersen, G. Top Value Added Chemical from Biomass; U.S. Department of Energy: Washington, DC, USA, 2004. [Google Scholar]
- Chotani, G.; Dodge, T.; Hsu, A.; Kumar, M.; LaDuca, R.; Trimbur, D.; Weyler, W.; Sanford, K. The commercial production of chemicals using pathway engineering. Biochim. Biophys. Acta 2000, 1543, 434–455. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, Z.; Du, C.; Li, Y.; Cao, Z. Introduction of an NADH regeneration system into Klebsiella oxytoca leads to an enhanced oxidative and reductive metabolism of glycerol. Metab. Eng. 2009, 11, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Celińska, E.; Grajek, W. Biotechnological production of 2,3-butanediol—Current state and prospects. Biotechnol. Adv. 2009, 27, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Wolf-Dieter, D. Microbial conversion of glycerol to 1,3-propanediol. FEMS Microbiol. Rev. 1995, 16, 143–149. [Google Scholar]
- Durán, N.; Menck, C.F.M. Chromobacterium violaceum: A review of pharmacological and industiral perspectives. Crit. Rev. Microbiol. 2001, 27, 201–222. [Google Scholar] [CrossRef] [PubMed]
- Biebl, H.; Marten, S.; Hippe, H.; Deckwer, W.-D. Glycerol conversion to 1,3-propanediol by newly isolated Clostridia. Appl. Microbiol. Biotechnol. 1992, 36, 592–597. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, J.H.; Jang, S.H.; Nielsen, L.K.; Kim, J.; Jung, K.S. Fermentative butanol production by Clostridia. Biotechnol. Bioeng. 2008, 101, 209–228. [Google Scholar] [CrossRef] [PubMed]
- Boyaval, P.; Corre, C. Production of propionic acid. Lait 1995, 75, 453–461. [Google Scholar] [CrossRef]
- Danilo, Z. Production of Propionic Acid. U.S. Patent 8,053,602 B2, 8 November 2011. [Google Scholar]
- Cardoso, F.S.; Gaspar, P.; Hugenholtz, J.; Ramos, A.; Santos, H. Enhancement of trehalose production in dairy propionibacteria through manipulation of environmental conditions. Int. J. Food Microbiol. 2004, 91, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Schiraldi, C.; di Lernia, I.; de Rosa, M. Trehalose production: Exploiting novel approaches. Trends Biotechnol. 2002, 20, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Kongjan, P.; Angelidaki, I. Extreme thermophilic biohydrogen production from wheat straw hydrolysate using mixed culture fermentation: Effect of reactor configuration. Bioresour. Technol. 2010, 101, 7789–7796. [Google Scholar] [CrossRef] [PubMed]
- Kongjan, P.; Min, B.; Angelidaki, I. Biohydrogen production from xylose at extreme thermophilic temperatures (70 °C) by mixed culture fermentation. Water Res. 2009, 43, 1414–1424. [Google Scholar] [CrossRef]
- Pflügl, S.; Marx, H.; Mattanovich, D.; Sauer, M. 1,3-Propanediol production from glycerol with Lactobacillus diolivorans. Bioresour. Technol. 2012, 119, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Bioplastics with a green agenda. Curr. Opin. Microbiol. 2010, 13, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Valadi, Å.; Granath, K.; Gustafsson, L.; Adler, L. Distinct intracellular localization of Gpd1p and Gpd2p, the two yeast isoforms of NAD+-dependent Glycerol-3-phosphate dehydrogenase, explains their different contributions to redox-driven glycerol production. J. Biol. Chem. 2004, 279, 39677–39685. [Google Scholar] [CrossRef] [PubMed]
- Adler, L.; Blomberg, A.; Nilsson, A. Glycerol metabolism and osmoregulation in the salt-tolerant yeast Debaryomyces hansenii. J. Bacteriol. 1985, 162, 300–306. [Google Scholar] [PubMed]
- André, L.; Nilsson, A.; Adler, L. The role of glycerol in osmotolerance of the yeast Debaryomyces hansenii. J. Gen. Microbiol. 1988, 134, 669–677. [Google Scholar]
- Castro, I.M.; Loureiro-Dias, M.C. Glycerol utilization in Fusarium oxysporum var. lini: Regulation of transport and metabolism. J. Gen. Microbiol. 1991, 137, 1497–1502. [Google Scholar] [CrossRef]
- Gancedo, C.; Gancedo, J.M.; Sols, A. Glycerol metabolism in yeasts. Eur. J. Biochem. 1968, 5, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Nobre, M.F.; da Costa, M.S. The accumulation of polyols by the yeast Debaryomyces hansenii in response to water stress. Can. J. Microbiol. 1985, 31, 1061–1064. [Google Scholar] [CrossRef]
- Ferreira, C.; van Voorst, F.; Martins, A.; Neves, L.; Oliveira, R.; Kielland-Brandt, M.C.; Lucas, C.; Brandt, A. A member of the sugar transporter family, Stl1p is the glycerol/H+ symporter in Saccharomyces cerevisiae. Mol. Biol. Cell. 2005, 16, 2068–2076. [Google Scholar] [CrossRef] [PubMed]
- Kayingo, G.; Martins, A.; Andrie, R.; Neves, L.; Lucas, C.; Wong, B. A permease encoded by STL1 is required for active glycerol uptake by Candida albicans. Microbiology 2009, 155, 1547–1557. [Google Scholar] [CrossRef] [PubMed]
- Nicol, R.W.; Marchand, K.; Lubitz, W.D. Bioconversion of crude glycerol by fungi. Appl. Microbiol. Biotechnol. 2012, 93, 1865–1875. [Google Scholar] [CrossRef] [PubMed]
- Amaral, P.F.F.; Ferreira, T.F.; Fontes, G.C.; Coelho, M.A.Z. Glycerol valorization: New biotechnological routes. Food Bioprod. Process. 2009, 87, 179–186. [Google Scholar] [CrossRef]
- Finogenova, T.V.; Morgunov, I.G.; Kamzolova, S.V.; Chernyavskaya, O.G. Organic acid production by the yeast Yarrowia lipolytica: A review of prospects. Appl. Biochem. Microbiol. 2005, 41, 418–425. [Google Scholar] [CrossRef]
- Xu, D.-B.; Madrid, C.P.; Röhr, M.; Kubicek, C.P. The influence of type and concentration of the carbon source on production of citric acid by Aspergillus niger. Appl. Microbiol. Biotechnol. 1989, 30, 553–558. [Google Scholar]
- Amaral, P.F.F.; Coelho, M.A.Z.; Marrucho, I.M.J.; Coutinho, J.A.P. Biosurfactants from Yeasts: Characteristics, Production and Application. In Biosurfactants; Sen, R., Ed.; Springer: New York, NY, USA, 2010; Volume 672, pp. 236–249. [Google Scholar]
- Banat, I.M.; Franzetti, A.; Gandolfi, I.; Bestetti, G.; Martinotti, M.G.; Fracchia, L.; Smyth, T.J.; Marchant, R. Microbial biosurfactants production, applications and future potential. Appl. Microbiol. Biotechnol. 2010, 87, 427–444. [Google Scholar] [CrossRef] [PubMed]
- Makkar, R.S.; Cameotra, S.S.; Banat, I.M. Advances in utilization of renewable substrates for biosurfactant production. AMB Express 2011, 1. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Koh, C.M.J.; Ji, L. Bioconversion of crude glycerol to glycolipids in Ustilago maydi. Bioresour. Technol. 2011, 102, 3927–3933. [Google Scholar]
- Papanikolaou, S.; Komaitis, M.; Aggelis, G. Single cell oil (SCO) production by Mortierella isabellina grown on high-sugar content media. Bioresour. Technol. 2004, 95, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, L.; Empis, J. Relative stabilities of microalgal carotenoids in microalgal extracts, biomass and fish feed: Effect of storage conditions. Innov. Food Sci. Emerg. Technol. 2003, 4, 227–233. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Essential fatty acids in health and chronic disease. Am. J. Clin. Nutr. 1999, 70, 560S–569S. [Google Scholar] [PubMed]
- Hou, C.T. Production of arachidonic acid and dihomo-γ-linolenic acid from glycerol by oil-producing filamentous fungi, Mortierella in the ARS culture collection. J. Ind. Microbiol. Biotechnol. 2008, 35, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Tonon, T.; Harvey, D.; Larson, T.R.; Graham, I.A. Long chain polyunsaturated fatty acid production and partitioning to triacylglycerols in four microalgae. Phytochemistry 2002, 61, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Grima, E.M.; Pérez, J.A.S.; Camacho, F.G.; Medina, A.R.; Giménez, A.G.; López Alonso, D. The production of polyunsaturated fatty acids by microalgae: From strain selection to product purification. Process Biochem. 1995, 30, 711–719. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Ingram, L.O.; Calder, J.A.; van Baalen, C.; Plucker, F.E.; Parker, P.L. Role of reduced exogenous organic compounds in the physiology of the blue-green bacteria (algae): Photoheterotrophic growth of a “heterotrophic” blue-green bacterium. J. Bacteriol. 1973, 114, 695–700. [Google Scholar] [PubMed]
- Ingram, L.O.; Van Baalen, C.; Calder, J.A. Role of reduced exogenous organic compounds in the physiology of the blue-green bacteria (algae): Photoheterotrophic growth of an “autotrophic” blue-green bacterium. J. Bacteriol. 1973, 114, 701–705. [Google Scholar] [PubMed]
- Neilson, A.H.; Lewin, R.A. The uptake and utilization of organic carbon by algae: An essay in comparative biochemistry. Phycologia 1974, 13, 227–264. [Google Scholar] [CrossRef]
- Villadsen, J.; Nielsen, J.; Lidén, G. Bioreaction Engineering Principles; Springer: New York, NY, USA, 2011. [Google Scholar]
- Menzel, K.; Zeng, A.-P.; Deckwer, W.-D. High concentration and productivity of 1,3-propanediol from continuous fermentation of glycerol by Klebsiella pneumoniae. Enzym. Microb. Technol. 1997, 20, 82–86. [Google Scholar] [CrossRef]
- Gungormusler, M.; Gonen, C.; Azbar, N. Continuous production of 1,3-propanediol using raw glycerol with immobilized Clostridium beijerinckii NRRL B-593 in comparison to suspended culture. Bioprocess Biosyst. Eng. 2011, 34, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Gungormusler, M.; Gonen, C.; Azbar, N. Use of ceramic-based cell immobilization to produce 1,3-propanediol from biodiesel-derived waste glycerol with Klebsiella pneumoniae. J. Appl. Microbiol. 2011, 111, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
- Yen, H.-W.; Brune, D.E. Anaerobic co-digestion of algal sludge and waste paper to produce methane. Bioresour. Technol. 2007, 98, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, S.; Mattam, S.; Gonzalez, R. Fuel and Chemical Production from Glycerol, a Biodiesel Waste Product. In Biofuels from Agricultural Wastes and Byproducts; Blaschek, H.P., Ezeji, T.C., Scheffran, J., Eds.; Wiley-Blackwell: Oxford, UK, 2010. [Google Scholar] [CrossRef]
- Astals, S.; Nolla-Ardèvol, V.; Mata-Alvarez, J. Anaerobic co-digestion of pig manure and crude glycerol at mesophilic conditions: Biogas and digestate. Bioresour. Technol. 2012, 110, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Harnisch, F.; Schröder, U. From MFC to MXC: Chemical and biological cathodes and their potential for microbial bioelectrochemical systems. Chem. Soc. Rev. 2010, 39, 4433–4448. [Google Scholar] [CrossRef] [PubMed]
- Nevin, K.P.; Hensley, S.A.; Franks, A.E.; Summers, Z.M.; Ou, J.; Woodard, T.L.; Snoeyenbos-West, O.L.; Lovley, D.R. Electrosynthesis of organic compounds from carbon dioxide is catalyzed by a diversity of acetogenic microorganisms. Appl. Environ. Microbiol. 2011, 77, 2882–2886. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E. Exoelectrogenic bacteria that power microbial fuel cells. Nat. Rev. Microbiol. 2009, 7, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Malvankar, N.S.; Lovley, D.R. Microbial nanowires: A new paradigm for biological electron transfer and bioelectronics. ChemSusChem 2012, 5, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Roden, E.E.; Kappler, A.; Bauer, I.; Jiang, J.; Paul, A.; Stoesser, R.; Konishi, H.; Xu, H. Extracellular electron transfer through microbial reduction of solid-phase humic substances. Nat. Geosci. 2010, 3, 417–421. [Google Scholar] [CrossRef]
- Lovley, D.R. Powering microbes with electricity: Direct electron transfer from electrodes to microbes. Environ. Microbiol. Rep. 2011, 3, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Cheng, S.; Logan, B.E. Production of electricity from acetate or butyrate using a single-chamber microbial fuel cell. Environ. Sci. Technol. 2005, 39, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Hu, H.; Chignell, J.; Fan, Y. Microbial electrolysis: Novel technology for hydrogen production from biomass. Biofuels 2010, 1, 129–142. [Google Scholar] [CrossRef]
- Nevin, K.P.; Richter, H.; Covalla, S.F.; Johnson, J.P.; Woodard, T.L.; Orloff, A.L.; Jia, H.; Zhang, M.; Lovley, D.R. Power output and columbic efficiencies from biofilms of Geobacter sulfurreducens comparable to mixed community microbial fuel cells. Environ. Microbiol. 2008, 10, 2505–2514. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Li, H.; Gu, T. A state of the art review on microbial fuel cells: A promising technology for wastewater treatment and bioenergy. Biotechnol. Adv. 2007, 25, 464–482. [Google Scholar] [CrossRef] [PubMed]
- Dennis, P.G.; Harnisch, F.; Yeoh, Y.K.; Tyson, G.W.; Rabaey, K. Dynamics of cathode-associated microbial communities and metabolite profiles in a glycerol-fed bioelectrochemical system. Appl. Environ. Microbiol. 2013, 79, 4008–4014. [Google Scholar] [CrossRef] [PubMed]
- Kraus, G.A. Synthetic methods for the preparation of 1,3-propanediol. CLEAN—Soil Air Water 2008, 36, 648–651. [Google Scholar] [CrossRef]
- Saxena, R.K.; Anand, P.; Saran, S.; Isar, J. Microbial production of 1,3-propanediol: Recent developments and emerging opportunities. Biotechnol. Adv. 2009, 27, 895–913. [Google Scholar] [CrossRef] [PubMed]
- Emptage, M.; Haynie, S.L.; Laffend, L.A.; Pucci, J.P.; Whited, G. Process for the Biological Production of 1,3-Propanediol with High Titer. U.S. Patent 7,504,250 B2, 17 March 2009. [Google Scholar]
- Nakamura, C.E.; Whited, G.M. Metabolic engineering for the microbial production of 1,3-propanediol. Curr. Opin. Biotechnol. 2003, 14, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Zeng, A.-P.; Biebl, H. Bulk Chemicals from Biotechnology: The Case of 1,3-Propanediol Production and the New Trends. In Tools and Applications of Biochem. Engineering Science; Schügerl, K., Zeng, A.-P., Aunins, J.G., Bader, A., Bell, W., Biebl, H., Biselli, M., Carrondo, M.J.T., Castilho, L.R., Chang, H.N., et al., Eds.; Springer: Berlin, Germany, 2002; Volume 74, pp. 239–259. [Google Scholar]
- Sánchez, Ó.J.; Cardona, C.A. Trends in biotechnological production of fuel ethanol from different feedstocks. Bioresour. Technol. 2008, 99, 5270–5295. [Google Scholar] [CrossRef] [PubMed]
- Holladay, J.D.; Hu, J.; King, D.L.; Wang, Y. An overview of hydrogen production technologies. Catal. Today 2009, 139, 244–260. [Google Scholar] [CrossRef]
- Dillich, S.; Ramsden, T.; Melaina, M. Hydrogen Production Cost Using Low-Cost Natural Gas; U.S. Department of Energy: Washington, DC, USA, 2012. [Google Scholar]
- Hu, H.; Fan, Y.; Liu, H. Hydrogen production using single-chamber membrane-free microbial electrolysis cells. Water Res. 2008, 42, 4172–4178. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Call, D.; Cheng, S.; Hamelers, H.V.M.; Sleutels, T.H.J.A.; Jeremiasse, A.W.; Rozendal, R.A. Microbial electrolysis cells for high yield hydrogen gas production from organic matter. Environ. Sci. Technol. 2008, 42, 8630–8640. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Ren, N.; Xing, D.; Logan, B.E. Hydrogen production with effluent from an ethanol—H2-coproducing fermentation reactor using a single-chamber microbial electrolysis cell. Biosens. Bioelectron. 2009, 24, 3055–3060. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Grot, S.; Logan, B.E. Electrochemically assisted microbial production of hydrogen from acetate. Environ. Sci. Technol. 2005, 39, 4317–4320. [Google Scholar] [CrossRef] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, C.; Lesnik, K.L.; Liu, H. Microbial Conversion of Waste Glycerol from Biodiesel Production into Value-Added Products. Energies 2013, 6, 4739-4768. https://doi.org/10.3390/en6094739
Li C, Lesnik KL, Liu H. Microbial Conversion of Waste Glycerol from Biodiesel Production into Value-Added Products. Energies. 2013; 6(9):4739-4768. https://doi.org/10.3390/en6094739
Chicago/Turabian StyleLi, Cheng, Keaton L. Lesnik, and Hong Liu. 2013. "Microbial Conversion of Waste Glycerol from Biodiesel Production into Value-Added Products" Energies 6, no. 9: 4739-4768. https://doi.org/10.3390/en6094739
APA StyleLi, C., Lesnik, K. L., & Liu, H. (2013). Microbial Conversion of Waste Glycerol from Biodiesel Production into Value-Added Products. Energies, 6(9), 4739-4768. https://doi.org/10.3390/en6094739





