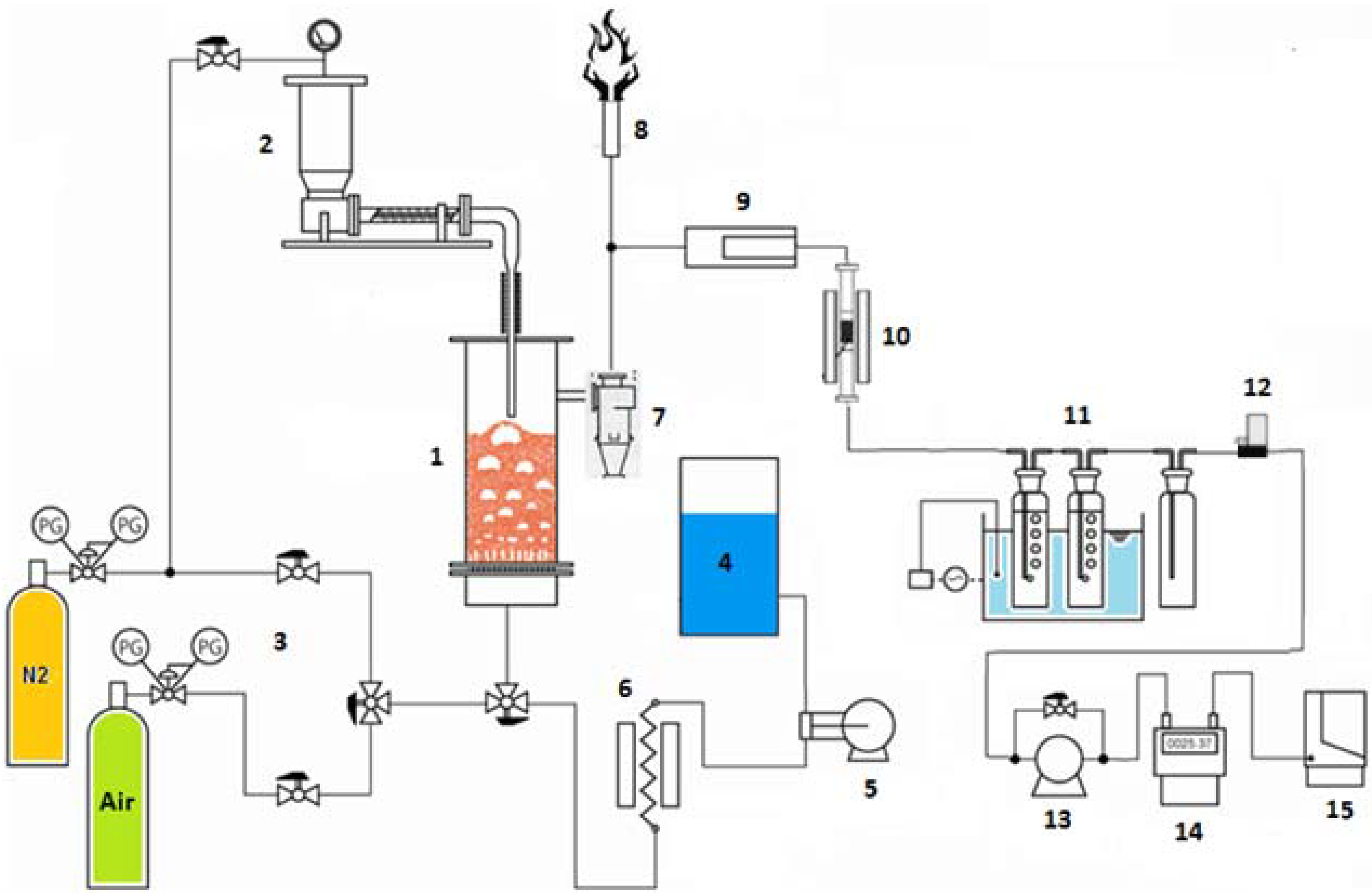
4.2. Sorbent/Catalyst Tests
The efficiency of the commercial nickel catalyst mixed with dolomite in both TAR conversion and CO
2 sorption has been evaluated by means of H
2 response curves. In case of sorption enhanced steam reforming, as described by Harrison
et al. [
33], response curves present a typical shape which can be divided in three sections. The first one is called “pre-breakthrough” and corresponds to the highest efficiency of CO
2 sorption, water gas shift and TAR reforming reactions together and is represented by a plateau until the reactions are at equilibrium. After some time, depending on the amount of CaO and on CO
2 partial pressure, carbonation reaction decreases its efficiency and the curve shows a rapid fall. This transient is the “breakthrough” section, characterized by an almost specular behaviour of H
2 (decreasing) and CO
2 (increasing). When carbonation offers no more contribution to TAR reforming reactions, hydrogen and carbon dioxide reach a new equilibrium, characterized by another plateau; this last section is called “post-breakthrough”.
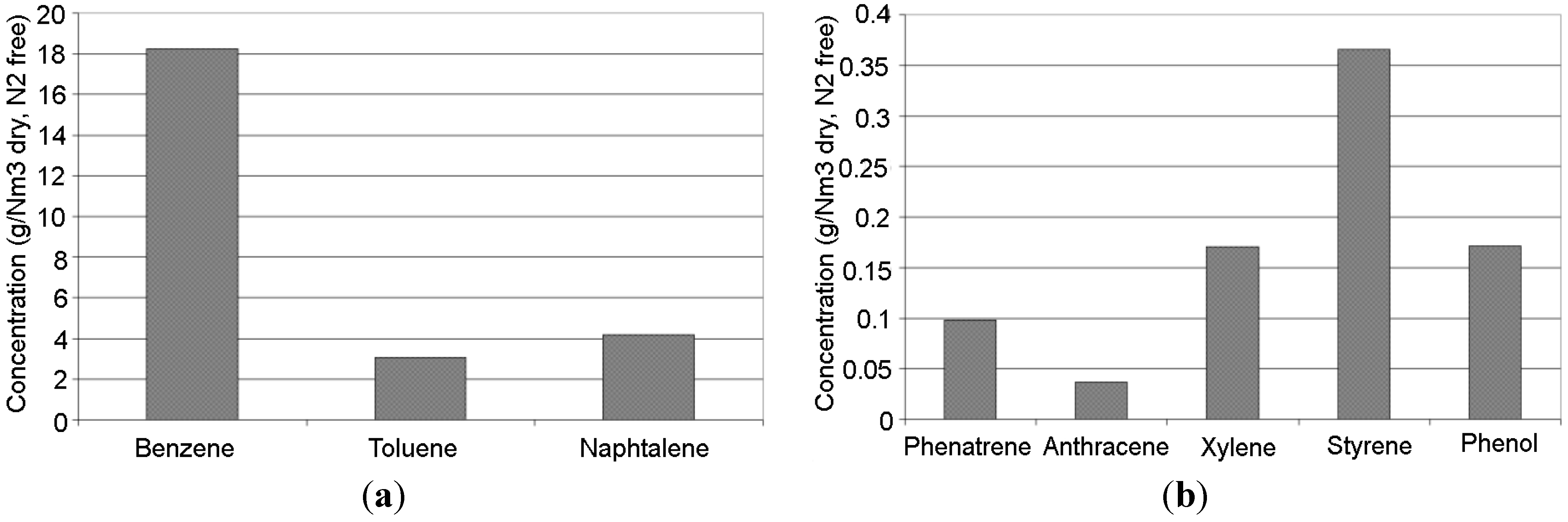
Figure 2.
Reference TAR concentration in the gas obtained from steam gasification of biomass at 850 °C (dry, N2 free): (a) benzene, toluene, naphthalene; (b) phenanthrene, anthracene, xylene, styrene, phenol.
Figure 2.
Reference TAR concentration in the gas obtained from steam gasification of biomass at 850 °C (dry, N2 free): (a) benzene, toluene, naphthalene; (b) phenanthrene, anthracene, xylene, styrene, phenol.
In
Figures 3, the behaviour of N
2-free content of raw gas principal components (H
2, CO
2, CO and CH
4) downstream Sorption Enhanced Steam Reforming is shown.
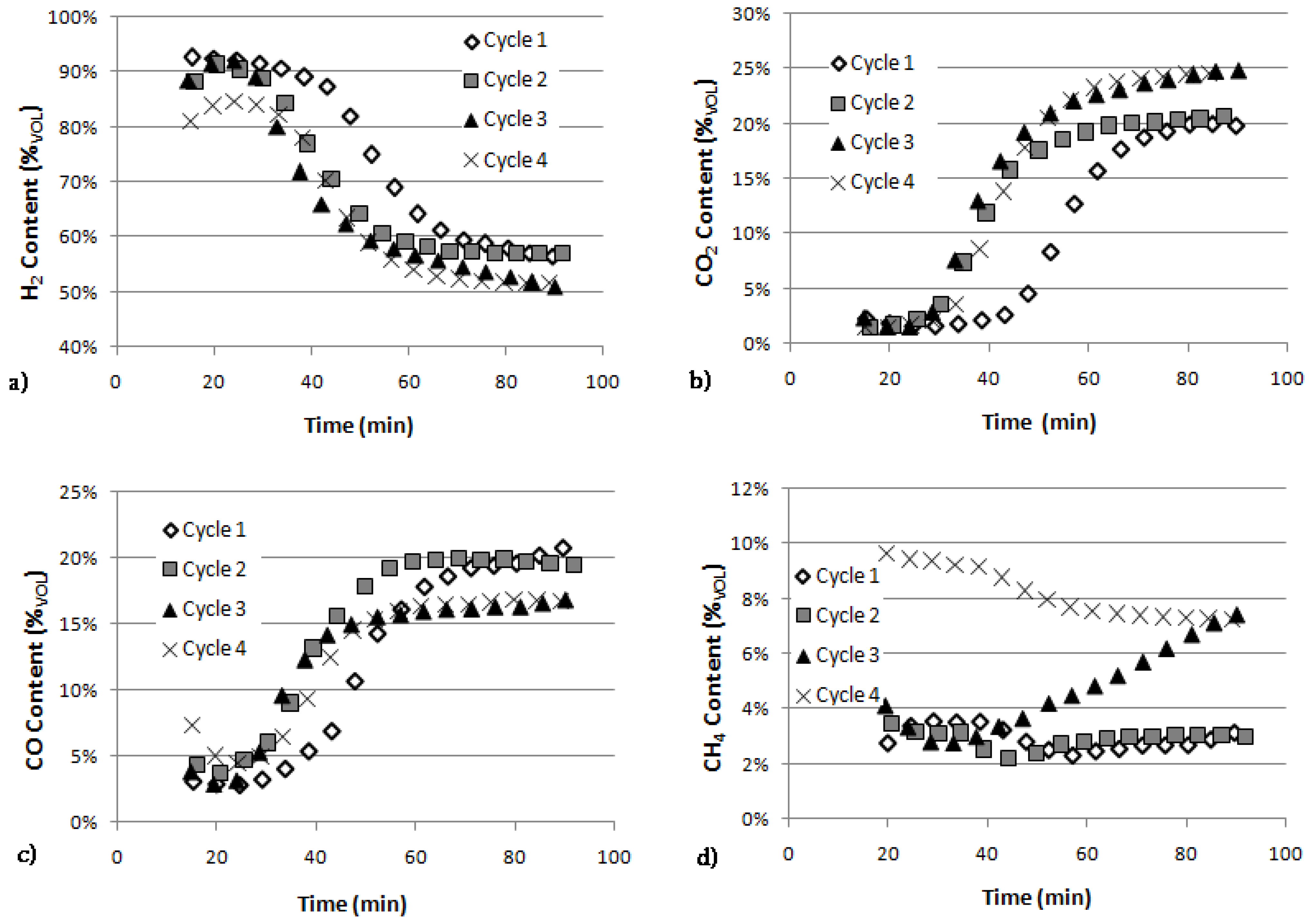
Figure 3a represents the H
2 response curves of Ni catalyst—Dolomite SESR during four subsequent cycles. For all the curves it is possible to distinguish well the three previously described sections. The first cycle curve shows the best behaviour in H
2 yield with values that reach 92% in the pre-breakthrough section (equal to the result achievable by thermodynamic analysis) and a successive softer decrease. The other cycles display instead an increasingly accentuated delay in reaching the initial plateau, a lower maximum value and a faster breakthrough transient.
The CO
2 trends for the different cycles (
Figure 3b) essentially confirm what already underlined by the H
2 response curves, showing a strong decay in sorption efficiency even between the first two cycles.
The CO content behaviour (
Figure 3c) proves the influence of the sorbent in water gas shift reaction activity. In the pre-breakthrough section the CO percentage is minimized due to both TAR reforming (that produces CO) and contemporary WGS reactions, with efficiency getting worse during the subsequent cycles. Then in the post-breakthrough section the lack of absorption activity causes a reduced effect of WGS and a gradual increase of the CO content. Furthermore a more rapid decrease in TAR reforming reactions activity compared to water gas shift’s one is observed during the cycles.
Finally,
Figure 3d confirms the behaviour of the other components content, highlighting the Ni catalyst decay in methane reforming during the subsequent cycles. Moreover CH
4 behaviour can be used as reference to evaluate high hydrocarbon steam reforming. 3
th cycle trend is the one that best demonstrates the catalyst activity decay.
Figure 3.
Content within the raw gas (expressed as %vol N2-free) during four subsequent cycles of SESR.
Figure 3.
Content within the raw gas (expressed as %vol N2-free) during four subsequent cycles of SESR.
This decay is also visible from
Figures 4 and
Figure 5, which represent the TARs conversion rate for the sum of the detected TARs and for each of the following TAR classes:
benzene;
1-ring: toluene (most abundant), xylene, styrene;
phenol;
2-rings: naphthalene;
3-rings: phenanthrene, anthracene;
4-rings: pyrene.
TARs conversion rate has been calculated by comparing it to a no-reforming test performed under the same gasifier steady state conditions.
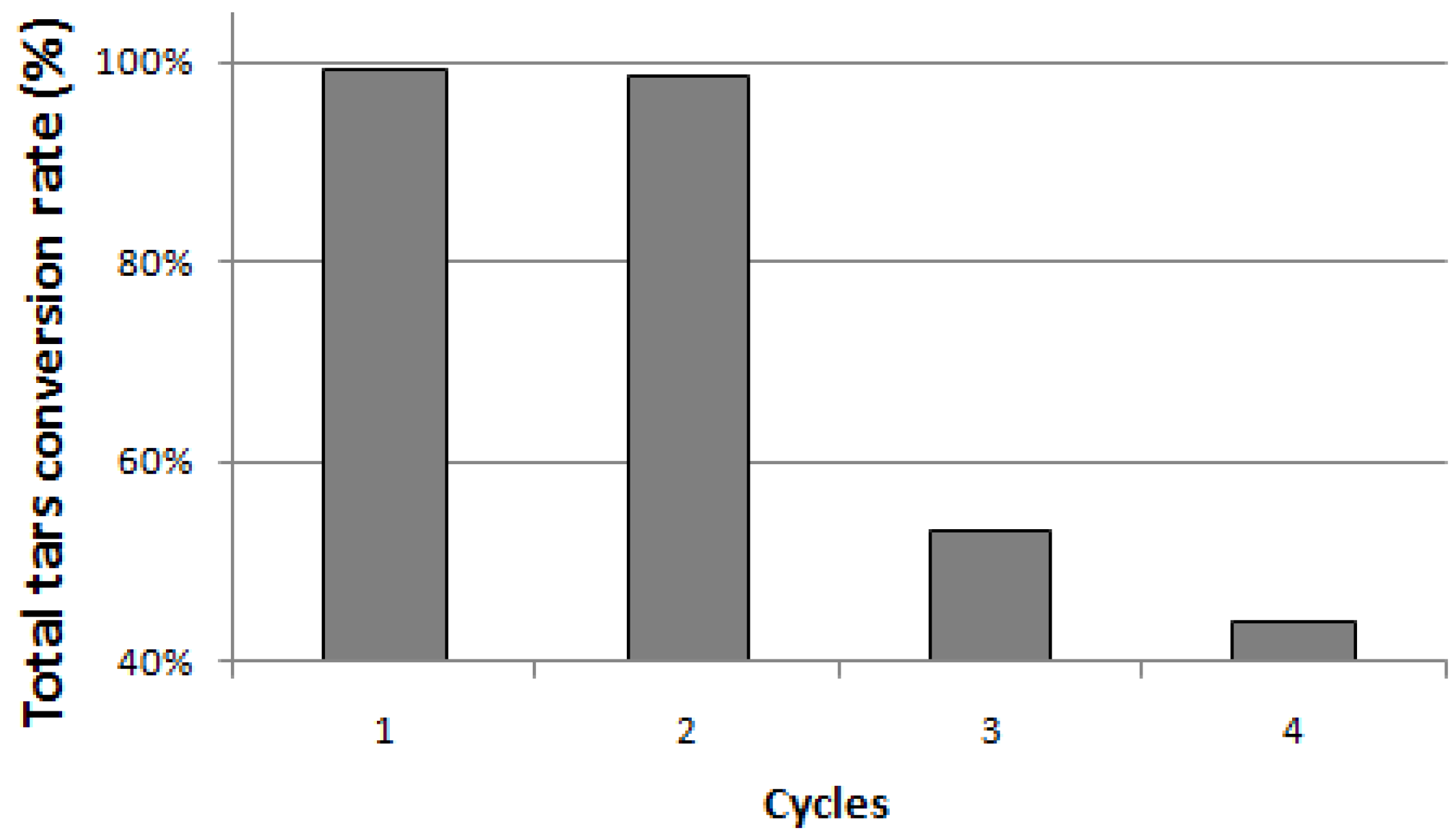
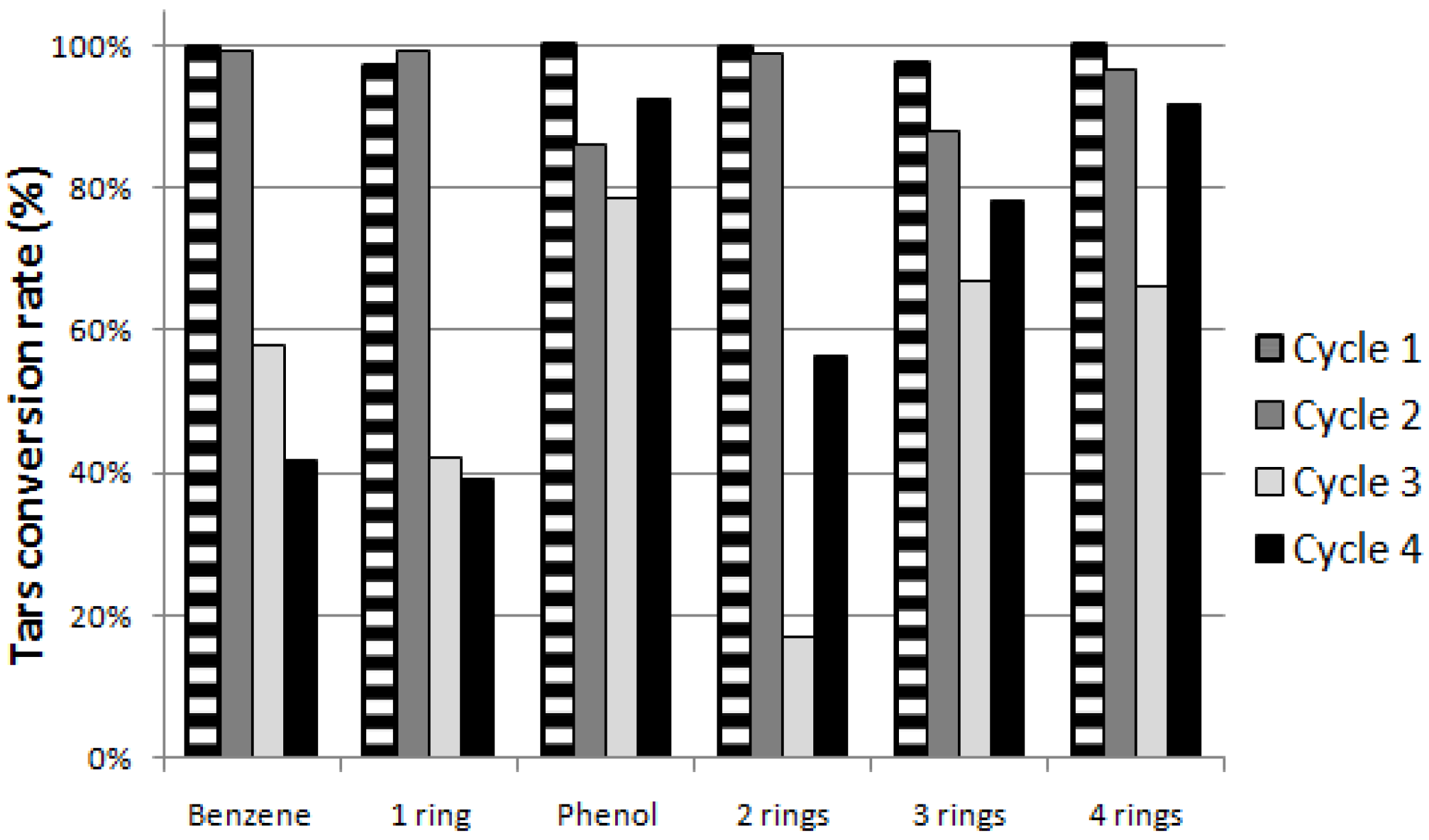
Figure 4 shows values of total TARs conversion rate close to 100% for both the first and the second cycle. On the contrary, between the second and the third cycle, in agreement with CH
4 content rise, a significant decrease (up to 53%) is observed.
The graph of
Figure 5 confirms the initial strong activity of Ni/Al
2O
3 for each TAR class, with values of conversion rate exceeding by 99% on average, but also shows a subsequent great decrease especially for benzene and 1 ring hydrocarbons (toluene, xylene, and styrene).
Figure 4.
Total TARs conversion rates during the cycles.
Figure 4.
Total TARs conversion rates during the cycles.
Figure 5.
TAR conversion rate (%) for single TAR classes during the cycles.
Figure 5.
TAR conversion rate (%) for single TAR classes during the cycles.
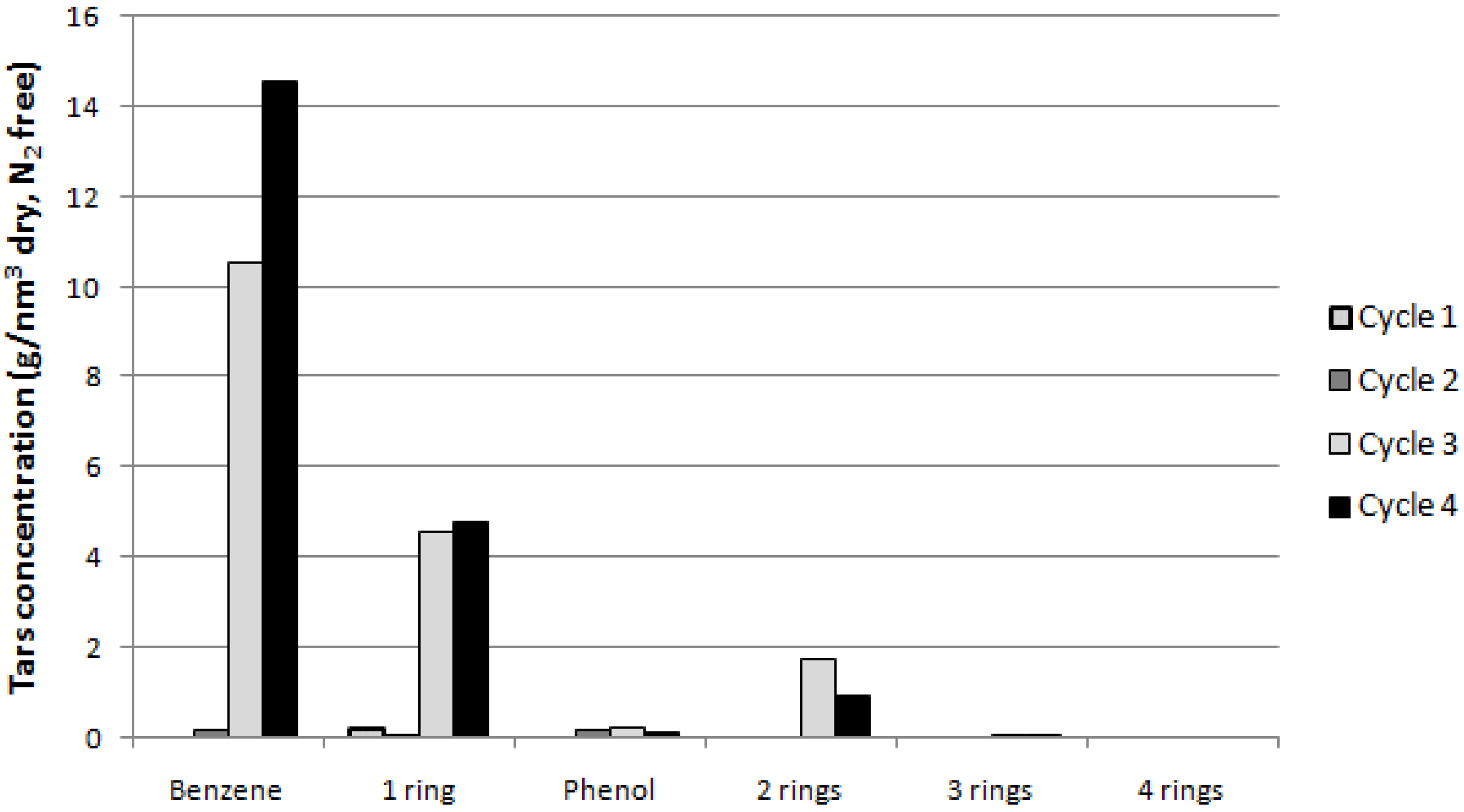
Then
Figure 6 (reporting the TAR concentration within the gas stream for each class), showing large amounts of benzene and 1-ring TAR in the last two cycles compared to the others, justifies the low values of the reported total TARs conversion rate.
Dolomite behaviour through multiple CO2 capture and release cycles, clearly described by the response curves, was also confirmed by the measurement of the CO2 released during calcinations.
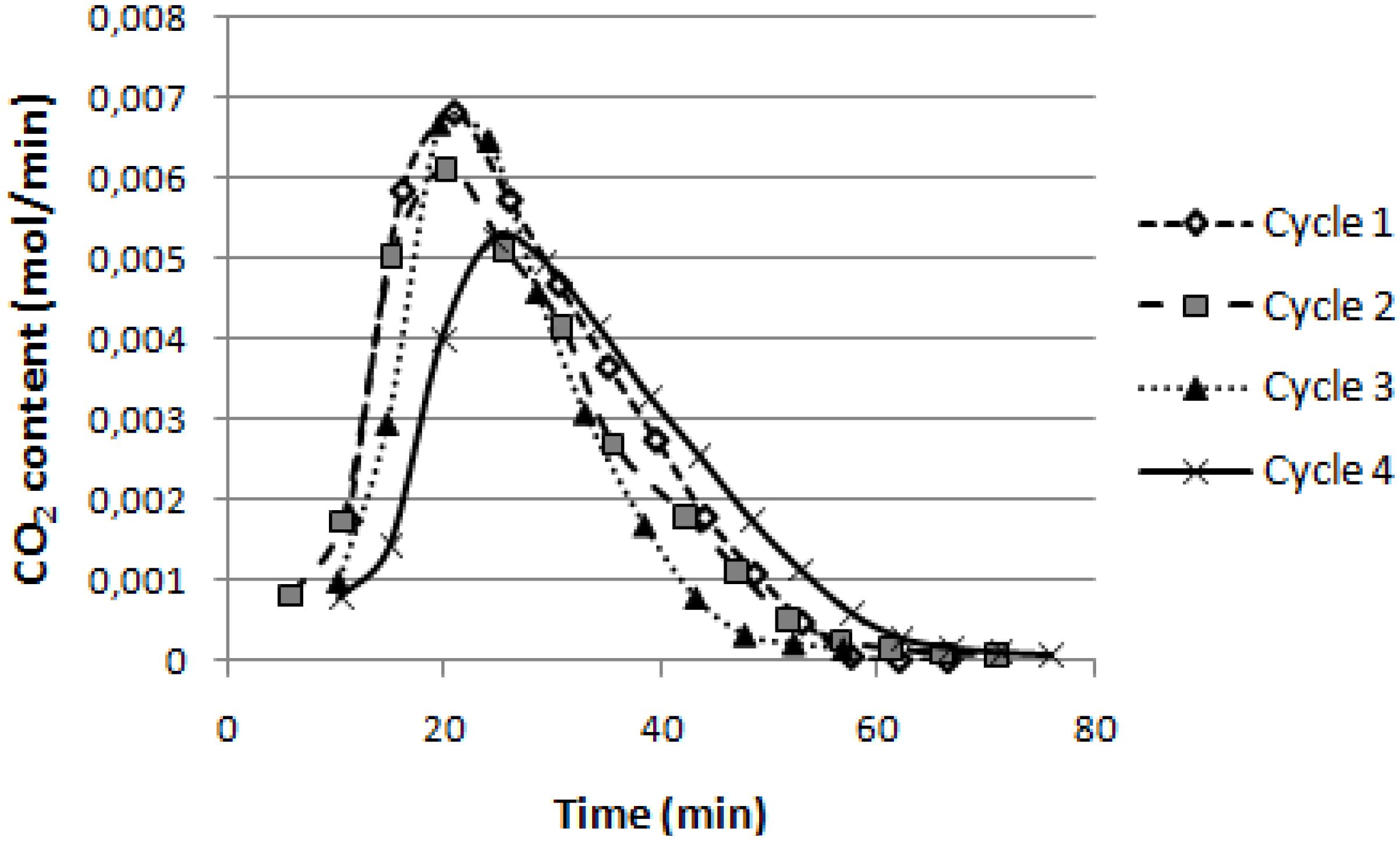
In
Figure 7, the CO
2 content behavior for each cycle expressed in moles per minute is reported. The total amount of released CO
2 (obtained as integral of each curve) can be related to the total amount of CO
2 captured during the previous phase without making significant mistakes. Furthermore calculation of the absorbed CO
2 moles by mean of
Figure 7 curves integral gives more accurate qualitative and quantitative information than the CO
2 response curve (
Figure 3b) because it’s not dependent on time and on any possible small fluctuation in the nitrogen flow rate. It is easy to notice that going forward with the cycles the area subtended to the curves decreases.
This result can also be expressed in terms of total moles of released—absorbed CO
2 and in terms of absorption efficiency, as summarized in
Table 7.
Table 7.
Results of calcination phase for all the cycles.
Table 7.
Results of calcination phase for all the cycles.
| Efficiency Expression | Cycle 1 | Cycle 2 | Cycle 3 | Cycle 4 |
|---|
| Moles of released CO2 (mol) | 0.165 | 0.147 | 0.143 | 0.144 |
| Sorbent conversion (%) | 81.3 | 72.2 | 70.6 | 71.0 |
| Moles or released CO (mol) | 0.010 | 0.024 | 0.008 | 0.018 |
Figure 6.
TAR concentration (g/Nm3 dry, N2 free) for single TAR classes during the cycles.
Figure 6.
TAR concentration (g/Nm3 dry, N2 free) for single TAR classes during the cycles.
Figure 7.
CO2 content within the N2 stream (expressed in moles per minute) for all cycles calcinations phase.
Figure 7.
CO2 content within the N2 stream (expressed in moles per minute) for all cycles calcinations phase.
Absorption efficiency was calculated as the ratio between the moles of released CO
2 and the total ones calculated stoichiometrically from carbonation reaction (equal to the moles of CaO in the bed material). Results show high efficiency during the first cycle (81.3% on average), followed by expected lower values (as already perceived in
Figure 3). Hence its trend confirms the dolomite progressive loss of sorption capacity during cycles, often accompanied with structural modification, pore volume and surface area reduction, as widely reported in literature [
13,
24].
Table 7 also reports the total amount of CO detected within the gas stream during the calcination phase. Furthermore traces of CO
2 within the air stream during the subsequent combustion phase have been detected. Presence of CO together with the detected CO
2 traces give information about catalyst deactivation by carbon deposition. As a matter of fact CO can be only generated during calcination by reaction of carbon with CO
2 in Boudouard’s equilibrium. As a consequence, as already verified in literature [
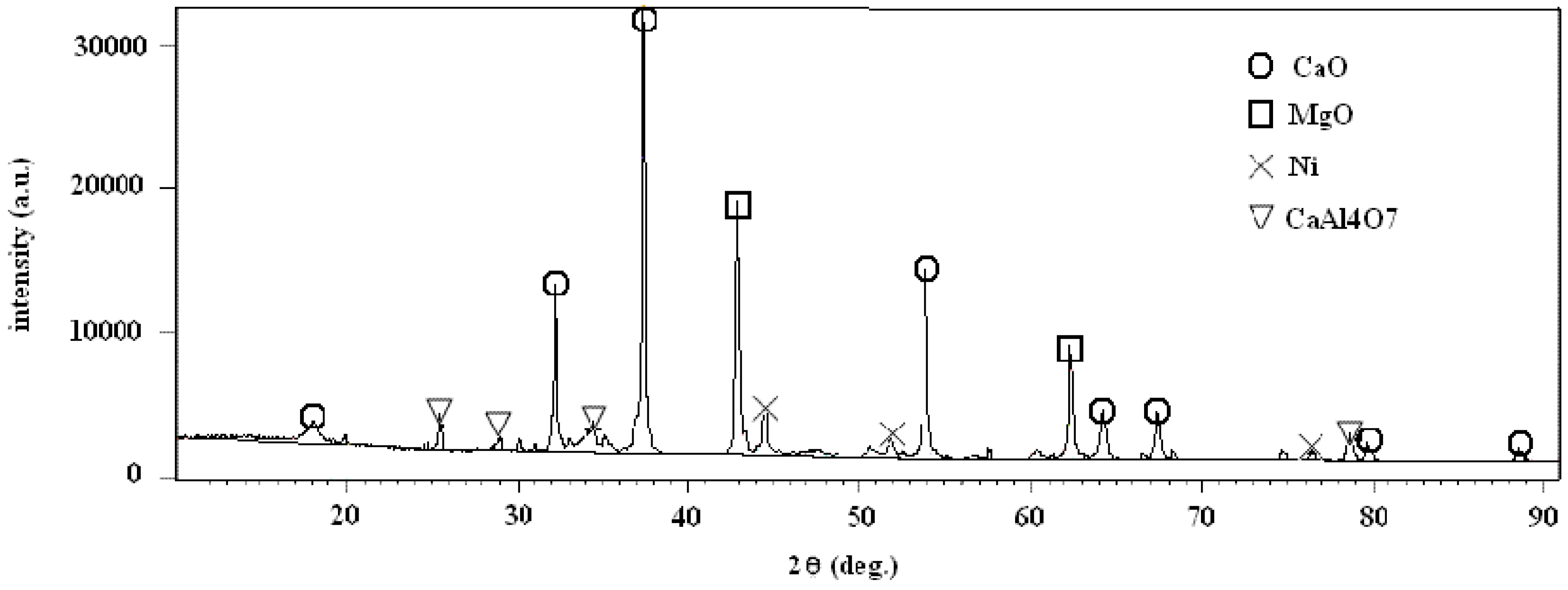
24], the presence of CO can partly justify the observed decay in catalyst efficiency. The use of a higher steam to carbon ratio could solve this problem. The decay of the catalyst activity could be due to the formation at high temperature of Ni aluminates that would not be reduced at 850 °C, the amount of metallic nickel available as catalyst would be thus lower. For this reason XRD analysis of the spent sorbent/catalyst after tests was carried out. The results of the analysis are shown in
Figure 8, no Ni aluminates were observed, the relevant phases are CaO, MgO metallic Ni and Grossite (CaAl
4O
7), and this last one is the binder of the commercial catalyst.
Figure 8.
XRD analysis of the spent sorbent/catalyst after tests.
Figure 8.
XRD analysis of the spent sorbent/catalyst after tests.
As a matter of fact it must be said that catalyst used for the tests has an amount of Ni of 16.6% that can be considered high.
Catalyst can suffer from metallic Ni thermal degradation and sintering as described in [
36], especially during the regeneration phase in an air environment where higher temperatures are reached on the Ni surface during carbon combustion. It is also important to underline that CO
2 absorption also decreases after four cycles which is a low number for a real industrial application.