Experimental Study on the Combustion and Microexplosion of Freely Falling Gelled Unsymmetrical Dimethylhydrazine (UDMH) Fuel Droplets
Abstract
:1. Introduction
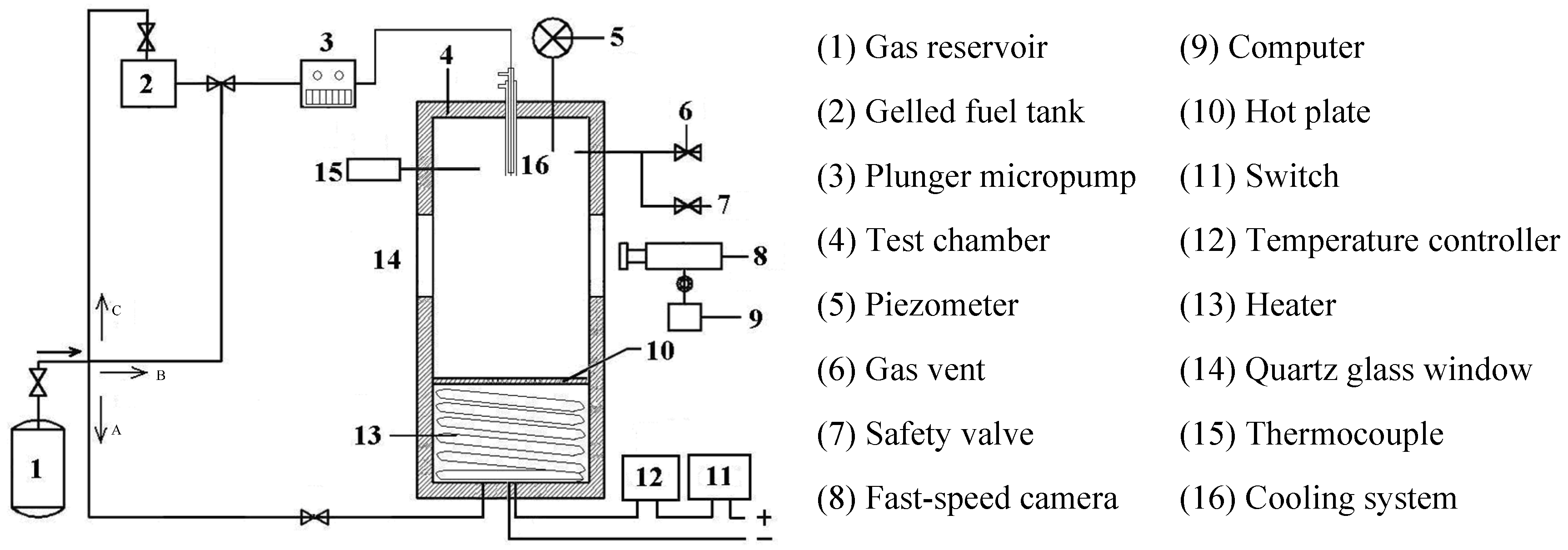
2. Experimental Setup
3. Results and Discussion
3.1. Freely Falling Gelled UDMH Droplets

3.2. Gelled UDMH Droplet on Hot Plate

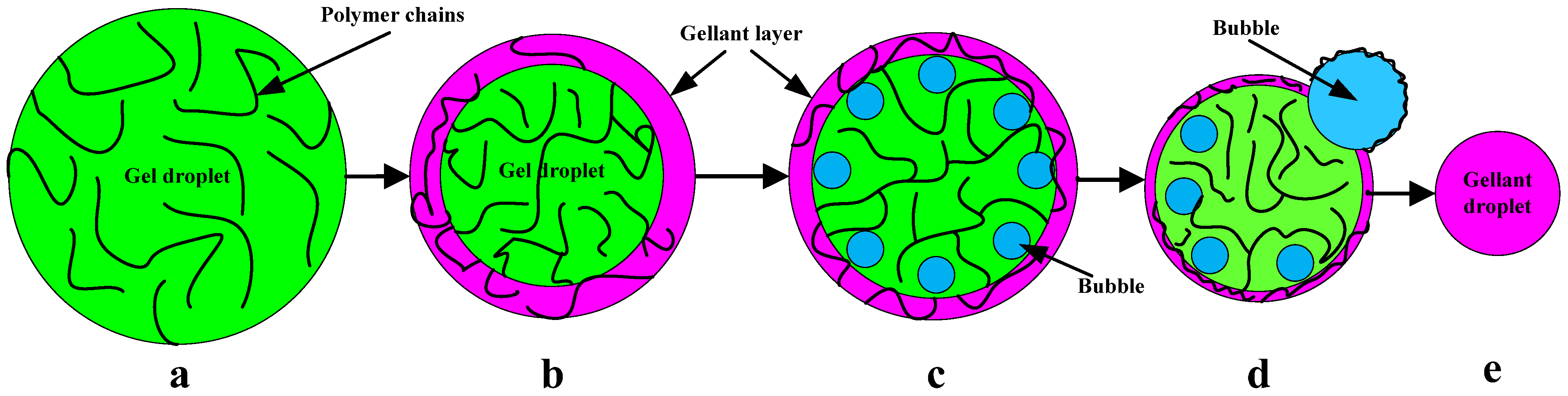
3.3. Burning Rate
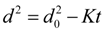
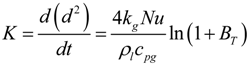
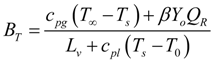
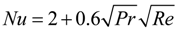
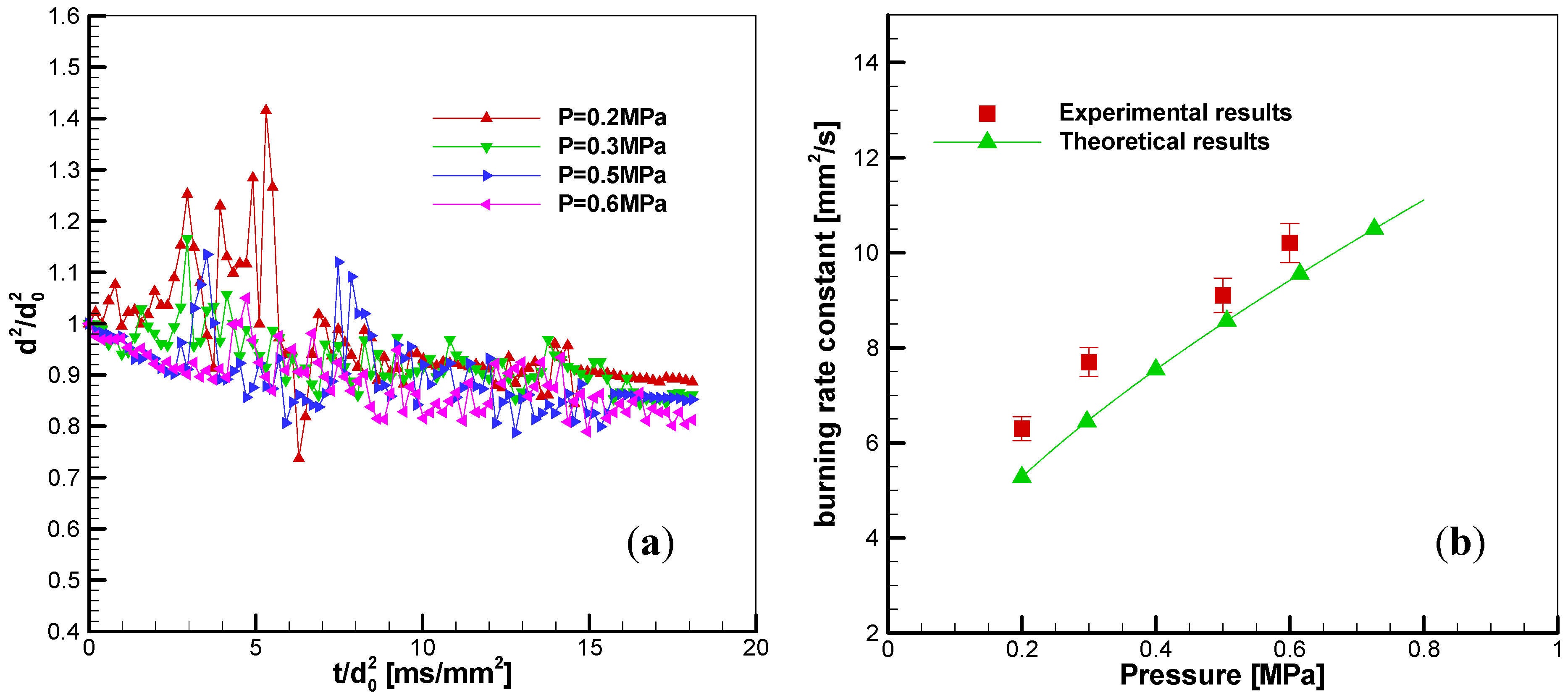
3.3.1. Effects of Pressure
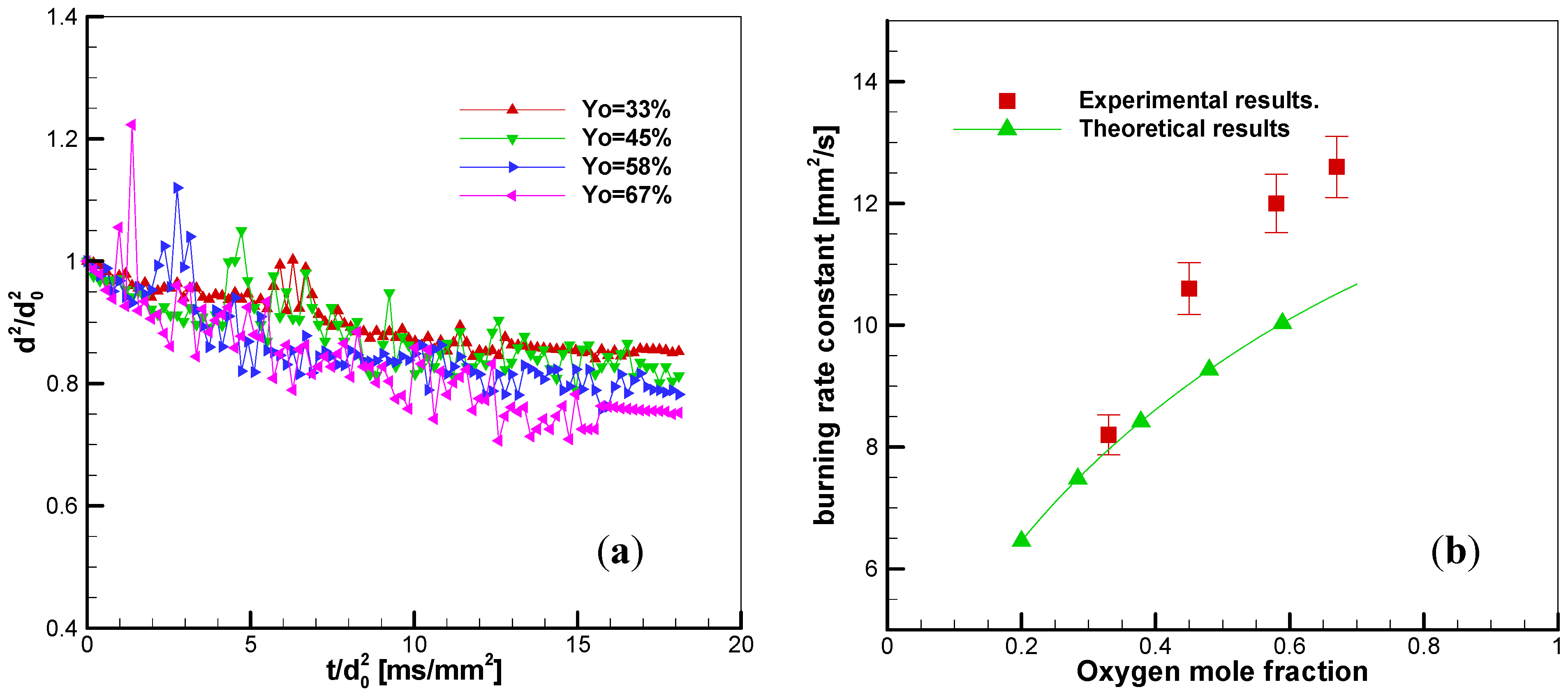
3.3.2. Effects of Oxygen Fraction
4. Conclusions
Acknowledgments
References
- Natan, B.; Rahimi, S. The status of gel propellants in year 2000. Int. J. Energ. Mater. Chem. Propuls. 2002, 5, 172–194. [Google Scholar]
- Sergei, S.S. Advanced models of fuel droplet heating and evaporation. Prog. Energy Combust. Sci. 2006, 32, 162–214. [Google Scholar] [CrossRef]
- Solomon, Y.; Natan, B. Combustion Characteristics of Organic-Gellant-Based Gel Fuels. In Rocket Propulsion: Present and Future, Proceedings of the 8th International Workshop on Combustion and Propulsion, Pozzuoli, Italy, 16–20 June 2003.
- Solomon, Y.; Natan, B. Experimental investigation of the organic gellant-based gel fuel droplets. Combust. Sci. Technol. 2006, 178, 1185–1199. [Google Scholar] [CrossRef]
- Ghassemi, H.; Seung, W.B.; Qasim, S.K. Experimental Study on Binary Droplet Evaporation at Elevated Pressure and Temperature. In Proceedings of the 43rd AIAA Aerospace Sciences Meeting and Exhibit, Reno, NV, USA, 10–13 January 2005. AIAA-2005-353.
- Nahamoni, G.; Natan, B. Investigation of the Combustion Process of Gel Propellants. In Proceedings of the 33rd AIAA/ASME/SAE/ASEE Joint Propulsion Conference and Exhibit, Seattle, WA, USA, 6–9 July 1997. AIAA-1997-2973.
- Nachmoni, G.; Natan, B. Combustion characteristics of gel fuels. Combust. Sci. Technol. 2000, 156, 139–157. [Google Scholar] [CrossRef]
- Arnold, R.; Santos, P.H.S.; Deridder, M.; Campanella, O.H.; Anderson, W.E. Comparison of Monomethylhydrazine/Hydroxypropylcellulose and Hydrocarbon/Silica Gels. In Proceedings of the 48th AIAA Aerospace Sciences Meeting Including the New Horizons Forum and Aerospace Exposition, Orlando, FL, USA, 4–7 January 2010. AIAA-2010-422.
- Arnold, R.; Anderson, W.E. Droplet burning of JP-8/Silica Gels. In Proceedings of the 48th AIAA Aerospace Sciences Meeting Including the New Horizons Forum and Aerospace Exposition, Orlando, FL, USA, 4–7 January 2010. AIAA-2010-421.
- Solomon, Y.; Natan, B.; Cohen, Y. Combustion of gel fuels based on organic gellants. Combust. Flame 2009, 156, 261–268. [Google Scholar] [CrossRef]
- Kunin, A.; Natan, B.; Greenberg, J.B. Preliminarily Model of the Transient Combustion of Organic-Gellant-Based Gel Fuel Droplets. In Proceedings of the 44th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Hartford, CT, USA, 21–23 July 2008. AIAA-2008-4871.
- Kunin, A.; Natan, B.; Greenberg, J.B. Modeling the evaporation of gel fuel droplets. Prog. Propuls. Phys. 2009, 1, 263–274. [Google Scholar]
- Kunin, A.; Natan, B.; Greenberg, J.B. Theoretical model of the transient combustion of organic-gellant-based gel fuel droplets. J. Propuls. Power 2010, 26, 765–771. [Google Scholar] [CrossRef]
- Mishra, D.P.; Patyal, A.; Padwal, M. Effects of gellant concentration on the burning and flame structure of organic gel propellant droplet. Fuel 2010, 90, 1805–1810. [Google Scholar] [CrossRef]
- Mishra, D.P.; Patyal, A. Effects of initial droplet diameter and pressure on burning of ATF gel propellant droplets. Fuel 2012, 95, 226–233. [Google Scholar] [CrossRef]
- Antaki, P. Transient processes in a rigid slurry droplet during liquid vaporization and combustion. Combust. Sci. Technol. 1986, 46, 113–135. [Google Scholar] [CrossRef]
- Yanan, G.; Qiao, L. Combustion characteristics of fuel droplet with addition of nano and micro-sized aluminum particles. Combust. Flame 2011, 158, 354–368. [Google Scholar] [CrossRef]
- Yanan, G.; Qiao, L. Burning characteristics of fuel droplet with addition of nanoparticles under nature and forced convections. Int. J. Heat Mass Transf. 2011, 54, 4913–4922. [Google Scholar] [CrossRef]
- Gan, Y.; Lim, Y.S.; Qiao, L. Combustion of nanofluid fuels with the addition of boron and iron particles at dilute and dense concentrations. Combust. Flame 2012, 159, 1732–1740. [Google Scholar] [CrossRef]
- Solomon, Y.; DeFini, S.J.; Poupoint, T.L.; Anderson, W.E. Gelled MMH hypergolic Droplet Investigation. In Proceedings of the 47th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, San Diego, CA, USA, 31 July–3 August 2011. AIAA-2011-5705.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, Z.; Hu, X.; He, Z.; Wu, J. Experimental Study on the Combustion and Microexplosion of Freely Falling Gelled Unsymmetrical Dimethylhydrazine (UDMH) Fuel Droplets. Energies 2012, 5, 3126-3136. https://doi.org/10.3390/en5083126
Liu Z, Hu X, He Z, Wu J. Experimental Study on the Combustion and Microexplosion of Freely Falling Gelled Unsymmetrical Dimethylhydrazine (UDMH) Fuel Droplets. Energies. 2012; 5(8):3126-3136. https://doi.org/10.3390/en5083126
Chicago/Turabian StyleLiu, Zejun, Xiaoping Hu, Zhen He, and Jianjun Wu. 2012. "Experimental Study on the Combustion and Microexplosion of Freely Falling Gelled Unsymmetrical Dimethylhydrazine (UDMH) Fuel Droplets" Energies 5, no. 8: 3126-3136. https://doi.org/10.3390/en5083126
APA StyleLiu, Z., Hu, X., He, Z., & Wu, J. (2012). Experimental Study on the Combustion and Microexplosion of Freely Falling Gelled Unsymmetrical Dimethylhydrazine (UDMH) Fuel Droplets. Energies, 5(8), 3126-3136. https://doi.org/10.3390/en5083126



