Deacidification of Pistacia chinensis Oil as a Promising Non-Edible Feedstock for Biodiesel Production in China
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Apparatus
2.2. Oil Extraction and Biodiesel Production Procedures
2.3. Analysis
2.3.1. Physicochemical Parameters of Crude Oil and Biodiesel
2.3.2. Biodiesel Chemical Composition
3. Results and Discussion
3.1. Crude Oil Extraction and Parameter Determination
| Extraction method | Seed/g | Oil/g | Yield/% |
|---|---|---|---|
| Leaching | 50 | 13.25 | 26.5 |
| Reflux | 30 | 7.50 | 25.0 |
| Soxhlet | 10 | 3.13 | 31.3 |
| Ultrasonic | 10 | 2.76 | 27.6 |
3.2. Deacidification of Refined Oil
| Acid value of crude oil | Alkali concentration/°Bé | Operating temperature/°C | |
|---|---|---|---|
| Start | End | ||
| <5 | 4~6 | 75~80 | 90~95 |
| 5~7 | 12~14 | 50~55 | 60~65 |
| 7~9 | 16~24 | 25~30 | 45~50 |
| >9 | >24 | 20~30 | 20~30 |

| Levels | The volume ratio of oil to ethanol (A) | Extraction temperature (B) | Extraction times (C) | Blank |
|---|---|---|---|---|
| 1 | 1:1 | 20 °C | 1 | 1 |
| 2 | 1:2 | 30 °C | 2 | 2 |
| 3 | 1:3 | 40 °C | 3 | 3 |
| Test No. | The volume ratio of oil to ethanol (A) | Extraction temperature (°C) (B) | Extraction times (C) | Blank | Acid value (mg KOH/g) |
|---|---|---|---|---|---|
| 1 | 1:1 | 20 | 1 | 1 | 22.51 |
| 2 | 1:1 | 30 | 2 | 2 | 9.87 |
| 3 | 1:1 | 40 | 3 | 3 | 4.37 |
| 4 | 1:2 | 20 | 2 | 3 | 4.38 |
| 5 | 1:2 | 30 | 3 | 1 | 0.55 |
| 6 | 1:2 | 40 | 1 | 2 | 13.62 |
| 7 | 1:3 | 20 | 3 | 2 | 1.13 |
| 8 | 1:3 | 30 | 1 | 3 | 9.03 |
| 9 | 1:3 | 40 | 2 | 1 | 1.65 |
| K1 | 36.75 | 28.02 | 45.16 | 24.71 | |
| K2 | 18.55 | 19.45 | 15.90 | 24.62 | |
| K3 | 11.81 | 19.64 | 6.05 | 17.78 | |
| 12.250 | 9.340 | 15.053 | 8.237 | ||
| 6.183 | 6.483 | 5.300 | 8.207 | ||
| 3.937 | 6.547 | 2.017 | 5.927 | ||
| R | 24.94 | 8.57 | 39.11 | 6.93 | |
| Priority factors | C > A > B | ||||
| Optimum solution | K3A < K2A < K1A K2B < K3B < K1B K3C < K2C < K1C | ||||
| A3B2C3 | |||||
3.3. Biodiesel Production and Analysis
3.4. Biodiesel Analysis

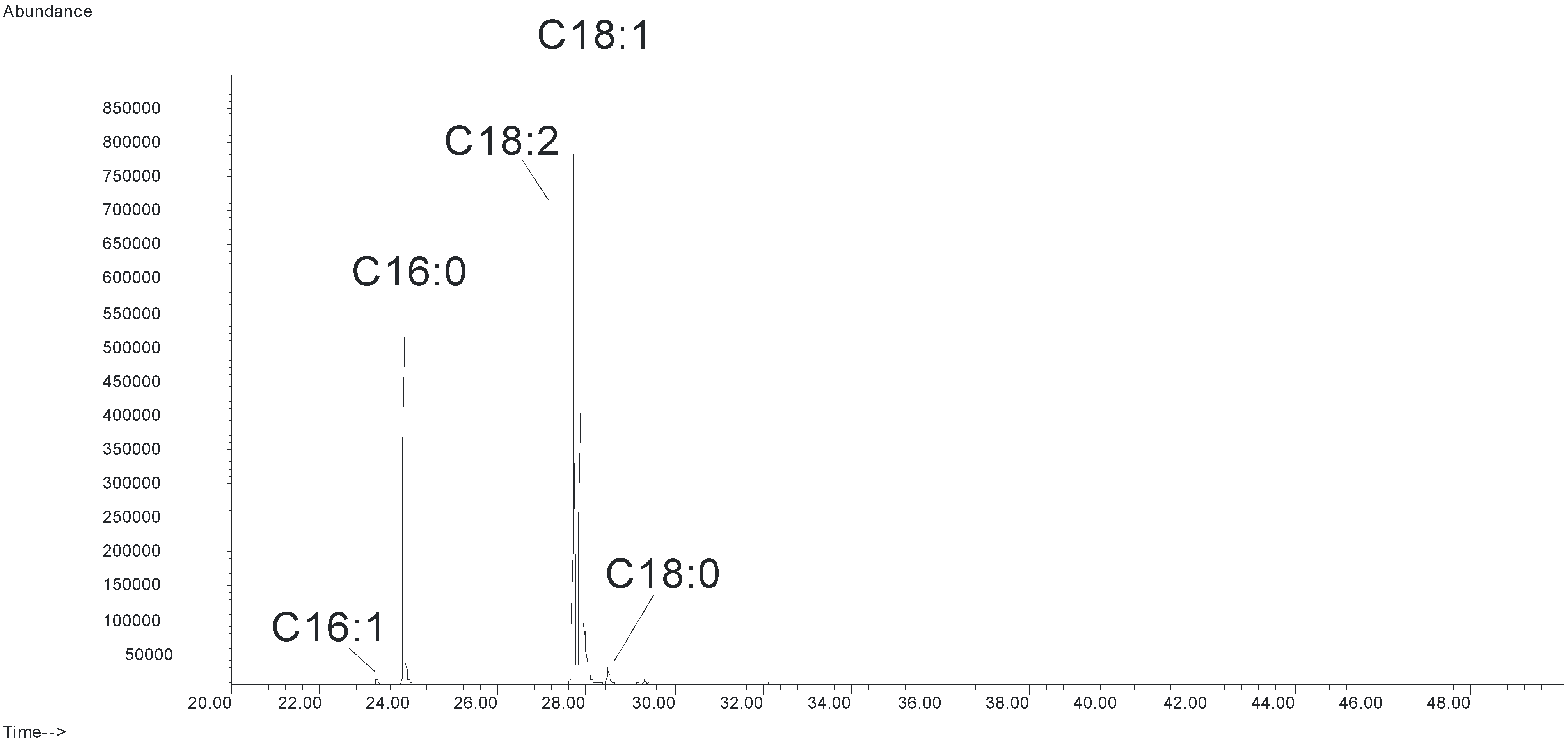
| Components | Methyl palmitate | Methyl linoleate | Methyl oleate | Methyl stearate |
|---|---|---|---|---|
| Formula | C16:0 | C18:2 | C18:1 | C18:0 |
| Composition (%) | 18.93 ± 0.28 | 27.42 ± 0.40 | 52.32 ± 1.15 | 1.33 ± 0.16 |
| Fuel property | Biodiesel | ASTM D6751 | EN 14214 |
|---|---|---|---|
| Density (15 °C), kg m−3 | 887 ± 9.6 * | - | 860–900 |
| Kinematic viscosity (mm2 s−1; 40 °C) | 5.24 ± 0.05 | 1.9–6.0 | 3.5–5.0 |
| Flash point (°C) | 166 ± 2.0 | 130 min | 120 min |
| Cloud point (°C) | 1.00 ± 0.12 | Report | - |
| Pour point (°C) | −2.00 ± 0.15 | - | - |
| Cold filter plugging point (°C) | 0.00 ± 0.10 | - | - |
| Ash content (%) | 0.013 ± 0.001 | 0.02 max | 0.02 max |
| Acid value (mg KOH g−1) | 0.35 ± 0.03 | 0.50 max | 0.50 max |
| Cetane number | 52.2 ± 1.30 | 47 min | 51 min |
4. Conclusions
Acknowledgments
References
- Leung, D.Y.C.; Xuan, W.; Leung, M.K.H. A review on biodiesel production using catalyzed transesterification. Appl. Energy 2010, 87, 1083–1095. [Google Scholar] [CrossRef]
- Gui, M.M.; Lee, K.T.; Bhatia, S. Feasibility of edible oil vs. non-edible oil vs. waste edible oil as biodiesel feedstock. Energy 2008, 33, 1646–1653. [Google Scholar] [CrossRef]
- Vyas, A.P.; Subrahmanyam, N.; Patel, P.A. Production of biodiesel through transesterification of Jatropha oil using KNO3/Al2O3 solid catalyst. Fuel 2009, 88, 625–628. [Google Scholar] [CrossRef]
- De Oliveira, J.S.; Leite, P.M.; De Souza, L.B.; Mello, V.M.; Silva, E.C.; Rubim, J.C.; Meneghetti, S.M.P.; Suarez, P.A.Z. Characteristics and composition of Jatropha gossypiifolia and Jatropha curcas L. oils and application for biodiesel production. Biomass Bioenergy 2009, 33, 449–453. [Google Scholar]
- Meher, L.C.; Dharmagadda, V.S.S.; Naik, S.N. Optimization of alkali-catalyzed transesterification of Pongamia pinnata oil for production of biodiesel. Bioresour. Technol. 2006, 97, 1392–1397. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.H.; Zhang, Y.J.; Xiao, Z.H.; Li, P.W.; Li, C.Z. Synthesis of biodiesel catalyzed by 1-(4-sulfonic acid) butyl-pyridinium hydrosulfate ionic liquid. Petrochem. Technol. 2009, 38, 47–51. [Google Scholar]
- Kansedo, J.; Lee, K.T.; Bhatia, S. Cerbera odollam (sea mango) oil as a promising non-edible feedstock for biodiesel production. Fuel 2009, 88, 1148–1150. [Google Scholar] [CrossRef]
- Anwar, F.; Rashid, U.; Ashraf, M.; Nadeem, M. Okra (Hibiscus esculentus) seed oil for biodiesel production. Appl. Energy 2010, 87, 779–785. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F.; Ashraf, M.; Saleem, M.; Yusup, S. Application of response surface methodology for optimizing transesterification of Moringa oleifera oil: Biodiesel production. Energ. Convers. Manag. 2011, 52, 3034–3042. [Google Scholar] [CrossRef]
- Rashid, U.; Anwar, F.; Knothe, G. Biodiesel from Milo (Thespesia populnea L.) seed oil. Biomass Bioenergy 2011, 35, 4034–4039. [Google Scholar] [CrossRef]
- Shao, H.B.; Chu, L.Y. Resource evaluation of typical energy plants and possible functional zone planning in China. Biomass Bioenergy 2008, 32, 283–288. [Google Scholar]
- Min, E.Z.; Tang, Z.; Du, Z.X.; Wu, W. Perspective of biodiesel industry in China. Eng. Sci. 2005, 7, 1–4. [Google Scholar]
- Hou, X.C.; Mou, H.X.; Yang, S.C.; Xing, Y.Z.; Jing, J. Research progress in woody energy plant Pistacia chinensis. J. Anhui Agri. Sci. 2007, 35, 3524–3525. [Google Scholar]
- Li, H.L.; Zhang, Z.X.; Lin, S.Z.; Li, X.X. Research advances in the study of Pistacia chinensis Bunge, a superior tree species for biomass energy. For. Study China 2007, 9, 164–168. [Google Scholar] [CrossRef]
- Niu, Z.T.; Li, T.; Jian, G.Z.; Zhang, Y.J. Resource survey, cultivation techniques and multipurpose utilization of Pistacia chinensis bunge. Nonwood For. Res. 2005, 23, 68–71. [Google Scholar]
- Qian, X.X.; Zhang, W.M.; Gu, G.P.; Zhang, G.L. The ultilization and cultivation of fuel plant Pistacia chinensis. Chin. Wild Plant Res. 2007, 26, 14–16. [Google Scholar]
- Hu, X.H.; Ni, W.S.; Zhou, Y.; Chen, Y.; Li, Q.; Deng, L. Analytical study on the physical and chemical index and the composition of FA of Pistacia chinensis bge seed oil. J. Wuhan Polytech. Univ. 2007, 26, 8–9. [Google Scholar]
- Chen, L.S.; Peng, F.R.; Liang, Y.W.; Zhang, C.X. Difference analysis of seed morphological characters and oil quality of Pistacia chinensis from different provenances. J. Plant Res. Env. 2009, 18, 16–21. [Google Scholar]
- Yu, X.H.; Wen, Z.; Li, H.L.; Tu, S.T.; Yan, J.Y. Transesterification of Pistacia chinensis oil for biodiesel catalyzed by CaO-CeO2 mixed oxides. Fuel 2011, 90, 1868–1874. [Google Scholar] [CrossRef]
- Qin, S.J.; Sun, Y.Z.; Meng, X.C.; Zhang, S.X. Production and analysis of biodiesel from non-edible seed oil of Pistacia chinensis. Energy Explor. Exploit. 2010, 28, 37–46. [Google Scholar] [CrossRef]
- Marchetti, J.M.; Miguel, V.U.; Errazu, A.F. Heterogeneous esterification of oil with high amount of free fatty acids. Fuel 2007, 86, 906–910. [Google Scholar] [CrossRef]
- Van Gerpen, J. Biodiesel processing and production. Fuel Process. Technol. 2005, 86, 1097–1107. [Google Scholar] [CrossRef]
- Kucek, K.T.; Cesar-Oliveira, M.A.F.; Wilhelm, H.M.; Ramos, L.P. Ethanolysis of refined soybean oil assisted by sodium and potassium hydroxides. J. Am. Oil Chem. Soc. 2007, 84, 385–392. [Google Scholar] [CrossRef]
- Li, C.Z.; Jiang, L.J.; Cheng, S.Q. Green Energy Resources-Biodiesel; Chemical Industrial Press: Beijing, China, 2005. [Google Scholar]
- Wang, Y.; Ou, S.Y.; Liu, P.Z.; Zhang, Z.S. Preparation of biodiesel from waste cooking oil via two-step catalyzed process. Energy Convers. Manag. 2007, 48, 184–188. [Google Scholar] [CrossRef]
- Dorado, M.P.; Ballesteros, E.; Mittelbach, M.; Lopez, F.J. Kinetic parameters affecting the alkali-catalyzed transesterification process of used olive oil. Energy Fuels 2004, 18, 1457–1462. [Google Scholar] [CrossRef]
- Encinar, J.M.; Gonzalez, J.F.; Rodriguez-Reinares, A. Ethanolysis of used frying oil. Biodiesel preparation and characterization. Fuel Process. Technol. 2007, 88, 513–522. [Google Scholar] [CrossRef]
- Monteiro, M.R.; Ambrozin, A.R.P.; Liao, L.M.; Ferreira, A.G. Critical review on analytical methods for biodiesel characterization. Talanta 2008, 77, 593–605. [Google Scholar] [CrossRef]
- Siatis, N.G.; Kimbaris, A.C.; Pappas, C.S.; Tarantilis, P.A.; Polissiou, M.G. Improvement of biodiesel production based on the application of ultrasound: Monitoring of the procedure by FTIR spectroscopy. J. Am. Oil Chem. Soc. 2006, 83, 53–57. [Google Scholar] [CrossRef]
- Knothe, G.; Steidley, K.R. Kinematic viscosity of biodiesel fuel components and related compounds: Influence of compound structure and comparison to petrodiesel fuel components. Fuel 2005, 84, 1059–1065. [Google Scholar] [CrossRef]
© 2012 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Qin, S.; Sun, Y.; Shi, C.; He, L.; Meng, Y.; Ren, X. Deacidification of Pistacia chinensis Oil as a Promising Non-Edible Feedstock for Biodiesel Production in China. Energies 2012, 5, 2759-2770. https://doi.org/10.3390/en5082759
Qin S, Sun Y, Shi C, He L, Meng Y, Ren X. Deacidification of Pistacia chinensis Oil as a Promising Non-Edible Feedstock for Biodiesel Production in China. Energies. 2012; 5(8):2759-2770. https://doi.org/10.3390/en5082759
Chicago/Turabian StyleQin, Shenjun, Yuzhuang Sun, Changlin Shi, Leqin He, Yuan Meng, and Xiaohui Ren. 2012. "Deacidification of Pistacia chinensis Oil as a Promising Non-Edible Feedstock for Biodiesel Production in China" Energies 5, no. 8: 2759-2770. https://doi.org/10.3390/en5082759
APA StyleQin, S., Sun, Y., Shi, C., He, L., Meng, Y., & Ren, X. (2012). Deacidification of Pistacia chinensis Oil as a Promising Non-Edible Feedstock for Biodiesel Production in China. Energies, 5(8), 2759-2770. https://doi.org/10.3390/en5082759




