Heat Transfer Analysis of Methane Hydrate Sediment Dissociation in a Closed Reactor by a Thermal Method
Abstract
:1. Introduction
2. Apparatus and Materials
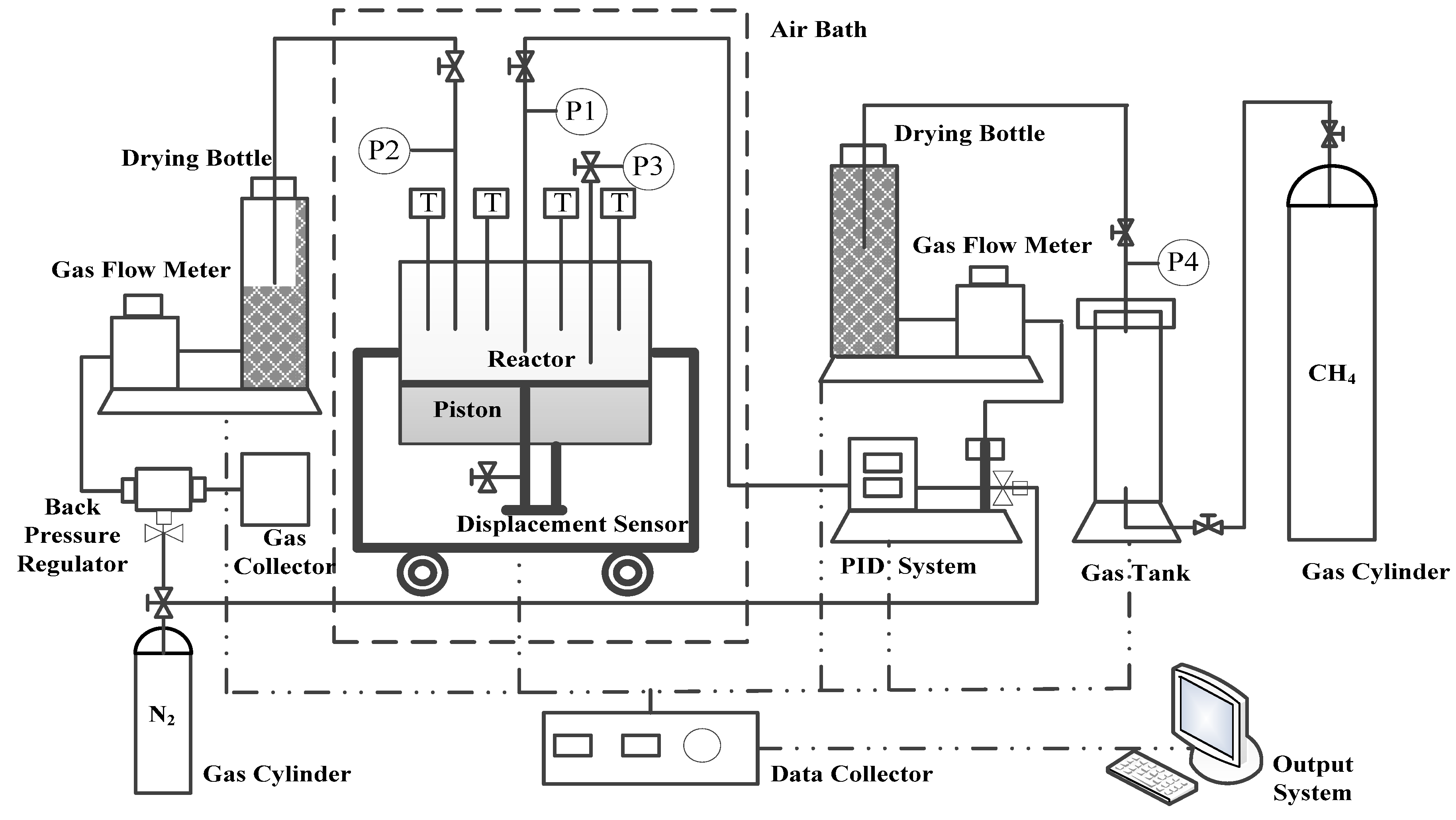
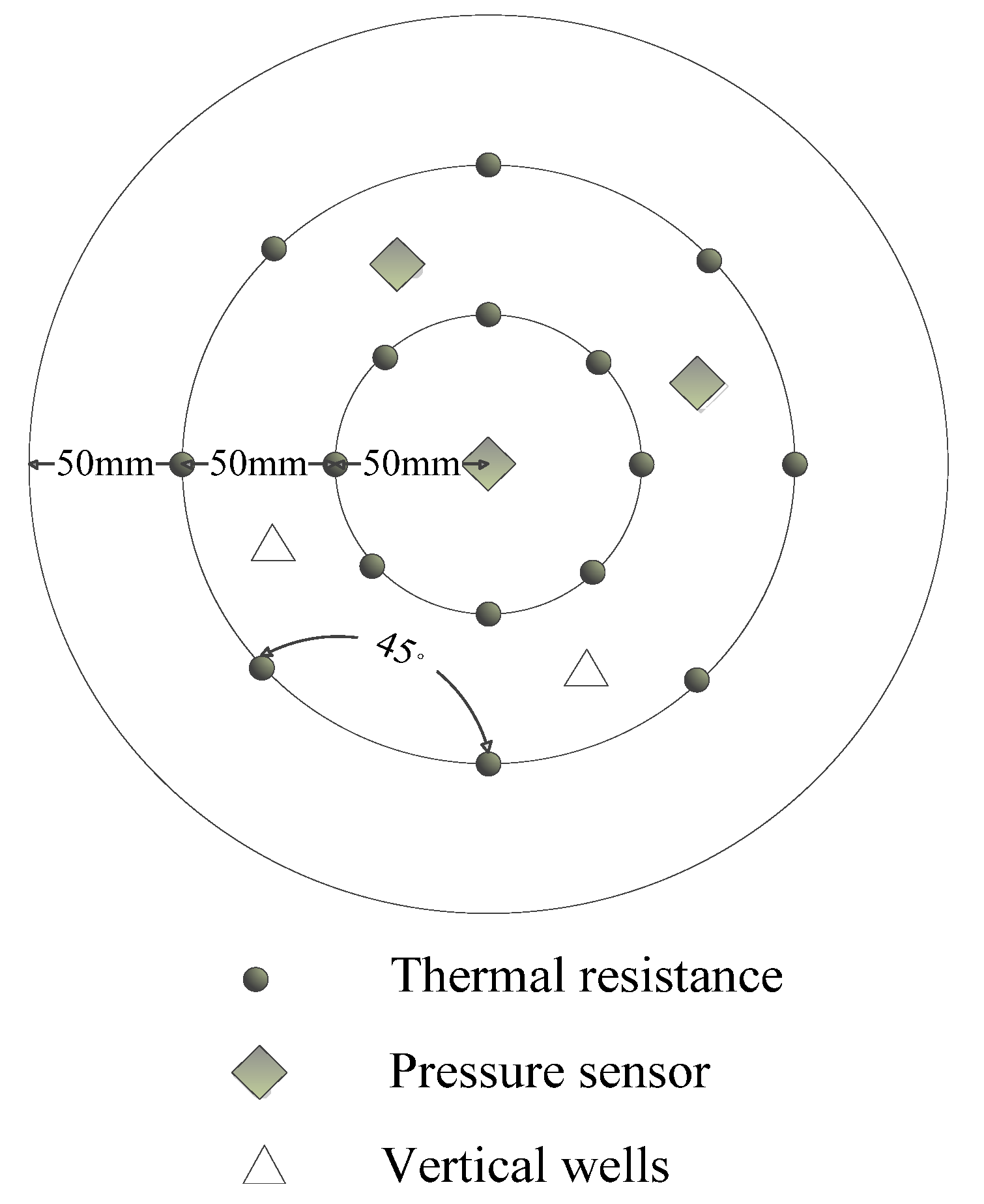
3. Procedure
3.1. Hydrate Formation
| Sample | Specification | Supply |
|---|---|---|
| CH4 | 99.999% | Dalian DATE special gas |
| Deionized water | Pure | Laboratory |
| Quartz sand | BZ-04, Φ-0.361, ρ-2.18 g/cm3 | AS-ONE corporation (Japan) |
3.2. Hydrate Dissociation
4. Results and Discussion
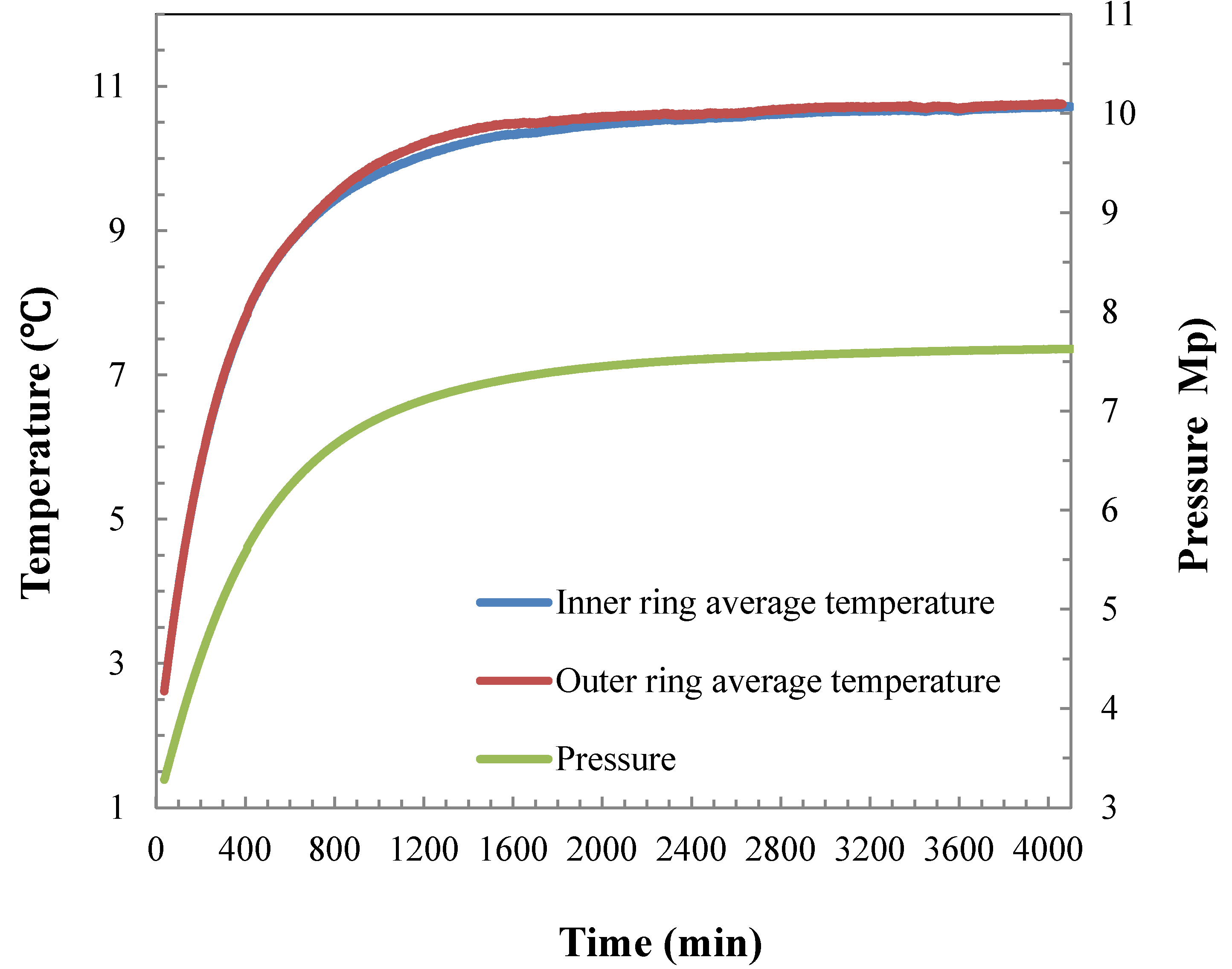
4.1. Hydrate Formation Characteristics in the 5 L Reactor
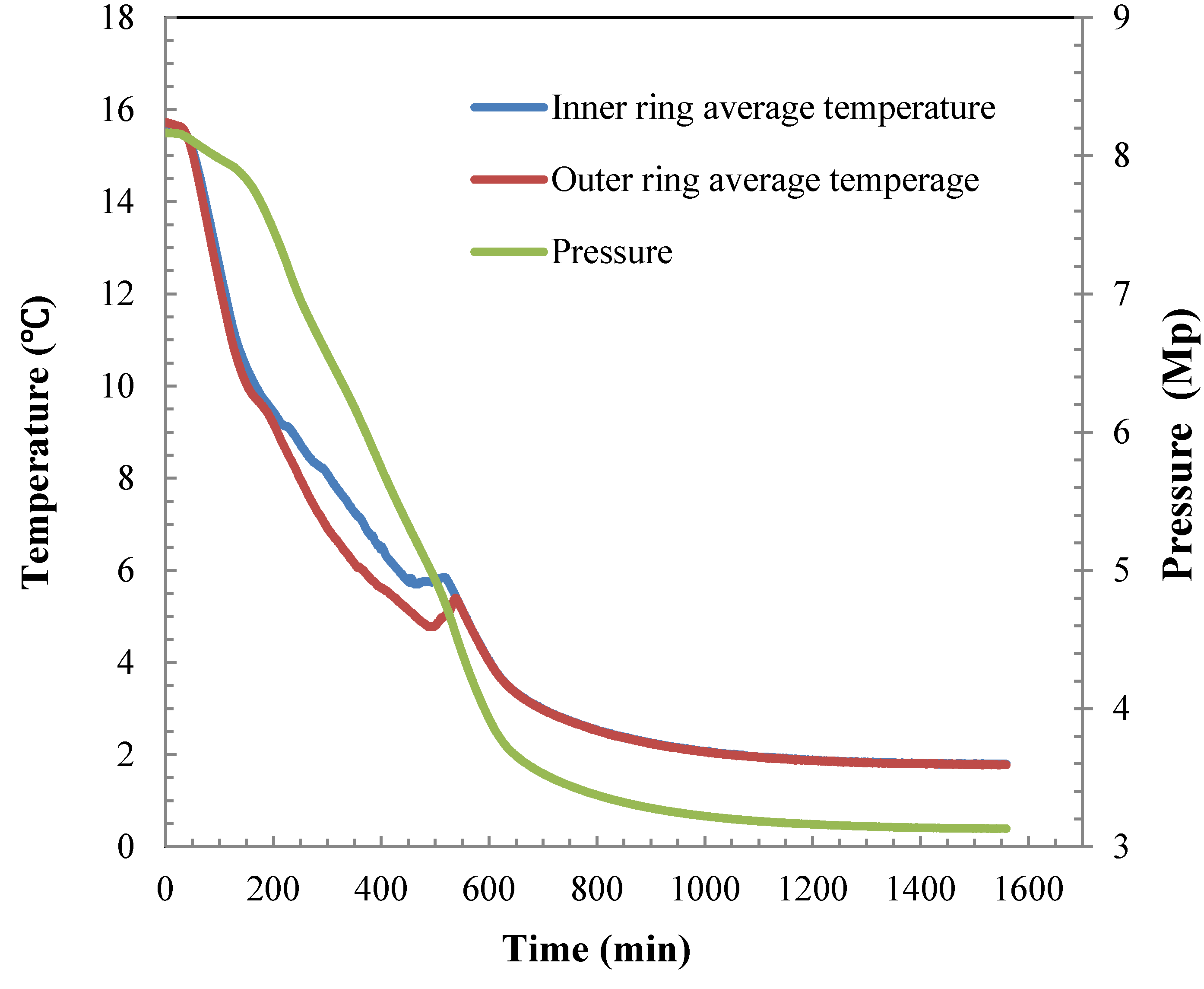
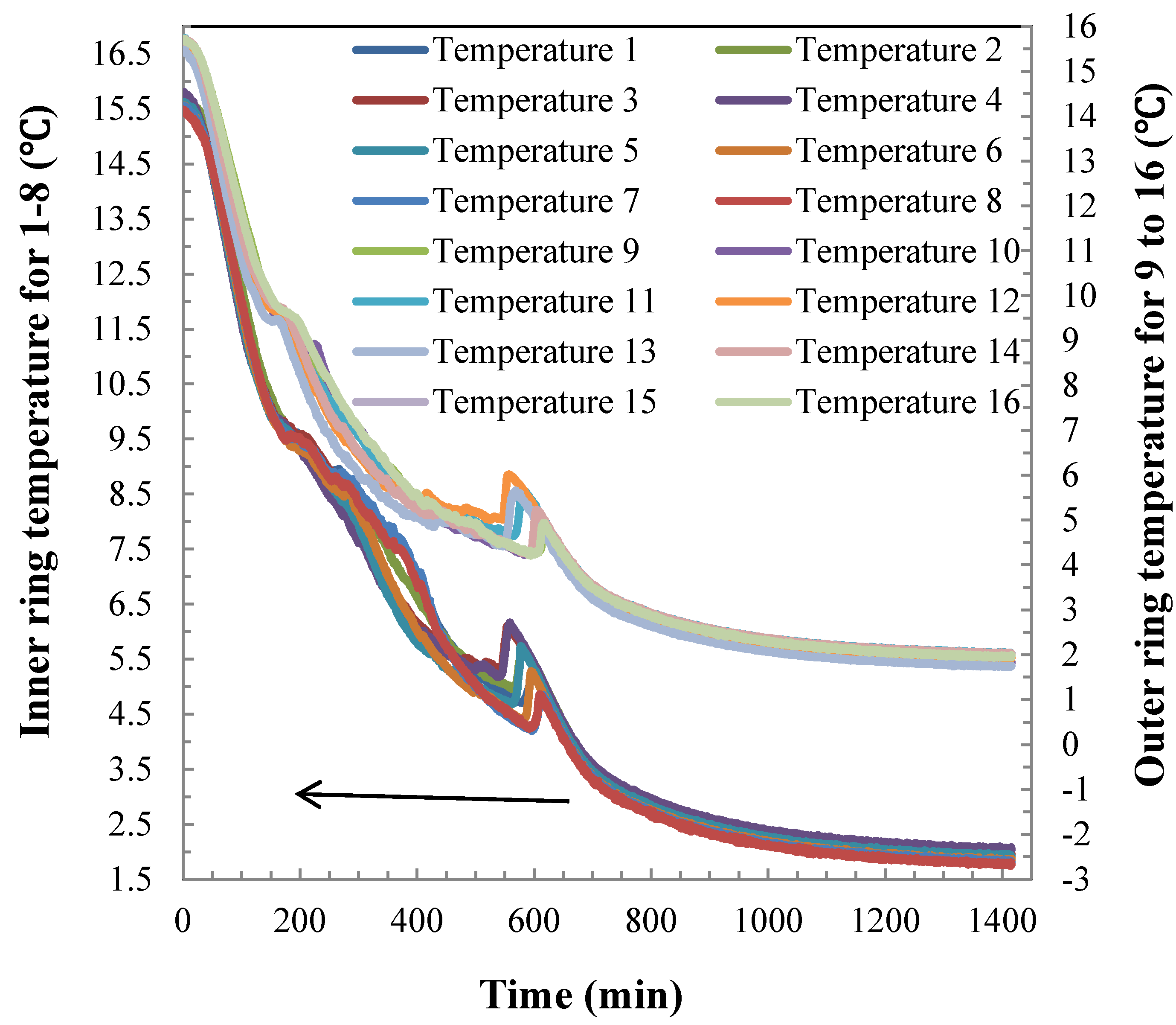
4.2. Hydrate Dissociation Process
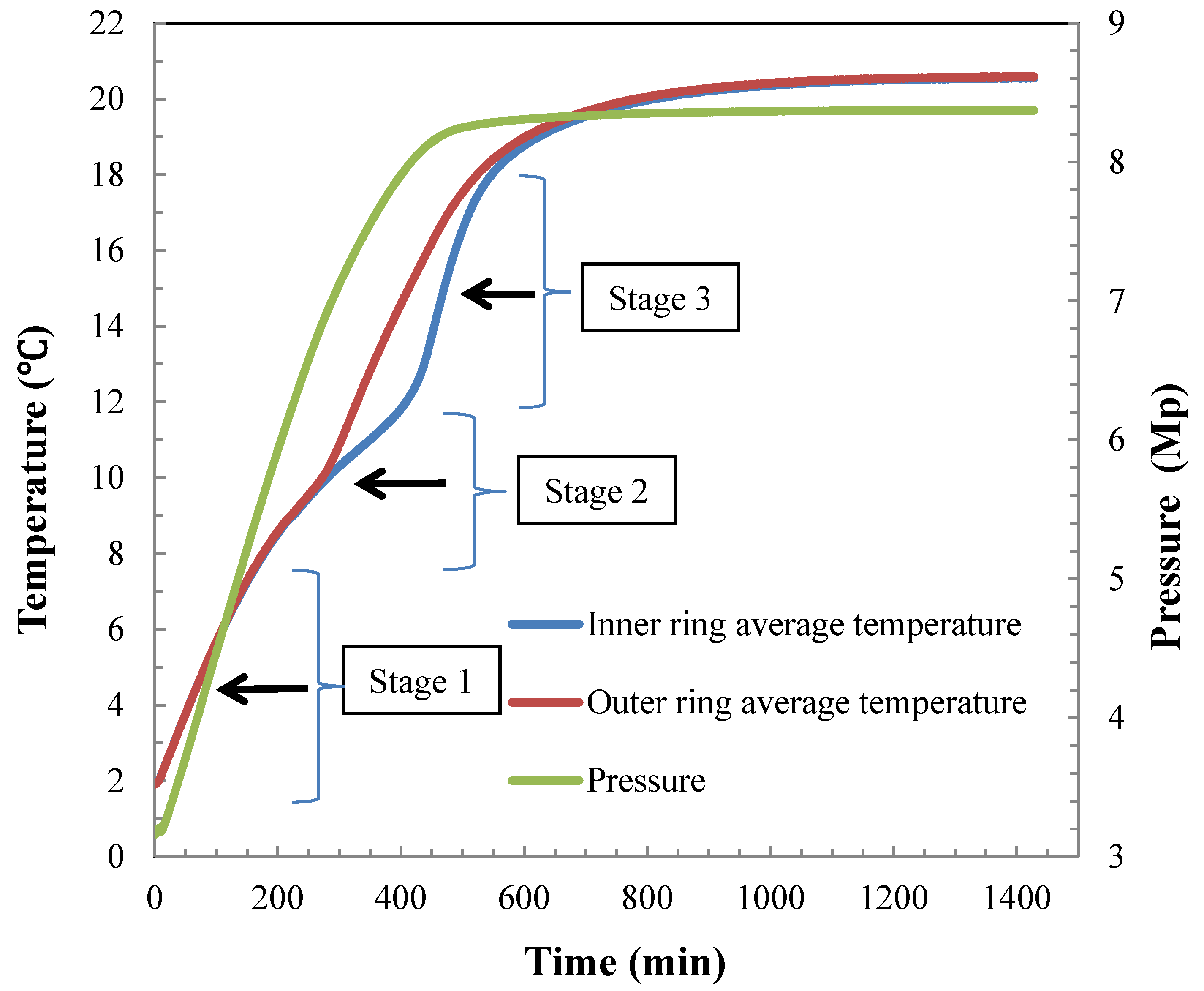
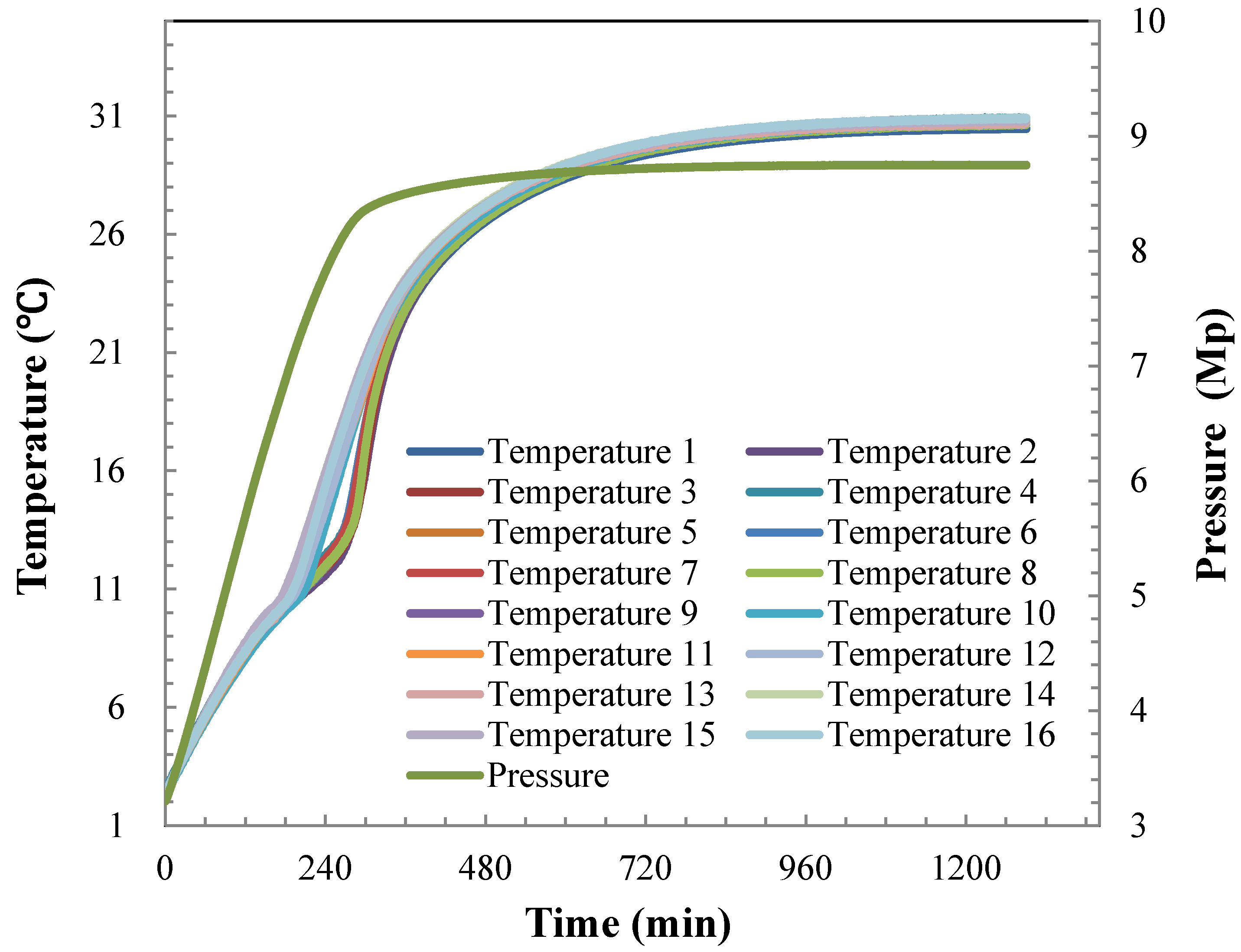
4.3. Heat Transfer Analysis during Hydrate Dissociation
- = Rayleigh number
- = Characteristic length (in this case, the height of hydrate porous media)
- g = acceleration due to gravity
- = Surface temperature (temperature of the wall of reactor)
- = Quiescent temperature (temperature in the dissociated zone)
- ν = Kinematic viscosity of water
- = Effective thermal diffusivity
- = Thermal expansion coefficient
- = Effective thermal conductivity
- = Thermal conductivity of sand
- = Thermal conductivity of water
- = Porosity of glass beads
- = Density (kg/m³)
- = Specific heat capacity (J/(kg·K))
4.4. Temperature Difference during the Hydrate Dissociation
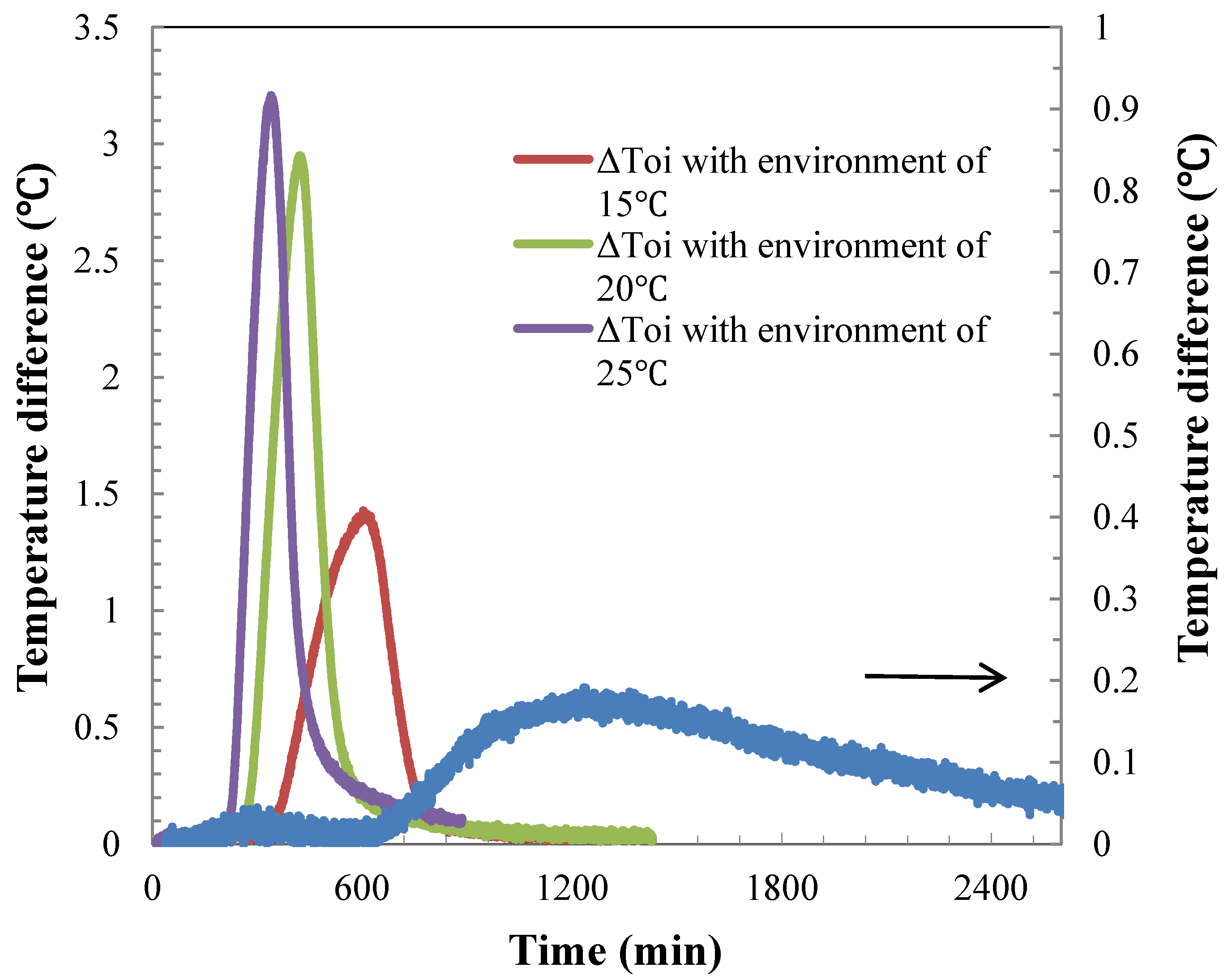
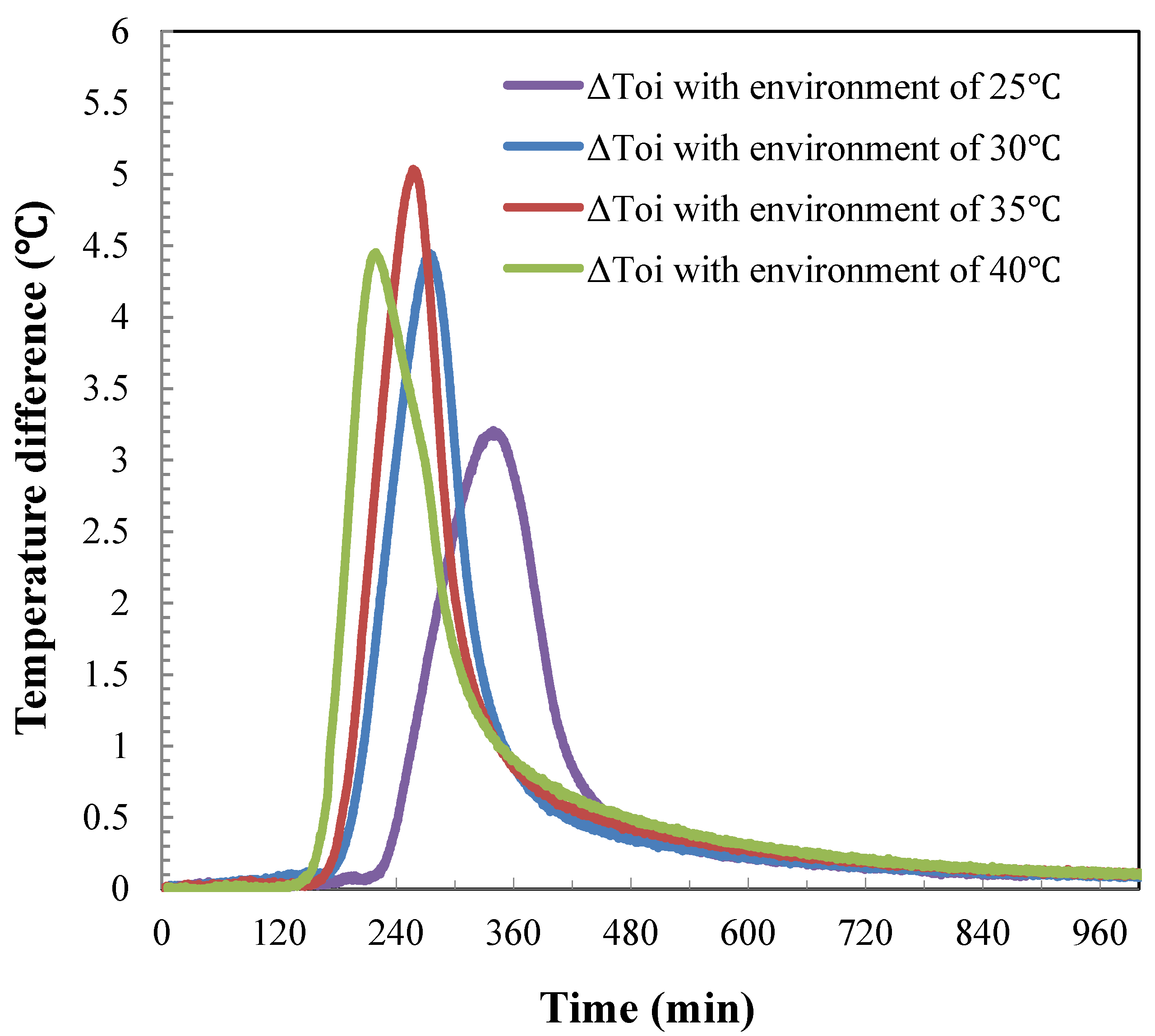
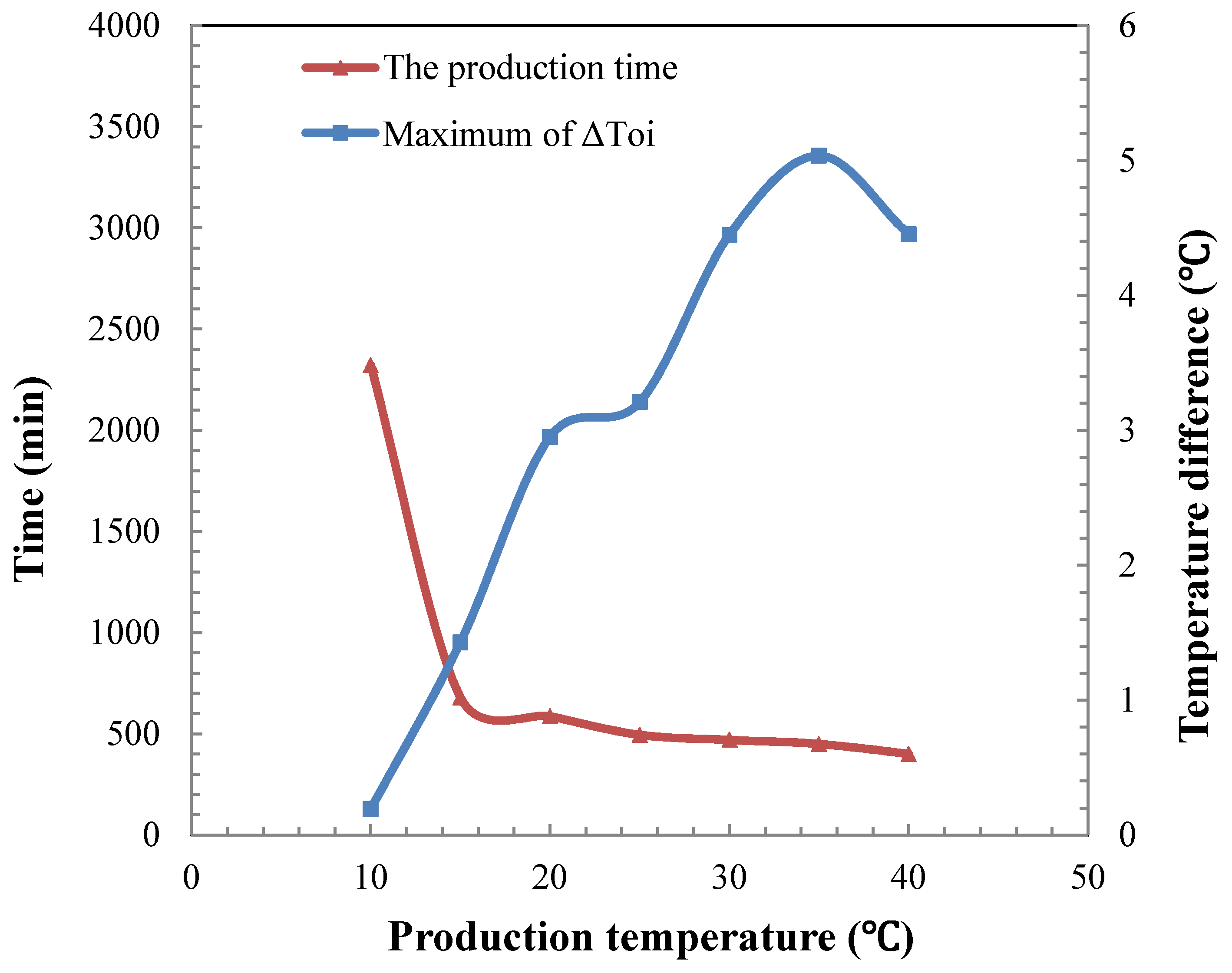
5. Conclusions
Acknowledgments
References
- Kerkar, P.; Jones, K.W.; Kleinberg, R.; Lindquist, W.B.; Tomov, S.; Feng, H.; Mahajan, D. Direct observations of three dimensional growth of hydrates hosted in porous media. Appl. Phys. Lett. 2009, 95, 024102:1–024102:3. [Google Scholar]
- Sloan, E.D. Clathrate Hydrates of Natural Gas, 2nd ed.; Marcel Dekker Inc: New York, NY, USA, 1998. [Google Scholar]
- Sloan, E.D. Fundamental principles and applications of natural gas hydrates. Nature 2003, 426, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Yao, L.; Song, Y.; Xue, K.; Cheng, C.; Liu, Y.; Zhang, Y. In situ observations by magnetic resonance imaging for formation and dissociation of tetrahydrofuran hydrate in porous media. Magn. Reson. Imaging 2011, 29, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.J.; Sun, C.Y.; Ma, Q.L. Science and Technology of Gas Hydrate; (in Chinese). Chemical Industry Press: Beijing, China, 2007; pp. 3–10. [Google Scholar]
- Gabitto, J.F.; Tsouris, C. Physical properties of gas hydrates: A review. J. Thermodyn. 2010, 2010, 1–12. [Google Scholar] [CrossRef]
- Selim, M.S.; Sloan, E.D. Heat and mass transfer during the dissociation of hydrates in porous media. AIChE J. 1989, 35, 1049–1052. [Google Scholar] [CrossRef]
- Fan, S.S. Storage and Transport Technology of Natural Gas Hydrate; (in Chinese). Chemical Industry Press: Beijing, China, 2005; pp. 2–10. [Google Scholar]
- Hong, H.; Pooladi-Darvish, M.; Bishnoi, P.R. Analytical modeling of gas production from hydrates in porous media. J. Canadian Petrol. Technol. 2003, 42, 45–56. [Google Scholar] [CrossRef]
- Pooladi-Darvish, M.; Hong, H. Effect of Conductive and Convective Heat Flow on Gas Production from Natural Hydrates by Depressurization, Advances in the Study of Gas Hydrates; Taylor, C., Kwan, J., Eds.; Springer: New York, NY, USA, 2004; pp. 43–65. [Google Scholar]
- Kamath, V.A.; Holder, G.D.; Angert, P.F. Three phase interfacial heat transfer during the dissociation of propane hydrates. Chem. Eng. Sci. 1984, 39, 1435–1442. [Google Scholar] [CrossRef]
- Selim, M.S.; Sloan, E.D. Modeling of the Dissociation of an In situ Hydrate. In SPE California Regional Meeting, Bakersfield, California, CA, USA, 27–29 March 1985; p. 13597.
- Ullerich, J.W.; Selim, M.S.; Sloan, E.D. Theory and measurement of hydrate dissociation. AIChE J. 1987, 33, 747–752. [Google Scholar] [CrossRef]
- Iida, M.; Mori, H.; Mochizuki, T.; Mori, Y.H. Formation and dissociation of clathrate hydrate in stoichiometric tetrahydrofuran—water mixture subjected to one-dimensional cooling or heating. Chem. Eng. Sci. 2001, 56, 4747–4758. [Google Scholar] [CrossRef]
- Katsuki, D.; Ohmura, R.; Ebinuma, T.; Narita, H. Formation, growth and ageing of clathrate hydrate crystals in a porous medium. Phil. Mag. 2006, 86, 1753–1761. [Google Scholar] [CrossRef]
- Katsuki, D.; Ohmura, R.; Ebinuma, T.; Narita, H. Methane hydrate crystal growth in a porous medium filled with methane-saturated liquid water. Phil. Mag. 2007, 87, 1057–1069. [Google Scholar] [CrossRef]
- Katsuki, D.; Ohmura, R.; Ebinuma, T.; Narita, H. Visual observation of dissociation of methane hydrate crystals in a glass micro model: Production and transfer of methane. J. Appl. Phys. 2008, 104, 083514:1–083514:9. [Google Scholar] [CrossRef]
- Yoon, Y.S.; Lee, S.H.; Seong, K. Convective heat transfer coefficient at hydrate pellet surface during regasification. In Proceedings of the 7th International Conference on Gas Hydrates, Edinburgh, Scotland, UK, 17–21 July 2011.
- Tanaka, S. Heat Transfer Analysis of Re-gasification of Packed Bed of Hydrates. In Proceedings of the 7th International Conference on Gas Hydrates, Edinburgh, UK, 17–21 July 2011.
- Liu, B.; Pang, W.; Peng, B.Z.; Sun, C.Y.; Chen, G.J. Heat transfer related to gas hydrate formation/dissociation. In Developments in Heat Transfer; InTech: Rijeka, Croatia, 2010; pp. 477–502. [Google Scholar]
- Pang, W.X.; Xu, W.Y.; Sun, C.Y.; Zhang, C.L.; Chen, G.J. Methane hydrate dissociation experiment in a middle-sized quiescent reactor using thermal method. Fuel 2009, 88, 497–503. [Google Scholar] [CrossRef]
- Zhang, X.; Lu, X.; Li, Q.; Yao, H. Thermally induced evolution of phase transformations in gas hydrate sediment. Sci. China Phys. Mech. Astron. 2010, 53, 1530–1535. [Google Scholar] [CrossRef]
- Kamth, V.A. Study of heat transfer characteristics during dissociation of gas hydrates in porous media. Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, USA, January 1984. [Google Scholar]
- Sloan, E.D. Clathrate Hydrates of Natural Gases; CRC Press: Boca Raton, FL, USA, 2008; pp. 609–615. [Google Scholar]
- Makogon, Y.F. Hydrates of Natural Gas; Penn Well Publishing Company: Tulsa, OK, USA, 1987. [Google Scholar]
- Su, K.; Sun, C.; Yang, X.; Chen, G.; Fan, S. Experimental investigation of methane hydrate decomposition by depressurizing in porous media with 3-Dimension device. J. Nat. Gas Chem. 2010, 19, 210–216. [Google Scholar] [CrossRef]
- Makogon, Y.F. Hydrates of Hydrocarbons; PennWell Publishing Company: Tulsa, OK, USA, 1997; pp. 245–248. [Google Scholar]
- Wang, X. Engineering of Heat and Mass Transfer; (in Chinese). Science Press: Beijing, China, 1998; Volume 2, pp. 322–342. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, J.; Cheng, C.; Song, Y.; Liu, W.; Liu, Y.; Xue, K.; Zhu, Z.; Yang, Z.; Wang, D.; Yang, M. Heat Transfer Analysis of Methane Hydrate Sediment Dissociation in a Closed Reactor by a Thermal Method. Energies 2012, 5, 1292-1308. https://doi.org/10.3390/en5051292
Zhao J, Cheng C, Song Y, Liu W, Liu Y, Xue K, Zhu Z, Yang Z, Wang D, Yang M. Heat Transfer Analysis of Methane Hydrate Sediment Dissociation in a Closed Reactor by a Thermal Method. Energies. 2012; 5(5):1292-1308. https://doi.org/10.3390/en5051292
Chicago/Turabian StyleZhao, Jiafei, Chuanxiao Cheng, Yongchen Song, Weiguo Liu, Yu Liu, Kaihua Xue, Zihao Zhu, Zhi Yang, Dayong Wang, and Mingjun Yang. 2012. "Heat Transfer Analysis of Methane Hydrate Sediment Dissociation in a Closed Reactor by a Thermal Method" Energies 5, no. 5: 1292-1308. https://doi.org/10.3390/en5051292
APA StyleZhao, J., Cheng, C., Song, Y., Liu, W., Liu, Y., Xue, K., Zhu, Z., Yang, Z., Wang, D., & Yang, M. (2012). Heat Transfer Analysis of Methane Hydrate Sediment Dissociation in a Closed Reactor by a Thermal Method. Energies, 5(5), 1292-1308. https://doi.org/10.3390/en5051292






