Experimental Study on Methane Hydrate Dissociation by Depressurization in Porous Sediments
Abstract
:1. Introduction
2. Experimental Section
2.1. Experimental Apparatus
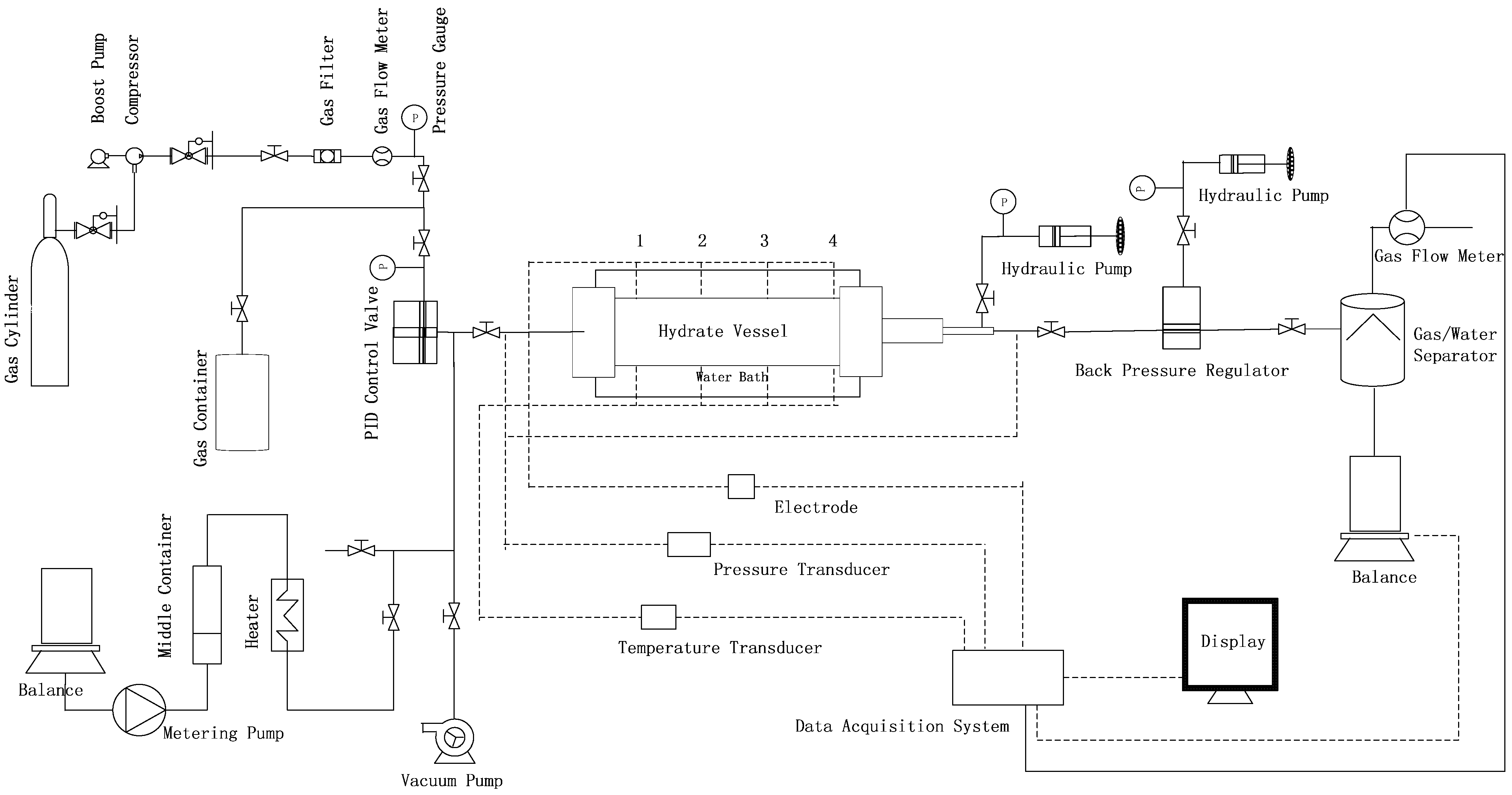
2.2. Preparation of the Methane Hydrate Samples
2.3. Depressurization Dissociation
3. Results and Discussion
| Run 1 | Run 2 | Run 3 | Run 4 | Run 5 | Run 6 | |
|---|---|---|---|---|---|---|
| Initial pressure (MPa) | 12.8 | 13.5 | 15.2 | 13.7 | 13.7 | 13.8 |
| Inner temperature (°C) | 9.1 | 9.1 | 9.0 | 9.0 | 8.9 | 9.0 |
| Equilibrium pressure (MPa) | 6.3 | 6.3 | 6.3 | 6.3 | 6.3 | 6.3 |
| Hydrate saturation (%) | 35.5 | 23.3 | 15.6 | 25.3 | 27.0 | 25.7 |
| Water saturation (%) | 12.3 | 21.8 | 28.2 | 31.5 | 29.2 | 30.5 |
| Production pressure (MPa) | 6.0 | 6.0 | 6.0 | 5.0 | 5.5 | 6.0 |
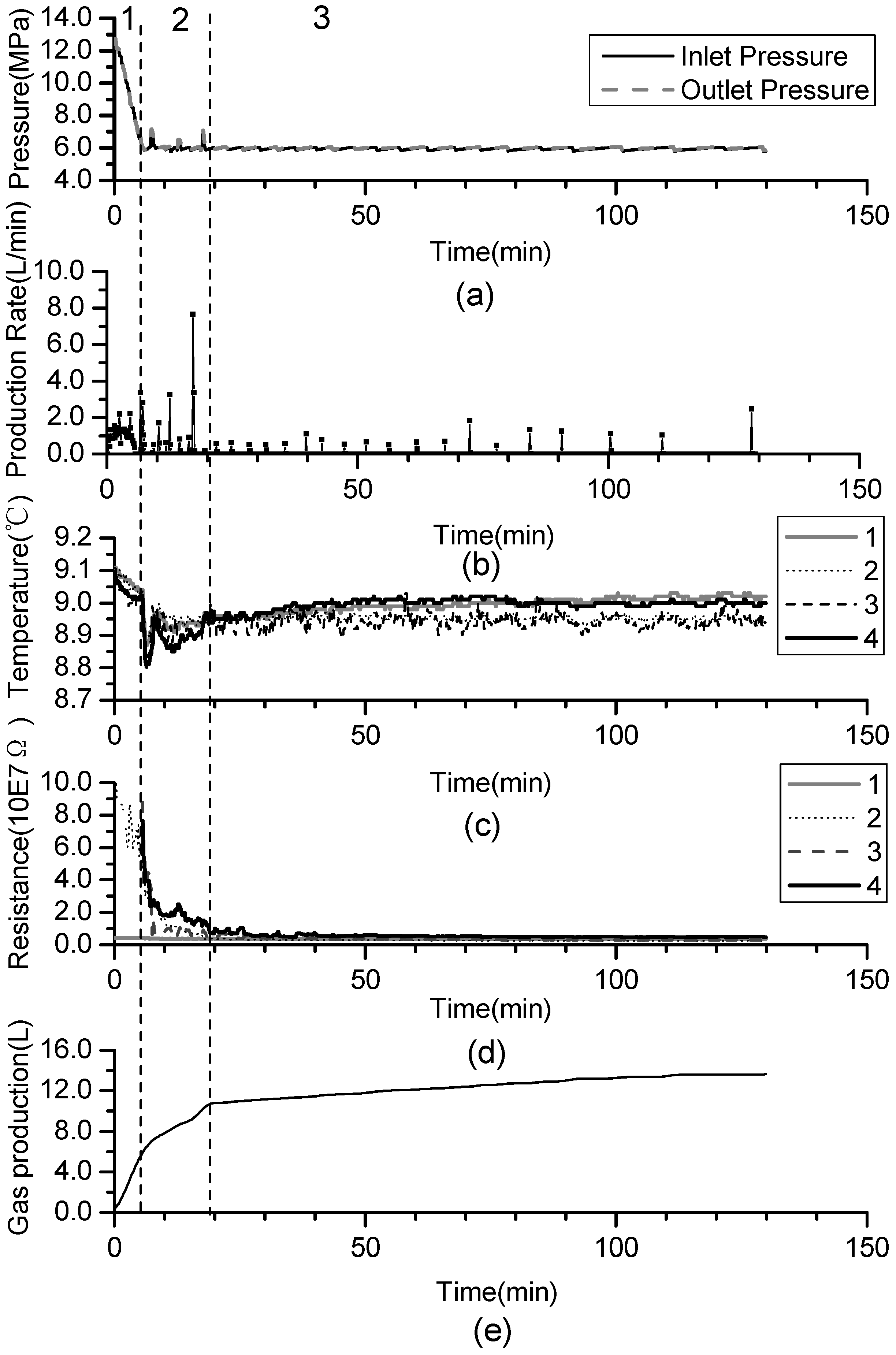
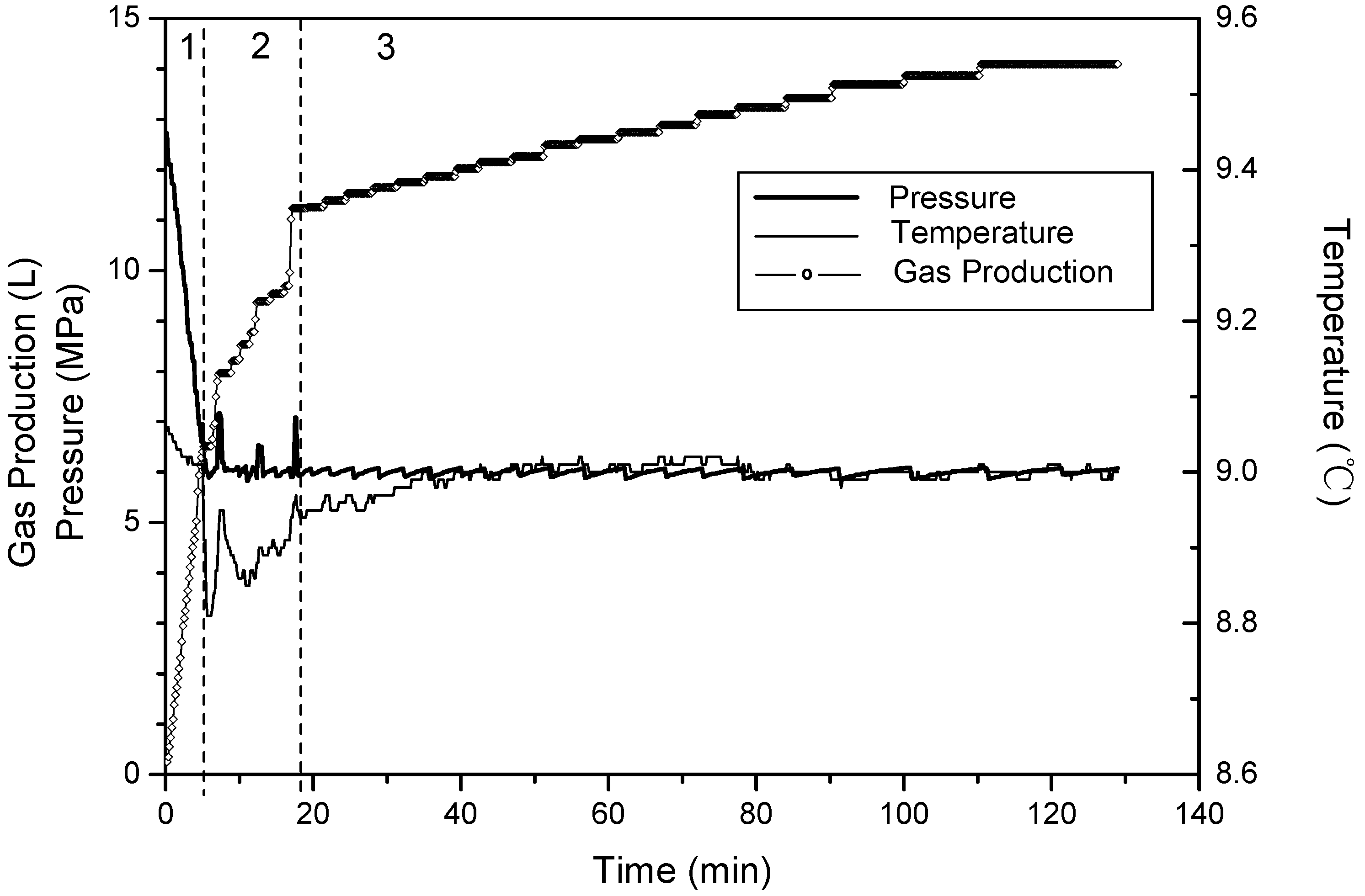
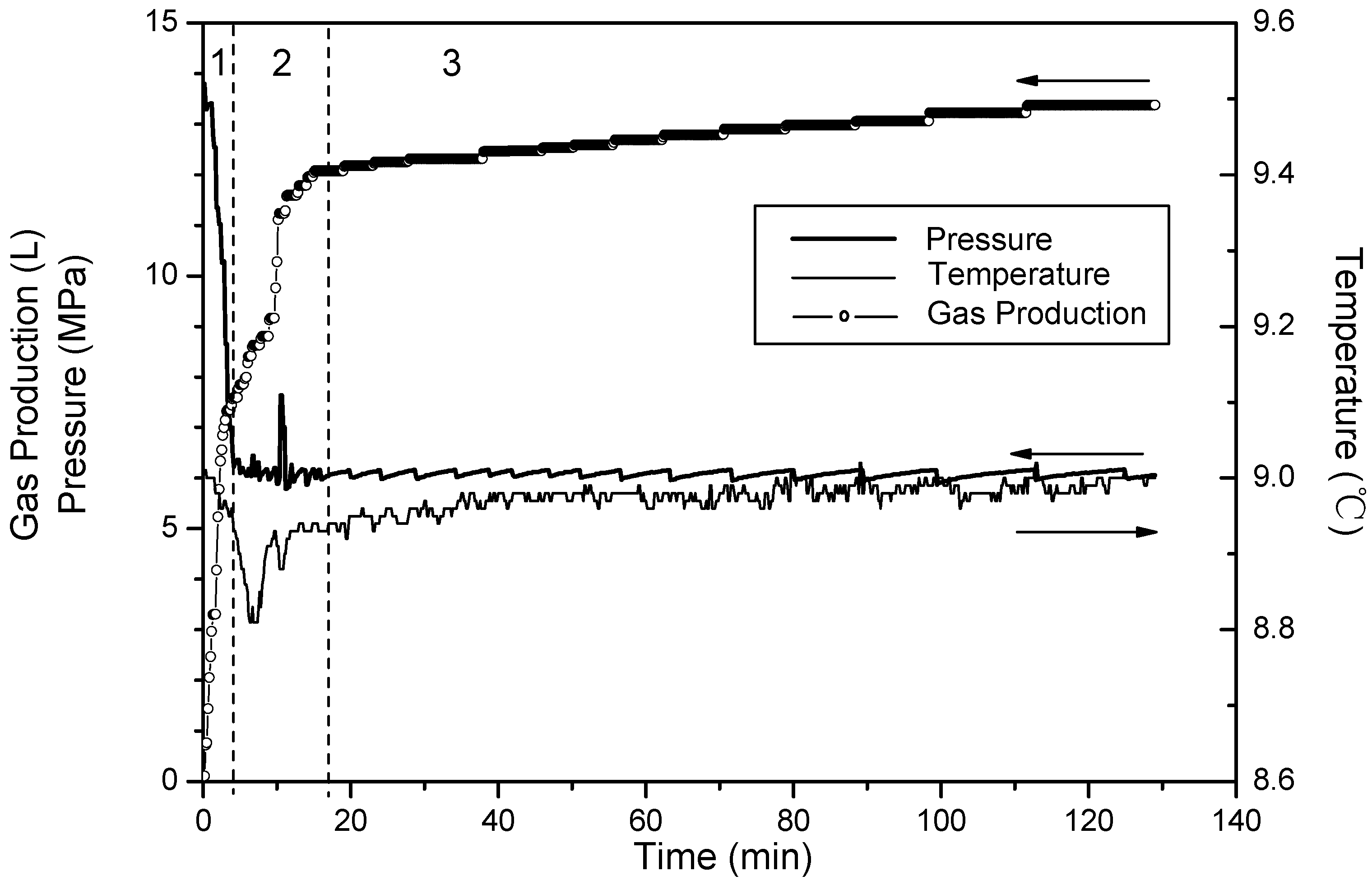
- (1)
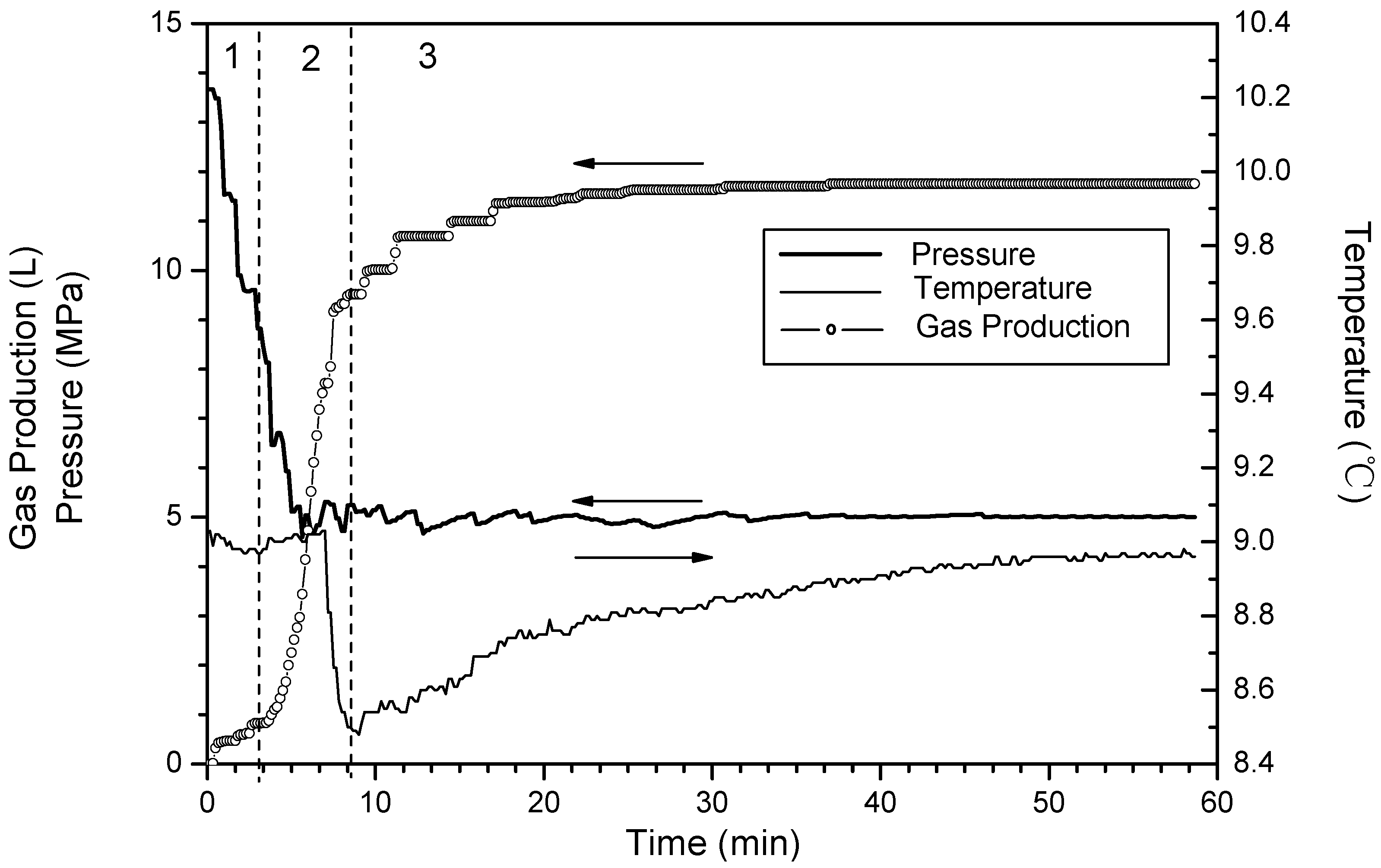
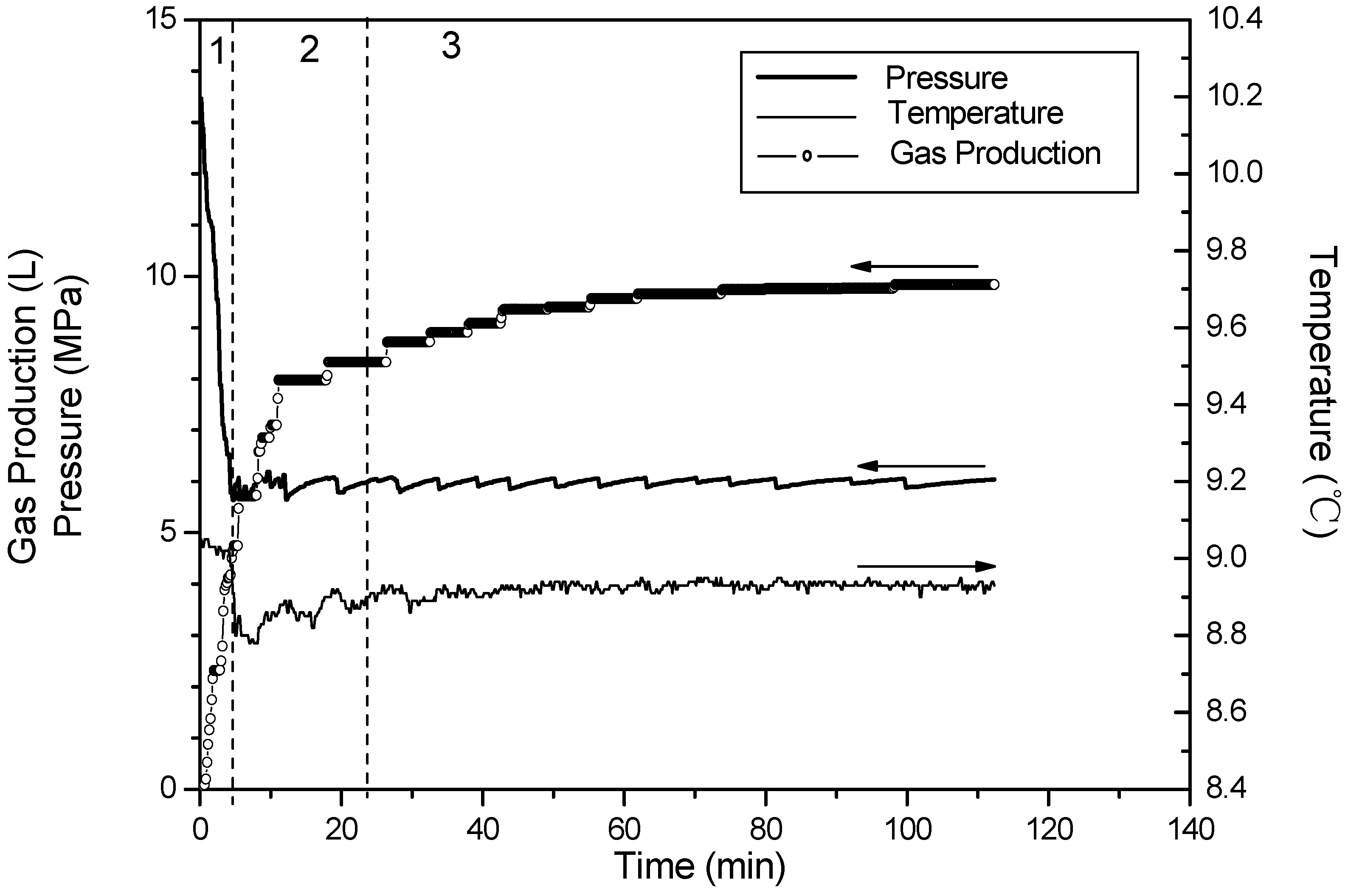
- Phase 1: free gas release process. With depressurization, the free gas is released at first, then temperatures and resistances decline slightly, while the gas production increases sharply. About 40% of the cumulative gas is produced in this section.
- (2)
- Phase 2: rapid hydrate dissociation process. When the pressure drops rapidly to 6 MPa, the gas production rate increases suddenly, while temperatures and resistances simultaneously decrease sharply. The drop of the temperature accompanies with that gas production rate increases suddenly and the curve of the cumulative gas production rises sharply. Then, temperatures increase gradually because of the heat transferred from the water bath. Meanwhile, the resistances drop continuously, and this indicates that the hydrate keeps dissociating. About 40% of cumulative gas is produced in this phase.
- (3)
- Phase 3: slow hydrate dissociation process. Gas production is unconspicuous in this phase. Temperatures and resistances display few changes, and the curve of the cumulative gas production tends to flatten. About 20% of cumulative gas is produced in this section, and the phase lasts for a long time.
| Run 1 | Run 2 | Run 3 | |
|---|---|---|---|
| Gas production in Phase 1 (%) | 40 | 60 | 70 |
| Gas production in Phase 2 (%) | 40 | 30 | 25 |
| Gas production in Phase 3 (%) | 20 | 10 | 5 |


4. Conclusions
- (1)
- The experimental process of hydrate dissociation induced by depressurization can be divided into three phases. The first one is the free gas release phase. In this phase, the gas production increases sharply, temperature and resistance decrease slightly as depressurization occurs. The second one is the rapid hydrate rapid dissociation phase. In this phase temperature and resistance decrease sharply at the beginning, then temperature recovers gradually because of the heat transfer, while resistance goes down gradually, and the production rate is large. The third one is the slow hydrate dissociation phase. In this phase, temperature and resistance experience few changes, but the gas production increases slowly.
- (2)
- The initial hydrate saturation can affect the amount of hydrate dissociation in the different phases. It shows that as the saturation increases, the proportion of hydrate dissociating during the rapid dissociation phase will decrease, thus, it allows more hydrates to dissociate in the third stage with slower rate and the temperature recovers slowly as well.
- (3)
- The dissociation rate increases by lowering the dissociation pressure, and this tendency is more apparent when the dissociation pressure approaches the equilibrium. At a lower dissociation pressure, more heat in the core can be applied for dissociation. Moreover, the dissociation pressure can also affect the temperature-drop during the hydrate dissociation process. Furthermore, the decrease of dissociation pressure may negatively affect the secondary hydrate formation.
Acknowledgments
References
- Sloan, E.D. Clathrate Hydrates of Nature Gas; Marcel Dekker: New York, NY, USA, 1998. [Google Scholar]
- Milkov, A.V. Global estimates of hydrate-bound gas in marine sediments: How much is really out there? Earth Sci. Rev. 2004, 66, 183–197. [Google Scholar] [CrossRef]
- Collett, T.S. Gas hydrates as a future energy resource. Geotimes 2004, 49, 24–27. [Google Scholar]
- Klauda, J.B.; Sandler, S.I. Predictions of gas hydrate phase equilibria and amounts in natural sediment porous media. Mar. Petrol. Geol. 2003, 20, 459–470. [Google Scholar] [CrossRef]
- Moridis, G.J.; Reagan, M.T. Strategies for Gas Production From Oceanic Class 3 Hydrate Accumulations. In Proceedings of the Offshore Technology Conference, Houston, TX, USA, 30 April–3 May 2007.
- Kurihara, M.; Funatsu, K.; Ouchi, H.; Masuda, Y.; Narita, H. Investigation on applicability of methane hydrate production methods to reservoirs with diverse characteristics. In Proceedings of the 5th International Conference on Gas Hydrates, Trondheim, Norway, 13–16 June 2005; pp. 714–725.
- Yousif, M.H.; Abass, H.H.; Selim, M.S.; Sloan, E.D. Experimental and theoretical investigation of methane-gas-hydrate dissociation in porous media. SPE Reserv. Eng. 1991, 6, 69–76. [Google Scholar] [CrossRef]
- Moridis, G.J.; Collett, T.S.; Boswell, R.; Kurihara, M.; Reagan, M.T.; Koh, C.; Sloan, E.D. Toward production from gas hydrates: Current status, assessment of resources, and simulation-based evaluation of technology and potential. SPE Reserv. Eval. Eng. 2009, 12, 745–771. [Google Scholar] [CrossRef]
- Su, K.H.; Sun, C.Y.; Yang, X.; Chen, G.J.; Fan, S.S. Experimental investigation of methane hydrate decomposition by depressurizing in porous media with 3-Dimension device. J. Nat. Gas Chem. 2010, 19, 210–216. [Google Scholar] [CrossRef]
- Yang, X.; Sun, C.Y.; Su, K.H.; Yuan, Q.; Li, Q.P.; Chen, G.J. A three-dimensional study on the formation and dissociation of methane hydrate in porous sediment by depressurization. Energy Convers. Manag. 2012, 56, 1–7. [Google Scholar] [CrossRef]
- Li, G.; Tang, L.; Huang, C.; Feng, Z.; Fan, S. Thermodynamic evaluation of hot brine stimulation for natural gas hydrate dissociation. J. Chem. Ind. Eng. (China) 2006, 57, 2033–2038. [Google Scholar]
- Li, G.; Li, X.S.; Tang, L.G.; Li, Q.P. Control Mechanisms for Methane Hydrate Production by Thermal Stimulation. In Proceedings of the 6th International Conference on Gas Hydrates (ICGH 2008), Vancouver, Canada, 6–10 July 2008.
- Kawamura, T.; Ohtake, M.; Sakamoto, Y.; Yamamoto, Y.; Haneda, H.; Komai, T.; Higuchi, S. Experimental Study on Steam Injection Method Using Methane Hydrate Core Samples. In Proceedings of the Seventh (2007) ISOPE Ocean Mining Symposium, Lisbon, Portugal, 1–6 July 2007; International Society Offshore& Polar Engineers: Lisbon, Portugal, 2007; pp. 83–86. [Google Scholar]
- Pang, W.X.; Xu, W.Y.; Sun, C.Y.; Zhang, C.L.; Chen, G.J. Methane hydrate dissociation experiment in middle-sized quiescent reactor using thermal method. Fuel 2009, 88, 497–503. [Google Scholar] [CrossRef]
- Yang, X.; Sun, C.Y.; Yuan, Q.; Ma, P.C.; Chen, G.J. Experimental study on gas production from methane hydrate-bearing sand by hot-water cyclic injection. Energy Fuel 2010, 24, 5912–5920. [Google Scholar] [CrossRef]
- Kawamura, T.; Yamamoto, Y.; Ohtake, M.; Sakamoto, Y.; Komai, T.; Haneda, H. Experimental Study on Dissociation of Hydrate Core Sample Accelerated by Thermodynamic Inhibitors for Gas Recovery from Natural Gas Hydrate. In Proceedings of the 5th International Conference on Gas Hydrate, Trondheim, Norway, 12–16 June 2005; pp. 3023–3028.
- Li, G.; Li, X.S.; Tang, L.G.; Zhang, Y. Experimental investigation of production behavior of methane hydrate under ethylene glycol injection in unconsolidated sediment. Energy Fuel 2007, 21, 3388–3393. [Google Scholar] [CrossRef]
- Najibi, H.; Chapoy, A.; Haghighi, H.; Tohidi, B. Experimental determination and prediction of methane hydrate stability in alcohols and electrolyte solutions. Fluid Phase Equilibria 2009, 275, 127–131. [Google Scholar] [CrossRef]
- Li, G.; Li, X.S.; Tang, L.; Zhang, Y.; Feng, Z.; Fan, S. experimental investigation of production behavior of methane hydrate under ethylene glycol injection. J. Chem. Ind. Eng. (China) 2007, 58, 2067–2074. [Google Scholar]
- Ota, M.; Morohashi, K.; Abe, Y.; Watanabe, M.; Smith, R.L.; Inomata, H. Replacement of CH4 in the hydrate by use of liquid CO2. Energy Convers. Manag. 2005, 46, 1680–1691. [Google Scholar] [CrossRef]
- Zhou, X.T.; Fan, S.S.; Liang, D.Q.; Du, J.W. Determination of appropriate condition on replacing methane from hydrate with carbon dioxide. Energy Convers. Manag. 2008, 49, 2124–2129. [Google Scholar] [CrossRef]
- Ota, M.; Abe, Y.; Watanabe, M.; Smith, R.L.; Inomata, H. Methane recovery from methane hydrate using pressurized CO2. Fluid Phase Equilibria 2005, 228, 553–559. [Google Scholar] [CrossRef]
- Hamaguchi, R.; Nishimura, Y.; Matsukuma, Y.; Minemoto, M. A Fluid Dynamic Study on Recovery System of Methane Hydrate. In Proceedings of the 5th International Conference on Gas Hydrate, Trondheim, Norway; 2005; p. 1005. [Google Scholar]
- Li, X.S.; Wang, Y.; Li, G.; Zhang, Y.; Chen, Z.Y. experimental investigation into methane hydrate decomposition during three-dimensional thermal huff and puff. Energy Fuel 2011, 25, 1650–1658. [Google Scholar] [CrossRef]
- Yousif, M.H.; Li, P.M.; Selim, M.S.; Sloan, E.D. Depressurization of natural gas hydrates in Berea sandstone cores. J. Inclus. Phenom. Mol. Recog. Chem. 1990, 8, 71–88. [Google Scholar] [CrossRef]
- Oyama, H.; Konno, Y.; Masuda, Y.; Narita, H. Dependence of depressurization-induced dissociation of methane hydrate bearing laboratory cores on heat transfer. Energy Fuel 2009, 23, 4995–5002. [Google Scholar] [CrossRef]
- Ji, C.; Ahmadi, G.; Smith, D.H. Natural gas production from hydrate decomposition by depressurization. Chem. Eng. Sci. 2001, 56, 5801–5814. [Google Scholar] [CrossRef]
- Sun, X.; Nanchary, N.; Mohanty, K.K. 1-D modeling of hydrate depressurization in porous media. Transp. Porous Media 2005, 58, 315–338. [Google Scholar] [CrossRef]
- Kono, H.O.; Narasimhan, S.; Song, F.; Smith, D.H. Synthesis of methane gas hydrate in porous sediments and its dissociation by depressurizing. Powder Technol. 2002, 122, 239–246. [Google Scholar] [CrossRef]
- Lu, J.A.; Yang, S.X.; Wu, N.Y.; Zhang, G.X.; Zhang, M.; Liang, J.Q. Well logging evaluation of gas hydrates in Shenhu area, south China sea. Geoscience 2008, 22, 447–451. [Google Scholar]
- Wu, N.Y.; Zhang, H.G.; Yang, S.X.; Liang, J.Q.; Wang, H.B. Preliminary discussion on natural gas hydrate (NGH) reservoir system of Shenhu area, north slope of south China sea. Nat. Gas Ind. 2007, 27, 1–6. [Google Scholar]
- Li, X.S.; Zhang, Y.; Li, G.; Chen, Z.Y.; Yan, K.F.; Li, Q.P. Gas hydrate equilibrium dissociation conditions in porous media using two thermodynamic approaches. J. Chem. Thermodyn. 2008, 40, 1464–1474. [Google Scholar] [CrossRef]
- Zhou, X.T.; Fan, S.S.; Liang, D.Q.; Wang, D.L.; Huang, N.S. Use of electrical resistance to detect the formation and decomposition of methane hydrate. J. Nat. Gas Chem. 2007, 16, 399–403. [Google Scholar] [CrossRef]
- Kim, H.C.; Bishnoi, P.R.; heidemann, R.A.; Rivzi, S.S.H. kinetics of methane hydrate decomposition. Chem. Eng. Sci. 1987, 42, 1645–1653. [Google Scholar] [CrossRef]
- Kneafsey, T.J.; Tomutsa, L.; Moridis, G.J.; Seol, Y.; Freifeld, B.M.; Taylor, C.E.; Gupta, A. Methane hydrate formation and dissociation in a partially saturated core-scale sand sample. J. Pet. Sci. Eng. 2007, 56, 108–126. [Google Scholar] [CrossRef]
- Stern, L.A.; Kirby, S.H.; Durham, W.B. Peculiarities of methane clathrate hydrate formation and rheology, and the associated superheating of water ice. Science 1996, 273, 1843–1848. [Google Scholar] [CrossRef]
- Li, X.S.; Zhang, Y.; Li, G.; Chen, Z.Y.; Wu, H.J. Experimental investigation into the production behavior of methane hydrate in porous sediment by depressurization with a novel three dimensional cubic hydrate simulator. Energy Fuel 2011, 25, 4497–4505. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xiong, L.; Li, X.; Wang, Y.; Xu, C. Experimental Study on Methane Hydrate Dissociation by Depressurization in Porous Sediments. Energies 2012, 5, 518-530. https://doi.org/10.3390/en5020518
Xiong L, Li X, Wang Y, Xu C. Experimental Study on Methane Hydrate Dissociation by Depressurization in Porous Sediments. Energies. 2012; 5(2):518-530. https://doi.org/10.3390/en5020518
Chicago/Turabian StyleXiong, Lijun, Xiaosen Li, Yi Wang, and Chungang Xu. 2012. "Experimental Study on Methane Hydrate Dissociation by Depressurization in Porous Sediments" Energies 5, no. 2: 518-530. https://doi.org/10.3390/en5020518
APA StyleXiong, L., Li, X., Wang, Y., & Xu, C. (2012). Experimental Study on Methane Hydrate Dissociation by Depressurization in Porous Sediments. Energies, 5(2), 518-530. https://doi.org/10.3390/en5020518






