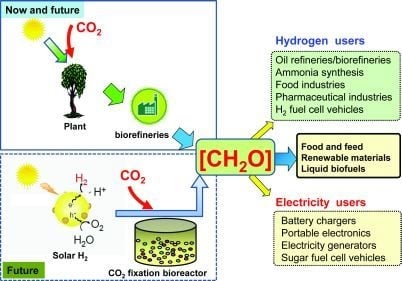
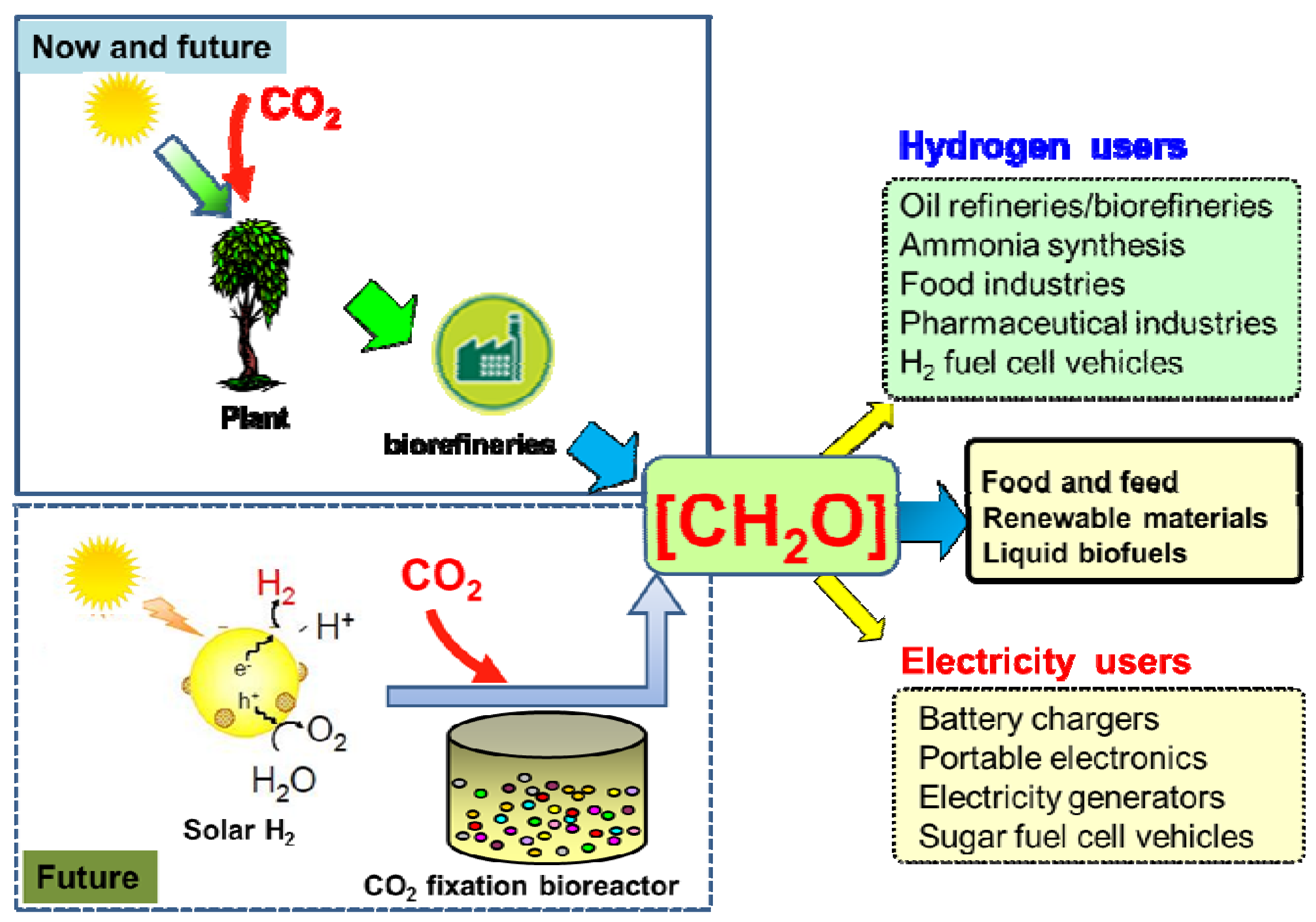
Renewable Hydrogen Carrier — Carbohydrate: Constructing the Carbon-Neutral Carbohydrate Economy
Abstract
:1. Introduction
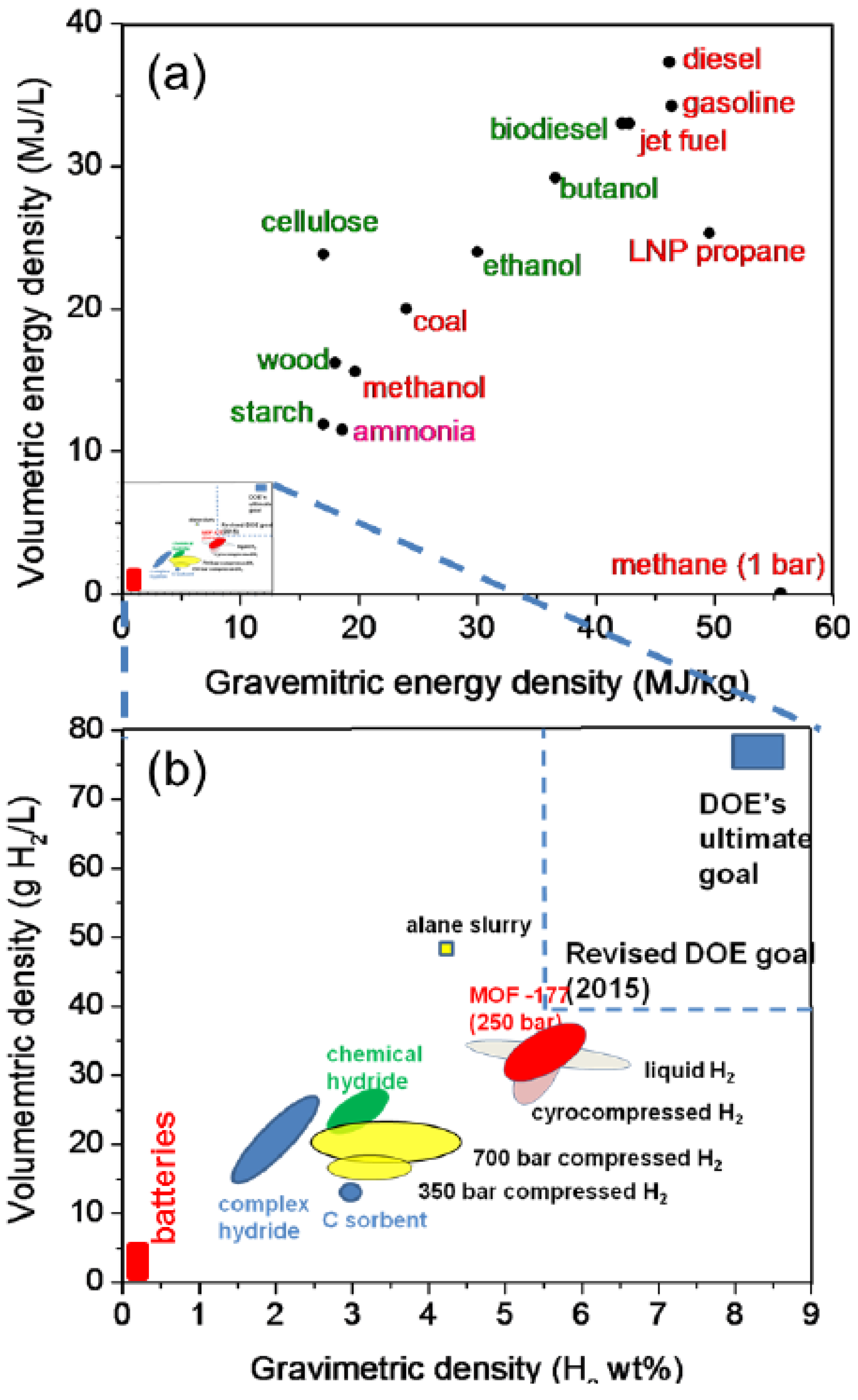
2. Hydrogen Production from Carbohydrate by Cell-Free Enzymatic Technology

3. The Carbon-Neutral Carbohydrate Economy
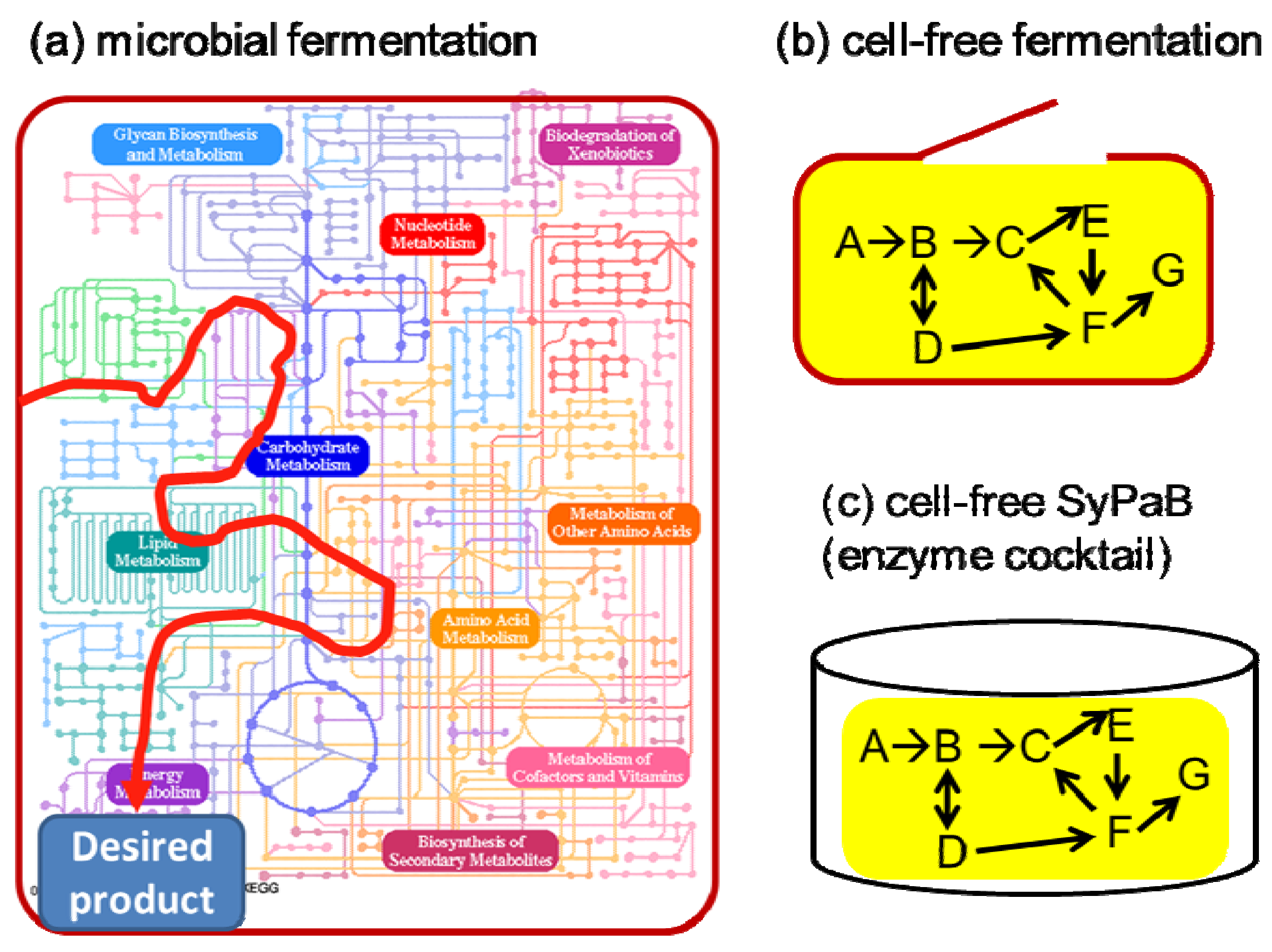
4. SyPaB Challenges and Opportunities
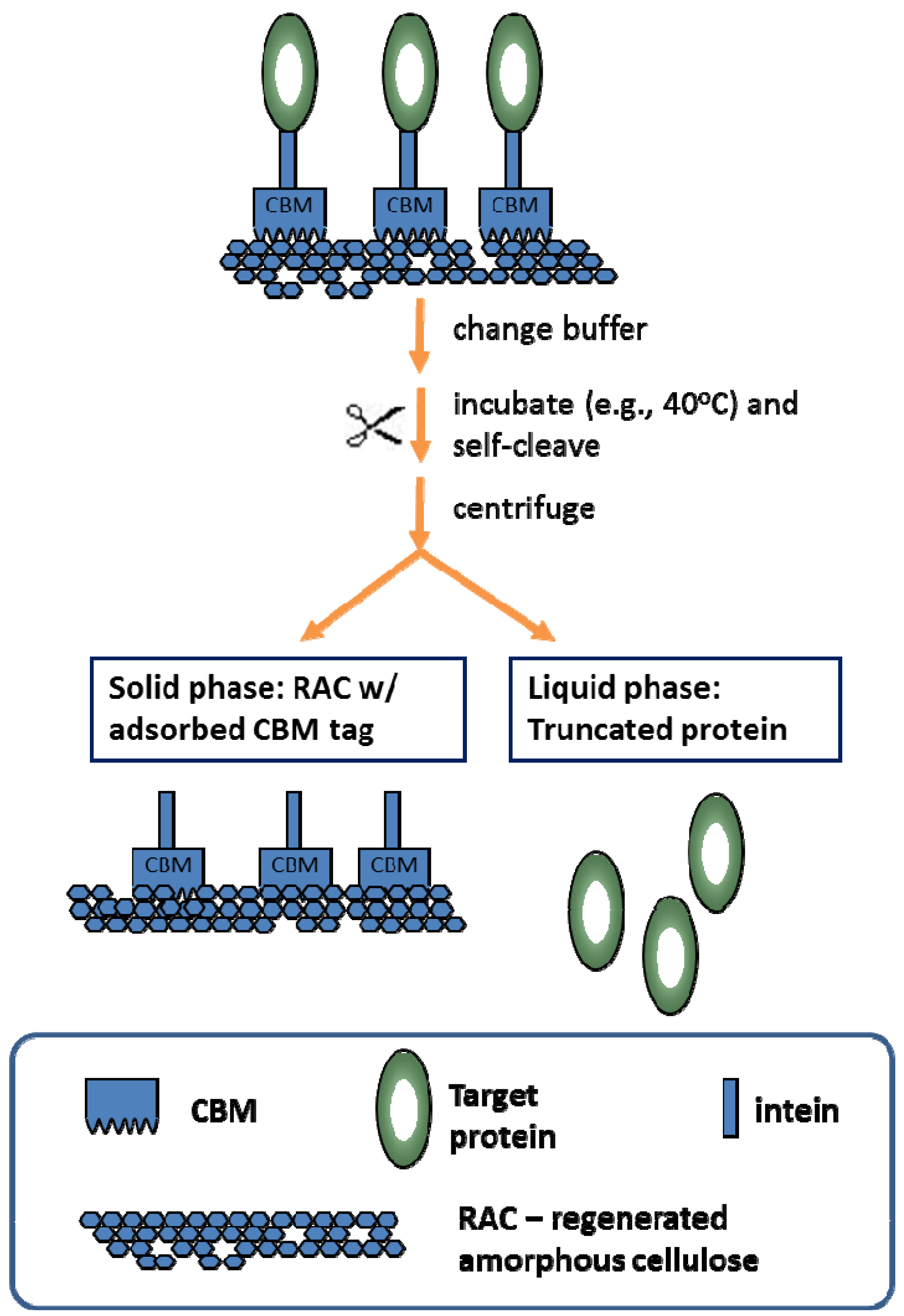
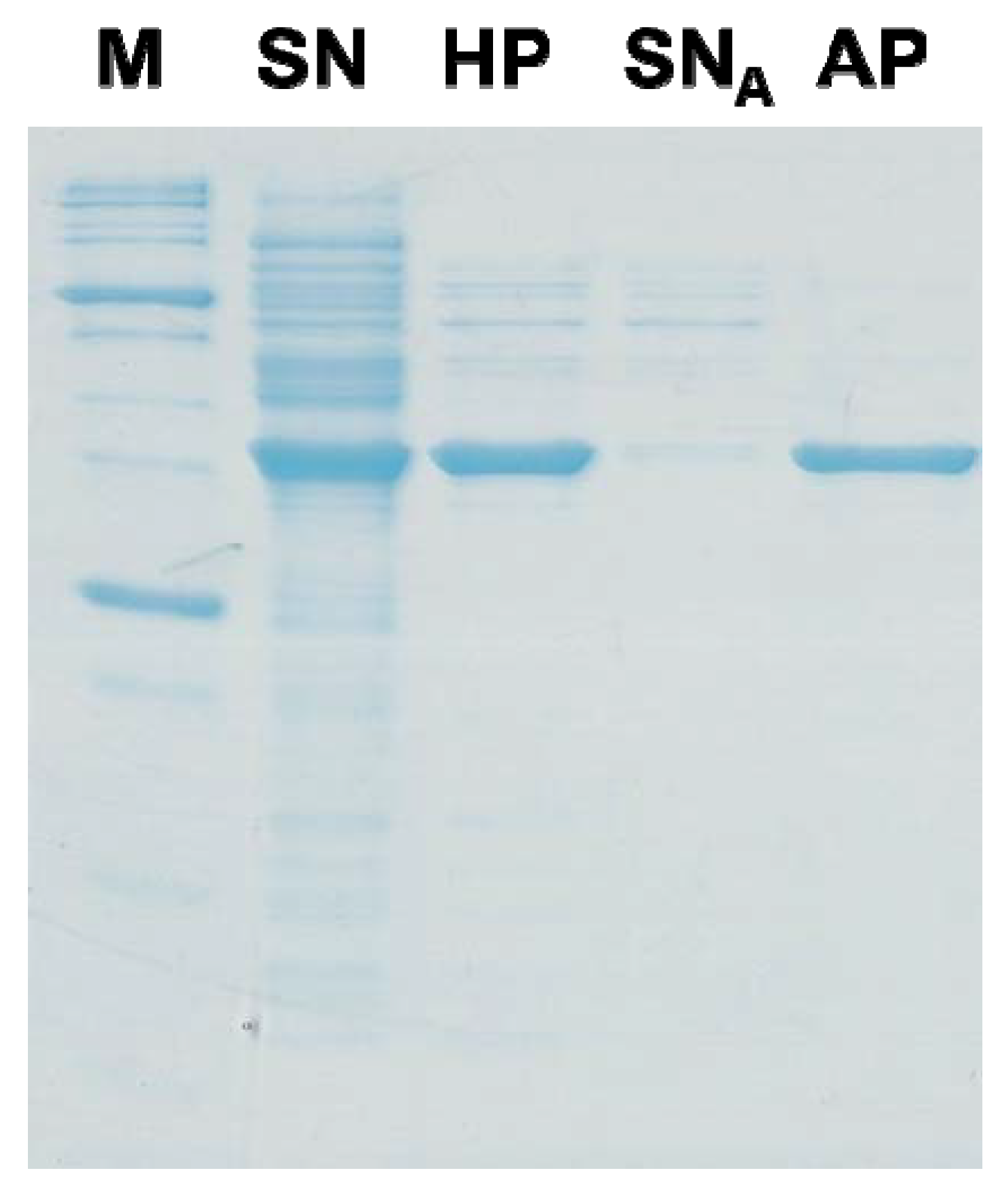
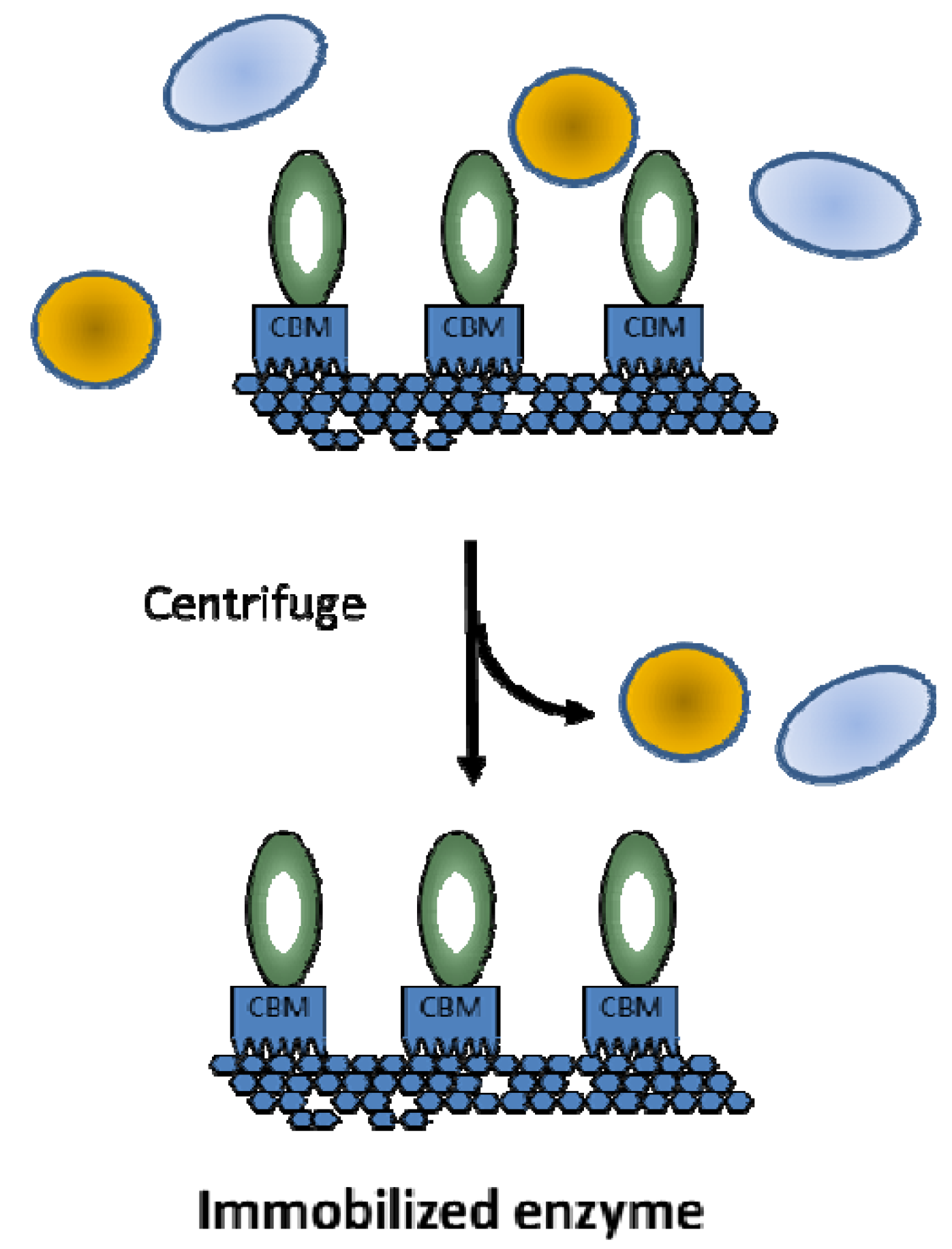

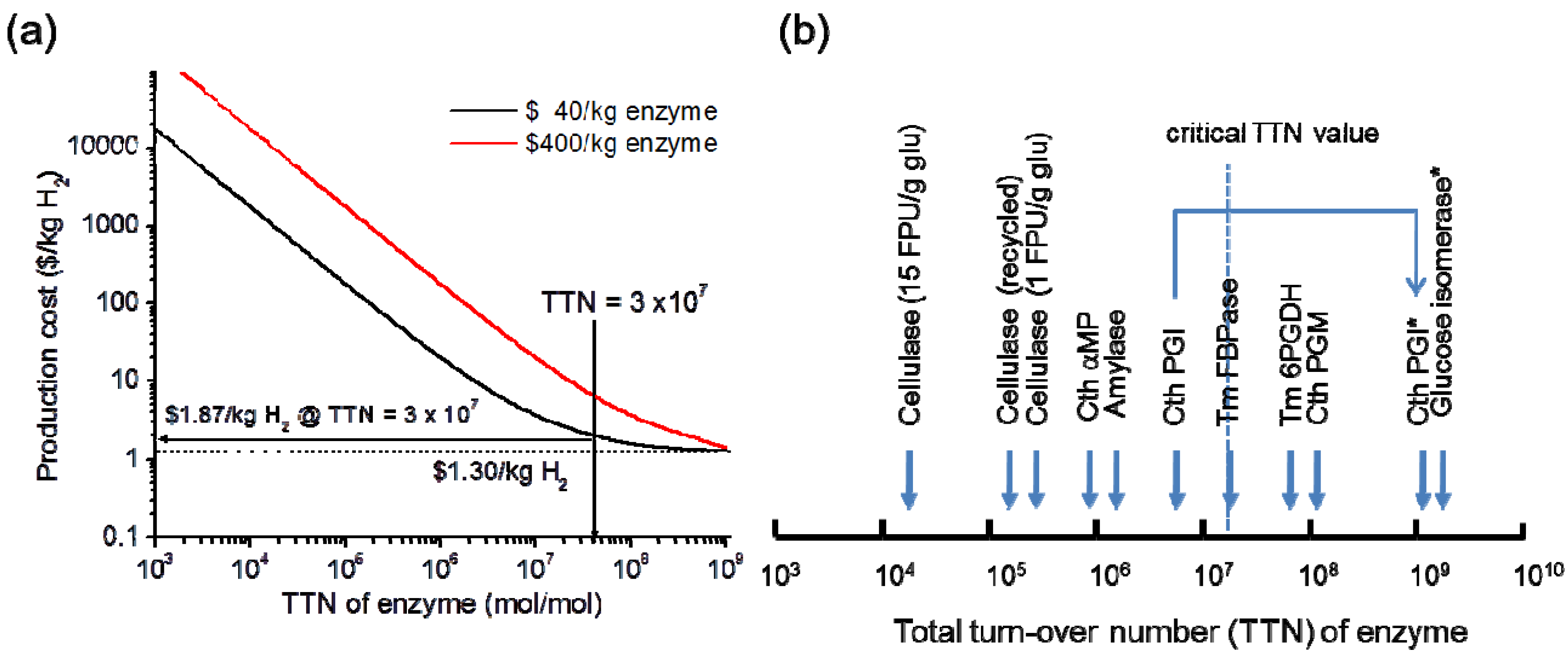
5. Conclusion
Acknowledgments
References
- Bockris, J.O.M. Energy, the Solar Hydrogen Alternative; Halsted Press: New York, NY, USA, 1975. [Google Scholar]
- Zhang, Y.-H.P. A sweet out-of-the-box solution to the hydrogen economy: Is the sugar-powered car science fiction? Energy Environ. Sci. 2009, 2, 272–282. [Google Scholar] [CrossRef]
- Zhang, Y.-H.P. Renewable carbohydrates are a potential high density hydrogen carrier. Int. J. Hydrogen Energy 2010, 35, 10334–10342. [Google Scholar] [CrossRef]
- DOE. Basic Research Needs for the Hydrogen Economy. Available online: http://www.sc.doe.gov/bes/hydrogen.pdf (accessed on 28 January 2011).
- Graetz, J. New approaches to hydrogen storage. Chem. Soc. Rev. 2009, 38, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Orimo, S.i.; Nakamori, Y.; Eliseo, J.R.; Zuttel, A.; Jensen, C.M. Complex hydrides for hydrogen storage. Chem. Rev. 2007, 107, 4111–4132. [Google Scholar] [CrossRef] [PubMed]
- Struzhkin, V.V.; Militzer, B.; Mao, W.L.; Mao, H.k.; Hemley, R.J. Hydrogen storage in molecular clathrates. Chem. Rev. 2007, 107, 4133–4151. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Ma, L.; Fang, Z.; Kang, X.; Wang, P. Improved hydrogen storage property of Li–Mg–B–H system by milling with titanium trifluoride. Energy Environ. Sci. 2009, 2, 120–123. [Google Scholar] [CrossRef]
- Lynd, L.R.; de Brito Cruz, C.H. Make way for ethanol. Science 2010, 330, 1176. [Google Scholar] [CrossRef] [PubMed]
- Dillon, A.C.; Heben, M.J. Hydrogen storage using carbon adsorbents: Past, present and future. Appl. Phys. A Mater. Sci. Process. 2001, 72, 133–142. [Google Scholar] [CrossRef]
- Sakintuna, B.; Lamari-Darkrim, F.; Hirscher, M. Metal hydride materials for solid hydrogen storage: A review. Int. J. Hydrogen Energy 2007, 32, 1121–1140. [Google Scholar] [CrossRef]
- Homepage of H-prize. Available online: http://www.hydrogenprize.org/ (accessed on 28 January 2011).
- Christensen, C.H.; Johannessen, T.; Sørensen, R.Z.; Nørskov, J.K. Towards an ammonia-mediated hydrogen economy? Catalysis Today 2006, 111, 140–144. [Google Scholar] [CrossRef]
- Deluga, G.A.; Salge, J.R.; Schmidt, L.D.; Verykios, X.E. Renewable hydrogen from ethanol by autothermal reforming. Science 2004, 303, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Olah, G.A. Beyond Oil and Gas: The Methanol Economy. Angew. Chem. Int. Ed. 2005, 44, 2636–2639. [Google Scholar] [CrossRef]
- Smil, V. Energy in Nature and Society; MIT Press: Cambridge, MA, USA, 2008. [Google Scholar]
- Moore, R.B.; Raman, V. Hydrogen infrastructure for fuel cell transportation. Int. J. Hydrogen Energy 1998, 23, 617–620. [Google Scholar] [CrossRef]
- Grant, P.M.; Starr, C.; Overbye, T.J. A power grid for the hydrogen economy. Sci. Am. 2006, 295, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Rezaiyan, J.; Cheremisinoff, N.P. Gasification Technologies: A Primer for Engineers and Scientist; CRC press: Boca Raton, FL, USA, 2005; pp. 119–145. [Google Scholar]
- Navarro, R.M.; Pena, M.A.; Fierro, J.L.G. Hydrogen Production Reactions from Carbon Feedstocks: Fossil Fuels and Biomass. Chem. Rev. 2007, 108, 3952–3991. [Google Scholar] [CrossRef]
- Cortright, R.D.; Davda, R.R.; Dumesic, J.A. Hydrogen from catalytic reforming of biomass-derived hydrocarbons in liquid water. Nature 2002, 418, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Thauer, K.; Jungermann, K.; Decker, K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol. Rev. 1977, 41, 100–180. [Google Scholar] [PubMed]
- Adams, M.W.W.; Stiefel, E.I. Biological hydrogen production: Not so elementary. Science 1998, 282, 1842–1843. [Google Scholar] [CrossRef] [PubMed]
- Hallenbeck, P.C.; Benemann, J.R. Biological hydrogen production: Fundamentals and limiting processes. Int. J. Hydrogen Energy 2002, 27, 1185–1193. [Google Scholar] [CrossRef]
- Kleerebezem, R.; van Loosdrecht, M.C.M. Mixed culture biotechnology for bioenergy production. Curr. Opin. Biotechnol. 2007, 18, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.-J.; Jenney, F.E., Jr.; Adams, M.W.W.; Kelly, R.M. Hydrogenesis in hyperthermophilic microorganisms: Implications for biofuels. Metab. Eng. 2008, 10, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Lardon, L.; Helias, A.; Sialve, B.; Stayer, J.P.; Bernard, O. Life-Cycle Assessment Of Biodiesel Production From Microalgae. Environ. Sci. Technol. 2009, 43, 6475–6481. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Regan, J.M. Microbial fuel cells—Challenges and applications. Environ. Sci. Technol. 2006, 40, 5172–5180. [Google Scholar] [CrossRef] [PubMed]
- Haryanto, A.; Fernando, S.; Murali, N.; Adhikari, S. Current status of hydrogen production techniques by steam reforming of ethanol: A review. Energy Fuels 2005, 19, 2098–2106. [Google Scholar] [CrossRef]
- Chheda, J.; Huber, G.; Dumesic, J. Liquid-phase catalytic processing of biomass-derived oxygenated hydrocarbons to fuels and chemicals. Angew. Chem. Int. Ed. 2007, 46, 7164–7183. [Google Scholar] [CrossRef]
- Zhang, Y.-H.P. Production of biocommodities and bioelectricity by cell-free synthetic enzymatic pathway biotransformations: Challenges and opportunities. Biotechnol. Bioeng. 2010, 105, 663–677. [Google Scholar] [PubMed]
- Zhang, Y.-H.P. Using extremophile enzymes to generate hydrogen for electricity. Microbe 2009, 4, 560–565. [Google Scholar]
- Zhang, Y.-H.P.; Sun, J.-B.; Zhong, J.-J. Biofuel production by in vitro synthetic pathway transformation. Curr. Opin. Biotechnol. 2010, 21, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Y.-H.P. Cell-free protein synthesis energized by slowly-metabolized maltodextrin. BMC Biotechnol. 2009, 9, 58. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, W.; Sathitsuksanoh, N.; Zhu, Z.; Zhang, Y.-H.P. Biohydrogenation from biomass sugar mediated by in vitro synthetic enzymatic pathways. Chem. Biol. 2011. accepted. [Google Scholar]
- Huang, W.D.; Zhang, Y.-H.P. Analysis of biofuels production from sugar based on three criteria: Thermodynamics, bioenergetics, and product separation. Energy Environ. Sci. 2011. [Google Scholar] [CrossRef]
- Zehentgruber, D.; Hannemann, F.; Bleif, S.; Bernhardt, R.; Lütz, S. Towards Preparative Scale Steroid Hydroxylation with Cytochrome P450 Monooxygenase CYP106A2. ChemBioChem. 2010, 11, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Cooney, M.J.; Svoboda, V.; Lau, C.; Martin, G.; Minteer, S.D. Enzyme catalysed biofuel cells. Energy Environ. Sci. 2008, 1, 320–337. [Google Scholar] [CrossRef]
- Ye, X.; Wang, Y.; Hopkins, R.C.; Adams, M.W.W.; Evans, B.R.; Mielenz, J.R.; Zhang, Y.-H.P. Spontaneous high-yield production of hydrogen from cellulosic materials and water catalyzed by enzyme cocktails. ChemSusChem. 2009, 2, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.P.; Evans, B.R.; Mielenz, J.R.; Hopkins, R.C.; Adams, M.W.W. High-yield hydrogen production from starch and water by a synthetic enzymatic pathway. PLoS One 2007, 2, e456. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Veziroglu, T.N. Hydrogen production by biological processes: A survey of literature. Int. J. Hydrogen Energy 2001, 26, 13–28. [Google Scholar] [CrossRef]
- Cheng, S.; Logan, B.E. Sustainable and efficient biohydrogen production via electrohydrogenesis. Proc. Nat. Acad. Sci. USA 2007, 104, 18871–18873. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.P.; Lynd, L.R. Toward an aggregated understanding of enzymatic hydrolysis of cellulose: Noncomplexed cellulase systems. Biotechnol. Bioeng. 2004, 88, 797–824. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.P. Reviving the carbohydrate economy via multi-product biorefineries. J. Ind. Microbiol. Biotechnol. 2008, 35, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.-G.; Long, S.P.; Ort, D.R. What is the maximum efficiency with which photosynthesis can convert solar energy into biomass? Curr. Opin. Biotechnol. 2008, 19, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.P. Artificial photosynthesis would unify the electricity-carbohydrate-hydrogen cycle for sustainability. Nature Precedings 2010. [Google Scholar] [CrossRef]
- Hightower, M.; Pierce, S.A. The energy challenge. Nature 2008, 452, 285–286. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Faus, R.; Powers, S.E.; Burken, J.G.; Alvarez, P.J. The Water Footprint of Biofuels: A Drink or Drive Issue? Environm. Sci. Technol. 2009, 43, 3005–3010. [Google Scholar] [CrossRef]
- Berner, R.A. The long-term carbon cycle, fossil fuels and atmospheric composition. Nature 2003, 426, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Falkowski, P.; Scholes, R.J.; Boyle, E.; Canadell, J.; Canfield, D.; Elser, J.; Gruber, N.; Hibbard, K.; Hogberg, P.; Linder, S.; Mackenzie, F.T.; Moore, B., III; Pedersen, T.; Rosenthal, Y.; Seitzinger, S.; Smetacek, V.; Steffen, W. The global carbon cycle: A test of our knowledge of earth as a system. Science 2000, 290, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Hermann, W.A. Quantifying global exergy resources. Energy 2006, 31, 1685–1702. [Google Scholar] [CrossRef]
- Balzani, V.; Credi, A.; Venturi, M. Photochemical Conversion of Solar Energy. ChemSusChem. 2008, 1, 26–58. [Google Scholar] [CrossRef] [PubMed]
- Kalyanasundaram, K.; Graetzel, M. Artificial photosynthesis: Biomimetic approaches to solar energy conversion and storage. Curr. Opin. Biotechnol. 2010, 21, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.S. Toward cost-effective solar energy use. Science 2007, 315, 798–801. [Google Scholar] [CrossRef] [PubMed]
- Gust, D.; Moore, T.A.; Moore, A.L. Solar Fuels via Artificial Photosynthesis. Acc. Chem. Res. 2009, 42, 1890–1898. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.S.; Nocera, D.G. Powering the planet: Chemical challenges in solar energy utilization. Proc. Nat. Acad. Sci. USA 2006, 103, 15729–15735. [Google Scholar] [CrossRef] [PubMed]
- Chin, J.W. Modular approaches to expanding the functions of living matter. Nat. Chem. Biol. 2006, 2, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.K.; Bernhard, S.A. Metabolite transfer via enzyme-enzyme complexes. Science 1986, 234, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Y.; Choi, P.J.; Li, G.-W.; Chen, H.; Babu, M.; Hearn, J.; Emili, A.; Xie, X.S. Quantifying E. coli Proteome and Transcriptome with Single-Molecule Sensitivity in Single Cells. Science 2010, 329, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Conrado, R.J.; Varner, J.D.; DeLisa, M.P. Engineering the spatial organization of metabolic enzymes: Mimicking nature’s synergy. Curr. Opin. Biotechnol. 2008, 19, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Agapakis, C.; Ducat, D.; Boyle, P.; Wintermute, E.; Way, J.; Silver, P. Insulation of a synthetic hydrogen metabolism circuit in bacteria. J. Biol. Eng. 2010, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Vasic-Racki, D. History of industrial biotransformations—Dreams and realities. In Industrial Biotransformations; Liese, A., Seebald, S., Wandrey, C., Eds.; Wiley-VCH: Weinheim, Germany, 2006; pp. 1–37. [Google Scholar]
- Wang, Y.; Zhang, Y.-H.P. A highly active phosphoglucomutase from Clostridium thermocellum: Cloning, purification, characterization, and enhanced thermostability. J. Appl. Microbiol. 2010, 108, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Myung, S.; Zhang, X.-Z.; Zhang, Y.-H.P. Ultra-stable phosphoglucose isomerase through immobilization of cellulose-binding module-tagged thermophilic enzyme on low-cost high-capacity cellulosic adsorbent. Biotechnol. Prog. 2011. submitted. [Google Scholar]
- Wang, Y.; Zhang, Y.-H.P. Overexpression and simple purification of the Thermotoga maritima 6-phosphogluconate dehydrogenase in Escherichia coli and its application for NADPH regeneration. Microb. Cell Fact. 2009, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Myung, S.; Wang, Y.R.; Zhang, Y.-H.P. Fructose-1,6-bisphosphatase from a hyper-thermophilic bacterium Thermotoga maritima: Characterization, metabolite stability and its implications. Process Biochem. 2010, 45, 1882–1887. [Google Scholar] [CrossRef]
- Taylor, L.E.I.; Dai, Z.; Decker, S.R.; Brunecky, R.; Adney, W.S.; Ding, S.-Y.; Himmel, M.E. Heterologous expression of glycosyl hydrolases in planta: A new departure for biofuels. Trends Biotechnol. 2008, 26, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.P.; Himmel, M.; Mielenz, J.R. Outlook for cellulase improvement: Screening and selection strategies. Biotechnol. Adv. 2006, 24, 452–481. [Google Scholar] [CrossRef] [PubMed]
- Sathitsuksanoh, N.; Zhu, Z.; Templeton, N.; Rollin, J.; Harvey, S.; Zhang, Y.-H.P. Saccharification of a potential bioenergy crop, Phragmites australis (common reed), by lignocellulose fractionation followed by enzymatic hydrolysis at decreased cellulase loadings. Ind. Eng. Chem. Res. 2009, 48, 6441–6447. [Google Scholar] [CrossRef]
- Zhang, X.-Z.; Zhang, Y.-H.P. One-step production of biocommodities from lignocellulosic biomass by recombinant cellulolytic Bacillus subtilis: Opportunities and challenges. Eng. Life Sci. 2010, 10, 398–406. [Google Scholar] [CrossRef]
- Shuler, M.; Kargi, F. Bioprocess Engineering: Basic Concepts, 2nd ed.; Prentice-Hall: Englewood Cliffs, NJ, USA, 2001. [Google Scholar]
- Zhang, Y.-H.; Lynd, L.R. Quantification of cell and cellulase mass concentrations during anaerobic cellulose fermentation: Development of an ELISA-based method with application to Clostridium thermocellum batch cultures. Anal. Chem. 2003, 75, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Rollin, J.; Zhang, Y.-H.P. Thermophilic α-glucan phosphorylase from Clostridium thermocellum: Cloning, Characterization and Enhanced thermostability. J. Mol. Cat. B Enzymatic 2010, 65, 110–116. [Google Scholar] [CrossRef]
- Hong, J.; Wang, Y.; Ye, X.; Zhang, Y.-H.P. Simple protein purification through affinity adsorption on regenerated amorphous cellulose followed by intein self-cleavage. J. Chromatogr. A 2008, 1194, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Ye, X.; Wang, Y.; Zhang, Y.-H.P. Bioseparation of recombinant cellulose binding module-protein by affinity adsorption on an ultra-high-capacity cellulosic adsorbent. Anal. Chim. Acta 2008, 621, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.; Wang, J.; Bachas, L. Cloning, expression, and characterization of the gsdA gene encoding thermophilic glucose-6-phosphate dehydrogenase from Aquifex aeolicus. Extremophiles 2002, 6, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Scopes, R.K. Protein Purification: Principles and Practice, 3rd ed.; Springer-Verleg: New York, NY, USA, 1993. [Google Scholar]
- Cao, L.; Langen, L.V.; Sheldon, R.A. Immobilised enzymes: Carrier-bound or carrier-free? Curr. Opin. Biotechnol. 2003, 14, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Palomo, J.M.; van Langen, L.M.; van Rantwijk, F.; Sheldon, R.A. A new, mild cross-linking methodology to prepare cross-linked enzyme aggregates. Biotechnol. Bioeng. 2004, 86, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R. Cross-linked enzyme aggregates (CLEAs): Stable and recyclable biocatalysts. Biochem. Soc. Trans. 2007, 35, 1583–1587. [Google Scholar] [CrossRef] [PubMed]
- Ong, E.; Gilkes, N.R.; Warren, R.A.J.; Miller, R.C.; Kilburn, D.G. Enzyme immobilization using the cellulose-binding domain of a cellulomonas fimi exoglucanase. Nat. Biotechnol. 1989, 7, 604–607. [Google Scholar] [CrossRef]
- Tomme, P.; Boraston, A.B.; McLean, B.; Kormos, J.M.; Creagh, A.L.; Sturch, K.; Gilkes, N.R.; Haynes, C.A.; Warren, R.A.; Kilburn, D.G. Characterization and affinity applications of cellulose-binding domains. J. Chromatogr. B. 1998, 715, 283–296. [Google Scholar] [CrossRef]
- Shoseyov, O.; Shani, Z.; Levy, I. Carbohydrate binding modules: Biochemical properties and novel applications. Microbiol. Mol. Biol. Rev. 2006, 70, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Hsieh, Y.-L. Enzyme immobilization on ultrafine cellulose fibers via poly(acrylic acid) electrolyte grafts. Biotechnol. Bioeng. 2005, 90, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, P. Cofactor regeneration for sustainable enzymatic biosynthesis. Biotechnol. Adv. 2007, 25, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Kazandjian, R.; Klibanov, A. Regioselective Oxidation of Phenols Catalyzed by Polyphenol Oxidase in Chloroform. J. Am. Chem. Soc. 1985, 107, 5448–5450. [Google Scholar] [CrossRef]
- Moore, J.C.; Pollard, D.J.; Kosjek, B.; Devine, P.N. Advances in the enzymatic reduction of ketones. Acc. Chem. Res. 2007, 40, 1412–1419. [Google Scholar] [CrossRef] [PubMed]
- Wildeman, S.M.A.D.; Sonke, T.; Schoemaker, H.E.; May, O. Biocatalytic reductions: From lab curiosity to "first choice". Acc. Chem. Res. 2007, 40, 1260–1266. [Google Scholar] [CrossRef] [PubMed]
- Lutz, J.; Hollmann, F.; Ho, T.V.; Schnyder, A.; Fish, R.H.; Schmid, A. Bioorganometallic chemistry: Biocatalytic oxidation reactions with biomimetic NAD+/NADH co-factors and [Cp*Rh(bpy)H]+ for selective organic synthesis. J. Organomet. Chem. 2004, 689, 4783–4790. [Google Scholar] [CrossRef]
- Ansell, R.J.; Lowe, C.R. Artificial redox coenzymes: Biomimetic analogues of NAD+. Appl. Microbiol. Biotechnol. 1999, 51, 703–710. [Google Scholar] [CrossRef]
- Lo, H.C.; Fish, R.H. Biomimetic NAD+ Models for Tandem Cofactor Regeneration, Horse Liver Alcohol Dehydrogenase Recognition of 1,4-NADH Derivatives, and Chiral Synthesis. Angew. Chem. Int. Ed. 2002, 41, 478–481. [Google Scholar] [CrossRef]
- Ryan, J.D.; Fish, R.H.; Clark, D.S. Engineering Cytochrome P450 Enzymes for Improved Activity towards Biomimetic 1,4-NADH Cofactors. ChemBioChem. 2008, 9, 2579–2582. [Google Scholar] [CrossRef] [PubMed]
- Campbell, E.; Wheeldon, I.R.; Banta, S. Broadening the cofactor specificity of a thermostable alcohol dehydrogenase using rational protein design introduces novel kinetic transient behavior. Biotechnol. Bioeng. 2010, 107, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.W.W.; Kelly, R.M. Finding and using hyperthermophilic enzymes. Trends Biotechnol. 1998, 16, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Auernik, K.S.; Cooper, C.R.; Kelly, R.M. Life in hot acid: Pathway analyses in extremely thermoacidophilic archaea. Curr. Opin. Biotechnol. 2008, 19, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Blumer-Schuette, S.E.; Kataeva, I.; Westpheling, J.; Adams, M.W.W.; Kelly, R.M. Extremely thermophilic microorganisms for biomass conversion: Status and prospects. Curr. Opin. Biotechnol. 2008, 19, 210–217. [Google Scholar] [CrossRef] [PubMed]
- WCPDB—Thermus thermophilus HB8 Whole Cell Project Database. Available online: http://www.srg.harima.riken.jp/ (accessed on 30 January 2011).
- Cava, F.; Hidalgo, A.; Berenguer, J. Thermus thermophilus as biological model. Extremophiles 2009, 13, 213–231. [Google Scholar] [CrossRef] [PubMed]
- Henne, A.; Bruggemann, H.; Raasch, C.; Wiezer, A.; Hartsch, T.; Liesegang, H.; Johann, A.; Lienard, T.; Gohl, O.; Martinez-Arias, R.; Jacobi, C.; Starkuviene, V.; Schlenczeck, S.; Dencker, S.; Huber, R.; Klenk, H.-P.; Kramer, W.; Merkl, R.; Gottschalk, G.; Fritz, H.-J. The genome sequence of the extreme thermophile Thermus thermophilus. Nat. Biotechnol. 2004, 22, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.E.; Clayton, R.A.; Gill, S.R.; Gwinn, M.L.; Dodson, R.J.; Haft, D.H.; Hickey, E.K.; Peterson, J.D.; Nelson, W.C.; Ketchum, K.A.; McDonald, L.; Utterback, T.R.; Malek, J.A.; Linher, K.D.; Garrett, M.M.; Stewart, A.M.; Cotton, M.D.; Pratt, M.S.; Phillips, C.A.; Richardson, D.; Heidelberg, J.; Sutton, G.G.; Fleischmann, R.D.; Eisen, J.A.; White, O.; Salzberg, S.L.; Smith, H.O.; Venter, J.C.; Fraser, C.M. Evidence for lateral gene transfer between Archaea and Bacteria from genome sequence of Thermotoga maritima. Nature 1999, 399, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Kawarabayasi, Y.; Sawada, M.; Horikawa, H.; Haikawa, Y.; Hino, Y.; Yamamoto, S.; Sekine, M.; Baba, S.-I.; Kosugi, H.; Hosoyama, A.; Nagai, Y.; Sakai, M.; Ogura, K.; Otsuka, R.; Nakazawa, H.; Takamiya, M.; Ohfuku, Y.; Funahashi, T.; Tanaka, T.; Kudoh, Y.; Yamazaki, J.; Kushida, N.; Oguchi, A.; Aoki, K.-I.; Yoshizawa, T.; Nakamura, Y.; Robb, F.T.; Horikoshi, K.; Masuchi, Y.; Shizuya, H.; Kikuchi, H. Complete Sequence and Gene Organization of the Genome of a Hyper-thermophilic Archaebacterium, Pyrococcus horikoshii OT3. DNA Res. 1998, 5, 55–76. [Google Scholar] [CrossRef] [PubMed]
- Bult, C.J.; White, O.; Olsen, G.J.; Zhou, L.; Fleischmann, R.D.; Sutton, G.G.; Blake, J.A.; FitzGerald, L.M.; Clayton, R.A.; Gocayne, J.D.; Kerlavage, A.R.; Dougherty, B.A.; Tomb, J.-F.; Adams, M.D.; Reich, C.I.; Overbeek, R.; Kirkness, E.F.; Weinstock, K.G.; Merrick, J.M.; Glodek, A.; Scott, J.L.; Geoghagen, N.S.M.; Weidman, J.F.; Fuhrmann, J.L.; Nguyen, D.; Utterback, T.R.; Kelley, J.M.; Peterson, J.D.; Sadow, P.W.; Hanna, M.C.; Cotton, M.D.; Roberts, K.M.; Hurst, M.A.; Kaine, B.P.; Borodovsky, M.; Klenk, H.-P.; Fraser, C.M.; Smith, H.O.; Woese, C.R.; Venter, J.C. Complete genome sequence of the methanogenic archaeon, Methanococcus jannaschii. Science 1996, 273, 1058–1073. [Google Scholar] [CrossRef] [PubMed]
- Deckert, G.; Warren, P.V.; Gaasterland, T.; Young, W.G.; Lenox, A.L.; Graham, D.E.; Overbeek, R.; Snead, M.A.; Keller, M.; Aujay, M.; Huber, R.; Feldman, R.A.; Short, J.M.; Olsen, G.J.; Swanson, R.V. The complete genome of the hyperthermophilic bacterium Aquifex aeolicus. Nature 1998, 392, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Hong, J.; Bevan, D.R.; Zhang, Y.-H.P. Fast identification of thermostable beta-glucosidase mutants on cellobiose by a novel combinatorial selection/screening approach. Biotechnol. Bioeng. 2009, 103, 1087–1094. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, X.-Z.; Zhang, Z.-M.; Zhang, Y.-H.P. Engineering of Clostridium phytofermentans Endoglucanase Cel5A for Improved Thermostability. Appl. Environ. Microbiol. 2010, 76, 4914–4917. [Google Scholar] [CrossRef] [PubMed]
- Arnold, F.H. Combinatorial and computational challenges for biocatalyst design. Nature 2001, 409, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Z.; Zhang, Y.-H.P. Simple, fast and high-efficiency transformation system for directed evolution of cellulase in Bacillus subtilis. Microb. Biotechnol. 2011, 4, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Hopkins, R.C.; Jenney, F.E.; McTernan, P.M.; Adams, M.W.W. Heterologous Expression and Maturation of an NADP-Dependent [NiFe]-Hydrogenase: A Key Enzyme in Biofuel Production. PLoS One 2010, 5, e10526. [Google Scholar] [CrossRef] [PubMed]
- Lynd, L.R.; Wyman, C.E.; Gerngross, T.U. Biocommodity engineering. Biotechnol. Prog. 1999, 15, 777–793. [Google Scholar] [CrossRef] [PubMed]
- The Royal Society of the UK Synthetic biology: Call for views. Available online: http://royalsociety.org/page.asp?changes=0&latest=1&id=6731 (accessed on 28 January 2011).
- Yang, H.; Xu, Z.; Fan, M.; Gupta, R.; Slimane, R.B.; Bland, A.E.; Wright, I. Progress in carbon dioxide separation and capture: A review. J. Environ. Sci. 2008, 20, 14–27. [Google Scholar] [CrossRef]
- Melamu, R.; von Blottnitz, H. A comparison of environmental benefits of transport and electricity applications of carbohydrate derived ethanol and hydrogen. Int. J. Hydrogen Energy 2009, 34, 1126–1134. [Google Scholar] [CrossRef]
- Thomas, C.E. Fuel cell and battery electric vehicles compared. Int. J. Hydrogen Energy 2009, 34, 6005–6020. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Y.-H.P.; Mielenz, J.R. Renewable Hydrogen Carrier — Carbohydrate: Constructing the Carbon-Neutral Carbohydrate Economy. Energies 2011, 4, 254-275. https://doi.org/10.3390/en4020254
Zhang Y-HP, Mielenz JR. Renewable Hydrogen Carrier — Carbohydrate: Constructing the Carbon-Neutral Carbohydrate Economy. Energies. 2011; 4(2):254-275. https://doi.org/10.3390/en4020254
Chicago/Turabian StyleZhang, Y.-H. Percival, and Jonathan R. Mielenz. 2011. "Renewable Hydrogen Carrier — Carbohydrate: Constructing the Carbon-Neutral Carbohydrate Economy" Energies 4, no. 2: 254-275. https://doi.org/10.3390/en4020254
APA StyleZhang, Y.-H. P., & Mielenz, J. R. (2011). Renewable Hydrogen Carrier — Carbohydrate: Constructing the Carbon-Neutral Carbohydrate Economy. Energies, 4(2), 254-275. https://doi.org/10.3390/en4020254