A Study on the Hydrotreating of Coal Hydroliquefaction Residue and its Kinetics
Abstract
:1. Introduction
2. Result and Discussion
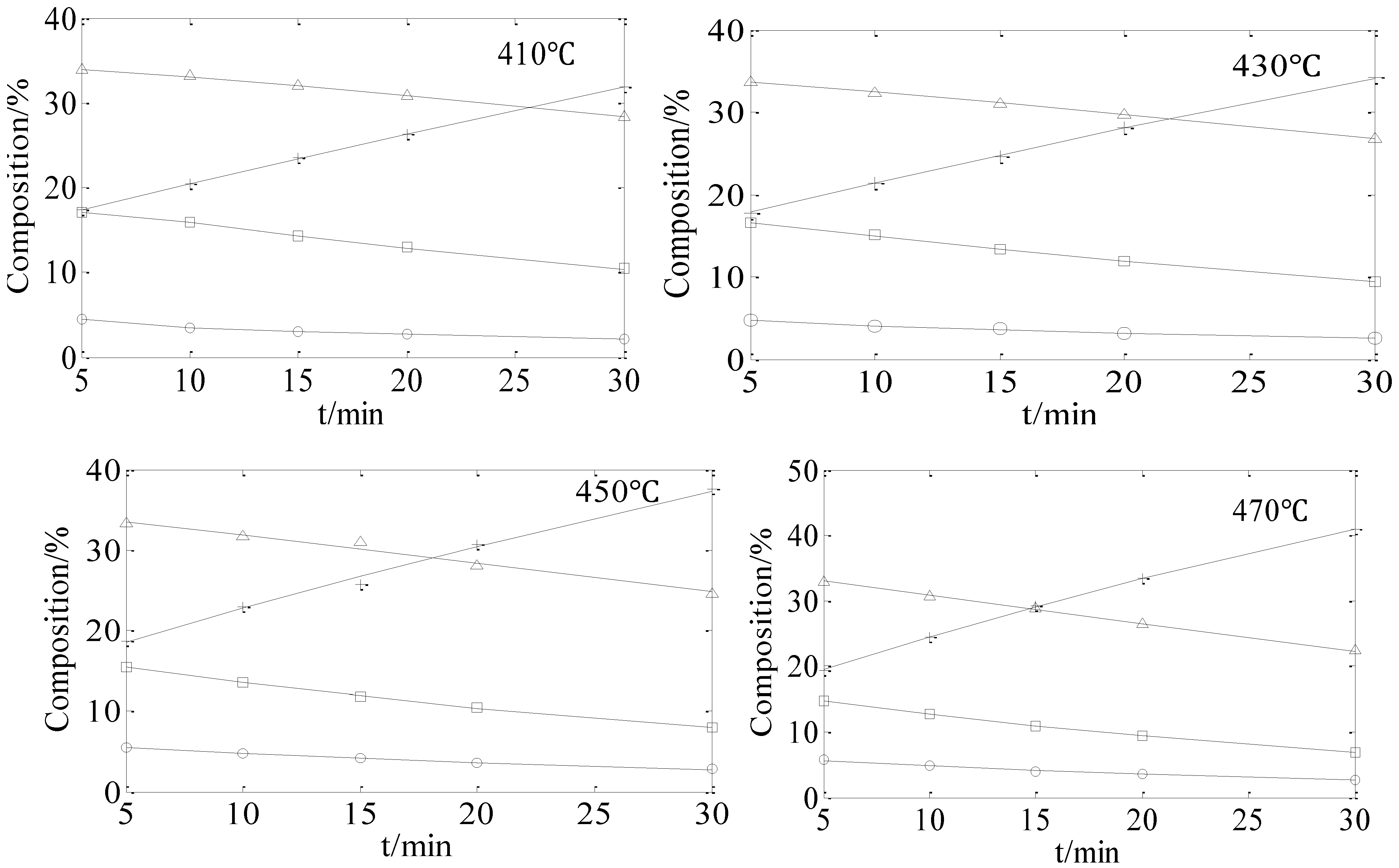
2.1. Hydro-Conversion of CHLR
| Proximate analysis/wt% | Ultimate analysis(daf)/wt% | |||||
|---|---|---|---|---|---|---|
| Mad | Ad | Vdaf | C | H | S | N+O |
| 0 | 15.89 | 45.02 | 91.44 | 5.47 | 2.4 | 0.69 |
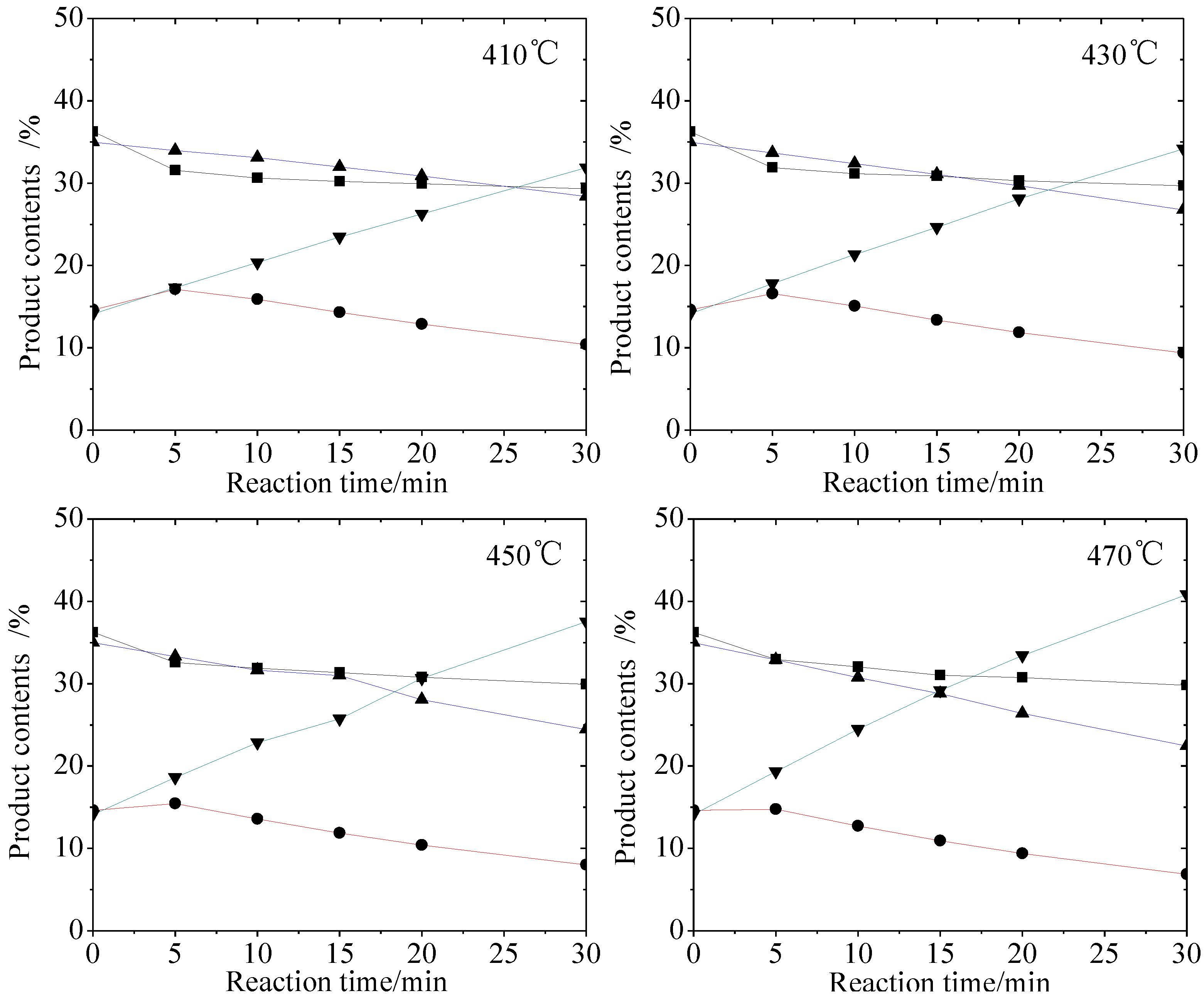
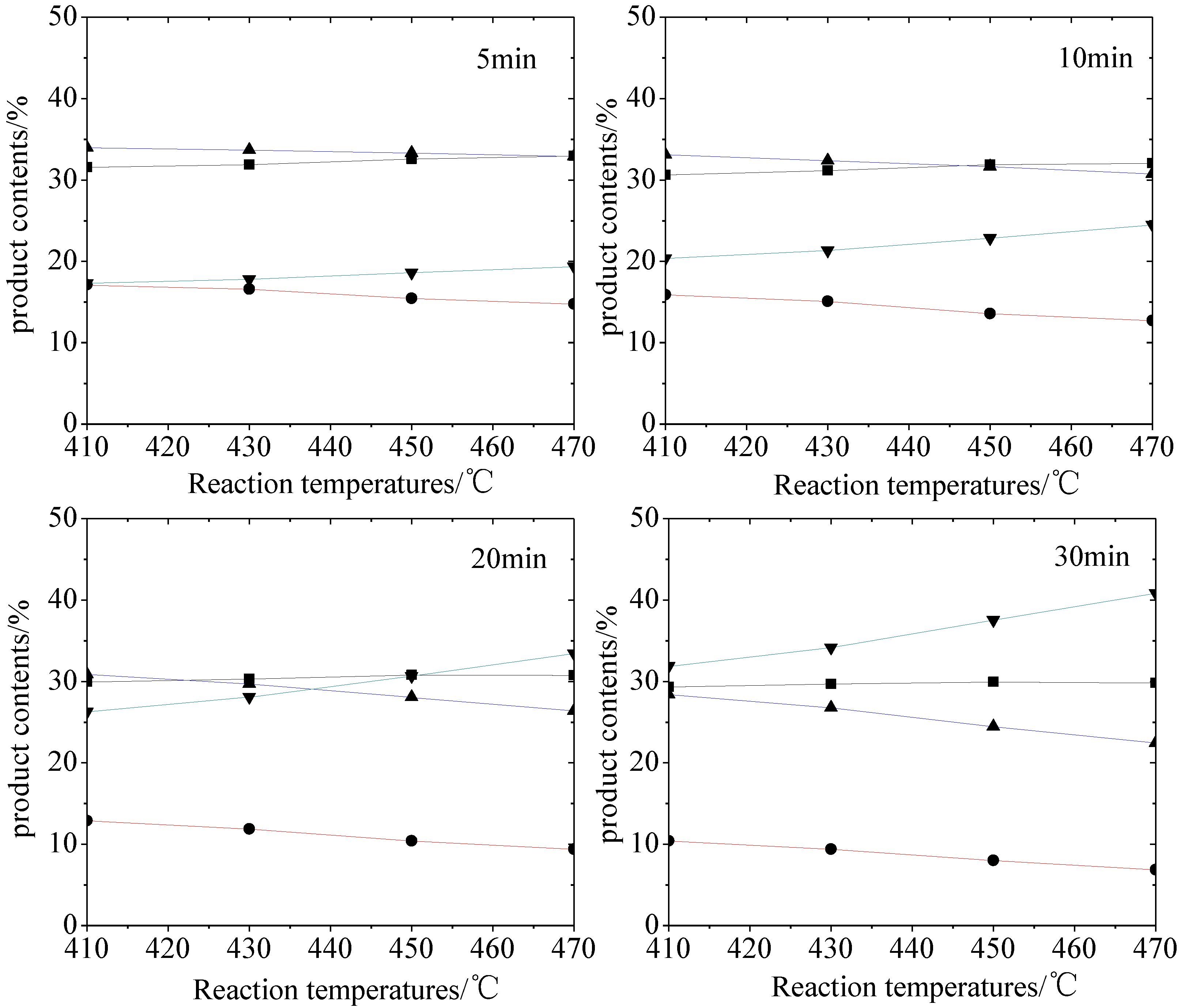
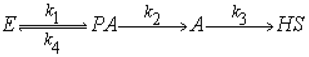
2.2. Hydro-Conversion Kinetic Model Establishment and Solution
- PA: the toluene insoluble/tetrahydrofuran soluble fractions
- A: the n-hexane insoluble/toluene soluble fractions
- HS: oil and gas
- E: Easily reactive part in THFIS
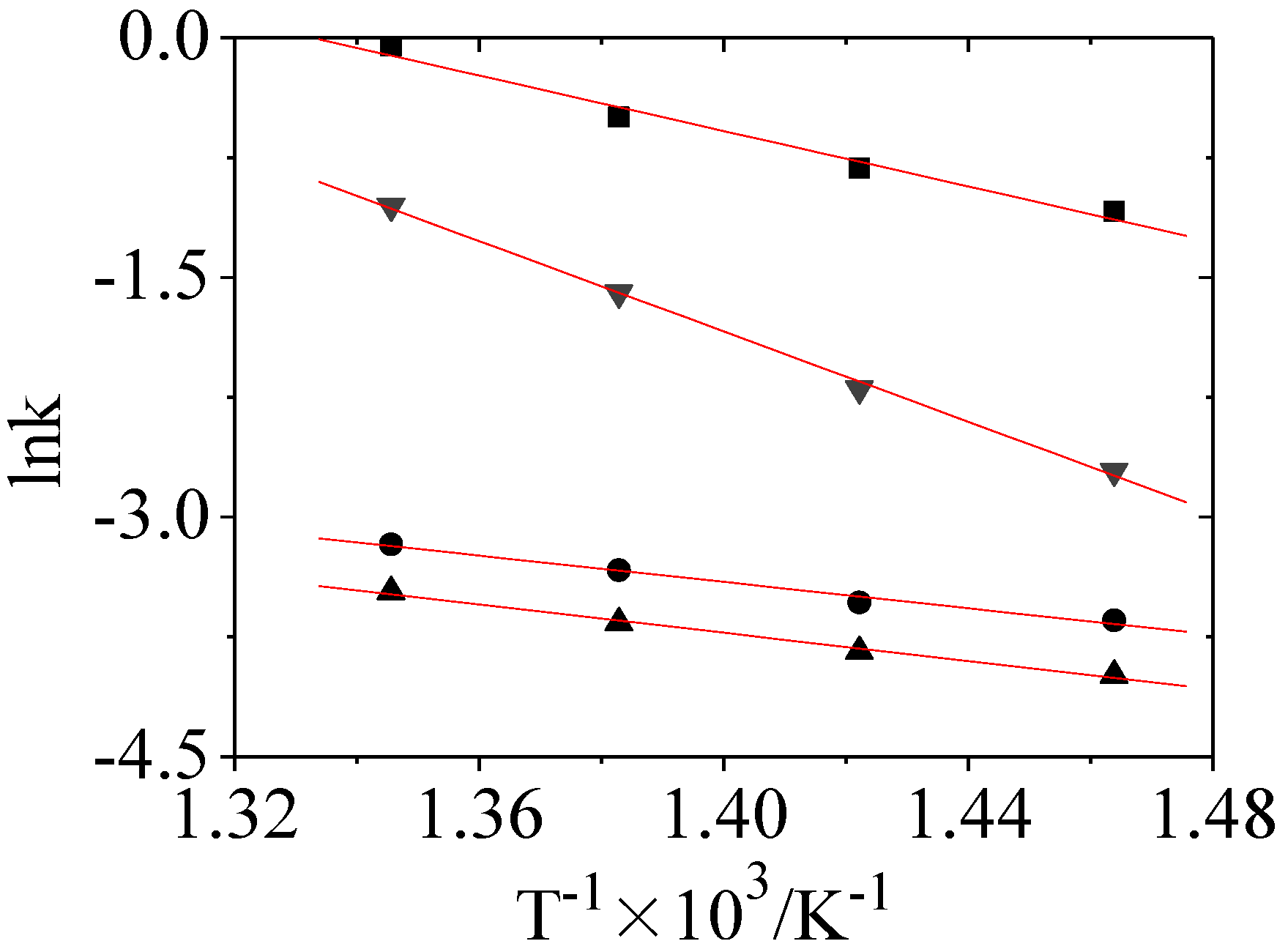
| Temperature/°C | 410 | 430 | 450 | 470 | Ea/kJ•mol−1 |
|---|---|---|---|---|---|
| k1 | 0.3362 | 0.4406 | 0.6077 | 0.9472 | 72.13 |
| k2 | 0.0261 | 0.0292 | 0.0357 | 0.0420 | 34.29 |
| k3 | 0.0185 | 0.0215 | 0.0257 | 0.0312 | 36.79 |
| k4 | 0.0665 | 0.1112 | 0.2023 | 0.3489 | 117.49 |
2.3 Verification of the Model
3. Experimental Section
4. Conclusions
- (1)
- CHLR obtained from coal hydroliquefaction reactor could be converted to lighter fractions under moderate hydrotreating conditions. At 470 °C and 30 min, HS was enhanced from 14.14% to 40.86% suggesting that the total HS yield in CHL could increase if CHLR is recycled as a co-reactant in CHL. It also indicates that a moderate extension of coal residence time in CHL is beneficial to the increase of oil yield.
- (2)
- Reverse reactions of PA were not significant under the hydrotreating conditions and the temperature of CHL was almost identical with this hydrotreating condition, which indicated that there could be no severe polymerization caused by recycling of CHLR in CHL.
- (3)
- Unlike the highly condensed poly-aromatic hydrocarbons and unreacted coal contained in THFIS, mildly condensed PA and A generated in high-temperature separator of the CHL process could easily depolymerize in a short time under the hydro-conversion which was also proved by a kinetic study.
- (4)
- On the basis of the non-uniform constitution of CHLR, the moderate hydrotreating performance of catalyst and the non-severity of temperature, the kinetics of the CHLR hydrogenation could be conveniently described by a 4-lumped kinetic model.
- (5)
- Our kinetic study of CHLR hydro-conversion shows that a large quantity of total catalyst consisting of remaining liquefaction catalyst, part of the minerals from raw coal and the Fe-based catalyst additive could considerably reduce the apparent activation energies of hydro-conversion for PA and A, which is beneficial to the recycling of CHLR in CHL.
Acknowledgements
References and Notes
- Chu, X.; Li, W.; Chen, H. Gasification property of direct coal liquefaction residue with steam. Process Saf. Environ. Prot. 2006, 84, 440–445. [Google Scholar] [CrossRef]
- Chu, X.J.; Li, W.; Li, B.Q.; Chen, H.K. Sulfur transfers from pyrolysis and gasification of direct liquefaction residue of Shenhua coal. Fuel 2008, 87, 211–215. [Google Scholar] [CrossRef]
- Zhou, Y.; Xiao, N.; Qiu, J.S.; Sun, Y.F.; Sun, T.J.; Zhao, Z.B.; Zhang, Y.; Tsubaki, N. Preparation of carbon microfibers from coal liquefaction residue. Fuel 2008, 87, 3474–3476. [Google Scholar] [CrossRef]
- Yang, J.L.; Wang, Z.X.; Liu, Z.Y.; Zhang, Y.Z. Novel Use of Residue from Direct Coal Liquefaction Process. Energy Fuels 2009, 23, 4717–4722. [Google Scholar] [CrossRef]
- Xiao, N.; Zhou, Y.; Qiu, J.S.; Wang, Z.H. Preparation of carbon nanofibers/carbon foam monolithic composite from coal liquefaction residue. Fuel 2010, 89, 1169–1171. [Google Scholar] [CrossRef]
- Benito, A.M.; Callejas, M.A.; Martinez, M.T.; Ana, M.B.; Maria, A.C.; Maria, T.M. Kinetics of asphaltene hydroconversion: 2. Catalytic hydrocracking of a coal residue. Fuel 1997, 76, 907–911. [Google Scholar] [CrossRef]
- Martinez, M.T.; Benito, A.M.; Callejas, M.A. Kinetics of asphaltene hydroconversion: 1. Thermal hydrocracking of a coal residue. Fuel 1997, 76, 899–905. [Google Scholar] [CrossRef]
- Sugano, M.; Ikemizu, R.; Mashimo, K. Effects of the oxidation pretreatment with hydrogen peroxide on the hydrogenolysis reactivity of coal liquefaction residue. Fuel Process. Technol. 2002, 77, 67–73. [Google Scholar] [CrossRef]
- Sugano, M.; Tamaru, T.; Hirano, K.; Mashimo, K. Additive effect of tyre constituents on the hydrogenolyses of coal liquefaction residue. Fuel 2005, 84, 2248–2255. [Google Scholar] [CrossRef]
- Sugano, M.; Onda, D.; Mashimo, K. Additive effect of waste tire on the hydrogenolysis reaction of coal liquefaction residue. Energy Fuels 2006, 20, 2713–2716. [Google Scholar] [CrossRef]
- Li, J.; Yang, J.L.; Liu, Z.Y. Hydro-treatment of a direct coal liquefaction residue and its components. Catal. Today 2008, 130, 389–394. [Google Scholar] [CrossRef]
- Wang, T.X.; Zong, Z.M.; Zhang, J.W.; Wei, Y.B.; Zhao, W.; Li, B.M.; Wei, X.Y. Microwave-assisted hydroconversions of demineralized coal liquefaction residues from Shenfu and Shengli coals. Fuel 2008, 87, 498–507. [Google Scholar] [CrossRef]
- Li, J.; Yang, J.L.; Liu, Z.Y. Hydrogenation of heavy liquids from a direct coal liquefaction residue for improved oil yield. Fuel Process. Technol. 2009, 90, 490–495. [Google Scholar] [CrossRef]
- Wei, Y.B.; Zong, Z.M.; Xie, R.L.; Peng, Y.L.; Mou, J.; Ma, Y.M.; Zhou, X.; Huang, Y.; Wu, L.; Wei, X.Y. Solid Superacid-catalyzed Hydroconversion of Demineralized Shengli Coal Liquefaction Residue under Microwave Irradiation. Energy Sources A 2010, 32, 551–558. [Google Scholar] [CrossRef]
- Ikeda, K.; Sakawaki, K.; Nogami, Y.; Inokuchi, K.; Imada, K. Kinetic evaluation of progress in coal liquefaction in the 1t/d PSU for the NEDOL process. Fuel 2000, 79, 373–378. [Google Scholar] [CrossRef]
- Lange, T.; Kopsel, R.; Kuchling, T.; Klose, E. Hydrogenation behaviour of a German brown coal: 2. Non-isothermic kinetics. Fuel 1989, 68, 366–370. [Google Scholar] [CrossRef]
- Shui, H.F.; Chen, Z.X.; Wang, Z.C.; Zhang, D.X. Kinetics of Shenhua coal liquefaction catalyzed by SO42=/ZrO2 solid acid. Fuel 2010, 89, 67–72. [Google Scholar] [CrossRef]
- Li, X.; Hu, H.Q.; Zhu, S.W.; Hu, S.X.; Wu, B.; Meng, M. Kinetics of coal liquefaction during heating-up and isothermal stages. Fuel 2008, 87, 508–513. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, J.; Lu, X.; Zhang, D.; Gao, J. A Study on the Hydrotreating of Coal Hydroliquefaction Residue and its Kinetics. Energies 2010, 3, 1576-1585. https://doi.org/10.3390/en3091576
Huang J, Lu X, Zhang D, Gao J. A Study on the Hydrotreating of Coal Hydroliquefaction Residue and its Kinetics. Energies. 2010; 3(9):1576-1585. https://doi.org/10.3390/en3091576
Chicago/Turabian StyleHuang, Jue, Xilan Lu, Dexiang Zhang, and Jinsheng Gao. 2010. "A Study on the Hydrotreating of Coal Hydroliquefaction Residue and its Kinetics" Energies 3, no. 9: 1576-1585. https://doi.org/10.3390/en3091576
APA StyleHuang, J., Lu, X., Zhang, D., & Gao, J. (2010). A Study on the Hydrotreating of Coal Hydroliquefaction Residue and its Kinetics. Energies, 3(9), 1576-1585. https://doi.org/10.3390/en3091576




