Impact of Plastic Blends on the Gaseous Product Composition from the Co-Pyrolysis Process
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Analytical Methods
2.2. Analytical Procedures
2.3. Experimental Design with Analysis of Variance (ANOVA)
2.4. Co-Pyrolysis Process
3. Results
3.1. The Co-Pyrolysis of Biomass and Plastic Waste Blends—Product Mass Balance
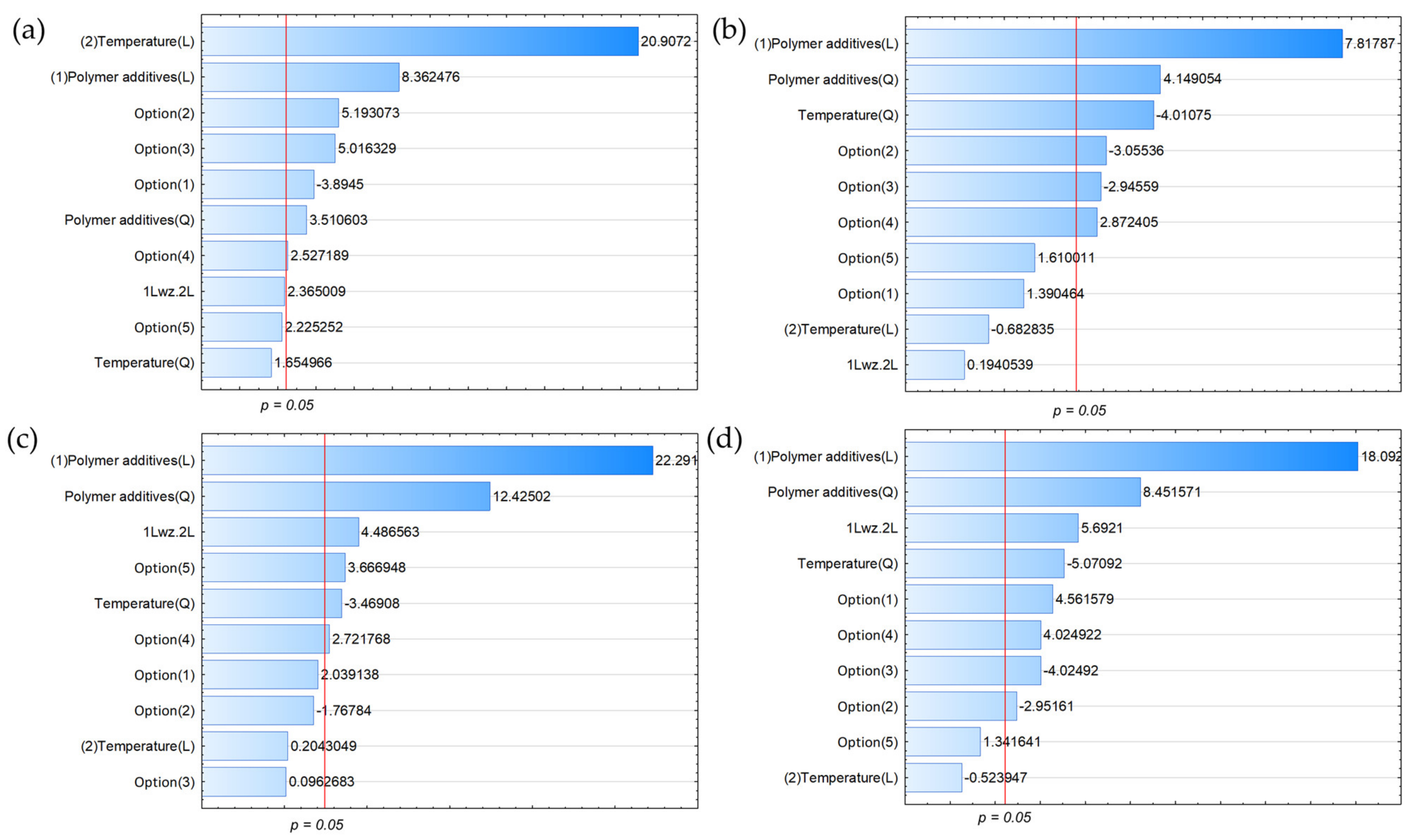
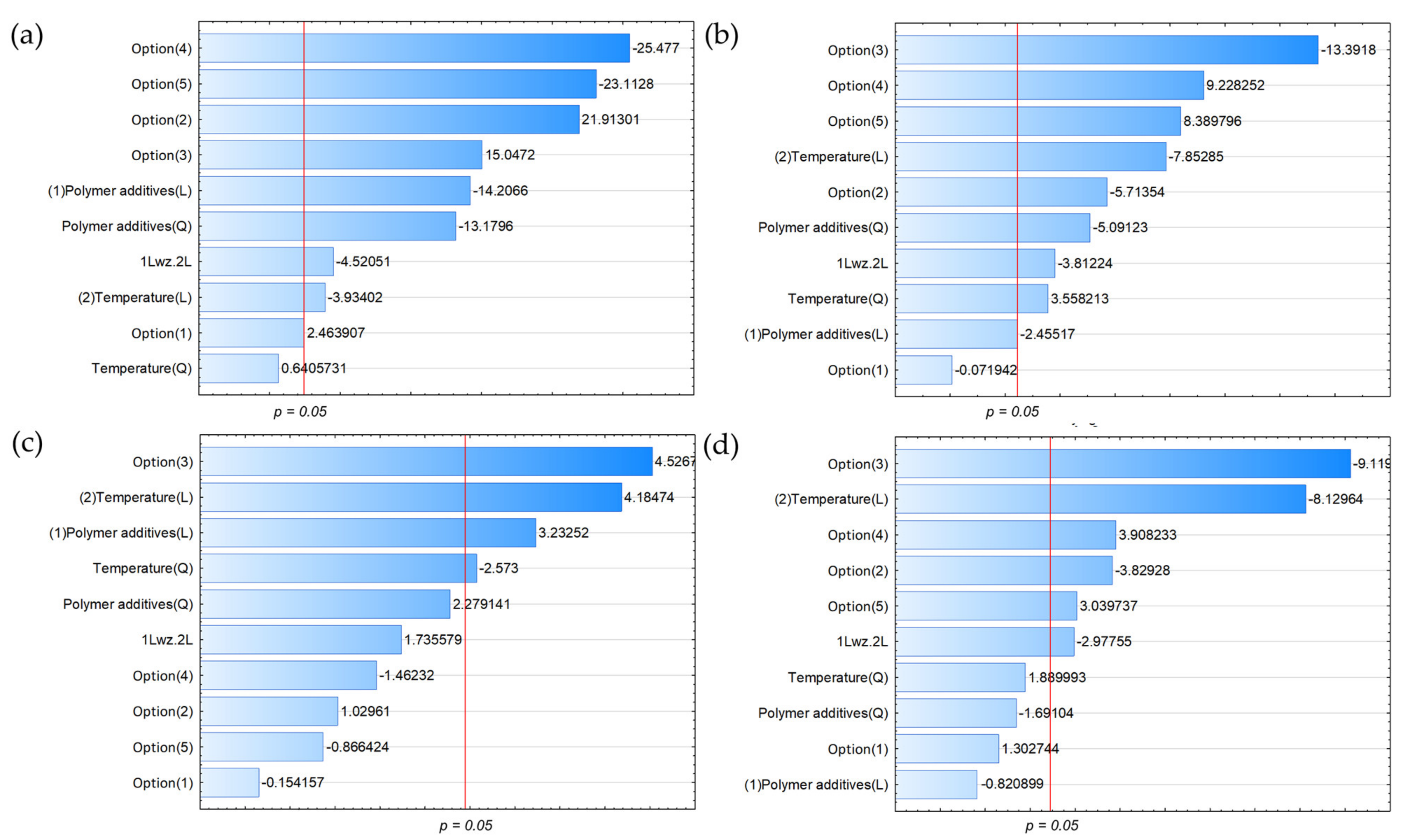
3.2. Variable Effect on the Chemical Composition of the Gaseous Products
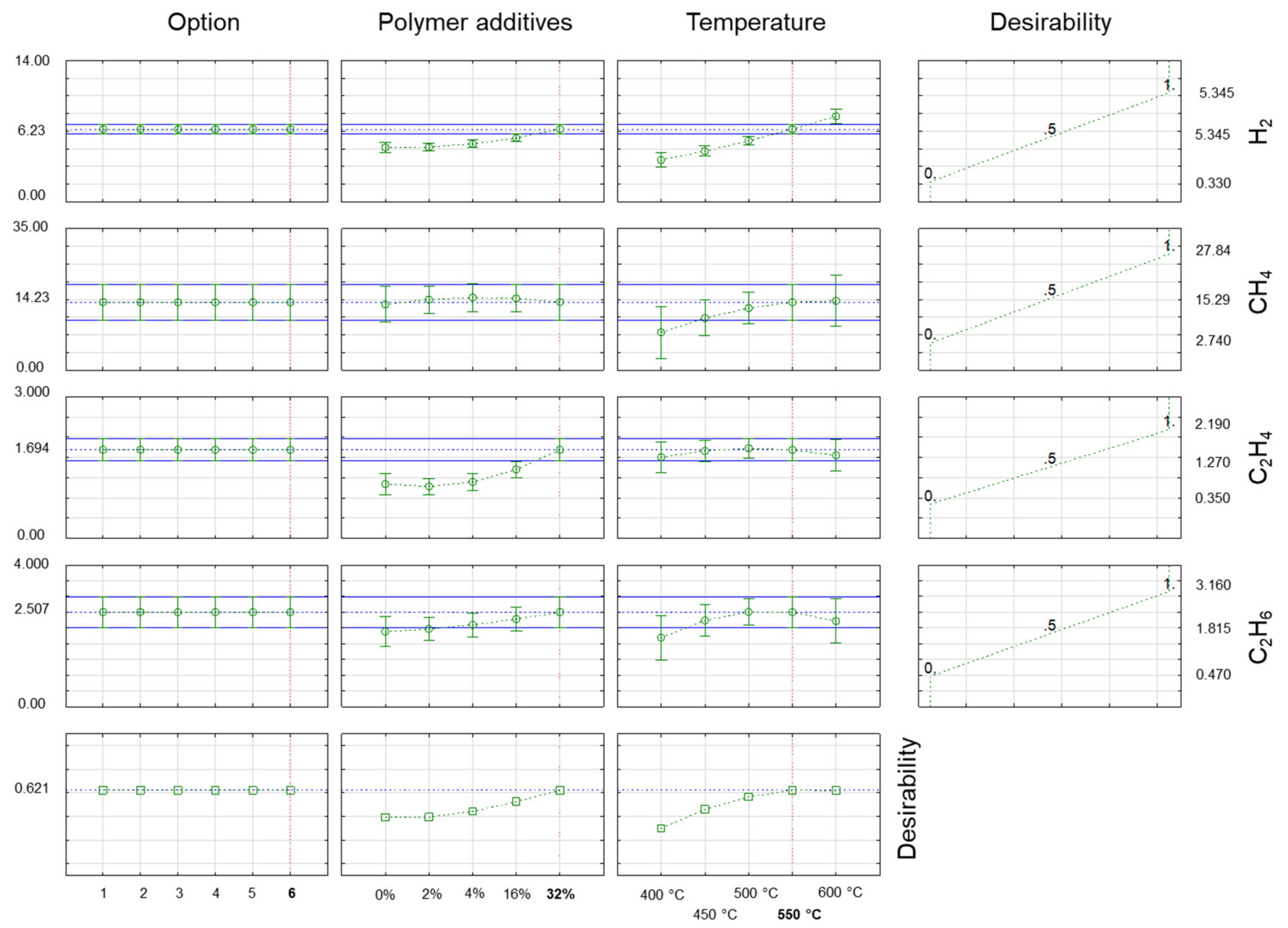
3.3. Determination of Optimal Co-Pyrolysis Parameters
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations Environment Programme, Converting Waste Plastics into a Resource—Compendium of Technologies. 2009. Available online: https://wedocs.unep.org/20.500.11822/8638 (accessed on 19 December 2022).
- Basu, P. Biomass Gasification and Pyrolysis: Practical Design and Theory; Academic Press: Cambridge, MA, USA, 2010; pp. 1–365. ISBN 9780123749888. [Google Scholar] [CrossRef]
- Scheirs, J.; Kaminsky, W. Feedstock Recycling and Pyrolysis of Waste Plastics: Converting Waste Plastics into Diesel and Other Fuels. Feedstock Recycl. Pyrolysis Waste Plast. Convert. Waste Plast. Into Diesel Other Fuels 2006, 1–785. [Google Scholar] [CrossRef]
- Gałko, G.; Rejdak, M.; Tercki, D.; Bogacka, M.; Sajdak, M. Evaluation of the Applicability of Polymeric Materials to BTEX and Fine Product Transformation by Catalytic and Non-Catalytic Pyrolysis as a Part of the Closed Loop Material Economy. J. Anal. Appl. Pyrolysis 2021, 154, 105017. [Google Scholar] [CrossRef]
- Gałko, G.; Mazurek, I.; Rejdak, M.; Jagustyn, B.; Hrabak, J.; Ouadi, M.; Jahangiri, H.; Sajdak, M. Evaluation of Alternative Refuse-Derived Fuel Use as a Valuable Resource in Various Valorised Applications. Energy 2023, 263 Pt D, 125920. [Google Scholar] [CrossRef]
- Scott, D.S.; Czernik, S.R.; Piskorz, J.; Radlein, D.S.A.G. Fast Pyrolysis of Plastic Wastes. Energy Fuels 1990, 4, 407–411. [Google Scholar] [CrossRef]
- Achilias, D.S.; Roupakias, C.; Megalokonomos, P.; Lappas, A.A.; Antonakou, V. Chemical Recycling of Plastic Wastes Made from Polyethylene (LDPE and HDPE) and Polypropylene (PP). J. Hazard. Mater. 2007, 149, 536–542. [Google Scholar] [CrossRef]
- Miskolczi, N.; Angyal, A.; Bartha, L.; Valkai, I. Fuels by Pyrolysis of Waste Plastics from Agricultural and Packaging Sectors in a Pilot Scale Reactor. Fuel Process. Technol. 2009, 90, 1032–1040. [Google Scholar] [CrossRef]
- Demirbas, A. Pyrolysis of Municipal Plastic Wastes for Recovery of Gasoline-Range Hydrocarbons. J. Anal. Appl. Pyrolysis 2004, 72, 97–102. [Google Scholar] [CrossRef]
- Babu, B.V.; Chaurasia, A.S. Modeling for Pyrolysis of Solid Particle: Kinetics and Heat Transfer Effects. Energy Convers Manag 2003, 44, 2251–2275. [Google Scholar] [CrossRef]
- Chen, D.; Yin, L.; Wang, H.; He, P. Pyrolysis Technologies for Municipal Solid Waste: A Review. Waste Manag. 2014, 34, 2466–2486. [Google Scholar] [CrossRef]
- Stanmore, B.R. The Formation of Dioxins in Combustion Systems. Combust. Flame 2004, 136, 398–427. [Google Scholar] [CrossRef]
- McKay, G. Dioxin Characterisation, Formation and Minimisation during Municipal Solid Waste (MSW) Incineration: Review. Chem. Eng. J. 2002, 86, 343–368. [Google Scholar] [CrossRef]
- Kanniche, M.; Gros-Bonnivard, R.; Jaud, P.; Valle-Marcos, J.; Amann, J.M.; Bouallou, C. Pre-Combustion, Post-Combustion and Oxy-Combustion in Thermal Power Plant for CO2 Capture. Appl. Eng. 2010, 30, 53–62. [Google Scholar] [CrossRef]
- Crocker, M. Thermochemical Conversion of Biomass to Liquid Fuels and Chemicals. The Royal Society of Chemistry: London, UK, 2011; Available online: https://pubs.rsc.org/en/content/ebook/978-1-84973-035-8 (accessed on 10 October 2022)ISBN 978-1-84973-035-8.
- Bagri, R.; Williams, P.T. Catalytic Pyrolysis of Polyethylene. J. Anal. Appl. Pyrolysis 2002, 63, 29–41. [Google Scholar] [CrossRef]
- Williams, P.T.; Slaney, E. Analysis of Products from the Pyrolysis and Liquefaction of Single Plastics and Waste Plastic Mixtures. Resour. Conserv. Recycl. 2007, 51, 754–769. [Google Scholar] [CrossRef]
- Serrano, D.P.; Aguado, J.; Escola, J.M.; Garagorri, E. Performance of a Continuous Screw Kiln Reactor for the Thermal and Catalytic Conversion of Polyethylene-Lubricating Oil Base Mixtures. Appl. Catal. B 2003, 44, 95–105. [Google Scholar] [CrossRef]
- Manos, G.; Garforth, A.; Dwyer, J. Catalytic Degradation of High-Density Polyethylene over Different Zeolitic Structures. Ind. Eng. Chem. Res. 2000, 39, 1198–1202. [Google Scholar] [CrossRef]
- del Remedio Hernández, M.; García, Á.N.; Marcilla, A. Study of the Gases Obtained in Thermal and Catalytic Flash Pyrolysis of HDPE in a Fluidized Bed Reactor. J. Anal. Appl. Pyrolysis 2005, 73, 314–322. [Google Scholar] [CrossRef]
- Muradov, N.Z. CO2-Free Production of Hydrogen by Catalytic Pyrolysis of Hydrocarbon Fuel. Energy Fuels 1998, 12, 41–48. [Google Scholar] [CrossRef]
- Miskolczi, N.; Ateş, F.; Borsodi, N. Comparison of Real Waste (MSW and MPW) Pyrolysis in Batch Reactor over Different Catalysts. Part II: Contaminants, Char and Pyrolysis Oil Properties. Bioresour. Technol. 2013, 144, 370–379. [Google Scholar] [CrossRef]
- Muhammad, C.; Onwudili, J.A.; Williams, P.T. Thermal Degradation of Real-World Waste Plastics and Simulated Mixed Plastics in a Two-Stage Pyrolysis-Catalysis Reactor for Fuel Production. Energy Fuels 2015, 29, 2601–2609. [Google Scholar] [CrossRef]
- Balakrishnan, R.K.; Guria, C. Thermal Degradation of Polystyrene in the Presence of Hydrogen by Catalyst in Solution. Polym. Degrad. Stab. 2007, 92, 1583–1591. [Google Scholar] [CrossRef]
- Syamsiro, M.; Saptoadi, H.; Norsujianto, T.; Noviasri, P.; Cheng, S.; Alimuddin, Z.; Yoshikawa, K. Fuel Oil Production from Municipal Plastic Wastes in Sequential Pyrolysis and Catalytic Reforming Reactors. Energy Procedia 2014, 47, 180–188. [Google Scholar] [CrossRef]
- Chung, S.H.; Park, J.J.; Jeon, S.G.; Kim, D.C. Pyrolysis of Waste Plastics Using Synthesized Catalysts from Fly Ash. J. Ind. Eng. Chem. 2003, 9, 181–187. [Google Scholar]
- Shah, J.; Jan, M.R.; Hussain, Z. Catalytic Pyrolysis of Low-Density Polyethylene with Lead Sulfide into Fuel Oil. Polym. Degrad. Stab. 2005, 87, 329–333. [Google Scholar] [CrossRef]
- Mastral, J.F.; Berrueco, C.; Gea, M.; Ceamanos, J. Catalytic Degradation of High Density Polyethylene over Nanocrystalline HZSM-5 Zeolite. Polym. Degrad. Stab. 2006, 91, 3330–3338. [Google Scholar] [CrossRef]
- Xiong, S.; Zhuo, J.; Zhou, H.; Pang, R.; Yao, Q. Study on the Co-Pyrolysis of High Density Polyethylene and Potato Blends Using Thermogravimetric Analyzer and Tubular Furnace. J. Anal. Appl. Pyrolysis 2015, 112, 66–73. [Google Scholar] [CrossRef]
- Čepelioğullar, Ö.; Pütün, A.E. Products Characterization Study of a Slow Pyrolysis of Biomass-Plastic Mixtures in a Fixed-Bed Reactor. J. Anal. Appl. Pyrolysis 2014, 110, 363–374. [Google Scholar] [CrossRef]
- Sajdak, M.; Kmieć, M.; Micek, B.; Hrabak, J. Determination of the Optimal Ratio of Coal to Biomass in the Co-Firing Process: Feed Mixture Properties. Int. J. Environ. Sci. Technol. 2019, 16, 2989–3000. [Google Scholar] [CrossRef]
- Sajdak, M.; Piotrowski, O. C&RT Model Application in Classification of Biomass for Energy Production and Environmental Protection. Cent. Eur. J. Chem. 2013, 11, 259–270. [Google Scholar] [CrossRef]
- Sajdak, M. Application of Chemometrics to Identifying Solid Fuels and Their Origin. Cent. Eur. J. Chem. 2013, 11, 151–159. [Google Scholar] [CrossRef]
- Sajdak, M.; Słowik, K. Use of Plastic Waste as a Fuel in the Co-Pyrolysis of Biomass: Part II. Variance Analysis of the Co-Pyrolysis Process. J. Anal. Appl. Pyrolysis 2014, 109, 152–158. [Google Scholar] [CrossRef]
- Sajdak, M.; Muzyka, R.; Hrabak, J.; Słowik, K. Use of Plastic Waste as a Fuel in the Co-Pyrolysis of Biomass: Part III: Optimisation of the Co-Pyrolysis Process. J. Anal. Appl. Pyrolysis 2015, 112, 298–305. [Google Scholar] [CrossRef]
- Sajdak, M.; Muzyka, R. Use of Plastic Waste as a Fuel in the Co-Pyrolysis of Biomass. Part I: The Effect of the Addition of Plastic Waste on the Process and Products. J. Anal. Appl. Pyrolysis 2014, 107, 267–275. [Google Scholar] [CrossRef]
- Sajdak, M. Impact of Plastic Blends on the Product Yield from Co-Pyrolysis of Lignin-Rich Materials. J. Anal. Appl. Pyrolysis 2017, 124, 415–425. [Google Scholar] [CrossRef]
- Çepelioǧullar, Ö.; Pütün, A.E. Thermal and Kinetic Behaviors of Biomass and Plastic Wastes in Co-Pyrolysis. Energy Convers Manag. 2013, 75, 263–270. [Google Scholar] [CrossRef]
- Chin, B.L.F.; Yusup, S.; al Shoaibi, A.; Kannan, P.; Srinivasakannan, C.; Sulaiman, S.A. Kinetic Studies of Co-Pyrolysis of Rubber Seed Shell with High Density Polyethylene. Energy Convers Manag. 2014, 87, 746–753. [Google Scholar] [CrossRef]
- Narobe, M.; Golob, J.; Klinar, D.; Francetič, V.; Likozar, B. Co-Gasification of Biomass and Plastics: Pyrolysis Kinetics Studies, Experiments on 100 KW Dual Fluidized Bed Pilot Plant and Development of Thermodynamic Equilibrium Model and Balances. Bioresour. Technol. 2014, 162, 21–29. [Google Scholar] [CrossRef]
- Alvarez, J.; Kumagai, S.; Wu, C.; Yoshioka, T.; Bilbao, J.; Olazar, M.; Williams, P.T. Hydrogen Production from Biomass and Plastic Mixtures by Pyrolysis-Gasification. Int. J. Hydrog. Energy 2014, 39, 10883–10891. [Google Scholar] [CrossRef]
- Aboulkas, A.; el Harfi, K.; el Bouadili, A. Pyrolysis of Olive Residue/Low Density Polyethylene Mixture: Part I Thermogravimetric Kinetics. J. Fuel Chem. Technol. 2008, 36, 672–678. [Google Scholar] [CrossRef]
- Brebu, M.; Ucar, S.; Vasile, C.; Yanik, J. Co-Pyrolysis of Pine Cone with Synthetic Polymers. Fuel 2010, 89, 1911–1918. [Google Scholar] [CrossRef]
- Jakab, E.; Várhegyi, G.; Faix, O. Thermal Decomposition of Polypropylene in the Presence of Wood-Derived Materials. J. Anal. Appl. Pyrolysis 2000, 56, 273–285. [Google Scholar] [CrossRef]
- Kumagai, S.; Fujita, K.; Kameda, T.; Yoshioka, T. Interactions of Beech Wood–Polyethylene Mixtures during Co-Pyrolysis. J. Anal. Appl. Pyrolysis 2016, 122, 531–540. [Google Scholar] [CrossRef]
- Ohmukai, Y.; Hasegawa, I.; Mae, K. Pyrolysis of the Mixture of Biomass and Plastics in Countercurrent Flow Reactor Part I: Experimental Analysis and Modeling of Kinetics. Fuel 2008, 87, 3105–3111. [Google Scholar] [CrossRef]
- Yoshioka, T.; Grause, G.; Eger, C.; Kaminsky, W.; Okuwaki, A. Pyrolysis of Poly(Ethylene Terephthalate) in a Fluidised Bed Plant. Polym. Degrad. Stab. 2004, 86, 499–504. [Google Scholar] [CrossRef]
- Williams, P.T.; Williams, E.A. Interaction of Plastics in Mixed-Plastics Pyrolysis. Energy Fuels 1998, 13, 188–196. [Google Scholar] [CrossRef]
- The Pyrolysis of Individual Plastics and a Plastic Mixture in a Fixed Bed Reactor—Williams—1997—Journal of Chemical Technology & Biotechnology—Wiley Online Library. Available online: https://onlinelibrary.wiley.co. (accessed on 6 November 2022).
- Fukushima, K.; Coady, D.J.; Jones, G.O.; Almegren, H.A.; Alabdulrahman, A.M.; Alsewailem, F.D.; Horn, H.W.; Rice, J.E.; Hedrick, J.L. Unexpected Efficiency of Cyclic Amidine Catalysts in Depolymerizing Poly (Ethylene Terephthalate). J. Polym. Sci. Part A Polym. Chem. 2013, 51, 1606–1611. [Google Scholar] [CrossRef]
- Kumagai, S.; Hasegawa, I.; Grause, G.; Kameda, T.; Yoshioka, T. Thermal Decomposition of Individual and Mixed Plastics in the Presence of CaO or Ca(OH)2. J. Anal. Appl. Pyrolysis 2015, 113, 584–590. [Google Scholar] [CrossRef]
- Samperi, F.; Puglisi, C.; Alicata, R.; Montaudo, G. Thermal Degradation of Poly(Butylene Terephthalate) at the Processing Temperature. Polym. Degrad. Stab. 2004, 83, 11–17. [Google Scholar] [CrossRef]
- Montaudo, G.; Puglisi, C.; Samperi, F. Primary Thermal Degradation Mechanisms of PET and PBT. Polym. Degrad. Stab. 1993, 42, 13–28. [Google Scholar] [CrossRef]
- Holland, B.J.; Hay, J.N. The Thermal Degradation of PET and Analogous Polyesters Measured by Thermal Analysis-Fourier Transform Infrared Spectroscopy. Polym. (Guildf) 2002, 43, 1835–1847. [Google Scholar] [CrossRef]
- Muzyka, R.; Chrubasik, M.; Dudziak, M.; Ouadi, M.; Sajdak, M. Pyrolysis of Tobacco Waste: A Comparative Study between Py-GC/MS and Fixed-Bed Reactors. J. Anal. Appl. Pyrolysis 2022, 167, 105702. [Google Scholar] [CrossRef]
- Sajdak, M.; Muzyka, R.; Hrabak, J.; Rózycki, G. Biomass, Biochar and Hard Coal: Data Mining Application to Elemental Composition and High Heating Values Prediction. J. Anal. Appl. Pyrolysis 2013, 104, 153–160. [Google Scholar] [CrossRef]
- Montgomery, D.C.; Wiley, J. Design and Analysis of Experiments, 8th ed; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- TIBCO Software. TIBCO® Statistica. Available online: https://www.tibco.com/resources/product-download/tibco-statistica-trial-download-for-windows (accessed on 13 January 2023).
- Sajdak, M. Characteristics of Chars from Biomass and Waste Co-Pyrolysis. 2021. Version 2. Available online: https://data.mendeley.com/datasets/rjg7j5wzrj/2 (accessed on 4 October 2021).
- Wang, Z.; Burra, K.G.; Lei, T.; Gupta, A.K. Co-Pyrolysis of Waste Plastic and Solid Biomass for Synergistic Production of Biofuels and Chemicals-A Review. Prog. Energy Combust. Sci. 2021, 84, 100899. [Google Scholar] [CrossRef]
- Sajdak, M.; Muzyka, R. Gaseous Products from Biomass and Waste Co-Pyrolysis—DOE. Version 1. 2022. Available online: https://data.mendeley.com/datasets/ny8bxjkhyk/1 (accessed on 14 November 2022).


| Parameter | Unit | ABS | PET | PP | SBR |
|---|---|---|---|---|---|
| Ash content—Ash | wt.% | 0.00 | 0.00 | 0.00 | 14.01 |
| Volatile Matter—VM | 99.95 | 86.84 | 99.92 | 61.61 | |
| Fixed carbon—FC | 0.00 | 13.11 | 0.00 | 24.36 | |
| Moisture content—Wa | 0.05 | 0.05 | 0.08 | 0.02 | |
| Carbon content-Cta | 75.44 | 62.48 | 84.8 | 70.42 | |
| Hydrogen content—Hta | 8.19 | 4.17 | 14.5 | 6.46 | |
| Oxygen content—Ota | 3.44 | 33.3 | 0.25 | 7.22 | |
| Nitrogen content—Na | 4.74 | 0.00 | 0.14 | 0.43 | |
| Sulphur content—Sta | 8.14 | 0.00 | 0.23 | 1.44 | |
| High heating value—HHV | MJ/kg | 38.09 | 30.85 | 45.80 | 33.92 |
| Parameter | Unit | Alder Wood | Straw Biomass | Furniture Waste |
|---|---|---|---|---|
| Ash | wt.% | 0.35 | 4.78 | 2.02 |
| VM | 78.55 | 70.50 | 73.87 | |
| FC | 14.78 | 17.18 | 17.78 | |
| Wa | 6.32 | 7.54 | 6.24 | |
| Cta | 45.90 | 43.80 | 47.70 | |
| Hta | 5.67 | 5.48 | 5.83 | |
| Ota | 37.40 | 38.69 | 41.79 | |
| Na | 4.47 | 0.62 | 0.26 | |
| Sta | 0.08 | 0.10 | 0.40 | |
| HHV | MJ/kg | 17.835 | 17.523 | 19.072 |
| Parameters/Levels | Symbols | ||
|---|---|---|---|
| Coding—Classical Experimental Design | A—Temperature (°C) | B—Polymer Blends Addition (M1, M2) (%) | |
| Lowest | −1.414 | 400 | 0.0 |
| Low | −1 | 450 | 2.0 |
| Centre | 0 | 500 | 4.0 |
| High | +1 | 550 | 16.0 |
| Highest | +1.414 | 600 | 32.0 |
| Pyrolysis Temperature | Char (% m/m) | Liquid Fraction (% m/m) | Gaseous Fraction (% m/m) |
|---|---|---|---|
| Straw biomass | |||
| 400 | 54.8 | 37.5 | 7.60 |
| 450 | 38.0 | 40.4 | 21.6 |
| 500 | 37.6 | 39.8 | 22.6 |
| 550 | 32.4 | 43.0 | 24.6 |
| 600 | 29.3 | 47.5 | 23.2 |
| Alder wood | |||
| 400 | 39.0 | 44.9 | 16.1 |
| 450 | 37.8 | 47.7 | 14.5 |
| 500 | 27.8 | 55.0 | 17.1 |
| 550 | 27.2 | 55.2 | 17.6 |
| 600 | 25.3 | 55.9 | 18.8 |
| Waste furniture | |||
| 400 | 53.5 | 32.1 | 14.4 |
| 450 | 38.5 | 44.2 | 17.3 |
| 500 | 32.7 | 47.5 | 19.8 |
| 550 | 30.3 | 48.9 | 20.8 |
| 600 | 29.0 | 49.6 | 21.4 |
| Addition (% m/m) | Pyrolysis Temperature (°C) | Char (% m/m) | Liquid Fraction (% m/m) | Gaseous Fraction (% m/m) | Char (% m/m) | Liquid Fraction (% m/m) | Gaseous Fraction (% m/m) |
|---|---|---|---|---|---|---|---|
| Abder wood + M1 | Alder wood + M2 | ||||||
| 2 | 450 | 28.00 | 54.40 | 17.61 | 30.50 | 54.02 | 15.48 |
| 2 | 550 | 25.25 | 56.56 | 18.19 | 23.25 | 58.59 | 18.17 |
| 16 | 450 | 38.50 | 48.24 | 19.68 | 31.75 | 54.67 | 13.58 |
| 16 | 550 | 29.25 | 50.25 | 20.51 | 26.00 | 58.99 | 15.01 |
| 0 | 500 | 27.67 | 55.17 | 17.16 | 27.67 | 55.17 | 17.16 |
| 32 | 500 | 29.50 | 54.12 | 16.39 | 28.75 | 56.16 | 15.09 |
| 4 | 400 | 36.50 | 49.50 | 14.01 | 35.00 | 51.33 | 13.67 |
| 4 | 600 | 23.50 | 58.20 | 18.31 | 23.25 | 58.72 | 18.04 |
| 4 | 500 | 24.75 | 57.61 | 17.64 | 25.75 | 57.46 | 16.79 |
| 4 | 500 | 26.25 | 57.09 | 16.67 | 26.25 | 56.92 | 16.83 |
| Straw + M1 | Straw + M2 | ||||||
| 2 | 450 | 34.00 | 44.96 | 21.04 | 31.25 | 47.78 | 20.98 |
| 2 | 550 | 29.50 | 47.81 | 22.69 | 27.50 | 50.16 | 22.34 |
| 16 | 450 | 37.25 | 40.79 | 21.97 | 33.75 | 47.97 | 18.29 |
| 16 | 550 | 29.75 | 50.21 | 20.05 | 27.00 | 54.11 | 18.90 |
| 0 | 500 | 38.94 | 39.28 | 21.77 | 38.94 | 39.28 | 21.77 |
| 32 | 500 | 34.25 | 41.58 | 19.43 | 30.00 | 51.38 | 18.62 |
| 4 | 400 | 39.00 | 41.58 | 19.43 | 38.25 | 43.25 | 18.50 |
| 4 | 600 | 27.25 | 49.82 | 22.94 | 26.00 | 51.81 | 22.19 |
| 4 | 500 | 29.75 | 49.17 | 21.09 | 30.25 | 49.36 | 20.40 |
| 4 | 500 | 32.25 | 46.43 | 21.33 | 31.00 | 45.56 | 23.44 |
| Waste furniture + M1 | Waste furniture + M2 | ||||||
| 2 | 450 | 41.00 | 41.21 | 17.79 | 36.00 | 46.54 | 17.46 |
| 2 | 550 | 28.00 | 51.88 | 20.12 | 28.00 | 51.99 | 20.01 |
| 16 | 450 | 42.00 | 33.64 | 24.36 | 36.00 | 46.96 | 17.04 |
| 16 | 550 | 29.00 | 50.42 | 20.58 | 29.00 | 50.73 | 20.27 |
| 0 | 500 | 36.92 | 45.08 | 18.00 | 36.92 | 45.08 | 18.00 |
| 32 | 500 | 38.50 | 38.95 | 22.55 | 40.00 | 47.89 | 12.74 |
| 4 | 400 | 43.00 | 42.20 | 14.80 | 41.50 | 44.34 | 14.16 |
| 4 | 600 | 27.00 | 52.25 | 20.75 | 23.00 | 55.33 | 21.67 |
| 4 | 500 | 30.00 | 47.86 | 22.14 | 30.50 | 50.25 | 19.25 |
| 4 | 500 | 33.00 | 48.00 | 19.00 | 29.00 | 50.19 | 20.81 |
| Variable | Temperature | Polymer Additives | Interactions | Pyrolysis Option |
| Parameter | ||||
| H2 | + | + | - | + |
| CO | + | + | + | + |
| CH4 | + | - | - | - |
| CO2 | + | + | + | + |
| C2H4 | - | + | - | - |
| C2H6 | + | + | - | - |
| C3H8 | + | + | - | + |
| C3H6 | + | + | + | + |
| C4H10 | + | + | + | + |
| HHV | + | + | - | + |
| LHV | + | + | - | + |
| Density | + | - | + | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muzyka, R.; Gałko, G.; Ouadi, M.; Sajdak, M. Impact of Plastic Blends on the Gaseous Product Composition from the Co-Pyrolysis Process. Energies 2023, 16, 947. https://doi.org/10.3390/en16020947
Muzyka R, Gałko G, Ouadi M, Sajdak M. Impact of Plastic Blends on the Gaseous Product Composition from the Co-Pyrolysis Process. Energies. 2023; 16(2):947. https://doi.org/10.3390/en16020947
Chicago/Turabian StyleMuzyka, Roksana, Grzegorz Gałko, Miloud Ouadi, and Marcin Sajdak. 2023. "Impact of Plastic Blends on the Gaseous Product Composition from the Co-Pyrolysis Process" Energies 16, no. 2: 947. https://doi.org/10.3390/en16020947
APA StyleMuzyka, R., Gałko, G., Ouadi, M., & Sajdak, M. (2023). Impact of Plastic Blends on the Gaseous Product Composition from the Co-Pyrolysis Process. Energies, 16(2), 947. https://doi.org/10.3390/en16020947









