Abstract
As greenhouse gas emissions are continuously increasing, research is now privileging greener and more sustainable human activities. An attractive strategy in the pursuit of sustainability is the valorization of lignocellulosic biomasses for the production of bioethanol. This approach relies on the bioconversion of wood and agricultural waste, which are abundant globally. They represent considerable sources of fermentable sugars that can be recovered through enzymatic hydrolysis. However, the presence of lignin in wood waste makes it more recalcitrant to enzymatic hydrolysis, and reduces the efficiency of the bioconversion process. Therefore, a pretreatment preceding hydrolysis is highly necessary in order to disrupt the resistant structure of woody biomass. The type and severity of the pretreatment affect the outcomes of the hydrolysis and fermentation steps, just as they strongly influence the overall process costs. Given this context, bioenergy production from this biomass is a promising alternative method of sustainably responding to energy demands while reducing the amounts of waste left in nature. The aim of this review is to thoroughly compare the recent bioconversion processes performed on woody substrates over the past five years, with a focus on thermomechanical pretreatments. Moreover, this review will discuss the outcomes of their separate steps, their impact on the overall process, and their energetic aspects.
1. Introduction
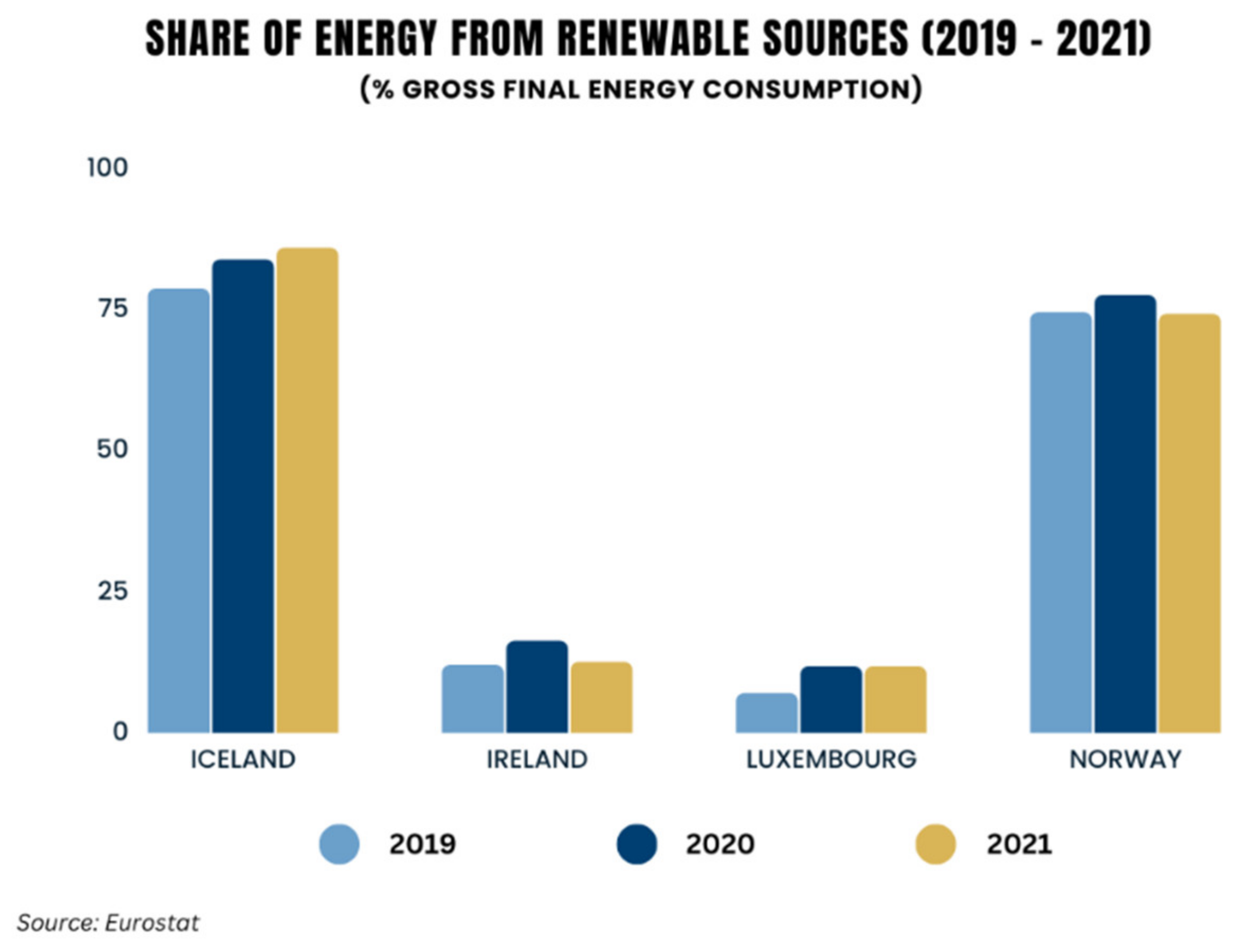
The availability of energy is essential for the economic development and prosperity of states. Global energy demand is increasing due to population growth and to the developing needs of emerging economies [1,2]. The current surge in fossil fuel prices is having an impact on most of the world’s economies, and raises the issue of energy security. The adoption of renewable energy can not only help in mitigating climate change, but can also enhance resilience to price volatility and reduce energy costs [3,4]. Many industries are responsible for high energy demand, with a significant contribution coming from the transportation sector. In fact, this sector’s consumption accounts for approximately 25% of the world’s energy use [5], while the need for green and renewable energy sources has increased [6]. Moreover, the total global biofuel demand is expected to expand by 20% between 2022 and 2027 [7]. Another important source of global greenhouse gas emissions is the heat and electricity sector, as it was notably responsible for the largest increase in carbon dioxide (CO2) emissions in 2021 [8]. However, as in the transportation sector, the need for renewable and greener energy is now greater than ever, in order to meet energy demands while ensuring sustainability. It is hence worthwhile to note that such shifts have already begun; the share of energy coming from renewable sources in many European Union countries increased between 2004 and 2021, most notably in Iceland and Norway [9] (Figure 1).
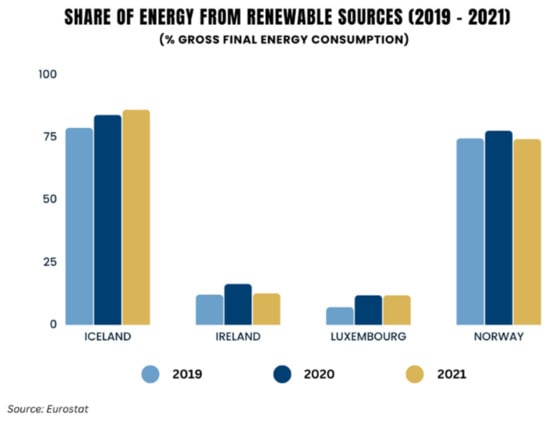
Figure 1.
Percentage share of energy from renewable sources in EU countries (2019–2021).
Nonetheless, political instability and financial scarcity in many countries obstruct access to renewable and greener energies by discouraging potential investors, whilst the current fossil fuel crises further push populations to find short-term solutions to increased energy prices [10,11]. A promising alternative is the production of biofuels from lignocellulosic biomass (LCB), a plant resource named after its three main components: lignin, cellulose, and hemicellulose [12,13,14]. The choice of LCB is based on the geographic distribution of attractive crops. From this point of view, forests account for 31% of the world’s land area [15], and 35% of the total area of Europe.
Wood is a natural resource classified into two types, hardwoods and softwoods, which come from angiosperms and gymnosperms trees, respectively. From an environmental perspective, both living hardwood and softwood forests play a role in carbon sequestration. While this may help in reducing the effects of global warming, the processing of their woods generates various polluting byproducts. Such byproducts are abundant and low in cost, as they are mainly derived from agricultural and forestry waste and residues [12,14,16,17], and are not part of the food chain. Although forest and wood residues are commonly studied, there also exist agricultural wastes of a woody nature, such as the Chinese chaste tree (Vitex negundo), kenaf (Hibiscus cannabinus L.), and industrial hemp (Cannabis sativa). In fact, the use of industrial hemp for bioethanol production gained popularity because this residue is derived from a sustainable crop; it grows rapidly, requires little water and pesticides, can be cultivated in a variety of climates, and eliminates contaminants from soils [18,19,20]. Furthermore, hemp cultures play an important role in carbon sequestration, as one hectare can capture about 9–20 tons of CO2, which is comparable to the levels sequestered by forests [19,21,22]. This globally cultivated crop yields large biomass and has a variety of uses, including textiles, cosmetics, food and beverages, medicine, and construction [23,24,25]. Regarding hemp hurds, they originate from the defibrillation of industrial hemp [26,27]. The chemical composition of this byproduct was compared to that of hardwood in a study conducted by Naithani et al. [28], and their results showed a similar composition in lignin, cellulose, and hemicellulose, which underlines the potential of using hemp hurds in bioethanol production.
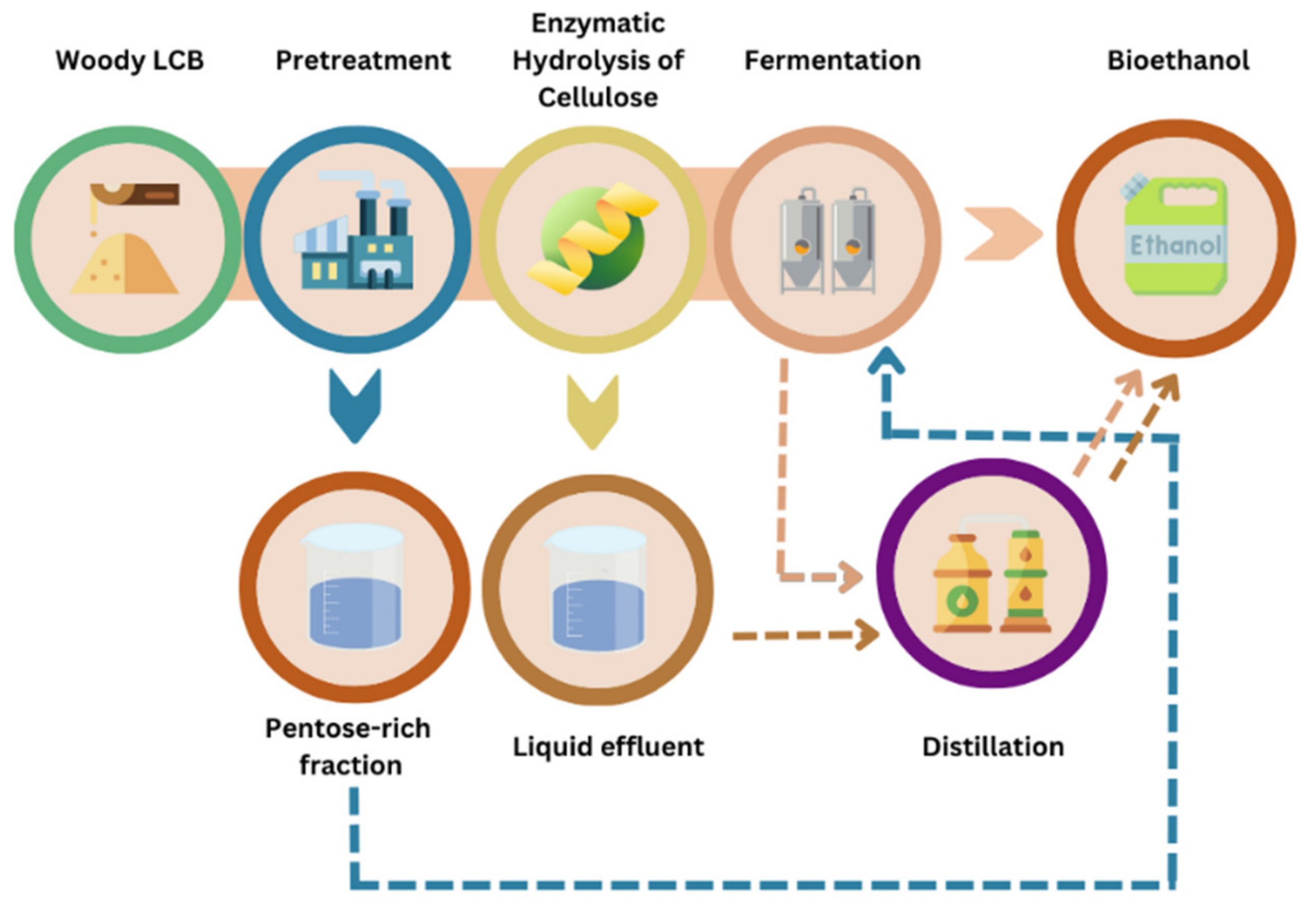
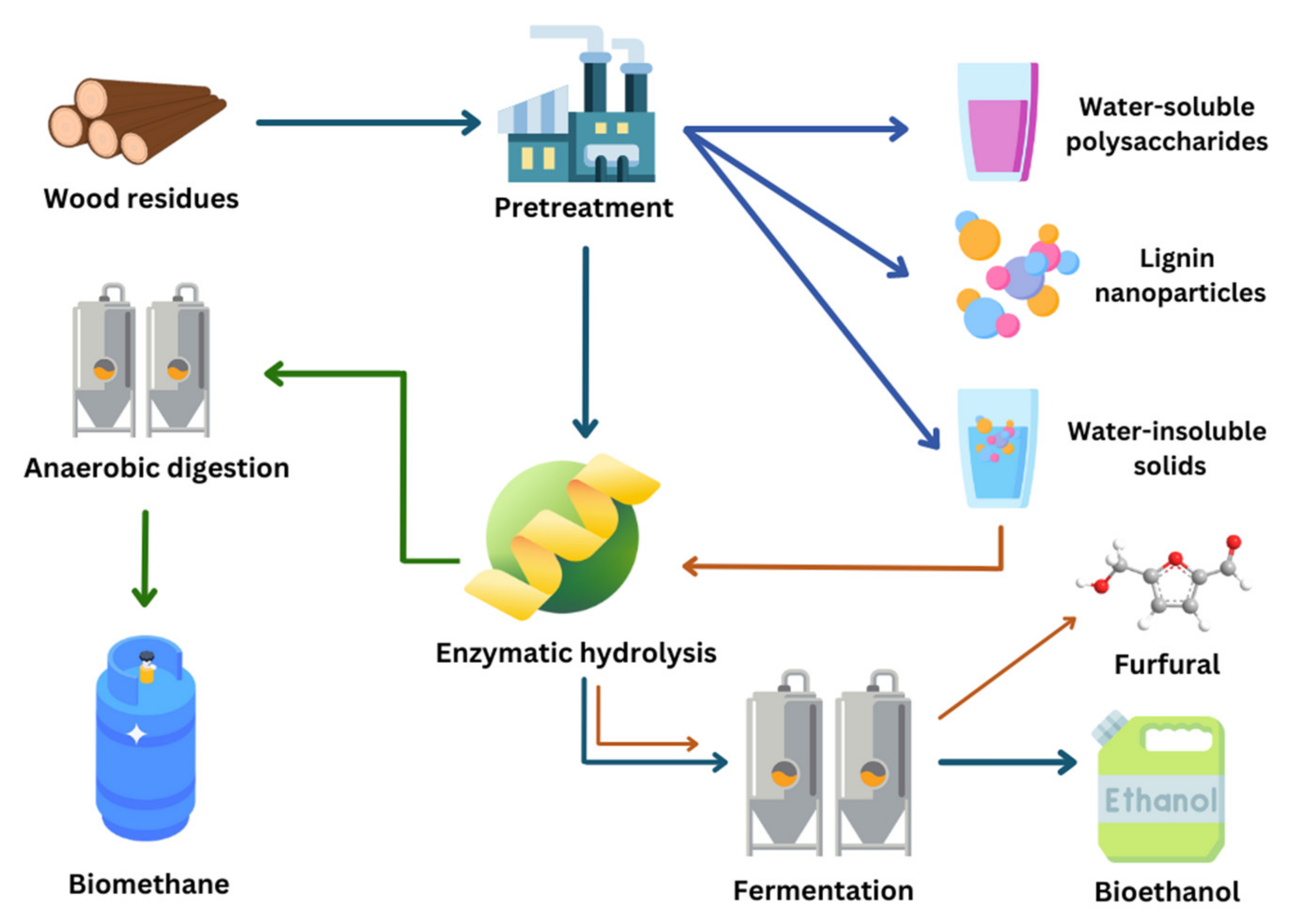
On the other hand, it is critical to note that the structure of lignocellulosic biomasses, whether woody or herbaceous, is complex and highly recalcitrant to enzymatic hydrolysis, the step of the bioconversion process that enables the recovery of fermentable sugars for the subsequent production of bioethanol [29,30,31]. Therefore, a pretreatment step is required to disrupt the complex structure and increase enzyme accessibility to cellulose and hemicellulose. Pretreatment can take several forms: chemical, biological, mechanical, or even a combination of two or more types of pretreatment (thermochemical, thermomechanical, etc.). Following pretreatment and enzymatic hydrolysis is the fermentation step, which aims to employ the recovered monomeric sugars and convert them into bioethanol and/or other products [32]. Finally, the inclusion of a distillation step allows the separation of the alcohol from the fermentation mixture, and the retrieval of pure ethanol [33,34]. Diverse woody and agricultural biomasses have been subjected to various pretreatments, both in previous and more recent studies, and previously reviewed [35]. The diagram in Figure 2 illustrates the different steps in a lignocellulosic biomass biorefinery. The viability of a bioconversion process, based on a sustainable and environmentally friendly approach, is necessarily associated with the maximum sugar yield that can be achieved with minimum inhibitors’ production, energy, and chemical consumption.

Figure 2.
Bioconversion process for bioethanol production.
The techno-economic assessment plays an important role in the scale-up and large-scale implementation of bioethanol production from lignocellulosic biomasses. Many studies have discussed the techno-economic performance of biofuel production by considering the overall process, including pretreatment, detoxification, hydrolysis, and fermentation [36,37,38]. Knowing that the pretreatment is a crucial step which considerably affects the production cost, some studies have focused only on the pretreatment step [39]. The techno-economic assessment discussed in a recent review [40] established that the overall cost of operation essentially comprises chemical cost and energy consumption. Hence, pretreatment and detoxification are key steps in determining the economics of the process. The environmental sustainability of bioethanol production is also an important aspect that has to be estimated. Among physico-chemical pretreatments such as liquid hot water, the AFEX process, or wet air oxidation, steam explosion affects the physiochemical properties of lignocellulosic biomass by breaking down the lignocellulosic structure and hydrolyzing the hemicellulosic fraction with lesser environmental consequences [41]. Zhao et al. [42] compared the integrated environmental impacts of different technological configurations, including pretreatment, hydrolysis, and fermentation steps, for bioethanol production from corn stover. It was pointed out that based on the cumulative probabilities of overall environmental performance together with the bioethanol amount produced, the pretreatment technologies that should be prioritized for further development are steam explosion and ammonia-based processes.
The objective of this review is to comprehensively analyze recent advances in the bioconversion processes of various woody substrates, summarizing studies on the matter that have been published in the past five years. Special attention will be given to the significance of thermomechanical pretreatments, as well as their influence on bioconversion and overall environmental impact. The environmental and economic aspects are discussed in the last section of this review.
2. Pretreatment of Woody Biomass
The type of pretreatment (PT) and its severity are crucial for an efficient delignification of the substrates in order to increase the effectiveness of enzymatic hydrolysis. The severity of a PT is assessed based on a combined severity factor (CSF), which is determined considering the PT temperature, its duration, and the pH when a catalyst is being used [26,43,44]. This factor allows us to compare the intensity of the PTs performed under defined conditions; its three parameters are correlated according to the equation below:
where T: temperature (°C) and t: time (min)
It is also important to note that particle size reduction and drying are often performed before PT. Reducing the size of the sample particles increases the surface area accessibility of the substrate, thus enhancing the subsequent process [26,45,46]. In fact, a study on steam-exploded beech wood has concluded that an excessive reduction in particle size (0.16 mm) leads to more advanced cooking of the biomass and more pronounced xylan (the major component of hemicellulose) [47] degradation. Meanwhile, a relatively large particle size (2 mm) led to incomplete autohydrolysis and a less efficient xylan extraction [48]. Therefore, particle size reduction is a pre-PT that must also be taken into account when considering the PT severity. On the other hand, drying also increases PT effectiveness, since it reduces the moisture content of the substrate, thus allowing it to be more homogenous [26].
The chemical compositions of some of the commonly utilized woody biomasses for the production of bioethanol are summarized in Table 1. The slight discrepancies observed in cellulose, hemicellulose, and lignin contents between different lignocellulosic substrates are typically attributed to differences in cultivars and species.

Table 1.
Chemical composition of commonly studied woody biomasses. Hardwoods are highlighted in green, and softwoods in yellow.
Hardwoods and softwoods exhibit distinct differences, not only in their origin, but also at the molecular level. To further understand the difference in PT outcomes, it should first be reminded that lignin is a biopolymer derived from three subunits: syringyl (S), guaiacyl (G), and hydroxyphenyl (H) [72,73,74]. The lignin composition, or the relative contents of S/G/H subunits, has various effects on the efficiency of the bioconversion process. In fact, the literature [75] states that differences in S/G ratios influence the way lignin and other plant cell wall components are linked, which affects the recalcitrance of a substrate to enzymatic hydrolysis. Additionally, since softwoods have a lower S/G ratio than hardwoods, they are less reactive to the PT step [76]. A higher S/G ratio increases PT efficiency due to the presence of more β-O-4′ linkages that are more easily cleavable during PT, just as a higher ratio means that there are more β-β’ linkages, resulting in a facilitated lignin removal [77]. However, some other studies have reported a negative or an insignificant correlation [75,77], which confirms the varied effect of this ratio on efficiency, and incites further research. Therefore, the adequacy or the inefficiency of a PT in delignifying a substrate can be reflected by these two factors. In addition, it is important to mention that under acidic, alkaline, and neutral conditions, it is possible for new C-C linkages to form intermolecularly between lignin fractions, leading to the formation of undesired new lignin molecules [78].
2.1. Thermomechanical Pretreatments
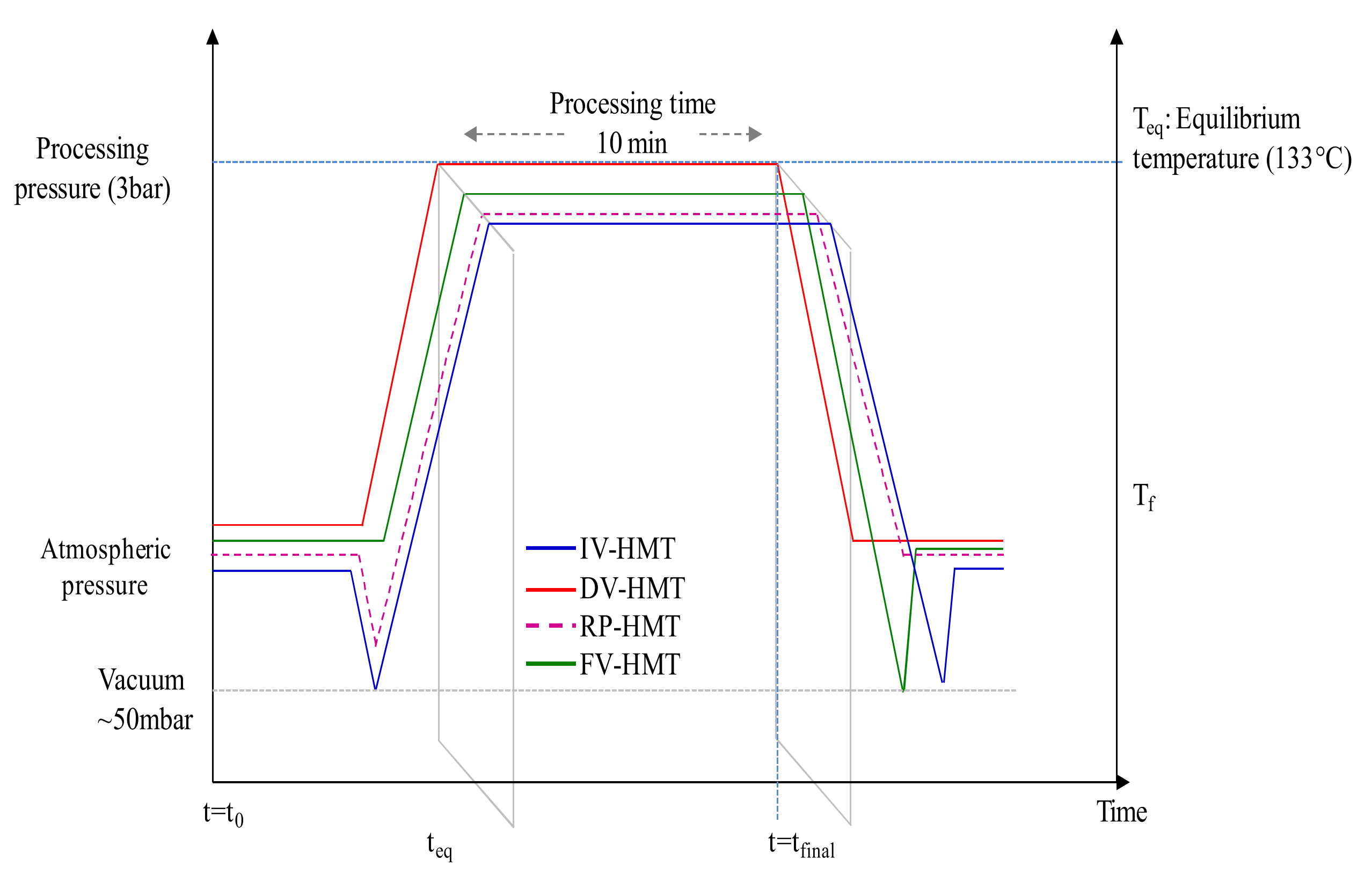
Since the carbohydrates present in wood are not readily accessible to enzymes, pretreatment plays a crucial role in disrupting the lignocellulosic matrix, thus rendering polysaccharides available to enzymatic actions. Widely investigated in the literature as a non-chemical process, steam explosion (SE) is considered by several authors to be an environmentally friendly technique [79,80,81]. It is recognized as one of the most effective PTs for breaking the crystalline structure of lignocellulose through chemical effects and mechanical shear, which is induced by sudden explosive decompression [82,83]. Other variants of SE have been studied, such as heat–moisture treatment (HMT) which is based on the hydrothermal effect obtained through the simultaneous action of heat (>165 °C) and moisture (<40%) of direct steaming for a defined duration [84]. Summarized in Figure 3, these variants correspond to four thermomechanical processes with or without an initial vacuum, and differing in the pressure towards which the decompression occurs [85]. During direct vapor (DV-HM) and final vapor (FV-HMT) heat–moisture treatments, the reactor is subjected to saturated steam injection, gradually increasing from atmospheric to the designated processing steam pressure (SP). As the SP is injected, the biomass undergoes initial heating from ambient temperature to reach a steam equilibrium temperature. In contrast, the reduced pressurized (RP-HMT) [86] and intensive vacuum (IV-HMT) heat–moisture treatments follow a different approach. The reactor is first subjected to a vacuum pressure of 50 mbar before the introduction of saturated steam. The presence of an initial vacuum in the latter two processes intensifies the diffusion of steam into the biomass due to the decrease in air resistance [87], and serves to generate a larger amount of condensed steam than in the case of DV or FV-HMT. For the same SP, an equivalent volume of steam replaces the volume of air. It should be noted that the IV-HMT and FV-HMT processes are distinguished by an abrupt decompression to vacuum at the end of the treatment, instead of atmospheric pressure, as in the DV-HMT and RP-HMT processes. The sudden pressure drop is accompanied by an abrupt self-evaporation of water from the LCB, causing rapid cooling. The mechanical constraints and intense shear caused by the sudden explosion disrupt the fibers in the biomass structure; some of the mechanical energy is converted into internal energy. It is important to highlight that the DV-HMT process corresponds to the SE process, mainly because it does not feature any drop towards vacuum pressure, neither before the rise to the desired pretreatment pressure nor during the final decompression phase.

Figure 3.
Schematic diagram of the typical pressure time of four HMT processes [85].
In research carried out by Semhaoui et al. [30], the impact of IV-HMT PT conditions on hemp hurds was investigated using the combined severity factor (Equation (1)). The results revealed that a CSF of 1.2 was favorable, not only for hemicellulose removal, but also for enhancing enzymatic hydrolysis, allowing around an 81.4% conversion yield. SE, on the other hand, was used as a PT of Eucalyptus wood [59,88,89,90], poplar wood [82,91], hornbeam wood [92], Aucoumea wood [93], aspen wood [94], and spruce wood [82,95]. Chemical catalysts are sometimes used prior to SE and HMT to improve hydrolysis, and to reduce the treatment time and energy costs [96]. Before SE PT of the aforementioned substrates, impregnation using NaOH [59,94], H2SO4 [26,82,93], or water [82,91] was performed, and the notion of PT severity is discussed in those studies regardless of the presence or absence of the impregnation step [26,82,88,89,92,93,94,95].
The PT temperature plays an elemental role in biomass delignification. It has been reported that temperatures that are too low do not allow an enhanced enzyme accessibility, while temperatures that are too high induce significant degradation of both lignin and sugars, as well as the formation of inhibitors [97,98]. Pielhop et al. [95], who performed SE on softwood, concluded that a temperature as high as 210 °C was not severe enough to change the structure of spruce wood chips. Such a severe pretreatment is fully understandable when treating large-sized particles (30 mm) without prior impregnation or the use of a catalyst. The study also revealed the significance of the pressure difference during the explosion, specifically between the processing pressure and atmospheric pressure. It concluded that a minimum pressure difference of 5 bar should be applied to achieve a notable impact on enzymatic digestibility.
Using a catalyst prior to SE is very important, as it enables the initial solubilization of lignin and hemicellulose. This, in turn, reduces energy costs by allowing for less severe treatment conditions. Barbanera et al. [92] obtained higher sugar mass recovery from hornbeam wood pretreated with SE at 200 °C for 13.75 min, corresponding to a severity of R0 = 4.08 when preceded by an impregnation step. Moreover, high temperatures increase the concentration of hydronium (H3O+) ions in water, which allows the latter to act as a catalyst and to solubilize hemicelluloses [99]. This could explain the use of water as a more environmentally friendly catalyst in the study conducted by He et al. [82]. Nevertheless, this catalyst could obviously modify the initial moisture content of the biomass [91]. In this sense, Cullis et al. [100] investigated the effect of the moisture content and chip size of SE-pretreated Douglas fir wood, and observed that a higher moisture content (30%) and smaller chip size rendered the substrates more reactive in the subsequent process. Additionally, the greatest lignolytic activity and highest lignin degradation in biologically delignified cotton stalks occurred at a considerably high initial moisture content of 75% [101], just as in the work of Pažitný et al., who pretreated their poplar wood at an initial moisture content of 85%. Conversely, increasing the initial moisture content has been shown to increase the specific energy requirement of a pretreatment [102], which encourages drying before PT. Based on this observation, particle size and initial moisture content acted antagonistically in these studies; when moisture content was at 85%, the particle size was 0.7 mm [91], while when moisture content was 75%, the particle size was 5 mm [101]. Alternatively, it was observed that greater severity resulted in greater mass loss, attributed to hemicellulose degradation, when using H2SO4 as a catalyst [93] (40% wood solubilization for R0 = 4.1, i.e., T = 210 °C, t = 7.5 min, 0.5% H2SO4). What is particularly noteworthy is that samples pretreated at R0 = 3.6 (210 °C, 2.5 min, 0.5% H2SO4) and R0 = 2.9 (170 °C, 5 min, 0.25% H2SO4) exhibited similar Klason lignin, glucose, and xylose contents in the solid fraction. This aspect results in environmental and economic advantages, as it allows for significant cost reduction. The run at 190 °C, performed for 5 min on wood impregnated with 0.5% H2SO4, showed the second most reduction in Klason lignin and xylose contents, while maintaining a high glucose content.
On another note, the experiments led by Rochón et al. [59], in which impregnated and non-impregnated Eucalyptus sawdust samples were treated with SE, revealed that the latter yielded greater outcomes in the subsequent process. Referring to the structure of the substrates’ polymers, it is important to consider the degree of acetylation of the substrate, i.e., the ratio between total acetyl group content and the total number of monomers that can carry them [103]. Studies have described a negative correlation between the degree of acetylation and hydrolysis efficiency [104,105]. Hence, deacetylation could be a key to enhancing the process. While Singh et al. [106] reported that alkalis effectively remove acetyl groups from hemicellulose, Rochón et al. showed that 7–9% of xylans were still detectable in NaOH-impregnated samples, and were undetectable in the non-impregnated solids. Xylose, xylo-oligosaccharides, and acetic acid were detected in the liquid fraction post-PT, in addition to furfural and lignin fragments. Moreover, three washing stages were required to rid the solid fractions of any inhibitors. Mihiretu et al. [94] reached the conclusion that the omission of a catalyst resulted in higher levels of xylan degradation, leading to increased formation of furfural. In contrast, when a 5% alkaline catalyst was used, furfural was not detectable. All of this indicates that deacetylated biomass positively influences the overall process. However, Bay et al. [53] reported that the removal of those acetyl groups with an alkaline PT will limit enzyme accessibility and partially depolymerize glucose, which then highlights the importance of further understanding the influence of xylose acetylation on the efficiency of the bioconversion process. Finally, when it comes to the PT’s duration, coupling either high temperature and short treatment time (e.g., 270 °C, 1 min) or low temperature and long duration time (e.g., 190 °C—10 min) has been highlighted as a way to effectively solubilize hemicelluloses [26,107]. Generally, treatment time is correlated to the amount of product degradation, which should be minimized in such processes [108].
Additionally, since SE can be performed under diverse conditions, it is comprehensible that sugar degradation depends on the severity of the process. Jacquet et al. [109] performed SE at different severities on bleached cellulose, delignified and free from hemicelluloses, obtained from wood pulp. The purpose of the study was mainly to investigate the effect of SE on the cellulose composition and structure. The findings revealed that cellulose undergoes thermal degradation when the severities exceeded 5.2 (i.e., T ≥ 248.4 °C, P ≥ 4.03, and t = 2–5 min). In parallel, Auxenfans et al. [110] performed SE at mild severities (R0 = 2–2.8) on three types of lignocellulosic biomass: Miscanthus, poplar, and wheat straw. Their results demonstrated an increase in glucose content in all samples treated at all severities, which confirms the observations of Jacquet et al. [109]. Nevertheless, as mentioned by Semhaoui [26], the inclusion of an acid catalyst such as H2SO4 can help mitigate cellulose loss following SE. Using a concentration of H2SO4 exceeding 3% w/v has been found to result in sugar loss. This observation further supports the notion that the preservation or degradation of cellulose is influenced by the severity of the pretreatment.
Typically, physical pretreatments aim to increase the specific surface area of lignocellulosic biomasses. To reach this goal, mechanical comminution processes were used to disperse massive lignocellulosic material into small pieces. Milling can significantly reduce the particle size and the degree of crystallinity of lignocellulosic materials, and consequently improve enzymatic hydrolysis. Alas, a high energy input is necessary to achieve the particle size required to reach a high enzymatic hydrolysis yield. In this respect, steam explosion is considered a cost-effective process. In a recent review on the steam explosion of lignocellulosic biomass, Yu et al. [111] emphasized that this process effectively enhances the structure of biomass by increasing its specific surface area and porosity. The authors concluded that steam explosion, being universally applicable and industrially scalable, holds tremendous potential as a key procedure in the bioenergy industry.
2.2. Chemical Pretreatments
Chemical PTs involve the use of organic or inorganic compounds that disrupt the recalcitrant structure of lignocellulosic substrates, and that reduce lignin content through interaction with the bonds between the three main constituents of the biomasses [112,113]. Over the years, various chemical PTs have been tested on woody lignocellulosic biomasses. For instance, five different chemical solvents were used for the PT of poplar wood (hardwood), pine wood (softwood), and rice straw (herbaceous) [53]. In this study, the samples were subjected to distilled water (180 °C, 30 min), dilute sulfuric acid (H2SO4, 0.5% w/w H2SO4/H2O), concentrated phosphoric acid (H3PO4, 85% w/w H3PO4/H2O), sodium hydroxide (NaOH, 2 M), and sodium carbonate (Na2CO3, 0.5 M). The delignification of the wood substrates was relatively mild, but notable variations were observed in the reaction of poplar, pine, and corn stalk to the different chemical agents. Poplar wood exhibited mild delignification in all PT conditions except autohydrolysis, whereas pine wood showed mild delignification only after autohydrolysis and cold NaOH PT. Wan et al. [68] tested another solvent, a phosphoric acid + hydrogen peroxide (PHP) mixture, on poplar sawdust, pine sawdust, and corn stalk. The PHP mixture showed more efficiency in the deconstruction of poplar rather than pine, successfully delignifying poplar at a rate of 83.59%. Similarly, studies carried out by Zhao et al. [114] and Ajayo et al. [115] employed PHP PT for the delignification of furniture boards and paper mulberry wood, respectively. In Zhao et al.’s study, 78.9–92.1% of the recovered cellulose was retrieved from the cellulose-rich fraction, which is a higher recovery rate than that reported by Wan et al. (74.9% for poplar wood). The PHP PT applied to paper mulberry wood in the works of Ajayo et al. resulted in a cellulose-rich substrate, and revealed that under optimal conditions defined by the response surface methodology, 97% delignification can be achieved. S/G ratios can account for these results; Santos et al. [116] determined that the significant differences in S and G subunit contents between wood species were strongly correlated with delignification rate. Indeed, as the S subunit content increases, the condensed lignin structure is reduced. Regarding pine wood, Normark et al. [117] estimated the S/G ratios of different fractions of pine wood (bark, heartwood, etc.), and the results were all relatively low (S/G ratio < 0.1), which may explain why pine is not as reactive to PTs as poplar or herbaceous biomasses. Another common element in the four above-mentioned studies is the use of H3PO4. This acid is a recurrent agent employed in the PT of lignocellulosic biomass, namely poplar wood [114,118,119]. It is non-toxic, inexpensive, and allows dissolution of cellulose at lower temperatures [120], which helps in reducing PT costs. Hydrogen peroxide (H2O2), however, when combined with an alkaline agent such as NaOH or aqueous ammonia, has effectively shown enhancement of substrate digestibility following the PT of corn stover [121], sugarcane bagasse [122], oil palm trunk [123], and rice husks [124]. This is because, under alkaline conditions, H2O2 breaks down and forms intermediates such as hydroxyl radicals, then forms water and oxygen. Part of the latter plays a role in lignin degradation and the production of oxidized lignin products [125]. Alkaline hydrogen peroxide is also considered more environmentally friendly than alkaline PT, as it requires lower PT temperatures and less water for the washing phase [123].
On another note, a mixture of fir sawdust, spruce sawdust, Sylvester pine sawdust, and Douglas fir sawdust was subjected to an optimized microwave-assisted organosolv PT [126]. Generally, this PT aims to solubilize lignin for the enhancement of enzymatic hydrolysis and fermentation [16,127,128]. The PT allowed for a 50% delignification rate and a preservation of 82% of initial cellulose. It is interesting to note that Ethanol Organosolv (EOS) PTs, which are based on the solubilization of lignin in organic solvent (ethanol in this case), can lead to the repolymerization of lignin [127,129,130], which could explain the results related to the extent of delignification. Likewise, olive wood, among other olive waste, was also subjected to organosolv, acid-catalyzed organosolv, and liquid hot water (LHW) PTs [131]. The compositional results reveal the inefficiency of organsolv and acid-catalyzed organosolv in the delignification of olive wood and other olive wastes, as the lignin contents were all greater than the ones noted for untreated samples. It could be possible that the lignin composition, as well as the formation of new lignin molecules following organosolv and acid-catalyzed PTs, led to the increase in lignin content. A study carried out on olive tree pruning biomass has demonstrated that a high lignin content of 25% along with a low S/G ratio and the recondensation of lignin underline the biomass’ low reactivity to PT [132]. However, the results proved the PT’s efficiency in solubilizing hemicellulose, which in turn enhances enzymatic hydrolysis. A hybrid organosolv–steam explosion PT was performed on spruce bark chips [133]. The highest delignification and hemicellulose solubilization were observed for the following conditions: t = 30 min, ethanol = 52% v/v E/W, 1% H2SO4. These parameters have resulted in almost 80% lignin removal and about 90% hemicellulose solubilization. It is relevant to underline that when studying the effect of PT time alone in the steam explosion reactor, formation of pseudo-lignin can be observed when the duration increases to 60 min. Since pseudo-lignin is formed from carbohydrates, especially under severe PT conditions [134,135,136], it can be deduced that a PT performed at 200 °C using 65% v/v E/W and lasting for 60 min is severe enough to induce the formation of pseudo-lignin. Nonetheless, an increase in ethanol concentration induces higher water chemical activity, thus leading to increased hemicellulose degradation and greater delignification. It has previously been described that hemicellulose is soluble in water at temperatures greater than 180 °C [137], but can also be precipitated by ethanol, usually after alkaline extraction [138]. Therefore, it is possible that the combination of high temperature for steam explosion PT and ethanol use for organosolv PT has led to enhanced and increased removal of both hemicellulose and lignin.
Alkaline PT using NaOH was performed on five species of poplar [50] as well as four hemp varieties [139]. All poplar species had glucan contents ranging between 32 and 40%, xylan ranging between 12 and 15%, and lignin contents varying between 21 and 26.5%. In contrast, the four hemp varieties had considerably more cellulose (46.5–51%) and hemicellulose (27.5–32%) than the poplar species, yet a lower lignin content (14.5–17%). The concentration of NaOH was identical in both studies (2%), but the PT time of hemp [139] was almost twice the time dedicated to poplar PT [50]. Additionally, the particle size used for hemp hurds was 2–4 mm, which is much greater than the particle size chosen for the poplar species (0.25–0.84 mm). Alkaline PT of the poplar species resulted in a maximum lignin removal of 52.3%, and the lignin content of the samples decreased or remained similar after PT. On the other hand, the alkaline PT adopted by Wawro et al. [139] resulted in slight delignification in only one of the four hemp varieties, whereas the opposite was observed in the remaining three, which is explained by the presence of strong C-C linkages and functional groups. Therefore, while one substrate may exhibit a lower lignin content compared to another, it’s important to consider that the severity of both PTs was different, which could influence the efficiency of lignin removal. Hemp was also chemically pretreated using 1% H2SO4 [140]. Interestingly, the study included a deacetylation step using 1% NaOH prior to dilute acid PT. This procedure has aimed to decrease acetate inhibition, as acetate is one of the most important inhibitors of lignocellulosic hydrolysates [141]. The use of 1% NaOH allowed for the least glucose and xylose loss, and had a positive impact on the subsequent process.
Another novel PT using p-toluenesulfonic acid/pentanol (TsOH/pentanol) was performed on poplar chips [142]. Moreover, the PT efficiency was boosted by adding different concentrations of mannitol (MT), which is a carbocation scavenger (i.e., an additive that reacts with carbocations formed within lignin under acidic conditions). These carbocations supposedly cause lignin repolymerization reactions [143]; their removal aims to enhance the PT outcomes. The PT performed by Madadi et al. [142] targeted biomass fractionation of poplar chips and resulted in three fractions: a cellulose-rich fraction, furfural fraction, and a native lignin fraction. The addition of 1–5% MT increased delignification from 70% (without MT) to 92.79% (at 5% MT). Chen et al. [144] have also reported a 90% delignification rate of poplar wood chips treated using only TsOH solutions of various concentrations. With this in mind, further research is highly necessary to explore the impact of mannitol assistance on TsOH pretreatment, particularly in the context of a process that involves fermentation. It is worth noting that neither of the aforementioned two studies have conducted this specific step, emphasizing the importance of filling this knowledge gap.
2.3. Thermal/Thermochemical Pretreatments
Thermal and thermochemical PTs have been used for the production of bioethanol from hemp hurds [145], aspen wood [67], pine wood [67,146,147], beech chips [146], teak wood residues [148], sal sawdust [149], and acacia wood [150,151]. The work of Dessie et al. [53] described three PTs: autohydrolysis, thermal hydrolysis (121 °C), and a thermochemical PT that consisted of an oxalic acid-assisted thermal hydrolysis at 121 °C. Thermochemical PTs of pine and aspen wood [67] have further confirmed pine’s resistance to PT [117] due to its high lignin content, just as these studies have determined that temperatures above 200 °C are essential for the efficient PT of aspen wood. In addition, Wang et al. [64] reported that untreated aspen wood had an S/G ratio of 1.68, as opposed to untreated birch wood, in which the S/G ratio was about 3.25, which could indicate why aspen wood requires a more severe PT. On the other hand, various thermochemical PTs tested on pine wood [146] led to different outcomes. While the use of NaCS 40% w/v (NaCS/H2O) or ethanol 60% v/v (E/W) + 1% v/v H2SO4 in combination with the microwave PT resulted in a biomass with the highest cellulose content and lowest lignin content, H2SO4 charred the biomass and hindered further analyses. NaCS is a surfactant that reduces the surface tension of liquids [152]. Surfactants have been proven to positively influence and enhance enzyme hydrolysis (Section 3).
High temperatures (200 °C) were applied by Lee et al. [151] for 5 min with 0.05% H2SO4 in order to obtain a maximum glucose yield. This type of PT was also followed by further mechanical particle size reduction, with the aim of further enhancing enzymatic hydrolysis. Lee et al. [150] also performed a hydrothermal PT, followed by an optimized lime PT to treat acacia wood. The results revealed a widened surface area of acacia wood pretreated hydrothermally. These experiments highlight a form of non-antagonistic effect between both high temperatures and the presence of a chemical agent, which presents a similarity with steam explosion; the chemical catalyst used in steam explosion assists the delignification process as well as reducing energy costs. Chemical assistance of thermal PTs also plays a role in process enhancement as well as energy savings.
Madadi et al. [147] performed three PTs for the delignification of pine sawdust: liquid hot water (thermal), green liquor (Na2CO3 + Na2S; chemical), and (liquid hot water + green liquor) (thermochemical). This study investigated the effect of assisting the thermochemical PT with three carbocation scavengers: syringic acid, 2-naphtol-7-sulfonate (NS), 2-naphtol (2N), and mannitol (MT). The results showed that NS had the most positive influence on mitigating lignin repolymerization, due to its greater hydrophilicity. Less repolymerized lignin increased the porosity of the pine biomass, which consequently increased cellulose accessibility, as well as the sugar and ethanol yields. The study led by Pielhop et al. [143] reached a similar conclusion, wherein the addition of 2-naphtol improved the cellulose digestibility of different hardwood and softwood substrates by 113%. On another note, Domínguez et al. [153] conducted a serial two-stage autohydrolysis as a PT of Paulownia wood. This approach serves as a viable and environmentally friendly alternative to using autohydrolysis solely as a first-stage delignification strategy. The biomass, which consisted of 39.7 g glucan/100 g raw material and of 21.8 g Klason lignin/100 g raw material, was subjected to two PTs: single-stage and two-stage autohydrolysis. After delignification, it was subjected to EH and fermentation. Autohydrolysis offers several environmental advantages; it eliminates the need for additional chemicals relying solely on water, has a minimal corrosive effect on equipment, enhances the EH rate, and lastly, preserves cellulose [153,154]. This two-step PT performed by Domínguez et al. allowed an 85% recovery of xylan in the form of xylooligosaccharides at an R0 = 4.08 within the first autohydrolysis step, which then enhanced the enzymatic digestibility of the second autohydrolysis. It is important to note that while the process was performed at temperatures ranging between 204 and 222 °C, it still provides other environmental benefits.
Finally, it should be highlighted that PTs will not be entirely green. Every PT has its own requirements and outcomes, and all of these come at specific costs. However, when comparing PTs, thermomechanical PTs present an advantage, not only in allowing for important bioethanol production, but also in reducing the generation of chemical waste. Additionally, Semhaoui [26] underlined that the sudden decompression in SE stops degradation reactions, due to the sudden decrease in temperature, which also presents an advantage under mild PT conditions.
3. Enzymatic Hydrolysis and Fermentation of Woody Biomass
Enzymatic hydrolysis (EH) is the step of the bioconversion process during which fermentable sugar monomers are liberated from the biomasses’ structural carbohydrates, cellulose and hemicellulose [155,156]. Cellulases target cellulose and cleave β-1,4-D-glucan linkages in the polymer, while hemicellulases breakdown galactan, xylan, mannan, and araban, having similar activities to cellulases, because the same linkages can be found in the hemicellulose structure [157,158,159]. One type of hemicellulase, known as endoxylanase, targets the β-d xylano pyranosyl bonds within xylan to release xylo-oligosaccharides [160,161]. Cellulose–glucose conversion generally takes place at temperatures of 40–50 °C and a pH of around 4.8, yet the efficiency of the process relies on other influential factors, namely lignin removal, solubilization of hemicellulose, acetylation of hemicellulose, hydrolysis duration, enzyme loading, cellulose crystallinity, the presence of surfactants, biomass particle size, pore volume, and accessible surface area [159,162,163]. This confirms, once again that PT operating conditions have a considerable impact on the subsequent process. Mussatto et al. [164] have evaluated the influence of enzyme loading, substrate concentration, and agitation speed on the outcomes of EH. While agitation speed had the lowest impact, the results showed that enzyme loading had the highest impact. Solid loading also plays an important role in the efficiency of EH; loadings less than 6–10% w/v make water more available, thus decreasing the reaction–diffusion limitations. The only drawback of low-solids EH is the increased production cost and energy demand [165,166]. Increasing substrate loading has its own disadvantages too, which include increased viscosity and the restraint of heat-mass transfers [167].
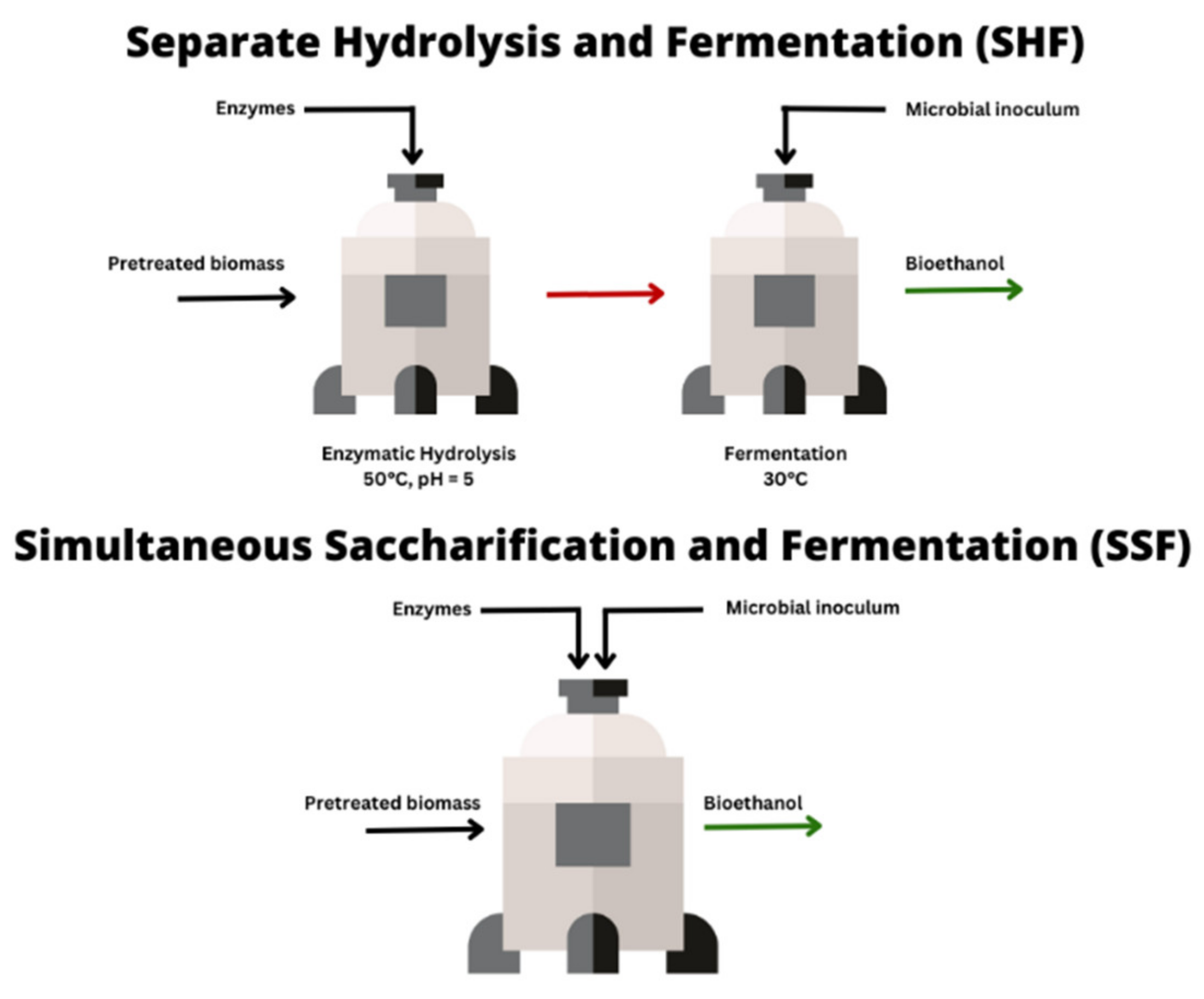
When the bioconversion process comprises both EH and fermentation, the former can either be conducted separately from the latter step (separate hydrolysis and fermentation; SHF) or together in one step (simultaneous saccharification and fermentation; SSF). Generally, studies have reported that SSF is more efficient than EH followed by fermentation, because it limits inhibitions that can be observed during SHF and it helps to reduce costs [168,169,170] (Figure 4).

Figure 4.
Comparing SHF and SSF.
When it comes to fermentation, Saccharomyces cerevisiae is the most commonly used microorganism for the conversion of glucose into ethanol. However, a limitation of S. cerevisiae is its inability to ferment pentoses, which are released during the EH of hemicellulose [171]. This challenge has prompted the development of genetically engineered strains capable of metabolizing these sugars, thereby enabling more efficient bioconversion processes.
3.1. Enzymatic Hydrolysis of Woody Biomass after Thermomechanical PT
Thermo-mechanically treated eucalyptus sawdust [59,88,90], poplar wood [91], spruce wood [95], Aucoumea wood [93], hornbeam wood [92], aspen wood [94], and hemp hurds [26] were subjected to enzymatic hydrolysis. The severity of the PT conditions had various effects on EH across different studies. For instance, when NaOH-impregnated and non-impregnated eucalyptus sawdust [59] were hydrolyzed, the results showed that the enzymatic digestibility of the non-impregnated solids attained an efficiency of 96% at 200 °C after optimization, producing 134 g/L of glucose. Interestingly, in this particular case, the impregnation step with a catalyst solution did not promote enzymatic hydrolysis, which contradicts previous findings and warrants further investigation. Another example would be the results obtained in the works of Romaní et al. on SE-treated eucalyptus wood [88], in which a conversion efficiency of 94.5% following PT optimization (195 °C, 5.87 min) was reached, and the resulting (xylose + oligosaccharides) concentration was about 18.1 g/100 g oven-dried wood. On the other hand, the PT of spruce wood [95] allowed the liberation of both glucose and mannose, which increased with PTs even at lower severities, and without any degradation of pentoses. This indicates that sugar molecules are preserved at specific severities, allowing for their recovery through EH, whereas higher PT severities may induce their degradation. However, and as already discussed, Pielhop et al. concluded that a ΔP > 5 bar is required for enhanced enzymatic digestibility, which was confirmed by the highest total sugar yield obtained (62%) at a PT severity of 4.7 (without catalyst). Zhou et al. [172] proved that increased PT severity influences the enzyme digestibility of corn stover, but not necessarily positively for all hydrolysis durations. They reported that their highest glucose yield (89.2%) was obtained at a severity of 3.716 (210 °C, 3 min), while just increasing the time from 3 to 10 min (severity of 4.239) led to a degradation of cellulose, thus slightly lowering the glucose yield. While the dependency of sugar yield and inhibitor production on temperature was observed, the correlation with PT severity was determined by the nature or the composition of the biomasses. Generally exhibiting a lower recalcitrance than woods, corn stover and other herbaceous biomasses are more sensitive to PT conditions, and have a higher risk of sugar degradation. Nitsos et al. [173] pretreated poplar residues and pine sawdust chemically, and tested several PT severities. Their results revealed low concentrations of major byproducts for the whole series of severities (R0 = 3.8–4.1), which further confirms the influence of the biomass’s nature. In contrast, Besserer et al. [93] concluded that lower severities (170–190 °C, 2.5–5 min, 0.25–0.5% H2SO4) resulted in low glucose yields, while an increase to 210 °C for 5 min with the addition of 0.25% H2SO4 resulted in 23.4 g glucose/100 g DW of wood. Notably, the washing step practiced by Besserer et al. provided an advantage by reducing the inhibitors, which consequently impacted the EH outcomes. In contrast, Mihiretu et al. [94] achieved their highest glucose yield at 204 °C over 10 min (approximately 82 g/L of glucose), with alkaline impregnation (5% w/w NaOH). Both furfural and HMF were detectable at higher severities (200 °C, 15 min, 5% w/w NaOH), but were significantly higher at lower severity (190 °C, 10 min, no alkali agent). Further investigation on the correlation between sugar yield, inhibitor formation, and PT severity is mandatory.
On another note, the works of Pažitný et al. [91] have highlighted that SE-treated poplar heartwood yielded the highest concentrations of both glucose and xylose (90 g/L after 72 h), and of glucose alone (70.4 g/L at 48 h). Studies performed on both larch wood [174] and pine wood [175] have both stated that sapwood possesses a higher cellulose content than heartwood, whereas heartwood has a higher lignin content. A similar distribution was also underlined in poplar tree parts [91]. While one might anticipate that the elevated lignin content in poplar heartwood would lead to decreased glucose yields, it is possible that the lower lignin content in sapwood potentially facilitated cellulose degradation. This is evidenced by the maximum glucose yield obtained from poplar sapwood, which reached 65 g/L. Finally, the study of Semhaoui [26] showed that the use of 0.66% H2SO4 significantly influenced the production of glucose and xylose. With 2% H2SO4, the maximum glucose and xylose concentrations obtained from 10 g of thermomechanically treated hemp hurds were 6.36 g/L and 2.1 g/L, respectively. The results of this study highlighted that non-catalyzed thermomechanical treatment (IV-HMT, 165 °C, 30 min) does not favor inhibitor production, the detected concentrations of which were negligible. The use of an acidic catalyst, as well as the increase in acid loading, led to greater formation of inhibitors. The project of Semhaoui [90] has also emphasized that the use of an H2SO4 catalyst as an impregnation solution resulted in an overall greater reduction in sugar concentrations than using NaOH. At optimal PT conditions, 0.8% H2SO4 enabled a 90% yield in reducing sugars versus a reduction of 83.2% for alkaline-impregnated hemp hurds. These results suggest that NaOH not only solubilized lignin and hemicellulose, but also induced the formation of other particles that hindered EH. Furthermore, a reduction in specific surface area was noted for NaOH-impregnated hemp hurds. This phenomenon can be attributed to the likelihood of lignin condensation occurring in the alkaline environment at elevated temperatures, thereby obstructing access to open pores.
Last but not least, another attempt at process optimization was performed by Schneider et al. [90]. After SE of eucalyptus wood, a biological post-PT using manufactured laccases from a strain of the saprophytic fungus Marasmiellus palmivorus [176] was conducted. The biological treatment aimed to detoxify the treated wood from the inhibitors produced, and was able to decrease the free phenolic compounds content found in soluble fraction by 70%. However, ethanol yield was approximately 10% greater when laccase treatment was performed after EH rather than after PT. Pretreated biomass detoxification has already been suggested as a promising additional step for an enhanced process; both biological and chemical PTs can be performed on LCB hydrolysates and slurries [177,178,179].
3.2. Enzymatic Hydrolysis of Woody Biomass after Chemical PT
Chemically pretreated pine and poplar wood [53], sawdust mixture [126], hemp [126], spruce [133], olive wastes [131], and paper mulberry wood [115] were subjected to an enzymatic hydrolysis step separate from the fermentation phase, while chemically pretreated poplar wood, pine wood [50,68], and furniture boards [114] were subjected to SSF. In the study led by Bay et al. [53], the structural analyses confirmed that amorphous parts are broken down by enzymes before the crystalline parts [180,181,182]. This is due to the fact that glycosidic bonds exist within the crystalline fraction and are less reactive to hydrolysis [183]. The results also revealed that the PT of wood samples with H3PO4 and cold NaOH had the lowest α (1510/900) values (i.e., the lowest lignin-to-cellulose ratios), which indicates the greatest delignification. It can also be concluded that among all five PTs, H3PO4 and cold NaOH PTs allowed for the greatest glucose production from pretreated poplar and pine (493.3 g/kg substrate (H3PO4) and 459.2 g/kg substrate (cold NaOH) vs. 446 g/kg substrate (H3PO4) and 340.5 g/kg substrate (cold NaOH), respectively). Similarly, PHP PT of furniture boards [114] further draws attention to the efficiency of H3PO4 in the enhancement of the bioconversion process, as 208–241 g glucose/kg of substrate was liberated from the hydrolysis of fiberboard, chipboard, and blockboard, using a high enzymatic loading of 20 mg protein/g cellulose. The experiments led by Zhao et al. [114] also revealed that PHP PT decreased both crystal size and crystallinity index (CrI), through the dissolution and swelling of cellulose, which is originally crystalline [184]. This reduction in crystallinity has been proven to increase surface area, thus enhancing enzymatic hydrolysis [185,186]. Furthermore, the effect of PHP PT on the saccharification of woody LCB was showcased in the experiments of Wan et al. [68], in which the cellulose-to-glucose conversion was 40.4% for poplar and 27.5% for pine within the first 4 h. Wan et al. [68] found that an almost complete conversion could be achieved within 48 h, while Zhao et al. [181] reported that a 72 h incubation was necessary to achieve the maximum conversion yield. This could be explained by the difference in composition of the furniture boards compared to poplar and pine, as well as by the crystallinity post-PT of the substrates. Although PHP treatment reduces the CrI, some crystallinity could remain in the pretreated substrates, which consequently affect the rate of hydrolysis.
Alternatively, the preliminary hydrolysis tests of an organosolv-treated sawdust mixture [126] revealed the influence of the substrate’s particle size on the overall process. Oven-dried particles greater than 0.5 mm produced less glucose (1.6 g/L) and had a slower cellulose-to-glucose conversion rate (12.4% within 8 days), as opposed to larger particles, which produced 3.1 g/L of glucose and had a conversion rate of 24%. Zhao et al. [181] concluded that smaller particle sizes are expected to enhance enzyme digestibility, but this is not the case for particles smaller than 350–590 µm, with which no further significant enhancement was observed.
As previously mentioned, EH is influenced by multiple factors. Alio et al. [126] tackled other influential parameters, which were solid and enzyme loading. The results demonstrated that under the chosen experimental conditions, an increase in enzyme loading (from 50 FPU/g to 70 FPU/g) for the same solid loading of 1.5% m/v improved the digestibility, as did an increase in solid loading (from 1.5% m/v to 5.2% m/v) for a fixed enzyme loading of 50 FPU/g. Moreover, a particularly high glucose yield of 443 mg glucose/g of pretreated wood was obtained in the study of Ajayo et al. [115] after RSM optimization (H3PO4 71.3% w/w + H2O2 4.84% w/w, 34.7 °C, 3.3 h). The study underlines that 100% hydrolysis could be achieved at a low H3PO4 fraction (60%) and high temperature (50 °C), while a higher acid fraction (80%) and lower-level temperature (30 °C) decreased hydrolysis efficiency to 69%. The study also showed the interaction between temperature and time, and its influence on hydrolysis yield, which reached 100% when both parameters were increased (41 °C, 2 h). Han et al. [187] deduced that while prolonged time increased hydrolysis efficiently, PT durations exceeding 1.5 h led to a decrease in efficiency due to the possible degradation of the sugar polymers. It is worth mentioning that the PT duration was also influential in this case, as the NaOH PT was performed at 121 °C, with varying NaOH concentrations (0.25–1.5% w/v). From here, the consideration of PT severity remains critical for the preservation of the sugar polymers. Finally, the influence of surfactants on enzymatic hydrolysis was investigated in pretreated spruce wood [188], and more recently in pretreated sugarcane bagasse, cypress sawdust, African coral wood [189], and bamboo [190]. These studies have tested the use of diverse surfactants, all of which had a positive effect on enzymatic hydrolysis. Nonetheless, since numerous previously used surfactants are not particularly environmentally friendly, the use of biosurfactants has been encouraged [191,192] as a more sustainable alternative.
3.3. Enzymatic Hydrolysis of Woody Biomass after Thermal/Thermochemical PT
Enzymatic hydrolysis was performed following the PT of aspen wood [67], pine wood [67,146], beech chips [146], teak wood [148], hemp hurds [145], sal sawdust [149], and acacia wood [151]. The double enzymatic hydrolysis performed by Sjulander et al. did not exhibit high efficiencies (overall glucose yield in aspen wood = 29.19% vs. 5.19% for pine wood) due to the reported repolymerization of lignin, as well as the possible inhibition of cellulases by mannan polysaccharides from pine wood. In contrast, the teak wood EH [148] was followed by a detoxification step via two different methods: chemical, using Ca(OH)2, and biological, using laccases (as observed in the study of Schneider et al. [90]). After 36 h, the obtained glucose and xylose concentrations were 60 g/L and 20 g/L, respectively. While it was reported that the biological detoxification did not influence the fermentable sugars’ concentration, it did lead to a 40% decrease in soluble phenolic compounds, which act as inhibitors. Lime detoxification negatively influenced the sugar concentrations, reducing the glucose concentration by 7% and xylose concentration by 27%. This might be caused by the reaction between Ca(OH)2 and the cleaved glucose molecules, as it produces the water-soluble molecule calcium glucosate [193].
Mikulski et al. [146] observed an increased susceptibility of two woody substrates to enzymatic hydrolysis following microwave-assisted PT using two solvents (NaCS and ethanol/H2SO4). The hydrolysis yield of pine chips following the ethanol/H2SO4 PT was 30.54%, as opposed to 56.84% for beech chips using the same solvent. However, the hydrolysis yield of beech chips was slightly greater using 1% NaOH (62.21%). Since cellulose and lignin contents were close in both NaCS-treated and NaOH-treated beech chips, the enhanced enzyme digestibility observed in the latter could be attributed to the lower extractive contents, which have been reported to have a positive effect on enzyme hydrolysis [194].
The work of Dessie et al. [145] took a completely different approach as part of their study. This approach consisted of a “one-pot PT and saccharification”, which finally achieved a maximal reducing sugar concentration of 39.49 g/L. The approach consisted of combining the thermochemically pretreated hemp hurds with mashes from the SSF step performed in the study; the chosen PT was OAA thermochemical PT using 2% oxalic acid. Surprisingly, not only did the greater acid concentration (3%) further reduce sugar concentrations, but it also increased the processing cost and formation of inhibitors. The one-pot PT and saccharification approach was also adopted by Shi et al. [195], and more recently by Sriariyanun et al. [196]. This “one-pot” approach would have been interesting with regard to time and space efficiency, as fewer specific units are required for each step of the bioconversion. However, if the pretreated substrate were not to exhibit successful delignification, or if microbial inhibitions were still present [196], then perhaps the approach would not be of greater efficiency than a classic PT.
Finally, Lee et al. [151] highlighted the efficiency of a mechanical refining post-PT of acacia wood, as well as the addition of soy protein, which increased the efficiency of the hydrolysis to 73.8%. The study explains how soy protein plays a role in enzymatic hydrolysis by preventing the irreversible binding of the enzyme to lignin [197,198].
4. Simultaneous Saccharification and Fermentation of Pretreated Woody Biomass
Diverse techniques have been employed for the fermentation of sugars recovered from wood PT. In addition to SHF and SSF which were previously compared, there exists a strategy known as simultaneous saccharification and co-fermentation (SSCF), which aims to ferment both hexoses and pentoses recovered from the EH step [199]. Another strategy, known as consolidated bioprocessing (CBP), hydrolyses and ferments the pretreated substrate in a single step with the help of a microbial culture that collectively has both abilities [200]. SSCF was employed by Chen et al. [201] for the production of bioethanol from wood dust via SE and supercritical fluid extraction (SFE), and also by Nurdin et al. [202] for the fermentation of chemically treated oil palm empty fruit bunches. CBP, on the other hand, was used for bioethanol production from olive mill wastes [203], in a study in which different fungal strains were tested. The final ethanol concentration (2.47 g/L) was not as significant as studies that have performed SSF or even SHF. However, the resulting ethanol yield (0.84 g ethanol/g treated biomass) was superior to that of SSCF-treated wood dust [201], which was 0.069 g ethanol/g treated biomass (for SFE) and 0.049 g ethanol/g treated biomass (for SE). Besides, CBP has been used for the production of other final products, such as hydrogen [204,205] and lactic acid [206]. Its application for the production of bioethanol is to be further investigated.
Studies that have investigated SSF following PT of woody biomasses are numerous [41,44,50,59,78,84,100,116,124,125,179]. The ethanol titers obtained as a result of SSF, notably Eucalyptus (75.6 g/L and 51 g/L) [59,88], poplar (12.2 g/L to 15.8 g/L and 22.12 g/L) [50,53], hemp (7.5 g/L and 18.9 g/L) [139,140], oil palm and furniture boards (8.1 to 10.4 g/L) [114], underline the efficiency of such a procedure. Ethanol yields varied between 12.3 and 79.4% [68,94].
The ethanol yield obtained from SSF is influenced by several factors, namely temperature, enzyme loading, yeast concentration, and pH [207,208]. Zhang et al. [208] focused on the optimization of the SSF of chemically treated wheat straw, and the results detailed the effect of each parameter. Optimal conditions, which include a high enzyme loading of 35 FPU/g, allowed for mean experimental yields of 37.84 g ethanol/L. Furthermore, the impact of the yeast cultivation procedure on the SSF outcome was studied by Alkasrawi et al. [209]. Yeasts cultivated in different ways influenced SSF. More robust and tolerant yeasts are substantially more advantageous, not only for greater ethanol yields, but also for making use of high solid loadings and recirculating process streams. Zhang et al. [210] compared SSF and SHF processes with regard to the production of bioethanol from raw sweet potato (starchy resource). The authors found that SSF had a shorter reaction time (27 h vs. 33 h for SHF) and a higher productivity (4.76 g/L/h vs. 2.84 g/L/h for SHF), amounting to a greater ethanol yield. Finally, a study on the impact of stirring speed on the glucose and ethanol yields in both SHF and SSF revealed that increasing the stirring speed (200 rpm vs. 150 rpm for SSF) was not more productive, nor was it more productive than a stirring speed of 100 rpm used in SHF [211].
SSF, SHF, and pre-saccharification followed by simultaneous saccharification and fermentation (PSSF) were tested for the hydrolysis of SE-treated Eucalyptus wood [59], with all three processes achieving relatively high ethanol concentrations. Both solid loading and enzyme dose were evaluated. It can be noted that the presence of a large quantity of water-soluble particles negatively influences enzyme diffusion by increasing the viscosity. SSF and PSSF reached similar productivities (2.1 g/Lh and 2 g/Lh, respectively) and close ethanol titers (75 g/L and 70.2 g/L, respectively). Although SSF was the most performant among the three processes, the concept of PSSF is advantageous for processes achieved at high solid loadings; this fact was also confirmed by Tareen et al. [212]. The conditions and results of the studies that have conducted SSF for bioethanol production are summarized in Table 2.

Table 2.
Summary of SSF results of pretreated woody biomass.
On a related note, and since S. cerevisiae cannot ferment pentoses, some studies have chosen to genetically modify it to generate a hexose- and pentose-fermenting strain. The genetic modification of yeasts also aims to render these living organisms more tolerant to inhibitors. A prime example would be the works of Ko et al. [214], in which an engineered strain of S. cerevisiae was able to ferment xylose recovered from treated sugarcane bagasse as well as tolerate acetic acid, which is basically a yeast growth and performance inhibitor. Their engineered strain produced up to 8.9 g/L of ethanol from an original xylose concentration of 20 g/L, in the presence of 4 g/L of acetic acid. The observed mechanisms indicated that the carbon source provided by the fermentation medium was not utilized for cell biomass generation, but rather for ethanol production.
5. Overview of the Bioconversion Processes of Woody Biomass into Bioethanol and Other Products
While wood LCBs are certainly promising for the production of bioethanol, there remains a possibility of valorizing them into value-added products. For instance, in the study led by Wan et al. [68], the PHP PT not only allowed for the production of bioethanol, but also allowed the recovery of water-soluble polysaccharides and lignin nanoparticles. On the other hand, the classical process of bioconversion can be followed with a different final product. For example, Hashemi et al. [215] pretreated birch wood with SE and performed EH, with the final purpose of producing biomethane. In another study, chemically treated birch wood resulted in water-insoluble solids, which in turn underwent EH and fermentation, separately, to finally produce furfural. The solid residue from EH was further fractionated for the product of lignin nanoparticles [216].
It is promising to visualize how the classical steps for the production of bioethanol from LCBs can coincidentally give different products, but also how a slight change in one parameter, such as the microbial inoculum, for example, may lead either to the same or to a totally different end product. As mentioned in Section 3, S. cerevisiae is the most commonly used microorganism for the fermentation of glucose into bioethanol. However, Sierra-Ibarra et al. [148] conducted a study on thermochemically pretreated teak wood that was subjected to enzymatic hydrolysis (at enzyme loading of 15 FPU/g), and ultimately fermented using an ethanologenic strain of E. coli, as it can ferment both hexoses and pentoses. In fact, genetically modified E. coli has also been employed for the production of bioethanol from other LCBs such as chemically pretreated brewery spent grain [217], wheat straw, and corn stover [218], and SE-treated barley straw [219]. Figure 5 summarizes the above-mentioned bioconversion pathways.
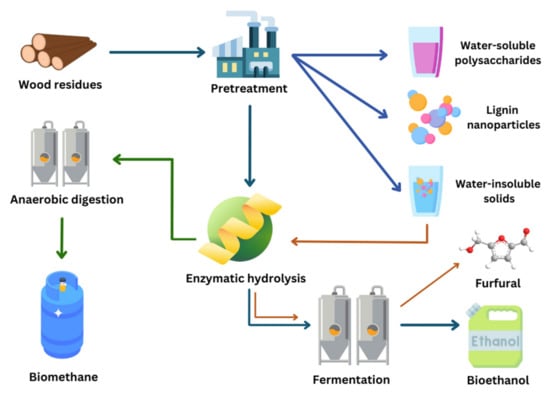
Figure 5.
Complete bioconversion pathways of woody biomass.
6. Energy, Economic, and Environmental Aspects
As mentioned earlier, in the context of a biorefinery, PT is a crucial step in the breaking down of the recalcitrant lignocellulosic structure of woody biomass. The primary objective of a PT is to achieve optimal conversion of cellulose and hemicellulose components into fermentable sugars, including glucose, xylose, arabinose, galactose, and mannose. As a result, the degradation of LCB enhances the rate of chemical reactions and facilitates heat and mass transfer during the subsequent biochemical conversion process. This improved efficiency contributes to the production of high-added-value products, such as biofuels. The overall process is efficient if the delignification is significant, the subsequent formation of inhibitors (furfural and hydroxymethyl furfural) is low, and if the operational cost, enzymes consumed, and amount of waste produced are reduced. The techno-economic assessment of a form of PT depends primarily on the composition of the used feedstock, on the type of PT, on the operating conditions (including temperature, processing time, type and quantity of chemicals), and on other parameters such as raw material grinding. Ab Rasid et al. [220] compared five environmentally friendly PT methods of LCB (ozonolysis, ionic liquids, deep eutectic solvents, organosolv, and SE), considering various criteria including energy consumption. For several LCBs including beech wood, eucalyptus wood, or pine wood, the authors reported that SE was the most effective PT, since it achieved a high delignification rate (up to 98% wt) and did not consume large quantities of chemical reagent, thus reducing environment pollution and cost. The same trends were observed by Baral and Shah [221] in their comparative techno-economic analysis of SE, H2SO4 PT, ammonia fiber explosion (AFEX), and biological PTs of corn stover before its transformation into fermentable sugars. The authors focused their work on the minimization of sugar production cost through the selection of optimal values of the most sensitive input parameters for each PT process. Their results indicated that the most competitive PTs are SE and H2SO4, with 0.43 and 0.42 US $/kg, followed by AFEX, with 0.65 $/kg. The highest sugar production cost was attributed to the biological PT (1.41 US $/kg). The authors postulate that these values could be improved by optimizing/intensifying factors such as feedstock cost, feedstock-to-sugar conversion rate, and enzyme cost. In recent years, enhancing the cost-effectiveness of cellulase and reducing greenhouse gas emissions have attracted global interest from multiple research teams. Malik et al. [222] indicated that cellulase constitutes the primary cost component in the bioethanol production process, irrespective of the selected strategy, be it SHF or SSF. In another context, Kuittinen et al. [223] showed that ethanol and butanol produced from barley straw reduced greenhouse gas emissions by 77.6% and 72.1%, respectively, as compared to the emissions released when produced from fossil fuel gasoline. Bioethanol production from barley straw has an advantage since it is a well-established and mature technology, whereas biobutanol production faces techno-economic challenges that are mainly associated with the low yield and the high recovery cost of butanol. For corn stalk feedstocks, Liu et al. [224] estimated the energy (MJ/kg) required to obtain m (kg) of treated samples (EI) following the ICSE process (instant catapult steam explosion). The use of the “Antoine equation” Equation (2) allows the pressure (P in Pa) to be related to the temperature (T in °C), since the medium is not a pure gas, but rather a mixture of air and steam. The developed equation involves pressure in the vapor generator before and after pressure release (P1 and P2), quantity of vapor (Δn in mol), and ideal gas constant (R in J.mol−1K−1):
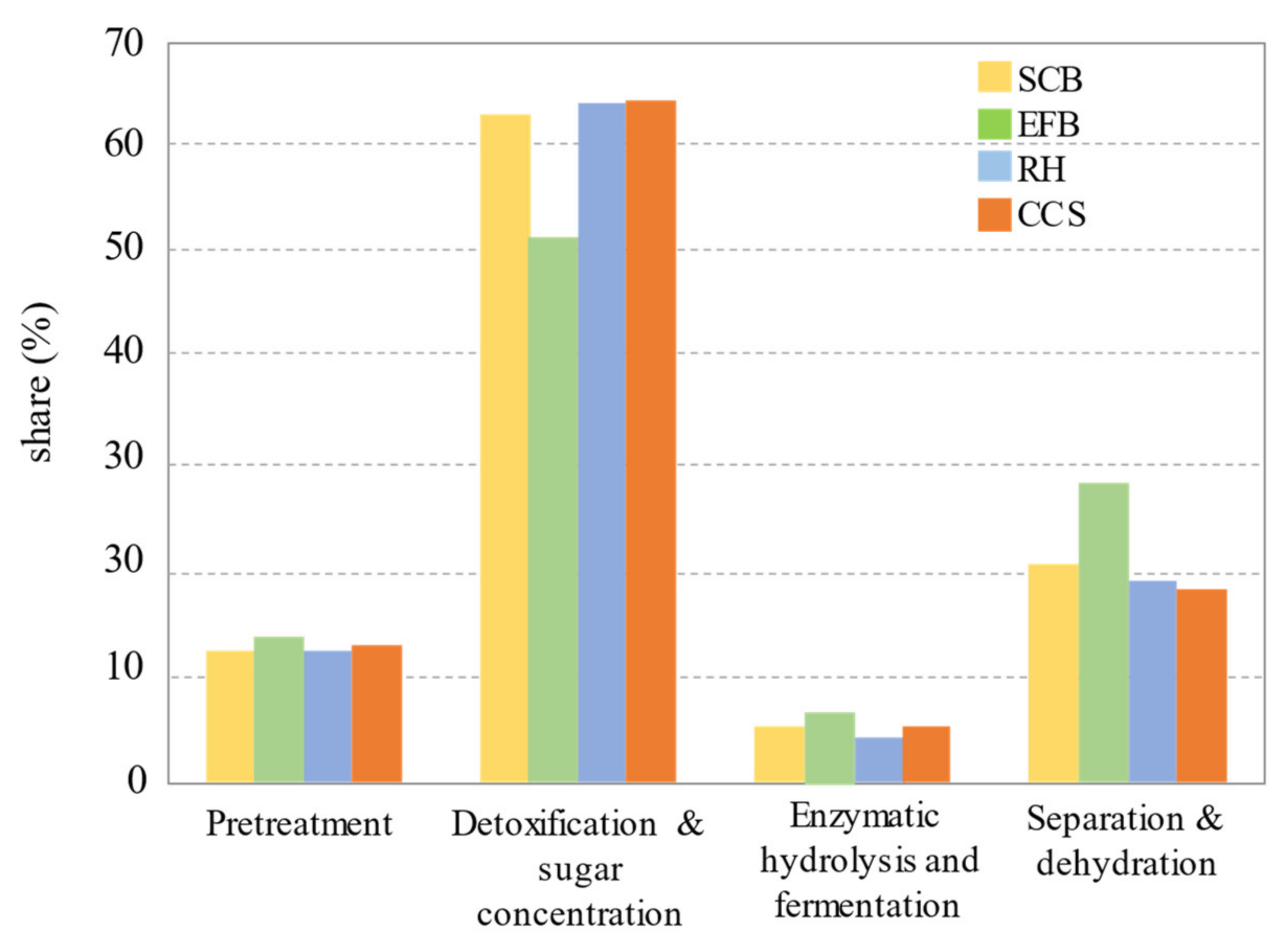
The authors concluded that the rapid explosion time in ICSE process, as compared to conventional SE processes, resulted in a significant reduction in processing time, from 15–20 min to 90–120 s, and a decrease in steam consumption from 0.9 tons to 0.25 tons per 1 ton of biomass treated. This study builds upon another previous one [225] in which the SE of corn stalk was divided into three stages, consisting of an initial pressure boost stage, a holding pressure stage, and a decompression stage. The most energy-consuming stage was the initial pressure rise, from atmospheric to holding pressure. During the latter step, the total energy consumption is the sum of five heating energies, namely steam heating (Q1), air heating (Q2), material heating (Q3), reactor heating (Q4), and reactor radiation energy (Q5), which were all calculated by multiplying the mass considered by enthalpy (Q1) or by multiplying the specific heat by the mass considered and then by the difference in temperature (Q2, Q3, and Q4). During the holding pressure stage, the energy consumption was attributed only to the heat radiation of SE reactor and calculated using the Stefan–Bolzmann law [226]. A modeling attempt using a central composite design concluded, as expected, that holding temperature and time were found to have a significant effect, along with the moisture content of the material (W). Steam consumption per unit mass of dry material marked an increase from 0.53 to 3.1 kg/kg−1 when W was raised from 10% to 90%, while keeping the other parameters constant at their central level. It is important to emphasize that in the process developed by Semhaoui et al. [30] for the PT of hemp hurds before enzymatic hydrolysis, the air heating energy (Q2) was not taken into account, since a preliminary vacuum step was carried out, which resulted in an acceleration of the heat transfer and a direct relationship between pressure and temperature. The calculation of the total energy consumption in this case becomes simplified. Quintero et al. [227] analyzed the production of bioethanol from four Colombian lignocellulosic materials: sugar-cane bagasse (SCB), coffee cut-stems (CCS), rice husk (RH), and empty fruit bunches (EFB), using Aspen plus software. In this study, a five-step process scheme was employed: (1) PT with dilute acid (135 °C, 4 h); (2) detoxification, consisting of Ca(OH)2 treatment at 60 °C for 30 min to eliminate inhibitory compounds (furfural and HMF); (3) enzymatic saccharification, involving treating the solid-pretreated fraction with cellulases at 50 °C for 96 h to convert solid cellulose into hexoses; (4) fermentation, consisting of the conversion of obtained reducing sugars into ethanol by a recombinant bacterium (Zymomonas mobilis) at 33 °C for 30 h; and (5) a separation, including ethanol distillation and dehydration. For all substrates, the authors showed that the most energy-consuming steps were detoxification and sugar concentration (Figure 6). This is caused by the evaporator system used to concentrate the substrate to a reasonable sugar (xylose and glucose) concentration prior to the fermentation process. It was underlined that the sugar concentration is a key parameter in the fermentation process.

Figure 6.
Energy consumption in each process stage for fuel ethanol production [227].
Besides, the economic performance could also be associated with the composition of the lignocellulosic material, as well as its moisture content. It was found that EFB presented the highest ethanol yield from a dry basis point of view (313.83 L/t), while RH produced less ethanol (250.56 L/t). The better economic performance of EFB could be related to the higher holocellulose content (67.2%), as opposed to RH (53.7%). It was also pointed out that EFB has the highest moisture content and RH a lower one, suggesting that the substrate moisture content favors ethanol production due to a higher flowrate of fresh (wet) material. Raw material costs have consistently emerged as one of the most significant cost factors, as highlighted in various techno-economic studies [227,228]. In the above study [227], they were 15 US $/t for SCB and 5 US $/t for EFB. By taking into account all considerations, the lowest bioethanol production cost was 0.578 US $ for EFB and the highest was 0.766 US $ for SCB. In recent years, intensive investigations have focused on second-generation biofuel production. This intensity in research we can attribute to the sustainability of lignocellulosic sources (crops and wood residues, corn stover, wheat and barley straw, agricultural wastes, etc.), and to non-competitiveness with food sector, unlike the case of first-generation bioethanol. Techno-economic analyses have shown that the obtained biofuels remain economically challenged compared to fossil fuels. Beside using inexpensive and abundant feedstocks according to different countries, the coproduction of high-added-value products has been shown to be a promising way to improve the energy output and economic feasibility of biofuel production [228,229]. To foster economic profitability, lignin residues could be used for heat or heat and power generation in a cogeneration strategy [227,230,231]. Moreover, the environmental impact of the use of woody biomass, and more broadly of lignocellulosic materials, when producing bioethanol is worth delving into in order to ensure its sustainability. Recently, Bello et al. [232] explored bioenergy with carbon capture and storage system (BECCS) as applied to a biorefinery. In this investigation, the biorefinery converted wood waste materials into bioethanol, which was then employed for internal combustion and engine vehicles. The bioethanol was studied in blends with gasoline, and the scenarios defined took into account the heating source for the CO2 compression step (bio-source: sugarcane bagasse vs. fossil source: natural gas). The conclusions indicate that it is possible to attain negative emissions by utilizing blends with bioethanol fractions of 85%. In fact, such a blend enables a net balance of −2.74 kg CO2 eq/100 km traveled in Europe, −4.62 kg CO2 eq/100 km traveled in France, and −5.01 kg CO2 eq/100 km traveled in Norway to be achieved. This holds true for those countries, as their shares of renewable energy are higher. Cavalaglio et al. [233] have concluded that the implementation of catalyst-assisted SE in cardoon biomass contributes to a reduction in energy costs, effectively mitigating environmental impact in terms of global warming potential (3.18 kg CO2 eq/kg of fermentable sugars) and cumulative energy demand (43.3 MJ/kg fermentable sugars). The above further confirms the importance of biofuels in reducing the carbon footprint of biorefineries.
7. Conclusions
In conclusion, woody biomass remains an abundant and attractive resource for the production of cellulosic bioethanol, notably thanks to its fermentable sugar content. These biomasses, however, do present a recalcitrance to enzymatic digestibility, therefore requiring a pretreatment (PT) step of various types. Biomass PT is an important phase that plays a significant role in the total process cost and sustainability, since it involves the consumption of energy for temperature increases, maintenance, the use of chemical agents, etc. Thermomechanical PTs such as steam explosion have been described as cost-effective and eco-friendly, as they allow for considerable delignification. These PTs have repeatedly been shown to be successful favorable options for the delignification of LCBs, and are therefore encouraged. Additionally, the impregnation step using a chemical catalyst helps in solubilizing the polymers responsible for recalcitrance, thus allowing a decrease in overall process cost. As for the saccharification and fermentation steps, it has been concluded that SSF is a more performant method, because not only it does decrease the risk of inhibitor formation, but it also contributes to reducing process costs through the use of one reactor instead of two. Both the saccharification and fermentation steps are influenced by various factors. Finally, the energetic, economic, and environmental studies conducted so far have drawn attention to the energetic consumption of the various phases of the bioconversion process. These studies confirm that the choice of raw material, PT type and conditions, and EH/fermentation conditions are all important contributors to the cost and sustainability of bioethanol production.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| CBP | Consolidated bioprocessing |
| CSF (or R0) | Combined severity factor |
| DB | Dry basis |
| E/W | Ethanol/water |
| EH | Enzymatic hydrolysis |
| IV-HMT | Intensive vacuum heat–moisture treatment |
| LCB | Lignocellulosic biomass |
| MT | Mannitol |
| 2N | 2-naphtol |
| NS | 2-naphtol-7-sulfonate |
| NaCS | Sodium cumene sulfonate solution |
| PHP | Phosphoric acid + hydrogen peroxide |
| PSSF | Pre-saccharification followed by simultaneous saccharification and fermentation |
| PT | Pretreatment |
| S/G Ratio | Ratio of S-to-G subunits |
| SE | Steam explosion |
| SHF | Separate hydrolysis and fermentation |
| SP | Steam pressure |
| SSCF | Simultaneous saccharification and co-fermentation |
| SSF | Simultaneous saccharification and fermentation |
| TsOH | p-toluenesulfonic acid |
| w/v | Weight on volume |
| w/w | Weight on weight |
References
- U.S. Energy Information Administration (EIA). U.S. Energy Information Administration—EIA—Independent Statistics and Analysis. Available online: https://www.eia.gov/todayinenergy/detail.php?id=56040 (accessed on 22 May 2023).
- ExxonMobil Energy Demand: Three Drivers. Available online: https://corporate.exxonmobil.com/what-we-do/energy-supply/outlook-for-energy/energy-demand#Residentialandcommercial (accessed on 22 May 2023).
- Morris, J. Renewable Energy. Available online: https://climate.mit.edu/explainers/renewable-energy (accessed on 22 May 2023).
- International Renewable Energy Agency (IRENA). Renewable Power Remains Cost-Competitive amid Fossil Fuel Crisis. Available online: https://www.irena.org/news/pressreleases/2022/Jul/Renewable-Power-Remains-Cost-Competitive-amid-Fossil-Fuel-Crisis (accessed on 22 May 2023).
- Rodrigue, J.-P. The Geography of Transport Systems. In The Geography of Transport Systems; Routledge: London, UK, 2020; ISBN 978-0-367-36463-2. [Google Scholar]
- International Energy Agency Renewables 2022—Analysis. Available online: https://www.iea.org/reports/renewables-2022/executive-summary (accessed on 28 April 2023).
- IEA. Transport Biofuels—Renewables 2022—Analysis. Available online: https://www.iea.org/reports/renewables-2022/transport-biofuels (accessed on 3 April 2023).
- International Energy Agency. Global Energy Review: CO2 Emissions in 2021; International Energy Agency: Paris, France, 2021. [Google Scholar]
- Eurostat Renewable Energy Statistics. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Renewable_energy_statistics#Share_of_renewable_energy_more_than_doubled_between_2004_and_2021 (accessed on 10 March 2023).
- Papathanasiou, D. The Developing Countries Leading the Way on Renewable Energy. World Economic Forum. Available online: https://www.weforum.org/agenda/2022/07/renewables-are-the-key-to-green-secure-affordable-energy (accessed on 10 March 2023).
- International Energy Agency. World Energy Outlook 2022; International Energy Agency: Paris, France, 2022; p. 524. [Google Scholar]
- Xu, F.; Li, Y. Biomass Digestion. Encycl. Sustain. Technol. 2017, 3, 197–204. [Google Scholar] [CrossRef]
- Zoghlami, A.; Paës, G. Lignocellulosic Biomass: Understanding Recalcitrance and Predicting Hydrolysis. Front. Chem. 2019, 7, 874. [Google Scholar] [CrossRef] [PubMed]
- Niju, S.; Swathika, M.; Balajii, M. Pretreatment of Lignocellulosic Sugarcane Leaves and Tops for Bioethanol Production. In Lignocellulosic Biomass to Liquid Biofuels; Elsevier: Amsterdam, The Netherlands, 2020; pp. 301–324. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). The State of the World’s Forests 2022; FAO: Rome, Italy, 2022; ISBN 9789251359846. [Google Scholar]
- Merklein, K.; Fong, S.S.; Deng, Y. Biomass Utilization. Biotechnology for Biofuel Production and Optimization; Elsevier: Amsterdam, The Netherlands, 2016; pp. 291–324. [Google Scholar] [CrossRef]
- Yousuf, A.; Pirozzi, D.; Sannino, F. Fundamentals of Lignocellulosic Biomass. Lignocellulosic Biomass to Liquid Biofuels; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–15. [Google Scholar] [CrossRef]
- Yano, H.; Fu, W. Hemp: A Sustainable Plant with High Industrial Value in Food Processing. Foods 2023, 12, 651. [Google Scholar] [CrossRef]
- European Commission. Hemp Production in the EU. Available online: https://agriculture.ec.europa.eu/farming/crop-productions-and-plant-based-products/hemp_en (accessed on 12 March 2023).
- United Nations Conference on Trade and Development Hemp’s Versatility and Sustainability Offer Huge Opportunities for Developing Countries. UNCTAD. Available online: https://unctad.org/news/hemps-versatility-and-sustainability-offer-huge-opportunities-developing-countries (accessed on 12 March 2023).
- Vosper, J. The Role of Industrial Hemp in Carbon Farming; GoodEarth Resources PTY Ltd.: Newport, NSW, Australia, 2011; pp. 1–6. [Google Scholar]
- University of York. Hemp-30 Phase I Final Report; University of York: Heslington, UK, 2022. [Google Scholar]
- Tutek, K.; Masek, A. Hemp and Its Derivatives as a Universal Industrial Raw Material (with Particular Emphasis on the Polymer Industry)—A Review. Materials 2022, 15, 2565. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.T.M.F.; Islam, M.Z.; Mahmud, M.S.; Sarker, M.E.; Islam, M.R. Hemp as a Potential Raw Material toward a Sustainable World: A Review. Heliyon 2022, 8, e08753. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Chanet, G.; Morin-Crini, N. Applications of Hemp in Textiles, Paper Industry, Insulation and Building Materials, Horticulture, Animal Nutrition, Food and Beverages, Nutraceuticals, Cosmetics and Hygiene, Medicine, Agrochemistry, Energy Production and Environment: A Review. Environ. Chem. Lett. 2020, 18, 1451–1476. [Google Scholar] [CrossRef]
- Semhaoui, I. Etude de la Bioconversion de la Chènevotte (Cannabis Sativa) et de l’alfa (Stipa Tenacissima) par Prétraitement Thermomécanique en Présence d’un Catalyseur Acide ou Alcalin. Ph.D. Thesis, Université de La Rochelle, La Rochelle, France, 2019. [Google Scholar]
- Bruker Extracting High-Value Products from Hemp Waste. Available online: https://www.bruker.com/es/resources/library/application-notes-mr/extracting-high-value-products-from-hemp-waste.html (accessed on 13 March 2023).
- Naithani, V.; Tyagi, P.; Jameel, H.; Lucia, L.A.; Pal, L. Ecofriendly and Innovative Processing of Hemp Hurds Fibers for Tissue and Towel Paper. BioResources 2020, 15, 706–720. [Google Scholar] [CrossRef]
- Periyasamy, S.; Beula Isabel, J.; Kavitha, S.; Karthik, V.; Mohamed, B.A.; Gizaw, D.G.; Sivashanmugam, P.; Aminabhavi, T.M. Recent Advances in Consolidated Bioprocessing for Conversion of Lignocellulosic Biomass into Bioethanol—A Review. Chem. Eng. J. 2023, 453, 139783. [Google Scholar] [CrossRef]
- Semhaoui, I.; Maugard, T.; Zarguili, I.; Rezzoug, S.A.; Zhao, J.M.Q.; Toyir, J.; Nawdali, M.; Maache-Rezzoug, Z. Eco-Friendly Process Combining Acid-Catalyst and Thermomechanical Pretreatment for Improving Enzymatic Hydrolysis of Hemp Hurds. Bioresour. Technol. 2018, 257, 192–200. [Google Scholar] [CrossRef]
- Paramasivan, S.; Sankar, S.; Velavan, R.S.; Krishnakumar, T.; Batcha, R.S.I.; Muthuvelu, K.S. Assessing the Potential of Lignocellulosic Energy Crops as an Alternative Resource for Bioethanol Production Using Ultrasound Assisted Dilute Acid Pretreatment. Mater. Today Proc. 2021, 45, 3279–3285. [Google Scholar] [CrossRef]
- Smullen, E.; Finnan, J.; Dowling, D.; Mulcahy, P. The Environmental Performance of Pretreatment Technologies for the Bioconversion of Lignocellulosic Biomass to Ethanol. Renew. Energy 2019, 142, 527–534. [Google Scholar] [CrossRef]
- Roni, K.A.; Hastarina, M.; Herawati, N. Effect of Time and Concentration of Sulfuric Acid on Yield Bioethanol Produced In Making Bioethanol from Peat Soil. J. Phys. Conf. Ser. 2019, 1167, 012056. [Google Scholar] [CrossRef]
- Achinas, S.; Euverink, G.J.W. Consolidated Briefing of Biochemical Ethanol Production from Lignocellulosic Biomass. Electron. J. Biotechnol. 2016, 23, 44–53. [Google Scholar] [CrossRef]
- El Hage, M.; Rajha, N.H.; Maache-Rezzoug, Z.; Koubaa, M. Intensification of Bioethanol Production from Different Lignocellulosic Biomasses, Induced by Various Pretreatment Methods: An Updated Review. Energies 2022, 15, 6912. [Google Scholar] [CrossRef]
- Demichelis, F.; Laghezza, M.; Chiappero, M.; Fiore, S. Technical, Economic and Environmental Assessement of Bioethanol Biorefinery from Waste Biomass. J. Clean. Prod. 2020, 277, 124111. [Google Scholar] [CrossRef]
- Silva Ortiz, P.A.; Maréchal, F.; de Oliveira Junior, S. Exergy Assessment and Techno-Economic Optimization of Bioethanol Production Routes. Fuel 2020, 279, 118327. [Google Scholar] [CrossRef]
- Araghi, M.K.; Barkhordari, S.; Hasannia, R. Economic Impacts of Producing Bioethanol in Iran: A CGE Approach. Energy 2023, 263, 125765. [Google Scholar] [CrossRef]
- Ruiz, H.A.; Sganzerla, W.G.; Larnaudie, V.; Veersma, R.J.; van Erven, G.; Shiva; Ríos-González, L.J.; Rodríguez-Jasso, R.M.; Rosero-Chasoy, G.; Ferrari, M.D.; et al. Advances in Process Design, Techno-Economic Assessment and Environmental Aspects for Hydrothermal Pretreatment in the Fractionation of Biomass under Biorefinery Concept. Bioresour. Technol. 2023, 369, 128469. [Google Scholar] [CrossRef] [PubMed]
- Gandam, P.K.; Chinta, M.L.; Pabbathi, N.P.P.; Baadhe, R.R.; Sharma, M.; Thakur, V.K.; Sharma, G.D.; Ranjitha, J.; Gupta, V.K. Second-Generation Bioethanol Production from Corncob—A Comprehensive Review on Pretreatment and Bioconversion Strategies, Including Techno-Economic and Lifecycle Perspective. Ind. Crops Prod. 2022, 186, 115245. [Google Scholar] [CrossRef]
- Shukla, A.; Kumar, D.; Girdhar, M.; Kumar, A.; Goyal, A.; Malik, T.; Mohan, A. Strategies of Pretreatment of Feedstocks for Optimized Bioethanol Production: Distinct and Integrated Approaches. Biotechnol. Biofuels Bioprod. 2023, 16, 44. [Google Scholar] [CrossRef]
- Zhao, Y.; Damgaard, A.; Liu, S.; Chang, H.; Christensen, T.H. Bioethanol from Corn Stover—Integrated Environmental Impacts of Alternative Biotechnologies. Resour. Conserv. Recycl. 2020, 155, 104652. [Google Scholar] [CrossRef]
- Lancha, J.P. A Multiscale Approach to Understand and Predict the Effects of Hydrothermal Treatment on Lignocellulosic Biomass; Université Paris-Saclay: Paris, France, 2020. [Google Scholar]
- Keskin, T.; Abubackar, H.N.; Arslan, K.; Azbar, N. Biohydrogen Production From Solid Wastes. Biohydrogen 2019, 321–346. [Google Scholar] [CrossRef]
- Tabil, L.; Adapa, P.; Kashaninejad, M. Biomass Feedstock Pre-Processing—Part 1: Pre-Treatment. In Biofuel’s Engineering Process Technolog; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Anukam, A.; Berghel, J. Biomass Pretreatment and Characterization: A Review. In Biotechnology Application Biomass; IntechOpen: London, UK, 2020. [Google Scholar] [CrossRef]
- Bajpai, P. Xylanases. Encycl. Microbiol. 2009, 600–612. [Google Scholar] [CrossRef]
- Simangunsong, E.; Ziegler-Devin, I.; Chrusciel, L.; Girods, P.; Wistara, N.; Brosse, N. Steam Explosion of Beech Wood-Effect of the Particle Size on the Xylans Recovery. HAL Id: Hal-02125966. Waste Biomass Valorization 2019, 11, 625. [Google Scholar] [CrossRef]
- Guo, M.; Littlewood, J.; Joyce, J.; Murphy, R. The Environmental Profile of Bioethanol Produced from Current and Potential Future Poplar Feedstocks in the EU. Green Chem. 2014, 16, 4680–4695. [Google Scholar] [CrossRef]
- Wu, P.; Li, L.; Zhou, Y.; Wang, W.; Sun, Y.; Guo, Y.; Kang, X. Biorefining of Ethanol and Methane from NaOH Pretreated Poplar Residues: Mass Balance and Energy Flow Analyses. Fuel 2023, 333, 126293. [Google Scholar] [CrossRef]
- Kizhakkepurakkal, A.R. Opportunities and Challenges Associated with Development of Wood Biomass Energy Production in Louisiana; Kerala Agricultural University: Thrissur, India, 2008. [Google Scholar]
- Geng, W.; Venditti, R.A.; Pawlak, J.J.; Chang, H.M. Effect of Delignification on Hemicellulose Extraction from Switchgrass, Poplar, and Pine and Its Effect on Enzymatic Convertibility of Cellulose-Rich Residues. BioResources 2019, 13, 4946–4963. [Google Scholar] [CrossRef]
- Bay, M.S.; Eslami, F.; Karimi, K. The Relationship between Structural Features of Lignocellulosic Materials and Ethanol Production Yield. Designs 2022, 6, 119. [Google Scholar] [CrossRef]
- Houghton, R.A. Biomass. Biofuels Biorefining 2008, 1, 448–453. [Google Scholar]
- Paul, S.K.; Chakraborty, S. Microwave-Assisted Ionic Liquid-Mediated Rapid Catalytic Conversion of Non-Edible Lignocellulosic Sunn Hemp Fibres to Biofuels. Bioresour. Technol. 2018, 253, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Buck, M.; Senn, T. Energy Self-Sufficient Production of Bioethanol from a Mixture of Hemp Straw and Triticale Seeds: Life-Cycle Analysis. Biomass Bioenergy 2016, 95, 99–108. [Google Scholar] [CrossRef]
- Isikgor, F.H.; Becer, C.R. Lignocellulosic Biomass: A Sustainable Platform for the Production of Bio-Based Chemicals and Polymers. Polym. Chem. 2015, 6, 4497–4559. [Google Scholar] [CrossRef]
- Jesús RANGEL, M.; Hornus, M.; Felissia, F.E.; Area, M.C. Hydrothermal Treatment of Eucalyptus Sawdust for a Forest Biorefinery. Cellul. Chem. Technol. 2016, 50, 521–528. [Google Scholar]
- Rochón, E.; Cabrera, M.N.; Scutari, V.; Cagno, M.; Guibaud, A.; Martínez, S.; Böthig, S.; Guchin, N.; Ferrari, M.D.; Lareo, C. Co-Production of Bioethanol and Xylosaccharides from Steam-Exploded Eucalyptus Sawdust Using High Solid Loads in Enzymatic Hydrolysis: Effect of Alkaline Impregnation. Ind. Crops Prod. 2022, 175, 114253. [Google Scholar] [CrossRef]
- Bodîrlău, R.; Teacă, C.A.; Spiridon, I. Chemical Modification of Beech Wood: Effect on Thermal Stability. BioResources 2008, 3, 789–800. [Google Scholar] [CrossRef]
- Szczepkowski, A.; Nicewicz, D.; Koczoń, P. The Relationship Between Tree Health and Chemical Composition Of Beech (Fagus Sylvatica L.) and Oak (Quercus Robur L.) Wood of Polish Provenances. Acta Sci. Pol. Tecnol. Aliment. 2007, 6, 77–88. [Google Scholar]
- Bari, E.; Mohebby, B.; Naji, H.R.; Oladi, R.; Yilgor, N.; Nazarnezhad, N.; Ohno, K.M.; Nicholas, D.D. Monitoring the Cell Wall Characteristics of Degraded Beech Wood by White-Rot Fungi: Anatomical, Chemical, and Photochemical Study. Maderas Cienc. Tecnol. 2018, 20, 35–56. [Google Scholar] [CrossRef]
- Rowell, R.M.; Keany, F.M. Fiberboards Made From Acetylated Bagasse Fiber. Wood Fiber Sci. 1991, 23, 15–22. [Google Scholar]
- Wang, Z.; Winestrand, S.; Gillgren, T.; Jönsson, L.J. Chemical and Structural Factors Influencing Enzymatic Saccharification of Wood from Aspen, Birch and Spruce. Biomass Bioenergy 2018, 109, 125–134. [Google Scholar] [CrossRef]
- Borovkova, V.S.; Malyar, Y.N.; Sudakova, I.G.; Chudina, A.I.; Zimonin, D.V.; Skripnikov, A.M.; Miroshnikova, A.V.; Ionin, V.A.; Kazachenko, A.S.; Sychev, V.V.; et al. Composition and Structure of Aspen (Pópulus Trémula) Hemicelluloses Obtained by Oxidative Delignification. Polymers 2022, 14, 4521. [Google Scholar] [CrossRef]
- Buzala, K.P.; Kalinowska, H.; Malachowska, E.; Boruszewski, P.; Krajewski, K.; Przybysz, P. The Effect of Lignin Content in Birch and Beech Kraft Cellulosic Pulps on Simple Sugar Yields from the Enzymatic Hydrolysis of Cellulose. Energies 2019, 12, 2952. [Google Scholar] [CrossRef]
- Sjulander, N.; Kikas, T. Two-Step Pretreatment of Lignocellulosic Biomass for High-Sugar Recovery from the Structural Plant Polymers Cellulose and Hemicellulose. Energies 2022, 15, 8898. [Google Scholar] [CrossRef]
- Wan, X.; Liu, J.; Zhang, Y.; Tian, D.; Liu, Y.; Zhao, L.; Huang, M.; Hu, J.; Shen, F. Conversion of Agricultural and Forestry Biomass into Bioethanol, Water-Soluble Polysaccharides, and Lignin Nanoparticles by an Integrated Phosphoric Acid plus Hydrogen Peroxide Process. Ind. Crops Prod. 2023, 191, 115969. [Google Scholar] [CrossRef]
- Geffert, A.; Geffertova, J.; Dudiak, M. Direct Method of Measuring the PH Value of Wood. Forests 2019, 10, 852. [Google Scholar] [CrossRef]
- Kučerová, V.; Hrčka, R.; Hýrošová, T. Relation of Chemical Composition and Colour of Spruce Wood. Polymers 2022, 14, 5333. [Google Scholar] [CrossRef]
- Čabalová, I.; Bélik, M.; Kučerová, V.; Jurczyková, T. Chemical and Morphological Composition of Norway Spruce Wood (Picea Abies L.) in the Dependence of Its Storage. Polymers 2021, 13, 1619. [Google Scholar] [CrossRef]
- Yoo, C.G.; Meng, X.; Pu, Y.; Ragauskas, A.J. The Critical Role of Lignin in Lignocellulosic Biomass Conversion and Recent Pretreatment Strategies: A Comprehensive Review. Bioresour. Technol. 2020, 301, 122784. [Google Scholar] [CrossRef]
- University of Knoxville Tennessee. Typical H:G:S Ratio for Lignin from Biomass. Available online: https://static.igem.org/mediawiki/2018/c/c2/T--CCU_Taiwan--CCUlignin_HGS.pdf (accessed on 22 June 2023).
- Paliwal, R.; Giri, K.; Rai, J.P. Microbial Ligninolysis: Avenue for Natural Ecosystem Management; IGI Global: Hershey, PE, USA, 2015; pp. 120–144. ISBN 9781466686830. [Google Scholar]
- Papa, G.; Varanasi, P.; Sun, L.; Cheng, G.; Stavila, V.; Holmes, B.; Simmons, B.A.; Adani, F.; Singh, S. Exploring the Effect of Different Plant Lignin Content and Composition on Ionic Liquid Pretreatment Efficiency and Enzymatic Saccharification of Eucalyptus Globulus L. Mutants. Bioresour. Technol. 2012, 117, 352–359. [Google Scholar] [CrossRef]
- Wagner, A.; Tobimatsu, Y.; Phillips, L.; Flint, H.; Geddes, B.; Lu, F.; Ralph, J. Syringyl Lignin Production in Conifers: Proof of Concept in a Pine Tracheary Element System. Proc. Natl. Acad. Sci. USA 2015, 112, 6218–6223. [Google Scholar] [CrossRef]
- Li, M.; Pu, Y.; Ragauskas, A.J. Current Understanding of the Correlation of Lignin Structure with Biomass Recalcitrance. Front. Chem. 2016, 4, 45. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, C.S. Recent Efforts to Prevent Undesirable Reactions from Fractionation to Depolymerization of Lignin: Toward Maximizing the Value from Lignin. Front. Energy Res. 2018, 6, 92. [Google Scholar] [CrossRef]
- Eom, T.; Chaiprapat, S.; Charnnok, B. Enhanced Enzymatic Hydrolysis and Methane Production from Rubber Wood Waste Using Steam Explosion. J. Environ. Manag. 2019, 235, 231–239. [Google Scholar] [CrossRef]
- Li, W.C.; Han, L.J.; Peng, T.B.; Xie, Y.Y.; Zou, Y.; Li, L.Z.; Jia, S.R.; Zhong, C. Structural and Behavior Changes of Herbaceous and Hardwood Biomass during Steam Explosion Pretreatment and Enzymatic Hydrolysis. BioResources 2020, 15, 691–705. [Google Scholar] [CrossRef]
- Singh, J.; Suhag, M.; Dhaka, A. Augmented Digestion of Lignocellulose by Steam Explosion, Acid and Alkaline Pretreatment Methods: A Review. Carbohydr. Polym. 2015, 117, 624–631. [Google Scholar] [CrossRef]
- He, Q.; Hou, Q.; Hong, L.; Lu, X.; Ziegler-Devin, I.; Chrusciel, L.; Besserer, A.; Brosse, N. Effect of Highly Efficient Steam Explosion Treatment on Beech, Poplar and Spruce Solid Wood Physicochemical and Permeable Performances. Ind. Crops Prod. 2022, 182, 114901. [Google Scholar] [CrossRef]
- Gao, H.; Wang, Y.; Yang, Q.; Peng, H.; Li, Y.; Zhan, D.; Wei, H.; Lu, H.; Bakr, M.M.A.; EI-Sheekh, M.M.; et al. Combined Steam Explosion and Optimized Green-Liquor Pretreatments Are Effective for Complete Saccharification to Maximize Bioethanol Production by Reducing Lignocellulose Recalcitrance in One-Year-Old Bamboo. Renew. Energy 2021, 175, 1069–1079. [Google Scholar] [CrossRef]
- Adawiyah, D.R.; Akuzawa, S.; Sasaki, T.; Kohyama, K. A Comparison of the Effects of Heat Moisture Treatment (HMT) on Rheological Properties and Amylopectin Structure in Sago (Metroxylon Sago) and Arenga (Arenga Pinnata) Starches. J. Food Sci. Technol. 2017, 54, 3404. [Google Scholar] [CrossRef] [PubMed]
- Aklouche, L.; Monteau, J.-Y.; Rezzoug, S.-A.; Maugard, T.; Guihard, L.; Cohendoz, S.; Maache-Rezzoug, Z. Prediction of Thermal Conductivity and Specific Heat of Native Maize Starch and Comparison with Hmt Treated Starch. J. Renew. Mater. 2019, 7, 535–546. [Google Scholar] [CrossRef]
- Maruta, I.; Kurahashi, Y.; Takano, R.; Hayashi, K.; Yoshino, Z. Reduced-Pressurized Heat-Moisture Treatment: A New Method for Heat-Moisture Treatment of Starch. Available online: https://agris.fao.org/agris-search/search.do?recordID=DE94S0213 (accessed on 28 April 2023).
- Bahrani, S.A.; Monteau, J.Y.; Rezzoug, S.A.; Loisel, C.; Maache-Rezzoug, Z. Physics-Based Modeling of Simultaneous Heat and Mass Transfer Intensification during Vacuum Steaming Processes of Starchy Material. Chem. Eng. Process. Process Intensif. 2014, 85, 216–226. [Google Scholar] [CrossRef]
- Romaní, A.; Garrote, G.; Ballesteros, I.; Ballesteros, M. Second Generation Bioethanol from Steam Exploded Eucalyptus Globulus Wood. Fuel 2013, 111, 66–74. [Google Scholar] [CrossRef]
- Troncoso-Ortega, E.; Castillo, R.D.P.; Reyes-Contreras, P.; Castaño-Rivera, P.; Mendonça, R.T.; Schiappacasse, N.; Parra, C. Effects on Lignin Redistribution in Eucalyptus Globulus Fibres Pre-Treated by Steam Explosion: A Microscale Study to Cellulose Accessibility. Biomolecules 2021, 11, 507. [Google Scholar] [CrossRef] [PubMed]
- Schneider, W.D.H.; Fontana, R.C.; Baudel, H.M.; de Siqueira, F.G.; Rencoret, J.; Gutiérrez, A.; de Eugenio, L.I.; Prieto, A.; Martínez, M.J.; Martínez, Á.T.; et al. Lignin Degradation and Detoxification of Eucalyptus Wastes by On-Site Manufacturing Fungal Enzymes to Enhance Second-Generation Ethanol Yield. Appl. Energy 2020, 262, 114493. [Google Scholar] [CrossRef]
- Pažitný, A.; Russ, A.; Boháček, Š.; Stankovská, M.; Ihnát, V. Effect of Steam Explosion on Enzymatic Hydrolysis of Various Parts of Poplar Tree. Wood Res. 2020, 65, 579–590. [Google Scholar] [CrossRef]
- Barbanera, M.; Lascaro, E.; Foschini, D.; Cotana, F.; Buratti, C. Optimization of Bioethanol Production from Steam Exploded Hornbeam Wood (Ostrya Carpinifolia) by Enzymatic Hydrolysis. Renew. Energy 2018, 124, 136–143. [Google Scholar] [CrossRef]
- Besserer, A.; Obame, S.N.; Safou-Tchima, R.; Saker, S.; Ziegler-Devin, I.; Brosse, N. Biorefining of Aucoumea Klaineana Wood: Impact of Steam Explosion on the Composition and Ultrastructure the Cell Wall. Ind. Crops Prod. 2022, 177, 114432. [Google Scholar] [CrossRef]
- Mihiretu, G.T.; Chimphango, A.F.; Görgens, J.F. Steam Explosion Pre-Treatment of Alkali-Impregnated Lignocelluloses for Hemicelluloses Extraction and Improved Digestibility. Bioresour. Technol. 2019, 294, 122121. [Google Scholar] [CrossRef]
- Pielhop, T.; Amgarten, J.; Von Rohr, P.R.; Studer, M.H. Steam Explosion Pretreatment of Softwood: The Effect of the Explosive Decompression on Enzymatic Digestibility. Biotechnol. Biofuels 2016, 9, 152. [Google Scholar] [CrossRef]
- Shrotri, A.; Kobayashi, H.; Fukuoka, A. Catalytic Conversion of Structural Carbohydrates and Lignin to Chemicals. In Advances in Catalysis; Elsevier: Amsterdam, The Netherlands, 2017; Volume 60, pp. 59–123. [Google Scholar]
- Duque, A.; Manzanares, P.; Ballesteros, I.; Ballesteros, M. Steam Explosion as Lignocellulosic Biomass Pretreatment; Elsevier Inc.: Amsterdam, The Netherlands, 2016; ISBN 9780128025611. [Google Scholar]
- Russ, A.; Fišerová, M.; Letko, M.; Opálen, E. Effect of Steam Explosion Temperature on Wheat Straw Enzymatic Hydrolysis. Wood Res. 2016, 61, 65–74. [Google Scholar]
- Jeong, S.Y.; Lee, J.W. Hydrothermal Treatment. Pretreat. Biomass Process. Technol. 2015, 61–74. [Google Scholar] [CrossRef]
- Cullis, I.F.; Saddler, J.N.; Mansfield, S.D. Effect of Initial Moisture Content and Chip Size on the Bioconversion Efficiency of Softwood Lignocellulosics. Biotechnol. Bioeng. 2004, 85, 413–421. [Google Scholar] [CrossRef]
- Meehnian, H.; Jana, A.K.; Jana, M.M. Effect of Particle Size, Moisture Content, and Supplements on Selective Pretreatment of Cotton Stalks by Daedalea Flavida and Enzymatic Saccharification. 3 Biotech 2016, 6, 235. [Google Scholar] [CrossRef] [PubMed]
- Barakat, A.; Monlau, F.; Solhy, A.; Carrere, H. Mechanical Dissociation and Fragmentation of Lignocellulosic Biomass: Effect of Initial Moisture, Biochemical and Structural Proprieties on Energy Requirement. Appl. Energy 2015, 142, 240–246. [Google Scholar] [CrossRef]
- Pawar, P.M.A.; Derba-Maceluch, M.; Chong, S.L.; Gandla, M.L.; Bashar, S.S.; Sparrman, T.; Ahvenainen, P.; Hedenström, M.; Özparpucu, M.; Rüggeberg, M.; et al. In Muro Deacetylation of Xylan Affects Lignin Properties and Improves Saccharification of Aspen Wood. Biotechnol. Biofuels 2017, 10, 98. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Peng, H.; Li, H.; Xu, J. Effect of Acetylation/Deacetylation on Enzymatic Hydrolysis of Corn Stalk. Biomass Bioenergy 2014, 71, 294–298. [Google Scholar] [CrossRef]
- Chen, X.; Shekiro, J.; Franden, M.A.; Wang, W.; Zhang, M.; Kuhn, E.; Johnson, D.K.; Tucker, M.P. The Impacts of Deacetylation Prior to Dilute Acid Pretreatment on the Bioethanol Process. Biotechnol. Biofuels 2012, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Shukla, A.; Tiwari, S.; Srivastava, M. A Review on Delignification of Lignocellulosic Biomass for Enhancement of Ethanol Production Potential. Renew. Sustain. Energy Rev. 2014, 32, 713–728. [Google Scholar] [CrossRef]
- Kumar, A.K.; Sharma, S. Recent Updates on Different Methods of Pretreatment of Lignocellulosic Feedstocks: A Review. Bioresour. Bioprocess. 2017, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Onyenwoke, C.; Tabil, L.G.; Dumonceaux, T.; Cree, D.; Mupondwa, E.; Adapa, P.; Karunakaran, C. Investigation of Steam Explosion Pretreatment of Sawdust and Oat Straw to Improve Their Quality as Biofuel Pellets. Energies 2022, 15, 7168. [Google Scholar] [CrossRef]
- Jacquet, N.; Vanderghem, C.; Danthine, S.; Quiévy, N.; Blecker, C.; Devaux, J.; Paquot, M. Influence of Steam Explosion on Physicochemical Properties and Hydrolysis Rate of Pure Cellulose Fibers. Bioresour. Technol. 2012, 121, 221–227. [Google Scholar] [CrossRef]
- Auxenfans, T.; Crônier, D.; Chabbert, B.; Paës, G. Understanding the Structural and Chemical Changes of Plant Biomass Following Steam Explosion Pretreatment. Biotechnol. Biofuels 2017, 10, 36. [Google Scholar] [CrossRef]
- Yu, Y.; Wu, J.; Ren, X.; Lau, A.; Rezaei, H.; Takada, M.; Bi, X.; Sokhansanj, S. Steam Explosion of Lignocellulosic Biomass for Multiple Advanced Bioenergy Processes: A Review. Renew. Sustain. Energy Rev. 2022, 154, 111871. [Google Scholar] [CrossRef]
- Norrrahim, M.N.F.; Ilyas, R.A.; Nurazzi, N.M.; Rani, M.S.A.; Atikah, M.S.N.; Shazleen, S.S. Chemical Pretreatment of Lignocellulosic Biomass for the Production of Bioproducts: An Overview. Appl. Sci. Eng. Prog. 2021, 14, 588–605. [Google Scholar] [CrossRef]
- Jȩdrzejczyk, M.; Soszka, E.; Czapnik, M.; Ruppert, A.M.; Grams, J. Physical and Chemical Pretreatment of Lignocellulosic Biomass; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 9780128151624. [Google Scholar]
- Zhao, J.; Tian, D.; Shen, F.; Hu, J.; Zeng, Y.; Huang, C. Valorizing Waste Lignocellulose-Based Furniture Boards by Phosphoric Acid and Hydrogen Peroxide (Php) Pretreatment for Bioethanol Production and High-Value Lignin Recovery. Sustainability 2019, 11, 6175. [Google Scholar] [CrossRef]
- Ajayo, P.C.; Huang, M.; Zhao, L.; Tian, D.; He, J.; Zou, J.; Fang, D.; Zeng, Y.; Shen, F. High Yield of Fermentable Sugar from Paper Mulberry Woods Using Phosphoric Acid plus Hydrogen Peroxide Pretreatment: Multifactorial Investigation and Optimization. Ind. Crops Prod. 2022, 180, 114771. [Google Scholar] [CrossRef]
- Santos, R.B.; Jameel, H.; Chang, H.M.; Hart, P.W. Impact of Lignin and Carbohydrate Chemical Structures on Degradation Reactions during Hardwood Kraft Pulping Processes. BioResources 2013, 8, 158–171. [Google Scholar] [CrossRef]
- Normark, M.; Winestrand, S.; Lestander, T.A.; Jönsson, L.J. Analysis, Pretreatment and Enzymatic Saccharification of Different Fractions of Scots Pine. BMC Biotechnol. 2014, 14, 20. [Google Scholar] [CrossRef]
- Mirmohamadsadeghi, S.; Karimi, K.; Horváth, I.S. Improvement of Solid-State Biogas Production from Wood by Concentrated Phosphoric Acid Pretreatment. BioResources 2016, 11, 3230–3243. [Google Scholar] [CrossRef]
- Tong, D.; Zhan, P.; Zhang, W.; Zhou, Y.; Huang, Y.; Qing, Y.; Chen, J. A Novel Surfactant-Assisted Dilute Phosphoric Acid plus Steam Explosion Pretreatment of Poplar Wood for Fermentable Sugar Production. Res. Sq. 2021, 18. [Google Scholar] [CrossRef]
- Qin, W.; Wu, L.; Zheng, Z.; Dong, C.; Yang, Y. Lignin Hydrolysis and Phosphorylation Mechanism during Phosphoric Acid–Acetone Pretreatment: A DFT Study. Molecules 2014, 19, 21335. [Google Scholar] [CrossRef]
- Banerjee, G.; Car, S.; Liu, T.; Williams, D.L.; Meza, S.L.; Walton, J.D.; Hodge, D.B. Scale-up and Integration of Alkaline Hydrogen Peroxide Pretreatment, Enzymatic Hydrolysis, and Ethanolic Fermentation. Biotechnol. Bioeng. 2012, 109, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Huang, S.; Wei, W.; Zhang, J.; Xie, J. Investigation of Alkaline Hydrogen Peroxide Pretreatment and Tween 80 to Enhance Enzymatic Hydrolysis of Sugarcane Bagasse. Biotechnol. Biofuels 2019, 12, 107. [Google Scholar] [CrossRef] [PubMed]
- Tareen, A.K.; Punsuvon, V.; Parakulsuksatid, P. Investigation of Alkaline Hydrogen Peroxide Pretreatment to Enhance Enzymatic Hydrolysis and Phenolic Compounds of Oil Palm Trunk. 3 Biotech 2020, 10, 179. [Google Scholar] [CrossRef] [PubMed]
- Novia, N.; Hasanudin, H.; Hermansyah, H.; Fudholi, A. Kinetics of Lignin Removal from Rice Husk Using Hydrogen Peroxide and Combined Hydrogen Peroxide–Aqueous Ammonia Pretreatments. Fermentation 2022, 8, 157. [Google Scholar] [CrossRef]
- Costa, C.A.E.; Vega-Aguilar, C.A.; Rodrigues, A.E. Added-Value Chemicals from Lignin Oxidation. Molecules 2021, 26, 4602. [Google Scholar] [CrossRef]
- Abdou Alio, M.; Tugui, O.C.; Rusu, L.; Pons, A.; Vial, C. Hydrolysis and Fermentation Steps of a Pretreated Sawmill Mixed Feedstock for Bioethanol Production in a Wood Biorefinery. Bioresour. Technol. 2020, 310, 123412. [Google Scholar] [CrossRef]
- Chu, Q.; Tong, W.; Chen, J.; Wu, S.; Jin, Y.; Hu, J.; Song, K. Organosolv Pretreatment Assisted by Carbocation Scavenger to Mitigate Surface Barrier Effect of Lignin for Improving Biomass Saccharification and Utilization. Biotechnol. Biofuels 2021, 14, 136. [Google Scholar] [CrossRef]
- Carvalheiro, F.; Duarte, L.C.; Pires, F.; Van-Dúnem, V.; Sanfins, L.; Roseiro, L.B.; Gírio, F. Effective Mild Ethanol-Based Organosolv Pre-Treatment for the Selective Valorization of Polysaccharides and Lignin from Agricultural and Forestry Residues. Energies 2022, 15, 5654. [Google Scholar] [CrossRef]
- Bhalla, A.; Cai, C.M.; Xu, F.; Singh, S.K.; Bansal, N.; Phongpreecha, T.; Dutta, T.; Foster, C.E.; Kumar, R.; Simmons, B.A.; et al. Performance of Three Delignifying Pretreatments on Hardwoods: Hydrolysis Yields, Comprehensive Mass Balances, and Lignin Properties. Biotechnol. Biofuels 2019, 12, 213. [Google Scholar] [CrossRef]
- Michelin, M.; Liebentritt, S.; Vicente, A.A.; Teixeira, J.A. Lignin from an Integrated Process Consisting of Liquid Hot Water and Ethanol Organosolv: Physicochemical and Antioxidant Properties. Int. J. Biol. Macromol. 2018, 120, 159–169. [Google Scholar] [CrossRef]
- Najafi, E.; Castro, E.; Karimi, K. Biorefining for Olive Wastes Management and Efficient Bioenergy Production. Energy Convers. Manag. 2021, 244, 114467. [Google Scholar] [CrossRef]
- Rencoret, J.; Gutiérrez, A.; Castro, E.; Del Río, J.C. Structural Characteristics of Lignin in Pruning Residues of Olive Tree (Olea Europaea L.). Holzforschung 2019, 73, 25–34. [Google Scholar] [CrossRef]
- Matsakas, L.; Raghavendran, V.; Yakimenko, O.; Persson, G.; Olsson, E.; Rova, U.; Olsson, L.; Christakopoulos, P. Lignin-First Biomass Fractionation Using a Hybrid Organosolv—Steam Explosion Pretreatment Technology Improves the Saccharification and Fermentability of Spruce Biomass. Bioresour. Technol. 2019, 273, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Jung, S.; Ragauskas, A. Pseudo-Lignin Formation and Its Impact on Enzymatic Hydrolysis. Bioresour. Technol. 2012, 117, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Jiang, B.; Chen, H.; Wu, W.; Wu, S.; Jin, Y.; Xiao, H. Recent Advances in Understanding the Effects of Lignin Structural Characteristics on Enzymatic Hydrolysis. Biotechnol. Biofuels 2021, 14, 205. [Google Scholar] [CrossRef]
- He, J.; Huang, C.; Lai, C.; Huang, C.; Li, X.; Yong, Q. Elucidation of Structure-Inhibition Relationship of Monosaccharides Derived Pseudo-Lignin in Enzymatic Hydrolysis. Ind. Crops Prod. 2018, 113, 368–375. [Google Scholar] [CrossRef]
- Brunner, G. Processing of Biomass with Hydrothermal and Supercritical Water. Supercrit. Fluid Sci. Technol. 2014, 5, 395–509. [Google Scholar] [CrossRef]
- Peng, F.; Ren, J.L.; Xu, F.; Bian, J.; Peng, P.; Sun, R.C. Fractional Study of Alkali-Soluble Hemicelluloses Obtained by Graded Ethanol Precipitation from Sugar Cane Bagasse. J. Agric. Food Chem. 2009, 58, 1768–1776. [Google Scholar] [CrossRef] [PubMed]
- Wawro, A.; Batog, J.; Gieparda, W. Polish Varieties of Industrial Hemp and Their Utilisation in the Efficient Production of Lignocellulosic Ethanol. Molecules 2021, 26, 6467. [Google Scholar] [CrossRef]
- Ju, Y.; Kim, I.J.; Kim, S.; Olawuyi, I.F.; Kim, K.M.; Kim, S.R. Deacetylation Kinetics of Promising Energy Crops, Hemp and Kenaf, for Cellulosic Ethanol Production. GCB Bioenergy 2022, 14, 1150–1161. [Google Scholar] [CrossRef]
- Yang, S.; Franden, M.A.; Yang, Q.; Chou, Y.C.; Zhang, M.; Pienkos, P.T. Identification of Inhibitors in Lignocellulosic Slurries and Determination of Their Effect on Hydrocarbon-Producing Microorganisms. Front. Bioeng. Biotechnol. 2018, 6, 23. [Google Scholar] [CrossRef]
- Madadi, M.; Elsayed, M.; Sun, F.; Wang, J.; Karimi, K.; Song, G.; Tabatabaei, M.; Aghbashlo, M. Sustainable Lignocellulose Fractionation by Integrating P-Toluenesulfonic Acid/Pentanol Pretreatment with Mannitol for Efficient Production of Glucose, Native-like Lignin, and Furfural. Bioresour. Technol. 2023, 371, 128591. [Google Scholar] [CrossRef]
- Pielhop, T.; Reinhard, C.; Hecht, C.; Del Bene, L.; Studer, M.H.; Rudolf von Rohr, P. Application Potential of a Carbocation Scavenger in Autohydrolysis and Dilute Acid Pretreatment to Overcome High Softwood Recalcitrance. Biomass Bioenergy 2017, 105, 164–173. [Google Scholar] [CrossRef]
- Chen, L.; Dou, J.; Ma, Q.; Li, N.; Wu, R.; Bian, H.; Yelle, D.J.; Vuorinen, T.; Fu, S.; Pan, X.; et al. Rapid and Near-Complete Dissolution of Wood Lignin at ≤80 °C by a Recyclable Acid Hydrotrope. Sci. Adv. 2017, 3, e1701735. [Google Scholar] [CrossRef] [PubMed]
- Dessie, W.; Tang, J.; Wang, M.; Luo, X.; Liu, X.; Qin, Z. One-Pot Conversion of Industrial Hemp Residue into Fermentable Feedstocks Using Green Catalyst and Enzyme Cocktails Generated by Solid-State Fermentation. Ind. Crops Prod. 2022, 182, 114885. [Google Scholar] [CrossRef]
- Mikulski, D.; Kłosowski, G. High-Pressure Microwave-Assisted Pretreatment of Softwood, Hardwood and Non-Wood Biomass Using Different Solvents in the Production of Cellulosic Ethanol. Biotechnol. Biofuels Bioprod. 2023, 16, 19. [Google Scholar] [CrossRef]
- Madadi, M.; Bakr, M.M.A.; Abdulkhani, A.; Zahoor; Asadollahi, M.A.; Sun, C.; Sun, F.; Abomohra, A.E.F. Alleviating Lignin Repolymerization by Carbocation Scavenger for Effective Production of Fermentable Sugars from Combined Liquid Hot Water and Green-Liquor Pretreated Softwood Biomass. Energy Convers. Manag. 2022, 251, 114956. [Google Scholar] [CrossRef]
- Sierra-Ibarra, E.; Alcaraz-Cienfuegos, J.; Vargas-Tah, A.; Rosas-Aburto, A.; Valdivia-López, Á.; Hernández-Luna, M.G.; Vivaldo-Lima, E.; Martinez, A. Ethanol Production by Escherichia Coli from Detoxified Lignocellulosic Teak Wood Hydrolysates with High Concentration of Phenolic Compounds. J. Ind. Microbiol. Biotechnol. 2022, 49, kuab077. [Google Scholar] [CrossRef]
- Raina, N.; Slathia, P.S.; Sharma, P. Experimental Optimization of Thermochemical Pretreatment of Sal (Shorea Robusta) Sawdust by Central Composite Design Study for Bioethanol Production by Co-Fermentation Using Saccharomyces Cerevisiae (MTCC-36) and Pichia Stipitis (NCIM-3498). Biomass Bioenergy 2020, 143, 105819. [Google Scholar] [CrossRef]
- Lee, I.; Yu, J.H. Design of Hydrothermal and Subsequent Lime Pretreatment for Fermentable Sugar and Bioethanol Production from Acacia Wood. Renew. Energy 2021, 174, 170–177. [Google Scholar] [CrossRef]
- Lee, I.; Yu, J.H. The Production of Fermentable Sugar and Bioethanol from Acacia Wood by Optimizing Dilute Sulfuric Acid Pretreatment and Post Treatment. Fuel 2020, 275, 117943. [Google Scholar] [CrossRef]
- Augustyn, A. Surfactant. Definition, Properties, Examples, & Facts. Britannica. Available online: https://www.britannica.com/science/surfactant (accessed on 3 April 2023).
- Domínguez, E.; Nóvoa, T.; del Río, P.G.; Garrote, G.; Romaní, A. Sequential Two-Stage Autohydrolysis Biorefinery for the Production of Bioethanol from Fast-Growing Paulownia Biomass. Energy Convers. Manag. 2020, 226, 113517. [Google Scholar] [CrossRef]
- Chen, H. Lignocellulose Biorefinery Feedstock Engineering. Lignocellul. Biorefinery Eng. 2015, 37–86. [Google Scholar] [CrossRef]
- Fan, Z. Consolidated Bioprocessing for Ethanol Production. In Biorefineries Integrated Biochemical Processes for Liquid Biofuels; Elsevier: Amsterdam, The Netherlands, 2014; pp. 141–160. [Google Scholar] [CrossRef]
- Stickel, J.J.; Elander, R.T.; MCmillan, J.D.; Brunecky, R. Enzymatic Hydrolysis of Lignocellulosic Biomass. In Bioprocessing of Renewable Resources to Commodity Bioproducts; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 77–103. ISBN 9781118175835. [Google Scholar] [CrossRef]
- Al-Ghanayem, A.A.; Joseph, B.; Alhussaini, M.S.; Ramteke, P.W. Current Applications and Future Trends of Extremozymes in Detergent Industries. Microb. Extrem. 2022, 223–230. [Google Scholar] [CrossRef]
- Yi, Y. Tiny Bugs Play Big Role: Microorganisms’ Contribution to Biofuel Production. In Advances in 2nd Generation of Bioethanol Production; Elsevier: Amsterdam, The Netherlands, 2021; pp. 113–136. [Google Scholar] [CrossRef]
- Vasić, K.; Knez, Ž.; Leitgeb, M. Bioethanol Production by Enzymatic Hydrolysis from Different Lignocellulosic Sources. Molecules 2021, 26, 753. [Google Scholar] [CrossRef]
- Meena, M.; Zehra, A.; Dubey, M.K.; Aamir, M.; Upadhyay, R.S. Penicillium Enzymes for the Food Industries. In New and Future Developments in Microbial Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2018; pp. 167–186. [Google Scholar] [CrossRef]
- Purkait, M.K.; Haldar, D. Enzymatic Hydrolysis of Lignocellulosic Biomass: Mechanistic Insight and Advancement. In Lignocellulosic Biomass to Value-Added Products: Fundamental Strategies and Technological Advancements; Elsevier: Amsterdam, The Netherlands, 2021; pp. 79–94. [Google Scholar] [CrossRef]
- Van Dyk, J.S.; Pletschke, B.I. A Review of Lignocellulose Bioconversion Using Enzymatic Hydrolysis and Synergistic Cooperation between Enzymes--Factors Affecting Enzymes, Conversion and Synergy. Biotechnol. Adv. 2012, 30, 1458–1480. [Google Scholar] [CrossRef]
- Laureano-Perez, L.; Teymouri, F.; Alizadeh, H.; Dale, B.E. Understanding Factors That Limit Enzymatic Hydrolysis of Biomass. In Twenty-Sixth Symposium on Biotechnology for Fuels and Chemicals; Humana Press: Totowa, NJ, USA, 2009; Volume 121, pp. 1081–1099. [Google Scholar]
- Mussatto, S.I.; Dragone, G.; Fernandes, M.; Milagres, A.M.F.; Roberto, I.C. The Effect of Agitation Speed, Enzyme Loading and Substrate Concentration on Enzymatic Hydrolysis of Cellulose from Brewer’s Spent Grain. Cellulose 2008, 15, 711–721. [Google Scholar] [CrossRef]
- Shiva; Climent Barba, F.; Rodríguez-Jasso, R.M.; Sukumaran, R.K.; Ruiz, H.A. High-Solids Loading Processing for an Integrated Lignocellulosic Biorefinery: Effects of Transport Phenomena and Rheology—A Review. Bioresour. Technol. 2022, 351, 127044. [Google Scholar] [CrossRef] [PubMed]
- Zanuso, E.; Ruiz, H.A.; Domingues, L.; Teixeira, J.A. Oscillatory Flow Bioreactor Operating at High Solids Loading for Enzymatic Hydrolysis of Lignocellulosic Biomass. Biochem. Eng. J. 2022, 187, 108632. [Google Scholar] [CrossRef]
- Lu, M.; He, D.; Li, J.; Han, L.; Xiao, W. Rheological Characterization of Ball-Milled Corn Stover with Different Fragmentation Scales at High-Solids Loading. Ind. Crops Prod. 2021, 167, 113517. [Google Scholar] [CrossRef]
- Olofsson, K.; Bertilsson, M.; Lidén, G. A Short Review on SSF—An Interesting Process Option for Ethanol Production from Lignocellulosic Feedstocks. Biotechnol. Biofuels 2008, 1, 7. [Google Scholar] [CrossRef]
- Rana, V.; Eckard, A.D.; Ahring, B.K. Comparison of SHF and SSF of Wet Exploded Corn Stover and Loblolly Pine Using In-House Enzymes Produced from T. Reesei RUT C30 and A. Saccharolyticus. Springerplus 2014, 3, 516. [Google Scholar] [CrossRef] [PubMed]
- Dahnum, D.; Tasum, S.O.; Triwahyuni, E.; Nurdin, M.; Abimanyu, H. Comparison of SHF and SSF Processes Using Enzyme and Dry Yeast for Optimization of Bioethanol Production from Empty Fruit Bunch. Energy Procedia 2015, 68, 107–116. [Google Scholar] [CrossRef]
- Nijland, J.G.; Driessen, A.J.M. Engineering of Pentose Transport in Saccharomyces Cerevisiae for Biotechnological Applications. Front. Bioeng. Biotechnol. 2020, 7, 464. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, Y.; Wan, C.; Li, D.; Mao, Z. Effect of Hot Water Pretreatment Severity on the Degradation and Enzymatic Hydrolysis of Corn Stover. Trans. ASABE 2010, 53, 1929–1934. [Google Scholar] [CrossRef]
- Nitsos, C.K.; Choli-Papadopoulou, T.; Matis, K.A.; Triantafyllidis, K.S. Optimization of Hydrothermal Pretreatment of Hardwood and Softwood Lignocellulosic Residues for Selective Hemicellulose Recovery and Improved Cellulose Enzymatic Hydrolysis. ACS Sustain. Chem. Eng. 2016, 4, 4529–4544. [Google Scholar] [CrossRef]
- Bardak, S.; Nemli, G.; Bardak, T.; Bardak, S.; Nemli, G.; Bardak, T. The Quality Comparison of Particleboards Produced from Heartwood and Sapwood of European Larch. Maderas. Cienc. Tecnol. 2019, 21, 511–520. [Google Scholar] [CrossRef]
- Benouadah, N.; Aliouche, D.; Pranovich, A.; Willför, S. Chemical Characterization of Pinus Halepensis Sapwood and Heartwood. Wood Mater. Sci. Eng. 2018, 14, 157–164. [Google Scholar] [CrossRef]
- Chitambar, J. Marasmiellus Palmivorus. Pest Rating Proposals and Final Ratings. Available online: https://blogs.cdfa.ca.gov/Section3162/?p=4525 (accessed on 4 April 2023).
- Cavka, A.; Jönsson, L.J. Detoxification of Lignocellulosic Hydrolysates Using Sodium Borohydride. Bioresour. Technol. 2013, 136, 368–376. [Google Scholar] [CrossRef]
- Ujor, V.C.; Okonkwo, C.C. Microbial Detoxification of Lignocellulosic Biomass Hydrolysates: Biochemical and Molecular Aspects, Challenges, Exploits and Future Perspectives. Front. Bioeng. Biotechnol. 2022, 10, 1061667. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, Z.; Wang, X.; Wang, N.; Wang, W.; Bao, J. Biodetoxification of Toxins Generated from Lignocellulose Pretreatment Using a Newly Isolated Fungus, Amorphotheca Resinae ZN1, and the Consequent Ethanol Fermentation. Biotechnol. Biofuels 2010, 3, 26. [Google Scholar] [CrossRef]
- Cheng, Y.; Mondal, A.K.; Wu, S.; Xu, D.; Ning, D.; Ni, Y.; Huang, F. Study on the Anti-Biodegradation Property of Tunicate Cellulose. Polymers 2020, 12, 3071. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, L.; Liu, D. Biomass Recalcitrance. Part II: Fundamentals of Different Pre-Treatments to Increase the Enzymatic Digestibility of Lignocellulose. Biofuels Bioprod. Biorefining 2012, 6, 561–579. [Google Scholar] [CrossRef]
- Singh, N.; Devi, A.; Bishnoi, M.B.; Jaryal, R.; Dahiya, A.; Tashyrev, O.; Hovorukha, V.; Singh, N.; Devi, A.; Bishnoi, M.B.; et al. Overview of the Process of Enzymatic Transformation of Biomass. In Elements of Bioeconomy; Biernat, K., Ed.; IntechOpen: London, UK, 2019; ISBN 978-1-78923-862-4. [Google Scholar]
- Kumar, D.; Murthy, G.S. Stochastic Molecular Model of Enzymatic Hydrolysis of Cellulose for Ethanol Production. Biotechnol. Biofuels 2013, 6, 63. [Google Scholar] [CrossRef] [PubMed]
- Daicho, K.; Saito, T.; Fujisawa, S.; Isogai, A. The Crystallinity of Nanocellulose: Dispersion-Induced Disordering of the Grain Boundary in Biologically Structured Cellulose. ACS Appl. Nano Mater. 2018, 1, 5774–5785. [Google Scholar] [CrossRef]
- Xu, H.; Che, X.; Ding, Y.; Kong, Y.; Li, B.; Tian, W. Effect of Crystallinity on Pretreatment and Enzymatic Hydrolysis of Lignocellulosic Biomass Based on Multivariate Analysis. Bioresour. Technol. 2019, 279, 271–280. [Google Scholar] [CrossRef]
- Baruah, J.; Nath, B.K.; Sharma, R.; Kumar, S.; Deka, R.C.; Baruah, D.C.; Kalita, E. Recent Trends in the Pretreatment of Lignocellulosic Biomass for Value-Added Products. Front. Energy Res. 2018, 6, 141. [Google Scholar] [CrossRef]
- Han, L.; Feng, J.; Zhang, S.; Ma, Z.; Wang, Y.; Zhang, X. Alkali Pretreated of Wheat Straw and Its Enzymatic Hydrolysis. Braz. J. Microbiol. 2012, 43, 53–61. [Google Scholar] [CrossRef]
- Eriksson, T.; Börjesson, J.; Tjerneld, F. Mechanism of Surfactant Effect in Enzymatic Hydrolysis of Lignocellulose. Enzyme Microb. Technol. 2002, 31, 353–364. [Google Scholar] [CrossRef]
- Wang, W.; Wang, C.; Zahoor; Chen, X.; Yu, Q.; Wang, Z.; Zhuang, X.; Yuan, Z. Effect of a Nonionic Surfactant on Enzymatic Hydrolysis of Lignocellulose Based on Lignocellulosic Features and Enzyme Adsorption. ACS Omega 2020, 5, 15812–15820. [Google Scholar] [CrossRef]
- Huang, C.; Zhao, X.; Zheng, Y.; Lin, W.; Lai, C.; Yong, Q.; Ragauskas, A.J.; Meng, X. Revealing the Mechanism of Surfactant-Promoted Enzymatic Hydrolysis of Dilute Acid Pretreated Bamboo. Bioresour. Technol. 2022, 360, 127524. [Google Scholar] [CrossRef]
- Oliva-Taravilla, A.; Carrasco, C.; Jönsson, L.J.; Martín, C. Effects of Biosurfactants on Enzymatic Saccharification and Fermentation of Pretreated Softwood. Molecules 2020, 25, 3559. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, S.S.; Balbino, T.R.; Alba, E.M.; Barbosa, F.C.; de Pier, F.T.; de Almeida, A.L.M.; Zilla, A.H.B.; Antunes, F.A.F.; Hilares, R.T.; Balagurusamy, N.; et al. Surfactants in Biorefineries: Role, Challenges & Perspectives. Bioresour. Technol. 2022, 345, 126477. [Google Scholar] [CrossRef]
- Singh, S. Carbohydrates 10. Available online: https://www.uou.ac.in/lecturenotes/science/MSCCH-17/CHEMISTRY%20LN%204%20CARBOHYDRATES-converted%20(1).pdf (accessed on 22 June 2023).
- Frankó, B.; Carlqvist, K.; Galbe, M.; Lidén, G.; Wallberg, O. Removal of Water-Soluble Extractives Improves the Enzymatic Digestibility of Steam-Pretreated Softwood Barks. Appl. Biochem. Biotechnol. 2018, 184, 599. [Google Scholar] [CrossRef]
- Shi, J.; Gladden, J.M.; Sathitsuksanoh, N.; Kambam, P.; Sandoval, L.; Mitra, D.; Zhang, S.; George, A.; Singer, S.W.; Simmons, B.A.; et al. One-Pot Ionic Liquid Pretreatment and Saccharification of Switchgrass. Green Chem. 2013, 15, 2579–2589. [Google Scholar] [CrossRef]
- Sriariyanun, M.; Kitiborwornkul, N.; Tantayotai, P.; Rattanaporn, K.; Show, P.L. One-Pot Ionic Liquid-Mediated Bioprocess for Pretreatment and Enzymatic Hydrolysis of Rice Straw with Ionic Liquid-Tolerance Bacterial Cellulase. Bioengineering 2022, 9, 17. [Google Scholar] [CrossRef]
- Florencio, C.; Badino, A.C.; Farinas, C.S. Soybean Protein as a Cost-Effective Lignin-Blocking Additive for the Saccharification of Sugarcane Bagasse. Bioresour. Technol. 2016, 221, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Simões, I.R.; Brondi, M.G.; Farinas, C.S. In-House Extracted Soybean Protein Can Reduce the Enzyme Dosage in Biomass Saccharification. Fermentation 2023, 9, 142. [Google Scholar] [CrossRef]
- Manzanares, P. Integrated Hydrolysis, Fermentation and Co-Fermentation of Lignocellulosic Biomass. In Bioalcohol Production: Biochemical Conversion of Lignocellulosic Biomass; Woodhead Publishing Limited: Sawston, UK, 2010; pp. 205–223. ISBN 9781845695101. [Google Scholar]
- Joshi, A.; Kanthaliya, B.; Meena, S.; Khan, F.; Arora, J. Process Consolidation Approaches for Cellulosic Ethanol Production. Sustain. Biofuels Oppor. Challenges 2021, 43–72. [Google Scholar] [CrossRef]
- Chen, W.C.; Lin, Y.C.; Ciou, Y.L.; Chu, I.M.; Tsai, S.L.; Lan, J.C.W.; Chang, Y.K.; Wei, Y.H. Producing Bioethanol from Pretreated-Wood Dust by Simultaneous Saccharification and Co-Fermentation Process. J. Taiwan Inst. Chem. Eng. 2017, 79, 43–48. [Google Scholar] [CrossRef]
- Nurdin, M.; Abimanyu, H.; Cahyaningrum, R.; Arham, Z.; Natsir, M.; Maulidiyah, M. Bioethanol Production Based on OPEFB Biomass by Sulfuric Acid Followed by Saccharification and Co-Fermentation Simultaneously. AIP Conf. Proc. 2023, 2719, 030016. [Google Scholar] [CrossRef]
- Nait M’Barek, H.; Arif, S.; Taidi, B.; Hajjaj, H. Consolidated Bioethanol Production from Olive Mill Waste: Wood-Decay Fungi from Central Morocco as Promising Decomposition and Fermentation Biocatalysts. Biotechnol. Rep. 2020, 28, e00541. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, D.; Lee, D.J.; Chang, J.S. Recent Insights into Consolidated Bioprocessing for Lignocellulosic Biohydrogen Production. Int. J. Hydrog. Energy 2019, 44, 14362–14379. [Google Scholar] [CrossRef]
- Singhvi, M.; Kim, B.S. Green Hydrogen Production through Consolidated Bioprocessing of Lignocellulosic Biomass Using Nanobiotechnology Approach. Bioresour. Technol. 2022, 365, 128108. [Google Scholar] [CrossRef]
- Shahab, R.L.; Luterbacher, J.S.; Brethauer, S.; Studer, M.H. Consolidated Bioprocessing of Lignocellulosic Biomass to Lactic Acid by a Synthetic Fungal-Bacterial Consortium. Biotechnol. Bioeng. 2018, 115, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Devos, R.J.B.; Colla, L.M. Simultaneous Saccharification and Fermentation to Obtain Bioethanol: A Bibliometric and Systematic Study. Bioresour. Technol. Rep. 2022, 17, 100924. [Google Scholar] [CrossRef]
- Zhang, W.; Lin, Y.; Zhang, Q.; Wang, X.; Wu, D.; Kong, H. Optimisation of Simultaneous Saccharification and Fermentation of Wheat Straw for Ethanol Production. Fuel 2013, 112, 331–337. [Google Scholar] [CrossRef]
- Alkasrawi, M.; Rudolf, A.; Lidén, G.; Zacchi, G. Influence of Strain and Cultivation Procedure on the Performance of Simultaneous Saccharification and Fermentation of Steam Pretreated Spruce. Enzym. Microb. Technol. 2006, 38, 279–286. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, H.; Gan, M.; Jin, Y.; Gao, X.; Chen, Q.; Guan, J.; Wang, Z. Application of Simultaneous Saccharification and Fermentation (SSF) from Viscosity Reducing of Raw Sweet Potato for Bioethanol Production at Laboratory, Pilot and Industrial Scales. Bioresour. Technol. 2011, 102, 4573–4579. [Google Scholar] [CrossRef]
- Palniandy, T.; Nafsun, A.I.; Mohd Jamil, N.; Herz, F.; Azmi, N.A.A.; Muhammad Zaki, M.H. Influence of Stirring Speed on Glucose and Ethanol Production in Simultaneous Saccharification and Fermentation Process. J. Chem. Eng. Ind. Biotechnol. 2022, 8, 20–25. [Google Scholar] [CrossRef]
- Tareen, A.K.; Sultan, I.N.; Songprom, K.; Laemsak, N.; Sirisansaneeyakul, S.; Vanichsriratana, W.; Parakulsuksatid, P. Two-Step Pretreatment of Oil Palm Trunk for Ethanol Production by Thermotolerent Saccharomyces Cerevisiae SC90. Bioresour. Technol. 2021, 320, 124298. [Google Scholar] [CrossRef]
- Kooprasertying, P.; Vanichsriratana, W.; Sirisansaneeyakul, S.; Laemsak, N.; Tareen, A.K.; Ullah, Z.; Parakulsuksatid, P.; Sultan, I.N. Ethanol Production through Optimized Alkaline Pretreated Elaeis Guineensis Frond Waste from Krabi Province, Thailand. Fermentation 2022, 8, 648. [Google Scholar] [CrossRef]
- Ko, J.K.; Enkh-Amgalan, T.; Gong, G.; Um, Y.; Lee, S.M. Improved Bioconversion of Lignocellulosic Biomass by Saccharomyces Cerevisiae Engineered for Tolerance to Acetic Acid. GCB Bioenergy 2020, 12, 90–100. [Google Scholar] [CrossRef]
- Hashemi, S.; Joseph, P.; Mialon, A.; Moe, S.; Lamb, J.J.; Lien, K.M. Enzymatic Pretreatment of Steam-Exploded Birch Wood for Increased Biogas Production and Lignin Degradation. Bioresour. Technol. Rep. 2021, 16, 100874. [Google Scholar] [CrossRef]
- Zhu, J.; Jiao, N.; Cheng, J.; Zhang, H.; Xu, G.; Xu, Y.; Zhu, J.Y. Integrated Process for the Co-Production of Bioethanol, Furfural, and Lignin Nanoparticles from Birch Wood via Acid Hydrotropic Fractionation. Renew. Energy 2023, 204, 176–184. [Google Scholar] [CrossRef]
- Wagner, E.; Sierra-Ibarra, E.; Rojas, N.L.; Martinez, A. One-Pot Bioethanol Production from Brewery Spent Grain Using the Ethanologenic Escherichia Coli MS04. Renew. Energy 2022, 189, 717–725. [Google Scholar] [CrossRef]
- Lopez-Hidalgo, A.M.; Magaña, G.; Rodriguez, F.; De Leon-Rodriguez, A.; Sanchez, A. Co-Production of Ethanol-Hydrogen by Genetically Engineered Escherichia Coli in Sustainable Biorefineries for Lignocellulosic Ethanol Production. Chem. Eng. J. 2021, 406, 126829. [Google Scholar] [CrossRef]
- Díaz, M.J.; Moya, M.; Castro, E. Bioethanol Production from Steam-Exploded Barley Straw by Co-Fermentation with Escherichia Coli SL100. Agronomy 2022, 12, 874. [Google Scholar] [CrossRef]
- Ab Rasid, N.S.; Shamjuddin, A.; Abdul Rahman, A.Z.; Amin, N.A.S. Recent Advances in Green Pre-Treatment Methods of Lignocellulosic Biomass for Enhanced Biofuel Production. J. Clean. Prod. 2021, 321, 129038. [Google Scholar] [CrossRef]
- Baral, N.R.; Shah, A. Comparative Techno-Economic Analysis of Steam Explosion, Dilute Sulfuric Acid, Ammonia Fiber Explosion and Biological Pretreatments of Corn Stover. Bioresour. Technol. 2017, 232, 331–343. [Google Scholar] [CrossRef]
- Malik, K.; Sharma, P.; Yang, Y.; Zhang, P.; Zhang, L.; Xing, X.; Yue, J.; Song, Z.; Nan, L.; Yujun, S.; et al. Lignocellulosic Biomass for Bioethanol: Insight into the Advanced Pretreatment and Fermentation Approaches. Ind. Crops Prod. 2022, 188, 115569. [Google Scholar] [CrossRef]
- Kuittinen, S.; Hietaharju, J.; Bhattarai, I.; Hassan, M.K.; Kupiainen, L.; Kangas, J.; Tanskanen, J.; Pappinen, A. Technoeconomic Analysis and Environmental Sustainability Estimation of Bioalcohol Production from Barley Straw. Biocatal. Agric. Biotechnol. 2022, 43, 102427. [Google Scholar] [CrossRef]
- Liu, L.Y.; Chandra, R.P.; Tang, Y.; Huang, X.Y.; Bai, F.W.; Liu, C.G. Instant Catapult Steam Explosion: An Efficient Preprocessing Step for the Robust and Cost-Effective Chemical Pretreatment of Lignocellulosic Biomass. Ind. Crops Prod. 2022, 188, 115664. [Google Scholar] [CrossRef]
- Sui, W.; Chen, H. Multi-Stage Energy Analysis of Steam Explosion Process. Chem. Eng. Sci. 2014, 116, 254–262. [Google Scholar] [CrossRef]
- Quinn, T.J.; Martin, J.E. Radiometric Measurements of the Stefan-Boltzmann Constant and Thermodynamic Temperature between −40 °C and +100 °C. Metrologia 1984, 20, 163. [Google Scholar] [CrossRef]
- Quintero, J.A.; Moncada, J.; Cardona, C.A. Techno-Economic Analysis of Bioethanol Production from Lignocellulosic Residues in Colombia: A Process Simulation Approach. Bioresour. Technol. 2013, 139, 300–307. [Google Scholar] [CrossRef]
- Yong, K.J.; Wu, T.Y. Second-Generation Bioenergy from Oilseed Crop Residues: Recent Technologies, Techno-Economic Assessments and Policies. Energy Convers. Manag. 2022, 267, 115869. [Google Scholar] [CrossRef]
- Aghaei, S.; Karimi Alavijeh, M.; Shafiei, M.; Karimi, K. A Comprehensive Review on Bioethanol Production from Corn Stover: Worldwide Potential, Environmental Importance, and Perspectives. Biomass Bioenergy 2022, 161, 106447. [Google Scholar] [CrossRef]
- Kuglarz, M.; Gunnarsson, I.B.; Svensson, S.E.; Prade, T.; Johansson, E.; Angelidaki, I. Ethanol Production from Industrial Hemp: Effect of Combined Dilute Acid/Steam Pretreatment and Economic Aspects. Bioresour. Technol. 2014, 163, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.I.; Silva, C.C.; Mussatto, S.I.; Osseweijer, P.; van der Wielen, L.A.M.; Posada, J.A. Integrated 1st and 2nd Generation Sugarcane Bio-Refinery for Jet Fuel Production in Brazil: Techno-Economic and Greenhouse Gas Emissions Assessment. Renew. Energy 2018, 129, 733–747. [Google Scholar] [CrossRef]
- Bello, S.; Galán-Martín, Á.; Feijoo, G.; Moreira, M.T.; Guillén-Gosálbez, G. BECCS Based on Bioethanol from Wood Residues: Potential towards a Carbon-Negative Transport and Side-Effects. Appl. Energy 2020, 279, 115884. [Google Scholar] [CrossRef]
- Cavalaglio, G.; Gelosia, M.; Giannoni, T.; Barros Lovate Temporim, R.; Nicolini, A.; Cotana, F.; Bertini, A. Acid-Catalyzed Steam Explosion for High Enzymatic Saccharification and Low Inhibitor Release from Lignocellulosic Cardoon Stalks. Biochem. Eng. J. 2021, 174, 108121. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).