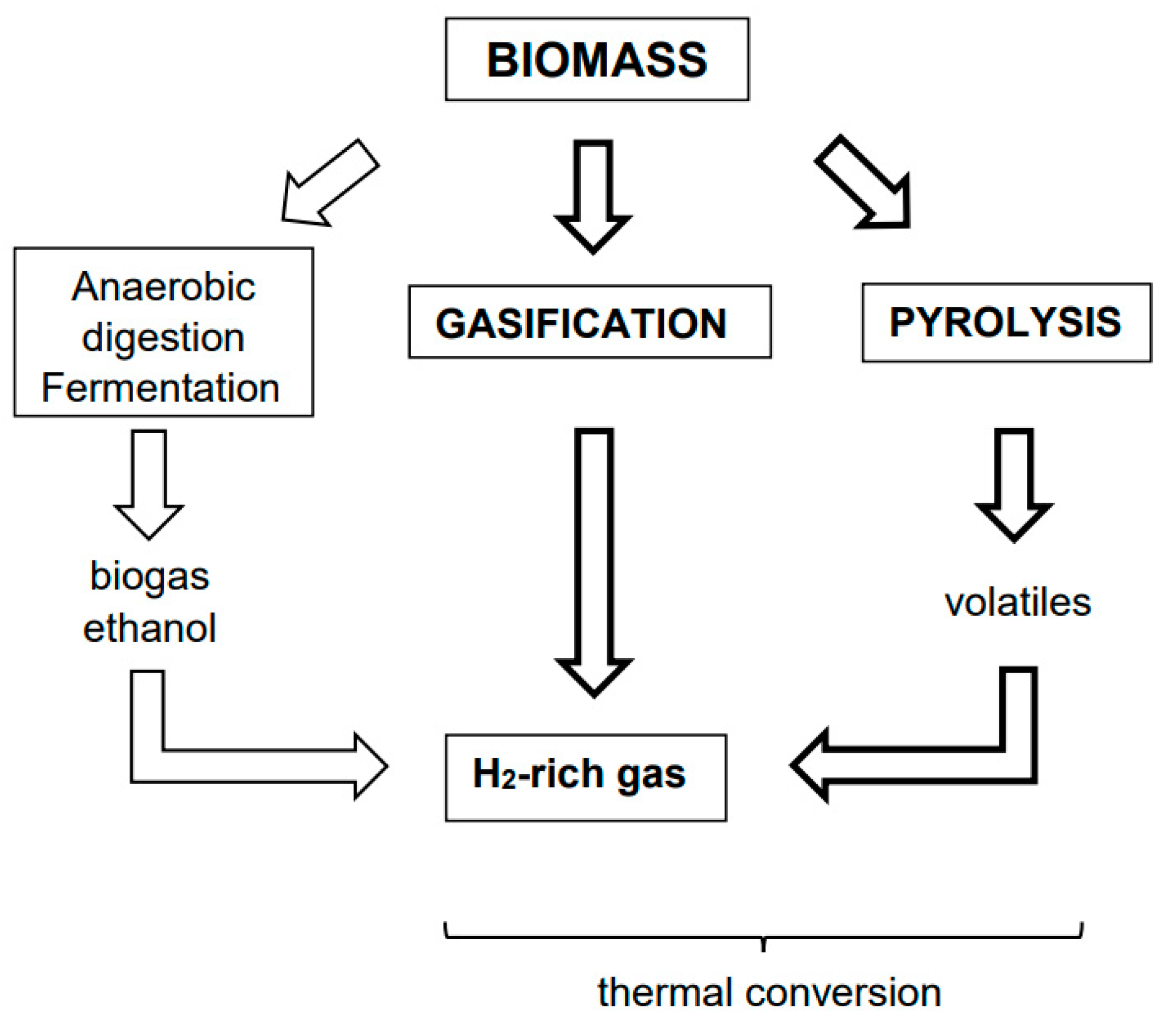
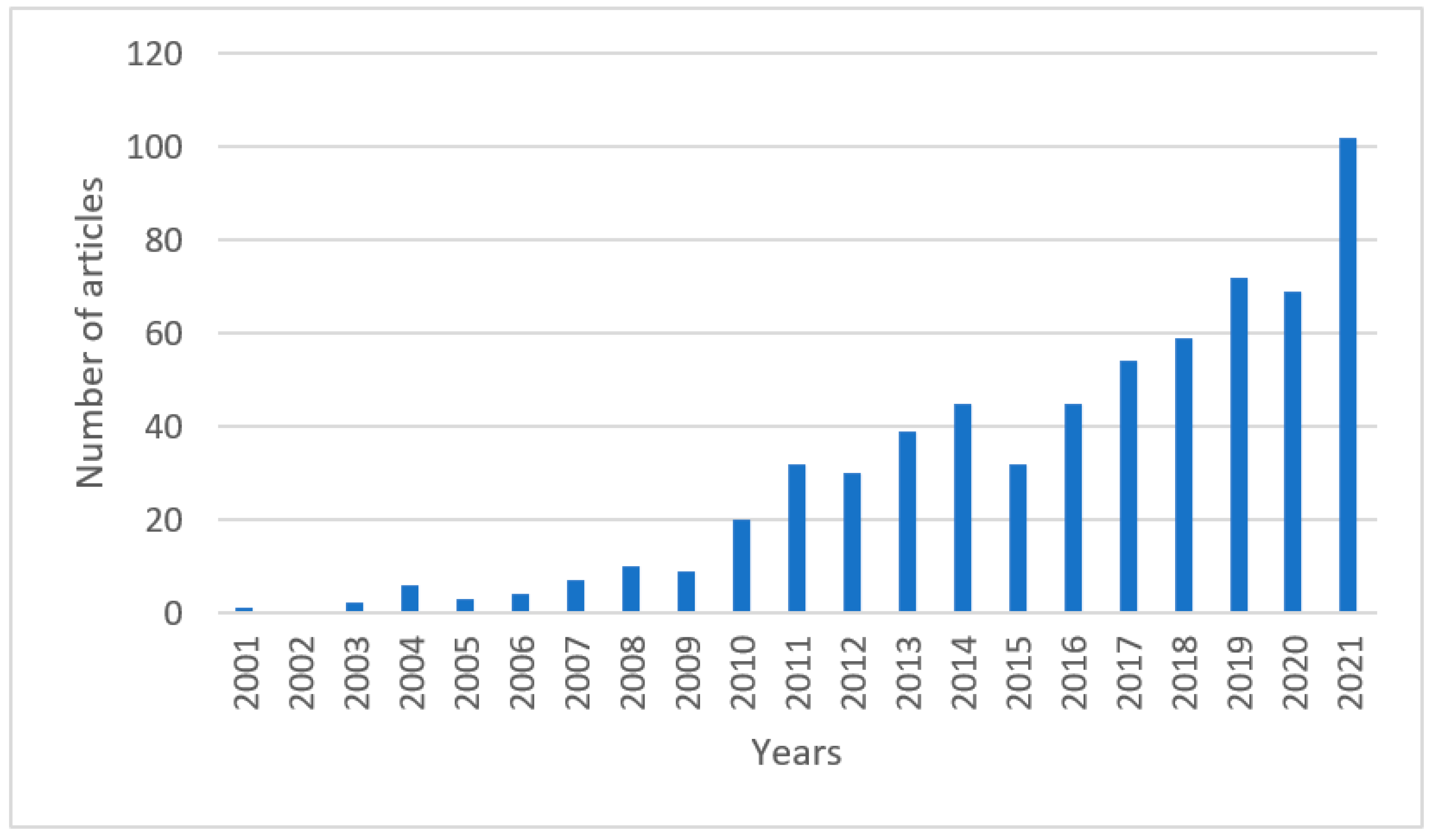
Upgrading of Lignocellulosic Biomass to Hydrogen-Rich Gas
Conflicts of Interest
References
- Dou, B.; Zhang, H.; Song, Y.; Zhao, L.; Jiang, B.; He, M.; Ruan, C.; Chen, H.; Xu, Y. Hydrogen production from the thermochemical conversion of biomass: Issues and challenges. Sustain. Energy Fuels 2019, 3, 314–342. [Google Scholar] [CrossRef]
- Setiabudi, H.; Aziz, M.; Abdullah, S.; Teh, L.; Jusoh, R. Hydrogen production from catalytic steam reforming of biomass pyrolysis oil or bio-oil derivatives: A review. Int. J. Hydrogen Energy 2020, 45, 18376–18397. [Google Scholar] [CrossRef]
- Glushkov, D.; Nyashina, G.; Shvets, A.; Pereira, A.; Ramanathan, A. Current Status of the Pyrolysis and Gasification Mechanism of Biomass. Energies 2021, 14, 7541. [Google Scholar] [CrossRef]
- Sarafraz, M.M.; Safaei, M.R.; Jafarian, M.; Goodarzi, M.; Arjomandi, M. High Quality Syngas Production with Supercritical Biomass Gasification Integrated with a Water–Gas Shift Reactor. Energies 2019, 12, 2591. [Google Scholar] [CrossRef]
- Chojnacki, J.; Najser, J.; Rokosz, K.; Peer, V.; Kielar, J.; Berner, B. Syngas Composition: Gasification of Wood Pellet with Water Steam through a Reactor with Continuous Biomass Feed System. Energies 2020, 13, 4376. [Google Scholar] [CrossRef]
- Stąsiek, J.; Szkodo, M. Thermochemical Conversion of Biomass and Municipal Waste into Useful Energy Using Advanced HiTAG/HiTSG Technology. Energies 2020, 13, 4218. [Google Scholar] [CrossRef]
- Marcantonio, V.; Müller, M.; Bocci, E. A Review of Hot Gas Cleaning Techniques for Hydrogen Chloride Removal from Biomass-Derived Syngas. Energies 2021, 14, 6519. [Google Scholar] [CrossRef]
- Ferreira, S.; Monteiro, E.; Brito, P.; Vilarinho, C. A Holistic Review on Biomass Gasification Modified Equilibrium Models. Energies 2019, 12, 160. [Google Scholar] [CrossRef]
- Marcantonio, V.; Bocci, E.; Monarca, D. Development of a Chemical Quasi-Equilibrium Model of Biomass Waste Gasification in a Fluidized-Bed Reactor by Using Aspen Plus. Energies 2020, 13, 53. [Google Scholar] [CrossRef]
- Marcantonio, V.; Monarca, D.; Villarini, M.; Di Carlo, A.; Del Zotto, L.; Bocci, E. Biomass Steam Gasification, High-Temperature Gas Cleaning, and SOFC Model: A Parametric Analysis. Energies 2020, 13, 5936. [Google Scholar] [CrossRef]
- González-Vázquez, M.P.; Rubiera, F.; Pevida, C.; Pio, D.; Tarelho, L. Thermodynamic Analysis of Biomass Gasification Using Aspen Plus: Comparison of Stoichiometric and Non-Stoichiometric Models. Energies 2021, 14, 189. [Google Scholar] [CrossRef]
- Moretti, L.; Arpino, F.; Cortellessa, G.; Di Fraia, S.; Di Palma, M.; Vanoli, L. Reliability of Equilibrium Gasification Models for Selected Biomass Types and Compositions: An Overview. Energies 2022, 15, 61. [Google Scholar] [CrossRef]
- Ayub, H.; Park, S.; Binns, M. Biomass to Syngas: Modified Non-Stoichiometric Thermodynamic Models for the Downdraft Biomass Gasification. Energies 2020, 13, 5668. [Google Scholar] [CrossRef]
- Sieradzka, M.; Gao, N.; Quan, C.; Mlonka-Mędrala, A.; Magdziarz, A. Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture. Energies 2020, 13, 1050. [Google Scholar] [CrossRef]
- Grams, J.; Ruppert, A.M. Development of heterogeneous catalysts for thermo-chemical conversion of lignocellulosic biomass. Energies 2017, 10, 545. [Google Scholar] [CrossRef]
- Grams, J.; Ruppert, A.M. Catalyst Stability—Bottleneck of Efficient Catalytic Pyrolysis. Catalysts 2021, 11, 265. [Google Scholar] [CrossRef]
- Yang, S.; Chen, L.; Sun, L.; Xie, X.; Zhao, B.; Si, H.; Zhang, X.; Hua, D. Novel Ni–Al nanosheet catalyst with homogeneously embedded nickel nanoparticles for hydrogen-rich syngas production from biomass pyrolysis. Int. J. Hydrogen Energy 2021, 46, 1762–1776. [Google Scholar] [CrossRef]
- Grams, J.; Ryczkowski, R.; Chalupka, K.A.; Sobczak, I.; Rzeznicka, I.I.; Przybysz, K. Impact of support (MCF, ZrO2, ZSM-5) on the efficiency of ni catalyst in high-temperature conversion of lignocellulosic biomass to hydrogen-rich gas. Materials 2019, 12, 3792. [Google Scholar] [CrossRef]
- Ryczkowski, R.; Jędrzejczyk, M.; Michalkiewicz, B.; Słowik, G.; Kwapiński, W.; Ruppert, A.M.; Grams, J. Impact of the modification method of Ni/ZrO2 catalyst by alkali and alkaline earth metals on its activity in thermo-chemical conversion of cellulose. Int. J. Hydrogen Energy 2018, 43, 22303–22314. [Google Scholar] [CrossRef]
- Ryczkowski, R.; Chałupka, K.; Kwapinski, W.; Przybysz, K.; Fridrichová, D.; Grams, J. Modification of Ni/ZrO2 catalyst by selected rare earth metals as a promising way for increase in the efficiency of thermocatalytic conversion of lignocellulosic biomass to hydrogen-rich gas. Fuel 2020, 276, 118110. [Google Scholar] [CrossRef]
- Ryczkowski, R.; Niewiadomski, M.; Michalkiewicz, B.; Skiba, E.; Ruppert, A.M.; Grams, J. Effect of alkali and alkaline earth metals addition on Ni/ZrO2 catalyst activity in cellulose conversion. J. Therm. Anal. Calorim. 2016, 126, 103–110. [Google Scholar] [CrossRef]
- Grams, J.; Ryczkowski, R.; Sadek, R.; Chałupka, K.; Przybysz, K.; Casale, S.; Dzwigaj, S. Hydrogen-rich gas production by upgrading of biomass pyrolysis vapors over NiBEA catalyst: Impact of dealumination and preparation method. Energy Fuels 2020, 34, 16936–16947. [Google Scholar] [CrossRef]
- Ryczkowski, R.; Goscianska, J.; Panek, R.; Franus, W.; Przybysz, K.; Grams, J. Sustainable nickel catalyst for the conversion of lignocellulosic biomass to H2-rich gas. Int. J. Hydrogen Energy 2021, 46, 10708–10722. [Google Scholar] [CrossRef]
- Santamaria, L.; Arregi, A.; Alvarez, J.; Artetxe, M.; Amutio, M.; Lopez, G.; Bilbao, J.; Olazar, M. Performance of a Ni/ZrO2 catalyst in the steam reforming of the volatiles derived from biomass pyrolysis. J. Anal. Appl. Pyrolysis 2018, 136, 222–231. [Google Scholar] [CrossRef]
- Santamaria, L.; Arregi, A.; Lopez, G.; Artetxe, M.; Amutio, M.; Bilbao, J.; Olazar, M. Effect of La2O3 promotion on a Ni/Al2O3 catalyst for H2 production in the in-line biomass pyrolysis-reforming. Fuel 2020, 262, 116593. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grams, J. Upgrading of Lignocellulosic Biomass to Hydrogen-Rich Gas. Energies 2023, 16, 72. https://doi.org/10.3390/en16010072
Grams J. Upgrading of Lignocellulosic Biomass to Hydrogen-Rich Gas. Energies. 2023; 16(1):72. https://doi.org/10.3390/en16010072
Chicago/Turabian StyleGrams, Jacek. 2023. "Upgrading of Lignocellulosic Biomass to Hydrogen-Rich Gas" Energies 16, no. 1: 72. https://doi.org/10.3390/en16010072
APA StyleGrams, J. (2023). Upgrading of Lignocellulosic Biomass to Hydrogen-Rich Gas. Energies, 16(1), 72. https://doi.org/10.3390/en16010072




