Preliminary Studies of Slag and Ash from Incinerated Municipal Waste for Prospective Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Waste Material
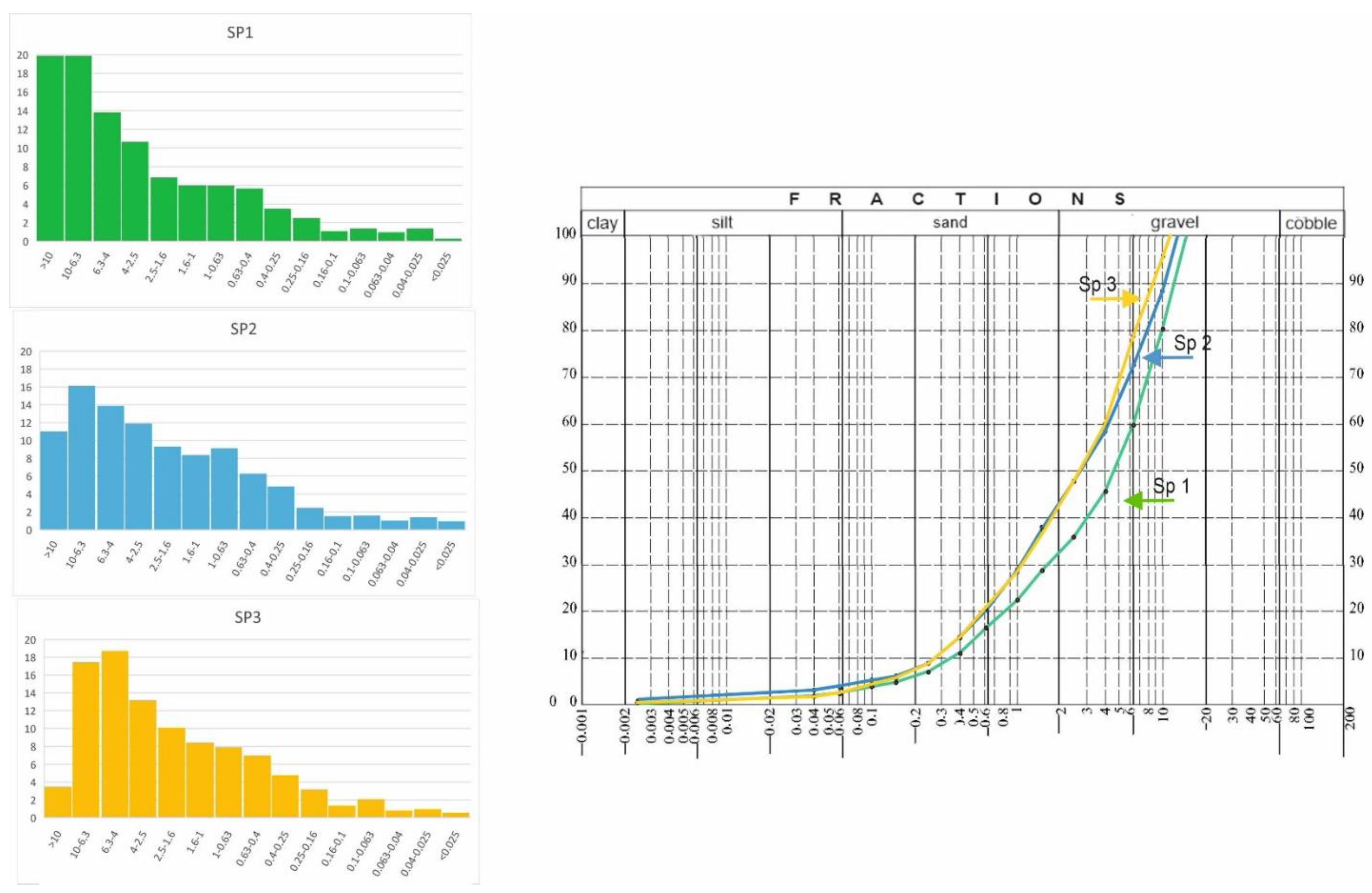
2.2. Granulometric Analysis
2.3. Microscopic Analysis
- —number of measurement points;
- —coefficient read from normal distribution tables;
- —permissible relative error;
- —the least numerous component of the analyzed phase (here: pore content).
2.4. BCR Sequential Extraction
2.5. Densimetric Studies
2.6. Gas Permeability Tests
- P1—pressure at the inlet of the sample, Pa;
- P0—pressure at the outlet of the sample, Pa;
- Q—gas flow rate, m3/s;
- —gas viscosity coefficient, Pa·s;
- L—sample length, m;
- A—ample cross-sectional area, m2.
2.7. Water Permeability Tests
- Q—filtration output, m3;
- A—sample cross-sectional area, m2;
- i—hydraulic gradient;
- t—measurement time, s.
3. Results
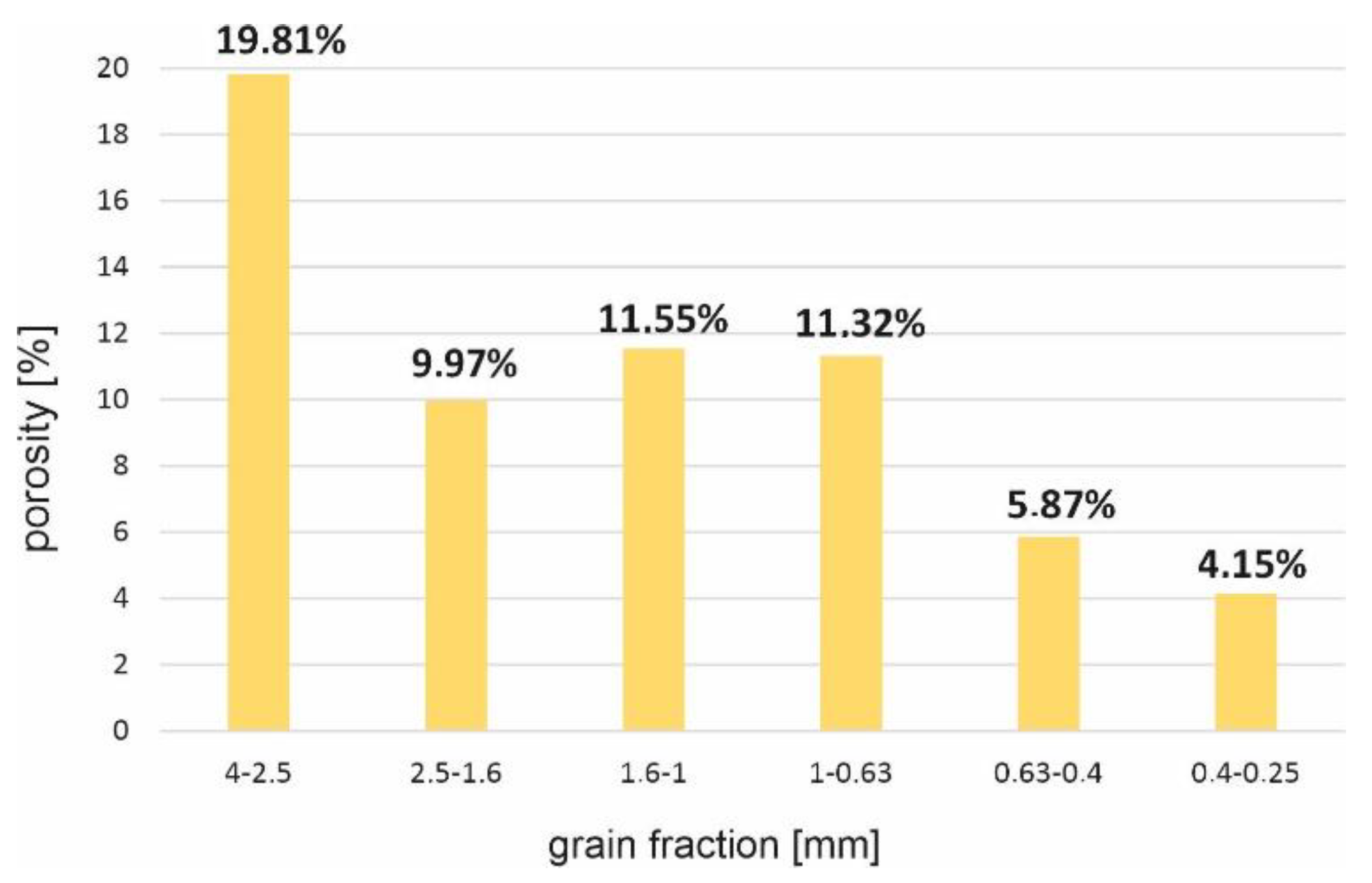
3.1. Granulometric Analysis
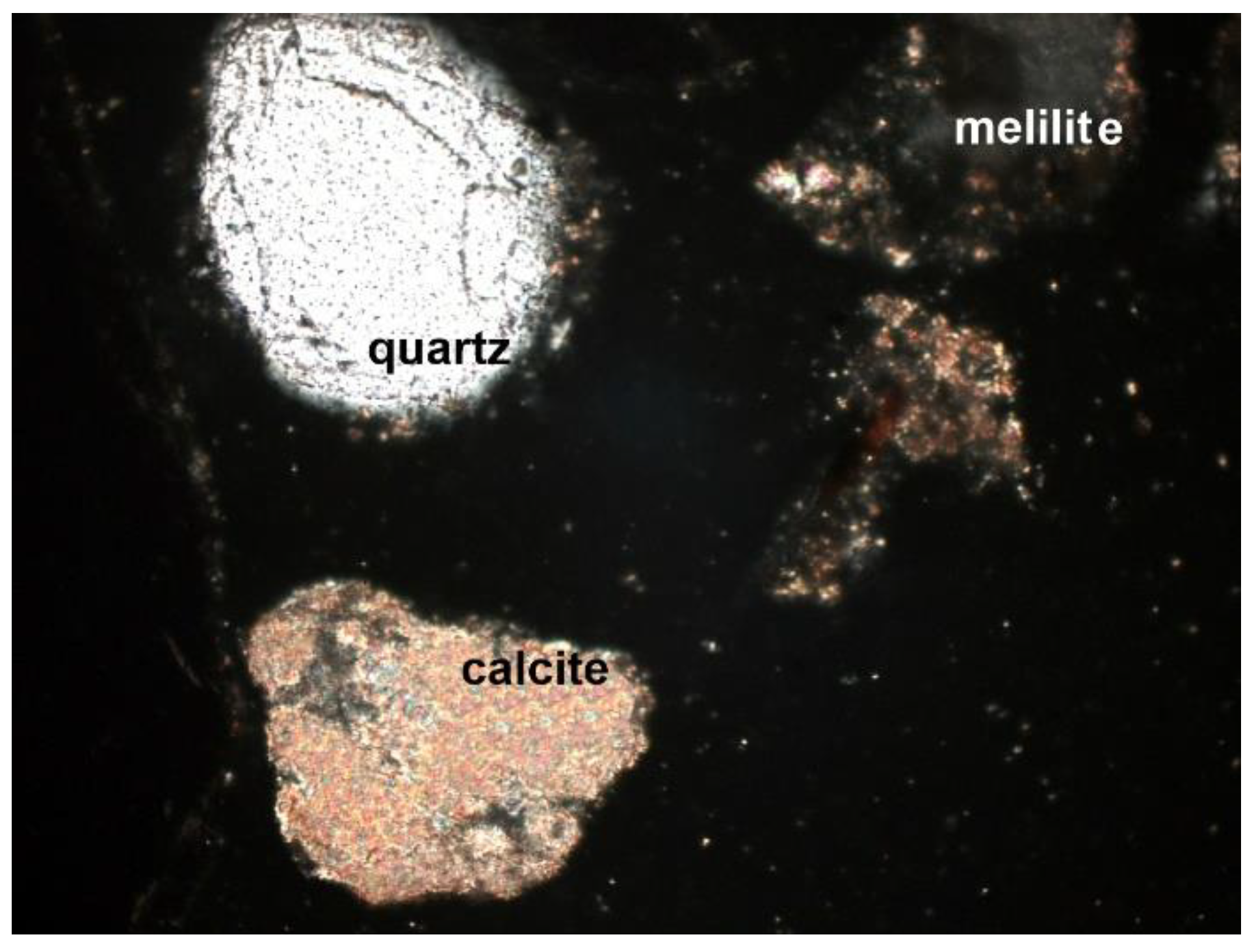
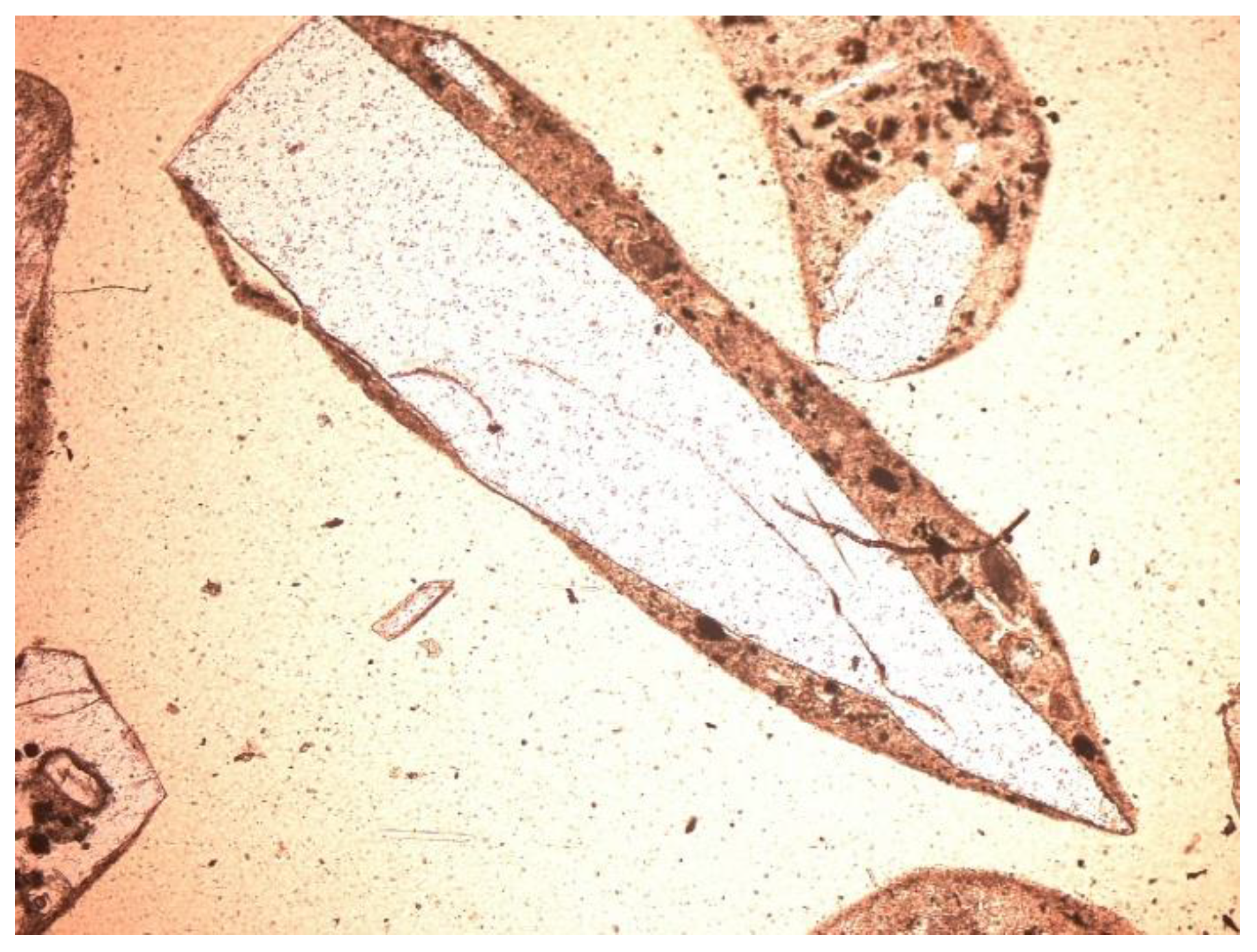
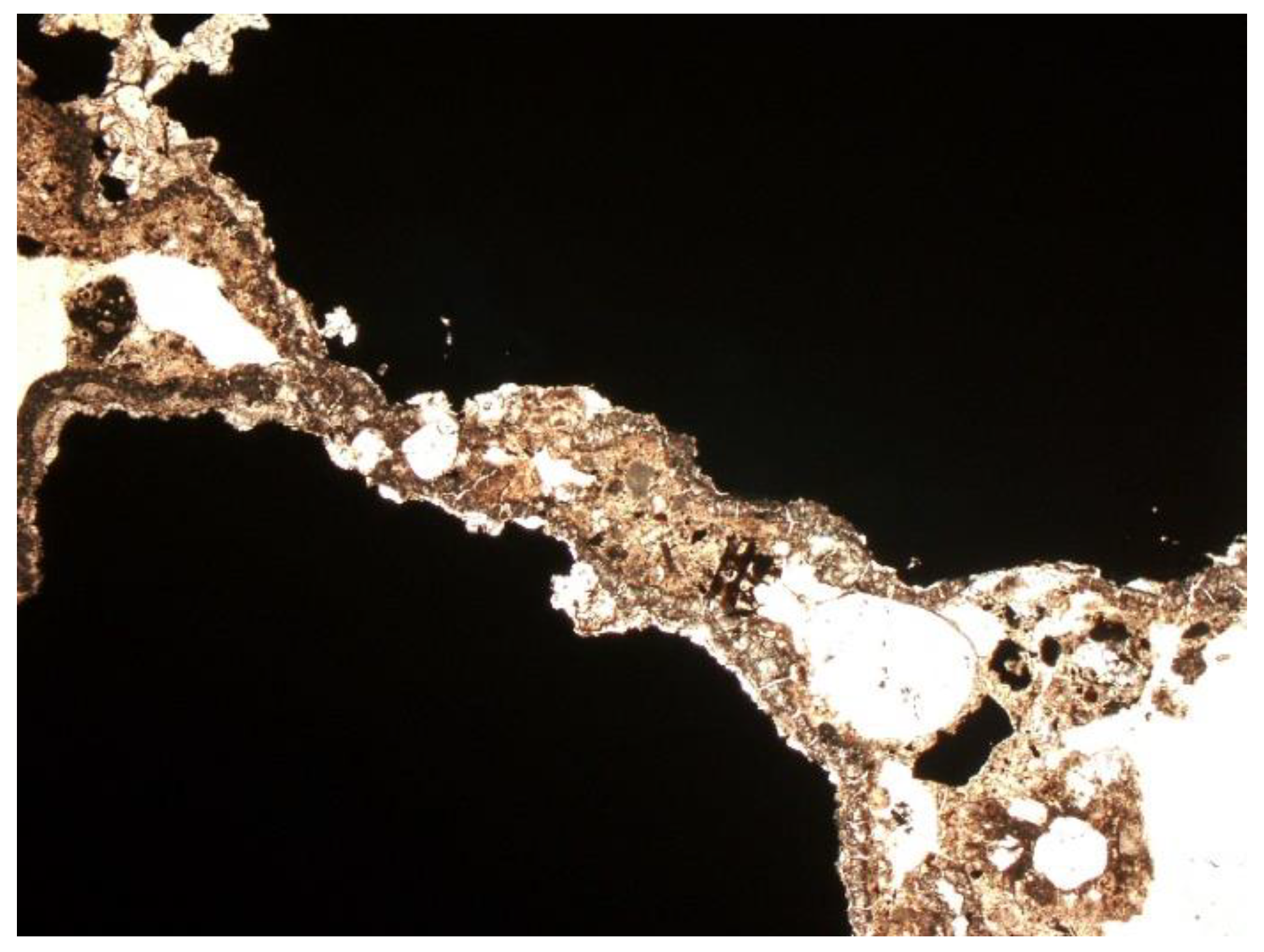
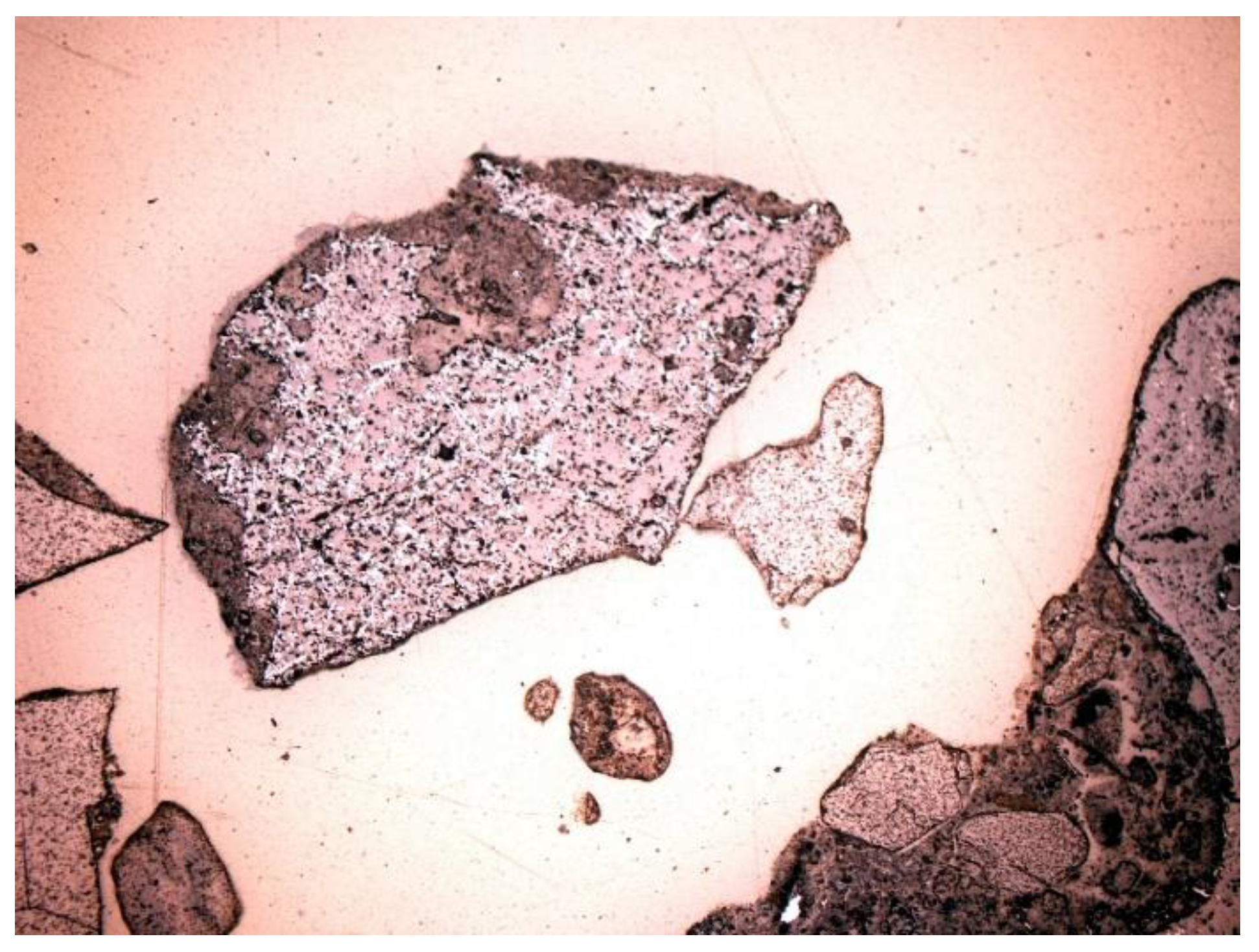
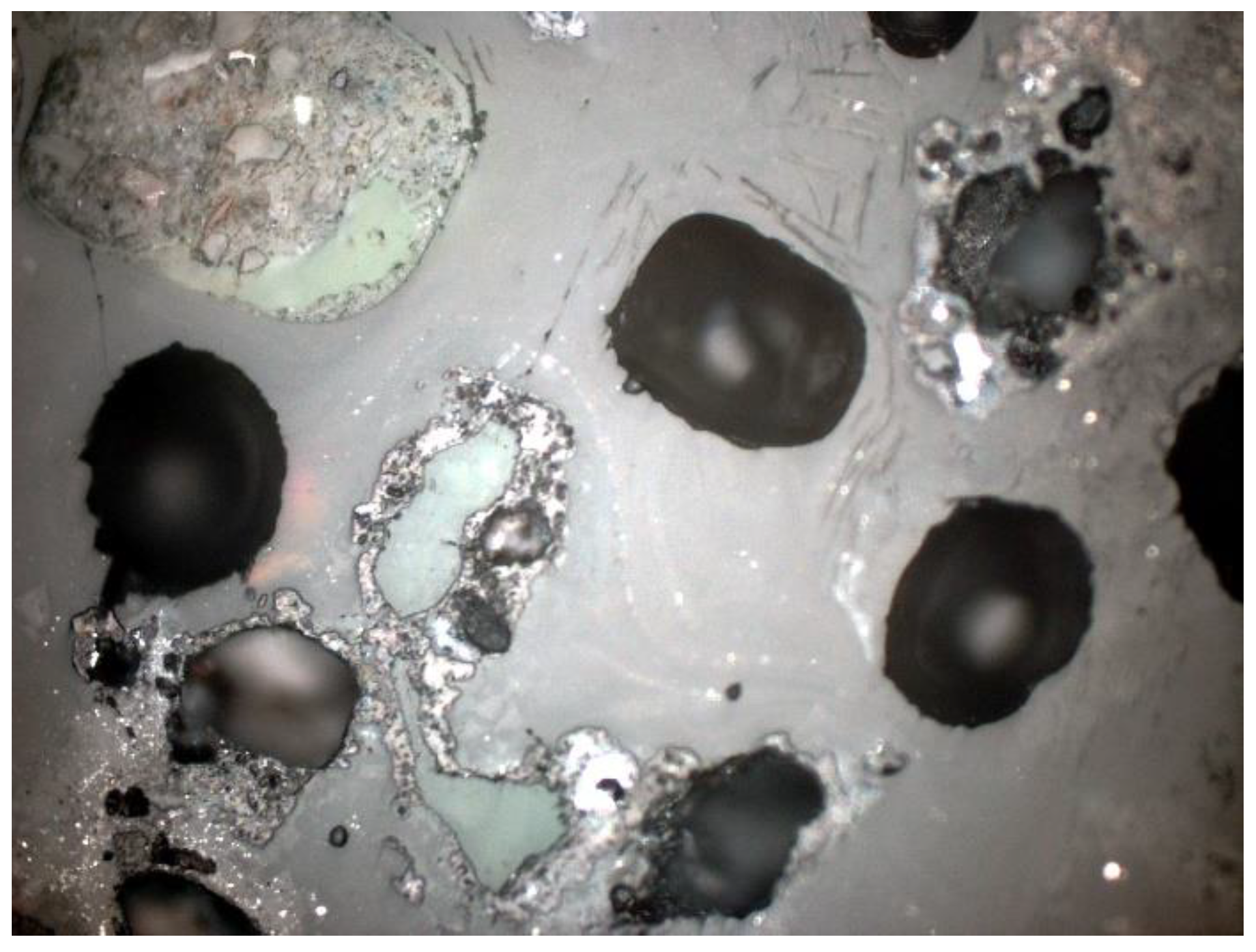
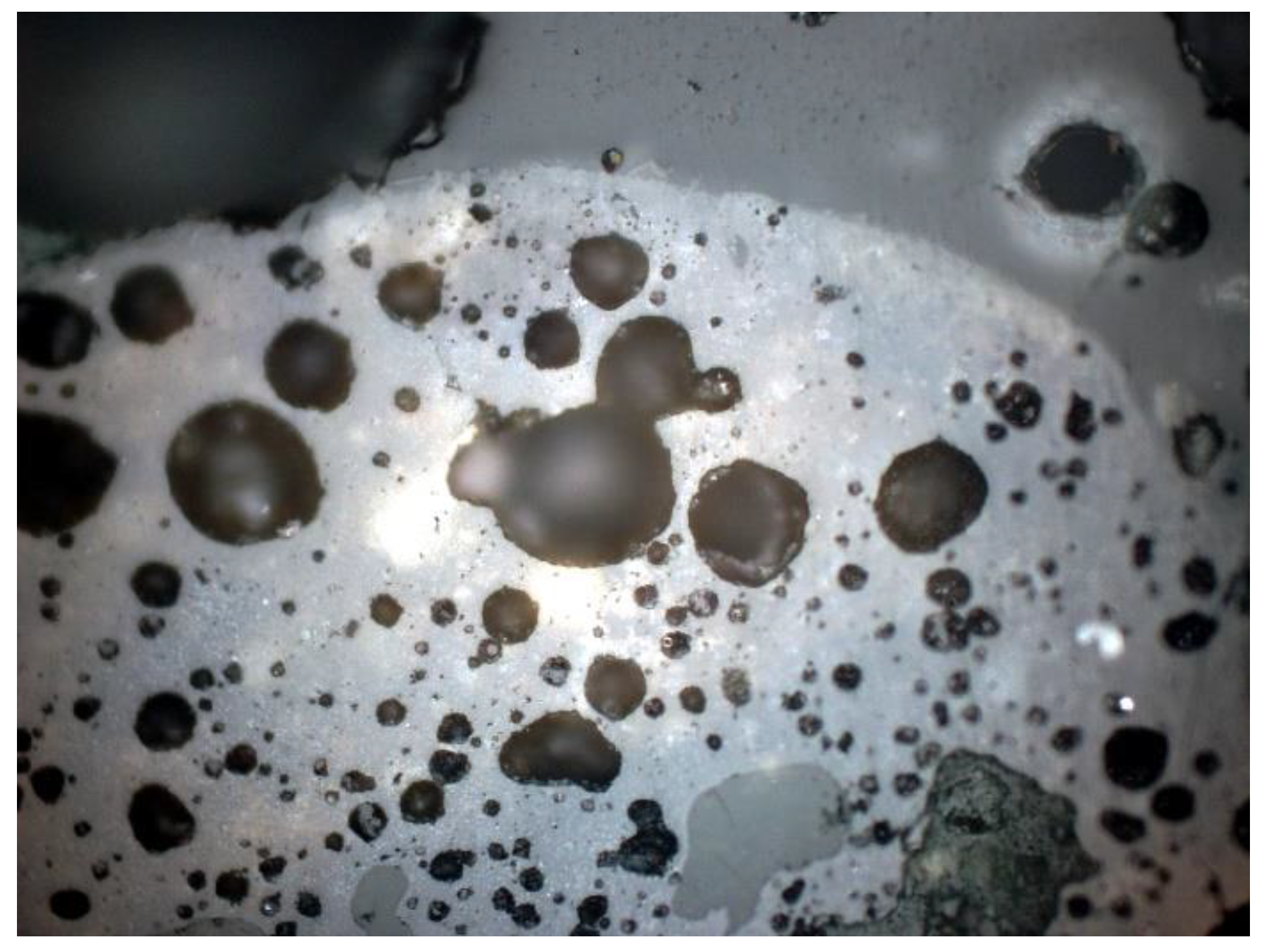
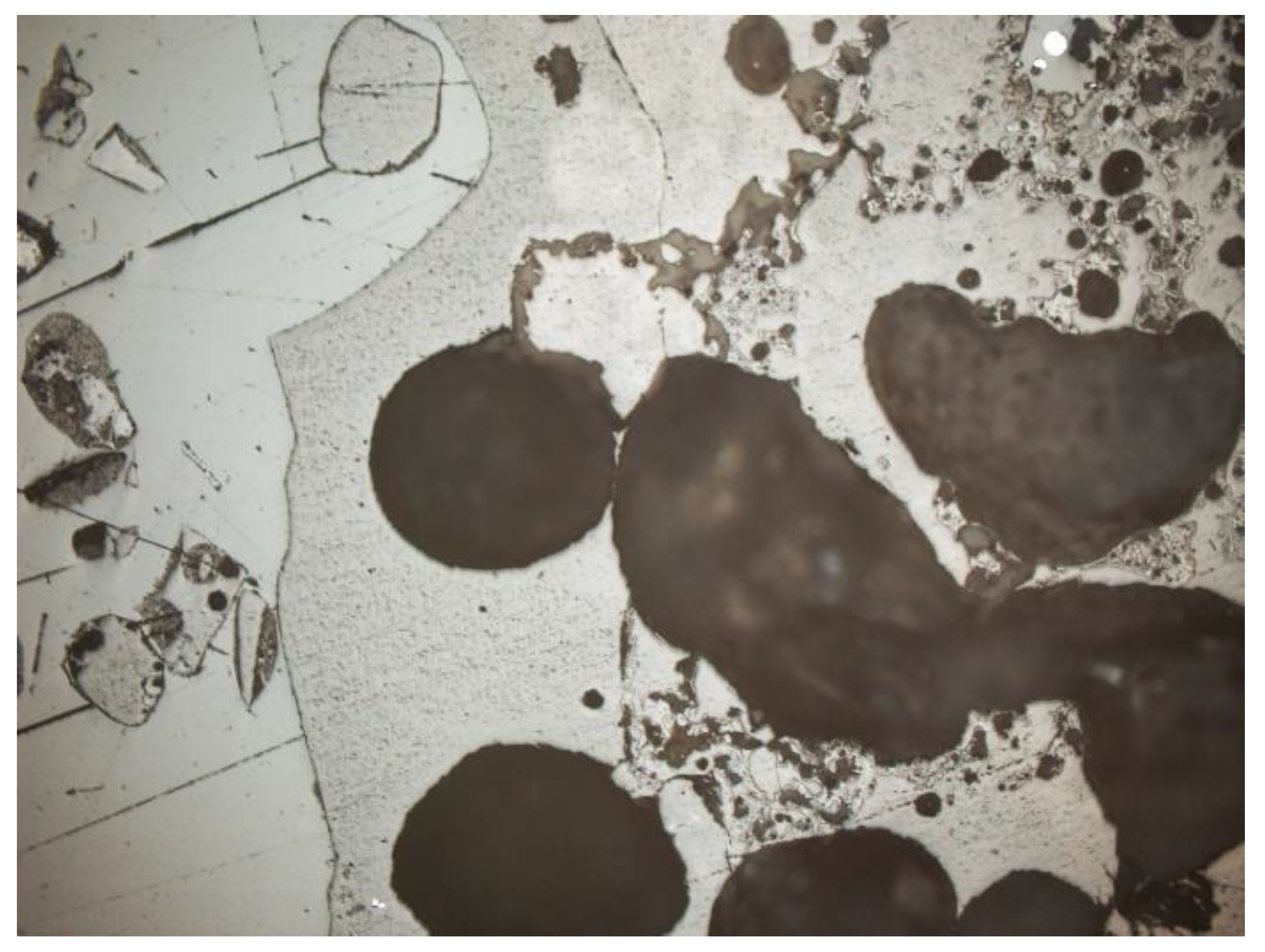
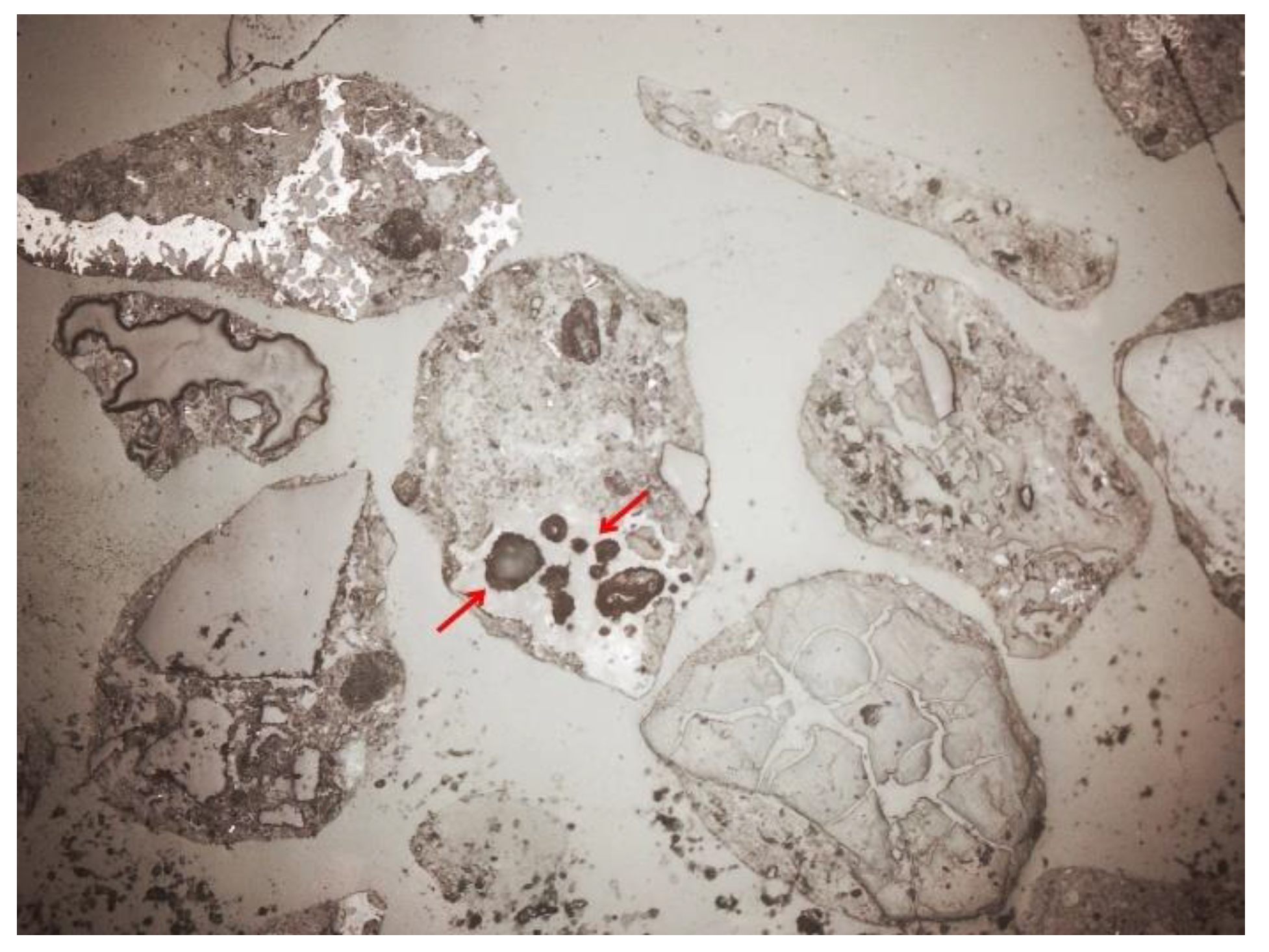
3.2. Microscopic Analysis—Qualitative Description
- -
- Quartz (SiO2): the dominant component of the mineral phase in each sample. It was in the form of oval, rounded, slightly cracked grains (Figure 6, Figure 7, Figure 8 and Figure 9). It showed gray interference colors of the first order. This mineral, due to the action of high temperature, showed wavy dimming.
- -
- Melilite: Ca2Mg and Ca2Al group silicates. The extreme links of the melilite series were akermanite Ca2Mg[Si2O7] and gehlenite Ca2Al[(SiAl)2O7]. Minerals formed at high temperature, found in all samples (Figure 7, Figure 8 and Figure 9). The mineral interference colors were low of the first order. There was an oblique dimming, and the surface was cracked, with little cleavage. This mineral is usually irregular or slightly oval in shape.
- -
- -
- Feldspars—spatial aluminosilicates of K, Na, and Ca, and less often Ba. Numerous grains of potassium feldspar and plagioclase were found in the samples. All feldspars had gray, low first-order interference colors, visible cleavage, and multiple or singly twinned crystals. The group of feldspars includes a number of minerals with different chemical compositions and different forms of crystals. In the analyzed slag and ash samples, sometimes numerous grains of potassium feldspars and plagioclases were found. The crystal habit was often subhedral, tabular. The material showed, for example, multiple twinned plagioclases (Figure 10) and potassium feldspars—microcline (Figure 11) and orthoclase (Figure 12 and Figure 13).
3.3. Microscopic Analysis—Quantitative Description
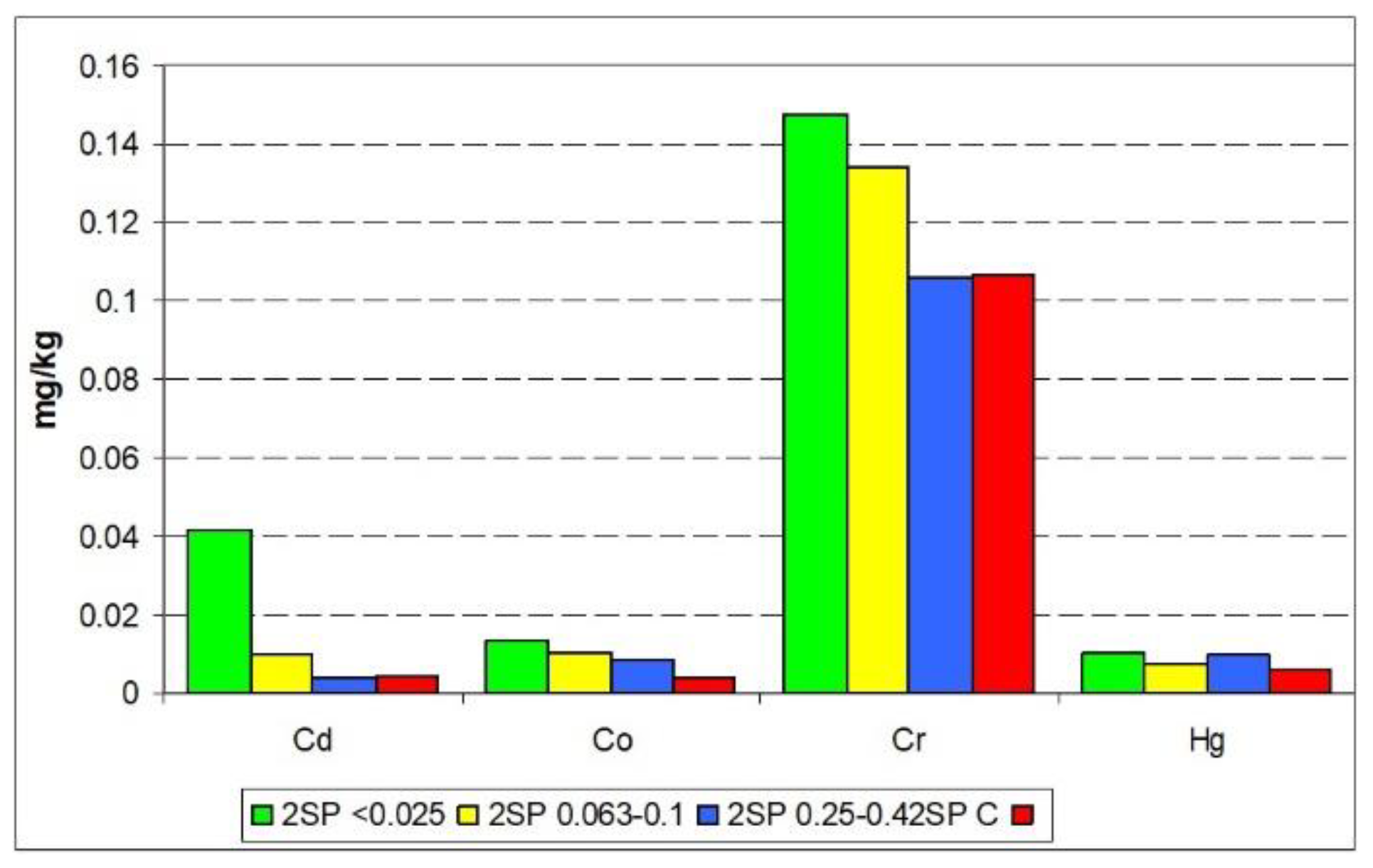
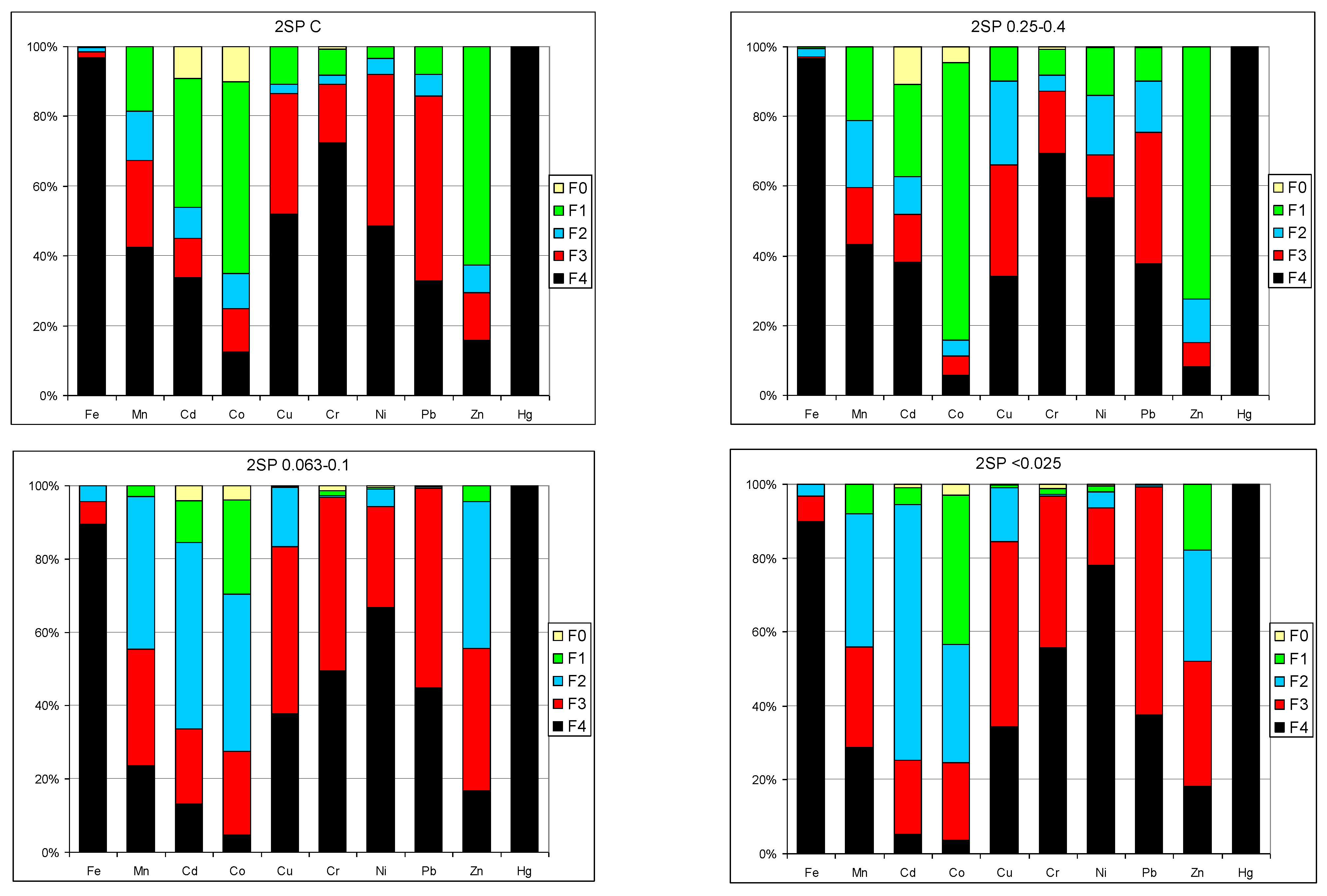
3.4. Sequential Extraction
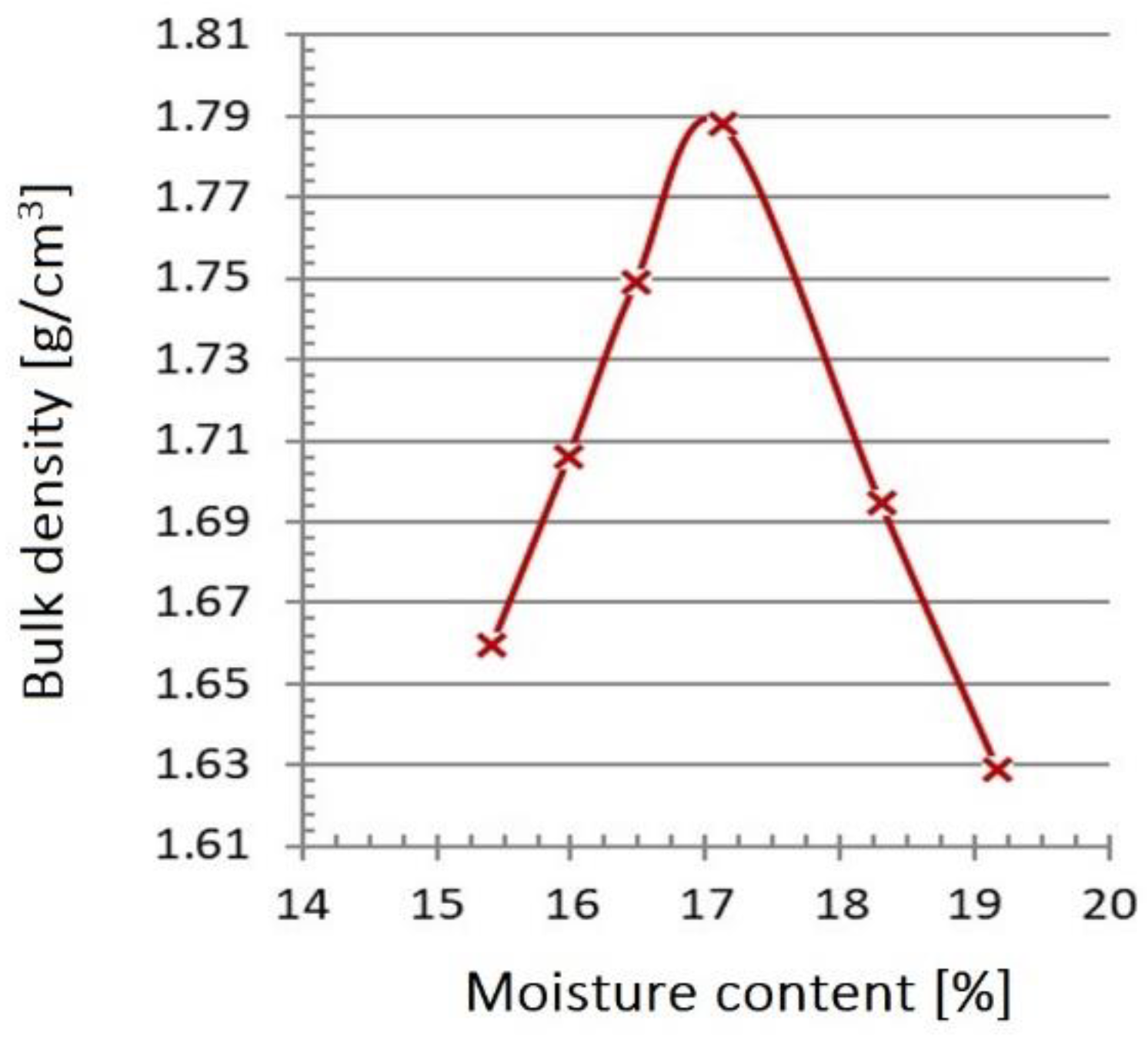
3.5. Densimetric Studies
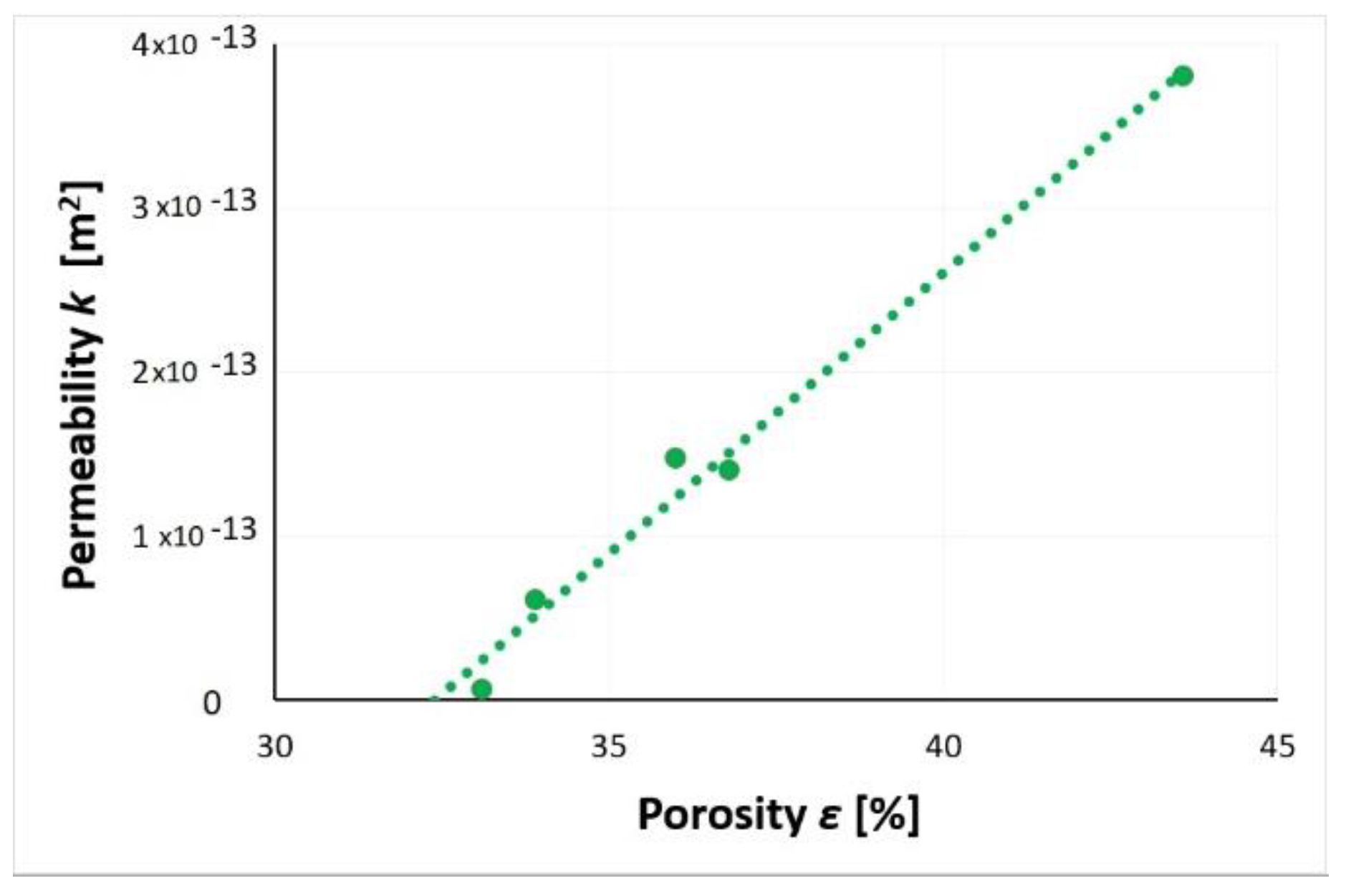
3.6. Gas Permeability
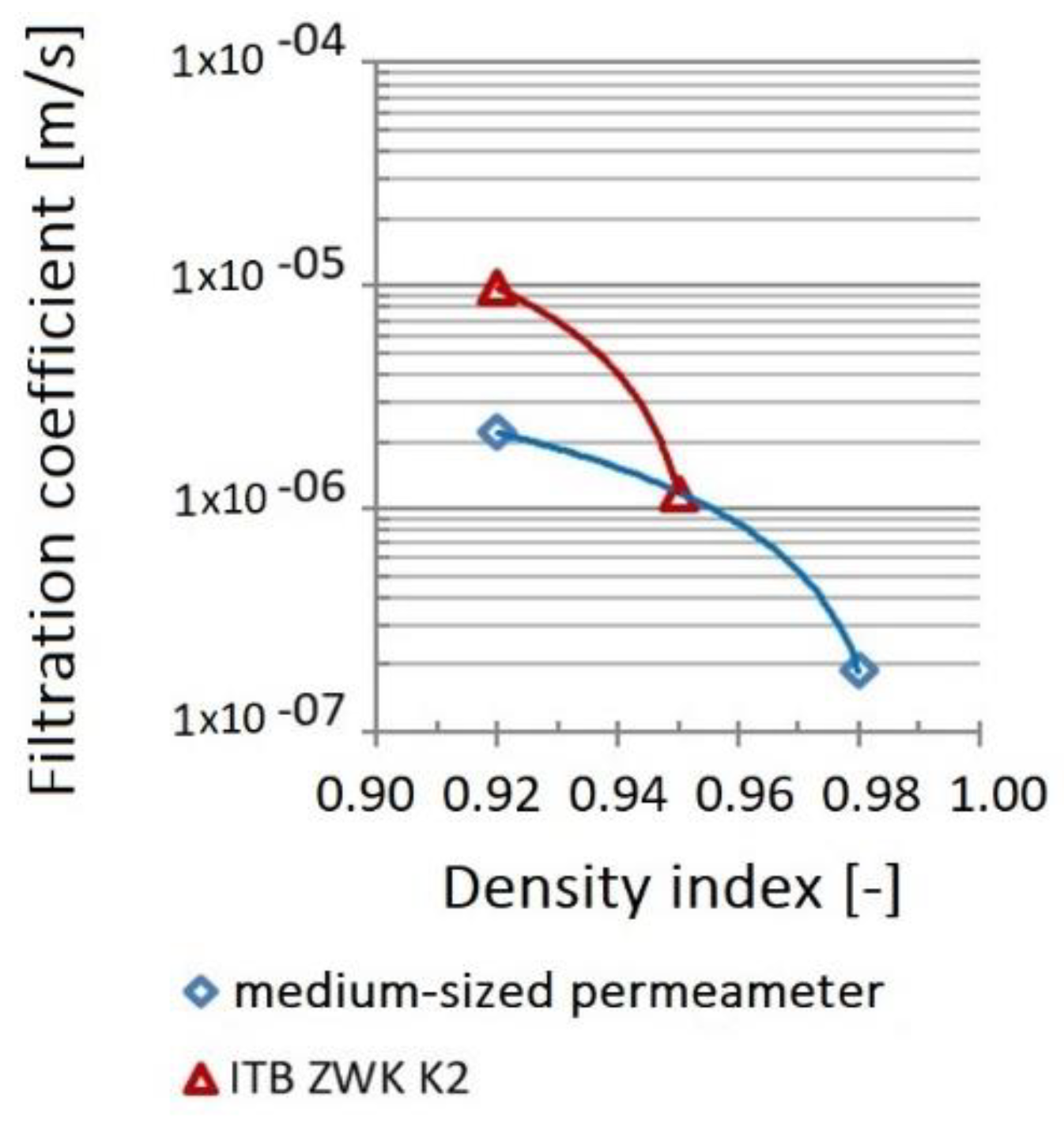
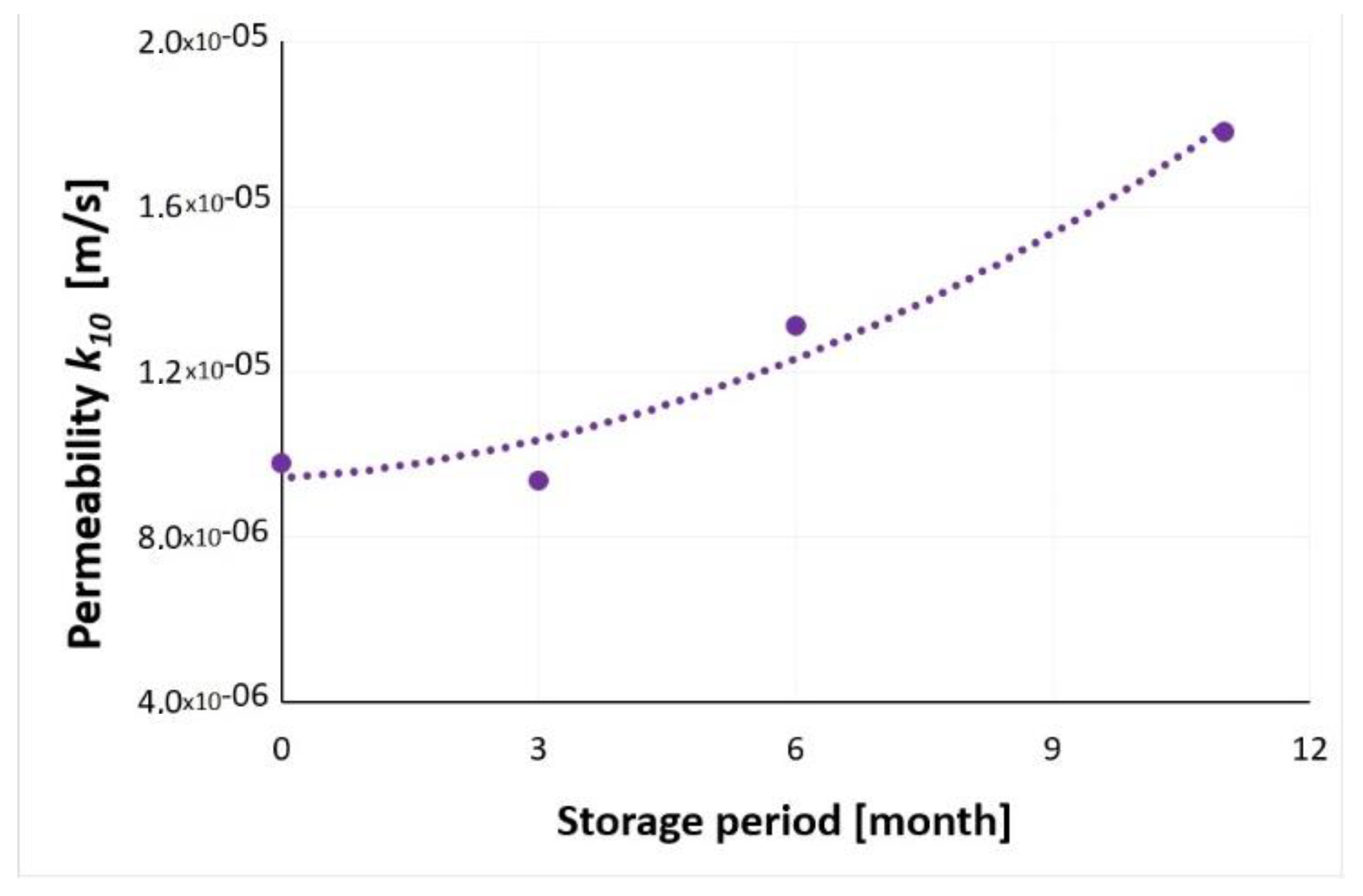
3.7. Water Permeability
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mądrawski, J.; Kostrzewski, W.; Smoczkiewicz-Wojciechowska, A. Problematyka wykorzystania odpadów typu żużle ze spalarni śmieci komunalnych do produkcji betonów. Przegląd. Bud. 2017, 88, 105–107. [Google Scholar]
- de Lorena Diniz Chaves, G.; Ribeiro Siman, R.; Mattos Ribeiro, G.; Chang, N.-B. Synergizing environmental, social, and economic sustainability factors for refuse derived fuel use in cement industry: A case study in Espirito Santo, Brazil. J. Environ. Manag. 2021, 288, 112401. [Google Scholar] [CrossRef] [PubMed]
- Musiał, M.; Pękala, A. Functioning of Heat Accumulating Composites of Carbon Recyclate and Phase Change Material. Materials 2022, 15, 2331. [Google Scholar] [CrossRef] [PubMed]
- Baran, P.; Nazarko, M.; Włosińska, E.; Kanciruk, A.; Zarębska, K. Synthesis of geopolymers derived from fly ash with an addition of perlite. J. Clean. Prod. 2021, 293, 126112. [Google Scholar] [CrossRef]
- Zarębska, K.; Zabierowski, P.; Gazda-Grzywacz, M.; Czuma, N.; Baran, P. Fly ash-based geopolymers with refractoriness properties. Clean Technol. Environ. Policy 2022, 24, 2161–2175. [Google Scholar] [CrossRef]
- 2021. Available online: https://khk.krakow.pl (accessed on 23 October 2022).
- Gluzińska, J.; Walawska, B.; Łuczkowska, D.; Pajdak, A. Properties of waste fly ash as a hard coal combustion by-product after the application of dry sodium sorbents to purify flue gases. Trans. Strat. Mech. Res. Inst. 2016, 18, 83–91. [Google Scholar]
- Pajdak, A.; Szymanek, A. The characteristics and the concept of the utilisation of post-production calcareous waste deposited in landfills. Mineralogia 2017, 48, 167–179. [Google Scholar] [CrossRef]
- Wielgosinski, G. Termiczne przekształcanie odpadów. Wydawnictwo Nowa Energia. 2020. Available online: https://nowa-energia.com.pl/wydawnictwa-ksiazkowe/ (accessed on 23 October 2022).
- Vehlow, J.; Bergfeldt, B.; Wilen, C.; Ran-ta, J.; Schwaiger, H.; Visser, H.J.M.; Gu, S.; Gyftopoulou, E.; Brammer, J. Management of Solid Residues in Waste-to-Energy and Biomass Systems. Forschungszentrum Karlsruhe (FZKA 7347), Karlsruhe. 2007. Available online: https://cris.vtt.fi/en/publications/management-of-solid-residues-in-waste-to-energy-and-biomass-syste (accessed on 23 October 2022).
- Blajer, M.; Stopkowicz, A.; Adamczyk, J.; Cała, M. The preliminary research of the physico-mechanical properties of aggregates based on the colliery shale, supplemented by fly ash. Arch. Min. Sci. 2019, 64, 21–34. [Google Scholar]
- PN-EN 12457-(1–4). Charakteryzowanie Odpadów. Wymywanie. Badanie Zgodności w Odniesieniu do Wymywania Ziarnistych Materiałów Odpadowych i Osadów. 2006. Available online: https://sklep.pkn.pl/pn-en-12457-4-2006p.html (accessed on 23 October 2022).
- Wielgosiński, G.; Wasiak, D. Politechnika Łódzka Wydział Inżynierii Procesowej i Ochrony Środowiska. 2014. Available online: https://sdr.gdos.gov.pl/Documents/GO/Spotkanie%2029.04.2014/WIELGOSINSKI---Badania.pdf (accessed on 23 October 2022).
- Tessier, A.; Campbell, P.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metal. Anal. Chem. Acta 1979, 286, 423–442. [Google Scholar] [CrossRef]
- Wielgosiński, G.; Wasiak, D.; Zawadzka, A. The Use of Sequential Extraction for Assessing Environmental Risks of Waste Incineration Bottom Ash. Ecol. Chem. Eng. S 2014, 21, 413–423. [Google Scholar] [CrossRef]
- van Herck, P.; Vandecasteele, C. Evaluation of the use of a sequential extraction procedure for the characterization and treatment of metal containing solid waste. Waste Manag. 2001, 21, 685–694. [Google Scholar] [CrossRef]
- Sutherland, R.A. BCR®-701: A review of 10-years of sequential extraction analyses. Anal. Chim. Acta 2010, 680, 10–20. [Google Scholar] [CrossRef]
- Sakita, S.; Nishimoto, J.; Nishimura, K. Porous Structure of Municipal Solid Waste Incineration Bottom Ash in Initial Stage of Landfill. J. Geosci. Environ. Prot. 2017, 5, 9–20. [Google Scholar] [CrossRef][Green Version]
- Ritzkowski, M.; Stegmann, R. Landfill Aeration within the Scope of Post-Closure Care and Its Completion. Waste Manag. 2013, 33, 2074–2082. [Google Scholar] [CrossRef]
- Pfrang-Stotz, G.; Schneider, J. Comparative studies of waste combustion bottom ashes from various grate and firing systems, conducted with respect to mineralogical and geochemical methods of examination. Waste Manag. Res. 1995, 13, 273–292. [Google Scholar] [CrossRef]
- Eusden, J.D.; Eighmy, T.T.; Hockert, K.; Holland, E.; Marsella, K. Petrogenesis of municipal solid waste combustion bottom ash. Appl. Geochem. 1999, 14, 1073–1091. [Google Scholar] [CrossRef]
- Bayuseno, A.P.; Schmahl, W.W. Understanding the chemical and mineralogical properties of the inorganic portion of MSWI bottom ash. Waste Manag. 2010, 30, 1509–1520. [Google Scholar] [CrossRef]
- Dutka, B.; Godyń, K. Coalification as a process determining the methane adsorption ability of coal seams. Arch. Min. Sci. 2021, 66, 181–195. [Google Scholar]
- Nowak-Winiarska, K.; Wrobel, S.; Sienkiewicz-Cholewa, U. Application of sequential analysis with the BCR method in the estimation of effects of chemical remediation of soil polluted with copper. Chem. Spec. Bioavailab. 2012, 24, 53. [Google Scholar] [CrossRef]
- Sutherland, R.A.; Tack, F.M.G. Determination of Al, Cu, Fe, Mn, Pb and Zn in certified reference materials using the optimized BCR sequential extraction procedure. Anal. Chim. Acta 2002, 454, 249. [Google Scholar] [CrossRef]
- Godyń, K.; Dutka, B.; Chuchro, M.; Młynarczuk, M. Synergy of parameters determining the optimal properties of coal as a natural sorbent. Energies 2020, 13, 1967. [Google Scholar] [CrossRef]
- Zeng, G.; Liu, L.; Xue, Q.; Wan, Y.; Ma, J.; Zhao, Y. Experimental study of the porosity and permeability of municipal solid waste. Environ. Prog. Sustain. Energy 2017, 36, 1694–1699. [Google Scholar] [CrossRef]
- Dutka, B. Effect of depth on the sorption capacity of coals affected by outburst hazard. Fuel 2021, 306, 121611. [Google Scholar] [CrossRef]
- Godyń, K.; Dutka, B. Slags and ashes generated in the process of incineration of municipal waste as one of the cogs of the circular economy. In Proceedings of SEEP 2022; Brunel University: London, UK, 2022; pp. 12–15. [Google Scholar]


























| Waste Sample | SP1 | SP2 | SP3 |
|---|---|---|---|
| Porosity (%) | 9.07 | 12.92 | 12.74 |
| Sample fraction (mm) | 4–2.5 | 2.5–1.6 | 1.6–1 | 1–0.63 | 0.63–0.4 | 0.4–0.25 |
| Porosity (%) | 19.81 | 9.97 | 11.55 | 11.32 | 5.87 | 4.15 |
| Slag and Ash | ε, % | Vp, cm3/g | ||
|---|---|---|---|---|
| SP1C | 2.564 | 1.951 | 23.9 | 0.123 |
| SP2C | 2.555 | 1.957 | 23.4 | 0.120 |
| SP3C | 2.562 | 1.965 | 23.3 | 0.119 |
| Sample Fraction (mm) | Vp, cm3/g | |||||
|---|---|---|---|---|---|---|
| 2 SP > 6.3 | 2.468 | 1.957 | 20.7 | 0.11 | 20.0 | 0.7 |
| 2 SP > 4 | 2.521 | 22.4 | 0.14 | 20.0 | 2.4 | |
| 2 SP > 2.5 | 2.527 | 22.6 | 0.16 | 19.8 | 2.8 | |
| 2 SP > 1.0 | 2.595 | 24.6 | 0.21 | 11.6 | 13.0 | |
| 2 SP > 0.63 | 2.564 | 23.7 | 0.18 | 11.3 | 12.4 | |
| 2 SP > 0.25 | 2.585 | 24.3 | 0.20 | 4.2 | 20.1 |
| Waste Sample | SP2 | ||||
|---|---|---|---|---|---|
| Consolidation force (kg) | 500 | 1000 | 1300 | 2600 | 4000 |
| Stress (MPa) | 6.7 | 13.5 | 17.5 | 35.0 | 53.0 |
| Layer density (g/cm3) | 1.44 | 1.62 | 1.63 | 1.69 | 1.71 |
| Porosity (%) | 43.6 | 36.8 | 36.0 | 33.9 | 33.1 |
| Permeability (m2) | 3.8 × 10−13 | 1.4 × 10−13 | 1.5 × 10−13 | 6.0 × 10−14 | 5.8 × 10−15 |
| Density Index | Apparatus | Storage Time | |||
|---|---|---|---|---|---|
| Without Storage | 3 Months | 6 Months | 11 Months | ||
| Filtration Coefficient, k10 [m/s] | |||||
| IS = 0.92 | Medium-dimensional permeameter | 2.20 × 10−6 | |||
| IS = 0.98 | 1.88 × 10−7 | ||||
| IS = 0.92 | ITB ZW K2 | 9.79 × 10−6 | 9.37 × 10−6 | 1.31 × 10−5 | 1.78 × 10−5 |
| IS = 0.95 | 9.46 × 10−6 | ||||
| IS = 0.98 | Water flow was not obtained | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Godyń, K.; Dutka, B. Preliminary Studies of Slag and Ash from Incinerated Municipal Waste for Prospective Applications. Energies 2023, 16, 117. https://doi.org/10.3390/en16010117
Godyń K, Dutka B. Preliminary Studies of Slag and Ash from Incinerated Municipal Waste for Prospective Applications. Energies. 2023; 16(1):117. https://doi.org/10.3390/en16010117
Chicago/Turabian StyleGodyń, Katarzyna, and Barbara Dutka. 2023. "Preliminary Studies of Slag and Ash from Incinerated Municipal Waste for Prospective Applications" Energies 16, no. 1: 117. https://doi.org/10.3390/en16010117
APA StyleGodyń, K., & Dutka, B. (2023). Preliminary Studies of Slag and Ash from Incinerated Municipal Waste for Prospective Applications. Energies, 16(1), 117. https://doi.org/10.3390/en16010117






