Biohydrogen Production in Microbial Electrolysis Cells Utilizing Organic Residue Feedstock: A Review
Abstract
1. Introduction
1.1. Hydrogen as the Product
1.1.1. Power-to-X and the Role of Hydrogen in Denmark
1.1.2. From Hydrogen to Ammonia
2. Current State-of-the-Art for MEC Technology
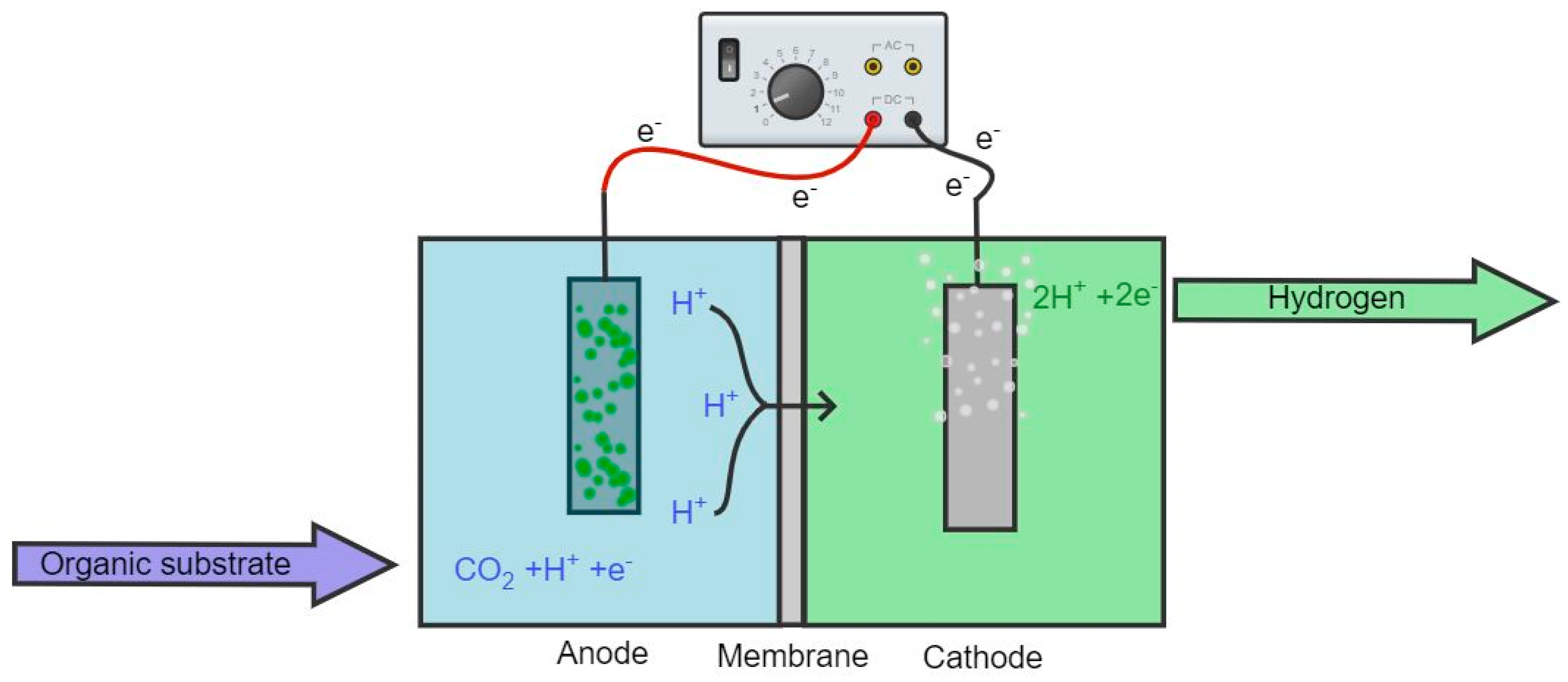
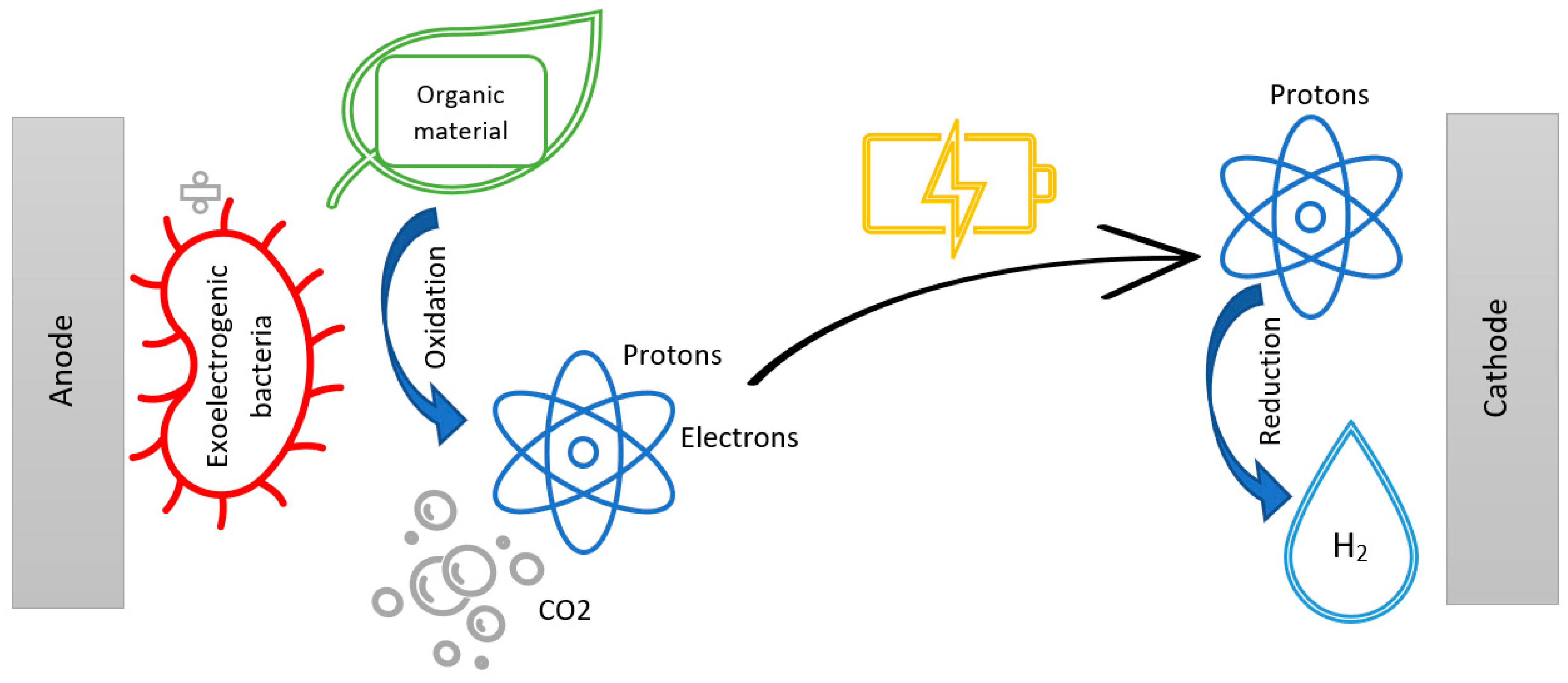
2.1. Operating Principle
2.2. Anode Material
2.3. Cathode Material
| Cathode Material | Catalyst Loading In mg · cm−2 | Advantages | Disadvantages | MEC/MFC Performance | Reference |
|---|---|---|---|---|---|
| Pt-Co/G (15 wt.% Pt) on carbon cloth | 2.5 | Less Pt needed compared with Pt/C coating | Pt is expensive and would need repleting | 1378 mW · m−2 | [153] |
| FePC-supported multiwalled carbon nanotubes on carbon cloth | 1 | Alternative to Pt, cheaper than Pt | Carbon nanotubes need further investigation to become a stable cathode | 601 mW · m−2 | [156] |
| MnOx on carbon paper | 0.1 | Alternative to Pt and cheaper than Pt, low catalyst loading needed | - | 772.8 mW · m−3 | [157] |
| Stainless steel 306 (12% Ni) | - | Ni incorporated into stainless steel enables the catalysis of HER | Investigated for water electrolysis | - | [158] |
| Ni/AC/PTFE-coated stainless steel mesh | 6.5 | Ni can substitute noble catalysts with high activity, high porosity | Ni ions can be poisonous | 1.88 L H2 · L−1 · day−1 | [151] |
| Ni2P/C coated on stainless steel mesh | 0.5 | Large chemical stability, HPR as high as Pt-based cathodes | 0.29 L H2 · L−1 · day−1 | [159] | |
| Pt-coated carbon cloth Pt-coated nickel foam | 0.5 0.5 | Reliable, efficient, long operation time Cheaper and higher HPR than similar Pt-coated carbon cloth, porous | Week base, expensive catalyst Not stable in the same duration as Pt-coated carbon cloth, relatively expensive | 0.67 L H2 · L−1 · day−1 0.71 L H2 · L−1 · day−1 | [152] |
| Nickel foam | - | High productivity, large surface area enables fast catalysis. | Problems connected to scaling, quick decrease in MEC performance | 50 L H2 · L−1 · day−1 | [150] |
| Biotic based on wastewater incorporated on stainless steel mesh | - | Cheap, environmentally friendly, long operation time | Not as effective as Pt or similar catalysts | 240 A · m−3 | [134] |
| Biotic based on urban wastewater and MFC inoculum incorporated on granular graphite | - | Cheap, environmentally friendly, long operation time, found to be as effective or more effective than carbon-based cathodes | Relatively low HPR | 0.9 L H2 · L−1 NCC · day−1 | [144] |
| Pd/GO-C incorporated on carbon paper | 0.25 | Cheap compared to Pt, efficient catalyst | Expensive compared to nickel or stainless steel | 901 mW · m−2 | [149] |
2.4. Membrane Configuration
2.5. Type of Configuration
3. Feedstocks for the Production of Biohydrogen
3.1. Wastewater
3.2. Solid Waste
4. Current Limitations and Improvement to MECs
Economic Viability and Life Cycle Assessment
5. Summary and Prospects
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- He, Y. How Population Growth Impacts Energy Consumption in Guangdong in China; Research Features: Stonehouse, UK, 2019. [Google Scholar]
- Hopfenberg, R.; Pimentel, D. Human Population Numbers as a Function of Food Supply. Environ. Dev. Sustain. 2001, 3, 1–15. [Google Scholar] [CrossRef]
- Shindell, D. Global Methane Assessment: Benefits and Costs of Mitigating Methane Emissions; Climate & Clean Air Coalition: Paris, France, 2021. [Google Scholar]
- Solomon, S.; Qin, D.; Manning, M.; Chen, Z.; Marquis, M.; Averyt, K.B.; Tignor, M.M.B.; Miller, H.L., Jr. Climate Change the Physical Science Basis; IPCC: Geneva, Switzerland, 2007. [Google Scholar]
- Malinauskaite, J.; Jouhara, H.; Czajczyńska, D.; Stanchev, P.; Katsou, E.; Rostkowski, P.; Thorne, R.J.; Colón, J.; Ponsá, S.; Al-Mansour, F.; et al. Municipal Solid Waste Management and Waste-to-Energy in the Context of a Circular Economy and Energy Recycling in Europe. Energy 2017, 141, 2013–2044. [Google Scholar] [CrossRef]
- Acar, C.; Dincer, I. 4.24 Hydrogen Energy Conversion Systems. In Comprehensive Energy Systems; Elsevier: Oxford, UK, 2018; Volume 4, pp. 947–984. ISBN 978-0-12-814925-6. [Google Scholar]
- International Energy Agency. The Future of Hydrogen; International Energy Agency: Paris, France, 2019. [Google Scholar]
- Dincer, I.; Acar, C. Review and Evaluation of Hydrogen Production Methods for Better Sustainability. Int. J. Hydrogen Energy 2015, 40, 11094–11111. [Google Scholar] [CrossRef]
- Pipitone, G.; Zoppi, G.; Pirone, R.; Bensaid, S. A Critical Review on Catalyst Design for Aqueous Phase Reforming. Int. J. Hydrogen Energy 2022, 47, 151–180. [Google Scholar] [CrossRef]
- Tak, S.S.; Shetye, O.; Muley, O.; Jaiswal, H.; Malik, S.N. Emerging Technologies for Hydrogen Production from Wastewater. Int. J. Hydrogen Energy, 2022; in press. [Google Scholar] [CrossRef]
- Brown, F.; Roberts, D. Green, Blue, Brown: The Colours of Hydrogen Explained. Available online: https://blog.csiro.au/green-blue-brown-hydrogen-explained/ (accessed on 5 October 2021).
- Nikolaidis, P.; Poullikkas, A. A Comparative Overview of Hydrogen Production Processes. Renew. Sustain. Energy Rev. 2017, 67, 597–611. [Google Scholar] [CrossRef]
- Daiyan, R.; MacGill, I.; Amal, R. Opportunities and Challenges for Renewable Power-to-X. ACS Energy Lett. 2020, 5, 3843–3847. [Google Scholar] [CrossRef]
- International Energy Agency. Energy Technology Perspectivies 2020; International Energy Agency: Paris, France, 2020. [Google Scholar]
- Lindstrand, N. Unlocking Ammonia’s Potential for Shipping. Available online: https://www.man-es.com/discover/two-stroke-ammonia-engine (accessed on 5 October 2021).
- International Energy Agency. Global Hydrogen Review 2021; International Energy Agency: Paris, France, 2021. [Google Scholar]
- European Union 2030 Climate Target Plan. Available online: https://ec.europa.eu/clima/eu-action/european-green-deal/2030-climate-target-plan_en (accessed on 7 October 2021).
- de Vasconcelos, B.; Lavoie, J.-M. Recent Advances in Power-to-X Technology for the Production of Fuels and Chemicals. Front. Chem. 2019, 7. [Google Scholar] [CrossRef]
- Thomas, G.; Parks, G. Potential Roles of Ammonia in a Hydrogen Economy; Office of Energy Efficiency & Renewable Energy: Washington, DC, USA, 2006. [Google Scholar]
- Ghavam, S.; Vahdati, M.; Wilson, I.A.G.; Styring, P. Sustainable Ammonia Production Processes. Front. Energy Res. 2021, 9, 34. [Google Scholar] [CrossRef]
- Smith, C.; Hill, A.K.; Torrente-Murciano, L. Current and Future Role of Haber–Bosch Ammonia in a Carbon-Free Energy Landscape. Energy Environ. Sci. 2020, 13, 331–344. [Google Scholar] [CrossRef]
- Amar, I.A.; Lan, R.; Petit, C.T.G.; Tao, S. Solid-State Electrochemical Synthesis of Ammonia: A Review. J. Solid State Electrochem. 2011, 15, 1845. [Google Scholar] [CrossRef]
- Tullo, A.H. Is Ammonia the Fuel of the Future? Chem. Eng. News 2021, 99, 20–22. [Google Scholar] [CrossRef]
- Minic, D.C.E.-D. Ammonia as a Hydrogen Source for Fuel Cells: A Review; IntechOpen: Rijeka, Croatia, 2012; p. 13. [Google Scholar]
- Lan, R.; Irvine, J.T.S.; Tao, S. Ammonia and Related Chemicals as Potential Indirect Hydrogen Storage Materials. Int. J. Hydrogen Energy 2012, 37, 1482–1494. [Google Scholar] [CrossRef]
- Eberle, U.; Felderhoff, M.; Schüth, F. Chemical and Physical Solutions for Hydrogen Storage. Angew. Chemie Int. Ed. 2009, 48, 6608–6630. [Google Scholar] [CrossRef]
- Grochala, W.; Edwards, P.P. Thermal Decomposition of the Non-Interstitial Hydrides for the Storage and Production of Hydrogen. Chem. Rev. 2004, 104, 1283–1316. [Google Scholar] [CrossRef]
- Satyapal, S.; Petrovic, J.; Read, C.; Thomas, G.; Ordaz, G. The U.S. Department of Energy’s National Hydrogen Storage Project: Progress towards Meeting Hydrogen-Powered Vehicle Requirements. Catal. Today 2007, 120, 246–256. [Google Scholar] [CrossRef]
- Sartbaeva, A.; Kuznetsov, V.L.; Wells, S.A.; Edwards, P.P. Hydrogen Nexus in a Sustainable Energy Future. Energy Environ. Sci. 2008, 1, 79–85. [Google Scholar] [CrossRef]
- Jain, I.P.; Jain, P.; Jain, A. Novel Hydrogen Storage Materials: A Review of Lightweight Complex Hydrides. J. Alloys Compd. 2010, 503, 303–339. [Google Scholar] [CrossRef]
- Hu, Y.H.; Zhang, L. Hydrogen Storage in Metal–Organic Frameworks. Adv. Mater. 2010, 22, E117–E130. [Google Scholar] [CrossRef]
- Graetz, J. New Approaches to Hydrogen Storage. Chem. Soc. Rev. 2009, 38, 73–82. [Google Scholar] [CrossRef]
- Makepeace, J.W.; He, T.; Weidenthaler, C.; Jensen, T.R.; Chang, F.; Vegge, T.; Ngene, P.; Kojima, Y.; de Jongh, P.E.; Chen, P.; et al. Reversible Ammonia-Based and Liquid Organic Hydrogen Carriers for High-Density Hydrogen Storage: Recent Progress. Int. J. Hydrogen Energy 2019, 44, 7746–7767. [Google Scholar] [CrossRef]
- Zamfirescu, C.; Dincer, I. Using Ammonia as a Sustainable Fuel. J. Power Sources 2008, 185, 459–465. [Google Scholar] [CrossRef]
- Christensen, C.H.; Johannessen, T.; Sørensen, R.Z.; Nørskov, J.K. Towards an Ammonia-Mediated Hydrogen Economy? Catal. Today 2006, 111, 140–144. [Google Scholar] [CrossRef]
- Zamfirescu, C.; Dincer, I. Ammonia as a Green Fuel and Hydrogen Source for Vehicular Applications. Fuel Process. Technol. 2009, 90, 729–737. [Google Scholar] [CrossRef]
- Cotterill, S.; Heidrich, E.; Curtis, T. Microbial electrolysis cells for hydrogen production. Microb. Electrochem. Fuel Cells 2016, 287–319. [Google Scholar] [CrossRef]
- Rahimnejad, M.; Adhami, A.; Darvari, S.; Zirepour, A.; Oh, S.-E. Microbial Fuel Cell as New Technology for Bioelectricity Generation: A Review. Alexandria Eng. J. 2015, 54, 745–756. [Google Scholar] [CrossRef]
- Kadier, A.; Simayi, Y.; Abdeshahian, P.; Azman, N.F.; Chandrasekhar, K.; Kalil, M.S. A Comprehensive Review of Microbial Electrolysis Cells (MEC) Reactor Designs and Configurations for Sustainable Hydrogen Gas Production. Alexandria Eng. J. 2016, 55, 427–443. [Google Scholar] [CrossRef]
- Kadier, A.; Yuliasni, R.; Sapuan, S.; Ilyas, R.A.; Rai, P.K.; Ma, P.-C.; Rajasekar, A.; Alabbosh, K.; Hamid, A.; Abu Hasan, H. The Role of Microbial Electrolysis Cell in Bioenergy Production: Current Applications and Pilot Plant Experiences. In Bioelectrochemical Systems; Springer: Singapore, 2021; pp. 323–342. [Google Scholar] [CrossRef]
- Adekunle, A.; Raghavan, V.; Tartakovsky, B. A Comparison of Microbial Fuel Cell and Microbial Electrolysis Cell Biosensors for Real-Time Environmental Monitoring. Bioelectrochemistry 2019, 126, 105–112. [Google Scholar] [CrossRef]
- Kadier, A.; Chaurasia, A.K.; Sapuan, S.M.; Ilyas, R.A.; Ma, P.C.; Alabbosh, K.F.S.; Rai, P.K.; Logroño, W.; Hamid, A.A.; Abu Hasan, H. Essential Factors for Performance Improvement and the Implementation of Microbial Electrolysis Cells (MECs). In Bioelectrochemical Systems; Springer: Singapore, 2021; pp. 139–168. ISBN 978-981-15-6871-8. [Google Scholar]
- Lim, S.S.; Fontmorin, J.-M.; Izadi, P.; Wan Daud, W.R.; Scott, K.; Yu, E.H. Impact of Applied Cell Voltage on the Performance of a Microbial Electrolysis Cell Fully Catalysed by Microorganisms. Int. J. Hydrogen Energy 2020, 45, 2557–2568. [Google Scholar] [CrossRef]
- Katuri, K.P.; Ali, M.; Saikaly, P.E. The Role of Microbial Electrolysis Cell in Urban Wastewater Treatment: Integration Options, Challenges, and Prospects. Curr. Opin. Biotechnol. 2019, 57, 101–110. [Google Scholar] [CrossRef]
- Sacco, N.J.; Bonetto, M.C.; Cortón, E. Isolation and Characterization of a Novel Electrogenic Bacterium, Dietzia Sp. RNV-4. PLoS ONE 2017, 12, e0169955. [Google Scholar] [CrossRef]
- Chabert, N.; Amin Ali, O.; Achouak, W. All Ecosystems Potentially Host Electrogenic Bacteria. Bioelectrochemistry 2015, 106, 88–96. [Google Scholar] [CrossRef]
- Call, D.F.; Wagner, R.C.; Logan, B.E. Hydrogen Production by Geobacter Species and a Mixed Consortium in a Microbial Electrolysis Cell. Appl. Environ. Microbiol. 2009, 75, 7579–7587. [Google Scholar] [CrossRef]
- Cusick, R.D.; Bryan, B.; Parker, D.S.; Merrill, M.D.; Mehanna, M.; Kiely, P.D.; Liu, G.; Logan, B.E. Performance of a Pilot-Scale Continuous Flow Microbial Electrolysis Cell Fed Winery Wastewater. Appl. Microbiol. Biotechnol. 2011, 89, 2053–2063. [Google Scholar] [CrossRef]
- Almatouq, A.; Babatunde, A.O.; Khajah, M.; Webster, G.; Alfodari, M. Microbial Community Structure of Anode Electrodes in Microbial Fuel Cells and Microbial Electrolysis Cells. J. Water Process Eng. 2020, 34, 101140. [Google Scholar] [CrossRef]
- UniProt Proteomes-Geobacter Sp. (Strain M18). Available online: https://www.uniprot.org/proteomes/UP000001442 (accessed on 8 March 2021).
- Guo, Y.; Wang, J.; Shinde, S.; Wang, X.; Li, Y.; Dai, Y.; Ren, J.; Zhang, P.; Liu, X. Simultaneous Wastewater Treatment and Energy Harvesting in Microbial Fuel Cells: An Update on the Biocatalysts. RSC Adv. 2020, 10, 25874–25887. [Google Scholar] [CrossRef]
- Babauta, J.; Renslow, R.; Lewandowski, Z.; Beyenal, H. Electrochemically Active Biofilms: Facts and Fiction. A Review. Biofouling 2012, 28, 789–812. [Google Scholar] [CrossRef]
- Hari, A.R.; Venkidusamy, K.; Katuri, K.P.; Bagchi, S.; Saikaly, P.E. Temporal Microbial Community Dynamics in Microbial Electrolysis Cells–Influence of Acetate and Propionate Concentration. Front. Microbiol. 2017, 8, 1371. [Google Scholar] [CrossRef]
- Proteomes-Shewanella Sp. (Strain ANA-3). Available online: https://www.uniprot.org/proteomes/UP000002589 (accessed on 8 March 2021).
- Hodgson, D.M.; Smith, A.; Dahale, S.; Stratford, J.P.; Li, J.V.; Grüning, A.; Bushell, M.E.; Marchesi, J.R.; Avignone Rossa, C. Segregation of the Anodic Microbial Communities in a Microbial Fuel Cell Cascade. Front. Microbiol. 2016, 7, 699. [Google Scholar] [CrossRef]
- Tahernia, M.; Plotkin-Kaye, E.; Mohammadifar, M.; Gao, Y.; Oefelein, M.R.; Cook, L.C.; Choi, S. Characterization of Electrogenic Gut Bacteria. ACS Omega 2020, 5, 29439–29446. [Google Scholar] [CrossRef]
- Croese, E.; Jeremiasse, A.W.; Marshall, I.P.G.; Spormann, A.M.; Euverink, G.-J.W.; Geelhoed, J.S.; Stams, A.J.M.; Plugge, C.M. Influence of Setup and Carbon Source on the Bacterial Community of Biocathodes in Microbial Electrolysis Cells. Enzym. Microb. Technol. 2014, 61–62, 67–75. [Google Scholar] [CrossRef]
- Walker, C.B.; He, Z.; Yang, Z.K.; Ringbauer, J.A.; He, Q.; Zhou, J.; Voordouw, G.; Wall, J.D.; Arkin, A.P.; Hazen, T.C.; et al. The Electron Transfer System of Syntrophically Grown Desulfovibrio vulgaris. J. Bacteriol. 2009, 191, 5793–5801. [Google Scholar] [CrossRef]
- Figueiredo, G.G.O.; Lopes, V.R.; Romano, T.; Camara, M.C. Clostridium. In Beneficial Microbes in Agro-Ecology; Academic Press: Cambridge, MA, USA, 2020; pp. 477–491. ISBN 978-0-12-823414-3. [Google Scholar]
- Juang, D.F.; Yang, P.C.; Lee, C.H.; Hsueh, S.C.; Kuo, T.H. Electrogenic Capabilities of Gram Negative and Gram Positive Bacteria in Microbial Fuel Cell Combined with Biological Wastewater Treatment. Int. J. Environ. Sci. Technol. 2011, 8, 781–792. [Google Scholar] [CrossRef]
- Milner, D.A.; Pecora, N.; Solomon, I.; Soong, T.R. (Eds.) Bacillus Species Infections. In Diagnostic Pathology: Infectious Diseases; Elsevier: Philadelphia, PA, USA, 2015; pp. II-2-2–II-2-5. ISBN 978-0-323-37677-8. [Google Scholar]
- Kamaraj, Y. Electrogenicity Assessment of Bacillus Subtilis and Bacillus Megaterium Using Microbial Fuel Cell Technology. Int. J. Appl. Res. 2015, 1, 435–438. [Google Scholar]
- Logan, B.E.; Call, D.; Cheng, S.; Hamelers, H.V.M.; Sleutels, T.H.J.A.; Jeremiasse, A.W.; Rozendal, R.A. Microbial Electrolysis Cells for High Yield Hydrogen Gas Production from Organic Matter. Environ. Sci. Technol. 2008, 42, 8630–8640. [Google Scholar] [CrossRef]
- Pham, T.H.; Boon, N.; Aelterman, P.; Clauwaert, P.; De Schamphelaire, L.; Vanhaecke, L.; De Maeyer, K.; Höfte, M.; Verstraete, W.; Rabaey, K. Metabolites Produced by Pseudomonas Sp. Enable a Gram-Positive Bacterium to Achieve Extracellular Electron Transfer. Appl. Microbiol. Biotechnol. 2008, 77, 1119–1129. [Google Scholar] [CrossRef]
- Gatidou, G.; Samanides, C.G.; Fountoulakis, M.S.; Vyrides, I. Microbial Electrolysis Cell Coupled with Anaerobic Granular Sludge: A Novel Technology for Real Bilge Water Treatment. Chemosphere 2022, 296, 133988. [Google Scholar] [CrossRef]
- Zecchin, S.; Mueller, R.C.; Seifert, J.; Stingl, U.; Anantharaman, K.; von Bergen, M.; Cavalca, L.; Pester, M. Rice Paddy Nitrospirae Carry and Express Genes Related to Sulfate Respiration: Proposal of the New Genus “Candidatus Sulfobium”. Appl. Environ. Microbiol. 2018, 84, e02224-17. [Google Scholar] [CrossRef]
- Gobalakrishnan, R.; Bhuvaneswari, R. Microbial fuel cells potential of marine actinobacteria Actinoalloteichus sp. MHA15 from the Havelock island of the Andamans, India. Biotechnol. Res. Innov. 2019, 3, 144–158. [Google Scholar] [CrossRef]
- Hasany, M.; Mardanpour, M.M.; Yaghmaei, S. Biocatalysts in Microbial Electrolysis Cells: A Review. Int. J. Hydrogen Energy 2016, 41, 1477–1493. [Google Scholar] [CrossRef]
- Ye, J.-Y.; Pan, Y.; Wang, Y.; Wang, Y.-C. Enhanced Hydrogen Production of Rhodobacter Sphaeroides Promoted by Extracellular H+ of Halobacterium Salinarum. Ann. Microbiol. 2021, 71, 15. [Google Scholar] [CrossRef]
- Salar-Garcia, M.J.; Obata, O.; Kurt, H.; Chandran, K.; Greenman, J.; Ieropoulos, I.A. Impact of Inoculum Type on the Microbial Community and Power Performance of Urine-Fed Microbial Fuel Cells. Microorganisms 2020, 8, 1921. [Google Scholar] [CrossRef]
- Cadirci, B.H. An electricity production study by Rhodobacter sphaeroides. Int. J. Hydrogen Energy 2018, 43, 18001–18006. [Google Scholar] [CrossRef]
- Cho, Y.K.; Donohue, T.J.; Tejedor, I.; Anderson, M.A.; McMahon, K.D.; Noguera, D.R. Development of a Solar-Powered Microbial Fuel Cell. J. Appl. Microbiol. 2008, 104, 640–650. [Google Scholar] [CrossRef]
- Holmes, D.E.; Nevin, K.P.; Lovley, D.R. Comparison of 16S RRNA, NifD, RecA, GyrB, RpoB and FusA Genes within the Family Geobacteraceae Fam. Nov. Int. J. Syst. Evol. Microbiol. 2004, 54, 1591–1599. [Google Scholar] [CrossRef]
- Lu, L.; Xing, D.; Ren, N. Pyrosequencing Reveals Highly Diverse Microbial Communities in Microbial Electrolysis Cells Involved in Enhanced H2 Production from Waste Activated Sludge. Water Res. 2012, 46, 2425–2434. [Google Scholar] [CrossRef] [PubMed]
- Uria, N.; Ferrera, I.; Mas, J. Electrochemical Performance and Microbial Community Profiles in Microbial Fuel Cells in Relation to Electron Transfer Mechanisms. BMC Microbiol. 2017, 17, 208. [Google Scholar] [CrossRef]
- Cao, Y.; Mu, H.; Liu, W.; Zhang, R.; Guo, J.; Xian, M.; Liu, H. Electricigens in the Anode of Microbial Fuel Cells: Pure Cultures versus Mixed Communities. Microb. Cell Fact. 2019, 18, 39. [Google Scholar] [CrossRef]
- Xie, X.; Yu, G.; Liu, N.; Bao, Z.; Criddle, C.S.; Cui, Y. Graphene–Sponges as High-Performance Low-Cost Anodes for Microbial Fuel Cells. Energy Environ. Sci. 2012, 5, 6862–6866. [Google Scholar] [CrossRef]
- Khilyas, I.V.; Sorokin, A.A.; Kiseleva, L.; Simpson, D.J.W.; Fedorovich, V.; Sharipova, M.R.; Kainuma, M.; Cohen, M.F.; Goryanin, I. Comparative Metagenomic Analysis of Electrogenic Microbial Communities in Differentially Inoculated Swine Wastewater-Fed Microbial Fuel Cells. Scientifica (Cairo) 2017, 2017, 7616359. [Google Scholar] [CrossRef]
- Yasri, N.; Roberts, E.P.L.; Gunasekaran, S. The Electrochemical Perspective of Bioelectrocatalytic Activities in Microbial Electrolysis and Microbial Fuel Cells. Energy Rep. 2019, 5, 1116–1136. [Google Scholar] [CrossRef]
- Aiyer, K.S. How Does Electron Transfer Occur in Microbial Fuel Cells? World J. Microbiol. Biotechnol. 2020, 36, 19. [Google Scholar] [CrossRef]
- Kadier, A.; Al-Shorgani, N.K.N.; Jadhav, D.A.; Sonawane, J.M.; Mathuriya, A.S.; Kalil, M.S.; Hasan, H.A.; Alabbosh, K.F.S. Microbial Electrolysis Cell (MEC): An Innovative Waste to Bioenergy and Value-Added By-product Technology. In Bioelectrosynthesis: Principles and Technologies for Value-Added Products; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2020; pp. 95–128. [Google Scholar] [CrossRef]
- Bajracharya, S.; ElMekawy, A.; Srikanth, S.; Pant, D. Cathodes for Microbial Fuel Cells. In Microbial Electrochemical and Fuel Cells; Woodhead Publishing: Boston, MA, USA, 2016; pp. 179–213. ISBN 978-1-78242-375-1. [Google Scholar]
- Jeremiasse, A.W. Cathode Innovations for Enhanced H2 Production through Microbial Electrolysis; Wageningen University: Wageningen, The Netherlands, 2011. [Google Scholar]
- Velasquez-Orta, S.B.; Yu, E.; Katuri, K.P.; Head, I.M.; Curtis, T.P.; Scott, K. Evaluation of Hydrolysis and Fermentation Rates in Microbial Fuel Cells. Appl. Microbiol. Biotechnol. 2011, 90, 789–798. [Google Scholar] [CrossRef] [PubMed]
- Montpart, N.; Baeza, M.; Baeza, J.A.; Guisasola, A. Low-Cost Fuel-Cell Based Sensor of Hydrogen Production in Lab Scale Microbial Electrolysis Cells. Int. J. Hydrogen Energy 2016, 41, 20465–20472. [Google Scholar] [CrossRef]
- Rozenfeld, S.; Hirsch, L.O.; Gandu, B.; Farber, R.; Schechter, A.; Cahan, R. Improvement of Microbial Electrolysis Cell Activity by Using Anode Based on Combined Plasma-Pretreated Carbon Cloth and Stainless Steel. Energies 2019, 12, 1968. [Google Scholar] [CrossRef]
- Borole, A.P.; Lewis, A.J. Proton Transfer in Microbial Electrolysis Cells. Sustain. Energy Fuels 2017, 1, 725–736. [Google Scholar] [CrossRef]
- Li, C.; Cheng, S. Functional Group Surface Modifications for Enhancing the Formation and Performance of Exoelectrogenic Biofilms on the Anode of a Bioelectrochemical System. Crit. Rev. Biotechnol. 2019, 39, 1015–1030. [Google Scholar] [CrossRef]
- Böl, M.; Ehret, A.E.; Bolea Albero, A.; Hellriegel, J.; Krull, R. Recent Advances in Mechanical Characterisation of Biofilm and Their Significance for Material Modelling. Crit. Rev. Biotechnol. 2013, 33, 145–171. [Google Scholar] [CrossRef]
- Santoro, C.; Arbizzani, C.; Erable, B.; Ieropoulos, I. Microbial Fuel Cells: From Fundamentals to Applications. A Review. J. Power Sources 2017, 356, 225–244. [Google Scholar] [CrossRef]
- Rinaldi, A.; Mecheri, B.; Garavaglia, V.; Licoccia, S.; Di Nardo, P.; Traversa, E. Engineering Materials and Biology to Boost Performance of Microbial Fuel Cells: A Critical Review. Energy Environ. Sci. 2008, 1, 417–429. [Google Scholar] [CrossRef]
- Zhen, G.; Lu, X.; Kumar, G.; Bakonyi, P.; Xu, K.; Zhao, Y. Microbial Electrolysis Cell Platform for Simultaneous Waste Biorefinery and Clean Electrofuels Generation: Current Situation, Challenges and Future Perspectives. Prog. Energy Combust. Sci. 2017, 63, 119–145. [Google Scholar] [CrossRef]
- Feng, Y.; Yang, Q.; Wang, X.; Liu, Y.; Lee, H.; Ren, N. Treatment of Biodiesel Production Wastes with Simultaneous Electricity Generation Using a Single-Chamber Microbial Fuel Cell. Bioresour. Technol. 2011, 102, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Gu, W.; Zhou, Q.; Zhang, L.; Wei, C.; Yang, Q.; Li, D. Ammonia Oxidation and Denitrification in a Bio-Anode Single-Chambered Microbial Electrolysis Cell. Bioresour. Technol. 2020, 310, 123466. [Google Scholar] [CrossRef] [PubMed]
- Tartakovsky, B.; Manuel, M.-F.; Wang, H.; Guiot, S.R. High Rate Membrane-Less Microbial Electrolysis Cell for Continuous Hydrogen Production. Int. J. Hydrogen Energy 2009, 34, 672–677. [Google Scholar] [CrossRef]
- Spiess, S.; Kucera, J.; Seelajaroen, H.; Sasiain, A.; Thallner, S.; Kremser, K.; Novak, D.; Guebitz, G.; Haberbauer, M. Impact of Carbon Felt Electrode Pretreatment on Anodic Biofilm Composition in Microbial Electrolysis Cells. Biosensors 2021, 11, 170. [Google Scholar] [CrossRef]
- Gandu, B.; Rozenfeld, S.; Ouaknin Hirsch, L.; Schechter, A.; Cahan, R. Immobilization of Bacterial Cells on Carbon-Cloth Anode Using Alginate for Hydrogen Generation in a Microbial Electrolysis Cell. J. Power Sources 2020, 455, 227986. [Google Scholar] [CrossRef]
- Chang, S.-H.; Huang, B.-Y.; Wan, T.-H.; Chen, J.-Z.; Chen, B.-Y. Surface Modification of Carbon Cloth Anodes for Microbial Fuel Cells Using Atmospheric-Pressure Plasma Jet Processed Reduced Graphene Oxides. RSC Adv. 2017, 7, 56433–56439. [Google Scholar] [CrossRef]
- Roubaud, E.; Lacroix, R.; Da Silva, S.; Etcheverry, L.; Bergel, A.; Basséguy, R.; Erable, B. Benchmarking of Industrial Synthetic Graphite Grades, Carbon Felt, and Carbon Cloth as Cost-Efficient Bioanode Materials for Domestic Wastewater Fed Microbial Electrolysis Cells. Front. Energy Res. 2019, 7, 106. [Google Scholar] [CrossRef]
- Clauwaert, P.; Verstraete, W. Methanogenesis in Membraneless Microbial Electrolysis Cells. Appl. Microbiol. Biotechnol. 2009, 82, 829–836. [Google Scholar] [CrossRef]
- Croese, E.; Keesman, K.J.; Widjaja-Greefkes, H.C.A.; Geelhoed, J.S.; Plugge, C.M.; Sleutels, T.H.J.A.; Stams, A.J.M.; Euverink, G.-J.W. Relating MEC Population Dynamics to Anode Performance from DGGE and Electrical Data. Syst. Appl. Microbiol. 2013, 36, 408–416. [Google Scholar] [CrossRef]
- Masoudi, M.; Rahimnejad, M.; Mashkour, M. Fabrication of Anode Electrode by a Novel Acrylic Based Graphite Paint on Stainless Steel Mesh and Investigating Biofilm Effect on Electrochemical Behavior of Anode in a Single Chamber Microbial Fuel Cell. Electrochim. Acta 2020, 344, 136168. [Google Scholar] [CrossRef]
- Todoroki, N.; Wadayama, T. Electrochemical Stability of Stainless-Steel-Made Anode for Alkaline Water Electrolysis: Surface Catalyst Nanostructures and Oxygen Evolution Overpotentials under Applying Potential Cycle Loading. Electrochem. Commun. 2021, 122, 106902. [Google Scholar] [CrossRef]
- Sonawane, J.M.; Patil, S.A.; Ghosh, P.C.; Adeloju, S.B. Low-Cost Stainless-Steel Wool Anodes Modified with Polyaniline and Polypyrrole for High-Performance Microbial Fuel Cells. J. Power Sources 2018, 379, 103–114. [Google Scholar] [CrossRef]
- Tahir, K.; Miran, W.; Jang, J.; Maile, N.; Shahzad, A.; Moztahida, M.; Ghani, A.A.; Kim, B.; Jeon, H.; Lim, S.-R.; et al. Nickel Ferrite/MXene-Coated Carbon Felt Anodes for Enhanced Microbial Fuel Cell Performance. Chemosphere 2021, 268, 128784. [Google Scholar] [CrossRef]
- Zakaria, B.S.; Barua, S.; Sharaf, A.; Liu, Y.; Dhar, B.R. Impact of Antimicrobial Silver Nanoparticles on Anode Respiring Bacteria in a Microbial Electrolysis Cell. Chemosphere 2018, 213, 259–267. [Google Scholar] [CrossRef]
- Banerjee, A.; Calay, R.K.; Mustafa, M. Review on Material and Design of Anode for Microbial Fuel Cell. Energies 2022, 15, 2283. [Google Scholar] [CrossRef]
- Zhu, X.; Logan, B.E. Copper anode corrosion affects power generation in microbial fuel cells. J. Chem. Technol. Biotechnol. 2013, 89, 471–474. [Google Scholar] [CrossRef]
- Zhao, Y.; Ma, Y.; Li, T.; Dong, Z.; Wang, Y. Modification of Carbon Felt Anodes Using Double-Oxidant HNO3/H2O2 for Application in Microbial Fuel Cells. RSC Adv. 2018, 8, 2059–2064. [Google Scholar] [CrossRef]
- Hidalgo, D.; Tommasi, T.; Bocchini, S.; Chiolerio, A.; Chiodoni, A.; Mazzarino, I.; Ruggeri, B. Surface Modification of Commercial Carbon Felt Used as Anode for Microbial Fuel Cells. Energy 2016, 99, 193–201. [Google Scholar] [CrossRef]
- Baek, G.; Saikaly, P.E.; Logan, B.E. Addition of a Carbon Fiber Brush Improves Anaerobic Digestion Compared to External Voltage Application. Water Res. 2021, 188, 116575. [Google Scholar] [CrossRef]
- Wang, X.; Cheng, S.; Feng, Y.; Merrill, M.D.; Saito, T.; Logan, B.E. Use of Carbon Mesh Anodes and the Effect of Different Pretreatment Methods on Power Production in Microbial Fuel Cells. Environ. Sci. Technol. 2009, 43, 6870–6874. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.-F.; Zhang, F.; Huang, B.-C.; Yu, H.-Q. Enhancing Electricity Generation of Microbial Fuel Cell for Wastewater Treatment Using Nitrogen-Doped Carbon Dots-Supported Carbon Paper Anode. J. Clean. Prod. 2019, 229, 412–419. [Google Scholar] [CrossRef]
- Santoro, C.; Guilizzoni, M.; Correa Baena, J.P.; Pasaogullari, U.; Casalegno, A.; Li, B.; Babanova, S.; Artyushkova, K.; Atanassov, P. The Effects of Carbon Electrode Surface Properties on Bacteria Attachment and Start up Time of Microbial Fuel Cells. Carbon N. Y. 2014, 67, 128–139. [Google Scholar] [CrossRef]
- Escapa, A.; Mateos, R.; Martínez, E.J.; Blanes, J. Microbial Electrolysis Cells: An Emerging Technology for Wastewater Treatment and Energy Recovery. From Laboratory to Pilot Plant and Beyond. Renew. Sustain. Energy Rev. 2016, 55, 942–956. [Google Scholar] [CrossRef]
- Saravanan, A.; Karishma, S.; Kumar, P.S.; Yaashikaa, P.R.; Jeevanantham, S.; Gayathri, B. Microbial Electrolysis Cells and Microbial Fuel Cells for Biohydrogen Production: Current Advances and Emerging Challenges. Biomass Convers. Biorefinery 2020. [Google Scholar] [CrossRef]
- Michaelidou, U.; ter Heijne, A.; Euverink, G.J.W.; Hamelers, H.V.M.; Stams, A.J.M.; Geelhoed, J.S. Microbial Communities and Electrochemical Performance of Titanium-Based Anodic Electrodes in a Microbial Fuel Cell. Appl. Environ. Microbiol. 2011, 77, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.; Kim, J.H.; Koo, K.; Kim, S. A Photo-Assisted Microbial Electrolysis Cell for the Exclusive Biohydrogen Production Using a MoS2-Coated p-Type Copper Oxide. J. Power Sources 2018, 373, 79–84. [Google Scholar] [CrossRef]
- Purushotham, K.G.; Sendilvelan, S. Analysis of Microbial Fuel Cell for Energy Harvesting with Waste Water and Molasses. Int. J. Pharma Bio Sci. 2017, 8, 63–69. [Google Scholar]
- Fuel Cell Store. Available online: https://www.fuelcellstore.com/isomolded-plate-024 (accessed on 7 October 2021).
- Delord, B.; Neri, W.; Bertaux, K.; Derre, A.; Ly, I.; Mano, N.; Poulin, P. Carbon Nanotube Fiber Mats for Microbial Fuel Cell Electrodes. Bioresour. Technol. 2017, 243, 1227–1231. [Google Scholar] [CrossRef]
- Amazon Microbial Fuel Cell Carbon Brush. Available online: https://www.amazon.com/Microbial-carbon-brush-conductive-5cm10cm15cm(LHW)/dp/B08154Z4C4 (accessed on 7 October 2021).
- Wei, J.; Liang, P.; Huang, X. Recent Progress in Electrodes for Microbial Fuel Cells. Bioresour. Technol. 2011, 102, 9335–9344. [Google Scholar] [CrossRef]
- Wu, X.; Qiao, Y.; Guo, C.; Shi, Z.; Li, C.M. Nitrogen Doping to Atomically Match Reaction Sites in Microbial Fuel Cells. Commun. Chem. 2020, 3, 68. [Google Scholar] [CrossRef]
- Logan, B.; Cheng, S.; Watson, V.; Estadt, G. Graphite Fiber Brush Anodes for Increased Power Production in Air-Cathode Microbial Fuel Cells. Environ. Sci. Technol. 2007, 41, 3341–3346. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.; Logan, B.E. High Hydrogen Production Rate of Microbial Electrolysis Cell (MEC) with Reduced Electrode Spacing. Bioresour. Technol. 2011, 102, 3571–3574. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.G.; Sarathi, V.G.S.; Nahm, K.S. Recent Advances and Challenges in the Anode Architecture and Their Modifications for the Applications of Microbial Fuel Cells. Biosens. Bioelectron. 2013, 43, 461–475. [Google Scholar] [CrossRef] [PubMed]
- Blanchet, E.; Erable, B.; De Solan, M.-L.; Bergel, A. Two-Dimensional Carbon Cloth and Three-Dimensional Carbon Felt Perform Similarly to Form Bioanode Fed with Food Waste. Electrochem. Commun. 2016, 66, 38–41. [Google Scholar] [CrossRef]
- Pocaznoi, D.; Calmet, A.; Etcheverry, L.; Erable, B.; Bergel, A. Stainless Steel Is a Promising Electrode Material for Anodes of Microbial Fuel Cells. Energy Environ. Sci. 2012, 5, 9645–9652. [Google Scholar] [CrossRef]
- Cheng, S.; Logan, B.E. Ammonia Treatment of Carbon Cloth Anodes to Enhance Power Generation of Microbial Fuel Cells. Electrochem. Commun. 2007, 9, 492–496. [Google Scholar] [CrossRef]
- Hu, H.; Fan, Y.; Liu, H. Hydrogen Production Using Single-Chamber Membrane-Free Microbial Electrolysis Cells. Water Res. 2008, 42, 4172–4178. [Google Scholar] [CrossRef]
- Jeremiasse, A.W.; Hamelers, H.V.M.; Buisman, C.J.N. Microbial Electrolysis Cell with a Microbial Biocathode. Bioelectrochemistry 2010, 78, 39–43. [Google Scholar] [CrossRef]
- Jafary, T.; Daud, W.R.W.; Ghasemi, M.; Kim, B.H.; Carmona-Martínez, A.A.; Bakar, M.H.A.; Jahim, J.M.; Ismail, M. A Comprehensive Study on Development of a Biocathode for Cleaner Production of Hydrogen in a Microbial Electrolysis Cell. J. Clean. Prod. 2017, 164, 1135–1144. [Google Scholar] [CrossRef]
- Call, D.; Logan, B. A Method for High Throughput Bioelectrochemical Research Based on Small Scale Microbial Electrolysis Cells. Biosens. Bioelectron. 2011, 26, 4526–4531. [Google Scholar] [CrossRef] [PubMed]
- Hassanein, A.; Witarsa, F.; Guo, X.; Yong, L.; Lansing, S.; Qiu, L. Next Generation Digestion: Complementing Anaerobic Digestion (AD) with a Novel Microbial Electrolysis Cell (MEC) Design. Int. J. Hydrogen Energy 2017, 42, 28681–28689. [Google Scholar] [CrossRef]
- Call, D.; Logan, B.E. Hydrogen Production in a Single Chamber Microbial Electrolysis Cell Lacking a Membrane. Environ. Sci. Technol. 2008, 42, 3401–3406. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Jain, A.; Aguilera, A.; He, Z. Effective Control of Biohythane Composition through Operational Strategies in an Innovative Microbial Electrolysis Cell. Appl. Energy 2017, 206, 879–886. [Google Scholar] [CrossRef]
- Lanas, V.; Ahn, Y.; Logan, B.E. Effects of Carbon Brush Anode Size and Loading on Microbial Fuel Cell Performance in Batch and Continuous Mode. J. Power Sources 2014, 247, 228–234. [Google Scholar] [CrossRef]
- Li, L.; Jiang, B.; Tang, D.; Zhang, X.; Yuan, K.; Zhang, Q. Alkaline Treatment of Used Carbon-Brush Anodes for Restoring Power Generation of Microbial Fuel Cells. RSC Adv. 2018, 8, 36754–36760. [Google Scholar] [CrossRef]
- Mohamed, H.O.; Sayed, E.T.; Obaid, M.; Choi, Y.-J.; Park, S.-G.; Al-Qaradawi, S.; Chae, K.-J. Transition Metal Nanoparticles Doped Carbon Paper as a Cost-Effective Anode in a Microbial Fuel Cell Powered by Pure and Mixed Biocatalyst Cultures. Int. J. Hydrogen Energy 2018, 43, 21560–21571. [Google Scholar] [CrossRef]
- Jiang, D.; Li, B. Granular Activated Carbon Single-Chamber Microbial Fuel Cells (GAC-SCMFCs): A Design Suitable for Large-Scale Wastewater Treatment Processes. Biochem. Eng. J. 2009, 47, 31–37. [Google Scholar] [CrossRef]
- Mu, Y.; Rabaey, K.; Rozendal, R.A.; Yuan, Z.; Keller, J. Decolorization of Azo Dyes in Bioelectrochemical Systems. Environ. Sci. Technol. 2009, 43, 5137–5143. [Google Scholar] [CrossRef]
- Abbas, A.A.; Farrag, H.H.; El-Sawy, E.; Allam, N.K. Microbial Fuel Cells with Enhanced Bacterial Catalytic Activity and Stability Using 3D Nanoporous Stainless Steel Anode. J. Clean. Prod. 2021, 285, 124816. [Google Scholar] [CrossRef]
- Batlle-Vilanova, P.; Puig, S.; Gonzalez-Olmos, R.; Vilajeliu-Pons, A.; Bañeras, L.; Balaguer, M.D.; Colprim, J. Assessment of Biotic and Abiotic Graphite Cathodes for Hydrogen Production in Microbial Electrolysis Cells. Int. J. Hydrogen Energy 2014, 39, 1297–1305. [Google Scholar] [CrossRef]
- Croese, E.; Pereira, M.A.; Euverink, G.-J.W.; Stams, A.J.M.; Geelhoed, J.S. Analysis of the Microbial Community of the Biocathode of a Hydrogen-Producing Microbial Electrolysis Cell. Appl. Microbiol. Biotechnol. 2011, 92, 1083–1093. [Google Scholar] [CrossRef] [PubMed]
- Dange, P.; Pandit, S.; Jadhav, D.; Shanmugam, P.; Gupta, P.K.; Kumar, S.; Kumar, M.; Yang, Y.-H.; Bhatia, S.K. Recent Developments in Microbial Electrolysis Cell-Based Biohydrogen Production Utilizing Wastewater as a Feedstock. Sustainability 2021, 13, 8796. [Google Scholar] [CrossRef]
- Tang, J.; Bian, Y.; Jin, S.; Sun, D.; Ren, Z.J. Cathode Material Development in the Past Decade for H2 Production from Microbial Electrolysis Cells. ACS Environ. Au 2022, 2, 20–29. [Google Scholar] [CrossRef]
- Kundu, A.; Sahu, J.N.; Redzwan, G.; Hashim, M.A. An Overview of Cathode Material and Catalysts Suitable for Generating Hydrogen in Microbial Electrolysis Cell. Int. J. Hydrogen Energy 2013, 38, 1745–1757. [Google Scholar] [CrossRef]
- Wang, J.; Mu, K.; Zhao, X.; Luo, D.; Yu, X.; Li, W.; Chu, J.; Yang, J.; Yang, Q. Uniform Distribution of Pd on GO-C Catalysts for Enhancing the Performance of Air Cathode Microbial Fuel Cell. Catalysts 2021, 11, 888. [Google Scholar] [CrossRef]
- Jeremiasse, A.W.; Hamelers, H.V.M.; Saakes, M.; Buisman, C.J.N. Ni Foam Cathode Enables High Volumetric H2 Production in a Microbial Electrolysis Cell. Int. J. Hydrogen Energy 2010, 35, 12716–12723. [Google Scholar] [CrossRef]
- Son, S.; Koo, B.; Chai, H.; Tran, H.V.H.; Pandit, S.; Jung, S.P. Comparison of Hydrogen Production and System Performance in a Microbial Electrolysis Cell Containing Cathodes Made of Non-Platinum Catalysts and Binders. J. Water Process Eng. 2021, 40, 101844. [Google Scholar] [CrossRef]
- Wang, L.; Liu, W.; He, Z.; Guo, Z.; Zhou, A.; Wang, A. Cathodic Hydrogen Recovery and Methane Conversion Using Pt Coating 3D Nickel Foam Instead of Pt-Carbon Cloth in Microbial Electrolysis Cells. Int. J. Hydrogen Energy 2017, 42, 19604–19610. [Google Scholar] [CrossRef]
- Yan, Z.H.; Wang, M.; Huang, B.X.; Zhao, J.S.; Liu, R.M. Carboxyl Multi-Wall Carbon Nanotubes Supported Pt-Ni Alloy Nanoparticles as Cathode Catalyst for Microbial Fuel Cells. Int. J. Electrochem. Sci. 2012, 7, 10825–10834. [Google Scholar]
- Wang, H.; Wang, G.; Ling, Y.; Qian, F.; Song, Y.; Lu, X.; Chen, S.; Tong, Y.; Li, Y. High Power Density Microbial Fuel Cell with Flexible 3D Graphene–Nickel Foam as Anode. Nanoscale 2013, 5, 10283–10290. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, R.; Krishnaraj, N.; Selvam, A.; Wong, J.W.-C.; Lee, P.K.H.; Leung, M.K.; Berchmans, S. Effect of composites based nickel foam anode in microbial fuel cell using Acetobacter aceti and Gluconobacter roseus as a biocatalysts. Bioresour. Technol. 2016, 217, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Zhao, B.; Jeon, Y.; Zhong, S.; Zhou, S.; Kim, S. Iron Phthalocyanine Supported on Amino-Functionalized Multi-Walled Carbon Nanotube as an Alternative Cathodic Oxygen Catalyst in Microbial Fuel Cells. Bioresour. Technol. 2011, 102, 5849–5854. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.-W.; Sun, X.-F.; Huang, Y.-X.; Sheng, G.-P.; Zhou, K.; Zeng, R.J.; Dong, F.; Wang, S.-G.; Xu, A.-W.; Tong, Z.-H.; et al. Nano-Structured Manganese Oxide as a Cathodic Catalyst for Enhanced Oxygen Reduction in a Microbial Fuel Cell Fed with a Synthetic Wastewater. Water Res. 2010, 44, 5298–5305. [Google Scholar] [CrossRef] [PubMed]
- Olivares-Ramírez, J.M.; Campos-Cornelio, M.L.; Uribe Godínez, J.; Borja-Arco, E.; Castellanos, R.H. Studies on the Hydrogen Evolution Reaction on Different Stainless Steels. Int. J. Hydrogen Energy 2007, 32, 3170–3173. [Google Scholar] [CrossRef]
- Kim, K.-Y.; Habas, S.E.; Schaidle, J.A.; Logan, B.E. Application of Phase-Pure Nickel Phosphide Nanoparticles as Cathode Catalysts for Hydrogen Production in Microbial Electrolysis Cells. Bioresour. Technol. 2019, 293, 122067. [Google Scholar] [CrossRef]
- Chae, K.-J.; Choi, M.-J.; Kim, K.-Y.; Ajayi, F.F.; Chang, I.-S.; Kim, I.S. Selective Inhibition of Methanogens for the Improvement of Biohydrogen Production in Microbial Electrolysis Cells. Int. J. Hydrogen Energy 2010, 35, 13379–13386. [Google Scholar] [CrossRef]
- Park, S.-G.; Chae, K.-J.; Lee, M. A Sulfonated Poly(Arylene Ether Sulfone)/Polyimide Nanofiber Composite Proton Exchange Membrane for Microbial Electrolysis Cell Application under the Coexistence of Diverse Competitive Cations and Protons. J. Memb. Sci. 2017, 540, 165–173. [Google Scholar] [CrossRef]
- Zamora, P.; Georgieva, T.; Ter Heijne, A.; Sleutels, T.H.J.A.; Jeremiasse, A.W.; Saakes, M.; Buisman, C.J.N.; Kuntke, P. Ammonia Recovery from Urine in a Scaled-up Microbial Electrolysis Cell. J. Power Sources 2017, 356, 491–499. [Google Scholar] [CrossRef]
- Lu, L.; Xing, D.; Xie, T.; Ren, N.; Logan, B.E. Hydrogen Production from Proteins via Electrohydrogenesis in Microbial Electrolysis Cells. Biosens. Bioelectron. 2010, 25, 2690–2695. [Google Scholar] [CrossRef]
- Shin, D.W.; Guiver, M.D.; Lee, Y.M. Hydrocarbon-Based Polymer Electrolyte Membranes: Importance of Morphology on Ion Transport and Membrane Stability. Chem. Rev. 2017, 117, 4759–4805. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Lin, Y.; Ma, H.; Jia, H.; Liu, X.; Lin, J. Preparation of Sulfonated Poly(Ether Ether Ketone) (SPEEK) Membrane Using Ethanol/Water Mixed Solvent. Mater. Lett. 2016, 169, 69–72. [Google Scholar] [CrossRef]
- Sleutels, T.H.J.A.; Hamelers, H.V.M.; Rozendal, R.A.; Buisman, C.J.N. Ion Transport Resistance in Microbial Electrolysis Cells with Anion and Cation Exchange Membranes. Int. J. Hydrogen Energy 2009, 34, 3612–3620. [Google Scholar] [CrossRef]
- Radhika, D.; Shivakumar, A.; Kasai, D.; Koutavarapu, R.; Peera, G. Microbial Electrolysis Cell as a Diverse Technology: Overview of Prospective Applications, Advancements, and Challenges. Energies 2022, 15, 2611. [Google Scholar] [CrossRef]
- Zuo, Y.; Cheng, S.; Logan, B. Ion Exchange Membrane Cathodes for Scalable Microbial Fuel Cells. Environ. Sci. Technol. 2008, 42, 6967–6972. [Google Scholar] [CrossRef]
- Cheng, G.; Li, Z.; Ren, S.; Han, D.; Xiao, M.; Wang, S.; Meng, Y. A Robust Composite Proton Exchange Membrane of Sulfonated Poly (Fluorenyl Ether Ketone) with an Electrospun Polyimide Mat for Direct Methanol Fuel Cells Application. Polymers 2021, 13, 523. [Google Scholar] [CrossRef]
- Amrut Pawar, A.; Karthic, A.; Lee, S.; Pandit, S.; Jung, S.P. Microbial Electrolysis Cells for Electromethanogenesis: Materials, Configurations and Operations. Environ. Eng. Res. 2022, 27, 200480–200484. [Google Scholar] [CrossRef]
- Rozendal, R.A.; Sleutels, T.H.J.A.; Hamelers, H.V.M.; Buisman, C.J.N. Effect of the Type of Ion Exchange Membrane on Performance, Ion Transport, and PH in Biocatalyzed Electrolysis of Wastewater. Water Sci. Technol. 2008, 57, 1757–1762. [Google Scholar] [CrossRef]
- Zhao, W.; Ci, S. Nanomaterials As Electrode Materials of Microbial Electrolysis Cells for Hydrogen Generation. In Nanomaterials for the Removal of Pollutants and Resource Reutilization; Elsevier: Amsterdam, The Netherlands, 2019; pp. 213–242. ISBN 978-0-12-814837-2. [Google Scholar]
- Kadier, A.; Kalil, M.; Abdeshahian, P.; Kuppam, C.; Mohamed, A.; Azman, N.F.; Logroño, W.; Simayi, Y.; Hamid, A. Recent Advances and Emerging Challenges in Microbial Electrolysis Cells (MECs) for Microbial Production of Hydrogen and Value-Added Chemicals. Renew. Sustain. Energy Rev. 2016, 61, 501–525. [Google Scholar] [CrossRef]
- Lu, L.; Xing, D.; Liu, B.; Ren, N. Enhanced Hydrogen Production from Waste Activated Sludge by Cascade Utilization of Organic Matter in Microbial Electrolysis Cells. Water Res. 2012, 46, 1015–1026. [Google Scholar] [CrossRef]
- Chookaew, T.; Prasertsan, P.; Ren, Z.J. Two-Stage Conversion of Crude Glycerol to Energy Using Dark Fermentation Linked with Microbial Fuel Cell or Microbial Electrolysis Cell. N. Biotechnol. 2014, 31, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Prévoteau, A.; Rabaey, K. A Novel Tubular Microbial Electrolysis Cell for High Rate Hydrogen Production. J. Power Sources 2017, 356, 484–490. [Google Scholar] [CrossRef]
- Fan, Y.; Sharbrough, E.; Liu, H. Quantification of the Internal Resistance Distribution of Microbial Fuel Cells. Environ. Sci. Technol. 2008, 42, 8101–8107. [Google Scholar] [CrossRef]
- Cerrillo, M.; Viñas, M.; Bonmatí, A. Anaerobic Digestion and Electromethanogenic Microbial Electrolysis Cell Integrated System: Increased Stability and Recovery of Ammonia and Methane. Renew. Energy 2018, 120, 178–189. [Google Scholar] [CrossRef]
- Ferraren-De Cagalitan, D.D.T.; Abundo, M.L.S. A Review of Biohydrogen Production Technology for Application towards Hydrogen Fuel Cells. Renew. Sustain. Energy Rev. 2021, 151, 111413. [Google Scholar] [CrossRef]
- Singh, L.; Miller, A.G.; Wang, L.; Liu, H. Scaling-up up-Flow Microbial Electrolysis Cells with a Compact Electrode Configuration for Continuous Hydrogen Production. Bioresour. Technol. 2021, 331, 125030. [Google Scholar] [CrossRef]
- Selembo, P.A.; Perez, J.M.; Lloyd, W.A.; Logan, B.E. High Hydrogen Production from Glycerol or Glucose by Electrohydrogenesis Using Microbial Electrolysis Cells. Int. J. Hydrogen Energy 2009, 34, 5373–5381. [Google Scholar] [CrossRef]
- Pendyala, B.; Chaganti, S.R.; Lalman, J.A.; Heath, D.D. Optimizing the Performance of Microbial Fuel Cells Fed a Combination of Different Synthetic Organic Fractions in Municipal Solid Waste. Waste Manag. 2016, 49, 73–82. [Google Scholar] [CrossRef]
- Damiano, L. Electricity Production from the Management of Municipal Solid Waste Leachate with Microbial Fuel Cells. Master’s Thesis, University of New Hampshire, Durham, NH, USA, 2009. [Google Scholar]
- Wang, W.; Zhang, B.; He, Z. Bioelectrochemical Deposition of Palladium Nanoparticles as Catalysts by Shewanella Oneidensis MR-1 towards Enhanced Hydrogen Production in Microbial Electrolysis Cells. Electrochim. Acta 2019, 318, 794–800. [Google Scholar] [CrossRef]
- Patil, K.; Kulkarni, B. Review of Recovery Methods for Acetic Acid from Industrial Waste Streams by Reactive Distillation. J. Water Pollut. Purif. Res. 2014, 1, 1–6. [Google Scholar]
- Liu, H.; Grot, S.; Logan, B.E. Electrochemically Assisted Microbial Production of Hydrogen from Acetate. Environ. Sci. Technol. 2005, 39, 4317–4320. [Google Scholar] [CrossRef] [PubMed]
- Chavez, D. Solid Waste Management; World Bank: Washington, DC, USA, 2019. [Google Scholar]
- Li, S.; Gang, C. Factors Affecting the Effectiveness of Bioelectrochemical System Applications: Data Synthesis and Meta-Analysis. Batteries 2018, 4, 34. [Google Scholar] [CrossRef]
- Hassan, M.; Fernandez, A.S.; Martin, I.S.; Xie, B.; Moran, A. Hydrogen evolution in microbial electrolysis cells treating landfill leachate: Dynamics of anodic biofilm. Int. J. Hydrogen Energy 2018, 43, 13051–13063. [Google Scholar] [CrossRef]
- Sonawane, J.; Adeloju, S.B.; Ghosh, P.C. Landfill leachate: A promising substrate for microbial fuel cells. Int. J. Hydrogen Energy 2017, 42, 23794–23798. [Google Scholar] [CrossRef]
- Cusick, R.D.; Kiely, P.D.; Logan, B.E. A Monetary Comparison of Energy Recovered from Microbial Fuel Cells and Microbial Electrolysis Cells Fed Winery or Domestic Wastewaters. Int. J. Hydrogen Energy 2010, 35, 8855–8861. [Google Scholar] [CrossRef]
- Heidrich, E.S.; Dolfing, J.; Scott, K.; Edwards, S.R.; Jones, C.; Curtis, T.P. Production of Hydrogen from Domestic Wastewater in a Pilot-Scale Microbial Electrolysis Cell. Appl. Microbiol. Biotechnol. 2013, 97, 6979–6989. [Google Scholar] [CrossRef]
- Min, B.; Kim, J.; Oh, S.; Regan, J.M.; Logan, B.E. Electricity Generation from Swine Wastewater Using Microbial Fuel Cells. Water Res. 2005, 39, 4961–4968. [Google Scholar] [CrossRef]
- Escapa, A.; Gil-Carrera, L.; García, V.; Morán, A. Performance of a Continuous Flow Microbial Electrolysis Cell (MEC) Fed with Domestic Wastewater. Bioresour. Technol. 2012, 117, 55–62. [Google Scholar] [CrossRef]
- Escapa, A.; Gómez, X.; Tartakovsky, B.; Morán, A. Estimating Microbial Electrolysis Cell (MEC) Investment Costs in Wastewater Treatment Plants: Case Study. Int. J. Hydrogen Energy 2012, 37, 18641–18653. [Google Scholar] [CrossRef]
- Fudge, T.; Bulmer, I.; Bowman, K.; Pathmakanthan, S.; Gambier, W.; Dehouche, Z.; Al-Salem, S.M.; Constantinou, A. Microbial Electrolysis Cells for Decentralised Wastewater Treatment: The Next Steps. Water 2021, 13, 445. [Google Scholar] [CrossRef]
- Guo, H.; Kim, Y. Stacked multi-electrode design of microbial electrolysis cells for rapid and low-sludge treatment of municipal wastewater. Biotechnol. Biofuels 2019, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kadier, A.; Jain, P.; Lai, B.; Kalil, M.S.; Kondaveeti, S.; Alabbosh, K.F.S.; Abu-Reesh, I.M.; Mohanakrishna, G. Biorefinery Perspectives of Microbial Electrolysis Cells (MECs) for Hydrogen and Valuable Chemicals Production through Wastewater Treatment. Biofuel Res. J. 2020, 7, 1128–1142. [Google Scholar] [CrossRef]
- Gurjar, R.; Behera, M. Treatment of Organic Fraction of Municipal Solid Waste in Bioelectrochemical Systems: A Review. J. Hazard. Toxic Radioact. Waste 2020, 24, 04020018. [Google Scholar] [CrossRef]
- Karluvalı, A.; Köroğlu, E.O.; Manav, N.; Çetinkaya, A.Y.; Özkaya, B. Electricity Generation from Organic Fraction of Municipal Solid Wastes in Tubular Microbial Fuel Cell. Sep. Purif. Technol. 2015, 156, 502–511. [Google Scholar] [CrossRef]
- Paritosh, K.; Yadav, M.; Mathur, S.; Balan, V.; Liao, W.; Pareek, N.; Vivekanand, V. Organic Fraction of Municipal Solid Waste: Overview of Treatment Methodologies to Enhance Anaerobic Biodegradability. Front. Energy Res. 2018, 6, 75. [Google Scholar] [CrossRef]
- Yu, E.H. Resource Recovery with Microbial Electrochemical Systems. In Microbial Electrochemical and Fuel Cells; Woodhead Publishing: Boston, MA, USA, 2016; pp. 321–339. ISBN 978-1-78242-375-1. [Google Scholar]
- Cotterill, S.E.; Dolfing, J.; Jones, C.; Curtis, T.P.; Heidrich, E.S. Low Temperature Domestic Wastewater Treatment in a Microbial Electrolysis Cell with 1 M2 Anodes: Towards System Scale-Up. Fuel Cells 2017, 17, 584–592. [Google Scholar] [CrossRef]
- Isabel San-Martín, M.; Mateos, R.; Carracedo, B.; Escapa, A.; Morán, A. Pilot-Scale Bioelectrochemical System for Simultaneous Nitrogen and Carbon Removal in Urban Wastewater Treatment Plants. J. Biosci. Bioeng. 2018, 126, 758–763. [Google Scholar] [CrossRef]
- Baeza, J.A.; Martínez-Miró, À.; Guerrero, J.; Ruiz, Y.; Guisasola, A. Bioelectrochemical Hydrogen Production from Urban Wastewater on a Pilot Scale. J. Power Sources 2017, 356, 500–509. [Google Scholar] [CrossRef]
- Lu, L.; Ren, Z.J. Microbial Electrolysis Cells for Waste Biorefinery: A State of the Art Review. Bioresour. Technol. 2016, 215, 254–264. [Google Scholar] [CrossRef]
- Rousseau, R.; Ketep, S.F.; Etcheverry, L.; Délia, M.-L.; Bergel, A. Microbial Electrolysis Cell (MEC): A Step Ahead towards Hydrogen-Evolving Cathode Operated at High Current Density. Bioresour. Technol. Reports 2020, 9, 100399. [Google Scholar] [CrossRef]
- San-Martín, M.I.; Sotres, A.; Alonso, R.M.; Díaz-Marcos, J.; Morán, A.; Escapa, A. Assessing Anodic Microbial Populations and Membrane Ageing in a Pilot Microbial Electrolysis Cell. Int. J. Hydrogen Energy 2019, 44, 17304–17315. [Google Scholar] [CrossRef]
- Segundo-Aguilar, A.; González-Gutiérrez, L.V.; Payá, V.C.; Feliu, J.; Buitrón, G.; Cercado, B. Energy and Economic Advantages of Simultaneous Hydrogen and Biogas Production in Microbial Electrolysis Cells as a Function of the Applied Voltage and Biomass Content. Sustain. Energy Fuels 2021, 5, 2003–2017. [Google Scholar] [CrossRef]
- Koul, Y.; Devda, V.; Varjani, S.; Guo, W.; Ngo, H.H.; Taherzadeh, M.J.; Chang, J.-S.; Wong, J.W.C.; Bilal, M.; Kim, S.-H.; et al. Microbial Electrolysis: A Promising Approach for Treatment and Resource Recovery from Industrial Wastewater. Bioengineered 2022, 13, 8115–8134. [Google Scholar] [CrossRef]
- Stoll, Z.A.; Ma, Z.; Trivedi, C.B.; Spear, J.R.; Xu, P. Sacrificing Power for More Cost-Effective Treatment: A Techno-Economic Approach for Engineering Microbial Fuel Cells. Chemosphere 2016, 161, 10–18. [Google Scholar] [CrossRef]
- Jourdin, L.; Sousa, J.; van Stralen, N.; Strik, D.P.B.T.B. Techno-Economic Assessment of Microbial Electrosynthesis from CO2 and/or Organics: An Interdisciplinary Roadmap towards Future Research and Application. Appl. Energy 2020, 279, 115775. [Google Scholar] [CrossRef]
- Chen, J.; Xu, W.; Wu, X.; E, J.; Lu, N.; Wang, T.; Zuo, H. System Development and Environmental Performance Analysis of a Pilot Scale Microbial Electrolysis Cell for Hydrogen Production Using Urban Wastewater. Energy Convers. Manag. 2019, 193, 52–63. [Google Scholar] [CrossRef]
- Pant, D.; Singh, A.; Van Bogaert, G.; Gallego, Y.A.; Diels, L.; Vanbroekhoven, K. An Introduction to the Life Cycle Assessment (LCA) of Bioelectrochemical Systems (BES) for Sustainable Energy and Product Generation: Relevance and Key Aspects. Renew. Sustain. Energy Rev. 2011, 15, 1305–1313. [Google Scholar] [CrossRef]
- Selembo, P.A.; Merrill, M.D.; Logan, B.E. Hydrogen Production with Nickel Powder Cathode Catalysts in Microbial Electrolysis Cells. Int. J. Hydrogen Energy 2010, 35, 428–437. [Google Scholar] [CrossRef]
- Shemfe, M.; Gadkari, S.; Yu, E.; Rasul, S.; Scott, K.; Head, I.M.; Gu, S.; Sadhukhan, J. Life Cycle, Techno-Economic and Dynamic Simulation Assessment of Bioelectrochemical Systems: A Case of Formic Acid Synthesis. Bioresour. Technol. 2018, 255, 39–49. [Google Scholar] [CrossRef]
- Foley, J.; Rozendal, R.; Hertle, C.; Lant, P.; Rabaey, K. Life Cycle Assessment of High-Rate Anaerobic Treatment, Microbial Fuel Cells, and Microbial Electrolysis Cells. Environ. Sci. Technol. 2010, 44, 3629–3637. [Google Scholar] [CrossRef]
- Savla, N.; Suman; Pandit, S.; Verma, J.P.; Awasthi, A.K.; Sana, S.S.; Prasad, R. Techno-Economical Evaluation and Life Cycle Assessment of Microbial Electrochemical Systems: A Review. Curr. Res. Green Sustain. Chem. 2021, 4, 100111. [Google Scholar] [CrossRef]
- Zhang, J.; Yuan, H.; Abu-Reesh, I.M.; He, Z.; Yuan, C. Life Cycle Environmental Impact Comparison of Bioelectrochemical Systems for Wastewater Treatment. Procedia CIRP 2019, 80, 382–388. [Google Scholar] [CrossRef]
- Öksüz, S. Life Cycle Assessment of Microbial Electrolysis Cells for Hydrogen Generation Using TRACI Methodology. Sak. Univ. J. Sci. 2022, 26, 620–632. [Google Scholar]

| Bacteria | Biological Description | Function in Relation to MECs | Electrogenic Properties | Source | |||
|---|---|---|---|---|---|---|---|
| Gram (+/−) | Oxygen | Other | Proteo Bacteria | ||||
| Geobacter genus | − | Anaerobic | Chemo-lithotrophic; Iron-reducing | Delta- probact | Geobacter transfers electrons onto the surface of electrodes, which can produce electricity out of waste organic matter. Commonly found species in MECs. | Present | [45,49,50,51,52,53] |
| Shewanella genus | − | Facultative anaerobic | Found in aquatic or marine life | Gamma- probact | The Shewanella species use a variety of compounds as electron acceptors, including oxygen, iron, manganese, uranium and nitrate. Commonly found species in MECs. | Present | [45,49,54,55,56] |
| Desulfovibrio genus | − | Anaerobic | Sulphate-reducing | Delta- probact | The Desulfovibrio species are identified in MFCs and MECs when the biocathode is enriched as an anode fed with H2 and acetate. Multiple articles suggest that the Desulfovibrio species may be a significant player in biohydrogen production. Consume and produce H2. | Present | [57,58] |
| Clostridium genus | + | Anaerobic | Mesophilic Known to produce H2 | - | Researchers have studied MFCthe power generation performance using Clostridium species of Gram-positive bacteria. | N/A | [53,55,59,60] |
| Firmicutes phylum | + (−) | Aerobic or facultative anaerobic | Phylum divided into clostridium and bacillus | - | Studies have shown that Bacillus subtilis and Bacillus megaterium are among the best-performing electrogenic bacteria. | Present | [61,62] |
| Pseudomonas genus | − | Aerobic and anaerobic | Rod-shaped | Gamma- probact | Transfer electrons to the electrode via self-produced phenazine-based mediators. | Present | [53,63,64] |
| Nitrospirota phylum | +/− | Aerobic | Chemo-litho-autotrophic, found in marine life | - | Nitrite-oxidizing. Nitrospirae spp. Are distantly related to the thermophilic and sulfate-reducing thermosdesulfovibrio spp. | N/A | [51,65,66] |
| Actinomycetota phylum | + | Anaerobic or aerobic. | Either terrestrial or aquatic | - | The marine actinobacterial strain Actinoalloteichus spp. is capable of generating bioelectricity. | N/A | [51,67,68] |
| Rhodobacter genus | − | Anaerobic or aerobic | Found in freshwater or marine life | Alpha-probact | Rhodobacter sphaeroides is known for its capability of using a wide variety of substrates and its high activity in hydrogen production under anaerobic conditions, including high electricity production in MFC. | N/A | [53,69,70,71,72] |
| Desulforomonas genus | − | Anaerobic | Sulfur/sulphate-reducing | Delta- probact | Desulfuromonas is a distinct phylogenetic cluster (one of three, the others being Geobacter and Dulfuromusa). | N/A | [65,73] |
| Bacteroidetes phylum | − | Anaerobic or aerobic | Rod-shaped | - | Bacteroides is one of the most abundant genera found in MEC and possesses extracellular electron transfer abilities. | Present | [51,53,55,56,74] |
| Material | Picture | Reference |
|---|---|---|
| Carbon felt |  | [110] |
| Carbon cloth |  | [119] |
| Graphite plate |  | [120] |
| Carbon fiber (often woven) |  | [121] |
| Carbon brush |  | [122] |
| Carbon mesh |  | [123] |
| Carbon paper |  | [123] |
| Activated carbon granules |  | [123] |
| Advantages | Disadvantages | Surface Modification | MEC/MFC Performance | Reference | |
|---|---|---|---|---|---|
| Carbon felt | Good conductivity, large porosity | Expensive, potential for clogging | Polyaniline doped Isopropanol and biohydrogen peroxide treatment | 460 mW · m2 COD removal: 69 mg · L−1 · day−1 0.44 mmol CH4 · L−1 · day−1 | [110] [96] |
| Carbon cloth | Flexible, large surface area, high mechanical strength, porous | Potential for clogging | Heated with ammonia gas None Heated with ammonia gas | 988 mW · m−2 0.69 L H2 · L−1 · day−1, 14 A · m−2 7.4 L H2 · L−1 · day−1, 607 A · m−2 | [112] [131] [126] |
| Graphite felt | Good conductivity, large porosity, chemical resistance | Expensive, potential for clogging | None Not stated | 3.3 A · m−2 1.85 L H2 · L−1 · day−1 | [132] [133] |
| Graphite plate | Good electric conductor, chemical resistance | Smooth surface, relatively expensive, low surface area | Treated with sandpaper and soaked in HCl 5.5–7.5% manganese incorporated | 120 A · m−3 17.9 L biogas · L−1 | [134] [135] |
| Graphite fiber (brush) | Porous, high surface area, good current collection, chemical resistance | Must be integrated correctly due to fiber distances, which can create dead zones, and due to electrode spacing, which can be hard to make sufficiently small, expensive | Heated with ammonia gas Heated with ammonia gas Heated with ammonia gas | 2400 mW · m−2 3.12 L H2 · L−1 · day−1, 292 A · m−3 17.8 L H2 · L−1 · day−1, 1830 A · m−3 | [125] [136] [126] |
| Carbon brush | Porous, high surface area, good current collection | Must be integrated correctly due to fiber distances, which can create dead zones, and due to electrode spacing, which can be hard to make sufficiently small, relatively expensive | None Heat-treated Soaked in an alkaline solution | 0.64 L biohythate · day−1 1270 mW · m−2 7.62 A · m−2 | [137] [138] [139] |
| Carbon mesh | Cheaper than carbon cloth but has many of the same properties, less potential for clogging | Pretreatment can be necessary | Heated with ammonia gas | 1015 mW · m−2 | [112] |
| Carbon paper | Porous, thin | Fragile, can be expensive, smooth surface | Nitrogen-doped carbon dots-supported Electrodeposition of cobalt | 0.32 mW 710 mA · m−2 | [113] [140] |
| Activated carbon granules | High surface area, relatively cheap, can be used for bioremediation | Potential for clogging, current collection can be challenging | None | 0.9 mW power output | [141] |
| Graphite granules | High surface area (smaller than carbon granules), relatively cheap, can be used for bioremediation | Potential for clogging, current collection can be challenging | Washed with diluted HCl | 137.37 mW · m−2 | [142] |
| Stainless steel | Can be combined with other materials for enhanced efficiency, a low cost, excellent conductivity, and resistance | Low surface area, smooth surface, low bacterial adhesion, surface treatment is necessary | None—microstructure stainless steel plate was used Anodized in mesh form | 21.5 A · m−2 430 mW · m−2 | [129] [143] |
| MEC Configuration | Anode | Cathode | Biohydrogen Productivity in L H2 · L−1 · day−1 | Externally Applied Voltage in V | Reference | |||
|---|---|---|---|---|---|---|---|---|
| Type | Dimensions | Material | Projected Surface Area in cm2 | Material | Projected Surface Area in cm2 | |||
| One chamber | 10 L | Carbon cloth-clamped to titanium current collector | 10,000 | Carbon cloth with 2.5 mg · cm−2 MoP clamped to the stainless steel 316 current collector | 2815 | 5.9 | 1.0 | [180] |
| Two-chamber tubular AMI membrane | 1 L | Stainless steel fiber felt, heat treated | 600 | Titanium mesh tube coated with platinum | - | 7.1 | 1.0 | [176] |
| Two-chamber H type separated by PEM | 300 mL in each chamber | Carbon cloth | 25 | Carbon cloth coated with 10% Pt | 25 | 0.28 | 1.1 | [49] |
| Two-chamber separated by a cation exchange membrane (CEM) | 100 mL | Graphite felt | 100 | Ni foam | 100 m2 · m2 | 2.2–2.7 | 0.7 | [57] |
| MEC Configuration | Anode Material | Cathode Material | Feedstock | Performance | Limitation/Problems for Scaleup | Reference | |
|---|---|---|---|---|---|---|---|
| Type and Size | Flow | ||||||
| Single chamber With 5 internal MEC setups 10 L | Continuous renewal 20 L · day−1 | Carbon cloth | Carbon cloth coated with MoP | Acetate and glucose | 5.9 L H2 · L−1 · day−1 | Setup proved scalable from 0.15 L to 10 L No industrially applicable feedstock was tried | [180] |
| Two-chamber AEM 10 cassettes 130 L Operated 150 days | Continuous internal recycle 65 L · day−1 | SS mesh wrapped with graphite fibers | SS wool | Glucose Glycerol Urban WW | 0.028 L H2 · L−1 · day−1 0.013 L H2 · L−1 · day−1 0.031 L H2 · L−1 · day−1 | Internal resistances limiting electrical potential received Material deterioration requiring maintenance Methane at the cathode | [205] |
| Two-chamber AEM 5 electrode pairs 150 L Operated 63 days | Continuous internal recycle 150 L · day−1 | Graphite felt | Graphite felt | Urban WW | Removed close to 70% of TOC | The setup aimed at removing carbon and nitrogen simultaneously, but only carbon removal was satisfactory Low hydrogen purity | [204] |
| Two-chamber 6 cassettes 120 L Operated 85 days | Continuous renewal 120 L · day−1 | Carbon felt | SS wool | Domestic WW | 0.015 L H2 · L−1 · day−1 | Loss of hydrogen gas, but pure hydrogen was produced High inefficiency and low COD removal | [192] |
| Single chamber 144 electrode pairs 1000 L Operated 100 days | Continuous renewal 1000 L · day−1 | Graphite fiber brushes | SS mesh | Winery WW | 0.027 L H2 · L−1 · day−1 62 % COD removed | Methanogens Low current density due to resistances Slow startup | [48] |
| Two-chamber 3 cassettes 175 L Operated 217 days | Continuous renewal 828 L · day−1 | Graphite felt | SS mesh combined with SS wool | Domestic WW | 0.003 L H2 · L−1 · day−1 60.6 % COD removed | Significant hydrogen loss. High inefficiency | [203] |
| Two-chamber CEM 16L Operated 103 days | 360 L · day−1 | Graphite felt | SS mesh | Pig slurry | 0.2 L H2 · L−1 · day−1 | Not optimal current density due to scaling of anode High solid content in the feed stream decrease MEC potential | [208] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jensen, L.S.; Kaul, C.; Juncker, N.B.; Thomsen, M.H.; Chaturvedi, T. Biohydrogen Production in Microbial Electrolysis Cells Utilizing Organic Residue Feedstock: A Review. Energies 2022, 15, 8396. https://doi.org/10.3390/en15228396
Jensen LS, Kaul C, Juncker NB, Thomsen MH, Chaturvedi T. Biohydrogen Production in Microbial Electrolysis Cells Utilizing Organic Residue Feedstock: A Review. Energies. 2022; 15(22):8396. https://doi.org/10.3390/en15228396
Chicago/Turabian StyleJensen, Line Schultz, Christian Kaul, Nilas Brinck Juncker, Mette Hedegaard Thomsen, and Tanmay Chaturvedi. 2022. "Biohydrogen Production in Microbial Electrolysis Cells Utilizing Organic Residue Feedstock: A Review" Energies 15, no. 22: 8396. https://doi.org/10.3390/en15228396
APA StyleJensen, L. S., Kaul, C., Juncker, N. B., Thomsen, M. H., & Chaturvedi, T. (2022). Biohydrogen Production in Microbial Electrolysis Cells Utilizing Organic Residue Feedstock: A Review. Energies, 15(22), 8396. https://doi.org/10.3390/en15228396







