Predicting Capacity Fade in Silicon Anode-Based Li-Ion Batteries
Abstract
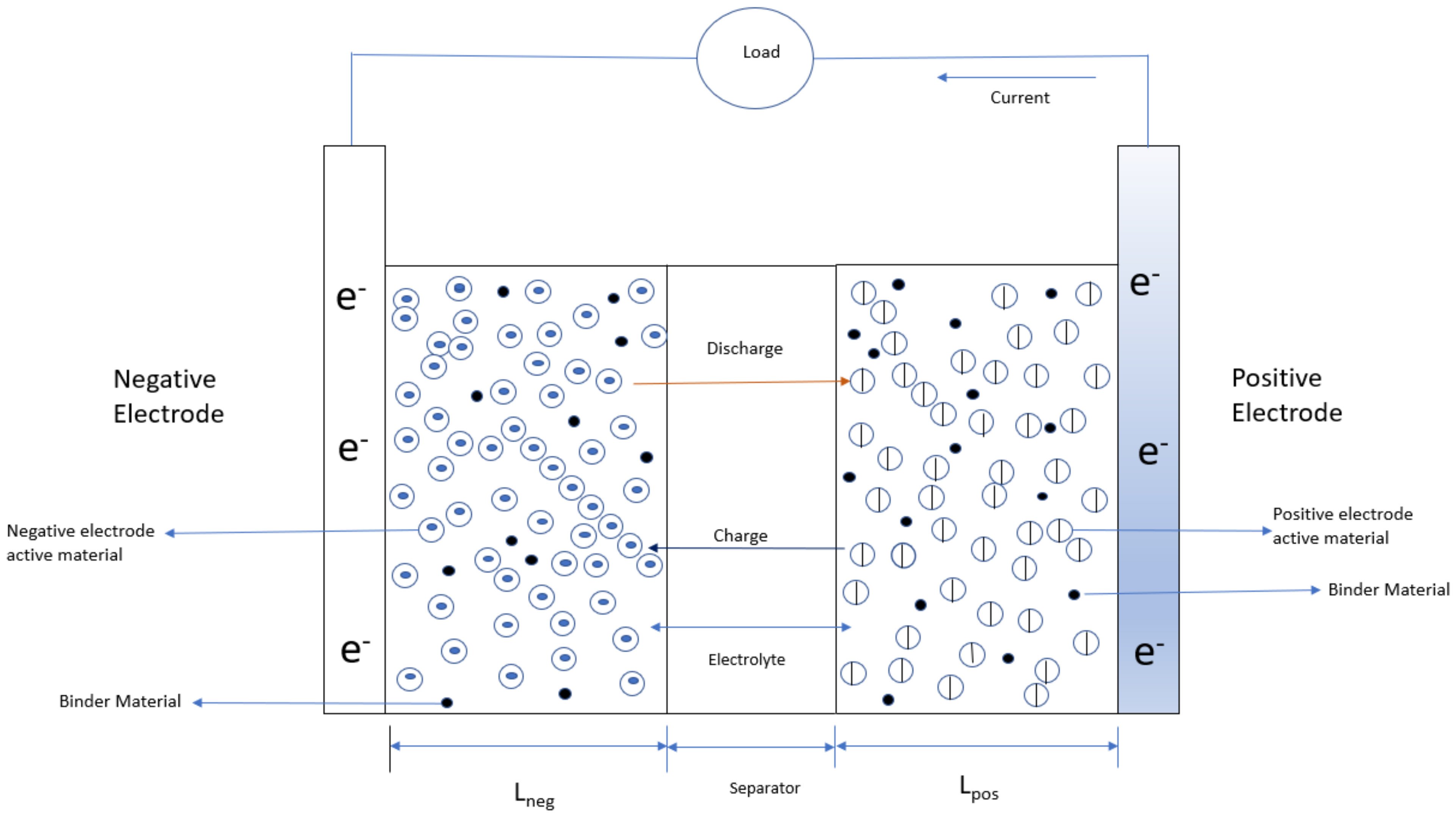
1. Introduction
2. Materials and Methods
2.1. Model Development
- = Solid phase volume fraction of the negative electrode
- = Solid phase volume fraction of the positive electrode
- = local current density,
- HK = dimensionless silicon expansion factor function, defined as zero during de–intercalation and the value depends on the state of charge of the negative electrode,
- J = exchange current density for the parasitic reaction (dimensionless),
- α = transfer coefficient of the electrochemical reduction reaction,
- = over-potential (assumes an equilibrium potential of 0 vs. lithium),
- = local accumulated charge from the SEI formation,
- f = parameter based on the SEI film properties (dimensionless)
- = reaction coefficient of the SEI species,
- n = number of electrons involved in the reaction,
- = electrode surface area (1/m)
- F = Faraday’s constant
- = SEI film conductivity (S/m)
2.2. Model Assumptions
3. Results
4. Discussion
4.1. Effect of Changing the Si Anode Particle Size
4.2. The Effect of Variations of the Electrolyte Volume Fraction in the Si Electrode
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| Specific surface area, m2/m3 | |
| Electronic current density in the solid phase (A m−2) | |
| Electrolyte current source | |
| The sum of all electrochemical current sources | |
| Li+ Transference number | |
| Electrolyte salt concentration (mol m−3) | |
| Average molar activity coefficient | |
| Current source term | |
| Flux of ions | |
| Total Li+ source term in the electrolyte | |
| r | Radius distance variable of the solid particles (m) |
| Local current density (A m−2) | |
| Exchange current density (A m−2) | |
| T | Battery Temperature (K) |
| R | Gas constant, 8.314 (J mol−1 K−1) |
| Electronic conductivity of solid phase (S m−1) | |
| Ionic conductivity of the electrolyte (S m−1) | |
| Solid phase volume fraction of the negative electrode | |
| Solid phase volume fraction of the positive electrode | |
| Electrolyte phase volume fraction negative electrode | |
| Electrolyte phase volume fraction positive electrode | |
| Electrolyte volume fraction | |
| Electrode volume fraction | |
| Li+ Transference number | |
| H | Overpotential, V |
| αa | αc Anodic and cathodic transfer coefficients |
| Electrolyte Potential | |
| Electric Potential | |
| T | Battery Temperature (K) |
| R | Gas constant, 8.314 (J mol−1 K−1) |
| Subscripts: | |
| l | Solution Phase |
| s | Solid Phase |
| eff | Effective value of transport property in porous medium |
Appendix A
Appendix B
References
- Yoshino, A. The Birth of the Lithium-Ion Battery. Angew. Chem. Int. Ed. 2012, 51, 5798–5800. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Han, X.; Li, J.; Hua, J.; Ouyang, M. A review on the key issues for lithium-ion battery management in electric vehicles. J. Power Sources 2013, 226, 272–288. [Google Scholar] [CrossRef]
- Bruce, G.C.; Marcoux, L. Large lithium ion batteries for aerospace and aircraft applications. IEEE AESS Syst. Mag. 2001, 16, 24–28. [Google Scholar] [CrossRef]
- Smith, K.; Wang, C.-Y. Power and thermal characterization of a lithium-ion battery pack for hybrid-electric vehi-cles. J. Power Sources 2006, 160, 662–673. [Google Scholar] [CrossRef]
- Casimir, A.; Zhang, H.; Ogoke, O.; Amine, J.C.; Lu, J.; Wu, G. Silicon-based anodes for lithium-ion batteries: Effectiveness of materials synthesis and electrode preparation. Nano Energy 2016, 27, 359–376. [Google Scholar] [CrossRef]
- Horiba, T. Li-ion battery systems. Proc. IEEE 2014, 102, 939–950. [Google Scholar] [CrossRef]
- Miao, Y.; Hynan, P.; von Jouanne, A.; Yokochi, A. Current Li-Ion Battery Technologies in Electric Vehicles and Op-portunities for Advancements. Energies 2019, 12, 1074. [Google Scholar] [CrossRef]
- Zhou, W. Effects of external mechanical loading on stress generation during lithiation in Li-ion battery electrodes. Electrochim. Acta 2015, 185, 28–33. [Google Scholar] [CrossRef]
- Zhou, W.; Hao, F.; Fang, D. The effects of elastic stiffening on the evolution of the stress field within a spherical electrode particle of lithium-ion batteries. Int. J. Appl. Mech. 2013, 5, 1350040. [Google Scholar] [CrossRef]
- Teki, R.; Datta, M.K.; Krishnan, R.; Parker, T.C.; Lu, T.-M.; Kumta, P.N.; Koratkar, N. Nanostructured Silicon Anodes for Lithium Ion Rechargeable Batteries. Small 2009, 5, 2236–2242. [Google Scholar] [CrossRef]
- Agubra, V.A.; Fergus, J.W. The formation and stability of the solid electrolyte interface on the graphite anode. J. Power Sources 2014, 268, 153–162. [Google Scholar] [CrossRef]
- Verma, P.; Maire, P.; Novak, P. A review of the features and analyses of the solid electrolyte interphase in Li-ion bat-teries. Electrochim. Acta 2010, 55, 6332–6341. [Google Scholar] [CrossRef]
- Hannan, M.A.; Hoque, M.; Hussain, A.; Yusof, Y.; Ker, P.J. State-of-the-Art and Energy Management System of Lithium-Ion Batteries in Electric Vehicle Applications: Issues and Recommendations. IEEE Access 2018, 6, 19362–19378. [Google Scholar] [CrossRef]
- Arora, P.; White, R.E.; Doyle, M. Capacity Fade Mechanisms and Side Reactions in Lithium-Ion Batteries. J. Electrochem. Soc. 1998, 145, 3647–3667. [Google Scholar] [CrossRef]
- Spotnitz, R. Simulation of capacity fade in lithium-ion batteries. J. Power Sources 2003, 113, 72–80. [Google Scholar] [CrossRef]
- Christensen, J.; Newman, J. Effect of Anode Film Resistance on the Charge/Discharge Capacity of a Lithium-Ion Battery. J. Electrochem. Soc. 2003, 150, A1416. [Google Scholar] [CrossRef]
- Ramadesigan, V.; Northrop, P.W.C.; De, S.; Santhanagopalan, S.; Braatz, R.D.; Subramanian, V.R. Multiscale Modeling and Simulation of Lithium-Ion Batteries from Systems Engineering Perspective. J. Electrochem. Soc. 2012, 159, R31–R45. [Google Scholar] [CrossRef]
- Jin, Y.; Zhu, B.; Lu, Z.; Liu, N.; Zhu, J. Challenges and Recent Progress in the Development of Si Anodes for Lithium-Ion Battery. Adv. Energy Mater. 2017, 7, 1700715. [Google Scholar] [CrossRef]
- Capacity Fade of a Lithium-Ion Battery; COMSOL Multiphysics® v. 5.5; COMSOL AB: Stockholm, Sweden, 2018.
- Ekström, H.; Lindbergh, G. A Model for Predicting Capacity Fade due to SEI Formation in a Commercial Graph-ite/LiFePO4 Cell. J. Electrochem. Soc. 2015, 162, A1003–A1007. [Google Scholar] [CrossRef]
- Safari, M.; Delacourt, C. Aging of a Commercial Graphite/LiFePO4 Cell. J. Electrochem. Soc. 2011, 158, A1123–A1135. [Google Scholar] [CrossRef]
- Salaha, M.; Murphy, P.; Hall, C.; Francis, F.; Kerr, R.; Fabretto, M. Pure silicon thin-film anodes for lithium-ion bat-teries: A review. J. Power Sources 2019, 414, 48–67. [Google Scholar] [CrossRef]
- Liu, X.H.; Zhong, L.; Huang, S.; Mao, S.X.; Zhu, T.; Huang, J.Y. Size-Dependent Fracture of Silicon Nanoparticles During Lithiation. ACS Nano 2012, 6, 1522–1531. [Google Scholar] [CrossRef]
- McDowell, M.T.; Ryu, I.; Lee, S.W.; Wang, C.; Nix, W.D.; Cui, Y. Studying the Kinetics of Crystalline Silicon Nano-particle Lithiation with In Situ Transmission Electron Microscopy. Adv. Mater. 2012, 24, 6034–6041. [Google Scholar] [CrossRef]
- Ma, Z.; Li, T.; Huang, Y.L.; Liu, J.; Zhou, Y.; Xue, D. Critical silicon-anode size for averting lithiation-induced me-chanical failure of lithium-ion batteries. RSC Adv. 2013, 3, 7398. [Google Scholar] [CrossRef]
- Mc Dowell, M.T.; Lee, S.W.; Nix, W.D.; Cui, Y. 25th anniversary article: Understanding the lithiation of silicon and other alloying anodes for lithium-ion batterries. Adv. Mater. 2013, 25, 4966–4985. [Google Scholar] [CrossRef]
- Kim, H.; Seo, M.; Park, M.-H.; Cho, J. A critical size of silicon nano-anodes for lithium rechargeable batteries. Angew. Chem. 2010, 122, 2192–2195. [Google Scholar] [CrossRef]
- Chen, Y.; Huo, W.; Lin, M.; Zhao, L. Simulation of electrochemical behavior in Lithium-ion battery during dis-charge process. PLoS ONE 2018, 13, e0189757. [Google Scholar]
- Wang, Y.; Li, H.; He, P.; Hosono, E.; Zhou, H. Nano active materials for lithium-ion batteries. Nanoscale 2010, 2, 1294–1305. [Google Scholar] [CrossRef] [PubMed]
- Mukharjee, R.; Krishnan, R.; Luc, T.-M.; Koratkar, N. Nanostructured electrodes for high-power lithium-ion batteries. Nano Energy 2012, 1, 518–533. [Google Scholar] [CrossRef]
- Roy, P.; Srivastava, S.K. Nanostructured anode materials for lithium ion batteries. J. Mater. Chem. A 2015, 3, 2454–2484. [Google Scholar] [CrossRef]
- Singh, M.; Kaiser, J.; Hahn, H. Effect of Porosity on the Thick Electrodes for High Energy Density Lithium-Ion Bat-teries for Stationary Applications. Batteries 2016, 2, 35. [Google Scholar] [CrossRef]
- Yuliya, P.; Heather, M.B.; Armando, F.; Daniel, L.C.; Jessica, R.-K.; Summer, R.F.; Babu, C. Degradation of Com-mercial Lithium-Ion Cells as a Function of Chemistry and Cycling Conditions. J. Electrochem. Soc. 2020, 167, 120532. [Google Scholar]
- Bryden, T.S.; Holland, A.; Hilton, G.; Dimitrov, B.; Albarrán, C.P.D.L.; Cruden, A. Lithium-ion degradation at varying discharge rates. Energy Procedia 2018, 151, 194–198. [Google Scholar] [CrossRef]
- Dubarry, M.; Truchot, C.; Liaw, B.Y. Cell degradation in commercial LiFePO4 cells with high-power and high-energy designs. J. Power Sources 2014, 258, 408–419. [Google Scholar] [CrossRef]
- Sun, S.; Guan, T.; Cheng, X.; Zuo, P.; Gao, Y.; Du, C.; Yin, G. Accelerated aging and degradation mechanism of LiFePO4/graphite batteries cycled at high discharge rates. RSC Adv. 2018, 8, 25695–25703. [Google Scholar] [CrossRef]
- Batteries and Fuel Cells Module User’s Guide; COMSOL Multiphysics® v. 5.5; COMSOL AB: Stockholm, Sweden, 2018; pp. 265–271.








| Name | Value | Units |
|---|---|---|
| α | 0.67 | 1 |
| J | 8.4 × 10−4 | 1 |
| f | 2 × 102 | 1/s |
| H | 6.7 | 1 |
| Parameter | Symbol | Value | Units |
|---|---|---|---|
| SEI layer conductivity | 5 × 10−6 | S/m | |
| Molar mass of product of side reaction | MP | 0.16 | Kg/mol |
| Density of product of side reaction | 1.6 × 103 | Kg/m3 |
| Parameter | Symbol | NMC | NCA | Separator | Silicon |
|---|---|---|---|---|---|
| Thickness () | L | 40 | 40 | 30 | 55 |
| Particle size (nm) | rP | 100assumed | 100assumed | Varied | |
| Volume fraction of the active material | Varied | Varied | |||
| Volume fraction of the electrolyte | Varied | Varied | |||
| Electrolyte phase volume fraction separator | 0.370 | ||||
| Maximum Lithium concentration in the solid phase (mol/m3) | cs, max | 49,000 | 48,000 | 278,000 | |
| Maximum electrode state of charge | 0.975 | 1 | - | 0.98 | |
| Minimum electrode state of charge | 0 | 0.25 | - | 0 | |
| Diffusion coefficient of electrodes (m2/S) | 5× 10−13 | 1.5× 10−15 | 1× 10−13 | ||
| Diffusion coefficient of electrolyte (m2/S) | Equation (A1) | ||||
| Transfer coefficient | 0.5 | 0.5 | 0.5 | ||
| Transport number | 0.363 | ||||
| Electrolyte Lithium concentration (mol/m3) | ce, max | 1200 | |||
| Bruggeman coefficient for tortuosity | 1.5 | 1.5 | 1.5 | ||
| Electronic conductivity (S/m) | 100 | 91 | Equation (A2) | 0.1 [22] | |
| Faraday’s Constant (C mole −1) | F | 96,487 | |||
| Universal gas constant (J/mol/K) | R | 8.314 | |||
| Temperature | T | 318.15 K |
| Particle Size | Effect on the Discharge Curve |
|---|---|
| 4m | Polarization persists |
| m | Polarization persists |
| m | Polarization persists |
| 150 nm | No Polarization |
| 100 nm | No Polarization |
| Particle Size | Effect on the Discharge Curve |
|---|---|
| 4m | Polarization persists |
| m | Polarization persists |
| m | Polarization persists |
| 150 nm | No Polarization |
| 100 nm | No Polarization |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dasari, H.; Eisenbraun, E. Predicting Capacity Fade in Silicon Anode-Based Li-Ion Batteries. Energies 2021, 14, 1448. https://doi.org/10.3390/en14051448
Dasari H, Eisenbraun E. Predicting Capacity Fade in Silicon Anode-Based Li-Ion Batteries. Energies. 2021; 14(5):1448. https://doi.org/10.3390/en14051448
Chicago/Turabian StyleDasari, Harika, and Eric Eisenbraun. 2021. "Predicting Capacity Fade in Silicon Anode-Based Li-Ion Batteries" Energies 14, no. 5: 1448. https://doi.org/10.3390/en14051448
APA StyleDasari, H., & Eisenbraun, E. (2021). Predicting Capacity Fade in Silicon Anode-Based Li-Ion Batteries. Energies, 14(5), 1448. https://doi.org/10.3390/en14051448





