The Effect of Antibiotics on Mesophilic Anaerobic Digestion Process of Cattle Manure
Abstract
1. Introduction
2. Materials and Methods
2.1. Anaerobic Digestion
2.2. Analyses of Antibiotic Concentration
2.3. Genomic DNA (gDNA) Isolation and Quantitative Analysis by qPCR
2.4. Illumina MiSeq Sequencing
2.5. Statistical Analyses
3. Results
3.1. Fate of Antibiotics During AD
3.2. Impact of AD on Microbial Community
3.3. Impact of AD on the Prevalence of mcrA, MSC and MST Genes
3.3.1. mcrA Gene
3.3.2. MST and MSC Genes
3.4. Impact of AD Process on the AAs of Analysed ARGs and Integrase Genes, and Their Prevalence in Bioreactors
3.4.1. tet Genes
3.4.2. bla Genes
3.4.3. sul1 Gene
3.4.4. MLS Group Genes
3.4.5. Fluoroquinolones Resistance Genes
3.4.6. Integrase Genes
3.5. Results of Statistical Analyses
3.6. Impact of Physicochemical Parameters on the Microbial Community in Bioreactors
3.7. Future Prospects
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adekunle, K.F.; Okolie, J.A. A Review of Biochemical Process of Anaerobic Digestion. Adv. Biosci. Biotechnol. 2015, 6, 205–212. [Google Scholar] [CrossRef]
- Spielmeyer, A. Occurrence and fate of antibiotics in manure during manure treatments: A short review. Sustain. Chem. Pharm. 2018, 9, 76–86. [Google Scholar] [CrossRef]
- Wallace, J.S.; Aga, D.S. Enhancing Extraction and Detection of Veterinary Antibiotics in Solid and Liquid Fractions of Manure. J. Environ. Qual. 2016, 45, 471–479. [Google Scholar] [CrossRef]
- Ahmad, M.; Vithanage, M.; Kim, K.; Cho, J.-S.; Lee, Y.H.; Joo, Y.K.; Lee, S.S.; Ok, Y.S. Inhibitory Effect of Veterinary Antibiotics on Denitrification in Groundwater: A Microcosm Approach. Sci. World J. 2014, 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gou, M.; Hu, H.-W.; Zhang, Y.-J.; Wang, J.-T.; Hayden, H.; Tang, Y.-Q.; He, J.-Z. Aerobic composting reduces antibiotic resistance genes in cattle manure and the resistome dissemination in agricultural soils. Sci. Total. Environ. 2018, 612, 1300–1310. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, M.; Ma, X.; Gao, Q.; Wang, T.; Shi, X.; Zhou, J.; Zuo, J.; Yang, Y. High variations of methanogenic microorganisms drive full-scale anaerobic digestion process. Environ. Int. 2019, 126, 543–551. [Google Scholar] [CrossRef]
- Tian, Z.; Zhang, Y.; Yu, Z.; Yang, M. Changes of resistome, mobilome and potential hosts of antibiotic resistance genes during the transformation of anaerobic digestion from mesophilic to thermophilic. Water Res. 2016, 98, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; Levin, S.A.; Goossens, H.; Laxminarayan, R. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463–E3470. [Google Scholar] [CrossRef]
- Arikan, O.A.; Sikora, L.J.; Mulbry, W.; Khan, S.U.; Rice, C.; Foster, G.D. The fate and effect of oxytetracycline during the anaerobic digestion of manure from therapeutically treated calves. Process. Biochem. 2006, 41, 1637–1643. [Google Scholar] [CrossRef]
- Lallai, A.; Mura, G.; Onnis, N. The efects of certain antibiotics on biogas production in the naerobic di-gestion. Bioresour. Technol. 2002, 82, 205–208. [Google Scholar] [CrossRef]
- Loftin, K.A.; Henny, C.; Adams, C.D.; Surampali, R.; Mormile, M.R. Inhibition of microbial metabolism in anaerobic lagoons by selected sulfonamides, tetracyclines, lincomycin, and tylosin tartrate. Environ. Toxicol. Chem. 2005, 24, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Chambers, L.; Yang, Y.; Littier, H.; Ray, P.; Zhang, T.; Pruden, A.; Strickland, M.; Knowlton, K. Metagenomic Analysis of Antibiotic Resistance Genes in Dairy Cow Feces following Therapeutic Administration of Third Generation Cephalosporin. PLoS ONE 2015, 10, e0133764. [Google Scholar] [CrossRef] [PubMed]
- Han, X.-M.; Hu, H.-W.; Chen, Q.-L.; Yang, L.-Y.; Li, H.-L.; Zhu, Y.-G.; Li, X.-Z.; Ma, Y.-B. Antibiotic resistance genes and associated bacterial communities in agricultural soils amended with different sources of animal manures. Soil Biol. Biochem. 2018, 126, 91–102. [Google Scholar] [CrossRef]
- Song, W.; Wang, X.; Gu, J.; Zhang, S.; Yin, Y.; Li, Y.; Qian, X.; Sun, W. Effects of different swine manure to wheat straw ratios on antibiotic resistance genes and the microbial community structure during anaerobic digestion. Bioresour. Technol. 2017, 231, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Du, K.; Yuan, R.; Chen, H.; Wang, F.; Zhou, B. Effects of sulfonamide antibiotics on digestion performance and microbial community during swine manure anaerobic digestion. Environ. Eng. Res. 2020, 26, 1. [Google Scholar] [CrossRef]
- Wang, Q.; Li, X.; Yang, Q.; Chen, Y.; Du, B. Evolution of microbial community and drug resistance during enrichment of tetracycline-degrading bacteria. Ecotoxicol. Environ. Saf. 2019, 171, 746–752. [Google Scholar] [CrossRef]
- Bajkacz, S.; Felis, E.; Kycia-Słocka, E.; Harnisz, M.; Korzeniewska, E. Development of a new SLE-SPE-HPLC-MS/MS method for the determination of selected antibiotics and their transformation products in anthropogenically altered solid environmental matrices. Sci. Total. Environ. 2020, 726, 138071. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Genet. 2010, 8, 423–435. [Google Scholar] [CrossRef]
- Czatzkowska, M.; Harnisz, M.; Korzeniewska, E.; Koniuszewska, I. Inhibitors of the methane fermentation process with particular emphasis on the microbiological aspect: A review. Energy Sci. Eng. 2020, 8, 1880–1897. [Google Scholar] [CrossRef]
- Wang, F.; Han, W.; Chen, S.; Dong, W.; Qiao, M.; Hu, C.; Liu, B. Fifteen-Year Application of Manure and Chemical Fertilizers Differently Impacts Soil ARGs and Microbial Community Structure. Front. Microbiol. 2020, 11, 62. [Google Scholar] [CrossRef]
- Aydin, S.; Cetecioglu, Z.; Arikan, O.; Ince, B.; Ozbayram, E.G.; Ince, O. Inhibitory effects of antibiotic combinations on syntrophic bacteria, homoacetogens and methanogens. Chemosphere 2015, 120, 515–520. [Google Scholar] [CrossRef]
- Morris, R. Relating Methanogen Community Structure to Function in Anaerobic Wastewater Digesters. Diss. 2009. Available online: http://epublications.marquette.edu/dissertations_mu/138 (accessed on 20 October 2020).
- Ghosh, P.; Kumar, M.; Kapoor, R.; Kumar, S.S.; Singh, L.; Vijay, V.; Vijay, V.K.; Kumar, V.; Thakur, I.S. Enhanced biogas production from municipal solid waste via co-digestion with sewage sludge and metabolic pathway analysis. Bioresour. Technol. 2020, 296, 122275. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.; Lizasoain, J.; Nettmann, E.; Bergmann, I.; Mundt, K.; Klocke, M.; Rincon, M.; Amon, T.; Piringer, G. Effects of the Antibiotics Chlortetracycline and Enrofloxacin on the Anaerobic Digestion in Continuous Experiments. BioEnergy Res. 2014, 7, 1244–1252. [Google Scholar] [CrossRef]
- Sun, J.P.; Zheng, P.; Hu, B.L. Combined effect of antibiotics on anaerobic digestion of piggery wastewater. Huan Jing Ke Xue 2009, 30, 19–24. [Google Scholar]
- Nuengjamnong, C.; Rachdawong, P.; Chalermchaikit, T. Effect of amoxicillin on biogas production and the Eschericia coli population in biogas systems treating swine wastewater. Thai J. Vet. Med. 2010, 40, 57–62. [Google Scholar]
- Qiao, M.; Ying, G.-G.; Singer, A.C.; Zhu, Y.-G. Review of antibiotic resistance in China and its environment. Environ. Int. 2018, 110, 160–172. [Google Scholar] [CrossRef]
- Massé, D.; Lu, D.; Masse, L.; Droste, R. Effect of antibiotics on psychrophilic anaerobic digestion of swine manure slurry in sequencing batch reactors. Bioresour. Technol. 2000, 75, 205–211. [Google Scholar] [CrossRef]
- Álvarez, J.; Otero, L.; Lema, J.; Omil, F. The effect and fate of antibiotics during the anaerobic digestion of pig manure. Bioresour. Technol. 2010, 101, 8581–8586. [Google Scholar] [CrossRef]
- Loke, M.L.; Jespersen, S.; Vreeken, R.; Halling-Sørensen, B.; Tjørnelund, J. Determination of oxytetracycline and its degradation products by high-performance liquid chromatography-tandem mass spectrometry in manure-containing anaerobic test systems. J. Chromatogr. B 2003, 783, 11–23. [Google Scholar] [CrossRef]
- Kuhne, M.; Ihnen, D.; Moller, G.; Agthe, O. Stability of tetracycline in water and liquid manure. J. Vet. Med. Ser. A 2000, 47, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Arikan, O.A. Degradation and metabolization of chlortetracycline during the anaerobic digestion of manure from medicated calves. J. Hazard. Mater. 2008, 158, 485–490. [Google Scholar] [CrossRef]
- Arsand, J.B.; Hoff, R.B.; Jank, L.; Meirelles, L.N.; Díaz-Cruz, M.S.; Pizzolato, T.M.; Barceló, D. Transformation products of amoxicillin and ampicillin after photolysis in aqueous matrices: Identification and kinetics. Sci. Total. Environ. 2018, 642, 954–967. [Google Scholar] [CrossRef] [PubMed]
- Keßler, D.N.; Fokuhl, V.K.; Petri, M.S.; Spielmeyer, A. Abiotic transformation products of tetracycline and chlortetracycline in salt solutions and manure. Chemosphere 2019, 224, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Gao, Y.; Yu, K.; Zhou, H.; De Costa, Y.G.; Yi, S.; Zhuang, W.-Q. Microbial community in in-situ waste sludge anaerobic digestion with alkalization for enhancement of nutrient recovery and energy generation. Bioresour. Technol. 2020, 295, 122277. [Google Scholar] [CrossRef] [PubMed]
- De Vrieze, J.; Hennebel, T.; Brande, J.V.D.; Bilad, R.M.; Bruton, T.A.; Vankelecom, I.F.; Verstraete, W.; Boon, N. Anaerobic digestion of molasses by means of a vibrating and non-vibrating submerged anaerobic membrane bioreactor. Biomass Bioenergy 2014, 68, 95–105. [Google Scholar] [CrossRef]
- Ning, J.; Zhou, M.; Pan, X.; Li, C.; Lv, N.; Wang, T.; Cai, G.; Wang, R.; Li, J.; Zhu, G. Simultaneous biogas and biogas slurry production from co-digestion of pig manure and corn straw: Performance optimization and microbial community shift. Bioresour. Technol. 2019, 282, 37–47. [Google Scholar] [CrossRef]
- De Vrieze, J.; Saunders, A.M.; He, Y.; Fang, J.; Nielsen, P.H.; Verstraete, W.; Boon, N. Ammonia and temperature determine potential clustering in the anaerobic digestion microbiome. Water Res. 2015, 75, 312–323. [Google Scholar] [CrossRef]
- Zhang, Y.-J.; Hu, H.-W.; Chen, Q.-L.; Singh, B.K.; Yan, H.; Chen, D.; He, J.-Z. Transfer of antibiotic resistance from manure-amended soils to vegetable microbiomes. Environ. Int. 2019, 130, 104912. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.; Sun, W.; Gu, J.; Wang, X.-J.; Sun, J.-J.; Yin, Y.-N.; Duan, M.-L. Variable effects of oxytetracycline on antibiotic resistance gene abundance and the bacterial community during aerobic composting of cow manure. J. Hazard. Mater. 2016, 315, 61–69. [Google Scholar] [CrossRef]
- Levén, L.; Eriksson, A.R.B.; Schnürer, A. Effect of process temperature on bacterial and archaeal communities in two methanogenic bioreactors treating organic household waste. FEMS Microbiol. Ecol. 2007, 59, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Limam, R.D.; Chouari, R.; Mazéas, L.; Wu, T.; Li, T.; Grossin-Debattista, J.; Guerquin-Kern, J.; Saidi, M.; Landoulsi, A.; Sghir, A.; et al. Members of the uncultured bacterial candidate division WWE 1 are implicated in anaerobic digestion of cellulose. MicrobiologyOpen 2014, 3, 157–167. [Google Scholar] [CrossRef]
- Delbès, C. Bacterial and archaeal 16S rDNA and 16S rRNA dynamics during an acetate crisis in an anaerobic digestor ecosystem. FEMS Microbiol. Ecol. 2001, 35, 19–26. [Google Scholar] [CrossRef]
- Morris, R.; Schauer-Gimenez, A.; Bhattad, U.; Kearney, C.; Struble, C.A.; Zitomer, D.; Maki, J.S. Methyl coenzyme M reductase (mcrA) gene abundance correlates with activity measurements of methanogenic H 2 / CO 2 -enriched anaerobic biomass. Microb. Biotechnol. 2014, 7, 77–84. [Google Scholar] [CrossRef]
- Aydin, S.; Ince, B.; Ince, O. Application of real-time PCR to determination of combined effect of antibiotics on Bacteria, Methanogenic Archaea, Archaea in anaerobic sequencing batch reactors. Water Res. 2015, 76, 88–98. [Google Scholar] [CrossRef]
- Cetecioglu, Z.; Ince, B.; Gros, M.; Rodriguez-Mozaz, S.; Barceló, D.; Orhon, D.; Ince, O. Chronic impact of tetracycline on the biodegradation of an organic substrate mixture under anaerobic conditions. Water Res. 2013, 47, 2959–2969. [Google Scholar] [CrossRef]
- Martinez, J.L. Environmental pollution by antibiotics and by antibiotic resistance determinants. Environ. Pollut. 2009, 157, 2893–2902. [Google Scholar] [CrossRef]
- Wu, X.; Wei, Y.; Zheng, J.; Zhao, X.; Zhong, W. The behavior of tetracyclines and their degradation products during swine manure composting. Bioresour. Technol. 2011, 102, 5924–5931. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.; Wang, J.; Zhu, L.; Yang, L.; Yang, R. Distribution characteristics of antibiotic resistant bacteria and genes in fresh and composted manures of livestock farms. Sci. Total Environ. 2019, 695, 133781. [Google Scholar] [CrossRef] [PubMed]
- Speece, R.E. Anaerobic biotechnology for industrial wastewater treatment a description of several installations. Environ. Sci. Technol. 1983, 17, 416–427. [Google Scholar] [CrossRef]
- Bauer, F.; Perrson, T.; Hulteberg, C.; Tamm, D. Perspective: Jatropha cultivation in southern India: Assessing farmers’ experiences. Biofuels Bioprod. Biorefining 2014, 7, 499–511. [Google Scholar] [CrossRef]
- Turker, G.; Ince, O.; Ertekin, E.; Akyol, C.; Ince, B. Changes in performance and active microbial communities due to single and multiple effects of mixing and solid content in anaerobic digestion process of otc medicated cattle manure. Int. J. Renew. Energy Res. 2013, 3, 144–148. [Google Scholar] [CrossRef]
- Santamaría, J.; López, L.; Soto, C.Y. Detection and diversity evaluation of tetracycline resistance genes in grassland-based production systems in Colombia, South America. Front. Microbiol. 2011, 2, 252. [Google Scholar] [CrossRef] [PubMed]
- Selvam, A.; Xu, D.; Zhao, Z.; Wong, J.W. Fate of tetracycline, sulfonamide and fluoroquinolone resistance genes and the changes in bacterial diversity during composting of swine manure. Bioresour. Technol. 2012, 126, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Baker-Austin, C.; Wright, M.S.; Stepanauskas, R.; McArthur, J. Co-selection of antibiotic and metal resistance. Trends Microbiol. 2006, 14, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Allen, H.K.; Looft, T.; Bayles, D.O.; Humphrey, S.; Levine, U.Y.; Alt, D.; Stanton, T.B. Antibiotics in Feed Induce Prophages in Swine Fecal Microbiomes. MBio 2011, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mroczkowska, J.E.; Barlow, M. Fitness Trade-Offs in blaTEM Evolution. Antimicrob. Agents Chemother. 2008, 52, 2340–2345. [Google Scholar] [CrossRef]
- Myllyniemi, A.-L.; Rannikko, R.; Lindfors, E.; Niemi, A.; Bäckman, C. Microbiological and chemical detection of incurred penicillin G, oxytetracycline, enrofloxacin and ciprofloxacin residues in bovine and porcine tissues. Food Addit. Contam. 2000, 17, 991–1000. [Google Scholar] [CrossRef]
- Miryala, S.K.; Anbarasu, A.; Ramaiah, S. Role of SHV-11, a Class A β-Lactamase, Gene in Multidrug Resistance among Klebsiella pneumoniae Strains and Understanding Its Mechanism by Gene Network Analysis. Microb. Drug Resist. 2020, 26, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.P.; Gooch, C.A.; Lansing, S.; Schueler, J.; Hurst, J.J.; Sassoubre, L.; Crossette, E.M.; Aga, D.S. Invited review: Fate of antibiotic residues, antibiotic-resistant bacteria, and antibiotic resistance genes in US dairy manure management systems. J. Dairy Sci. 2020, 103, 1051–1071. [Google Scholar] [CrossRef]
- Xie, S.; Wickham, R.; Nghiem, L.D. Synergistic effect from anaerobic co-digestion of sewage sludge and organic wastes. Int. Biodeterior. Biodegrad. 2017, 116, 191–197. [Google Scholar] [CrossRef]
- Bouanane-Darenfed, A.; Fardeau, M.-L.; Grégoire, P.; Joseph, M.; Kebbouche-Gana, S.; Benayad, T.; Hacène, H.; Cayol, J.-L.; Ollivier, B. Caldicoprobacter algeriensis sp. nov. a New Thermophilic Anaerobic, Xylanolytic Bacterium Isolated from an Algerian Hot Spring. Curr. Microbiol. 2010, 62, 826–832. [Google Scholar] [CrossRef]
- Nowara, A.; Burhenne, A.J.; Spiteller, M. Binding of Fluoroquinolone Carboxylic Acid Derivatives to Clay Minerals. J. Agric. Food Chem. 1997, 45, 1459–1463. [Google Scholar] [CrossRef]
- Qu, A.; Brulc, J.M.; Wilson, M.K.; Law, B.F.; Theoret, J.R.; Joens, L.A.; Konkel, M.E.; Angly, F.; Dinsdale, E.A.; Edwards, R.A.; et al. Comparative Metagenomics Reveals Host Specific Metavirulomes and Horizontal Gene Transfer Elements in the Chicken Cecum Microbiome. PLoS ONE 2008, 3, e2945. [Google Scholar] [CrossRef] [PubMed]
- Gaze, W.H.; Zhang, L.; Abdouslam, N.A.; Hawkey, P.M.; Calvo-Bado, L.A.; Royle, J.; Brown, H.; Davis, S.D.; Kay, P.; Boxall, A.B.A.; et al. Impacts of anthropogenic activity on the ecology of class 1 integrons and integron-associated genes in the environment. ISME J. 2011, 5, 1253–1261. [Google Scholar] [CrossRef]
- Nesme, J.; Simonet, P. The soil resistome: A critical review on antibiotic resistance origins, ecology and dissemination potential in telluric bacteria. Environ. Microbiol. 2015, 17, 913–930. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Yi, Y.; Liu, F.; Lv, N.; Yang, X.; Li, J.; Hu, Y.; Zhu, B. Distribution and molecular profiling of class 1 integrons in MDR Acinetobacter baumannii isolates and whole genome-based analysis of antibiotic resistance mechanisms in a representative strain. Microbiol. Res. 2014, 169, 811–816. [Google Scholar] [CrossRef]
- Luo, Y.; Mao, D.; Rysz, M.; Zhou, Q.; Zhang, H.; Xu, L.; Alvarez, P.J.J. Trends in Antibiotic Resistance Genes Occurrence in the Haihe River, China. Environ. Sci. Technol. 2010, 44, 7220–7225. [Google Scholar] [CrossRef]
- Gao, P.; Gu, C.; Wei, X.; Li, X.; Chen, H.; Jia, H.; Liu, Z.; Xue, G.; Ma, C. The role of zero valent iron on the fate of tetracycline resistance genes and class 1 integrons during thermophilic anaerobic co-digestion of waste sludge and kitchen waste. Water Res. 2017, 111, 92–99. [Google Scholar] [CrossRef]
- Zhang, X.; Gu, J.; Wang, X.; Zhang, K.; Yin, Y.; Zhang, R.; Zhang, S. Effects of tylosin, ciprofloxacin, and sulfadimidine on mcrA gene abundance and the methanogen community during anaerobic digestion of cattle manure. Chemosphere 2019, 221, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Mainardis, M.; Flaibani, S.; Trigatti, M.; Goi, D. Techno-economic feasibility of anaerobic digestion of cheese whey in small Italian dairies and effect of ultrasound pre-treatment on methane yield. J. Environ. Manag. 2019, 246, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Ternes, T.; Joss, A. Pharmaceuticals and personal care products in Wastewater Treatment. Environ. Sci. Technol. 2004, 10, 393–399. [Google Scholar]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Kim, J.; Jeon, S.; Rhie, H.; Lee, B.; Park, M.; Lee, H.; Lee, J.; Kim, S. Rapid Detection of Extended Spectrum β-Lactamase (ESBL) for Enterobacteriaceae by use of a Multiplex PCR-based Method. Infect. Chemother. 2009, 41, 181. [Google Scholar] [CrossRef]
- Eitel, Z.; Sóki, J.; Urbán, E.; Nagy, E. The prevalence of antibiotic resistance genes in Bacteroides fragilis group strains isolated in different European countries. Anaerobe 2013, 21, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, T.; Shao, B.; Shen, J.; Wang, S.; Wu, Y. Plasmid-mediated quinolone resistance genes and antibiotic residues in wastewater and soil adjacent to swine feedlots: Potential transfer to agricultural lands. Environ. Health Perspect. 2012, 120, 1144–1149. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Robicsek, A.; Jacoby, G.A.; Sahm, D.; Hooper, D.C. Prevalence in the United States of aac(6′)-Ib-cr encoding a ciprofloxacin-modifying enzyme. Antimicrob. Agents Chemother. 2006, 50, 3953–3955. [Google Scholar] [CrossRef]
- Nawaz, M.; Sung, K.; Khan, S.A.; Khan, A.A.; Steele, R. Biochemical and molecular characterization of tetracycline-resistant Aeromonas veronii isolates from catfish. Appl. Environ. Microbiol. 2006, 72, 6461–6466. [Google Scholar] [CrossRef]
- Ng, L.K.; Martin, I.; Alfa, M.; Mulvey, M. Multiplex PCR for the detection of tetracycline resistant genes. Mol. Cell. Probes 2001, 15, 209–215. [Google Scholar] [CrossRef]
- Nadkarni, M.A.; Martin, F.E.; Jacques, N.A.; Hunter, N. Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology 2002, 148, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, C.; Lee, M.D.; Sanchez, S.; Hudson, C.; Phillips, B.; Register, B.; Grady, M.; Liebert, C.; Summers, A.O.; White, D.G.; et al. Incidence of class 1 and 2 integrases in clinical and commensal bacteria from livestock, companion animals, and exotics. Antimicrob. Agents Chemother. 2001, 45, 723–726. [Google Scholar] [CrossRef] [PubMed]
- Luton, P.E.; Wayne, J.M.; Sharp, R.J.; Riley, P.W. The mcrA gene as an alternative to 16S rRNA in the phylogenetic analysis of methanogen populations in landfill. Microbiology 2002, 148, 3521–3530. [Google Scholar] [CrossRef] [PubMed]
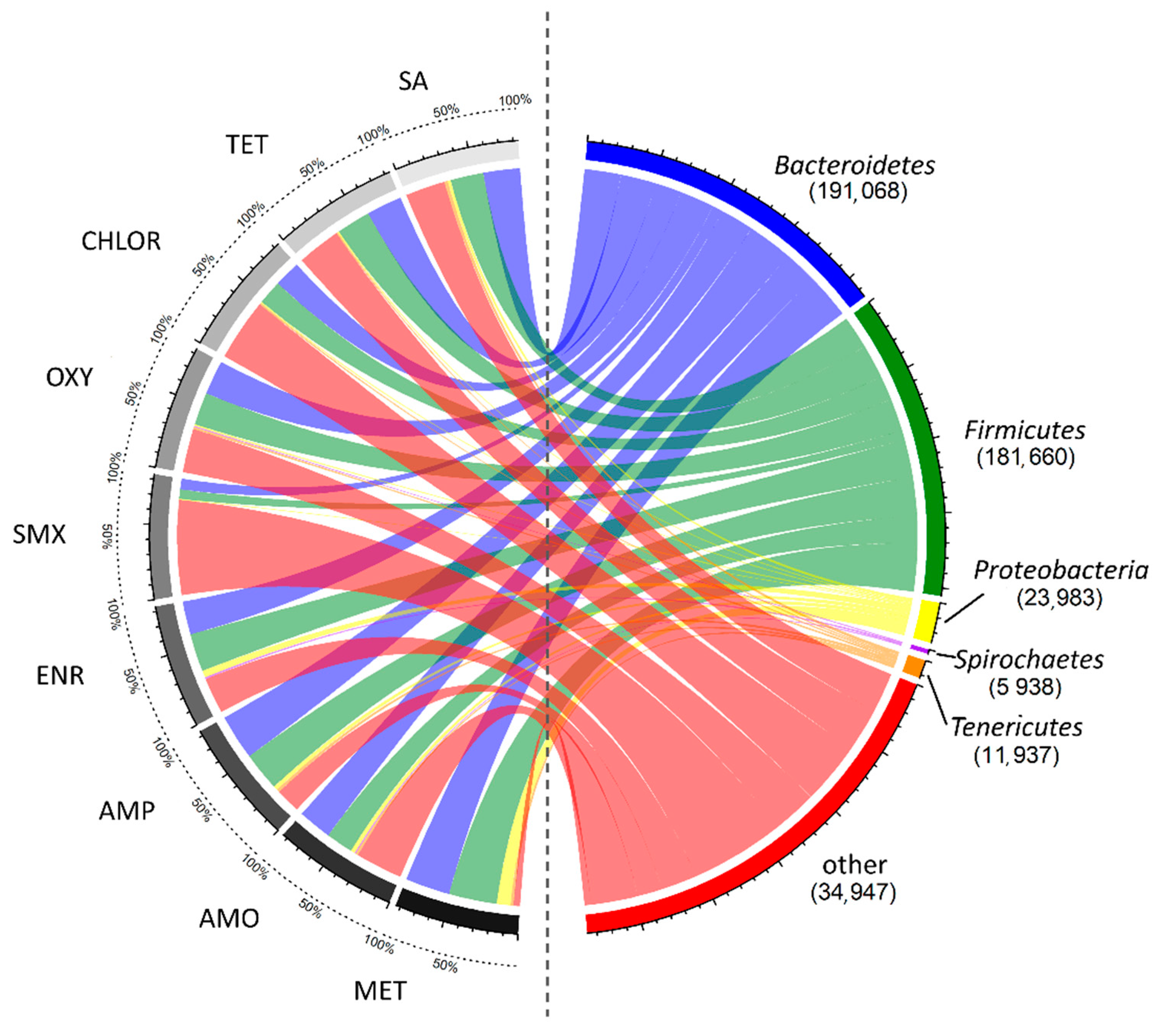
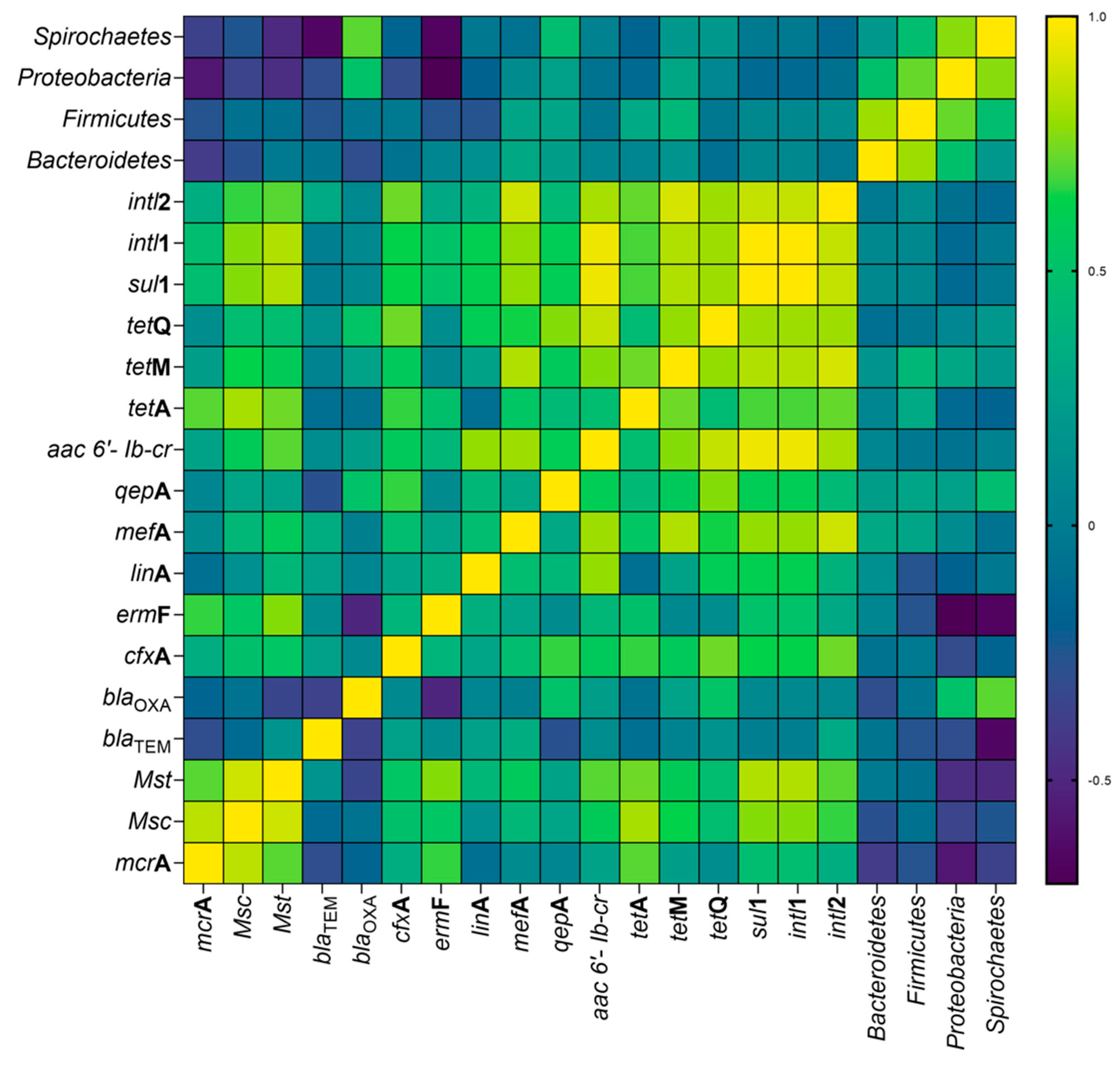
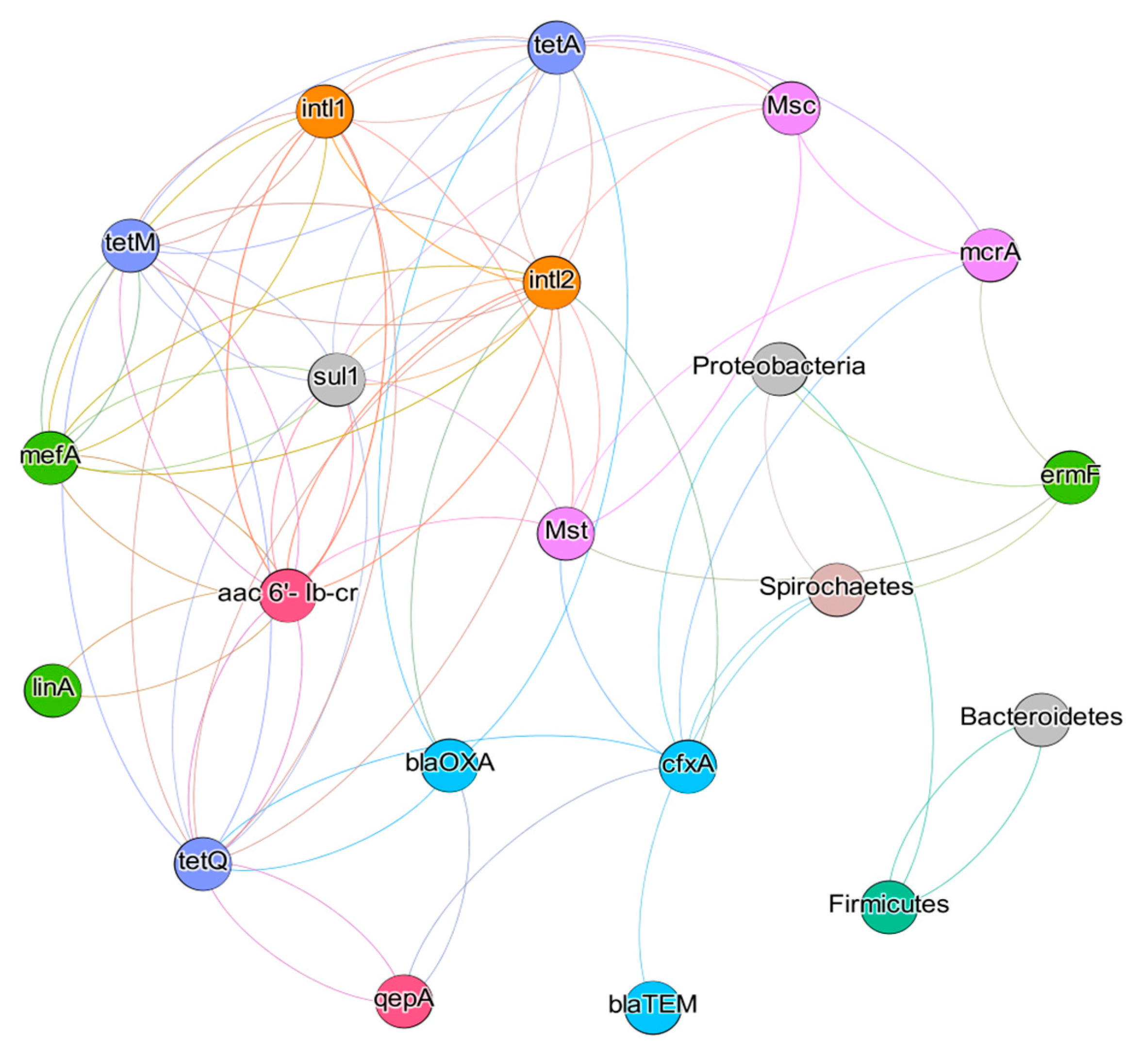
| Antibiotic | CH4 Production (L kg−1 VS) | CH4 Content in Biogas (%) | VFAs Concentration (g L−1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetic acid | Propionic acid | Iso-butyric acid | Butyric acid | Iso-valeric acid | Valeric acid | Iso-caproic acid | Caproic acid | Heptanoic acid | |||
| MET | 143.4 ± 44.0 | 70.7 ± 4.7 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| AMO | 51.2 ± 27.0 | 61.5 ± 11.4 | 16.11 ± 2.09 | 5.96 ± 0.99 | 6.46 ± 1.12 | 4.56 ± 0.89 | 7.75 ± 1.45 | 2.93 ± 0.43 | 4.34 ± 0.85 | 0.89 ± 0.29 | 0.23 ± 0.11 |
| AMP | 138.6 ± 19.6 | 69.1 ± 3.3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| ENR | 72.5 ± 14.7 | 72.9 ± 12.5 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| SMX | 183.1 ± 54.1 | 71.4 ± 3.9 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| OXY | 91.5 ± 33.1 | 68.2 ± 5.4 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| CHLOR | 107.0 ± 54.9 | 68.7 ± 4.1 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| TET | 79.2 ± 23.7 | 69.4 ± 3.8 | 6.35 ± 2.54 | 0.43 ± 0.09 | 0.48 ± 0.14 | 0.26 ± 0.07 | 2.35 ± 1.06 | 0.68 ± 0.22 | 0.35 ± 0.08 | 0.12 ± 0.04 | 0.03 ± 0.01 |
| SA | 201.2 ± 9.7 | 70.8 ± 3.5 | 0.26 ± 0.10 | 0.03 ± 0.03 | 0.03 ± 0.03 | 0.00 | 0.04 ± 0.03 | 0.02 ± 0.02 | 0.01 ± 0.01 | 0.01 ± 0.01 | 0.04 ± 0.02 |
| Genes Specific for Archaea | MLS Group | Fluoroquiolones | Tetracyclines | Sulfona-mides | Beta-Lactams | Integrase Genes | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mcrA | MSC | MST | ermF | linA | mefA | qepA | aac6’-Ib-cr | tetA | tetM | tetQ | sul1 | blaTEM | blaOXA | cfxA | intI1 | intI2 | |
| MET | 3.14 × 106 | 2.14 × 106 | 9.00 × 104 | 7.59 × 105 | 1.03 × 105 | 1.14 × 107 | 2.37 × 105 | 3.41 × 105 | 6.10 × 107 | 3.97 × 108 | 2.56 × 107 | 1.70 × 107 | 6.60 × 104 | 0 | 5.03 × 105 | 1.96 × 106 | 1.16 × 107 |
| AMO | 6.63 × 106 | 5.94 × 106 | 7.15 × 105 | 3.71 × 106 | 9.62 × 105 | 1.28 × 107 | 1.58 × 109 | 2.54 × 106 | 5.73 × 107 | 4.05 × 108 | 3.36 × 108 | 3.46 × 107 | 6.57 × 104 | 8.11 × 105 | 4.56 × 106 | 3.91 × 106 | 1.71 × 107 |
| AMP | 3.04 × 107 | 3.74 × 107 | 9.35 × 106 | 2.16 × 107 | 6.09 × 106 | 1.27 × 108 | 3.03 × 109 | 2.41 × 107 | 1.76 × 109 | 8.95 × 109 | 2.26 × 109 | 6.18 × 108 | 1.81 × 105 | 0 | 5.41 × 107 | 8.82 × 107 | 3.58 × 108 |
| ENR | 6.72 × 108 | 8.02 × 107 | 5.85 × 106 | 7.36 × 106 | 2.44 × 105 | 3.99 × 106 | 2.30 × 108 | 9.59 × 105 | 1.61 × 108 | 1.53 × 108 | 1.15 × 108 | 1.81 × 107 | 2.66 × 105 | 0 | 7.10 × 106 | 6.54 × 106 | 2.16 × 107 |
| SMX | 2.10 × 108 | 1.13 × 107 | 3.26 × 106 | 4.33 × 106 | 4.43 × 105 | 2.42 × 106 | 7.00 × 108 | 1.02 × 106 | 4.76 × 107 | 1.29 × 108 | 4.64 × 107 | 3.34 × 107 | 4.12 × 104 | 0 | 1.76 × 106 | 2.35 × 106 | 1.37 × 107 |
| OXY | 1.52 × 108 | 9.02 × 106 | 2.25 × 106 | 2.01 × 106 | 1.54 × 105 | 8.50 × 106 | 1.73 × 109 | 1.62 × 106 | 2.10 × 109 | 1.97 × 109 | 2.83 × 108 | 3.95 × 107 | 1.06 × 104 | 7.53 × 105 | 4.90 × 106 | 2.79 × 106 | 7.57 × 106 |
| CHLOR | 1.11 × 109 | 4.55 × 107 | 8.63 × 106 | 1.41 × 108 | 4.96 × 105 | 2.26 × 107 | 1.31 × 106 | 4.01 × 106 | 1.44 × 109 | 1.82 × 109 | 1.86 × 108 | 1.09 × 108 | 1.01 × 105 | 0 | 3.90 × 106 | 1.43 × 107 | 3.49 × 107 |
| TET | 1.05 × 109 | 9.75 × 107 | 2.30 × 107 | 3.37 × 108 | 3.24 × 105 | 2.12 × 107 | 1.30 × 109 | 2.13 × 106 | 2.36 × 109 | 1.74 × 109 | 1.30 × 108 | 7.70 × 107 | 5.05 × 104 | 0 | 7.40 × 106 | 1.58 × 107 | 3.75 × 107 |
| SA | 5.00 × 108 | 1.73 × 107 | 6.03 × 106 | 2.37 × 108 | 4.37 × 105 | 7.49 × 106 | 1.09 × 109 | 1.14 × 106 | 4.11 × 108 | 2.10 × 108 | 6.14 × 107 | 3.41 × 107 | 6.16 × 104 | 0 | 2.54 × 106 | 3.12 × 106 | 1.01 × 107 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koniuszewska, I.; Harnisz, M.; Korzeniewska, E.; Czatzkowska, M.; Jastrzębski, J.P.; Paukszto, Ł.; Bajkacz, S.; Felis, E.; Rusanowska, P. The Effect of Antibiotics on Mesophilic Anaerobic Digestion Process of Cattle Manure. Energies 2021, 14, 1125. https://doi.org/10.3390/en14041125
Koniuszewska I, Harnisz M, Korzeniewska E, Czatzkowska M, Jastrzębski JP, Paukszto Ł, Bajkacz S, Felis E, Rusanowska P. The Effect of Antibiotics on Mesophilic Anaerobic Digestion Process of Cattle Manure. Energies. 2021; 14(4):1125. https://doi.org/10.3390/en14041125
Chicago/Turabian StyleKoniuszewska, Izabela, Monika Harnisz, Ewa Korzeniewska, Małgorzata Czatzkowska, Jan Paweł Jastrzębski, Łukasz Paukszto, Sylwia Bajkacz, Ewa Felis, and Paulina Rusanowska. 2021. "The Effect of Antibiotics on Mesophilic Anaerobic Digestion Process of Cattle Manure" Energies 14, no. 4: 1125. https://doi.org/10.3390/en14041125
APA StyleKoniuszewska, I., Harnisz, M., Korzeniewska, E., Czatzkowska, M., Jastrzębski, J. P., Paukszto, Ł., Bajkacz, S., Felis, E., & Rusanowska, P. (2021). The Effect of Antibiotics on Mesophilic Anaerobic Digestion Process of Cattle Manure. Energies, 14(4), 1125. https://doi.org/10.3390/en14041125








