Current Status of the Pyrolysis and Gasification Mechanism of Biomass
Abstract
:1. Introduction
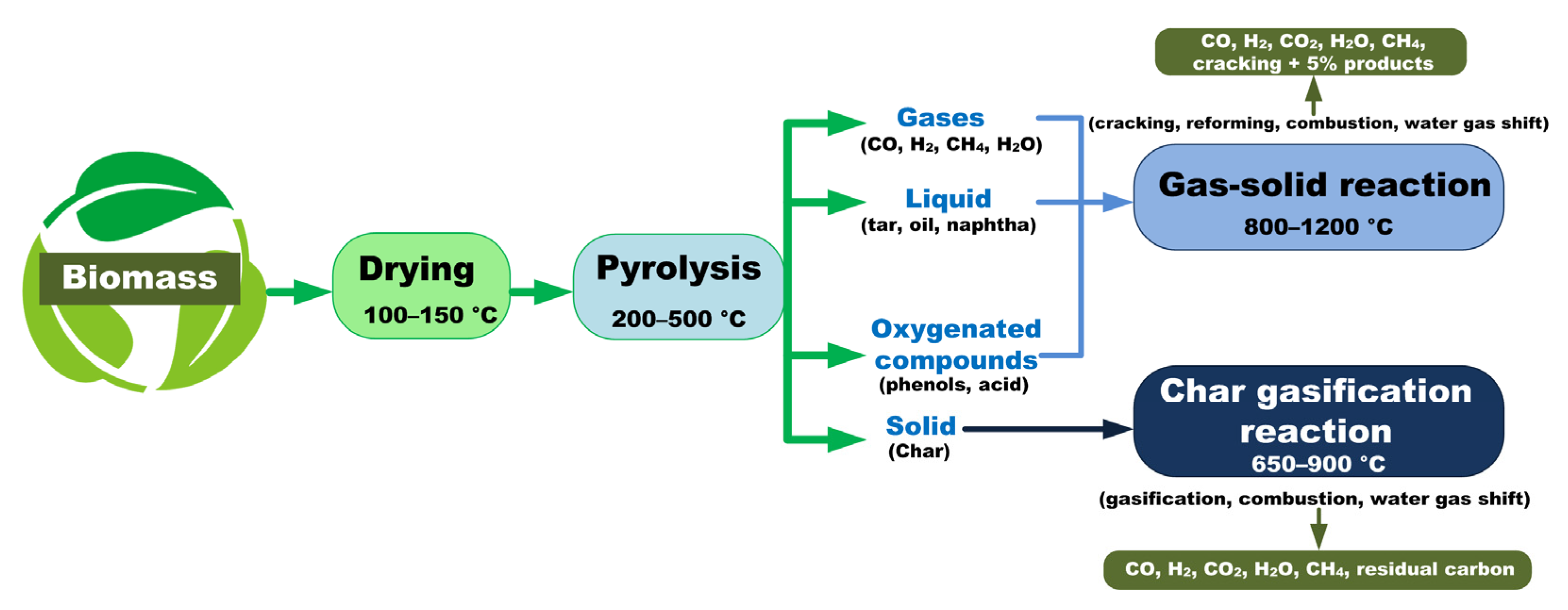
2. Mechanisms and Stages of Biomass Pyrolysis and Gasification
2.1. Drying
2.2. Pyrolysis
2.3. Gasification of Carbonaceous Residue
- C + CO2 → 2CO (absorbed heat, i.e., endothermic effect −14.6∙106 J/kg) [32],
- C + H2O → CO + H2 (absorbed heat, i.e., endothermic effect −10.9∙106 J/kg) [32],
- C + 2H2 → CH4 (proceeds only at temperatures above 500 °C with heat released, i.e., exothermic effect +8∙106 J/kg) [29]. These reactions develop on the surface and in the pores of biomass particles.
2.4. Pyrolysis and Gasification
3. Biomass Types Used for Pyrolysis and Gasification
4. Factors Influencing Pyrolysis and Gasification Efficiency
4.1. Effect of Biomass Composition
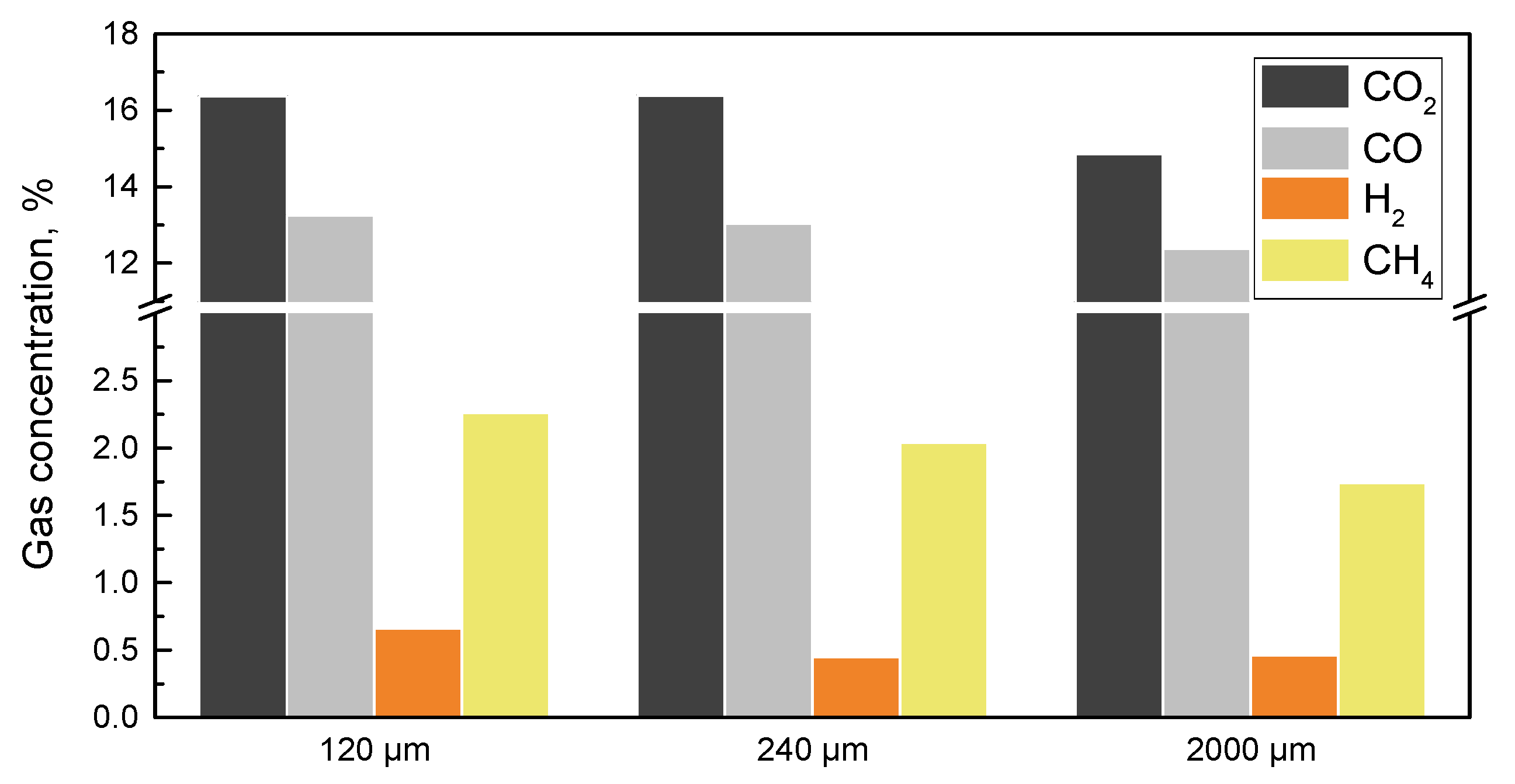
4.2. Effect of Biomass Particle Size
4.3. Effect of Biomass Structure
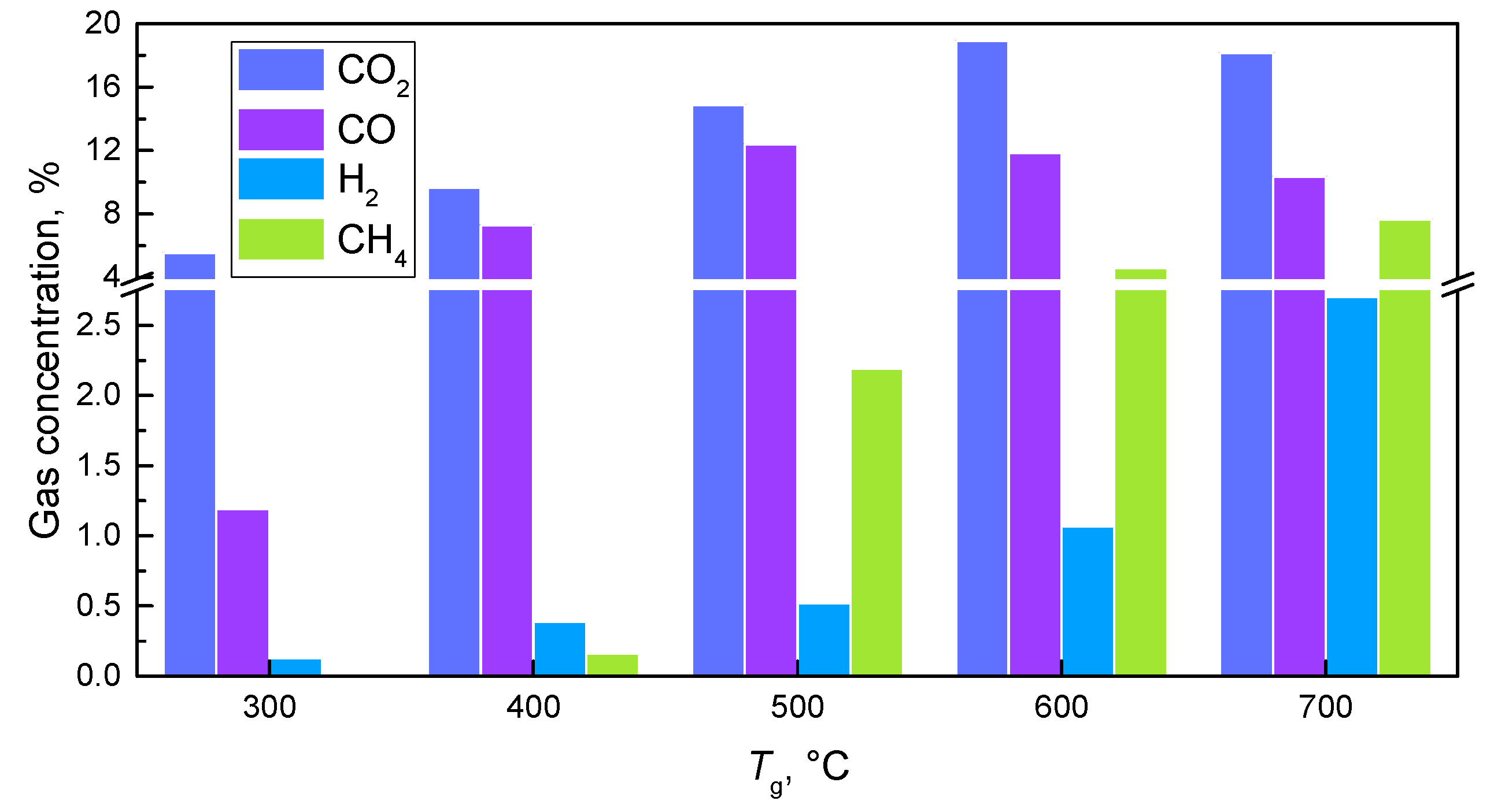
4.4. Effect of Temperature
4.5. Effect of Gasifying Agent
4.6. Effect of Catalysts
5. Mathematical Models of Pyrolysis and Gasification
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Qian, X.; Xue, J.; Yang, Y.; Lee, S.W. Thermal Properties and Combustion-Related Problems Prediction of Agricultural Crop Residues. Energies 2021, 14, 4619. [Google Scholar] [CrossRef]
- World Bioenergy Association. Global Bioenergy Statistics 2019; World Bioenergy Association: Stockholm, Sweden, 2019; 58p. [Google Scholar]
- Yogalakshmi, K.N.; Poornima Devi, T.; Sivashanmugam, P.; Kavitha, S.; Yukesh Kannah, R.; Varjani, S.; Adish Kumar, S.; Kumar, G.; Rajesh Banu, J. Lignocellulosic biomass-based pyrolysis: A comprehensive review. Chemosphere 2021, 286, 131824. [Google Scholar]
- Bhaskar, T.; Ruan, R.; Park, Y.K.; Yang, H.; Chen, G. Pyrolysis, combustion and gasification of biomass (PCGB-2020). Bioresour. Technol. 2020, 313, 123803. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Rajagopalan, K.; Lei, H.; Ruan, R.; Sharma, B.K. An overview of a novel concept in biomass pyrolysis: Microwave irradiation. Sustain. Energy Fuels 2017, 1, 1664–1699. [Google Scholar]
- Sikarwar, V.S.; Zhao, M.; Clough, P.; Yao, J.; Zhong, X.; Memon, M.Z.; Shah, N.; Anthony, E.J.; Fennell, P.S. An overview of advances in biomass gasification. Energy Environ. Sci. 2016, 9, 2939–2977. [Google Scholar]
- Li, H.; Wang, Y.; Zhou, N.; Dai, L.; Deng, W.; Liu, C.; Cheng, Y.; Liu, Y.; Cobb, K.; Chen, P.; et al. Applications of calcium oxide–based catalysts in biomass pyrolysis/gasification—A review. J. Clean. Prod. 2021, 291, 125826. [Google Scholar]
- Choe, K. Review of Wood Biomass Cyclone Burner. Energies 2021, 14, 4807. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Renewable fuels and chemicals by thermal processing of biomass. Chem. Eng. J. 2003, 91, 87–102. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Vassileva, C.G. An overview of the behaviour of biomass during combustion: Part I. Phase-mineral transformations of organic and inorganic matter. Fuel 2013, 112, 391–449. [Google Scholar] [CrossRef]
- Jach-Nocoń, M.; Pełka, G.; Luboń, W.; Mirowski, T.; Nocoń, A.; Pachytel, P. An Assessment of the Efficiency and Emissions of a Pellet Boiler Combusting Multiple Pellet Types. Energies 2021, 14, 4465. [Google Scholar] [CrossRef]
- Ren, J.; Cao, J.P.; Zhao, X.Y.; Liu, Y.L. Fundamentals and applications of char in biomass tar reforming. Fuel Process. Technol. 2021, 216, 106782. [Google Scholar] [CrossRef]
- Bulushev, D.A.; Ross, J.R.H. Catalysis for conversion of biomass to fuels via pyrolysis and gasification: A review. Catal. Today 2011, 171, 1–13. [Google Scholar] [CrossRef]
- Park, S.Y.; Oh, G.; Kim, K.; Seo, M.W.; Ra, H.W.; Mun, T.Y.; Lee, J.G.; Yoon, S.J. Deactivation characteristics of Ni and Ru catalysts in tar steam reforming. Renew. Energy 2017, 105, 76–83. [Google Scholar] [CrossRef]
- Oh, G.; Ra, H.W.; Yoon, S.M.; Mun, T.Y.; Seo, M.W.; Lee, J.G.; Yoon, S.J. Syngas production through gasification of coal water mixture and power generation on dual-fuel diesel engine. J. Energy Inst. 2019, 92, 265–274. [Google Scholar] [CrossRef]
- Rao, A.B.; Phadke, P.C. CO2 Capture and Storage in Coal Gasification Projects. IOP Conf. Ser. Earth Environ. Sci. 2017, 76, 012011. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, B.S.; Szklo, A. Integrated gasification combined cycle and carbon capture: A risky option to mitigate CO2 emissions of coal-fired power plants. Appl. Energy 2011, 88, 3917–3929. [Google Scholar] [CrossRef]
- Sofia, D.; Giuliano, A.; Poletto, M.; Barletta, D. Techno-economic analysis of power and hydrogen co-production by an IGCC plant with CO2 capture based on membrane technology. Comput. Aided Chem. Eng. 2015, 37, 1373–1378. [Google Scholar]
- Fahmi, R.; Bridgwater, A.V.; Donnison, I.; Yates, N.; Jones, J.M. The effect of lignin and inorganic species in biomass on pyrolysis oil yields, quality and stability. Fuel 2008, 87, 1230–1240. [Google Scholar] [CrossRef]
- Shihadeh, A.S.; Hochgreb, S. Diesel Engine Combustion of Biomass Pyrolysis Oils. Energy Fuels 2000, 14, 260–274. [Google Scholar] [CrossRef]
- Jahirul, M.I.; Rasul, M.G.; Chowdhury, A.A.; Ashwath, N. Biofuels Production through Biomass Pyrolysis—A Technological Review. Energies 2012, 5, 4952–5001. [Google Scholar] [CrossRef]
- Goyal, H.B.; Seal, D.; Saxena, R.C. Bio-fuels from thermochemical conversion of renewable resources: A review. Renew. Sustain. Energy Rev. 2008, 12, 504–517. [Google Scholar] [CrossRef]
- Giuliano, A.; Freda, C.; Catizzone, E. Techno-Economic Assessment of Bio-Syngas Production for Methanol Synthesis: A Focus on the Water–Gas Shift and Carbon Capture Sections. Bioenergy 2020, 7, 70. [Google Scholar] [CrossRef]
- Nkosi, N.; Muzenda, E.; Gorimbo, J.; Belaid, M. Developments in waste tyre thermochemical conversion processes: Gasification, pyrolysis and liquefaction. RSC Adv. 2021, 11, 11844–11871. [Google Scholar] [CrossRef]
- Neves, D.; Thunman, H.; Matos, A.; Tarelho, L.; Gómez-Barea, A. Characterization and prediction of biomass pyrolysis products. Prog. Energy Combust. Sci. 2011, 37, 611–630. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Glushkov, D.O.; Nyashina, G.S.; Anand, R.; Strizhak, P.A. Composition of gas produced from the direct combustion and pyrolysis of biomass. Process Saf. Environ. Prot. 2021, 156, 43–56. [Google Scholar] [CrossRef]
- Kuznetsov, G.V.; Syrodoy, S.V.; Gutareva, N.Y. Influence of a wet wood particle form on the characteristics of its ignition in the high-temperature medium. Renew. Energy 2020, 145, 1474–1486. [Google Scholar] [CrossRef]
- Wickramaarachchi, W.A.M.K.P.; Narayana, M. Pyrolysis of single biomass particle using three-dimensional Computational Fluid Dynamics modelling. Renew. Energy 2020, 146, 1153–1165. [Google Scholar] [CrossRef]
- Williams, P.T.; Besler, S. The influence of temperature and heating rate on the slow pyrolysis of biomass. Renew. Energy 1996, 7, 233–250. [Google Scholar] [CrossRef]
- Hanchate, N.; Ramani, S.; Mathpati, C.S.; Dalvi, V.H. Biomass gasification using dual fluidized bed gasification systems: A review. J. Clean. Prod. 2021, 280, 123148. [Google Scholar] [CrossRef]
- Duffy, N.T.M.; Eaton, J.A. Investigation of factors affecting channelling in fixed-bed solid fuel combustion using CFD. Combust. Flame 2013, 160, 2204–2220. [Google Scholar] [CrossRef]
- Sand, U.; Sandberg, J.; Larfeldt, J.; Bel Fdhila, R. Numerical prediction of the transport and pyrolysis in the interior and surrounding of dry and wet wood log. Appl. Energy 2008, 85, 1208–1224. [Google Scholar] [CrossRef]
- Haseli, Y.; van Oijen, J.A.; de Goey, L.P.H. A detailed one-dimensional model of combustion of a woody biomass particle. Bioresour. Technol. 2011, 102, 9772–9782. [Google Scholar] [CrossRef]
- Papadikis, K.; Gu, S.; Bridgwater, A.V.; Gerhauser, H. Application of CFD to model fast pyrolysis of biomass. Fuel Process. Technol. 2009, 90, 504–512. [Google Scholar] [CrossRef]
- Socci, J.; Saraeian, A.; Stefanidis, S.D.; Banks, S.W.; Shanks, B.H.; Bridgwater, T. The role of catalyst acidity and shape selectivity on products from the catalytic fast pyrolysis of beech wood. J. Anal. Appl. Pyrolysis 2019, 104710. [Google Scholar] [CrossRef]
- Riaza, J.; Mason, P.; Jones, J.M.; Gibbins, J.; Chalmers, H. High temperature volatile yield and nitrogen partitioning during pyrolysis of coal and biomass fuels. Fuel 2019, 248, 215–220. [Google Scholar] [CrossRef]
- Zubkova, V.; Strojwas, A.; Bielecki, M.; Kieush, L.; Koverya, A. Comparative study of pyrolytic behavior of the biomass wastes originating in the Ukraine and potential application of such biomass. Part 1. Analysis of the course of pyrolysis process and the composition of formed products. Fuel 2019, 254, 115688. [Google Scholar] [CrossRef]
- Johansson, A.C.; Sandström, L.; Öhrman, O.G.W.; Jilvero, H. Co-pyrolysis of woody biomass and plastic waste in both analytical and pilot scale. J. Anal. Appl. Pyrolysis 2018, 134, 102–113. [Google Scholar] [CrossRef]
- Korshunov, A.; Kichatov, B.; Melnikova, K.; Gubernov, V.; Yakovenko, I.; Kiverin, A.; Golubkov, A. Pyrolysis characteristics of biomass torrefied in a quiescent mineral layer. Energy 2019, 187, 116015. [Google Scholar] [CrossRef]
- Mishra, R.K.; Mohanty, K. Pyrolysis kinetics and thermal behavior of waste sawdust biomass using thermogravimetric analysis. Bioresour. Technol. 2018, 251, 63–74. [Google Scholar] [CrossRef]
- Nguyen, T.S.; He, S.; Raman, G.; Seshan, K. Catalytic hydro-pyrolysis of lignocellulosic biomass over dual Na2CO3/Al2O3 and Pt/Al2O3 catalysts using n-butane at ambient pressure. Chem. Eng. J. 2016, 299, 415–419. [Google Scholar] [CrossRef]
- Schneider, J.K.; da Cunha, M.E.; dos Santos, A.L.; Maciel, G.P.S.; Brasil, M.C.; Pinho, A.R.; Mendes, F.L.; Jacques, R.A.; Caramão, E.B. Comprehensive two dimensional gas chromatography with fast-quadrupole mass spectrometry detector analysis of polar compounds extracted from the bio-oil from the pyrolysis of sawdust. J. Chromatogr. A 2014, 1356, 236–240. [Google Scholar] [CrossRef]
- Mishra, R.K.; Mohanty, K. Thermal and catalytic pyrolysis of pine sawdust (Pinus ponderosa) and Gulmohar seed (Delonix regia) towards production of fuel and chemicals. Mater. Sci. Energy Technol. 2019, 2, 139–149. [Google Scholar] [CrossRef]
- He, J.; Strezov, V.; Kumar, R.; Weldekidan, H.; Jahan, S.; Dastjerdi, B.H.; Zhou, X.; Kan, T. Pyrolysis of heavy metal contaminated Avicennia marina biomass from phytoremediation: Characterisation of biomass and pyrolysis products. J. Clean. Prod. 2019, 234, 1235–1245. [Google Scholar] [CrossRef]
- Pighinelli, A.L.M.T.; Boateng, A.A.; Mullen, C.A.; Elkasabi, Y. Evaluation of Brazilian biomasses as feedstocks for fuel production via fast pyrolysis. Energy Sustain. Dev. 2014, 21, 42–50. [Google Scholar] [CrossRef]
- Makkawi, Y.; El Sayed, Y.; Salih, M.; Nancarrow, P.; Banks, S.; Bridgwater, T. Fast pyrolysis of date palm (Phoenix dactylifera) waste in a bubbling fluidized bed reactor. Renew. Energy 2019, 143, 719–730. [Google Scholar] [CrossRef]
- Li, J.; Qiao, Y.; Zong, P.; Qin, S.; Wang, C.; Tian, Y. Fast pyrolysis characteristics of two typical coastal zone biomass fuels by thermal gravimetric analyzer and down tube reactor. Bioresour. Technol. 2019, 283, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Liu, H.; Li, S.; Li, M.; Wang, G.; Man, G.; Yao, H. Catalytic pyrolysis of biomass wastes over Org-CaO/Nano-ZSM-5 to produce aromatics: Influence of catalyst properties. Bioresour. Technol. 2019, 294, 122186. [Google Scholar] [CrossRef]
- Akubo, K.; Nahil, M.A.; Williams, P.T. Pyrolysis-catalytic steam reforming of agricultural biomass wastes and biomass components for production of hydrogen/syngas. J. Energy Inst. 2019, 92, 1987–1996. [Google Scholar] [CrossRef]
- Liu, X.; Luo, Z.; Yu, C.; Xie, G. Conversion mechanism of fuel-N during pyrolysis of biomass wastes. Fuel 2019, 246, 42–50. [Google Scholar] [CrossRef]
- Pelaez-Samaniego, M.R.; Mesa-Pérez, J.; Cortez, L.A.B.; Rocha, J.D.; Sanchez, C.G.; Marín, H. Use of blends of gasoline with biomass pyrolysis-oil derived fractions as fuels in an Otto engine. Energy Sustain. Dev. 2011, 15, 376–381. [Google Scholar] [CrossRef]
- Pehlivan, E.; Özbay, N.; Yargıç, A.S.; Şahin, R.Z. Production and characterization of chars from cherry pulp via pyrolysis. J. Environ. Manage. 2017, 203, 1017–1025. [Google Scholar] [CrossRef]
- Maliutina, K.; Tahmasebi, A.; Yu, J.; Saltykov, S.N. Comparative study on flash pyrolysis characteristics of microalgal and lignocellulosic biomass in entrained-flow reactor. Energy Convers. Manag. 2017, 151, 426–438. [Google Scholar] [CrossRef]
- Ahmad, N.; Abnisa, F.; Wan Daud, W.M.A. Liquefaction of natural rubber to liquid fuels via hydrous pyrolysis. Fuel 2018, 218, 227–235. [Google Scholar] [CrossRef]
- Borel, L.D.M.S.; Lira, T.S.; Ribeiro, J.A.; Ataíde, C.H.; Barrozo, M.A.S. Pyrolysis of brewer’s spent grain: Kinetic study and products identification. Ind. Crops Prod. 2018, 121, 388–395. [Google Scholar] [CrossRef]
- Chang, Z.; Duan, P.; Xu, Y. Catalytic hydropyrolysis of microalgae: Influence of operating variables on the formation and composition of bio-oil. Bioresour. Technol. 2015, 184, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Chernova, N.I.; Kiseleva, S.V.; Larina, O.M.; Sytchev, G.A. Manufacturing gaseous products by pyrolysis of microalgal biomass. Int. J. Hydrogen Energy 2020, 45, 1569–1577. [Google Scholar] [CrossRef]
- Dhyani, V.; Bhaskar, T. A comprehensive review on the pyrolysis of lignocellulosic biomass. Renew. Energy 2018, 129, 695–716. [Google Scholar] [CrossRef]
- Yang, H.; Yan, R.; Chen, H.; Zheng, C.; Ho Lee, D.; Liang, D.T. In-Depth Investigation of Biomass Pyrolysis Based on Three Major Components: Hemicellulose, Cellulose and Lignin. Energy Fuels 2005, 20, 388–393. [Google Scholar] [CrossRef]
- Burhenne, L.; Messmer, J.; Aicher, T.; Laborie, M.P. The effect of the biomass components lignin, cellulose and hemicellulose on TGA and fixed bed pyrolysis. J. Anal. Appl. Pyrolysis 2013, 101, 177–184. [Google Scholar] [CrossRef]
- Zaman, C.Z.; Pal, K.; Yehye, W.A.; Sagadevan, S.; Shah, S.T.; Adebisi, G.A.; Marliana, E.; Rafique, R.F.; Johan, R.B. Pyrolysis: A Sustainable Way to Generate Energy from Waste. Pyrolysis 2017, 1–34. [Google Scholar] [CrossRef] [Green Version]
- Kirubakaran, V.; Sivaramakrishnan, V.; Nalini, R.; Sekar, T.; Premalatha, M.; Subramanian, P. A review on gasification of biomass. Renew. Sustain. Energy Rev. 2009, 13, 179–186. [Google Scholar] [CrossRef]
- Di Blasi, C.; Signorelli, G.; Di Russo, C.; Rea, G. Product Distribution from Pyrolysis of Wood and Agricultural Residues. Ind. Eng. Chem. Res. 1999, 38, 2216–2224. [Google Scholar] [CrossRef]
- Pang, Y.X.; Yan, Y.; Foo, D.C.Y.; Sharmin, N.; Zhao, H.; Lester, E.; Wu, T.; Pang, C.H. The influence of lignocellulose on biomass pyrolysis product distribution and economics via steady state process simulation. J. Anal. Appl. Pyrolysis 2021, 158, 104968. [Google Scholar] [CrossRef]
- Sahoo, K.; Kumar, A.; Chakraborty, J.P. A comparative study on valuable products: Bio-oil, biochar, non-condensable gases from pyrolysis of agricultural residues. J. Mater. Cycles Waste Manag. 2021, 23, 186–204. [Google Scholar] [CrossRef]
- Zhang, Z.; Pang, S. Experimental investigation of tar formation and producer gas composition in biomass steam gasification in a 100 kW dual fluidised bed gasifier. Renew. Energy 2019, 132, 416–424. [Google Scholar] [CrossRef]
- Couhert, C.; Commandre, J.M.; Salvador, S. Is it possible to predict gas yields of any biomass after rapid pyrolysis at high temperature from its composition in cellulose, hemicellulose and lignin? Fuel 2009, 88, 408–417. [Google Scholar] [CrossRef] [Green Version]
- Luo, S.; Xiao, B.; Guo, X.; Hu, Z.; Liu, S.; He, M. Hydrogen-rich gas from catalytic steam gasification of biomass in a fixed bed reactor: Influence of particle size on gasification performance. Int. J. Hydrogen Energy 2009, 34, 1260–1264. [Google Scholar] [CrossRef]
- Mohammed, M.A.A.; Salmiaton, A.; Wan Azlina, W.A.K.G.; Mohammad Amran, M.S.; Fakhru’l-Razi, A. Air gasification of empty fruit bunch for hydrogen-rich gas production in a fluidized-bed reactor. Energy Convers. Manag. 2011, 52, 1555–1561. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Zawawi, N.A.; Kasim, F.H.; Inayat, A.; Khasri, A. Assessing the gasification performance of biomass: A review on biomass gasification process conditions, optimization and economic evaluation. Renew. Sustain. Energy Rev. 2016, 53, 1333–1347. [Google Scholar] [CrossRef]
- Yahaya, A.Z.; Somalu, M.R.; Muchtar, A.; Sulaiman, S.A.; Wan Daud, W.R. Effect of particle size and temperature on gasification performance of coconut and palm kernel shells in downdraft fixed-bed reactor. Energy 2019, 175, 931–940. [Google Scholar] [CrossRef]
- Sibiya, N.T.; Oboirien, B.; Lanzini, A.; Gandiglio, M.; Ferrero, D.; Papurello, D.; Bada, S.O. Effect of different pre-treatment methods on gasification properties of grass biomass. Renew. Energy 2021, 170, 875–883. [Google Scholar] [CrossRef]
- Skoulou, V.; Swiderski, A.; Yang, W.; Zabaniotou, A. Process characteristics and products of olive kernel high temperature steam gasification (HTSG). Bioresour. Technol. 2009, 100, 2444–2451. [Google Scholar] [CrossRef] [PubMed]
- Zeng, B.; Shimizu, N. Hydrogen Generation from Wood Chip and Biochar by Combined Continuous Pyrolysis and Hydrothermal Gasification. Energies 2021, 14, 3793. [Google Scholar] [CrossRef]
- Lv, P.M.; Xiong, Z.H.; Chang, J.; Wu, C.Z.; Chen, Y.; Zhu, J.X. An experimental study on biomass air-steam gasification in a fluidized bed. Bioresour. Technol. 2004, 95, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Ismail, T.M.; Banks, S.W.; Yang, Y.; Yang, H.; Chen, Y.; Bridgwater, A.V.; Ramzy, K.; Abd El-Salam, M. Coal and biomass co-pyrolysis in a fluidized-bed reactor: Numerical assessment of fuel type and blending conditions. Fuel 2020, 275, 118004. [Google Scholar] [CrossRef]
- Chai, Y.; Wang, M.; Gao, N.; Duan, Y.; Li, J. Experimental study on pyrolysis/gasification of biomass and plastics for H2 production under new dual-support catalyst. Chem. Eng. J. 2020, 396, 125260. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Jiao, L.; Yan, B.; Li, Z.; Dong, X.; Chen, G. Microwave pyrolysis of herb residue for syngas production with in-situ tar elimination and nitrous oxides controlling. Fuel Process. Technol. 2021, 221, 106955. [Google Scholar] [CrossRef]
- Li, L.; Liu, G.; Li, Y.; Zhu, Z.; Xu, H.; Chen, J.; Ren, X. Release of Sulfur and Nitrogen during Co-pyrolysis of Coal and Biomass under Inert Atmosphere. ACS Omega 2020, 5, 30001–30010. [Google Scholar] [CrossRef]
- Mlonka-Mędrala, A.; Evangelopoulos, P.; Sieradzka, M.; Zajemska, M.; Magdziarz, A. Pyrolysis of agricultural waste biomass towards production of gas fuel and high-quality char: Experimental and numerical investigations. Fuel 2021, 296, 120611. [Google Scholar] [CrossRef]
- Hwang, I.H.; Kobayashi, J.; Kawamoto, K. Characterization of products obtained from pyrolysis and steam gasification of wood waste, RDF, and RPF. Waste Manag. 2014, 34, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Franco, C.; Pinto, F.; Gulyurtlu, I.; Cabrita, I. The study of reactions influencing the biomass steam gasification process. Fuel 2003, 82, 835–842. [Google Scholar] [CrossRef]
- Lv, P.; Yuan, Z.; Ma, L.; Wu, C.; Chen, Y.; Zhu, J. Hydrogen-rich gas production from biomass air and oxygen/steam gasification in a downdraft gasifier. Renew. Energy 2007, 32, 2173–2185. [Google Scholar] [CrossRef]
- Chang, A.C.C.; Chang, H.F.; Lin, F.J.; Lin, K.H.; Chen, C.H. Biomass gasification for hydrogen production. Int. J. Hydrogen Energy 2011, 36, 14252–14260. [Google Scholar] [CrossRef]
- Parthasarathy, P.; Narayanan, K.S. Hydrogen production from steam gasification of biomass: Influence of process parameters on hydrogen yield—A review. Renew. Energy 2014, 66, 570–579. [Google Scholar] [CrossRef]
- Cerone, N.; Zimbardi, F. Effects of Oxygen and Steam Equivalence Ratios on Updraft Gasification of Biomass. Energies 2021, 14, 2675. [Google Scholar] [CrossRef]
- Wongsiriamnuay, T.; Kannang, N.; Tippayawong, N. Effect of operating conditions on catalytic gasification of bamboo in a fluidized bed. Int. J. Chem. Eng. 2013, 297941. [Google Scholar] [CrossRef] [Green Version]
- Elshokary, S.; Farag, S.; Sayed Mohamed Abu-Elyazeed, O.; Hurisso, B.; Ismai, M. Downdraft gasifier design calculation for biomass gasification using exhaust gas as a gasification agent. Mater. Today Proc. 2021, 1–6. [Google Scholar] [CrossRef]
- Zhang, H.; Xiao, R.; Wang, D.; He, G.; Shao, S.; Zhang, J.; Zhong, Z. Biomass fast pyrolysis in a fluidized bed reactor under N2, CO2, CO, CH4 and H2 atmospheres. Bioresour. Technol. 2011, 102, 4258–4264. [Google Scholar] [CrossRef] [PubMed]
- Habibollahzade, A.; Ahmadi, P.; Rosen, M.A. Biomass gasification using various gasification agents: Optimum feedstock selection, detailed numerical analyses and tri-objective grey wolf optimization. J. Clean. Prod. 2021, 284, 124718. [Google Scholar] [CrossRef]
- Gayubo, A.G.; Aguayo, A.T.; Atutxa, A.; Prieto, R.; Bilbao, J. Deactivation of a HZSM-5 Zeolite Catalyst in the Transformation of the Aqueous Fraction of Biomass Pyrolysis Oil into Hydrocarbons. Energy Fuels 2004, 18, 1640–1647. [Google Scholar] [CrossRef]
- Samolada, M.C.; Papafotica, A.; Vasalos, I.A. Catalyst Evaluation for Catalytic Biomass Pyrolysis. Energy Fuels 2000, 14, 1161–1167. [Google Scholar] [CrossRef]
- Lin, Y.; Zhang, C.; Zhang, M.; Zhang, J. Deoxygenation of Bio-oil during Pyrolysis of Biomass in the Presence of CaO in a Fluidized-Bed Reactor. Energy Fuels 2010, 24, 5686–5695. [Google Scholar] [CrossRef]
- Florentino-Madiedo, L.; Díaz-Faes, E.; García, R.; Barriocanal, C. Influence of binder type on greenhouse gases and PAHs from the pyrolysis of biomass briquettes. Fuel Process. Technol. 2018, 171, 330–338. [Google Scholar] [CrossRef] [Green Version]
- Zeng, K.; Li, R.; Minh, D.P.; Weiss-Hortala, E.; Nzihou, A.; He, X.; Flamant, G. Solar pyrolysis of heavy metal contaminated biomass for gas fuel production. Energy 2019, 187, 116016. [Google Scholar] [CrossRef]
- Sutton, D.; Kelleher, B.; Ross, J.R.H. Review of literature on catalysts for biomass gasification. Fuel Process. Technol. 2001, 73, 155–173. [Google Scholar] [CrossRef]
- Matsuoka, K.; Shimbori, T.; Kuramoto, K.; Hatano, H.; Suzuki, Y. Steam Reforming of Woody Biomass in a Fluidized Bed of Iron Oxide-Impregnated Porous Alumina. Energy Fuels 2006, 20, 2727–2731. [Google Scholar] [CrossRef]
- Zhang, R.; Brown, R.C.; Suby, A.; Cummer, K. Catalytic destruction of tar in biomass derived producer gas. Energy Convers. Manag. 2004, 45, 995–1014. [Google Scholar] [CrossRef]
- Hepola, J.; McCarty, J.; Krishnan, G.; Wong, V. Elucidation of behavior of sulfur on nickel-based hot gas cleaning catalysts. Appl. Catal. B Environ. 1999, 20, 191–203. [Google Scholar] [CrossRef]
- Shen, Y.; Yoshikawa, K. Recent progresses in catalytic tar elimination during biomass gasification or pyrolysis—A review. Renew. Sustain. Energy Rev. 2013, 21, 371–392. [Google Scholar] [CrossRef]
- Rauch, R.; Hrbek, J.; Hofbauer, H. Biomass gasification for synthesis gas production and applications of the syngas. Wiley Interdiscip. Rev. Energy Environ. 2014, 3, 343–362. [Google Scholar] [CrossRef]
- Li, J.; Xu, K.; Yao, X.; Chen, S. Prediction and optimization of syngas production from steam gasification: Numerical study of operating conditions and biomass composition. Energy Convers. Manag. 2021, 236, 114077. [Google Scholar] [CrossRef]
- Wei, L.; Xu, S.; Zhang, L.; Liu, C.; Zhu, H.; Liu, S. Steam gasification of biomass for hydrogen-rich gas in a free-fall reactor. Int. J. Hydrogen Energy 2007, 32, 24–31. [Google Scholar] [CrossRef]
- Mazaheri, N.; Akbarzadeh, A.H.; Madadian, E.; Lefsrud, M. Systematic review of research guidelines for numerical simulation of biomass gasification for bioenergy production. Energy Convers. Manag. 2019, 183, 671–688. [Google Scholar] [CrossRef]
- Ahmed, T.Y.; Ahmad, M.M.; Yusup, S.; Inayat, A.; Khan, Z. Mathematical and computational approaches for design of biomass gasification for hydrogen production: A review. Renew. Sustain. Energy Rev. 2012, 16, 2304–2315. [Google Scholar] [CrossRef]
- Ramos, A.; Monteiro, E.; Rouboa, A. Numerical approaches and comprehensive models for gasification process: A review. Renew. Sustain. Energy Rev. 2019, 110, 188–206. [Google Scholar] [CrossRef]
- Mahishi, M.R.; Goswami, D.Y. Thermodynamic optimization of biomass gasifier for hydrogen production. Int. J. Hydrogen Energy 2007, 32, 3831–3840. [Google Scholar] [CrossRef]
- Li, X.T.; Grace, J.R.; Lim, C.J.; Watkinson, A.P.; Chen, H.P.; Kim, J.R. Biomass gasification in a circulating fluidized bed. Biomass Bioenergy 2004, 26, 171–193. [Google Scholar] [CrossRef]
- Karmakar, M.K.; Datta, A.B. Generation of hydrogen rich gas through fluidized bed gasification of biomass. Bioresour. Technol. 2011, 102, 1907–1913. [Google Scholar] [CrossRef] [PubMed]


| Type of Conversion | Benefits | Drawbacks |
|---|---|---|
| Thermochemical Conversion | ||
| Combustion | The scale of setups varies from small to industrial ones in the range of 50–3000 MW. Conversion efficiency is between 20% and 40%. Biomass can be co-fired with coal. | Biomass moisture content should be less than 50%. The process develops at a high temperature (800–1000 °C). |
| Gasification | The produced gas with a higher heating value of 4–6 MJ/m3 can be burned directly or used (after cleaning) as a fuel for gas engines and turbines. Syngas production from biomass makes it possible to obtain methanol and hydrogen, each of which can be used as a fuel for transportation. | The gas with a higher heating value of 4–6 MJ/m3 is not appropriate for pipeline transportation due to its low energy density. The production of methanol with a higher heating value of 9–11 MJ/m3 requires gasification involving oxygen. |
| Pyrolysis | Bio-oil can be used in engines and turbines; it also serves as feedstock for oil refineries. | Low heat stability and high corrosiveness. |
| Process | Conditions | Result | |||
|---|---|---|---|---|---|
| Pyrolysis | Fast | Moderate temperature (600–800 °C), short residence time particularly vapor (from 10 to 200 °C/s) | Liquid | Char | Gas |
| 75% | 12% | 13% | |||
| Slow | Low temperature (300–500 °C), very long residence time (under 1 °C/s) | 30% | 35% | 35% | |
| Gasification | High temperature (650–1200 °C), long residence times (from 1 to 100 °C/s) | 5% | 10% | 85% | |
| Plant Name | Location | Units | Capacity |
|---|---|---|---|
| Pyrolysis | |||
| Red arrow, WI | Canada | Circulating fluidized bed | 1700 kg/h |
| Dyna Motive | Canada | Bubbling fluidized bed | 400 kg/h |
| Bio-alternative | USA | Fixed bed | 2000 kg/h |
| Battelle | USA | Catalytic pyrolysis technology | 1000 kg/h |
| Empyro | Netherlands | Flash pyrolysis | 5000 kg/h |
| Bioliq | Germany | Fast pyrolysis | 500 kg/h |
| BEST Energy | Australia | Bubbling fluidized bed | 300 kg/h |
| Fortum | Finland | - | 350 kg/h |
| Unión Fenosa | Spain | Bubbling fluidized bed | 200 kg/h |
| IRR manufacturing | South Africa | - | 1000 kg/h |
| Gasification | |||
| Great plains synfuels plant | USA | Fixed bed dry bottom | 16,000 t/day |
| Energos Gasification Plant | Norway | two-stage thermal treatment process | 78,000 t/year |
| Red Rock Bio | USA | TCG Global steam reforming | 136,000 tons/year |
| Shaanxi Weihe Fertilizer Co | China | General Electric | 1500 t/day |
| Yunnan Yuntianhua Group Tian’an Chemical Co., Ltd. | China | - | 2700 t/day |
| Component | Country | Ultimate Analysis (wt %) | Proximate Analysis (wt %) | Ref. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | H | O | N | S | Moisture | Volatile Matter | Fixed Carbon | Ash | Heat of Combustion (MJ/kg) | |||
| Woody biomass | ||||||||||||
| Beech wood | Germany | 44.1 | 6.3 | 49.4 | 0.2 | 0 | 4.7 | 87.6 | 8 | 0.8 | 19.5 | [36] |
| Wood pellet | UK | 52.3 | 6.8 | 40.7 | 0.16 | - | 6.7 | 84.3 | 15.7 | 0.8 | 20.8 | [37] |
| Soft wood | Ukraine | 45.34 ± 0.13 | 5.86 ± 0.04 | 42.45 ± 0.04 | 0.58 ± 0.11 | 0.17 ± 0.07 | 5.15 | - | - | 5.60 ± 0.38 | 18.23 ± 0.13 | [38] |
| Woody biomass | Sweden | 51.3 | 6.2 | 42 | 0.1 | 0.021 | 4.3 | 83.8 | - | 0.3 | 19.36 | [39] |
| Pine | Russia | 47.88 | 6.34 | 45.69 | 0.09 | 0 | - | 72.5 | 27.0 | 0.5 | - | [40] |
| Pine sawdust | India | 50.3 | 6 | 42.99 | 0.69 | - | 6.09 ± 0.3 | 78.03 ± 0.2 | 12.16 ± 0.1 | 2.07 ± 0.03 | 18.44 ± 09 | [41] |
| Sal sawdust | India | 49.83 | 6.01 | 43.56 | 0.58 | - | 8.88 ± 0.2 | 76.03 ± 0.1 | 14.09 ± 0.2 | 1.14 ± 0.01 | 18.20 ± 09 | [41] |
| Pine wood chips | Canada | 48.3 | 5.8 | 45.4 | 0.5 | - | 4.5 | 78.4 | - | 2.6 | 16.1 | [42] |
| Sawdust | Ecuador | 46.1 | 6.3 | 46.7 | 0.3 | - | 7.4 | - | - | 0.6 | - | [43] |
| Pine sawdust | India | 53.5 | 6.93 | 32.55 | 3.33 | 0.66 | 7.85 ± 0.05 | 77.27 ± 0.65 | 12.20 ± 0.15 | 2.78 ± 0.12 | 18.55 ± 0.43 | [44] |
| Root of mango tree | Australia | 45.56 | 6.44 | 47.24 | 0.56 | 0.20 | 5.73 | 67.87 | 22.49 | 3.91 | 18.52 | [45] |
| Eucalyptus urophylla | Brazil | 45.03 | 4.78 | 38.46 | 0.11 | - | 11.37 | 75.34 | 13.04 | 0.27 | 17.16 | [46] |
| Herbaceous and agricultural biomass | ||||||||||||
| Miscanthus | Australia | 50.73 | 7.08 | 41.95 | 0.14 | 0.10 | 10.67 | 65.65 | 18.34 | 5.34 | 17.00 | [45] |
| Palm empty fruit bunches | UAE | 44.7 | 5.97 | 49.05 | 0.74 | 0.18 | 8.73 | 67.51 | 17.47 | 6.28 | 17.2 | [47] |
| Palm leaves | UAE | 40.76 | 5.55 | 52.14 | 1.32 | 0.24 | 12.03 | 58.17 | 15.41 | 14.4 | 18.9 | [47] |
| Palm leave stems | UAE | 42.67 | 5.83 | 50.78 | 0.58 | 0.15 | 11.65 | 68.84 | 10 | 9.51 | 16.5 | [47] |
| Corn stalks | Ukraine | 36.38 ± 1.36 | 5.40 ± 0.13 | 44.08 ± 0.38 | 1.68 ± 0.01 | 0.16 ± 0.02 | 8.13 | - | - | 12.30 ± 0.87 | 14.24 ± 0.46 | [38] |
| Jerusalem artichoke stalks | China | 45.36 | 6.11 | 47.26 | 0.75 | 0.52 | 15.76 | 67.4 | 13.5 | 3.34 | 15.69 | [48] |
| Cane | China | 42.78 | 5.17 | 50.51 | 1.33 | 0.21 | 5.89 | 72.12 | 13.52 | 8.47 | 16.16 | [48] |
| Gulmohar seed | India | 51.3 | 6 | 40.56 | 2.58 | - | 7.09 ± 0.05 | 75.56 ± 0.5 | 15.80 ± 0.2 | 2.07 ± 0.12 | 19.65 ± 0.11 | [44] |
| Corncob | China | 46.6 | 5.8 | 47.0 | 0.4 | 0.2 | - | 86.9 | 11.8 | 1.3 | - | [49] |
| Corn cob | India | 44.2 | 5.9 | 44.2 | 0.54 | 0.08 | 10.2 | 80 | 4.2 | 5.7 | 15.5 | [41] |
| Palm kernel shell | UK | 50.11 | 6.24 | 42.16 | 1.50 | 0 | 6.70 | 67.52 | 22.13 | 3.65 | - | [50] |
| Olive waste | UK | 52.8 | 6.5 | 39.1 | 1.6 | - | 5.9 | 80.1 | 19.9 | 7.6 | 20.1 | [37] |
| Palm kernel cake | China | 49.04 | 5.93 | 34.10 | 2.46 | 0.29 | 2.88 | 75.83 | 15.99 | 5.30 | - | [51] |
| Jatropha seeds cake | China | 45.3 | 6.2 | 43.8 | 4.5 | 0.2 | - | 73.5 | 18.2 | 7.3 | - | [49] |
| Bagasse | China | 46.4 | 6.7 | 45.8 | 0.7 | 0.4 | - | 87.4 | 9.7 | 2.9 | - | [49] |
| Sugarcane | India | 43.2 | 6.2 | 43.2 | 0.4 | 0.8 | 10 | 76 | 9.6 | 4.4 | 17.2 | [41] |
| Sugarcane | UK | 44.34 | 5.92 | 49.17 | 0.57 | 0 | 5.33 | 83.39 | 7.79 | 3.49 | - | [50] |
| Sugarcane | Brazil | 43.79 | 5.16 | 38.90 | 0.29 | - | 7.32 | 74.86 | 13.27 | 4.55 | 17.81 | [46] |
| Sugarcane trash | Brazil | 44.7 | 5.8 | 48.97 | 0.45 | 0.08 | 9.92 | 81.55 | 6.90 | 11.57 | 17.74 (16.50) | [52] |
| Cherry pulp | Turkey | 50.80 | 6.79 | 39.66 | 2.67 | - | 6.42 | 72.02 | 19.70 | 1.86 | 19.82 | [53] |
| Straw | ||||||||||||
| Wheat straw | Ukraine | 39.90 ± 0.15 | 5.75 ± 0.02 | 41.97 ± 0.07 | 0.65 ± 0.08 | 0.13 ± 0.05 | 6.84 | - | - | 11.59 ± 0.76 | 16.12 ± 0.19 | [37] |
| Wheat straw | UK | 40.58 | 4.84 | 53.84 | 0.74 | 0 | 5.19 | 64.24 | 15.60 | 14.97 | - | [50] |
| Wheat straw | China | 45.94 | 5.83 | 39.08 | 0.56 | 0.45 | 2.50 | 72.36 | 18.00 | 5.64 | - | [51] |
| Rice straw | China | 42.66 | 5.68 | 37.37 | 1.03 | 0.44 | 1.51 | 69.09 | 18.09 | 11.31 | - | [51] |
| Cotton stalk | India | 46.8 | 6.4 | 46.8 | 0.3 | 0.2 | 8.9 | 71 | 16.6 | 3.5 | 19.2 | [41] |
| Cotton stalk | UK | 43.10 | 6.24 | 49.07 | 1.59 | 0 | 7.33 | 69.54 | 19.47 | 3.67 | - | [50] |
| Rice Husk | UK | 37.60 | 5.26 | 55.45 | 1.69 | 0 | 8.02 | 61.43 | 12.53 | 18.02 | - | [50] |
| Nut husk and shells | ||||||||||||
| Sunflower husks | Ukraine | 45.82 ± 0.08 | 6.32 ± 0.02 | 38.31 ± 0.08 | 2.61 ± 0.05 | 0.14 ± 0.02 | 6.1 | - | - | 6.81 ± 0.51 | 19.31 ± 0.13 | [38] |
| Areca nut husk | India | 48.8 | 5.79 | 43.45 | 1.95 | 0.1 | 7.43 ± 0.1 | 74.05 ± 0.2 | 15.55 ± 0.3 | 2.48 ± 0.05 | 18.21 ± 09 | [41] |
| Peanut shell | China | 49.7 | 5.8 | 43.7 | 0.6 | 0.1 | - | 84.1 | 14.5 | 1.4 | - | [49] |
| Palm kernel shell | Malaysia | 48.82 | 5.68 | 45.08 | 0.42 | - | 13.65 | 75.32 | 20.81 | 3.87 | 14.88 (14.75) | [54] |
| Walnut shells | Ukraine | 43.41 ± 0.17 | 5.66 ± 0.06 | 48.44 ± 0.08 | 1.98 ± 0.06 | 0.11 ± 0.03 | 4.1 | - | - | 0.41 ± 0.11 | 16.79 ± 0.08 | [38] |
| Coconut shell | UK | 48.32 | 5.26 | 46.14 | 0.29 | 0 | 7.16 | 68.58 | 22.00 | 2.26 | - | [50] |
| Other | ||||||||||||
| Cellulose | UK | 41.61 | 5.63 | 52.64 | 0.11 | 0 | 4.74 | 84.16 | 9.85 | 1.25 | - | [50] |
| Natural rubber | Malaysia | 83.63 | 11.97 | 2.71 | 1.58 | 0.12 | 1.71 | 89.98 | 4.71 | 3.60 | 45 | [55] |
| Spent coffee grounds | China | 55.98 | 6.73 | 31.07 | 2.0 | 0.31 | 2.66 | 80.44 | 15.65 | 1.25 | - | [51] |
| Brewer’s spent grain | Brazil | 42.2 | 7.2 | 37.6 | 3.6 | 1.1 | 3.97 | 83.3 | 9.51 | 3.22 | 21.6 | [56] |
| Microalgae | China | 52.07 | 7.15 | 21.65 | 8.57 | 0.62 | - | 72.37 | 22.16 | 5.46 | 24.19 | [57] |
| Microalgae | China | 49.6 | 7.0 | 25.4 | 8.2 | 0.5 | 10 | 81 | 16 | 9 | - | [57] |
| Microalgae | Russia | 61.3 | 6.4 | 22.5 | 8.8 | 1.1 | 3.0 | - | - | 5.1 | 25.04 | [58] |
| Biomass Properties | Factors | Favorable Conditions for Pyrolysis and Gasification |
|---|---|---|
| Moisture content | There are two kinds of biomass moisture: inherent (the content of moisture in biomass not affected by the weather conditions), and external (the content of moisture in biomass considering the weather conditions). | A high content of moisture enhances pyrolysis and gasification. |
| Heating value | There are higher and lower heating values. The higher heating value is the maximum amount of energy potentially derived from this biomass source. It includes the content of energy released during the fuel combustion in the air, as well as latent heat in the water steam. The lower heating value is the minimum amount of energy released from biomass conversion. | The higher the heating value, the more rapid is pyrolysis and gasification. |
| Proportion of bound carbon and volatiles | The content of volatiles in the solid fuel, part of the fuel that is released as gas when it is heated. The content of bound carbon is a mass remaining after the release of volatiles, excluding the content of ash and moisture. The content of volatiles and bound carbon account for the rate of ignition and then gasification or oxidation. The elemental analysis, including the values of O, H, C, N, and S, indicates that a higher percentage of oxygen as compared with carbon reduces the heat of combustion of the fuel due to lower energy. | A high content of volatiles and carbon combined with a low content of oxygen. |
| Content of ash | The chemical degradation of a biomass-derived fuel as a result of thermochemical or biochemical processes produces a solid residue which is actually ash. The content of ash in biomass affects both the cost of processing and the total cost of biomass energy conversion. Depending on the ash content, the available energy of the fuel proportionally decreases. After combustion, ash can form slag, a liquid phase (at a high temperature), which impairs the performance of the facility and increases operating costs. | Low ash content. |
| Content of alkali metals | Alkali metals (Na, K, Mg, P, and Ca) in biomass lead to the formation of a sticky mobile liquid phase (slag) which may obstruct the flue gas path. | Lower content of alkali metals. |
| Cellulose/lignin ratio | Cellulose decomposes at lower temperatures than lignin does. Therefore, the total conversion of the carbon-containing plant matter in the form of cellulose is higher than that of plants with a higher proportion of lignin. | High content of cellulose and low content of lignin. |
| Feedstock | Lignin (%) | Cellulose (%) | Hemicellulose (%) | Ref. |
|---|---|---|---|---|
| Rice straw | 30 | 25 | 12 | [6] |
| Bagasse | 38 | 39 | 20 | [6] |
| Oat straw | 31–37 | 27–39 | 16–19 | [59] |
| Beech wood | 21.9 | 45.8 | 31.8 | [59] |
| Oak | 43.2 | 21.9 | 35.4 | [59] |
| Sawdust | 22.16 | 32.63 | 37.23 | [59] |
| Leaves | 4 | 15.5 | 80.5 | [59] |
| Straw | 28.90 | 36.70 | 34.4 | [59] |
| Wood | 25–30 | 35–50 | 20–30 | [62] |
| Wheat straw | 15–20 | 33–40 | 20–25 | [62] |
| Rice husk | 14.3 | 31.3 | 24.3 | [63] |
| Coconut shell | 28.7 | 36.3 | 25.1 | [63] |
| Corn stalks | 17.5 | 42.7 | 23.6 | [63] |
| Millet husk | 14 | 33.3 | 26.9 | [63] |
| Olive husk | 28.0 | 18.5 | 18.5 | [64] |
| Surface Structure | CO2, % | CO, % | H2, % | CH4, % |
|---|---|---|---|---|
| Level layer | 14.56 | 12.38 | 0.46 | 1.74 |
| Perforated area | 15.40 | 12.94 | 0.50 | 1.97 |
 | ||||
| Segments | 14.76 | 12.52 | 0.41 | 1.52 |
 | ||||
| Channels | 15.03 | 12.78 | 0.47 | 1.54 |
 |
| Type of Biomass | Particle Size | Biomass Structure | Catalyst | Pyrolysis/Gasification Air | Gas Concentration | Temperature | Ref. |
|---|---|---|---|---|---|---|---|
| Corn straw, biomass | 125–250 μm | Level layer | Without catalysts | N2 | SO2: 1.25–1.75 mg/g; CH4: 2–44 mg/g; CO: 12–85 mg/g | 300–900 °C | [80] |
| Biomass | - | Level layer | - | O2 (entrained flow) | H2: 15–20%; CO: 40–60%; CO2: 10–15%; CH4: 0–1%; N2: 0–1% | 1000 °C | [102] |
| - | Level layer | - | O2 (fluidized bed) | H2: 20–30%; CO: 20–30%; CO2: 25–40%CH4: 5–10%; N2: 0–1% | |||
| - | Level layer | - | Steam | H2: 30–45%; CO: 20–25%; CO2: 20–25%; CH4: 6–12%; N2: 0–1% | |||
| Herb residue | 20–40 μm | Level layer | Ni, Fe | Microwave pyrolysis in argon | H2: 30–37%; CO: 45–55%; CO2: 10–15%; CH4: 5–8% | 500–800 °C | [79] |
| TG in argon | H2: 15–27%; CO: 30–60%; CO2: 8–25%; CH4: 13–18% | ||||||
| Willow wood | 500–1000 μm | Pressurized level layer (pellets) | Cu Ni | Argon | H2: 0.5–12 mol/kg; CO: 1.7–13 mol/kg;CO2: 0.45–1.2 mol/kg; CH4: 0.2–1.75 mol/kg | 600–1600 °C | [96] |
| Bamboo | 100–250 μm | Level layer | Dolomite | Air | H2: 6.6–8.16%; CO: 23.5–30.6%; CO2: 59–63%; CH4: 4–5% | 400–500 °C | [88] |
| Agricultural wastes | 210–250 μm | Level layer | Without catalysts | Air | H2: 10–15%; CO: 20–28%; CO2: 40–50%; CH4: 8–10% | 700 °C | [103] |
| Corn stover | pellets of 10–15 mm long and 6 mm in diameter | Pressurized level layer (pellets) | Without catalysts | Water steam | H2: 23–26%; CO: 28–29%; CO2: 26–30%; CH4: 10–11%; C2H4: 7–8%; C2H6: 0.7–1.4% | 700–800 °C | [67] |
| Rice husk | H2: 22–28%; CO: 36–37%; CO2: 18–23%; CH4: 11–12%; C2H4: 4–5%; C2H6: 0.5–1% | ||||||
| Pine | H2: 25–31%; CO: 35–36%; CO2: 16–22%; CH4: 12–13%; C2H4: 3–4%; C2H6: 0.6–1% | ||||||
| Wood pellet | 5–20 mm size | Pressurized level layer (pellets) | Without catalysts | Air | H2: 10–12%; CO: 9–13%; CO2: 4–5%; CH4: 1–2% | 900 °C | [89] |
| 20–50 mm | Pressurized level layer (pellets) | CO2 | H2: 16–19%; CO: 14–19%; CO2: 8–10%; CH4: 2–4%. | ||||
| Corncob | 1000–2000 μm | Level layer | Without catalysts | N2 | H2: 0%; CO: 6.1%; CO2: 8.6%; CH4: 0.7% | 550 °C | [90] |
| CO2 | H2: 0.3%; CO: 6.1%; CO2: 7.7% CH4: 0.8% | ||||||
| CO | H2: 0%; CO: 5.8%; CO2: 14.3% CH4: 0.9% | ||||||
| CH4 | H2: 0.7%; CO: 4%; CO2: 6%; CH4: 0.3% | ||||||
| H2 | H2: 0%; CO: 4.2%; CO2: 8.5%; CH4: 0.5% | ||||||
| Biomass | - | - | - | Air | H2: 19.20%; CO: 18.30%; CO2: 11.09%; CH4: 0.89%; N2: 39.75%; H2O: 10.77% | 800 °C | [91] |
| - | - | - | O2 | H2: 33.31%; CO: 32.63%; CO2: 15.76%; CH4: 2.68%; N2: 0.74%; H2O: 14.87% | |||
| - | - | - | CO2 | H2: 23.51%; CO: 43.79%; CO2: 21.83%; CH4: 0.03%; N2: 0%; H2O: 10.84% | |||
| - | - | - | Steam | H2: 39.13%; CO: 18.45%; CO2: 13.08%; CH4: 3.70%; N2: 0%; H2O: 25.65% | |||
| Pine sawdust | 0.30–0.45 mm | Level layer | Limestone | Steam | H2: 35%; CO: 36%; CO2: 17%; CH4: 6%; C2H4: 4% | 800 °C | [104] |
| Olivine | H2: 35%; CO: 34%; CO2: 22%; CH4: 4%; C2H4: 2% | ||||||
| Dolomite | H2: 42%; CO: 30%; CO2: 16%; CH4: 10%; C2H4: 1% | ||||||
| Empty fruit bunch | 300–1000 μm | Level layer | Without catalysts | Air | H2: 10.27–38.02%; CO: 21.87–36.36%; CO2: 10–65%; CH4: 5.84–14.72% | 700–1000 °C | [70] |
| Pine sawdust | 75–1200 μm | Level layer | Dolomite | Steam | H2: 40–51.2%; CO: 15–22.4%; CO2: 12–40%; CH4: 2–5% | 900 °C | [69] |
| Coconut shell | 1–11 mm | Level layer | Without catalysts | Air | H2: 8.2–14.6%; CO: 13.0–17.4%; CO2: 14.7–16.7%; CH4: 2.82–4.23% | 700–900 °C | [72] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glushkov, D.; Nyashina, G.; Shvets, A.; Pereira, A.; Ramanathan, A. Current Status of the Pyrolysis and Gasification Mechanism of Biomass. Energies 2021, 14, 7541. https://doi.org/10.3390/en14227541
Glushkov D, Nyashina G, Shvets A, Pereira A, Ramanathan A. Current Status of the Pyrolysis and Gasification Mechanism of Biomass. Energies. 2021; 14(22):7541. https://doi.org/10.3390/en14227541
Chicago/Turabian StyleGlushkov, Dmitrii, Galina Nyashina, Anatolii Shvets, Amaro Pereira, and Anand Ramanathan. 2021. "Current Status of the Pyrolysis and Gasification Mechanism of Biomass" Energies 14, no. 22: 7541. https://doi.org/10.3390/en14227541
APA StyleGlushkov, D., Nyashina, G., Shvets, A., Pereira, A., & Ramanathan, A. (2021). Current Status of the Pyrolysis and Gasification Mechanism of Biomass. Energies, 14(22), 7541. https://doi.org/10.3390/en14227541








