Production and Evaluation of Fractionated Tamarind Seed Oil Methyl Esters as a New Source of Biodiesel
Abstract
1. Introduction
2. Materials and Methods
2.1. Materilas
2.2. Extraction of Oil
2.3. High Vacuum Fractionation of Tamarind Seed Oil
2.4. Transesterification
2.5. Determination of Fuel Properties
3. Results and Discussion
3.1. Effect of Catalysts on Biodiesel Yield (%)
3.2. Gas Chromatographic-Mass Spectroscopy (GC-MS) Analysis
3.3. Assessment of Fuel Quality Parameters
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Jaeger, W.K.; Egelkraut, T.M. Biofuel economics in a setting of multiple objectives and unintended consequences. Renew. Sustain. Energ. Rev. 2011, 15, 4320–4333. [Google Scholar] [CrossRef]
- Ismail, S.A.A.; Ali, R.F.M. Physico-chemical properties of biodiesel manufactured from waste frying oil using domestic adsorbents. Sci. Technol. Adv. Mater. 2015, 16, 034602. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G. “Designer” biodiesel: Optimizing fatty ester composition to improve fuel properties. Energy Fuels 2008, 22, 1358–1364. [Google Scholar] [CrossRef]
- Ge, J.G.; Kim, H.Y.; Yoon, S.K.; Choi, N.J. Optimization of palm oil biodiesel blends and engine operating parameters to improve performance and PM morphology in a common rail direct injection diesel engine. Fuel 2020, 260, 116326. [Google Scholar] [CrossRef]
- Mumtaz, M.W.; Adnan, A.; Anwar, F.; Mukhtar, H.; Raza, M.A.; Ahmad, F.; Rashid, U. Response surface methodology: An emphatic tool for optimized biodiesel production using rice bran and sunflower oils. Energies 2012, 5, 3307–3328. [Google Scholar] [CrossRef]
- Ge, J.C.; Kim, H.Y.; Yoon, S.K.; Choi, N.J. Reducing volatile organic compound emissions from diesel engines using canola oil biodiesel fuel and blends. Fuel 2018, 218, 266–274. [Google Scholar] [CrossRef]
- Moser, B.R. Biodiesel production, properties, and feedstocks. In Vitro Cell. Dev. Biol.–Plant 2009, 45, 229–266. [Google Scholar] [CrossRef]
- Haas, M.J.; McAloon, A.J.; Yee, W.C.; Foglia, T.A. A process model to estimate biodiesel production costs. Bioresour. Technol. 2006, 97, 671–678. [Google Scholar] [CrossRef]
- Zhang, Y.; Dubé, M.A.; McLean, D.D.; Kates, M. Biodiesel production from waste cooking oil: 2. Economic assessment and sensitivity analysis. Bioresour. Technol. 2003, 90, 229–240. [Google Scholar] [CrossRef]
- Canakci, M.; Sanli, H. Biodiesel production from various feedstocks and their effects on the fuel properties. J. Ind. Microbiol. Biotechnol. 2008, 35, 431–441. [Google Scholar] [CrossRef]
- Kansedo, J.; Lee, K.T.; Bhatia, S. Biodiesel production from palm oil via heterogeneous transesterification. Biomass Bioenerg. 2009, 33, 271–276. [Google Scholar] [CrossRef]
- Ahmad Farabi, M.S.; Ibrahim, M.L.; Rashid, U.; Taufiq-Yap, Y.H. Esterification of palm fatty acid distillate using sulfonated carbon-based catalyst derived from palm kernel shell and bamboo. Energ. Conver. Manag. 2019, 181, 562–570. [Google Scholar] [CrossRef]
- Tiwari, A.K.; Kumar, A.; Raheman, H. Biodiesel production from jatropha oil (Jatropha curcas) with high free fatty acids: An optimized process. Biomass Bioenergy 2007, 31, 569–575. [Google Scholar] [CrossRef]
- Juan, J.C.; Kartika, D.A.; Wu, T.Y.; Hin, T.-Y.Y. Biodiesel production from jatropha oil by catalytic and non-catalytic approaches: An overview. Bioresour. Technol. 2011, 102, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Meher, L.C.; Naik, S.N.; Naik, M.K.; Dalai, A.K. Biodiesel production using Karanja (Pongamia pinnata) and Jatropha (Jatropha curcas) seed oil. In Handbook of Plant-Based Biofuels; CRC Press: Boca Raton, FL, USA, 2008; pp. 255–266. [Google Scholar]
- Umaru, M.; Mohammed, I.A.; Sadiq, M.M.; Aliyu, A.M.; Suleiman, B.; Segun, T. Production and characterization of biodiesel from Nigerian mango seed oil. In World Congress on Engineering, WCE 2014; Ao, S.I., Gelman, L., Hukins, D., Hunter, A., Korsunsky, A.M., Eds.; Newswood Limited: Hong Kong, China, 2014; Volume 1, pp. 645–649. [Google Scholar]
- Dunn, R.O. Cold flow properties of biodiesel: A guide to getting an accurate analysis. Biofuels 2015, 6, 115–128. [Google Scholar] [CrossRef]
- Bello, E.I.; Adekanbi, I.T.; Akinbode, F.O. Production and characterization of coconut (Cocos nucifera) oil and its methyl ester. Euro. J. Eng. Technol. 2015, 3, 25–35. [Google Scholar]
- Keera, S.; El Sabagh, S.; Taman, A. Castor oil biodiesel production and optimization. Egypt. J. Petrol. 2018, 27, 979–984. [Google Scholar] [CrossRef]
- Shah, S.N.; Sharma, B.K.; Moser, B.R.; Erhan, S.Z. Preparation and evaluation of jojoba oil methyl esters as biodiesel and as a blend component in ultra-low sulfur diesel fuel. Bioenergy Res. 2010, 3, 214–223. [Google Scholar] [CrossRef][Green Version]
- Banu, H.D.; Shallangwa, T.B.; Joseph, I.; Magu, T.O.; Louis, H.; Ahmed, S. Biodiesel production from neem seed (Azadirachta indica) oil using calcium oxide as heterogeneous catalyst. J. Phys. Chem. Biophys. 2018, 8, 2. [Google Scholar] [CrossRef]
- Ali, O.M.; Mamat, R.; Abdullah, N.R.; Abdullah, A.A.; Khoerunnisa, F.; Sardjono, R.E. Effects of Different Chemical Additives on Biodiesel Fuel Properties and Engine Performance. A Comparison Review. In MATEC Web of Conferences; EDP Sciences: Les Ulis, France, 2016; p. 03002. [Google Scholar]
- Kumbhar, V.; Pandey, A.; Varghese, A.; Wanjari, S. An overview of production, properties and prospects of tamarind seed oil biodiesel as an engine fuel. Int. J. Ambient Energy 2020, 41, 1–9. [Google Scholar] [CrossRef]
- Shaisundaram, V.S.; Chandrasekaran, M.; Muraliraja, R.; Shanmugam, M.; Baskar, S.; Bhuvendran, A. Investigation of Tamarind seed oil biodiesel with aluminium oxide nanoparticle in CI engine. Mater. Today Proceed. 2021, 37, 1417–1421. [Google Scholar] [CrossRef]
- Raju, V.D.; Kishore, P.S.; Yamini, K. Experimental studies on four stroke diesel engine fuelled with Tamarind seed oil as potential alternate fuel for sustainable green environment. Eur. J. Sustain. Dev. 2018, 2, 10. [Google Scholar]
- Lokman, I.M.; Rashid, U.; Taufiq-Yap, Y.H. Production of biodiesel from palm fatty acid distillate using sulfonated-glucose solid acid catalyst: Characterization and optimization. Chin. J. Chem. Eng. 2015, 23, 1857–1864. [Google Scholar] [CrossRef]
- Azeem, M.W.; Hanif, M.A.; Al-Sabahi, J.N.; Khan, A.A.; Naz, S.; Ijaz, A. Production of biodiesel from low priced, renewable and abundant date seed oil. Renew. Energy 2016, 86, 124–132. [Google Scholar] [CrossRef]
- Mohammed, N.I.; Kabbashi, N.A.; Alam, M.Z.; Mirghani, M.E. Jatropha curcas oil characterization and its significance for feedstock selection in biodiesel production. Catalysts 2014, 6, 7. [Google Scholar]
- Ogbunugafor, H.; Eneh, F.; Ozumba, A.; Igwo-Ezikpe, M.; Okpuzor, J.; Igwilo, I.; Adenekan, S.; Onyekwelu, O. Physico-chemical and antioxidant properties of Moringa oleifera seed oil. Pak. J. Nutr. 2011, 10, 409–414. [Google Scholar] [CrossRef]
- Krisnangkura, K. A simple method for estimation of cetane index of vegetable oil methyl esters. J. Am. Oil Chem. Soc. 1986, 63, 552–553. [Google Scholar] [CrossRef]
- Bhatti, H.N.; Hanif, M.A.; Faruq, U.; Sheikh, M.A. Acid and base catalyzed transesterification of animal fats to biodiesel. Iran. J. Chem. Chem. Eng. 2008, 27, 41–48. [Google Scholar]
- Fernando, S.; Karra, P.; Hernandez, R.; Jha, S.K. Effect of incompletely converted soybean oil on biodiesel quality. Energy 2007, 32, 844–851. [Google Scholar] [CrossRef]
- Lam, M.K.; Jamlluddin, N.A.; Lee, K. Production of biodiesel using palm oil. In Biofuels: Alternative Feedstocks and Conversion Processes for the Production of Liquid and Gaseous Biofuels; Academic Press: Cambridge, MA, USA, 2011; pp. 539–574. [Google Scholar]
- Hossain, A.B.M.S.; Mazen, M.A. Effects of catalyst types and concentrations on biodiesel production from waste soybean oil biomass as renewable energy and environmental recycling process. Aust. J. Crop Sci. 2010, 4, 550–555. [Google Scholar]
- Wibowo, A.D.K. Study on production process of biodiesel from rubber seed (Hevea brasiliensis) by in situ (trans) esterification method with acid catalyst. Energy Procedia 2013, 32, 64–73. [Google Scholar]
- Barabás, I.; Todoruț, I.-A. Biodiesel quality, standards and properties. In Biodiesel-Quality, Emissions and By-Products; InTechOpen: London, UK, 2011; pp. 1–28. [Google Scholar]
- Baste, S.V.; Bhosale, A.V.; Chavan, S.B. Emission characteristics of Pongamia pinnata (Karanja) biodiesel and its blending up to 100% in a CI engine. Res. J. Agric. Fores. Sci. 2013, 1, 1–5. [Google Scholar]
- Indhumathi, P.; Shabudeen, P.S.S.; Shoba, U. A Method for production and characterization of biodiesel from green micro algae. Int. J. Bio-Sci. Bio-Tech. 2014, 6, 111–122. [Google Scholar] [CrossRef]
- Knothe, G.; Steidley, K.R. A comprehensive evaluation of the density of neat fatty acids and esters. J. Am. Oil Chem. Soc. 2014, 91, 1711–1722. [Google Scholar] [CrossRef]
- Bobade, S.; Khyade, V. Preparation of methyl ester (biodiesel) from karanja (Pongamia pinnata) oil. Res. J. Chem. Sci. 2012, 2, 43–50. [Google Scholar]
- Srivastava, A.; Prasad, R. Triglycerides-based diesel fuels. Renew. Sustain. Energy Rev. 2000, 4, 111–133. [Google Scholar] [CrossRef]
- Nielsen, S.S. Food analysis laboratory manual. In Food Science Text Series; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Ali, E.N.; Tay, C.I. Characterization of biodiesel produced from palm oil via base catalyzed transesterification. Procedia Eng. 2013, 53, 7–12. [Google Scholar] [CrossRef]
- Emmanuel, C.O.; Njoku, U.O. Production and characterization of biodiesel from fluted Pumpkin (Telfairia occidentalis Hook F) seed oil. Asian J. Res. Chem. 2011, 4, 1582–1586. [Google Scholar]
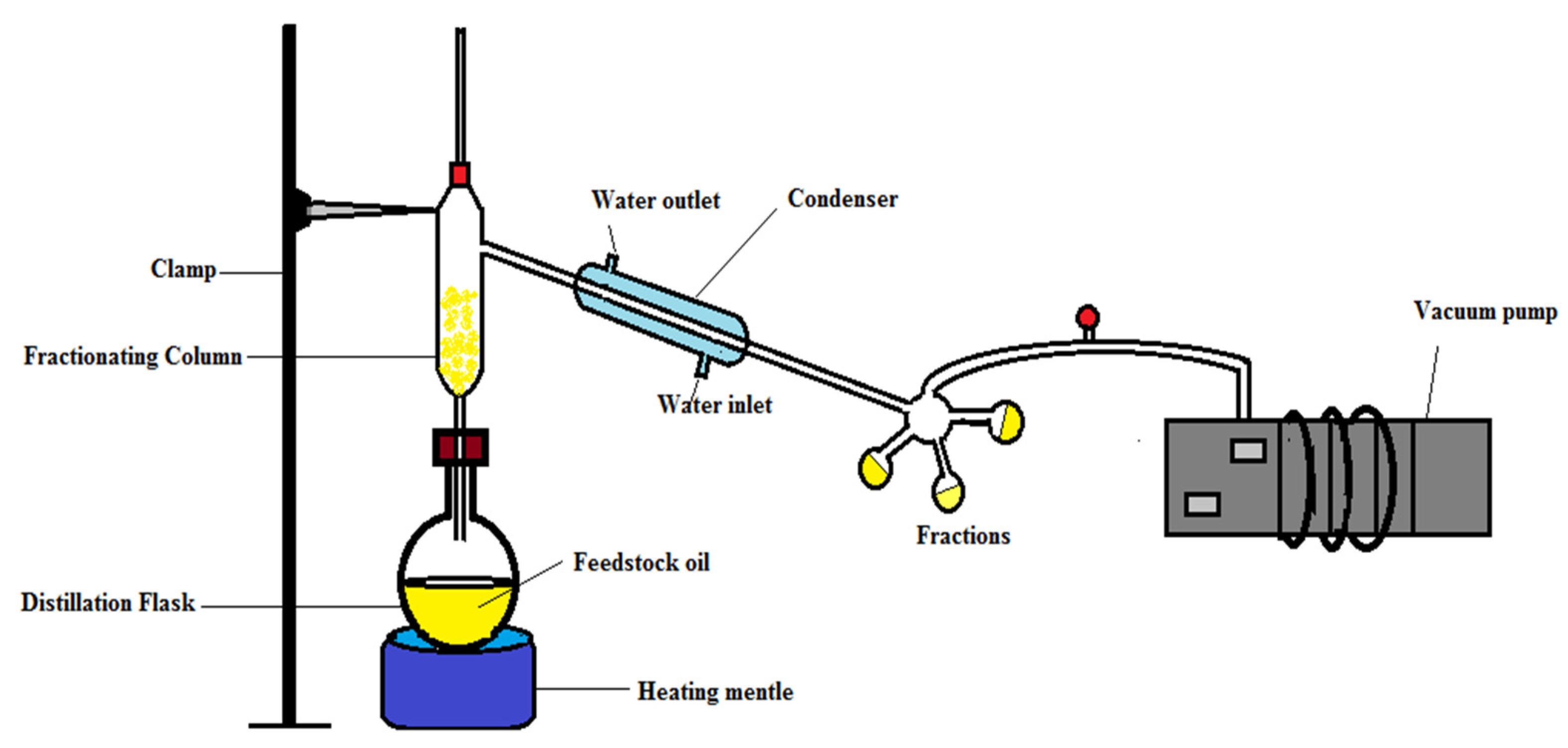
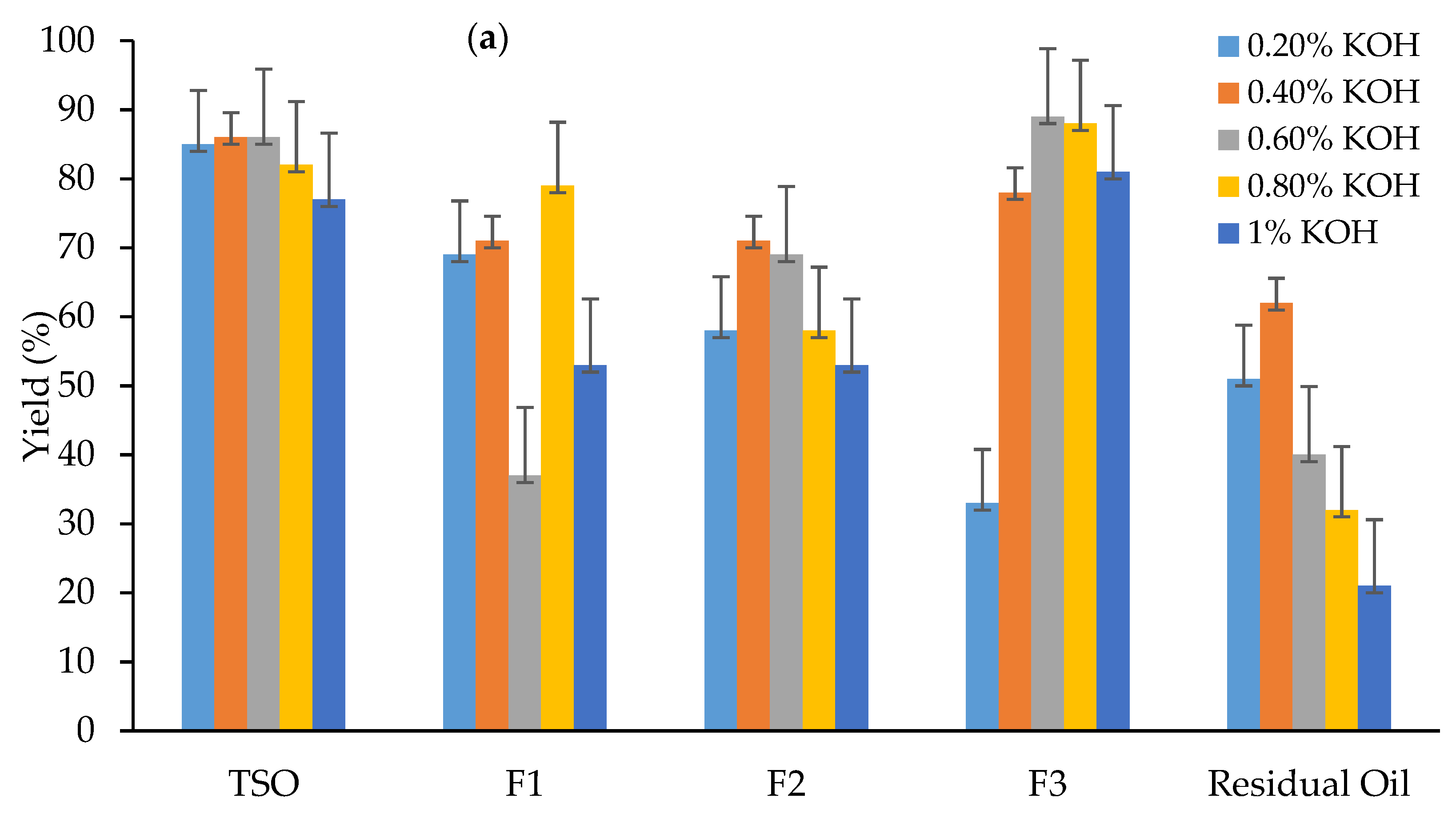
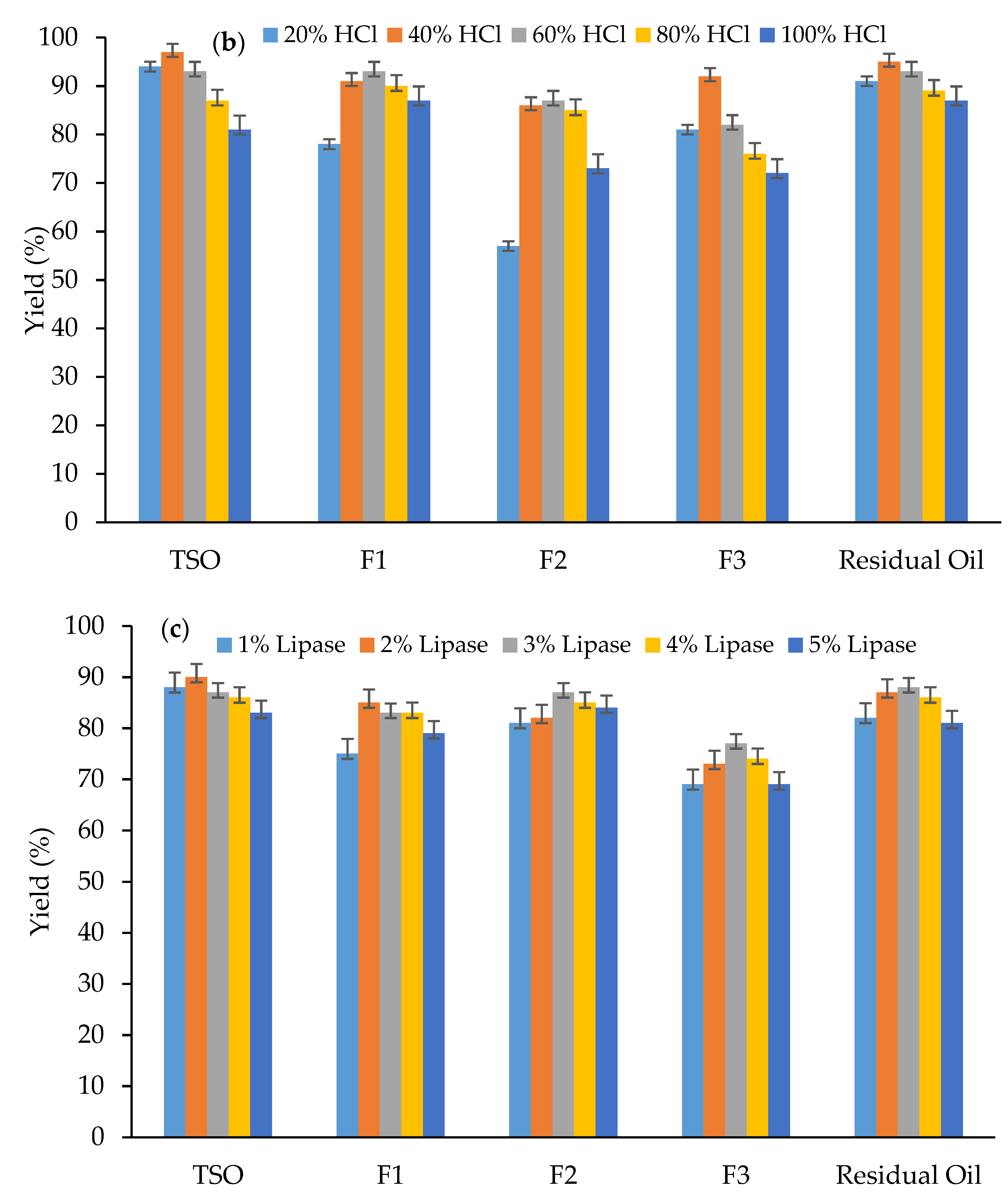
| Fraction Number | Boiling Range (°C) | wt. (g) | % of Total TSO |
|---|---|---|---|
| F1 | 147–210 | 215.87 | 21.6 |
| F2 | 230–268 | 280.09 | 28.0 |
| F3 | 260–290 | 330.12 | 33.0 |
| Residual oil | _ | 173.92 | 17.4 |
| Fatty Acid | Percentage Composition (%) | ||||
|---|---|---|---|---|---|
| F1 | F2 | F3 | TSO | Residual Oil | |
| Caprylic acid | 13.1 | 12.4 | - | 6.8 | - |
| Capric acid | 2.5 | 2.4 | 4.2 | 1.4 | 22.0 |
| Lauric acid | - | - | - | 12.0 | - |
| Myristic acid | 3.8 | 0 | 5.4 | 6.5 | - |
| Palmitic acid | 11.9 | 0 | 1.9 | 8.9 | 13.2 |
| Stearic acid | 20.2 | 4.7 | 3.4 | 1.3 | 25.9 |
| Arachidic acid | - | - | - | 1.0 | 15.9 |
| Behenic acid | - | 0.8 | 1.0 | 1.3 | - |
| Lignoceric acid | 22.7 | 3.5 | 2.3 | 1.6 | 16.1 |
| Oleic acid | 17.2 | 58.2 | 15.2 | 17.1 | 3.8 |
| Linoleic acid | 5.1 | 17.1 | 65.1 | 31.8 | 2.9 |
| Vaccenic acid | 1.2 | 0.8 | 7.1 | 9.4 | - |
| Gondoic acid | 0.5 | - | - | 1.0 | - |
| Catalyst | Conc. of Catalyst | Saponification Values (mg KOH/g) | |||
|---|---|---|---|---|---|
| F1 | F2 | F3 | TSO | ||
| KOH | 0.2 | 171.0 | 186.1 | 179.2 | 195 |
| 0.4 | 168.3 | 187.2 | 177.3 | 196 | |
| 0.6 | 159.0 | 190.2 | 179.2 | 210 | |
| 0.8 | 173.9 | 186.5 | 179.2 | 193 | |
| 1.0 | 169.2 | 190.2 | 176.0 | 193 | |
| HCl | 20 | 172.3 | 195.2 | 175.8 | 201 |
| 40 | 165.3 | 192.6 | 177.2 | 199 | |
| 60 | 168.9 | 191.4 | 180.8 | 194 | |
| 80 | 173.9 | 195.0 | 175.8 | 206 | |
| 100 | 169.2 | 197.1 | 183.0 | 195 | |
| Lipase | 1 | 172.3 | 196.8 | 180.0 | 193 |
| 2 | 171.0 | 196.8 | 185.2 | 192 | |
| 3 | 168.3 | 199.5 | 176.0 | 201 | |
| 4 | 169.0 | 197.1 | 185.8 | 207 | |
| 5 | 173.9 | 197.1 | 177.2 | 199 | |
| Catalyst | Conc. of Catalyst | Iodine Values (mg KOH/g) | |||
|---|---|---|---|---|---|
| F1 | F2 | F3 | TSO | ||
| KOH | 0.2 | 32.5 | 43.21 | 55.21 | 26.12 |
| 0.4 | 23.10 | 45.72 | 59.20 | 27.26 | |
| 0.6 | 23.14 | 39.72 | 52.90 | 32.06 | |
| 0.8 | 25.10 | 37.54 | 57.32 | 31.02 | |
| 1.0 | 22.99 | 39.21 | 52.20 | 26.09 | |
| HCl | 20 | 23.14 | 37.21 | 48.20 | 28.09 |
| 40 | 22.99 | 46.21 | 49.41 | 28.09 | |
| 60 | 24.57 | 38.45 | 50.21 | 32.12 | |
| 80 | 25.66 | 49.90 | 53.90 | 27.18 | |
| 100 | 22.10 | 38.45 | 51.20 | 31.02 | |
| Lipase | 1 | 25.10 | 45.91 | 48.21 | 30.16 |
| 2 | 23.14 | 39.90 | 49.01 | 29.12 | |
| 3 | 26.11 | 43.78 | 54.21 | 27.19 | |
| 4 | 22.99 | 45.12 | 49.21 | 30.16 | |
| 5 | 22.99 | 36.21 | 51.01 | 28.09 | |
| Catalyst | Conc. of Catalyst (%) | F1 | F2 | F3 | Residual Oil | TSO | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CP | PP | CP | PP | CP | PP | CP | PP | CP | PP | ||
| KOH | 0.2 | 11.6 | 2.7 | −2.9 | −6.1 | −3.9 | −6.1 | Room temperature | 1.5 | −1.2 | |
| 0.4 | 12.1 | 5.1 | −2.3 | −6.4 | −3.2 | −6.1 | Room temperature | 1.2 | −1.2 | ||
| 0.6 | 11.8 | 2.5 | −3.2 | −6.1 | −3.1 | −5.2 | Room temperature | 1.5 | −0.3 | ||
| 0.8 | 13.1 | 2.5 | −3.1 | −6.7 | −2.9 | −5.9 | Room temperature | 1.4 | −0.3 | ||
| 1.0 | 14.9 | 3.9 | −3.9 | −5.9 | −4.2 | −7.1 | Room temperature | 1.4 | −0.4 | ||
| HCl | 20 | 12.7 | 5.3 | −2.5 | −6.9 | −2.1 | −8.1 | Room temperature | −0.6 | −2.5 | |
| 40 | 12.1 | 4.9 | −2.0 | −6.2 | −2.9 | −7.3 | Room temperature | 0.7 | −2.3 | ||
| 60 | 13.3 | 6.6 | −2.7 | −6.2 | −2.2 | −6.9 | Room temperature | 1.6 | −2.2 | ||
| 80 | 15.2 | 5.1 | −3.1 | −6.5 | −3.2 | −6.5 | Room temperature | 0.3 | −0.7 | ||
| 100 | 12.2 | 6.2 | −2.9 | −5.9 | −3.3 | −6.9 | Room temperature | 0.1 | −0.4 | ||
| Lipase | 1 | 15.1 | 5.1 | −3.8 | −5.6 | −3.5 | −6.1 | Room temperature | 1.5 | −1.2 | |
| 2 | 13.3 | 5.5 | −3.5 | −6.1 | −3.5 | −6.1 | Room temperature | 1.4 | −2.2 | ||
| 3 | 14.9 | 4.8 | −2.9 | −4.4 | −3.3 | −5.6 | Room temperature | 1.2 | −2.5 | ||
| 4 | 12.4 | 6.1 | −2.3 | −6.1 | −2.2 | −6.1 | Room temperature | 0.6 | −2.1 | ||
| 5 | 13.1 | 5.9 | −3.9 | −6.3 | −2.8 | −5.8 | Room temperature | 0.2 | −1.9 | ||
| Catalyst | Conc. of Catalyst | Cetane Number | |||
|---|---|---|---|---|---|
| F1 | F2 | F3 | TSO | ||
| KOH | 0.2 | 70.9 | 65.9 | 64.3 | 68.41 |
| 0.4 | 73.5 | 67.4 | 63.8 | 68.01 | |
| 0.6 | 75.4 | 66.9 | 64.8 | 65.07 | |
| 0.8 | 72.0 | 66.0 | 63.8 | 67.60 | |
| 1.0 | 73.5 | 66.2 | 65.4 | 68.70 | |
| HCl | 20 | 72.7 | 65.8 | 66.5 | 67.13 |
| 40 | 74.1 | 64.2 | 65.1 | 67.40 | |
| 60 | 73.0 | 64.4 | 65.1 | 67.20 | |
| 80 | 71.9 | 63.0 | 65.2 | 66.07 | |
| 100 | 73.5 | 65.3 | 64.6 | 67.31 | |
| Lipase | 1 | 72.3 | 63.7 | 65.4 | 66.66 |
| 2 | 73.0 | 66.0 | 64.7 | 68.10 | |
| 3 | 72.8 | 63.8 | 65.1 | 67.33 | |
| 4 | 73.4 | 63.8 | 65.6 | 65.88 | |
| 5 | 72.5 | 65.1 | 65.6 | 67.40 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mushtaq, A.; Hanif, M.A.; Zahid, M.; Rashid, U.; Mushtaq, Z.; Zubair, M.; Moser, B.R.; Alharthi, F.A. Production and Evaluation of Fractionated Tamarind Seed Oil Methyl Esters as a New Source of Biodiesel. Energies 2021, 14, 7148. https://doi.org/10.3390/en14217148
Mushtaq A, Hanif MA, Zahid M, Rashid U, Mushtaq Z, Zubair M, Moser BR, Alharthi FA. Production and Evaluation of Fractionated Tamarind Seed Oil Methyl Esters as a New Source of Biodiesel. Energies. 2021; 14(21):7148. https://doi.org/10.3390/en14217148
Chicago/Turabian StyleMushtaq, Ayesha, Muhammad Asif Hanif, Muhammad Zahid, Umer Rashid, Zahid Mushtaq, Muhammad Zubair, Bryan R. Moser, and Fahad A. Alharthi. 2021. "Production and Evaluation of Fractionated Tamarind Seed Oil Methyl Esters as a New Source of Biodiesel" Energies 14, no. 21: 7148. https://doi.org/10.3390/en14217148
APA StyleMushtaq, A., Hanif, M. A., Zahid, M., Rashid, U., Mushtaq, Z., Zubair, M., Moser, B. R., & Alharthi, F. A. (2021). Production and Evaluation of Fractionated Tamarind Seed Oil Methyl Esters as a New Source of Biodiesel. Energies, 14(21), 7148. https://doi.org/10.3390/en14217148









