2.1. Volumetric Estimation of CO Storage Capacity
The gas volume factor of a component
X,
, is given as follows based on the equation of state of real gases:
where
V,
P,
T and
z are the volume, pressure, temperature and z-factor (compressibility factor), respectively, and subscripts
R and
S denote the reservoir and standard conditions (15
C and 1 Bar), respectively.
We consider the volume of CH
at the standard condition,
. This can be the amount of CH
produced by a reservoir. At the reservoir condition, this CH
occupies the following volume:
Hence, the same CO
volume that can be stored in the reservoir,
, can be calculated as follows:
Here, we used
. Finally, using Equation (
1) and
, we obtain:
The volume of CO
storage capacity estimated with this equation will be compared with the amount of stored CO
computed from the numerical simulations in
Section 3.
2.2. Numerical Model Descriptions
The ECLIPSE compositional simulator, a commercial reservoir simulator, was used. The mass conservation of each component is given by
where the first term denotes net mass accumulation of component
i; the second term denotes net mass flux; and the third term denotes source and sink terms. The simulator solves this differential equation based on finite difference methods (FDM). The second term, the net mass flux term, is determined from multiphase Darcy’s law, which is described as:
where the subscript
p denotes phase
p;
q is the Darcy velocity;
k and
are the absolute and relative permeability, respectively;
and
are the density and viscosity, respectively;
P is the hydrodynamic pressure; and
is the gravitational acceleration. More details on the numerical schemes of the simulator can be found in [
10,
11].
In this study, we considered the three components of CH
, CO
and water (aqueous phase) to model CO
injection in depleted dry gas reservoirs. In this numerical model, the phase separation of the components was modelled based on the modified Peng Robinson equation, proposed by Søreide and Whitson [
12], to obtain accurate gas solubilities in the aqueous phase [
11]. Hence, both CH
and CO
can be present in the aqueous phase as a dissolved component in water. In addition to mass transport by Darcy’s law, we modelled the diffusive mass transport of components in both the gas and aqueous phases based on Fick’s second law [
4]. The diffusion coefficient,
D, for the components in the gas and aqueous phases was set to 1.2
m
/s and 1.2
m
/s, respectively, based on the typical values for the molecular diffusion coefficient of the components in the gas and water phases [
13].
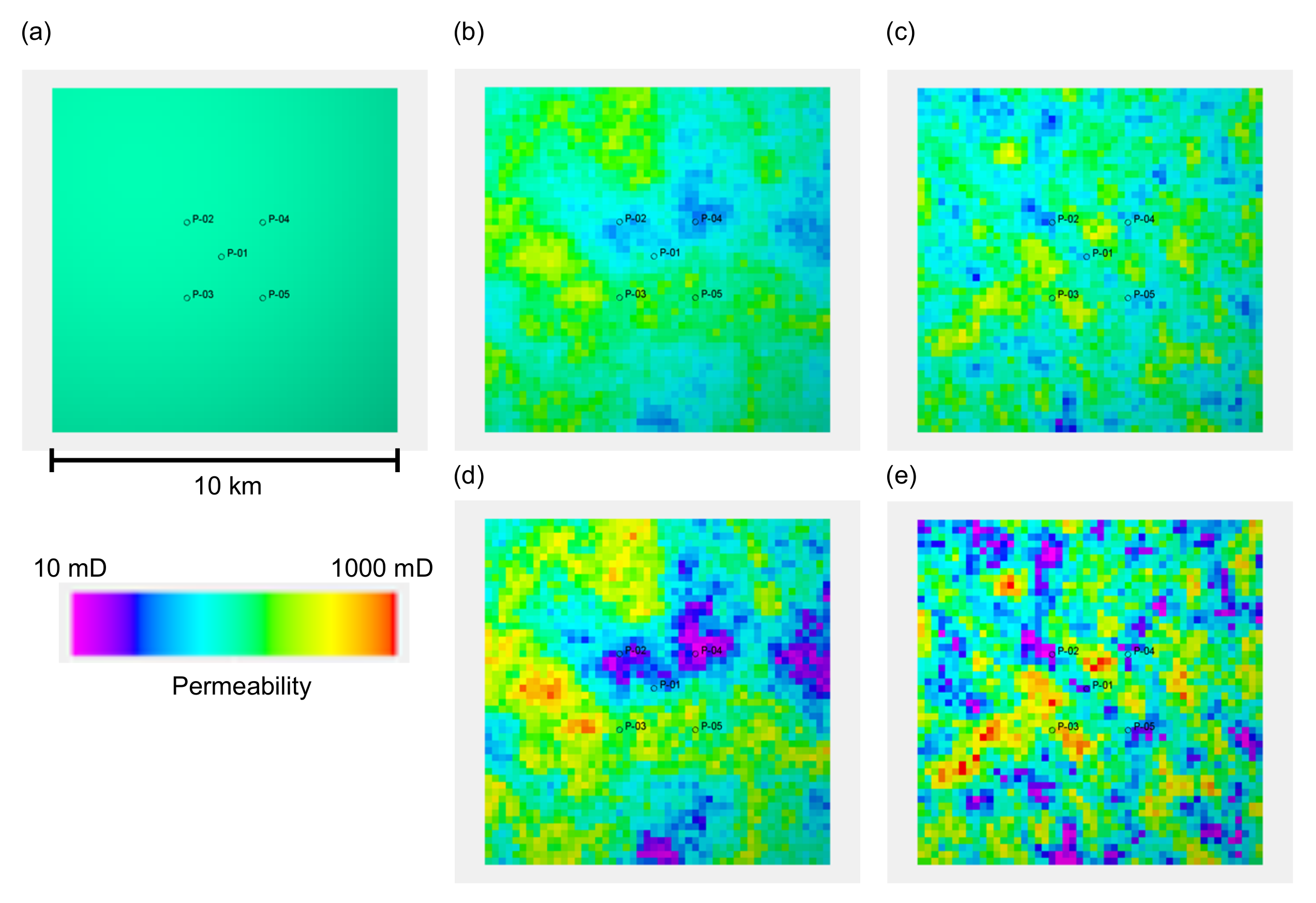
A four-way closure reservoir model, with the spherical shape of a reservoir top surface that had a horizontal extent of 10 km in the
x and
y directions, was used. A sand body with a uniform thickness of 50 m was considered as a reservoir sand. A cap rock layer with a uniform thickness of 5 m was modelled immediately above the reservoir section. This structure was modelled with a grid block with a size of 200 m × 200 m × 1 m, which resulted in 50 × 50 × 55 grid blocks. The depth of the reservoir top was 2000 m at the centre of the model. The gas–water contact was located at a depth of 2030 m. Hence, this gas reservoir was subjected to pressure support from the bottom aquifer below the gas–water contact. Five wells, composing a five-spot pattern with a 2 km side length, were placed in the model. These wells were completed for 10 m from the top reservoir surface.
Figure 1 depicts the reservoir model and the well location.
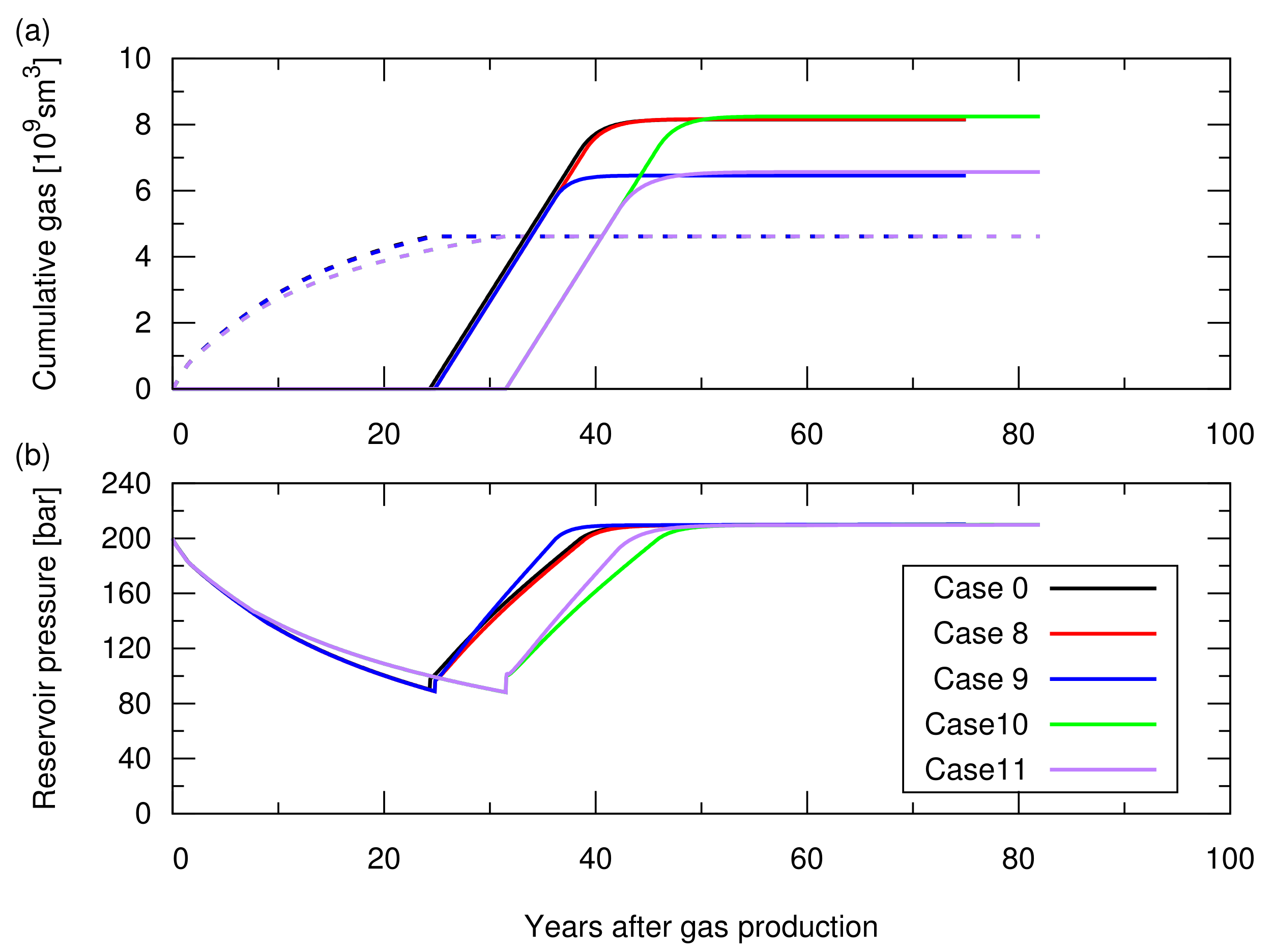
We assumed an isothermal temperature of 100
C and a hydrostatic pressure of 200 Bar at a datum depth of 2015 m (initial reservoir pressure varied with depth with the water gradient). As a base case (case 0), we considered the homogeneous porosity,
, and the permeability,
k, of 20% and 100 mD, respectively. We also considered heterogeneous
and
k distributions, which will be described in
Section 3.1.
For the saturation functions of the relative permeability,
, and the capillary pressure,
, of the reservoir sand, the hysteresis in drainage and imbibition was modelled as shown in
Figure 2. The relative permeability of the reservoir sand was determined based on the measured data of Krevor et al. [
14], preformed for a CO
/water system on Berea sandstone samples. These relative permeability curves had a connate water saturation,
, of 40% and a trapped gas saturation,
of 36%. The capillary pressure curves for drainage and imbibition were modelled based on the Van Genuchten model [
15]. The curve shape was determined based on the experimental data from Bottero et al. [
16], while the magnitude of the pressure was adjusted to provide a reasonable capillary pressure for a sandstone with a permeability of 100 mD—the maximum capillary pressure at
was 0.5 Bar, corresponding to a pore radius,
R, of ∼1
m for a CO
/water system that has an interfacial tension,
, of 35 mN/m and a contact angle,
, of 30
based on the Young–Laplace relationship:
. Furthermore, in
Section 3.2, we investigated the impact of the hysteresis on the saturation functions by changing the value of
, which will be described later.