Sugars Production from Municipal Forestry and Greening Wastes Pretreated by an Integrated Steam Explosion-Based Process
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Pretreatment
- Steam explosion (SE) pretreatment: Temperatures 185, 205, and 220 °C for 20 min.
- Metal salt combined with SE pretreatment (MS-SE): Metal salt, FeCl3 (0.1 M, 90 mg/g MFGW biomass); temperature, 180 and 195 °C; and residence time: 20 min.
- Organosolv combined with SE pretreatment (OS-SE): Solvent, ethanol (50%, v/v); temperature, 180 and 200 °C; and residence time, 20 min.
- Diluted-acid combined SE pretreatment (DA-SE): Acid, H2SO4 (60 mg/g MFGW biomass); temperature, 180–215 °C; and residence time, 5–15 min.
2.3. Enzymatic Hydrolysis Test
2.4. Fermentation Assay
2.5. Chemical Analysis
2.5.1. Extractives, Ash, Carbohydrate, and Lignin Content
2.5.2. Oligomers, Monomers, and Degradation Products
2.5.3. Ethanol
2.6. Statistical Analysis
3. Results
3.1. Raw Material Composition
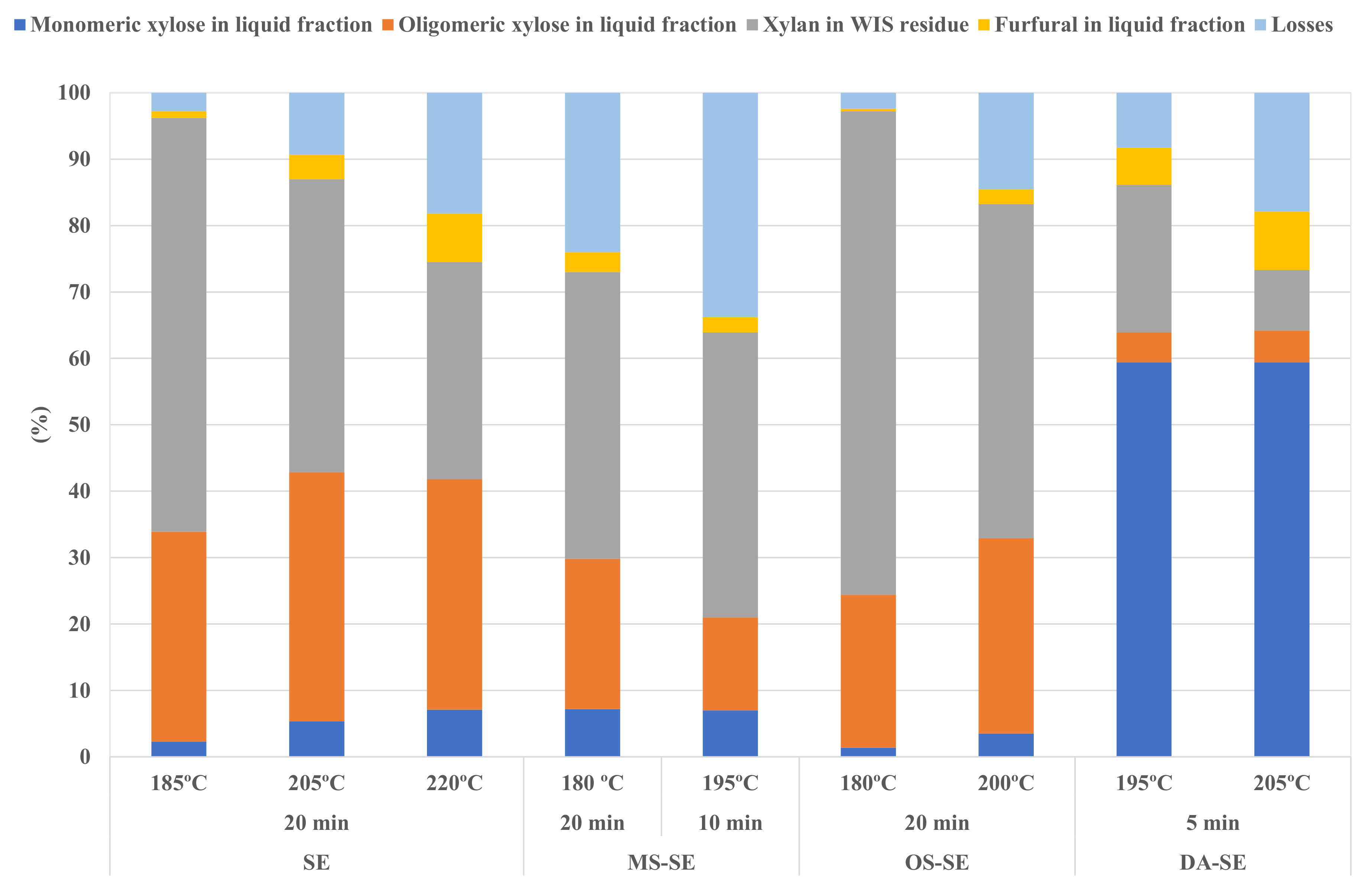
3.2. Effect of Pretreatment Conditions on WIS and Prehydrolysates
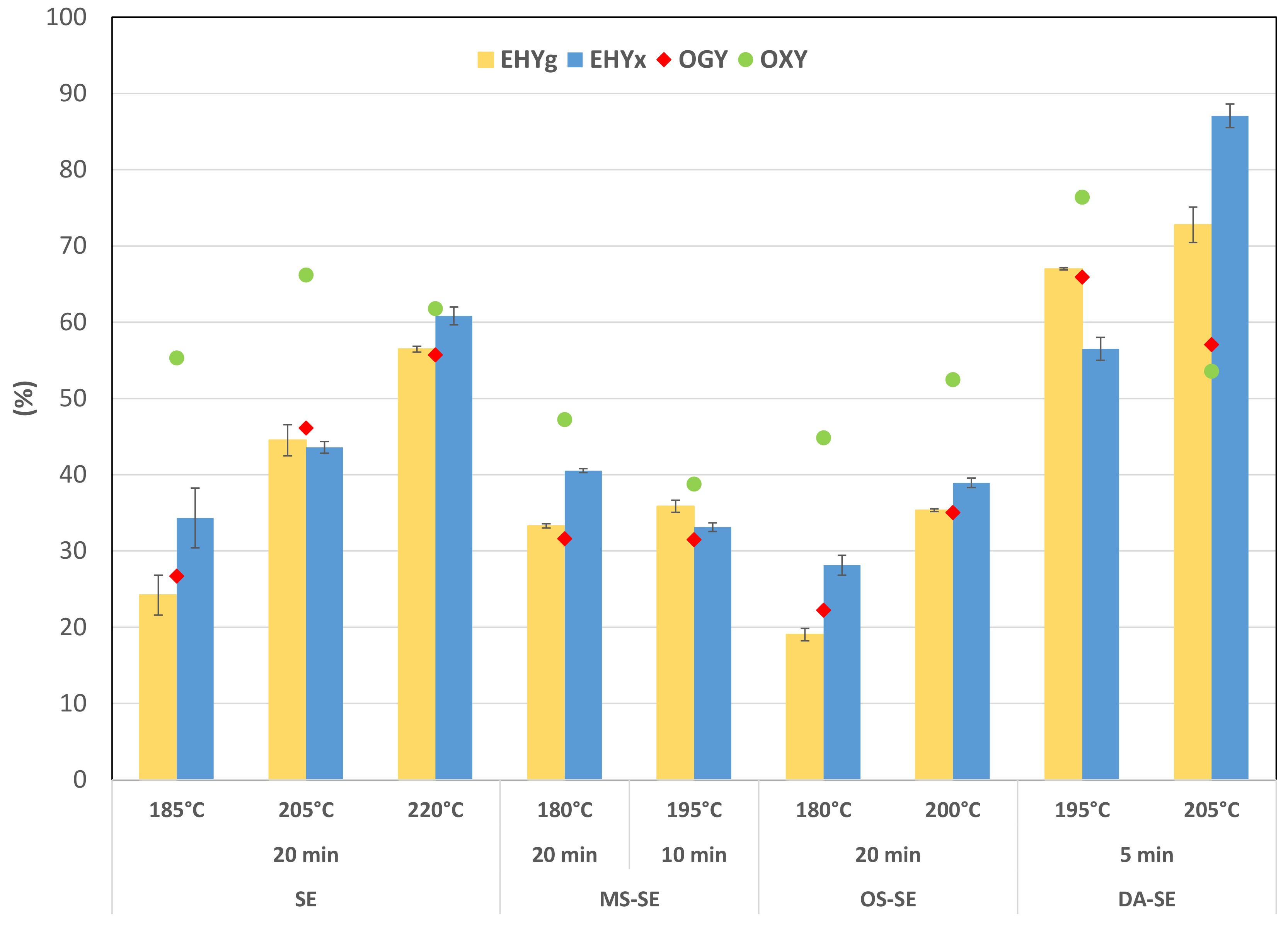
3.3. Evaluation of Potential Digestibility
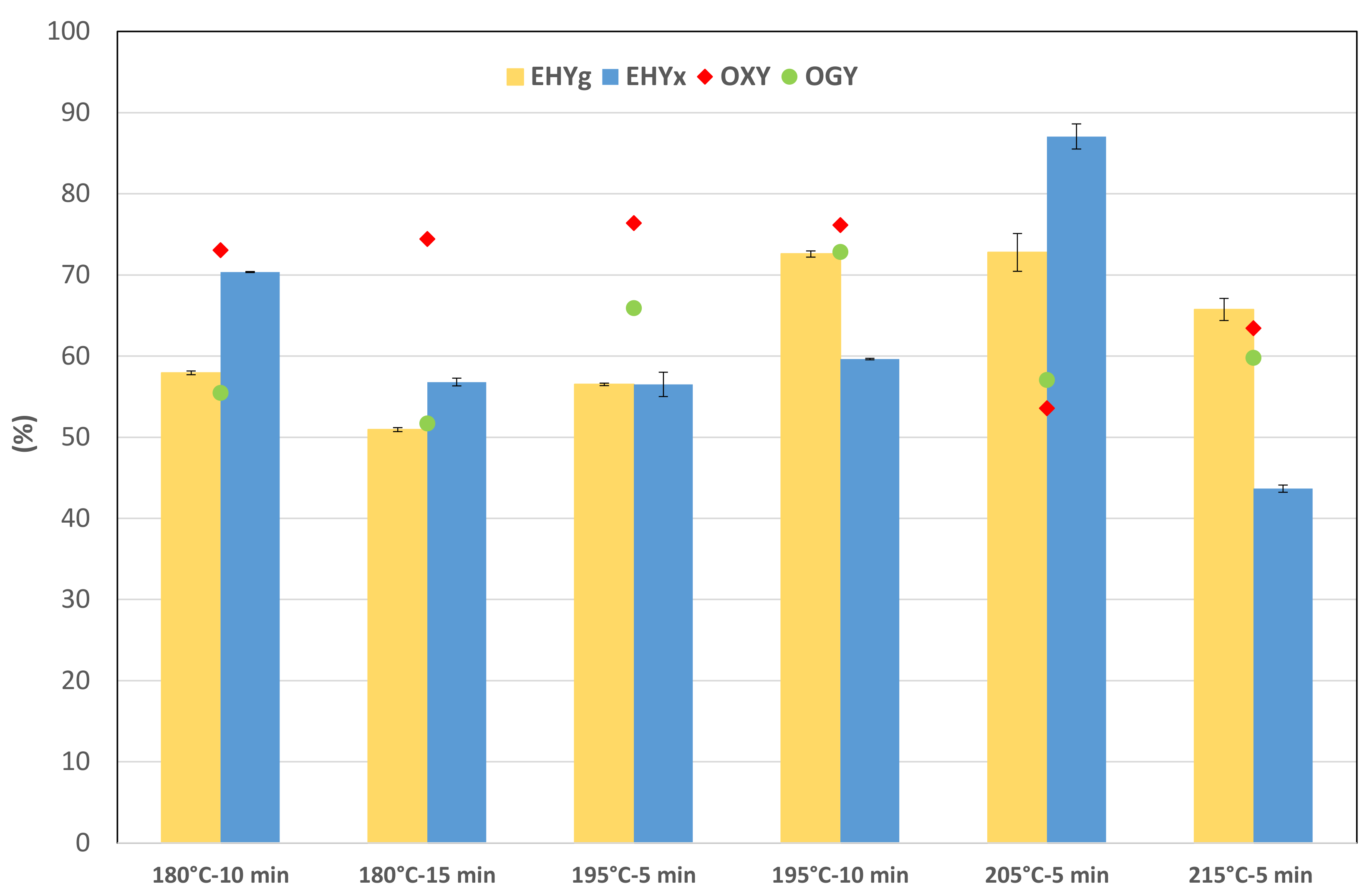
3.4. DA-SE Pretreatment Selection
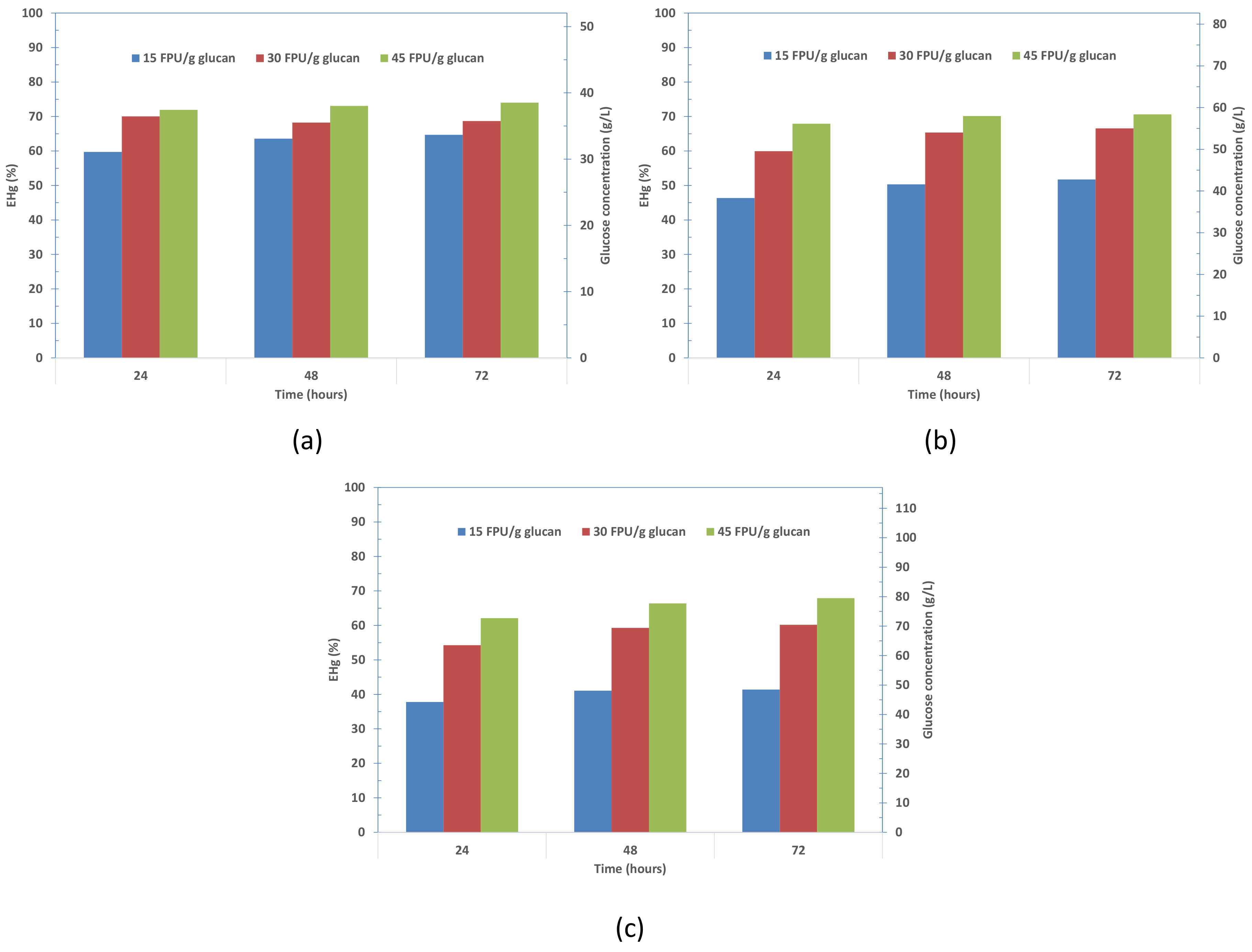
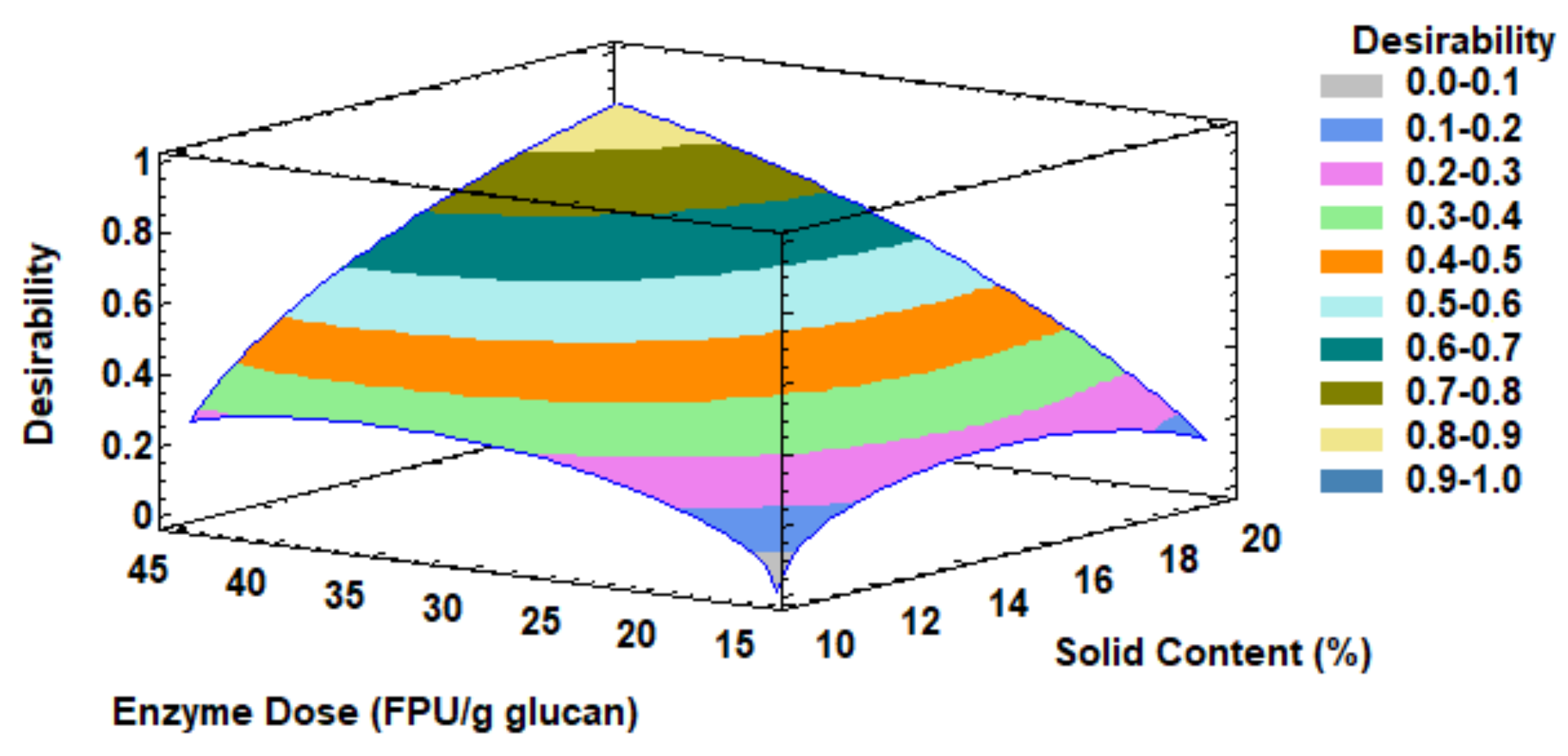
3.5. Study of the Effect of Solid Content and Enzyme Loading in Enzymatic Hydrolysis Step
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rojas-Rueda, D.; Nieuwenhuijsen, M.; Gascon, M.; Perez-Leon, D.; Mudu, P. Green spaces and mortality: A systematic review and meta-analysis of cohort studies. Lancet Planet. Health 2019, 3, 69–77. [Google Scholar] [CrossRef]
- Shi, Y.; Ge, Y.; Chang, J.; Shao, H.; Tang, Y. Garden waste biomass for renewable and sustainable energy production in China: Potential, challenges and development. Renew. Sust. Energy Rev. 2013, 22, 432–437. [Google Scholar] [CrossRef]
- Tetma. 2018. Available online: http://tetma.com/2018/03/28/tetma-y-centreverd-siguen-con-sucompromiso- (accessed on 24 May 2020).
- Benito, M.; Masaguer, A.; de Antonio, R.; Moliner, A. Blending green feedstocks at a Madrid composting facility (Spain). Biocycle 2005, 46, 72–74. [Google Scholar]
- Raud, M.; Mitt, M.; Oja, T.; Orupõld, K.; Kikas, T. The utilization potential of urban greening wastes: Tartu case study. Urban For. Urban Gree. 2017, 21, 96–101. [Google Scholar] [CrossRef]
- Kabir, M.J.; Chowdhury, A.A.; Rasul, M.G. Pyrolysis of municipal green waste: A modelling, simulation and experimental analysis. Energies 2015, 8, 7522–7541. [Google Scholar] [CrossRef]
- Arici, Ș.; Ersöz, Ö.; Bayrakci, A.G.; Eryaşar, A.; Koçar, G. Influence of thermal and alkali pretreatment to solubilisation and biomethane production of garden waste. Int. J. Glob. Warn. 2015, 7, 242–255. [Google Scholar] [CrossRef]
- Cubas-Cano, E.; González-Fernández, C.; Ballesteros, I.; Tomás-Pejó, E. Efficient utilization of hydrolysates from steam-exploded gardening residues for lactic acid production by optimization of enzyme addition and pH control. Waste Manag. 2020, 107, 235–243. [Google Scholar] [CrossRef]
- EU. Directive (EU) 2018/2001 of the European Parliament and of the Council of 11 December 2018 on the promotion of the use of energy from renewable sources. Off. J. Eur. Union 2018. Available online: https://eur-lex.europa.eu/eli/dir/2018/2001/oj (accessed on 28 May 2020).
- Moreno, D.; Tomás-Pejó, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for lignocellulosic biomass deconstruction within a biorefinery perspective. In Biomass, Biofuels, Biochemicals: Alternative Feedstocks and Conversion Processes, 2nd ed.; Pandey, P., Larroche, C., Dussap, C.G., Gnansounou, E., Khanal, S.-K., Ricke, S., Eds.; Academic Press: London, UK, 2019; pp. 379–399. [Google Scholar]
- Valdivia, M.; Galán, J.J.; Laffarga, J.; Ramos, J.L. Biofuels 2020: Biorefineries base on lignocellulosic materials. Micr. Biotechnol. 2016, 9, 585–594. [Google Scholar] [CrossRef]
- Alvira, P.; Tomás-Pejó, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef]
- Duque, A.; Manzanares, P.; Ballesteros, I.; Ballesteros, M. Steam explosion as lignocellulosic biomass pretreatment. In Biomass Fractionation Technologies for a Lignocellulosic Feedstock Based Biorefinery; Mussatto, S.I., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 349–368. [Google Scholar]
- Chen, H.Z.; Liu, Z.H. Steam explosion and its combinatorial pretreatment refining technology of plant biomass to bio-based products. Biotech. J. 2015, 10, 866–885. [Google Scholar] [CrossRef]
- Zhang, K.; Pei, Z.; Wang, D. Organic solvent pretreatment of lignocellulosic biomass and biochemicals: A review. Bioresour. Technol. 2016, 199, 21–33. [Google Scholar] [CrossRef]
- Matsakas, L.; Nitsos, C.; Raghavendran, V.; Yakimenko, O.; Personn, G.; Olsoon, E.; Rova, U.; Olsson, L.; Cristakopoulos, P. A novel hybrid organosolv: Steam explosion methods for the efficient fractionationand pretreatment of birch biomass. Biotechnol. Biofuels 2018, 11, 160. [Google Scholar] [CrossRef] [PubMed]
- Amarasekara, A.S.; Deng, F. Single reagent treatment and degradation of switchgrass using iron (III)chloride: The effects on hemicellulose, cellulose and lignin. Biomass Bioenergy 2019, 131, 105421. [Google Scholar] [CrossRef]
- López-Linares, J.C.; Romero, I.; Moya, M.; Cara, C.; Ruiz, E.; Castro, E. Pretreatment of olive tree biomass with FeCl3 prior enzymatic hydrolysis. Bioresour. Technol. 2013, 128, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Sun, J.; Li, M.; Wang, S.; Pei, H.; Zhang, J. Enhanced enzymatic hydrolysis and structural features of corn stover by FeCl3 pretreatment. Bioresour. Technol. 2009, 100, 5853–5858. [Google Scholar] [CrossRef] [PubMed]
- Sluiter, J.B.; Ruiz, R.O.; Scarlata, C.J.; Sluiter, A.D.; Templeton, D.W. Compositional analysis of lignocellulosic feedstocks. 1. Review and description of methods. J. Agric. Food Chem. 2010, 58, 9043–9053. [Google Scholar] [CrossRef]
- Álvarez, C.; González, A.; Negro, M.J.; Ballesteros, I.; Oliva, J.M.; Sáez, F. Optimized use of hemicelluloses within a biorefinery for processing high value-added xylooligosaccharides. Ind. Crops Prod. 2017, 99, 41–48. [Google Scholar] [CrossRef]
- Pawar, P.M.A.; Koutaniemi, S.; Tenkanen, M.; Mellerowicz, E.J. Acetylation of woody lignocellulose: Significance and regulation. Front. Plant Sci. 2013, 4, 1–8. [Google Scholar] [CrossRef]
- del Rio, J.C.; Marques, G.; Rencoret, J.; Martínez, A.T.; Gutiérrez, A. Occurrence of naturally acetylated lignin units. J. Agric. Food Chem. 2007, 55, 5461–5468. [Google Scholar] [CrossRef]
- Raud, M.; Kesperi, R.; Oja, T.; Olt, J. Utilization of urban waste in bioethanol production: Potential and technical solutions. Agron. Res. 2014, 12, 397–406. [Google Scholar]
- Cubas-Cano, E.; López-Gómez, J.P.; González-Fernández, C.; Ballesteros, I.; Tomas-Pejó, E. Towards sequential bioethanol and L-lactic acid co-generation: Improving xylose conversion to L-lactic acid in presence of lignocellulosic ethanol with an evolved Bacillus coagulans. Renew. Energy 2020, 153, 759–765. [Google Scholar]
- Wang, Q.; Wang, Z.; Shen, F.; Hu, J.; Sun, F.; Lin, L.; Yang, G.; Zhang, Y.; Deng, S. Pretreating lignocellulosic biomass by concentrated phosphoric acid plus hydrogen peroxide (PHP) for enzymatic hydrolysis: Evaluating the pretreatment flexibility on feedstocks and particle sizes. Bioresour. Technol. 2014, 160, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Boldrin, A.; Christensen, T.H. Seasonal generation and composition of garden waste in Aarthus (Denmark). Waste Manag. 2010, 30, 551–557. [Google Scholar] [CrossRef]
- Romaní, A.; Garrote, G.; Ballesteros, I.; Ballesteros, M. Second generation bioethanol from steam exploded Eucalyptus globulus wood. Fuel 2013, 111, 66–74. [Google Scholar] [CrossRef]
- López-Linares, J.C.; Ballesteros, I.; Tourán, J.; Cara, C.; Castro, E.; Ballesteros, M.; Romero, I. Optimization of uncatalyzed steam explosion pretreatment of rapeseed straw for biofuel production. Bioresour. Technol. 2015, 190, 97–105. [Google Scholar] [CrossRef]
- Romero, I.; Ruiz, E.; Castro, E. Pretreatment with metal salts. In Biomass Fractionation Technologies for a Lignocellulosic Feedstock Based Biorefinery; Mussatto, S.I., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 209–227. [Google Scholar]
- Ballesteros, I.; Negro, M.J.; Oliva, J.M.; Cabañas, A.; Manzanares, P.; Ballesteros, M. Ethanol production from steam-explosion pretreated wheat straw. Appl. Biochem. Biotechnol. 2006, 129–132, 496–508. [Google Scholar] [CrossRef]
- Modenbach, A.A.; Nokes, S.E. Enzymatic hydrolysis of biomass at high-solid loading—A review. Biomass Bioenerg. 2013, 56, 526–544. [Google Scholar] [CrossRef]
- da Silva, A.S.; Espinheira, R.P.; Teixeira, R.S.S.; Fernández de Sousa, M.; Ferreira–Leitão, V.; Bon, E.P.S. Constraints and advances in high-solids enzymatic hydrolysis of lignocellulosic biomass: A critical review. Biotechnol. Biofuels 2020, 13, 58. [Google Scholar] [CrossRef]
- da Silva, A.S.; Fernández de Souza, M.; Ballesteros, I.; Manzanares, P.; Ballesteros, M.; Bon, E.P.S. High-solids content enzymatic hydrolysis of hydrothermally pretreated sugarcane bagasse using a laboratory-made enzyme blend and commercial preparations. Process. Biochem. 2016, 51, 1561–1567. [Google Scholar] [CrossRef]
| Component | Percentage on Dry Weight Basis |
|---|---|
| Extracts | 9.2 |
| Glucan | 34.7 ± 1.4 |
| Hemicelluloses | 14.0 |
| Xylan | 9.6 ± 0.03 |
| Galactan | 2.2 ± 0.12 |
| Arabinan | 1.3 ± 0.02 |
| Mannan | 0.9 ± 0.0 |
| Acetyl groups | 4.7 ± 0.10 |
| Lignin | 29.9 |
| Whole Ash | 3.7 ± 0.1 |
| Pretreatment | Time (min) | Temperature (°C) | RY (%) | Contents (%) | Recovered Glucan | Removed Xylan | Removed AIL * | ||
|---|---|---|---|---|---|---|---|---|---|
| Glucan | Xylan | AIL | |||||||
| SE | 20 | 185 | 73.5 | 39.6 | 8.1 | 38.6 | 81.8 | 37.7 | 5.1 |
| 205 | 71.4 | 40.1 | 6.0 | 38.8 | 81.1 | 55.9 | 7.3 | ||
| 220 | 72.8 | 42.3 | 4.3 | 40.7 | 87.1 | 67.3 | 5.6 | ||
| MS-SE | 20 | 180 | 69.7 | 37.3 | 5.9 | 39.1 | 73.5 | 56.8 | 8.8 |
| 10 | 195 | 69.0 | 37.7 | 6.0 | 38.1 | 73.6 | 57.1 | 12.1 | |
| OS-SE | 20 | 180 | 73.4 | 37.0 | 9.5 | 36.1 | 76.8 | 27.2 | 11.5 |
| 200 | 69.4 | 38.5 | 6.9 | 36.5 | 75.6 | 49.7 | 15.3 | ||
| DA-SE | 5 | 195 | 64.8 | 41.4 | 3.3 | 43.8 | 75.8 | 77.8 | 5.1 |
| 205 | 57.5 | 41.6 | 1.5 | 47.6 | 67.8 | 90.7 | 8.5 | ||
| Pretreatment | SE | MS-SE | OS-SE | DA-SE | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (°C) | 185 | 205 | 220 | 180 | 195 | 180 | 200 | 195 | 205 | |
| Time (min) | 20 | 20 | 10 | 20 | 5 | |||||
| Main sugars | Monomeric glucose | 0.57 | 0.75 | 0.7 | 1.21 | 1.26 | 0.75 | 0.83 | 4.68 | 4.62 |
| Oligomeric Glucose | 2.06 | 2.57 | 1.85 | 1.58 | 1.33 | 2.21 | 2.43 | 0.98 | 0.01 | |
| Monomeric Xylose | 0.25 | 0.58 | 0.77 | 0.78 | 0.76 | 0.15 | 0.38 | 6.42 | 6.41 | |
| Oligomeric Xylose | 3.42 | 4.05 | 3.75 | 2.44 | 1.51 | 2.48 | 3.17 | 0.48 | 0.52 | |
| Degradation products | Furfural | 0.07 | 0.25 | 0.51 | 0.21 | 0.16 | 0.03 | 0.16 | 0.39 | 0.61 |
| HMF | 0.04 | 0.1 | 0.2 | 0.12 | 0.11 | 0.03 | 0.08 | 0.36 | 0.42 | |
| Acetic acid | 0.68 | 1.36 | 1.92 | 1.2 | 1.24 | 0.46 | 1.01 | 2.24 | 2.42 | |
| Formic acid | 0.28 | 0.45 | 0.47 | 0.63 | 0.64 | 0.24 | 0.33 | 0.54 | 0.4 | |
| Levulinic acid | 0.02 | 0.03 | 0 | 0.03 | 0.03 | 0.02 | 0.02 | 0.01 | 0.09 | |
| Vanillin | 3 | 8 | 15 | 3 | 2 | 3 | 4 | 4 | 7 | |
| Hydroxybenzoic acid | 20 | 40 | 74 | 20 | 17 | 21 | 31 | 36 | <0.5 | |
| Syringaldehyde | ND | 6 | 15 | 3 | 3 | 2 | 4 | 7 | 10 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Negro, M.J.; Álvarez, C.; Doménech, P.; Iglesias, R.; Ballesteros, I. Sugars Production from Municipal Forestry and Greening Wastes Pretreated by an Integrated Steam Explosion-Based Process. Energies 2020, 13, 4432. https://doi.org/10.3390/en13174432
Negro MJ, Álvarez C, Doménech P, Iglesias R, Ballesteros I. Sugars Production from Municipal Forestry and Greening Wastes Pretreated by an Integrated Steam Explosion-Based Process. Energies. 2020; 13(17):4432. https://doi.org/10.3390/en13174432
Chicago/Turabian StyleNegro, Maria José, Cristina Álvarez, Pablo Doménech, Raquel Iglesias, and Ignacio Ballesteros. 2020. "Sugars Production from Municipal Forestry and Greening Wastes Pretreated by an Integrated Steam Explosion-Based Process" Energies 13, no. 17: 4432. https://doi.org/10.3390/en13174432
APA StyleNegro, M. J., Álvarez, C., Doménech, P., Iglesias, R., & Ballesteros, I. (2020). Sugars Production from Municipal Forestry and Greening Wastes Pretreated by an Integrated Steam Explosion-Based Process. Energies, 13(17), 4432. https://doi.org/10.3390/en13174432







