Operational Parameters of Biogas Plants: A Review and Evaluation Study
Abstract
1. Introduction
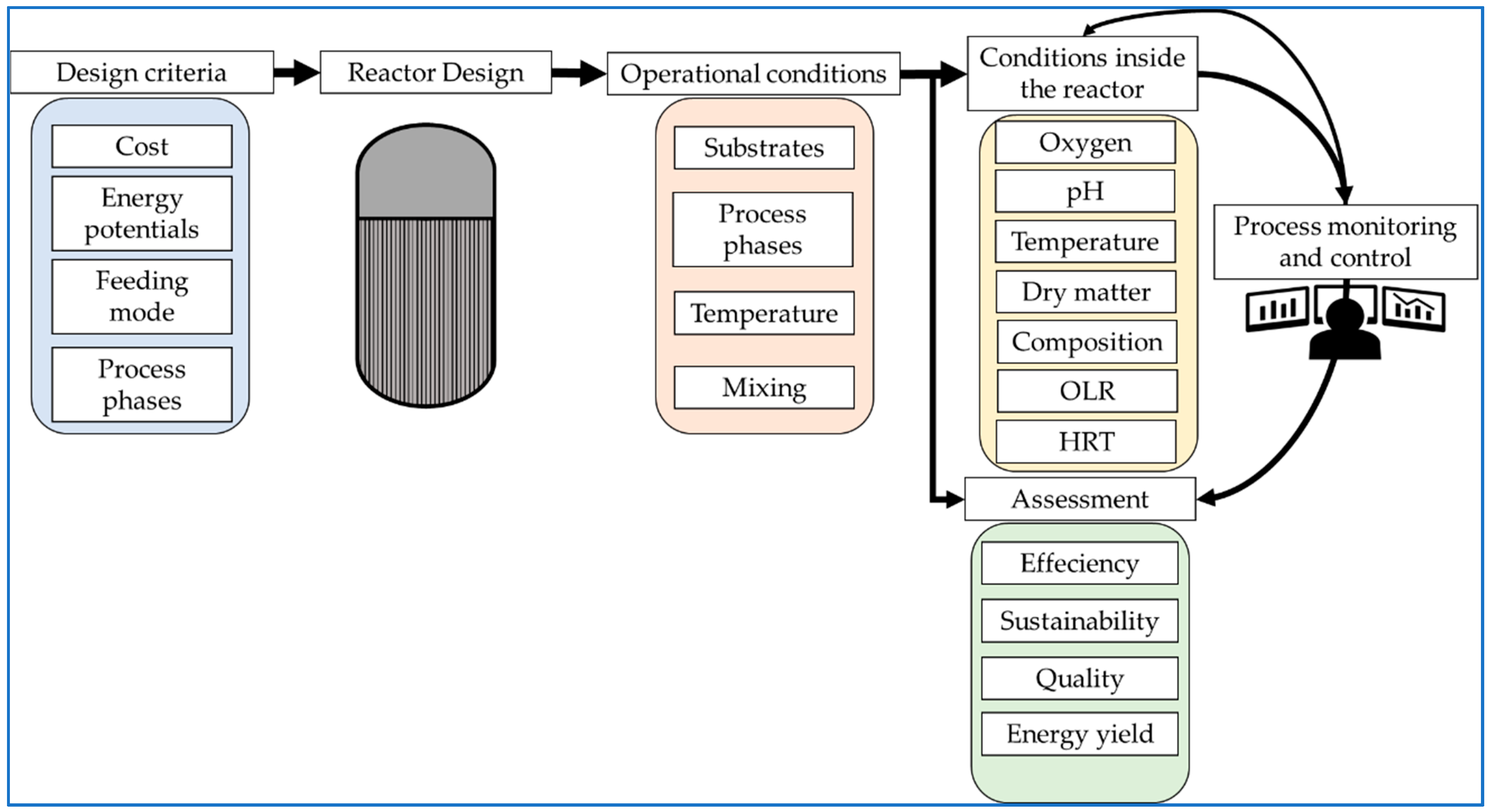
2. Reactor Design Considerations and Operational Conditions
- -
- Dry matter (DM) content of the substrate: Wet digestion (DM < 12%) and dry digestion (DM > 12%)
- -
- Mode of material feeding: Intermittent (no substrate addition during the dwell time), semi-continuous (at least once per working day) and continuous (flow)
- -
- Number of process phases: Single-phase (all steps take place in the same reactor) and two-phase (hydrolysis and methanogenesis take place in separate reactors)
- -
- Process temperature: Psychrophilic (<25 °C), mesophilic (37 to 42 °C) and thermophilic (50 to 60 °C) [3].
2.1. Substrates
- The feedstock’s composition determines the content of the volatile solids as well as the ammonia concentration inside the reactor [26].
- Based on the feedstock quality and quantity, sedimentation and floating layers could be obtained [27,28]. The high amount of impurities in the substrates leads to sedimentation. The size of the substrate and its biodegradability are factors which determine sedimentation potentials. The floating layers can be created by the surfactants.
- The anaerobic digestion stability is dependent on the feedstock, because of their different chemical and physical properties. Therefore, suitable feeding is required to ensure the anaerobic digestion process [29].
- The technology used for the anaerobic digestion, as well as the digester’s size and shape, are defined by the feedstock’s properties, e.g., DM, oDM, biogas formation potentials, as well as carbon to nitrogen ration (C:N ratio) [30]. Table 1 summarizes the main agricultural substrates that are globally utilized and their main properties.
2.2. Process Phases
- Cost: the installation and maintenance of the multi-stage system are more expensive than that for the one-stage system [51].
- Operational conditions: the optimal operation conditions (e.g., temperature and pH) for the microorganisms in the multi-stage systems are more demanding than that for single-stage systems, since the operational parameters in the different stages are diverse. On the other hand, due to the separation of the phases, there is a better process control.
- The stability of the anaerobic digestion process is improved through using the multi-stage systems. The methanogenesis step is very sensitive to the changes in the organic load rate, the heterogeneity of the biodegradable feedstock, and the changes in the environmental conditions. Hence, the multi-stage is more advantageous than the single-stage system, where the control of these conditions is more efficient, and the flow of the biodegradable feedstock from the first digester to the others is more homogeneous in quantity and quality [51,52,53,54,55,56,57].
- The single-stage systems are still the most used due to their simplicity [10].
2.3. Process Temperature
2.4. Mixing
- Reduce the creation of sediments on the bottom of the digesters to ensure the highest available volume for the anaerobic digestion process and reduce the need to clean the digester (on average, it is once every 4 to 7 years) [93].
- Break the foam layer on the top of the biodegradable substrate. The creation of this layer can inhibit 20% to 50% of the biogas production [94]. “Foam is generally a dispersion of a gas in a liquid consisting of a large proportion (approximately 95%) of gas. The liquid phase is located in a thin film which is present between the gas bubbles” [28,94]. Two groups of surface-active compounds are considered to be in charge of the foam formation, which are the surfactants and bio-surfactants. The surfactants are compounds inflowing the digester with the feeding, while the bio-surfactants are considered to be the outcomes of the activities of the microorganisms [27,28]. These foam layers have to be destroyed because their formation leads to a drop in the biogas yield, high-cost losses, and equipment damage, as well as operational disturbances [27,28,95,96,97].
- The size of the digester,
- The operating temperature inside the digesters,
- The used mixing technology,
- The used feedstock and the DM value of the biodegradable feedstock.
2.5. The Energetic Potential of the Biogas Plants
3. The Conditions Inside the Reactor
3.1. Oxygen
3.2. PH
3.3. Dry Matter Content of the Biodegradable Feedstock
- The researchers consider that the wet fermentation process occurs at dry matter content of less than 15%, while the dry fermentation takes place at higher DM values.
- The mixing inside the digesters (technology as well as duration) is highly dependent on the dry matter content, as mentioned in Section 2.4. The dry matter content of the biodegradable feedstock is a critical factor in controlling the Bingham viscosity and the yield stress [144].
3.4. Organic Loading Rate (OLR)
3.5. Hydraulic Retention Time (HRT)
3.6. Nutrients
- Carbon: For building cells’ structure.
- Nitrogen: For protein biosynthesis.
- Sulfur: For the growth of methanogens and component of amino acids.
3.7. Process Inhibitors
3.7.1. Ammonia
3.7.2. Sulfide
3.7.3. Light and Heavy Metals
3.7.4. Organics
3.7.5. Secondary Metabolites
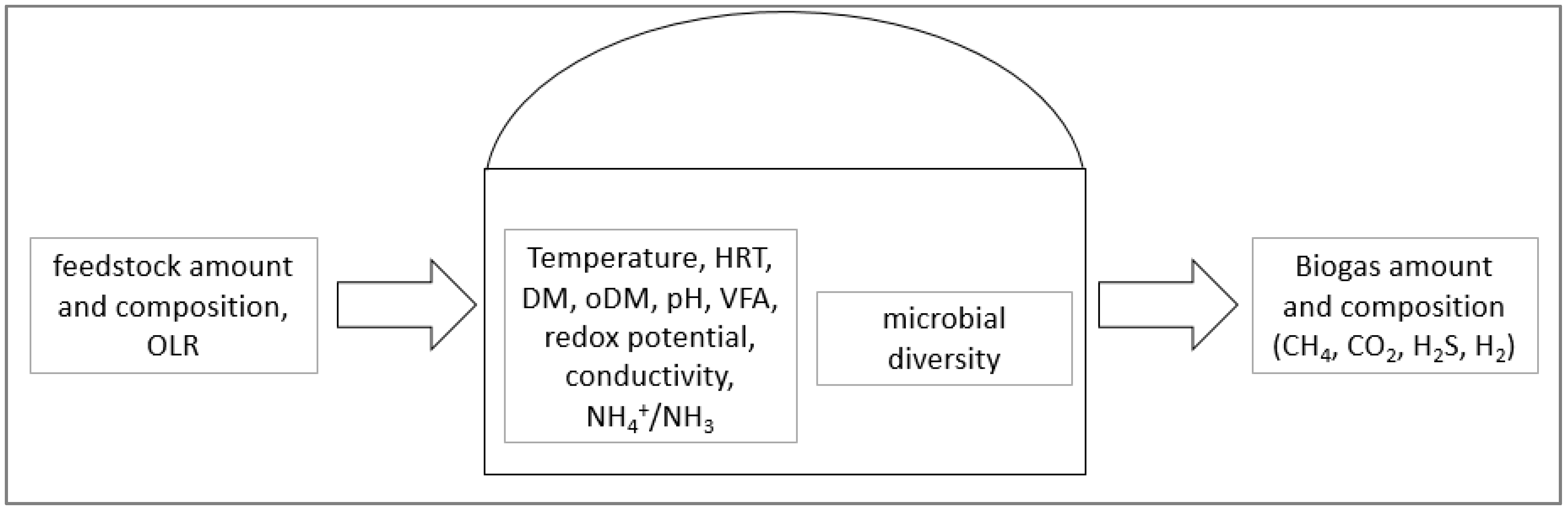
4. Monitoring of the Operational Conditions in Biogas Plants
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hren, R.; Petrovič, A.; Čuček, L.; Simonič, M. Determination of Various Parameters during Thermal and Biological Pretreatment of Waste Materials. Energies 2020, 13, 2262. [Google Scholar] [CrossRef]
- Falk, H.M.; Benz, H.C. Monitoring the Anaerobic Digestion Process; IRC-Library; Information Resource Center der Jacobs University Bremen: Bremen, Germany, 2011. [Google Scholar]
- Rohstoffe, F.N. Guide to Biogas from Production to Use; Federal Ministry of Food; Agriculture and Consumer Protection; Fachagentur Nachwachsende Rohstoffe E.V. (FNR): Gülzow, Germany, 2012. [Google Scholar]
- Refai, S. Development of Efficient Tools for Monitoring and Improvement of Biogas Production. Ph.D. Thesis, Universitäts-und Landesbibliothek Bonn, Bonn, Germany, 2016. [Google Scholar]
- Ashraf, M.T.; Fang, C.; Alassali, A.; Sowunmi, A.; Farzanah, R.; Brudecki, G.; Chaturvedi, T.; Haris, S.; Bochenski, T.; Cybulska, I.; et al. Estimation of Bioenergy Potential for Local Biomass in the United Arab Emirates. Emir. J. Food Agric. 2016, 28, 99. [Google Scholar] [CrossRef]
- Piwowar, A. Agricultural Biogas—An Important Element in the Circular and Low-Carbon Development in Poland. Energies 2020, 13, 1733. [Google Scholar] [CrossRef]
- Rosén, T.; Ödlund, L. System Perspective on Biogas Use for Transport and Electricity Production. Energies 2019, 12, 4159. [Google Scholar] [CrossRef]
- Gómez, D.; Ramos-Suárez, J.L.; Fernández, B.; Muñoz, E.; Tey, L.; Romero-Güiza, M.; Hansen, F. Development of a Modified Plug-Flow Anaerobic Digester for Biogas Production from Animal Manures. Energies 2019, 12, 2628. [Google Scholar] [CrossRef]
- Theuerl, S.; Herrmann, C.; Heiermann, M.; Grundmann, P.; Landwehr, N.; Kreidenweis, U.; Prochnow, A. The Future Agricultural Biogas Plant in Germany: A Vision. Energies 2019, 12, 396. [Google Scholar] [CrossRef]
- Sarker, S.; Lamb, J.J.; Hjelme, D.R.; Lien, K.M. A Review of the Role of Critical Parameters in the Design and Operation of Biogas Production Plants. Appl. Sci. 2019, 9, 1915. [Google Scholar] [CrossRef]
- Daniel-Gromke, J.; Rensberg, N.; Denysenko, V.; Stinner, W.; Schmalfuß, T.; Scheftelowitz, M.; Nelles, M.; Liebetrau, J. Current Developments in Production and Utilization of Biogas and Biomethane in Germany. Chem. Ing. Tech. 2017, 90, 17–35. [Google Scholar] [CrossRef]
- Gemmeke, B.; Rieger, C.; Weiland, P.; Schröder, J. Biogas-Messprogramm II, 61 Biogasanlagen im Vergleich; Fachagentur Nachwachsende Rohstoffe E.V. (FNR): Gülzow, Germany, 2009. [Google Scholar]
- Stolze, Y.; Bremges, A.; Maus, I.; Pühler, A.; Sczyrba, A.; Schlüter, A. Targeted in situ metatranscriptomics for selected taxa from mesophilic and thermophilic biogas plants. Microb. Biotechnol. 2017, 11, 667–679. [Google Scholar] [CrossRef]
- Annibaldi, V.; Cucchiella, F.; Gastaldi, M.; Rotilio, M.; Stornelli, V. Sustainability of Biogas Based Projects: Technical and Economic Analysis. In Proceedings of the E3S Web of Conferences, Kitahiroshima, Japan, 27–29 August 2018; EDP Sciences: Les Ulis, France, 2019; Volume 93, p. 03001. [Google Scholar]
- Heerenklage, J.; Rechtenbach, D.; Atamaniuk, I.; Alassali, A.; Raga, R.; Koch, K.; Kuchta, K. Development of a method to produce standardised and storable inocula for biomethane potential tests—Preliminary steps. Renew. Energy 2019, 143, 753–761. [Google Scholar] [CrossRef]
- Al-Addous, M.; Saidan, M.N.; Bdour, M.; Alnaief, M. Evaluation of Biogas Production from the Co-Digestion of Municipal Food Waste and Wastewater Sludge at Refugee Camps Using an Automated Methane Potential Test System. Energies 2018, 12, 32. [Google Scholar] [CrossRef]
- Pavi, S.; Kramer, L.E.; Gomes, L.P.; Miranda, L.A.S. Biogas production from co-digestion of organic fraction of municipal solid waste and fruit and vegetable waste. Bioresour. Technol. 2017, 228, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Campuzano, R.; González-Martínez, S. Characteristics of the organic fraction of municipal solid waste and methane production: A review. Waste Manag. 2016, 54, 3–12. [Google Scholar] [CrossRef]
- Li, W.; Loh, K.-C.; Zhang, J.; Tong, Y.W.; Dai, Y. Two-stage anaerobic digestion of food waste and horticultural waste in high-solid system. Appl. Energy 2018, 209, 400–408. [Google Scholar] [CrossRef]
- Chandra, R.; Takeuchi, H.; Hasegawa, T. Methane production from lignocellulosic agricultural crop wastes: A review in context to second generation of biofuel production. Renew. Sustain. Energy Rev. 2012, 16, 1462–1476. [Google Scholar] [CrossRef]
- Yang, Y.-Q.; Shen, D.-S.; Li, N.; Xu, N.; Long, Y.; Lu, X.-Y. Co-digestion of kitchen waste and fruit–vegetable waste by two-phase anaerobic digestion. Environ. Sci. Pollut. Res. 2013, 20, 2162–2171. [Google Scholar] [CrossRef]
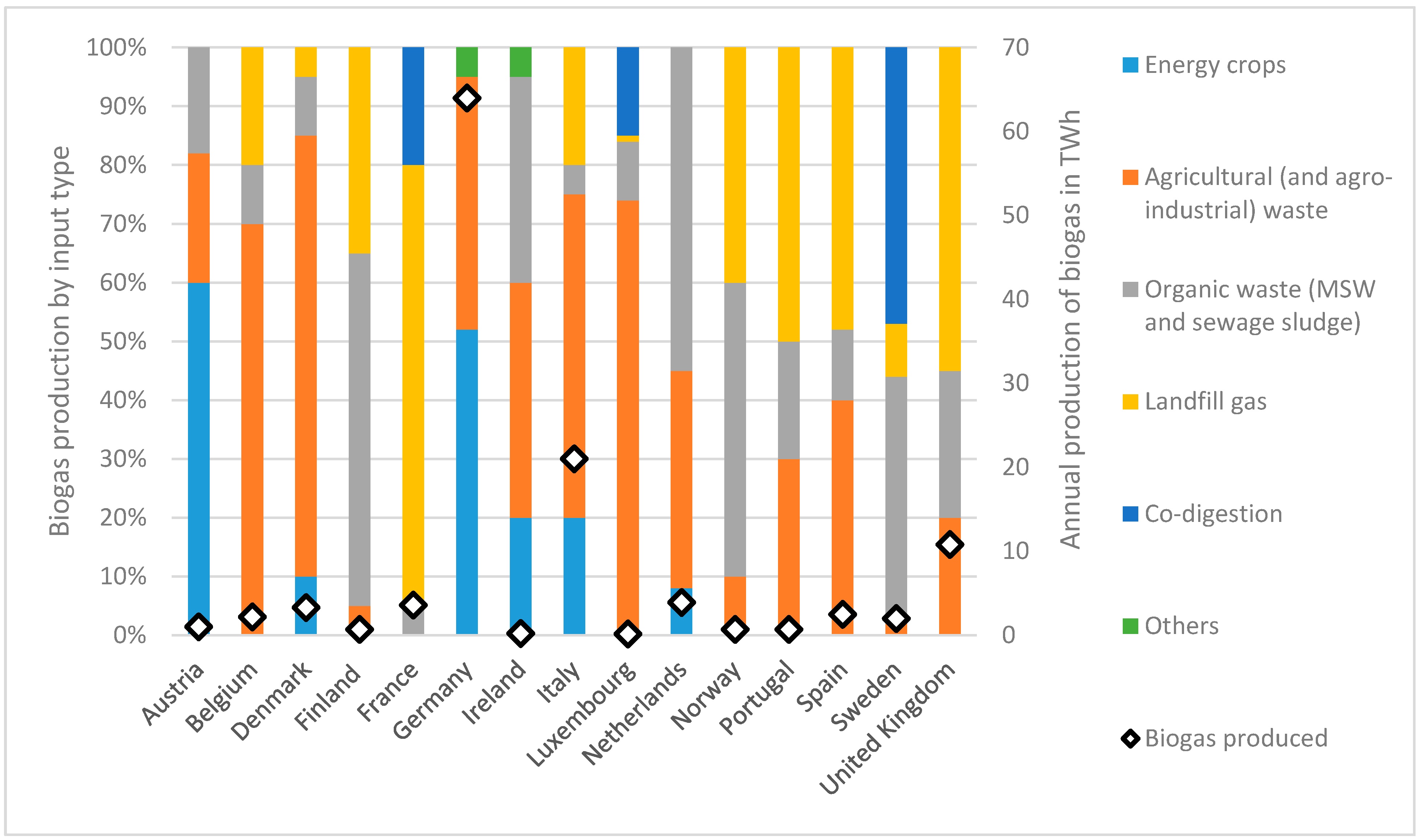
- Scarlat, N.; Dallemand, J.-F.; Fahl, F. Biogas: Developments and perspectives in Europe. Renew. Energy 2018, 129, 457–472. [Google Scholar] [CrossRef]
- Geerolf, L. The Biogas Sector Development: Current and Future Trends in Western and Northern Europe. In Master of Science; KTH School of Industrial Engineering and Management: Stockholm, Sweden, 2018. [Google Scholar]
- Tixier, N.; Guibaud, G.; Baudu, M. Determination of some rheological parameters for the characterization of activated sludge. Bioresour. Technol. 2003, 90, 215–220. [Google Scholar] [CrossRef]
- Björn, A.; de la Monja, P.S.; Karlsson, A.; Ejlertsson, J.; Svensson, B.H. Rheological characterization. In Biogas; IntechOpen: London, UK, 2012. [Google Scholar]
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2009, 85, 849–860. [Google Scholar] [CrossRef]
- Ganidi, N.; Tyrrel, S.; Cartmell, E. Anaerobic digestion foaming causes—A review. Bioresour. Technol. 2009, 100, 5546–5554. [Google Scholar] [CrossRef]
- Moeller, L.; Goersch, K.; Neuhaus, J.; Zehnsdorf, A.; Mueller, R.A. Comparative review of foam formation in biogas plants and ruminant bloat. Energy Sustain. Soc. 2012, 2, 12. [Google Scholar] [CrossRef]
- Li, Y.; Park, S.Y.; Zhu, J. Solid-state anaerobic digestion for methane production from organic waste. Renew. Sustain. Energy Rev. 2011, 15, 821–826. [Google Scholar] [CrossRef]
- Wang, X.; Yang, G.; Feng, Y.; Ren, G.; Han, X. Optimizing feeding composition and carbon–nitrogen ratios for improved methane yield during anaerobic co-digestion of dairy, chicken manure and wheat straw. Bioresour. Technol. 2012, 120, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Asam, Z.-U.-Z.; Poulsen, T.G.; Nizami, A.-S.; Rafique, R.; Kiely, G.; Murphy, J.D. How can we improve biomethane production per unit of feedstock in biogas plants? Appl. Energy 2011, 88, 2013–2018. [Google Scholar] [CrossRef]
- Vasmara, C.; Cianchetta, S.; Marchetti, R.; Galletti, S. Biogas production from wheat straw pre-treated with ligninolytic fungi and co-digestion with pig slurry. Environ. Eng. Manag. J. 2015, 14, 1751–1760. [Google Scholar] [CrossRef]
- Nsair, A.; Cinar, S.Ö.; Abu-Qdais, H.; Kuchta, K. Optimizing the performance of a large-scale biogas plant by controlling stirring process: A case study. Energy Convers. Manag. 2019, 198, 111931. [Google Scholar] [CrossRef]
- Nsair, A. Improving the performance of biogas systems. In Case Study: Applying Enhanced Stirring Strategies, 51st ed.; Abfall Aktuell: Hamburg, Germany, 2020. [Google Scholar]
- Al-Addous, M.; Alnaief, M.; Class, C.; Nsair, A.; Kuchta, K.; Alkasrawi, M. Technical possibilities of biogas production from Olive and Date Waste in Jordan. BioResources 2017, 12, 9383–9395. [Google Scholar]
- Kaparaju, P.; Serrano, M.; Thomsen, A.B.; Kongjan, P.; Angelidaki, I. Bioethanol, biohydrogen and biogas production from wheat straw in a biorefinery concept. Bioresour. Technol. 2009, 100, 2562–2568. [Google Scholar] [CrossRef]
- Risberg, K.; Sun, L.; Levén, L.; Horn, S.J.; Schnürer, A. Biogas production from wheat straw and manure—Impact of pretreatment and process operating parameters. Bioresour. Technol. 2013, 149, 232–237. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, R.; Chen, C.; Liu, G.; He, Y.; Liu, X. Biogas production from co-digestion of corn stover and chicken manure under anaerobic wet, hemi-solid, and solid-state conditions. Bioresour. Technol. 2013, 149, 406–412. [Google Scholar] [CrossRef]
- Fantozzi, F.; Buratti, C. Biogas production from different substrates in an experimental Continuously Stirred Tank Reactor anaerobic digester. Bioresour. Technol. 2009, 100, 5783–5789. [Google Scholar] [CrossRef] [PubMed]
- Hills, D.J. Effects of carbon: Nitrogen ratio on anaerobic digestion of dairy manure. Agric. Wastes 1979, 1, 267–278. [Google Scholar] [CrossRef]
- Yuan, Y.; Bian, A.; Zhang, L.; Chen, T.; Pan, M.; He, L.; Wang, A.; Ding, C. A combined process for efficient biomethane production from corn straw and cattle manure: Optimizing C/N Ratio of mixed hydrolysates. BioResources 2019, 14, 1347–1363. [Google Scholar]
- Zhang, Z.; Zhang, G.; Li, W.; Li, C.; Xu, G. Enhanced biogas production from sorghum stem by co-digestion with cow manure. Int. J. Hydrogen Energy 2016, 41, 9153–9158. [Google Scholar] [CrossRef]
- Zahan, Z.; Othman, M.Z.; Muster, T. Anaerobic digestion/co-digestion kinetic potentials of different agro-industrial wastes: A comparative batch study for C/N optimisation. Waste Manag. 2018, 71, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Song, Z.; Li, D.; Yuan, Y.; Liu, X.; Zheng, T. The effects of initial substrate concentration, C/N ratio, and temperature on solid-state anaerobic digestion from composting rice straw. Bioresour. Technol. 2015, 177, 266–273. [Google Scholar] [CrossRef]
- Riya, S.; Suzuki, K.; Terada, A.; Hosomi, M.; Zhou, S. Influence of C/N Ratio on Performance and Microbial Community Structure of Dry-Thermophilic Anaerobic Co-Digestion of Swine Manure and Rice Straw. J. Med. Bioeng. 2016, 5, 11–14. [Google Scholar] [CrossRef][Green Version]
- Fernandez-Bayo, J.D.; Yazdani, R.; Simmons, C.W.; Vander-Gheynst, J.S. Comparison of thermophilic anaerobic and aerobic treatment processes for stabilization of green and food wastes and production of soil amendments. Waste Manag. 2018, 77, 555–564. [Google Scholar] [CrossRef]
- Dębowski, M.; Kisielewska, M.; Kazimierowicz, J.; Rudnicka, A.; Dudek, M.; Romanowska-Duda, Z.; Zieliński, M. The effects of Microalgae Biomass Co-Substrate on Biogas Production from the Common Agricultural Biogas Plants Feedstock. Energies 2020, 13, 2186. [Google Scholar] [CrossRef]
- Guarino, G.; Carotenuto, C.; di Cristofaro, F.; Papa, S.; Morrone, B.; Minale, M. Does the C/N ratio really affect the Bio-methane Yield? A three years investigation of Buffalo Manure Digestion. Chem. Eng. Trans. 2016, 49, 463–468. [Google Scholar]
- Abu-Qdais, H.; Bani-Hani, K.A.; Shatnawi, N. Modeling and optimization of biogas production from a waste digester using artificial neural network and genetic algorithm. Resour. Conserv. Recycl. 2010, 54, 359–363. [Google Scholar] [CrossRef]
- Ganesh, R.; Torrijos, M.; Sousbie, P.; Lugardon, A.; Steyer, J.P.; Delgenes, J.P. Single-phase and two-phase anaerobic digestion of fruit and vegetable waste: Comparison of start-up, reactor stability and process performance. Waste Manag. 2014, 34, 875–885. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.J.; Hobbs, P.J.; Holliman, P.J.; Jones, D.L. Optimisation of the anaerobic digestion of agricultural resources. Bioresour. Technol. 2008, 99, 7928–7940. [Google Scholar] [CrossRef] [PubMed]
- Demirel, B.; Yenigun, O. Two-phase anaerobic digestion processes: A review. J. Chem. Technol. Biotechnol. 2002, 77, 743–755. [Google Scholar] [CrossRef]
- Mata-Alvarez, J. Biomethanization of the Organic Fraction of Municipal Solid Wastes; IWA publishing: London, UK, 2002. [Google Scholar]
- Voelklein, M.; Jacob, A.; Shea, R.O.; Murphy, J.D. Assessment of increasing loading rate on two-stage digestion of food waste. Bioresour. Technol. 2016, 202, 172–180. [Google Scholar] [CrossRef]
- Wu, L.-J.; Kobayashi, T.; Li, Y.-Y.; Xu, K.-Q. Comparison of single-stage and temperature-phased two-stage anaerobic digestion of oily food waste. Energy Convers. Manag. 2015, 106, 1174–1182. [Google Scholar] [CrossRef]
- Xu, F.; Li, Y.; Ge, X.; Yang, L.; Li, Y. Anaerobic digestion of food waste—Challenges and opportunities. Bioresour. Technol. 2018, 247, 1047–1058. [Google Scholar] [CrossRef]
- Bouallagui, H.; Touhami, Y.; Ben-Cheikh, R.; Hamdi, M. Bioreactor performance in anaerobic digestion of fruit and vegetable wastes. Process Biochem. 2005, 40, 989–995. [Google Scholar] [CrossRef]
- Liu, D.; Liu, D.; Zeng, R.J.; Angelidaki, I. Hydrogen and methane production from household solid waste in the two-stage fermentation process. Water Res. 2006, 40, 2230–2236. [Google Scholar] [CrossRef]
- Nielsen, H.B.; Mladenovska, Z.; Westermann, P.; Ahring, B. Comparison of two-stage thermophilic (68 °C/55 °C) anaerobic digestion with one-stage thermophilic (55 °C) digestion of cattle manure. Biotechnol. Bioeng. 2004, 86, 291–300. [Google Scholar] [CrossRef]
- Zhang, J.; Loh, K.-C.; Li, W.; Lim, J.W.; Dai, Y.; Tong, Y.W. Three-stage anaerobic digester for food waste. Appl. Energy 2017, 194, 287–295. [Google Scholar] [CrossRef]
- de Gioannis, G.; Muntoni, A.; Polettini, A.; Pomi, R.; Spiga, D. Energy recovery from one- and two-stage anaerobic digestion of food waste. Waste Manag. 2017, 68, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Drosg, B. Process Monitoring in Biogas Plants; IEA Bioenergy Paris: Paris, France, 2013. [Google Scholar]
- Adekunle, K.F.; Okolie, J.A. A Review of Biochemical Process of Anaerobic Digestion. Adv. Biosci. Biotechnol. 2015, 6, 205–212. [Google Scholar] [CrossRef]
- Jabłoński, S.; Rodowicz, P.; Łukaszewicz, M.; Ski, S.J.J.O.; Lukaszewicz, M. Methanogenic archaea database containing physiological and biochemical characteristics. Int. J. Syst. Evol. Microbiol. 2015, 65, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Mondal, C.; Biswas, G.K. Effect of Temperature on Kinetic Constants in Anaerobic Bio-digestion. Chitkara Chem. Rev. 2013, 1, 19–28. [Google Scholar] [CrossRef]
- Hans, B. Enzyme Kinetics Principles and Methods; Wiley Vch Valag: Weinheim, Germany, 2008. [Google Scholar]
- Caballero-Arzápalo, N. Untersuchungen zum Anaeroben Abbauprozess Ausgewählter Abfallsubstrate mit Hilfe Spezieller Mikroorganismen und Enzyme; Technische Universität München: Munich, Germany, 2015. [Google Scholar]
- Streitwieser, D.A. Comparison of the anaerobic digestion at the mesophilic and thermophilic temperature regime of organic wastes from the agribusiness. Bioresour. Technol. 2017, 241, 985–992. [Google Scholar] [CrossRef]
- Pandey, P.K.; Soupir, M.L. Impacts of Temperatures on Biogas Production in Dairy Manure Anaerobic Digestion. Int. J. Eng. Technol. 2012, 4, 629–631. [Google Scholar] [CrossRef]
- Zhang, J.-S.; Sun, K.-W.; Wu, M.-C.; Zhang, L. Influence of temperature on performance of anaerobic digestion of municipal solid waste. J. Environ. Sci. 2006, 18, 810–815. [Google Scholar]
- Hamzah, M.A.F.; Jahim, J.M.; Abdul, P.M.; Asis, A.J. Investigation of Temperature Effect on Start-Up Operation from Anaerobic Digestion of Acidified Palm Oil Mill Effluent. Energies 2019, 12, 2473. [Google Scholar] [CrossRef]
- Rohstoffe, F.N. Leitfaden Biogas: Von der Gewinnung zur Nutzung; Fachagentur Nachwachsende Rohstoffe E.V. (FNR): Gülzow-Prüzen, Germany, 2016; pp. 156–157. [Google Scholar]
- Wu, M.-C.; Sun, K.-W.; Zhang, Y. Influence of temperature fluctuation on thermophilic anaerobic digestion of municipal organic solid waste. J. Zhejiang Univ. Sci. B 2006, 7, 180–185. [Google Scholar] [CrossRef]
- el Mashad, H.M. Effect of temperature and temperature fluctuation on thermophilic anaerobic digestion of cattle manure. Bioresour. Technol. 2004, 95, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Ahring, B.K.; Sandberg, M.; Angelidaki, I. Volatile fatty acids as indicators of process imbalance in anaerobic digestors. Appl. Microbiol. Biotechnol. 1995, 43, 559–565. [Google Scholar] [CrossRef]
- Ahring, B.K.; Ibrahim, A.A.; Mladenovska, Z. Effect of temperature increase from 55 to 65 °C on performance and microbial population dynamics of an anaerobic reactor treating cattle manure. Water Res. 2001, 35, 2446–2452. [Google Scholar] [CrossRef]
- Pap, B.; Györkei, A.; Boboescu, I.Z.; Nagy, I.K.; Bíró, T.; Kondorosi, E.; Maróti, G. Temperature-dependent transformation of biogas-producing microbial communities’ points to the increased importance of hydrogenotrophic methanogenesis under thermophilic operation. Bioresour. Technol. 2015, 177, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Chae, K.-J.; Jang, A.; Yim, S.; Kim, I.S. The effects of digestion temperature and temperature shock on the biogas yields from the mesophilic anaerobic digestion of swine manure. Bioresour. Technol. 2008, 99, 1–6. [Google Scholar] [CrossRef]
- Kim, M.-S.; Kim, D.-H.; Yun, Y.-M. Effect of operation temperature on anaerobic digestion of food waste: Performance and microbial analysis. Fuel 2017, 209, 598–605. [Google Scholar] [CrossRef]
- Boušková, A.; Dohányos, M.; Schmidt, J.E.; Angelidaki, I. Strategies for changing temperature from mesophilic to thermophilic conditions in anaerobic CSTR reactors treating sewage sludge. Water Res. 2005, 39, 1481–1488. [Google Scholar] [CrossRef]
- Chachkhiani, M.; Dabert, P.; Abzianidze, T.; Partskhaladze, G.; Tsiklauri, L.; Dudauri, T.; Godon, J.-J. 16S rDNA characterisation of bacterial and archaeal communities during start-up of anaerobic thermophilic digestion of cattle manure. Bioresour. Technol. 2004, 93, 227–232. [Google Scholar] [CrossRef]
- Gerber, M. Ganzheitliche Stoffliche und Energetische Modellierung des Biogasbildungsprozesses; Ruhr-Universität Bochum: Bochum, Germany, 2010. [Google Scholar]
- Wang, B. Factors that Influence the Biochemical Methane Potential (BMP) Test; Lund University: Lund, Sweden, 2016. [Google Scholar]
- Al-Seadi, T.; Rutz, D.; Prassl, H.; Köttner, M.; Finsterwalder, T.; Volk, S.; Janssen, R. Biogas Handbook; ICRISAT: Esbjerg, Denmark, 2008. [Google Scholar]
- Besgen, S. Energie-und Stoffumsetzung in Biogasanlagen-Ergebnisse Messtechnischer Untersuchungen an Landwirtschaftlichen Biogasanlagen im Rheinland; Universitäts-und Landesbibliothek Bonn: Bonn, Germany, 2005. [Google Scholar]
- Gerardi, M.H. The Microbiology of Anaerobic Digesters; Wiley: Hoboken, NJ, USA, 2003. [Google Scholar]
- Nsair, A.; Bade, O.; Kuchta, K. Development of Velocity Sensor to Optimize the Energy Yield in a Biogas Plant. Environ. Sci. Technol. 2018, 51–56. [Google Scholar]
- Karim, K.; Hoffmann, R.; Klasson, T.; Al-Dahhan, M.; Klasson, K. Anaerobic digestion of animal waste: Waste strength versus impact of mixing. Bioresour. Technol. 2005, 96, 1771–1781. [Google Scholar] [CrossRef]
- Karim, K.; Klasson, K.; Hoffmann, R.; Drescher, S.; de Paoli, D.; Al-Dahhan, M. Anaerobic digestion of animal waste: Effect of mixing. Bioresour. Technol. 2005, 96, 1607–1612. [Google Scholar] [CrossRef] [PubMed]
- Karim, K.; Varma, R.; Vesvikar, M.; Al-Dahhan, M. Flow pattern visualization of a simulated digester. Water Res. 2004, 38, 3659–3670. [Google Scholar] [CrossRef] [PubMed]
- Lemmer, A.; Naegele, H.-J.; Sondermann, J. How Efficient are Agitators in Biogas Digesters? Determination of the Efficiency of Submersible Motor Mixers and Incline Agitators by Measuring Nutrient Distribution in Full-Scale Agricultural Biogas Digesters. Energies 2013, 6, 6255–6273. [Google Scholar] [CrossRef]
- Wiedemann, L.; Conti, F.; Janus, T.; Sonnleitner, M.; Zörner, W.; Goldbrunner, M. Mixing in Biogas Digesters and Development of an Artificial Substrate for Laboratory-Scale Mixing Optimization. Chem. Eng. Technol. 2016, 40, 238–247. [Google Scholar] [CrossRef]
- Last, S. The Anaerobic Digestion Biofuels Blog. Available online: https://blog.anaerobic-digestion.com/digester-cleaning-services/ (accessed on 28 January 2019).
- Nandi, R.; Saha, C.K.; Huda, M.S.; Alam, M.M. Effect of mixing on biogas production from cowdung. Eco-Friendly Agril J. 2017, 10, 7–13. [Google Scholar]
- Kopplow, O. Maßnahmen zur Minderung des Schäumens im Faulbehälter Unter Besonderer Berücksichtigung der Klärschlammdesintegration; Inst. für Umweltingenieurwesen: Rostock, Germany, 2006. [Google Scholar]
- Westlund, A.D.; Hagland, E.; Rothman, M. Foaming in anaerobic digesters caused by Microthrix parvicella. Water Sci. Technol. 1998, 37, 51–55. [Google Scholar] [CrossRef]
- Barjenbruch, M.; Hoffmann, H.; Kopplow, O.; Tränckner, J. Minimizing of foaming in digesters by pre-treatment of the surplus-sludge. Water Sci. Technol. 2000, 42, 235–241. [Google Scholar] [CrossRef]
- Mir, M.A.; Hussain, A.; Verma, C. Design considerations and operational performance of anaerobic digester: A review. Cogent Eng. 2016, 3, 795. [Google Scholar] [CrossRef]
- Hopfner-Sixt, K.; Amon, T. Monitoring of agricultural biogas plants in Austria—Mixing technology and specific values of essential process parameters. In Proceedings of the 15th European Biomass Conference and Exhibition, Berlin, Germany, 7–11 May 2007; Springer: Berlin/Heidelberg, Germany, 2007; Volume 711, p. 17181728. [Google Scholar]
- Thorin, E.; Nordlander, E.; Lindmark, J.; Dahlquist, E.; Yan, J.; Bel-Fdhila, R. Modeling of the Biogas Production process—A Review. In Proceedings of the International Conference on Applied Energy ICAE, Suzhou, China, 5–8 July 2012. [Google Scholar]
- Black, C.; United States Environmental Protection Agency, Office of Technology Transfer. Process Design Manual for Sludge Treatment and Disposal; US Environmental Protection Agency, Technology Transfer: Washington, DC, USA, 1979.
- Karim, K.; Thoma, G.J.; Al-Dahhan, M. Gas-lift digester configuration effects on mixing effectiveness. Water Res. 2007, 41, 3051–3060. [Google Scholar] [CrossRef]
- Weiland, P. Biomass Digestion in Agriculture: A Successful Pathway for the Energy Production and Waste Treatment in Germany. Eng. Life Sci. 2006, 6, 302–309. [Google Scholar] [CrossRef]
- Bártfai, Z.; Oldal, I.; Tóth, L.; Szabó, I.; Beke, J. Conditions of using propeller stirring in biogas reactors. Hung. Agric. Eng. 2015, 5–10. [Google Scholar] [CrossRef][Green Version]
- Hashimoto, A.G. Effect of mixing duration and vacuum on methane production rate from beef cattle waste. Biotechnol. Bioeng. 1982, 24, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Gollakota, K.; Meher, K. Effect of particle size, temperature, loading rate and stirring on biogas production from castor cake (oil expelled). Boil. Wastes 1988, 24, 243–249. [Google Scholar] [CrossRef]
- Chen, T.H.; Chynoweth, P.; Biljetina, R. Anaerobic digestion of municipal solid waste in a nonmixed solids concentrating digestor. Appl. Biochem. Biotechnol. 1990, 24, 533–544. [Google Scholar] [CrossRef]
- Madamwar, D.; Patel, A.; Patel, V. Effect of temperature and retention time on methane recovery from water hyacinth-cattle dung. J. Ferment. Bioeng. 1990, 70, 340–342. [Google Scholar] [CrossRef]
- Hamdi, M. Effects of agitation and pretreatment on the batch anaerobic digestion of olive mil. Bioresour. Technol. 1991, 36, 173–178. [Google Scholar] [CrossRef]
- Nasr, F.A. Treatment, and reuse of sewage sludge. Environmentalist 1997, 17, 109–113. [Google Scholar] [CrossRef]
- Rodriguez-Andara, A.; Esteban, J.L. Kinetic study of the anaerobic digestion of the solid fraction of piggery slurries. Biomass Bioenergy 1999, 17, 435–443. [Google Scholar] [CrossRef]
- Kim, M.; Ahn, Y.-H.; Speece, R.E. Comparative process stability and efficiency of anaerobic digestion; mesophilic vs. thermophilic. Water Res. 2002, 36, 4369–4385. [Google Scholar] [CrossRef]
- Kaparaju, P.; Buendia, I.; Ellegaard, L.; Angelidakia, I. Effects of mixing on methane production during thermophilic anaerobic digestion of manure: Lab-scale and pilot-scale studies. Bioresour. Technol. 2008, 99, 4919–4928. [Google Scholar] [CrossRef]
- Rojas, C.; Fang, S.; Uhlenhut, F.; Borchert, A.; Stein, I.; Schlaak, M. Stirring and biomass starter influences the anaerobic digestion of different substrates for biogas production. Eng. Life Sci. 2010, 10, 339–347. [Google Scholar] [CrossRef]
- Chen, J.; Li, X.; Liu, Y.; Zhu, B.; Yuan, H.; Pang, Y. Effect of mixing rates on anaerobic digestion performance of rice straw. Transact. CSAE 2011, 27, 144–148. [Google Scholar]
- Ghanimeh, S.; el Fadel, M.; Saikaly, P.E. Mixing effect on thermophilic anaerobic digestion of source-sorted organic fraction of municipal solid waste. Bioresour. Technol. 2012, 117, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Keanoi, N.; Hussaro, K.; Teekasap, S. Effect of with/without agitation of agricultural waste on biogas production from anaerobic co-digestion-a small scale. Am. J. Environ. Sci. 2014, 10, 74–85. [Google Scholar] [CrossRef][Green Version]
- Lindmark, J.; Thorin, E.; Fdhila, R.B.; Dahlquist, E. Effects of mixing on the result of anaerobic digestion: Review. Renew. Sustain. Energy Rev. 2014, 40, 1030–1047. [Google Scholar] [CrossRef]
- El-Bakhshwan, M.; El-Ghafar, S.A.; Zayed, M.; El-Shazly, A. Effect of mechanical stirring on biogas production efficiency in large scale digesters. J. Soil Sci. Agric. Eng. 2015, 6, 47–63. [Google Scholar] [CrossRef][Green Version]
- Zareei, S.; Khodaei, J. Modeling and optimization of biogas production from cow manure and maize straw using an adaptive neuro-fuzzy inference system. Renew. Energy 2017, 114, 423–427. [Google Scholar] [CrossRef]
- Abdullah, N.O.; Pandebesie, E.S. The Influences of Stirring and Cow Manure Added on Biogas Production from Vegetable Waste Using Anaerobic Digester. IOP Conf. Ser. Earth Environ. Sci. 2018, 135, 012005. [Google Scholar] [CrossRef]
- Aksay, M.V.; Ozkaymak, M.; Calhan, R. Co-digestion of cattle manure and tea waste for biogas production. Int. J. Energ. Res. 2018, 8, 1246–1353. [Google Scholar]
- Babaei, A.; Shayegan, J. Effects of temperature and mixing modes on the performance of municipal solid waste anaerobic slurry digester. J. Environ. Heal. Sci. Eng. 2020, 17, 1077–1084. [Google Scholar] [CrossRef]
- Ioelovich, M. Recent findings and the energetic potential of plant biomass as a renewable source of biofuels–a review. Bio. Resour. 2015, 10, 1879–1914. [Google Scholar]
- Agrahari, R.P.; Tiwari, G.N. The Production of Biogas Using Kitchen Waste. Int. J. Energy Sci. 2013, 3, 408. [Google Scholar] [CrossRef]
- Achinas, S.; Achinas, V.; Euverink, G.J.W. A Technological Overview of Biogas Production from Biowaste. BioRixv 2017, 3, 299–307. [Google Scholar] [CrossRef]
- Jaber, J.; Probert, S.; Williams, P.T. Gaseous fuels (derived from oil shale) for heavy-duty gas turbines and combined-cycle power generators. Appl. Energy 1998, 60, 1–20. [Google Scholar] [CrossRef]
- Frey, J.; Grüssing, F.; Nägele, H.-J.; Oechsner, H. Cutting the electric power consumption of biogas plants: The impact of new technologies. Landtechnik. Agric. Eng. 2013, 68, 58–63. [Google Scholar]
- Botheju, D. Oxygen Effects in Anaerobic Digestion—A Review. Open Waste Manag. J. 2011, 4, 1–19. [Google Scholar] [CrossRef]
- Jagadabhi, P.S.; Kaparaju, P.; Rintala, J. Effect of micro-aeration and leachate replacement on COD solubilization and VFA production during mono-digestion of grass-silage in one-stage leach-bed reactors. Bioresour. Technol. 2010, 101, 2818–2824. [Google Scholar] [CrossRef]
- Jenicek, P.; Keclik, F.; Máca, J.; Bindzar, J. Use of microaerobic conditions for the improvement of anaerobic digestion of solid wastes. Water Sci. Technol. 2008, 58, 1491–1496. [Google Scholar] [CrossRef]
- Nghiem, L.; Manassa, P.; Dawson, M.; Fitzgerald, S.K. Oxidation reduction potential as a parameter to regulate micro-oxygen injection into anaerobic digester for reducing hydrogen sulphide concentration in biogas. Bioresour. Technol. 2014, 173, 443–447. [Google Scholar] [CrossRef]
- Tabatabaei, M.; Ghanavati, H. Biogas: Fundamentals, process, and operation. In Prominent Parameters in Biogas Production Systems; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Sibiya, N.T.; Muzenda, E.; Tesfagiorgis, H.B. Effect of temperature and pH on the anaerobic digestion of grass silage. In Proceedings of the 6th International Conference on Green Technology, Renewable Energy and Environmental Engineering, Cape Town, South Africa, 15–16 April 2014. [Google Scholar]
- Zhang, C.; Su, H.; Baeyens, J.; Tan, T. Reviewing the anaerobic digestion of food waste for biogas production. Renew. Sustain. Energy Rev. 2014, 38, 383–392. [Google Scholar] [CrossRef]
- Voß, E. Prozessanalyse und Optimierung von Landwirtschaftlichen Biogasanlagen. Ph.D. Thesis, Institut für Siedlungswasserwirtschaft und Abfalltechnik, Hanover, Germany, 2015. [Google Scholar]
- Mpofu, A.B.; Welz, P.J.; Oyekola, O.O. Anaerobic Digestion of Secondary Tannery Sludge: Optimisation of Initial pH and Temperature and Evaluation of Kinetics. Waste Biomass Valorizat. 2019, 11, 873–885. [Google Scholar] [CrossRef]
- Ren, Y.; Yu, M.; Wu, C.; Wang, Q.; Gao, M.; Huang, Q.; Liu, Y. A comprehensive review on food waste anaerobic digestion: Research updates and tendencies. Bioresour. Technol. 2018, 247, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Önen, S.; Nsair, A.; Kuchta, K. Innovative operational strategies for biogas plant including temperature and stirring management. Waste Manag. Res. 2018, 37, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Murto, M.; Björnsson, L.; Mattiasson, B. Impact of food industrial waste on anaerobic co-digestion of sewage sludge and pig manure. J. Environ. Manag. 2004, 70, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Hong, F.; Lü, Y.; Li, X.; Liu, H. Improved biogas production and biodegradation of oilseed rape straw by using kitchen waste and duck droppings as co-substrates in two-phase anaerobic digestion. PLoS ONE 2017, 12, e0182361. [Google Scholar] [CrossRef] [PubMed]
- Boe, K. Online Monitoring and Control of the Biogas Process. Ph.D. Thesis, Technical University of Denmark, Copenhagen, Denmark, 2006. [Google Scholar]
- Cecchi, F.; Pavan, P.; Alvarez, J.M.; Bassetti, A.; Cozzolino, C. Anaerobic digestion of municipal solid waste: Thermophilic vs. mesophilic performance at high solids. Waste Manag. Res. 1991, 9, 305–315. [Google Scholar] [CrossRef]
- Baudez, J.-C.; Markis, F.; Eshtiaghi, N.; Slatter, P. The rheological behaviour of anaerobic digested sludge. Water Res. 2011, 45, 5675–5680. [Google Scholar] [CrossRef]
- Aboudi, K.; Álvarez-Gallego, C.J.; García, L.I.R. Semi-continuous anaerobic co-digestion of sugar beet byproduct and pig manure: Effect of the organic loading rate (OLR) on process performance. Bioresour. Technol. 2015, 194, 283–290. [Google Scholar] [CrossRef]
- Dhar, H.; Kumar, P.; Kumar, S.; Mukherjee, S.; Vaidya, A.N. Effect of organic loading rate during anaerobic digestion of municipal solid waste. Bioresour. Technol. 2016, 217, 56–61. [Google Scholar] [CrossRef]
- IRENA. Bioenergy. 2020. Available online: https://www.irena.org/bioenergy (accessed on 27 April 2020).
- Statista GmbH. Installierte Elektrische Leistung der Biogasanlagen in Deutschland in den Jahren 1999 bis 2019. 2020. Available online: https://de.statista.com/statistik/daten/studie/167673/umfrage/installierte-elektrische-leistung-von-biogasanlagen-seit-1999/ (accessed on 27 April 2020).
- Chiumenti, A.; da Borso, F.; Limina, S. Dry anaerobic digestion of cow manure and agricultural products in a full-scale plant: Efficiency and comparison with wet fermentation. Waste Manag. 2018, 71, 704–710. [Google Scholar] [CrossRef]
- Li, N.; Liu, S.; Mi, L.; Li, Z.; Yuan, Y.; Yan, Z.; Liu, X. Effects of feedstock ratio and organic loading rate on the anaerobic mesophilic co-digestion of rice straw and pig manure. Bioresour. Technol. 2015, 187, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.-T.; Fan, X.-L.; Zhao, X.-X.; Fu, S.; He, S.; Manasa, M.; Guo, R.-B. Effects of organic loading rate on biogas production from macroalgae: Performance and microbial community structure. Bioresour. Technol. 2017, 235, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Montingelli, M.; Tedesco, S.; Olabi, A.G. Biogas production from algal biomass: A review. Renew. Sustain. Energy Rev. 2015, 43, 961–972. [Google Scholar] [CrossRef]
- González-Fernández, C.; Sialve, B.; Bernet, N.; Steyer, J.-P. Effect of organic loading rate on anaerobic digestion of thermally pretreated Scenedesmus sp. biomass. Bioresour. Technol. 2013, 129, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Z.; Wu, S.; Zhang, W.; Dong, R. Effects of organic loading rate and effluent recirculation on the performance of two-stage anaerobic digestion of vegetable waste. Bioresour. Technol. 2013, 146, 556–561. [Google Scholar] [CrossRef]
- Liu, X.; Wang, W.; Shi, Y.; Zheng, L.; Gao, X.; Qiao, W.; Zhou, Y. Pilot-scale anaerobic co-digestion of municipal biomass waste and waste activated sludge in China: Effect of organic loading rate. Waste Manag. 2012, 32, 2056–2060. [Google Scholar] [CrossRef]
- Mähnert, P.; Linke, B. Kinetic study of biogas production from energy crops and animal waste slurry: Effect of organic loading rate and reactor size. Environ. Technol. 2009, 30, 93–99. [Google Scholar] [CrossRef]
- Luste, S.; Luostarinen, S. Anaerobic co-digestion of meat-processing by-products and sewage sludge—Effect of hygienization and organic loading rate. Bioresour. Technol. 2010, 101, 2657–2664. [Google Scholar] [CrossRef]
- Zhou, J.; Yang, J.; Yu, Q.; Yong, X.; Xie, X.; Zhang, L.; Wei, P.; Jia, H. Different organic loading rates on the biogas production during the anaerobic digestion of rice straw: A pilot study. Bioresour. Technol. 2017, 244, 865–871. [Google Scholar] [CrossRef]
- Nagao, N.; Tajima, N.; Kawai, M.; Niwa, C.; Kurosawa, N.; Matsuyama, T.; Yusoff, F.M.; Toda, T. Maximum organic loading rate for the single-stage wet anaerobic digestion of food waste. Bioresour. Technol. 2012, 118, 210–218. [Google Scholar] [CrossRef]
- Song, H.; Zhang, Y.; Kusch-Brandt, S.; Banks, C. Comparison of Variable and Constant Loading for Mesophilic Food Waste Digestion in a Long-Term Experiment. Energies 2020, 13, 1279. [Google Scholar] [CrossRef]
- Ezekoye, V.A.; Ezekoye, B.A.; Offor, P.O. Effect of retention time on biogas production from poultry droppings and cassava peels. Niger. J. Biotechnol. 2011, 22, 53–59. [Google Scholar]
- Li, C.; Champagne, P.; Anderson, B.C. Biogas production performance of mesophilic and thermophilic anaerobic co-digestion with fat, oil, and grease in semi-continuous flow digesters: Effects of temperature, hydraulic retention time, and organic loading rate. Environ. Technol. 2013, 34, 2125–2133. [Google Scholar] [CrossRef] [PubMed]
- Kaosol, T.; Sohgrathok, N. Influence of Hydraulic Retention Time on Biogas Production from Frozen Seafood Wastewater Using Decanter Cake as Anaerobic Co-digestion Material. Int. J. Environ. Eng. 2012, 20. [Google Scholar]
- Dareioti, M.A.; Kornaros, M. Anaerobic mesophilic co-digestion of ensiled sorghum, cheese whey and liquid cow manure in a two-stage CSTR system: Effect of hydraulic retention time. Bioresour. Technol. 2015, 175, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Dareioti, M.A.; Kornaros, M. Effect of hydraulic retention time (HRT) on the anaerobic co-digestion of agro-industrial wastes in a two-stage CSTR system. Bioresour. Technol. 2014, 167, 407–415. [Google Scholar] [CrossRef]
- Schmidt, T.; Ziganshin, A.M.; Nikolausz, M.; Scholwin, F.; Nelles, M.; Kleinsteuber, S.; Pröter, J. Effects of the reduction of the hydraulic retention time to 1.5 days at constant organic loading in CSTR, ASBR, and fixed-bed reactors—Performance and methanogenic community composition. Biomass Bioenergy 2014, 69, 241–248. [Google Scholar] [CrossRef]
- Shi, X.-S.; Dong, J.-J.; Yu, J.-H.; Yin, H.; Hu, S.-M.; Huang, S.-X.; Yuan, X.-Z. Effect of Hydraulic Retention Time on Anaerobic Digestion of Wheat Straw in the Semicontinuous Continuous Stirred-Tank Reactors. BioMed Res. Int. 2017, 1–6. [Google Scholar] [CrossRef]
- Krakat, N.; Schmidt, S.; Scherer, P. Mesophilic Fermentation of Renewable Biomass: Does Hydraulic Retention Time Regulate Methanogen Diversity? Appl. Environ. Microbiol. 2010, 76, 6322–6326. [Google Scholar] [CrossRef]
- Vintiloiu, A.; Lemmer, A.; Oechsner, H.; Jungbluth, T. Mineral substances and macronutrients in the anaerobic conversion of biomass: An impact evaluation. Eng. Life Sci. 2012, 12, 287–294. [Google Scholar] [CrossRef]
- Sibiya, N.T.; Tesfagiorgis, H.B.; Muzenda, E. Influence of nutrients addition for enhanced biogas production from energy crops: A review. Magnesium 2015, 1, 1–5. [Google Scholar]
- Demirel, B.; Scherer, P. Trace element requirements of agricultural biogas digesters during biological conversion of renewable biomass to methane. Biomass Bioenergy 2011, 35, 992–998. [Google Scholar] [CrossRef]
- Bougrier, C.; Dognin, D.; Laroche, C.; Gonzalez, V.; Benali-Raclot, D.; Rivero, J.A.C. Anaerobic digestion of Brewery Spent Grains: Trace elements addition requirement. Bioresour. Technol. 2018, 247, 1193–1196. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cheng, J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef] [PubMed]
- Kayhanian, M. Ammonia Inhibition in High-Solids Biogasification: An Overview and Practical Solutions. Environ. Technol. 1999, 20, 355–365. [Google Scholar] [CrossRef]
- Yenigun, O.; Demirel, B. Ammonia inhibition in anaerobic digestion: A review. Process. Biochem. 2013, 48, 901–911. [Google Scholar] [CrossRef]
- Chen, J.L.; Ortiz, R.; Steele, T.W.; Stuckey, D.C. Toxicants inhibiting anaerobic digestion: A review. Biotechnol. Adv. 2014, 32, 1523–1534. [Google Scholar] [CrossRef]
- McCartney, D.; Oleszkiewicz, J. Sulfide inhibition of anaerobic degradation of lactate and acetate. Water Res. 1991, 25, 203–209. [Google Scholar] [CrossRef]
- Fagbohungbe, M.O.; Herbert, B.M.; Hurst, L.; Ibeto, C.N.; Li, H.; Usmani, S.Q.; Semple, K.T. The challenges of anaerobic digestion and the role of biochar in optimizing anaerobic digestion. Waste Manag. 2017, 61, 236–249. [Google Scholar] [CrossRef]
- Thiele, J.H.; Wu, W.-M.; Jain, M.K.; Zeikus, J.G. Ecoengineering high rate anaerobic digestion systems: Analysis of improved syntrophic biomethanation catalysts. Biotechnol. Bioeng. 1990, 35, 990–999. [Google Scholar] [CrossRef]
- van Langerak, E.; Gonzalez-Gil, G.; van Aelst, A.; van Lier, J.; Hamelers, H.; Lettinga, G. Effects of high calcium concentrations on the development of methanogenic sludge in upflow anaerobic sludge bed (UASB) reactors. Water Res. 1998, 32, 1255–1263. [Google Scholar] [CrossRef]
- Dimroth, P.; Thomer, A. A primary respiratory Na+ pump of an anaerobic bacterium: The Na+-dependent NADH: Quinone oxidoreductase of Klebsiella pneumoniae. Arch. Microbiol. 1989, 151, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Cabirol, N.; Barragán, E.; Durán, A.; Noyola, A. Effect of aluminium and sulphate on anaerobic digestion of sludge from wastewater enhanced primary treatment. Water Sci. Technol. 2003, 48, 235–240. [Google Scholar] [CrossRef]
- Urriza-Arsuaga, I.; Bedoya, M.; Orellana, G. Tailored luminescent sensing of NH3 in biomethane productions. Sens. Actuators B Chem. 2019, 292, 210–216. [Google Scholar] [CrossRef]
- Romero-Güiza, M.; Vila, J.; Mata-Alvarez, J.; Simon, F.-G.; Astals, S. The role of additives on anaerobic digestion: A review. Renew. Sustain. Energy Rev. 2016, 58, 1486–1499. [Google Scholar] [CrossRef]
- Romero-Güiza, M.; Astals, S.; Mata-Alvarez, J.; Simon, F.-G. Feasibility of coupling anaerobic digestion and struvite precipitation in the same reactor: Evaluation of different magnesium sources. Chem. Eng. J. 2015, 270, 542–548. [Google Scholar] [CrossRef]
- Schattauer, A.; Abdoun, E.; Weiland, P.; Plöchl, M.; Heiermann, M. Abundance of trace elements in demonstration biogas plants. Biosyst. Eng. 2011, 108, 57–65. [Google Scholar] [CrossRef]
- Lo, H.; Chiang, C.; Tsao, H.; Pai, T.; Liu, M.; Kurniawan, T.; Chao, K.; Liou, C.; Lin, K.; Chang, C.; et al. Effects of spiked metals on the MSW anaerobic digestion. Waste Manag. Res. 2012, 30, 32–48. [Google Scholar] [CrossRef]
- Lo, H.-M.; Chiu, H.; Lo, S.; Lo, F. Effects of different SRT on anaerobic digestion of MSW dosed with various MSWI ashes. Bioresour. Technol. 2012, 125, 233–238. [Google Scholar] [CrossRef]
- Guo, Q.; Majeed, S.; Xu, R.; Zhang, K.; Kakade, A.; Khan, A.; Hafeez, F.Y.; Mao, C.; Liu, P.; Li, X. Heavy metals interact with the microbial community and affect biogas production in anaerobic digestion: A review. J. Environ. Manag. 2019, 240, 266–272. [Google Scholar] [CrossRef]
- Mueller, R.F.; Steiner, A. Inhibition of Anaerobic Digestion Caused by Heavy Metals. Water Sci. Technol. 1992, 26, 835–846. [Google Scholar] [CrossRef]
- Altaş, L. Inhibitory effect of heavy metals on methane-producing anaerobic granular sludge. J. Hazard. Mater. 2009, 162, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Shafy, H.I.; Mansour, M. Biogas production as affected by heavy metals in the anaerobic digestion of sludge. Egypt. J. Pet. 2014, 23, 409–417. [Google Scholar] [CrossRef]
- Li, C.; Fang, H.H. Inhibition of heavy metals on fermentative hydrogen production by granular sludge. Chemosphere 2007, 67, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Gagliano, M.C.; Sudmalis, D.; Temmink, H.; Plugge, C.M. Calcium effect on microbial activity and biomass aggregation during anaerobic digestion at high salinity. New Biotechnol. 2020, 56, 114–122. [Google Scholar] [CrossRef]
- Yuan, Z.; Yang, H.; Zhi, X.; Shena, J. Increased performance of continuous stirred tank reactor with calcium supplementation. Int. J. Hydrogen Energy 2010, 35, 2622–2626. [Google Scholar] [CrossRef]
- Tan, L.; Qu, Y.; Zhou, J.; Ma, F.; Li, A. Dynamics of microbial community for X-3B wastewater decolorization coping with high-salt and metal ions conditions. Bioresour. Technol. 2009, 100, 3003–3009. [Google Scholar] [CrossRef]
- Lin, C. Heavy metal effects on fermentative hydrogen production using natural mixed microflora. Int. J. Hydrogen Energy 2008, 33, 587–593. [Google Scholar] [CrossRef]
- Fermoso, F.G.; Bartacek, J.; Jansen, S.; Lens, P.N. Metal supplementation to UASB bioreactors: From cell-metal interactions to full-scale application. Sci. Total. Environ. 2009, 407, 3652–3667. [Google Scholar] [CrossRef]
- Gikas, P. Kinetic responses of activated sludge to individual and joint nickel (Ni (II)) and cobalt (Co (II)): An isobolographic approach. J. Hazard. Mater. 2007, 143, 246–256. [Google Scholar] [CrossRef]
- Kida, K.; Shigematsu, T.; Kijima, J.; Numaguchi, M.; Mochinaga, Y.; Abe, N.; Morimura, S. Influence of Ni2+ and Co2+ on Methanogenic Activity and the Amounts of Coenzymes Involved in Methanogenesis. J. Biosci. Bioeng. 2001, 91, 590–595. [Google Scholar] [CrossRef]
- Ma, J.; Mungoni, L.J.; Verstraete, W.; Carballa, M. Maximum removal rate of propionic acid as a sole carbon source in UASB reactors and the importance of the macro- and micro-nutrients stimulation. Bioresour. Technol. 2009, 100, 3477–3482. [Google Scholar] [CrossRef] [PubMed]
- Worm, P.; Fermoso, F.G.; Lens, P.N.; Plugge, C.M. Decreased activity of a propionate degrading community in a UASB reactor fed with synthetic medium without molybdenum, tungsten, and selenium. Enzym. Microb. Technol. 2009, 45, 139–145. [Google Scholar] [CrossRef]
- Chan, P.C.; Lu, Q.; Toledo, R.A.; Gu, J.-D.; Shim, H. Improved anaerobic co-digestion of food waste and domestic wastewater by copper supplementation—Microbial community change and enhanced effluent quality. Sci. Total Environ. 2019, 670, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.; Sparling, R.; Oleszkiewicz, J.A. VFA generation from waste activated sludge: Effect of temperature and mixing. Chemosphere 2011, 82, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Zheng, Z.; Zhao, Y.; Zhang, Y.; Guo, S.; Cui, Z.; Wang, X. Effects of molybdenum, selenium and manganese supplementation on the performance of anaerobic digestion and the characteristics of bacterial community in acidogenic stage. Bioresour. Technol. 2018, 266, 166–175. [Google Scholar] [CrossRef]
- Fang, C.; Boe, K.; Angelidaki, I. Anaerobic co-digestion of desugared molasses with cow manure; focusing on sodium and potassium inhibition. Bioresour. Technol. 2011, 102, 1005–1011. [Google Scholar] [CrossRef]
- Feijoo, G.; Soto, M.; Mendez, R.; Lema, J.M. Sodium inhibition in the anaerobic digestion process: Antagonism and adaptation phenomena. Enzym. Microb. Technol. 1995, 17, 180–188. [Google Scholar] [CrossRef]
- Stamatelatou, K.; Antonopoulou, G.; Lyberatos, G. Production of Biogas via Anaerobic Digestion; Elsevier BV: Amsterdam, The Netherlands, 2011; pp. 266–304. [Google Scholar]
- Yang, J.; Speece, R. The effects of chloroform toxicity on methane fermentation. Water Res. 1986, 20, 1273–1279. [Google Scholar] [CrossRef]
- Renard, P.; Bouillon, C.; Naveau, H.; Nyns, E.-J. Toxicity of a mixture of polychlorinated organic compounds towards an unacclimated methanogenic consortium. Biotechnol. Lett. 1993, 15, 195–200. [Google Scholar] [CrossRef]
- van Beelen, P.; van Vlaardingen, P. Toxic effects of pollutants on the mineralization of 4-chlorophenol and benzoate in methanogenic river sediment. Environ. Toxicol. Chem. 1994, 13, 1051–1060. [Google Scholar] [CrossRef]
- Sierra-Alvarez, R.; Lettinga, G. The effect of aromatic structure on the inhibition of acetoclastic methanogenesis in granular sludge. Appl. Microbiol. Biotechnol. 1991, 34, 544–550. [Google Scholar] [CrossRef]
- Soto, M.; Mendez, R.; Lema, J. Biodegradability and toxicity in the anaerobic treatment of fish canning wastewaters. Environ. Technol. 1991, 12, 669–677. [Google Scholar] [CrossRef]
- Fang, H.H.P.; Chen, T.; Chan, O.C. Toxic effects of phenolic pollutants on anaerobic benzoate-degrading granules. Biotechnol. Lett. 1995, 17, 117–120. [Google Scholar] [CrossRef]
- Shin, H.-S.; Kwon, J.-C. Degredation and interaction between organic concentrations and toxicity of 2,4,6-trichlorophenol in anaerobic system. Biotechnol. Tech. 1998, 12, 39–43. [Google Scholar] [CrossRef]
- Uberoi, V.; Bhattacharya, S.K. Toxicity, and degradability of nitrophenols in anaerobic systems. Water Environ. Res. 1997, 69, 146–156. [Google Scholar] [CrossRef]
- McCue, T.; Hoxworth, S.; Randall, A.A. Degradation of halogenated aliphatic compounds utilizing sequential anaerobic/aerobic treatments. Water Sci. Technol. 2003, 47, 79–84. [Google Scholar] [CrossRef]
- Mormile, M.R.; Suflita, J.M. The Toxicity of Selected Gasoline Components to Glucose Methanogenesis by Aquifer Microorganisms. Anaerobe 1996, 2, 299–303. [Google Scholar] [CrossRef]
- Stuckey, D.C.; Owen, W.F.; McCarty, P.L.; Parkin, G.F. Anaerobic toxicity evaluation by batch and semi-continuous assays. J. Water Pollut. Control Fed. 1980, 720–729. [Google Scholar]
- Demirer, G.; Speece, R. Anaerobic biotransformation of four3-carbon compounds (acrolein, acrylic acid, allyl alcohol and n-propanol) in UASB reactors. Water Res. 1998, 32, 747–759. [Google Scholar] [CrossRef]
- Gonzalez-Gil, G.; Kleerebezem, R.; Lettinga, G. Conversion and toxicity characteristics of formaldehyde in acetoclastic methanogenic sludge. Biotechnol. Bioeng. 2002, 79, 314–322. [Google Scholar] [CrossRef]
- Playne, M.J.; Smith, B.R. Toxicity of organic extraction reagents to anaerobic bacteria. Biotechnol. Bioeng. 1983, 25, 1251–1265. [Google Scholar] [CrossRef]
- Hayward, G.; Lau, I. Toxicity of organic solvents to fatty acid forming bacteria. Can. J. Chem. Eng. 1989, 67, 157–161. [Google Scholar] [CrossRef]
- Stergar, V.; Zagorc-Končan, J.; Zgajnar-Gotvanj, A. Laboratory scale and pilot plant study on treatment of toxic wastewater from the petrochemical industry by UASB reactors. Water Sci. Technol. 2003, 48, 97–102. [Google Scholar] [CrossRef]
- Liu, S.-M.; Wu, C.-H.; Huang, H.-J. Toxicity and anaerobic biodegradability of pyridine and its derivatives under sulfidogenic conditions. Chemosphere 1998, 36, 2345–2357. [Google Scholar] [CrossRef]
- Surerus, V.; Giordano, G.; Teixeira, L.A. Activated sludge inhibition capacity index. Braz. J. Chem. Eng. 2014, 31, 385–392. [Google Scholar] [CrossRef]
- Hwu, C.-S.; Lettinga, G. Acute toxicity of oleate to acetate-utilizing methanogens in mesophilic and thermophilic anaerobic sludges. Enzym. Microb. Technol. 1997, 21, 297–301. [Google Scholar] [CrossRef]
- Wikandari, R.; Gudipudi, S.; Pandiyan, I.; Millati, R.; Taherzadeh, M. Inhibitory effects of fruit flavors on methane production during anaerobic digestion. Bioresour. Technol. 2013, 145, 188–192. [Google Scholar] [CrossRef]
- Nie, Y.; Tian, X.; Zhou, Z.; Cheng, J. Impact of food to microorganism ratio and alcohol ethoxylate dosage on methane production in treatment of low-strength wastewater by a submerged anaerobic membrane bioreactor. Front. Environ. Sci. Eng. 2017, 11, 6. [Google Scholar] [CrossRef]
- Garcia-Bernet, D.; Loisel, D.; Guizard, G.; Buffiere, P.; Steyer, J.-P.; Escudie, R. Rapid measurement of the yield stress of anaerobically digested solid waste using slump tests. Waste Manag. 2011, 31, 631–635. [Google Scholar] [CrossRef]
- Bhattacharya, S.K.; Qu, M.; Madura, R.L. Effects of nitrobenzene and zinc on acetate utilizing methanogens. Water Res. 1996, 30, 3099–3105. [Google Scholar] [CrossRef]
- Blum, D.J.W.; Speece, R.E. A Database of Chemical Toxicity to Environmental Bacteria and Its Use in Interspecies Comparisons and Correlations. J. Water Pollut. Control Fed. 1991, 63, 198–207. [Google Scholar]
- Borja, R.; Alba, J.; Banks, C. Impact of the main phenolic compounds of olive mill wastewater (OMW) on the kinetics of acetoclastic methanogenesis. Process. Biochem. 1997, 32, 121–133. [Google Scholar] [CrossRef]
- Boucquey, J.-B.; Renard, P.; Amerlynck, P.; Filho, P.M.; Agathos, S.N.; Naveau, H.; Nyns, E.-J. High-rate continuous biodegradation of concentrated chlorinated aliphatics by a durable enrichment of methanogenic origin under carrier-dependent conditions. Biotechnol. Bioeng. 1995, 47, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Mahanty, B.; Zafar, M.; Han, M.J.; Park, H.-S. Optimization of co-digestion of various industrial sludges for biogas production and sludge treatment: Methane production potential experiments and modeling. Waste Manag. 2014, 34, 1018–1024. [Google Scholar] [CrossRef]
- Mohamed, S.D.Y. Influence of Oregano (Origanum vulgare L.), Fennel (Foeniculum vulgare L.) and Hop Cones (Humulus lupulus L.) on Biogas and Methane Production; Universitat Giessen: Giessen, Germany, 2014. [Google Scholar]
- Hess, J.; Bernard, O. Advanced dynamical risk analysis for monitoring anaerobic digestion process. Biotechnol. Prog. 2009, 25, 643–653. [Google Scholar] [CrossRef]
- Arthur, R.; Scherer, P. Application of total reflection X-Ray fluorescence spectrometry to quantify cobalt concentration in the presence of high iron concentration in biogas plants. Spectrosc. Lett. 2019, 53, 100–113. [Google Scholar] [CrossRef]
- Zheng, G.; Liu, J.; Shao, Z.; Chen, T. Emission characteristics and health risk assessment of VOC’s from a food waste anaerobic digestion plant: A case study of Suzhou, China. Environ. Pollut. 2019, 257, 113546. [Google Scholar] [CrossRef]
- Reinelt, T.; Liebetrau, J. Monitoring and Mitigation of Methane Emissions from Pressure Relief Valves of a Biogas Plant. Chem. Eng. Technol. 2019, 43, 7–18. [Google Scholar] [CrossRef]
- Wünscher, H.; Frank, T.; Cyriax, A.; Tobehn-Steinhäuser, I.; Ortlepp, T.; Kirner, T. Monitoring of Ammonia in Biogas. Chem. Eng. Technol. 2019, 43, 99–103. [Google Scholar] [CrossRef]
- Paul, A.; Schwind, B.; Weinberger, C.; Tiemann, M.; Wagner, T. Gas Responsive Nanoswitch: Copper Oxide Composite for Highly Selective H 2 S Detection. Adv. Funct. Mater. 2019, 29. [Google Scholar] [CrossRef]
- Kushkevych, I.; Kobzová, E.; Vítězová, M.; Vítěz, T.; Dordević, D.; Bartoš, M. Acetogenic microorganisms in operating biogas plants depending on substrate combinations. Boilogia 2019, 74, 1229–1236. [Google Scholar] [CrossRef]
- Mudaheranwa, E.; Rwigema, A.; Ntagwirumugara, E.; Masengo, G.; Singh, R.; Biziyaremye, J. Development of PLC based monitoring and control of pressure in Biogas Power Plant Digester. In Proceedings of the 2019 International Conference on Advances in Big Data, Computing and Data Communication Systems (icABCD), Winterton, South Africa, 5–6 August 2019; Institute of Electrical and Electronics Engineers (IEEE): Piscataway, NJ, USA, 2019; pp. 1–7. [Google Scholar]
- Logan, M.; Safi, M.; Lens, P.; Visvanathan, C. Investigating the performance of internet of things based anaerobic digestion of food waste. Process. Saf. Environ. Prot. 2019, 127, 277–287. [Google Scholar] [CrossRef]
- Selvaraj, R.; Vasa, N.J.; Nagendra, S.M.S. Off-Resonant Broadband Photoacoustic Spectroscopy for Online Monitoring of Biogas Concentration with a Wide Dynamic Range. In Proceedings of the Conference on Lasers and Electro-Optics, San Jose, CA, USA, 5–10 May 2019; The Optical Society: Washington, DC, USA, 2019; p. JW2A.20. [Google Scholar]
- Flexibierung von Biogasanlagen. Federal Ministry of Food, Agriculture and Consumer Protection; Fachagentur Nachwachsende Rohstoffe E.V. (FNR): Gülzow, Germany, 2018. [Google Scholar]
| Substrate | DM % | oDM % (In DM) | Biogas Yield NL kg−1 FM | Methane Content NL kg−1 FM | Electricity Produced kWh t−1 FM | References |
|---|---|---|---|---|---|---|
| Pig slurry | 4–19 | 73–86 | 20–35 | 10–21 | 40–71 | [3,31,32] |
| Cattle slurry | 6–11 | 75–82 | 20–30 | 11–19 | 40–61 | [3] |
| Cattle manure | 20–25 | 68–76 | 60–120 | 33–36 | 112–257 | [3,26] |
| Poultry manure | 34–50 | 60–75 | 130–270 | 70–140 | 257–551 | [3,33] |
| Maize silage | 28–39 | 85–98 | 170–230 | 68–120 | 347–469 | [3,26,31,33,34] |
| Grass silage | 15–50 | 70–95 | 102–200 | 46–109 | 208–408 | [3,26,31,33] |
| Sugar beets | 13–23 | 84–90 | 120–140 | 65–113 | 245–286 | [3,26,34] |
| Olive pomace | 57–90 | 55–86 | 92–147 | 65–104 | 188–300 | [35] |
| Wheat straw | 91–94 | 87–92 | 135–237 | 146–266 | [36,37] | |
| Corn (corn stover) | 66–89 | 83–99 | 261–402 | 293–451 | [33,34,38] | |
| Rye | 62–93 | 84–87 | 130 | 70 | 265 | [33,34] |
| Scale of The Plant | Substrate | Stirring Period Daily (h) | Stirring Intensity | Temperature (°C) | Remarks | References | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1–8 | 9–23 | 24 | High | Low | |||||
| A | Beef cattle waste | 0 | − | 55 | [105] | |||||
| B | Beef cattle waste | 0 | 0 | 55 | [105] | |||||
| A | Castor cake | 0 | + | + | 30 | Loading rates 4 and 8 g L−1 d−1 | [106] | |||
| A | Castor cake | 0 | − | − | 37 | Loading rates 4 and 12 g L−1 d−1 | [106] | |||
| A | Castor cake | 0 | + | + | 37 | Loading rate 8 g L−1 d−1 | [106] | |||
| B | Refuse-derived fuel and primary sludge | 0 | − | 35 | [107] | |||||
| A | Water hyacinth and cattle dung | 0 | + | − | 37 | [108] | ||||
| A | Unmodified olive mill wastewater | 0 | − | − | 0 | 35 | [109] | |||
| A | Fermented olive mill wastewater | 0 | 0 | 0 | 0 | 35 | [109] | |||
| A | Sewage sludge | 0 | − | 28 | Loading rates 2.4, 4.8, and 7.2 g COD L−1 d−1 | [110] | ||||
| B | Animal manure | 0 | + | 35 | [111] | |||||
| A | Dog food | 0 | − | 35 & 55 | Batch fed | [112] | ||||
| A | Manure slurry | 0 | + | 35 | [88] | |||||
| A | Animal waste | 0 | − | − | 0 | 35 | Biogas recirculation, low DM values | [89] | ||
| A | Cow manure | + | 0 | − | 55 | [113] | ||||
| B | Cow manure | X | 0 | 0 | + | 54 | [113] | |||
| A | Lipid-rich waste | 0 | + | 37 | Batch reactor | [114] | ||||
| A | Corn silage | 0 | + | 37 | Continuous reactor | [114] | ||||
| A | Rice straw | + | 0 | 35 | [115] | |||||
| A | Manure and MSW | 0 | + | 55 | [116] | |||||
| B | Natural water, cow dung, rice straw and water hyacinth | 0 | + | 31 | [117] | |||||
| A | MSW | 0 | X | − | + | 33 | [118] | |||
| C | Cow dung | X | + | − | 27 | Optimum mixing was for 3 h daily with 60 rpm | [119] | |||
| B | Cow manure and maize straw | 0 | X | − | + | Different DM and C:N ratios | [120] | |||
| A | Cow manure and vegetable waste | 0 | + | [121] | ||||||
| A | Cattle manure, tea waste | 0 | + | 37 | [122] | |||||
| A | Municipal solid waste | − | + | 0 | 34 | [123] | ||||
| OLR (kg m−3 d−1) | Stable/Optimum OLR | Reactor Type | Temperature | Substrate | Reference |
|---|---|---|---|---|---|
| 1.0, 2.0, 3.0 and 4.0 | 3 kg m−3 d−1 | CSTR, Semi-CSTR, 8 L | 35 °C | Maize, rye, fodder beets | [156] |
| 1.0 and 2.5 (COD) | 1 kg COD m−3 d−1 | CSTR, 1 L | 35 °C | Thermally pretreated microalgae | [153] |
| 1.8 to 4.0 (oDM) | 2 kg oDM m−3 d−1 | CSTR, 4 L | 35 °C | Animal by-products from the meat processing industry | [157] |
| Ranged from 1.2 to 8.0 (oDM) | 8 kg oDM [76] m−3 d−1 | CSTR, 1.6 m3 | 35 ± 2 °C | Municipal biomass waste and waste-activated sludge | [155] |
| 3.0, 3.6, 4.2, 4.8, 6.0, 8.0, and 12.0 (oDM) | 6–8 kg oDM m−3 d−1 | CSTR, 40 L | 37 ± 2 °C | Rice straw and pig manure | [150] |
| Ranged from 4.2 to 12.8 (oDM) | 1.12 g oDM L−1 d−1 | Semi-CSTR, 10 L | 37 ± 0.5 °C | Dried pellets of exhausted sugar beet cossettes and pig manure | [145] |
| 1.22, 1.46, 1.70, and 2.0 (oDM) | <2.00 kg oDM substrate.m−3 d−1 | Completely mixed bioreactor, 300 m3 | 39 ± 1 °C | Rice straw | [158] |
| 3.7 to 12.9 (oDM) | 9.2 kg oDM m−3 d−1 | Semi-CSTR, 3000 mL | 37 ± 1 °C | Food waste | [159] |
| Ranged from 0 to 10.0 (oDM) | CSTR, 5 L | 37 ± 0.5 °C | Food waste | [160] |
| Metal | Inhibitory Concentration in mg L−1 | Positive Concentration in mg L−1 | Reference |
|---|---|---|---|
| Aluminum | 1000–2500 | [188] | |
| Cadmium | 36–3400 | 0.1–0.3 | [189,190,191,192,193] |
| Calcium | 300–8000 | 100–1035 | [173,188,194,195,196] |
| Chromium | 27–2500 | 0.1–15 | [191,192,193,197] |
| Cobalt | 35–950 | 0.03–19 | [197,198,199,200,201,202] |
| Copper | 12.5–1000 | 0–10 | [190,191,193,197,201,203] |
| Iron | 0.3–4000 | [189,202] | |
| Lead | 67.2–8000 | 0.2 | [191,196,204] |
| Magnesium | 750–4000 | 0–720 | [183,188] |
| Mercury | 125 | ||
| Molybdenum | 1000 | 0–0.1 | [202,205] |
| Nickel | 35–1600 | 0.03–27 | [191,193,198,199,200,201] |
| Potassium | 400–28,934 | 0–400 | [188,196,206] |
| Sodium | 3000–16,000 | 100–350 | [188,206,207] |
| Zinc | 5–1500 | 0–5 | [189,191,193,198,201,202] |
| Parameter | Measurement Method | Reference |
|---|---|---|
| Cobalt concentration in the high presence of iron concentrations | Total reflection X-ray fluorescence spectroscopy | [238] |
| VOCs (volatile organic compounds) emitted from different units of food waste anaerobic digestion plant | Portable GC-MS (gas chromatography–mass spectroscopy) | [239] |
| CH4 emissions from pressure relief valves of an agricultural biogas plant | Flow velocity and temperature sensors | [240] |
| Ammonia in biogas | Impedance measurement of biogas condensate in the gas room above the digester | [241] |
| Dissolved active trace elements in biogas | Total reflection X-ray fluorescence spectroscopy in dried digester slurry | [238] |
| H2S in biogas | Gas responsive nano-switch (copper oxide composite) | [242] |
| Microbial communities depend on the substrate combinations | Sequencing of the 16S rRNA, biodegradable feedstock samples from eight different biogas plants | [243] |
| Controlling gas pressure in the digester | Programmable logic controller (PCL) | [244] |
| Ammonia in biomethane | Luminescent ammonia sensor based on an imidazole-containing Ru(II) polypyridyl complex immobilized on silica microspheres | [183] |
| pH, temperature, oxidation-reduction potential (ORP) | via electrodes, on-line monitoring with PCL | [245] |
| CO2, CH4, H2O | On-line monitoring with a Supercontinuum laser-based off-resonant broadband photoacoustic spectroscopy | [246] |
| Different volatile fatty acids | On-line monitoring with total-reflectance Fourier-transformed infrared spectroscopy (ATR-MIR-FTIR) | [2] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nsair, A.; Onen Cinar, S.; Alassali, A.; Abu Qdais, H.; Kuchta, K. Operational Parameters of Biogas Plants: A Review and Evaluation Study. Energies 2020, 13, 3761. https://doi.org/10.3390/en13153761
Nsair A, Onen Cinar S, Alassali A, Abu Qdais H, Kuchta K. Operational Parameters of Biogas Plants: A Review and Evaluation Study. Energies. 2020; 13(15):3761. https://doi.org/10.3390/en13153761
Chicago/Turabian StyleNsair, Abdullah, Senem Onen Cinar, Ayah Alassali, Hani Abu Qdais, and Kerstin Kuchta. 2020. "Operational Parameters of Biogas Plants: A Review and Evaluation Study" Energies 13, no. 15: 3761. https://doi.org/10.3390/en13153761
APA StyleNsair, A., Onen Cinar, S., Alassali, A., Abu Qdais, H., & Kuchta, K. (2020). Operational Parameters of Biogas Plants: A Review and Evaluation Study. Energies, 13(15), 3761. https://doi.org/10.3390/en13153761