1. Introduction
There is a growing demand for bioenergy in today’s world. Biomass as a renewable resource form is becoming an important element of sustainability and more attractive to the public due to its abundance and reduced air pollution in terms of criteria air pollutants and greenhouse gases as compared to most types of fossil fuels [
1]. In particular, a large amount of woody biomass (forest residues) can be harvested annually in North America to produce heat and power as well as a rather broad range of products that include biofuels, pulp and paper, panels, chemicals and other energy feedstocks [
2].
The immediate use of woody biomass after harvest is generally infeasible. Depending on the production cycles, the biomass is stored in the field for some time before transportation and processing. It is particularly necessary to store the biomass materials for an extended time period during the winter months in order to ensure their uninterrupted supply and delivery to the conversion facilities. The need for woody fuels storage, especially wood chips, is also in line with the increasing demand for bioenergy.
Nevertheless, the prolonged storage of waste biomass prior to utilization may cause some problems. The physicochemical and biological degradations of the biomass during storage will lead to gas emissions that have impact on the surrounding environment and human health [
3,
4,
5]. Gas emissions from stored biomass will also result in a loss of dry matter and the heating value of the materials, and hence a negative economic impact [
6,
7,
8]. In our previous study, dry matter loss and gas emissions were found to be positively correlated [
9]. Furthermore, the degradation process can lead to heat buildup that will favor the growth of microbes and generate higher temperatures in the pile; and in some cases, even self-ignition and spontaneous fires [
10]. Serious and fatal accidents have been reported in the past decade that are associated with self-heating and fires during the unloading of woody biomass after long time storage [
11,
12].
In the literature, gas emissions are assumed to arise from the biological decomposition and/or chemical oxidation of the organic compounds in wood [
10,
13]. Gas emissions from stored biomass have been identified to be mainly composed of CO
2, CO, CH
4 and non-methane volatile organic compounds (NMVOCs) [
3,
11,
14]. The emission of CO
2, CO and VOCs accumulated to toxic levels together with a large depletion in oxygen when the biomass were stored in confined spaces during sea transportation [
8]. For instance, it was shown that the concentration of CO emitted from pellets storage bins exceeded the guidance and regulatory limits for CO exposure [
15]. Tumuluru et al. suggested that CO emission from woody biomass was due to oxidation of chemical compounds in the materials [
16]. Sedlmayer et al. found that wood extractives like fatty and resin acids were correlated with NMVOC emissions, but not CO emissions, from wood pellets [
17]. According to Alakoski et al., chemical oxidation of fatty acids is the dominant mechanism for gas emissions from wood pellets, while biological degradation plays a more significant role in emissions from wood chips [
4].
Factors such as ambient temperature, precipitation, the size of the stockpile, duration of storage, moisture content and particle size were found to affect the emissions from the storage of wet woody biomass [
18,
19]. Results from a study on stored Douglas fir chips showed that higher temperature led to higher gas concentrations [
20]. Emery and Mosier investigated emissions from large-scale biomass storage and revealed that methane emissions were inversely correlated with material moisture [
21]. As for the storage of dried wood pellets (generally having less than 10% moisture content), the gas emission rates and the maximum concentrations were reported to be lower at lower temperatures [
22,
23]. Li et al. observed higher emission of aldehydes and ketones due to accelerated hydrolysis of glyceride in woody pellets with higher moisture contents [
24]. In other studies of stored wood pellets, the emissions were positively affected by the ambient relative humidity and oxygen availability [
4,
25].
Dry matter loss is affected by storage duration, biomass characteristics, and weather elements (temperature, relative humidity, precipitation) [
26,
27]. The annual dry matter loss from the storage of fresh biomass was reported to range from 3–21% [
28], while the monthly dry matter loss during the storage of wood chips ranged from 2–4% [
29]. The moisture content of biomass was found to be proportional to the dry matter loss; thus dried forest residues had 6.6% dry matter loss whereas fresh materials lost 15.6% dry mass after 6-month storage [
10]. It was reported that the dry matter losses were 5.8% and 7.3% from stored biomass with/without cover respectively after seven-month storage [
30]. In a study on the storage of wood chips for one year, Tumuluru et al. observed a reduction in the heating value of woody biomass by 25–55% due to biochemical reactions and moisture adsorption [
16]. Moisture content of wood chips lower than 20% (wet basis) can slow down the decomposition process and minimize the dry matter loss during storage. Yet, in practice the challenge to obtain such low moisture content can only be met by artificial (forced) drying rather than natural drying [
10,
18].
Western Red Cedar (WRC,
Thujaplicata) is a popular softwood species in British Columbia, Canada. The WRC residues can be used as alternative clean biomass in the form of chips or pellets for combustion, or converted to liquid biofuels [
31]. Chipped wood waste that originates from WRC and other softwood species are being used as solid biofuel in the biomass gasification plant (as part of a District Energy System) on the University of British Columbia campus. In fact, WRC chips can be one source of biomass feedstock for densification via pelletizing as some two-thirds of the wood pellets manufactured in North America (Canada and USA) are shipped to Europe and Asia [
32].
Based on the literature review, moisture content is an important factor that affects the gas emissions from stored woody biomass. However, the impact of moisture content on stored wood chips has not received much attention, especially the extent of its influence on gas emission and dry matter loss. In particular, emissions from stored WRC chips with different moisture contents have not been studied. The objective of this paper is to investigate the gas emission from stored WRC chips with different moisture contents and its effect on dry matter loss under different storage conditions. Findings from this study will be useful in developing safe storage and transportation of woody biomass, and contribute towards assessing the potential impact of gas emission on the surrounding environment and human health.
2. Materials and Methods
2.1. Materials
Western red cedar chips used in this study were obtained from Langley, BC, Canada (
Figure 1). The cedar logs originating from Vancouver Island were transported in water booms to sawmills in the Lower Mainland Vancouver area. The chips with a uniform size for pulp or bioenergy applications were produced from waste wood which was cut off to make commercial products. The chips were stored in a room at 4 °C at UBC laboratory before the experiment. The average size of the chips as measured by the Gilson Testing Screens (Gilson Company, Worthington, OH, USA) had a range of 10–20 mm in length, 5–10 mm in width and 1–3 mm in thickness. The moisture content of the wood chips was determined in triplicate in a forced-air convection oven at 103 °C for 24 hours according to ASABE Standards S358.2 [
33]. The initial moisture content of the chips was about 50% (wet basis). The wood chips were further dried in an oven to obtain the moisture content around 20% (wet basis) and 35% (wet basis), respectively. The moisture content of 35% simulates the value of naturally dried wood chips, while the value of 20% is after artificial drying. All materials were then stored at 4 °C before the test.
2.2. Experimental Setup
Wood chips with different initial moisture contents (20%, 35% and 50%, wet basis) were divided into three series in this study. Series 1 to 3 represented the WRC chips with initial moisture content of 20%, 35% and 50%, respectively. For each series, materials were separated to two groups, one group under aerobic condition and the other group under non-aerobic condition. This is to simulate the different environmental conditions that the materials in a pile experience during storage depending on the availability of oxygen.
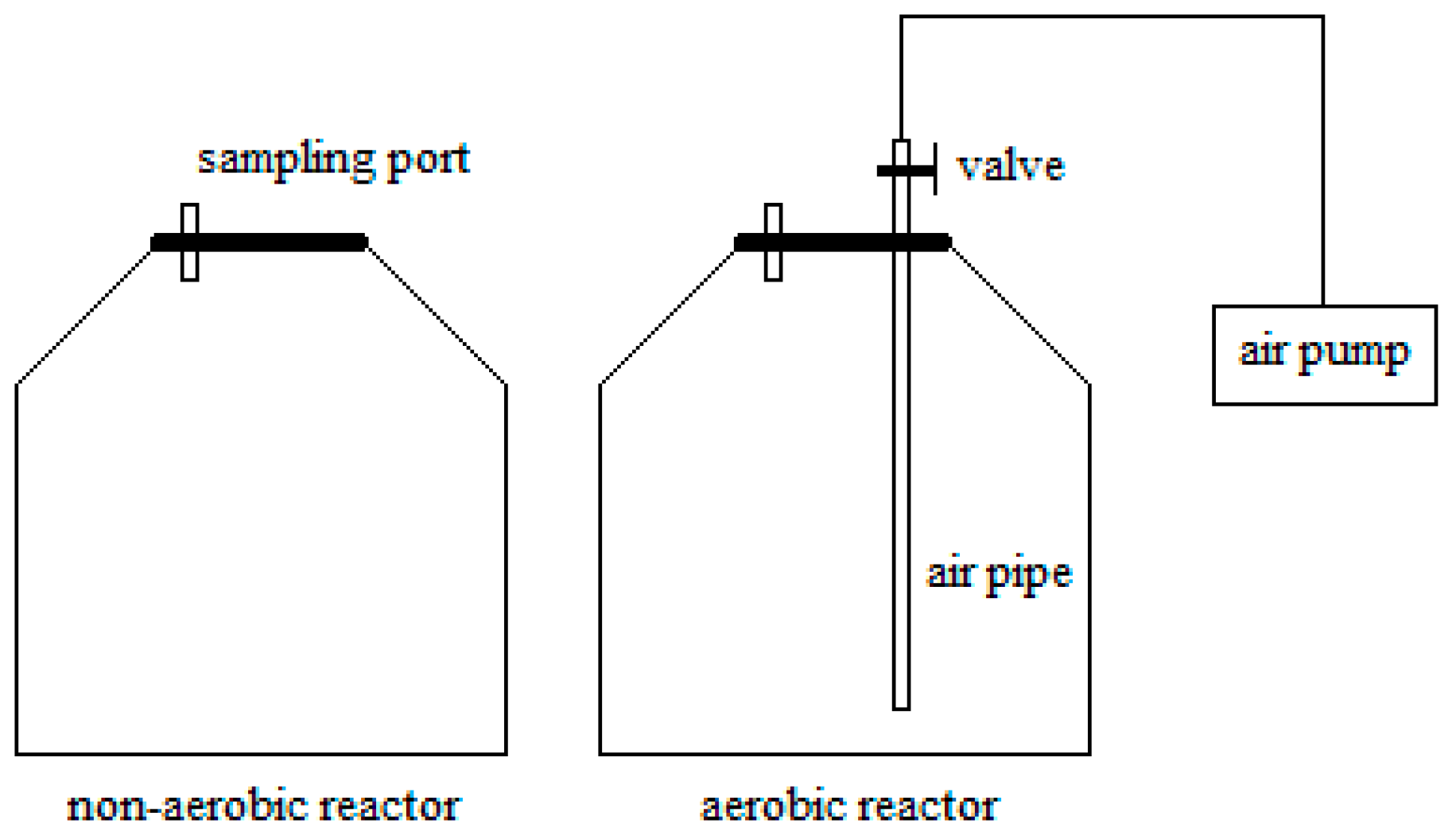
A number of 2 L glass containers were used to conduct the storage test. The containers were fitted with valves and sampling ports. Each assembled reactor was then loaded with 330 g wood chips. As shown in
Figure 2, non-aerobic reactors were sealed at all times to study gas emissions under airtight conditions. After the daily gas sampling, air was pumped into the aerobic reactors in order to replenish and maintain a high oxygen level in the reactors.
After all the reactors were loaded with the materials, they were sealed and placed under different temperatures at 5 °C, 20 °C, 35 °C and 50 °C. This range of storage temperature represents cool to hot climate conditions in different geographic locations, and involving seasonal variations. Two replicates were performed for each test. The average values of the results obtained from the two replicates were then presented. In all cases, the experiment period was approximately two months.
2.3. Gas Emission Measurements
The emissions of CO2 and CO from the tests were analyzed by gas chromatography (Model SRI8610 C, Mandel, Torrance, CA, USA). The packed column GC was equipped with FID/TCD detector, and using argon as carrier gas. The oven was heated up to 400 °C with a retention time of 15 min. The GC was calibrated regularly with the corresponding standard gases. During each sampling event, gas sample was drawn by using syringe from each reactor to measure the concentrations of CO2, CO and O2 concentrations. Gas emissions from the aerobic reactors were measured daily. Whereas, gas emissions from the non-aerobic reactors were measured more frequently at the beginning of the experiment, and every few days in the later period of the test. 250 μL gas sample was drawn from each reactor at the end of the experiment to analyze the NMVOC compositions by GC/MS analyzer (Model 5975B/6890N, Agilent Technologies, Santa Clara, CA, USA). The GC was equipped with capillary column and FID detector, and using helium as carrier gas. The oven was heated to 50 °C, and then ramped 20 °C/min till 200 °C for 3 min.
The total concentrations of NMVOCs (TVOC) from the aerobic reactors were measured by a portable VOC monitor (Model PGM-7240, RAE Systems, San Jose, CA, USA) on a daily basis. The ppbRAE photoionization detector was employed to measure the concentration by plugging the detection end to the reactor outlet after daily opening. The measured values were recorded after stabilization. For the reactors under non-aerobic conditions, the same procedure cannot be applied since this would cause a larger amount of gases to be released from the reactor, thus affecting the accuracy of gas analysis for the remaining test (storage) period. Thus, TVOC concentrations associated with the non-aerobic reactors were measured at the end of the test period.
2.4. Data Analysis
In this study, the emissions of gases are expressed as emission factors, in units of [gram gas species per kilogram dry matter DM]. Emission factor is a cumulative parameter. The measured gas concentrations in vol% were converted to emission factors by using the N
2 balance method [
13], assuming that nitrogen was not consumed during the test period. At constant temperature and pressure, the emission factor
f in [g/kg DM] is related to volumetric gas concentration
Ci as follows:
where
I is the gas species;
Ps is absolute pressure of gas in the container (Pa);
Ci is volumetric concentration of a particular gas (m
3 of gas species per m
3 of gas going through the GC);
V is gas volume in the reactor (m
3);
Mwt is gas molecular weight (g/mol);
Cn0 is initial concentration of nitrogen (%);
Cnt is the concentration of nitrogen at time
t (%);
R is universal gas constant (8.31 J/(mol·K));
T is temperature (K); and
m is the total mass of materials in the container (kg). The gas volume is the numeric difference between the volume of container and volume of chips. Change in pressure during the test period was assumed to be minimal in general.
4. Discussions
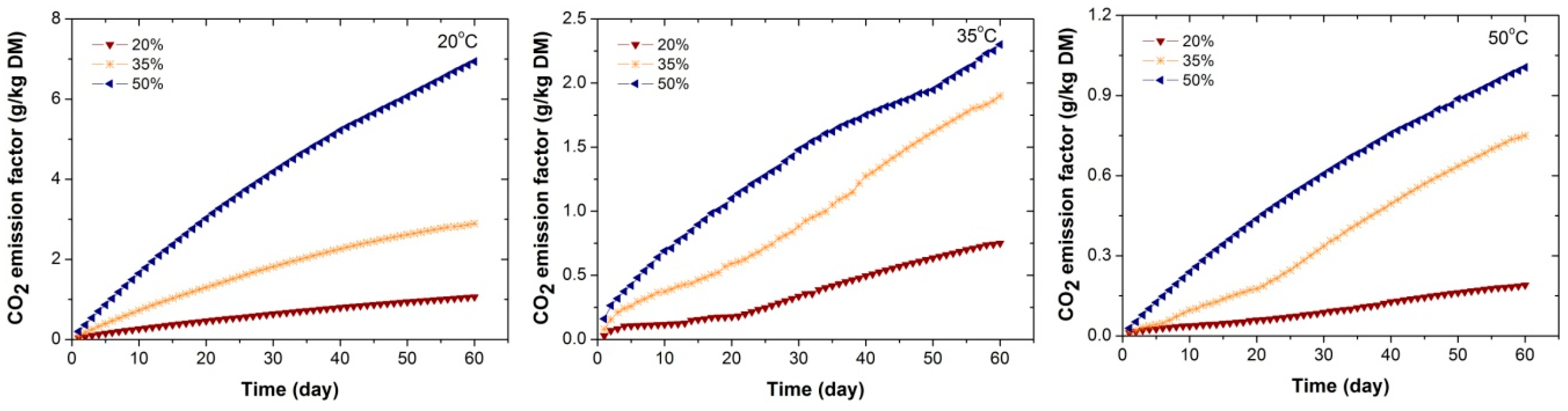
Biomass materials can be degraded both chemically and biologically. When chemical oxidation is dominant, the gas emissions increase with temperature according to the Arrhenius relationship for the temperature dependence of chemical reaction rates. Moreover, temperature is also one of the key factors that affect biological processes, which can be categorized with regard to three cardinal temperatures (minimum, optimum and maximum). When temperatures are lower or higher than the optimum, bacteria and fungi could be dormant or even killed. The highest fCO2 at 20 °C indicated that the generation of CO2 from WRC chips was dominated by biological mechanism rather than chemical mechanism. CO is produced from chemical reaction of the materials and promoted by high temperature generally. The increase in the CO emission factor with temperature and oxygen availability in this study implied dominance by chemical mechanism.
The variation in the emission factors of WRC chips with different moisture contents suggested that the moisture content is an important factor affecting the gas emissions from biomass materials during storage. In terms of biological aspect, microbial activities have a vital influence on the process. Moisture content is known to be a key factor that governs biological reactions. It was found to have a significant effect on the size and activity of the microbial community as the overall patterns of respiration were strongly controlled by moisture content. The respiration rate of the microbes had a positive correlation with percentage humidity [
34]. The microbial growth rate constant and the evolution of CO
2 increased linearly with increasing moisture content. And the microbial activity was inhibited under dry circumstance [
35].
One previous study reported that the fresh forest residues with 60% moisture content (wet basis) emitted more than twice the amount of CO
2 as compared to that emitted from naturally dried forest residues with 40% moisture content after 6-month storage [
29]. Other research also reported the importance of moisture content to the chemical reaction rate, as high moisture content favored the decomposition process and allowed rapid degradation circumstances [
13]. Our results are in accord with these studies, that WRC chips having higher moisture content generated greater gas emissions.
For the purpose of better understanding, we compared our results with other woody materials (moisture content ranging from 3% to 50%, wet basis) in the literature. Yazdanpanah [
36] measured the gas emissions from white pellets with moisture contents of 4%, 9%, 15%, 35% and 50% (wb) at temperatures of 25 °C, 40 °C and 60 °C for 9-week storage test period. The emission factors of CO
2 that ranged from 0.007 to 0.415 g/kg DM were positively correlated with temperature as well as moisture content. Our experimental results suggested that the CO
2 emissions from wood chips were an order of magnitude greater than those from the wood pellets. The differences in gas emissions may be due to the presence of live microorganisms in the wood chips, and the larger surface area of wood chips facilitate the microbial colonization. Furthermore, most of the microorganisms in wood pellets were killed during pelletization and pretreatment processes. Fresh wood chips could release more CO
2 than wood pellets in the beginning of storage, possibly as a result of biological reaction in addition to chemical oxidation. When oxygen was eventually depleted to zero, no more CO
2 was generated from biological process. Similar mechanism was also revealed in our earlier study on gas emissions from stored Douglas fir wood chips with moisture content of 50% (wet basis), whereby results indicated that CO
2 emissions increased from 3 g/kg DM to 18 g/kg DM as temperature rose from 5 °C to 50 °C [
9]. The highest CO
2 emission from Douglas fir chips was about three times higher than that from the WRC chips. Besides, CO emissions from stored WRC chips were significantly lower when compared to the corresponding emissions from wood pellets.
The gas emissions of CO
2 and CO may have safety and health implications, depending on the duration of exposure. The threshold limit value-time weighted average (TLV-TWA) values for 8 h exposure to CO
2 and CO are 0.5 and 0.0025%, respectively. The threshold limit value-short term exposure limit (TLV-STEL) values for 15 min exposure to CO
2 and CO are 3% and 0.01%, respectively. The corresponding concentrations that are deemed “Immediately Dangerous to Life and Health” for CO
2 and CO are 4% and 0.12%, respectively [
37]. Thus, the storage of biomass can be dangerous if it is not ventilated properly. In confined space, CO concentrations are several orders of magnitude higher than the occupational exposure limits. Serious accidents in Europe have been reported in the past decade, as the CO concentration was found to be high with a range of 14 ppm to 155 ppm (v/v) in the pellet storage bins [
38].
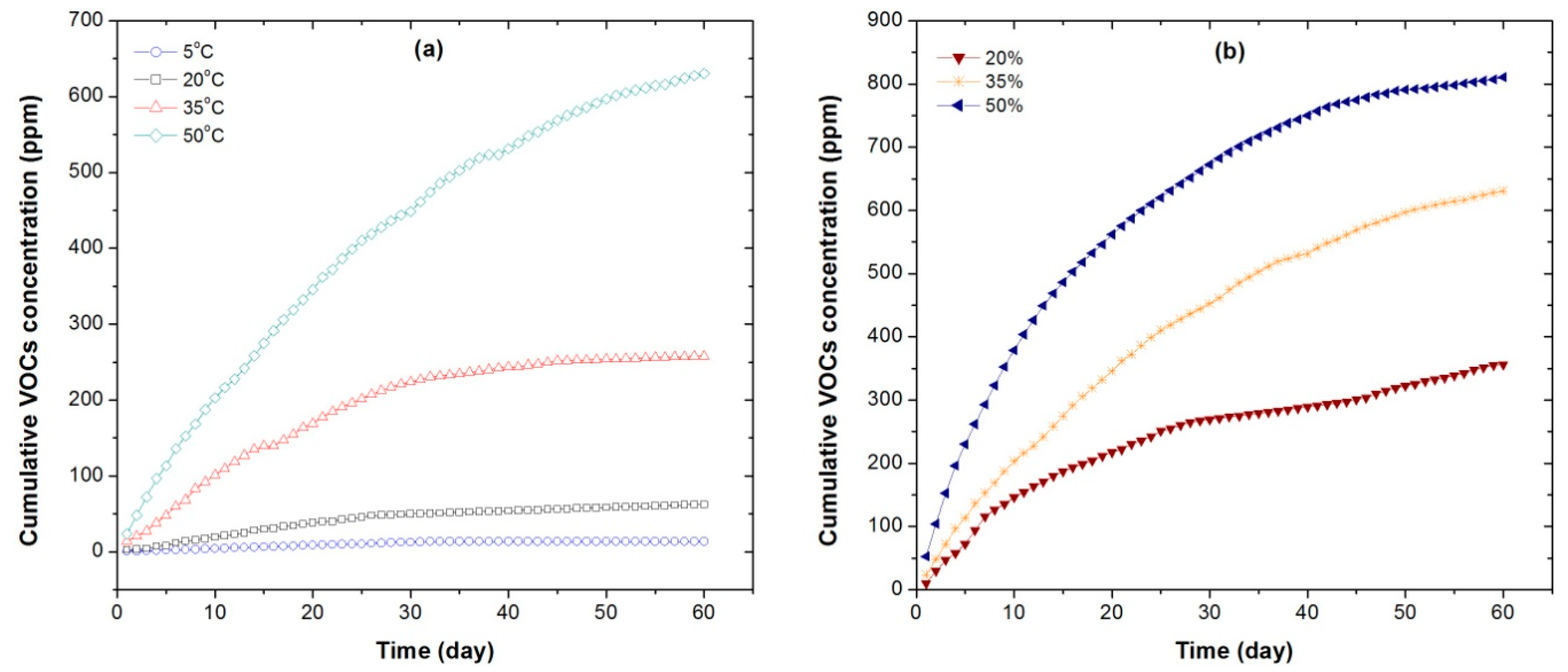
As products of the degradation of woody materials during storage, many of the NMVOCs are highly flammable and may lead to the self-ignition during storage. The odorous NMVOCs (for instance aldehydes, ketones and terpenes) may have odor nuisance and even health problems, as they can be inhaled by people who live nearby or work close to the storage area. Some of the NMVOCs detected in this study were also reported in the literature. Monoterpenes were found to be the primary NMVOCs from fresh spruce and pine wood [
4,
39]. Wang et al. reported the emissions of aldehydes and ketones from stored wood pellets [
25]. Aldehydes are the most abundant NMVOCs from pellet storage, in which hexanal accounts for 80% of emitted aldehydes. Other lab study also showed that the main NMVOCs including methanol, pentane, pentanal and hexanal were generated by the oxidation of stored pellets [
38]. Temperature was in accord with the TVOC concentrations from stored WRC chips. It reaffirmed that higher temperature could induce more release of VOCs as a result of organic matter degradation. This phenomenon was also found from other biomass materials [
25].
Dry matter loss is one of the major factors that attribute to the limitation of wood chips storage in large piles. Dry matter loss can affect the energy value of fuel, so that they have to be accounted for during the emission evaluations. The reported monthly dry matter loss from storage of fresh biomass chips ranged from 1–4% [
26,
28,
29], while the maximum mass loss of lower than 1% was found in this study after two months of storage. By comparison with other woody materials reported in the literature, small degree of dry matter loss from this storage test reaffirms the decay-resistance characteristics of WRC chips as extractives like thujaplicins were present in the wood [
3,
9].
The utilization of forest chips has been predicted to increase in the future. Therefore, the safe storage of biomass is of great importance. The risks of storing wood chips include spontaneous heating and self-ignition, reduction of fuel quality, dry matter losses, human health and environmental risks from gas emissions [
4]. To some extent, gas emissions during storage are much higher than those from the rest of the biomass supply chain. However, in practice, it may be difficult to monitor the gas emissions from wood chip piles. Results from this study can be used as a preliminary estimate to larger scale piles. In most cases, fresh wood chips contain high moisture when received, and the moisture content varies depending on sources and climate. Wood chips can be subject to drying process before storage, but it could have high energy consumption. Thus, it is desirable to grasp a good understanding of dry matter loss and the associated gas emissions.