In Situ Water Electrolyzer Stack for an Electrobioreactor
Abstract
1. Introduction
2. Theory and Concept Background
2.1. In Situ Water Electrolysis and Electrobioreactor Characteristics
2.2. Immersed Electrodes
- The electrode area must be increased to maximize the hydrogen production.
- The coil-shaped electrodes inside each other lead to inhomogeneous current densities in the electrode surface and the electrolyte.
- The rod electrodes also force most of the current to travel along an unnecessarily long path through the low-conductivity electrolyte.
- The electrodes make the mixing of the reactor more difficult.
- The internal resistance of the electrode limits the length and thus the surface area of the electrode.
- The serial connection of the immersed electrodes is not allowed to avoid excessive leakage currents, although it would be necessary to scale the system voltage to a favorable level for the industrial-scale AC/DC converters
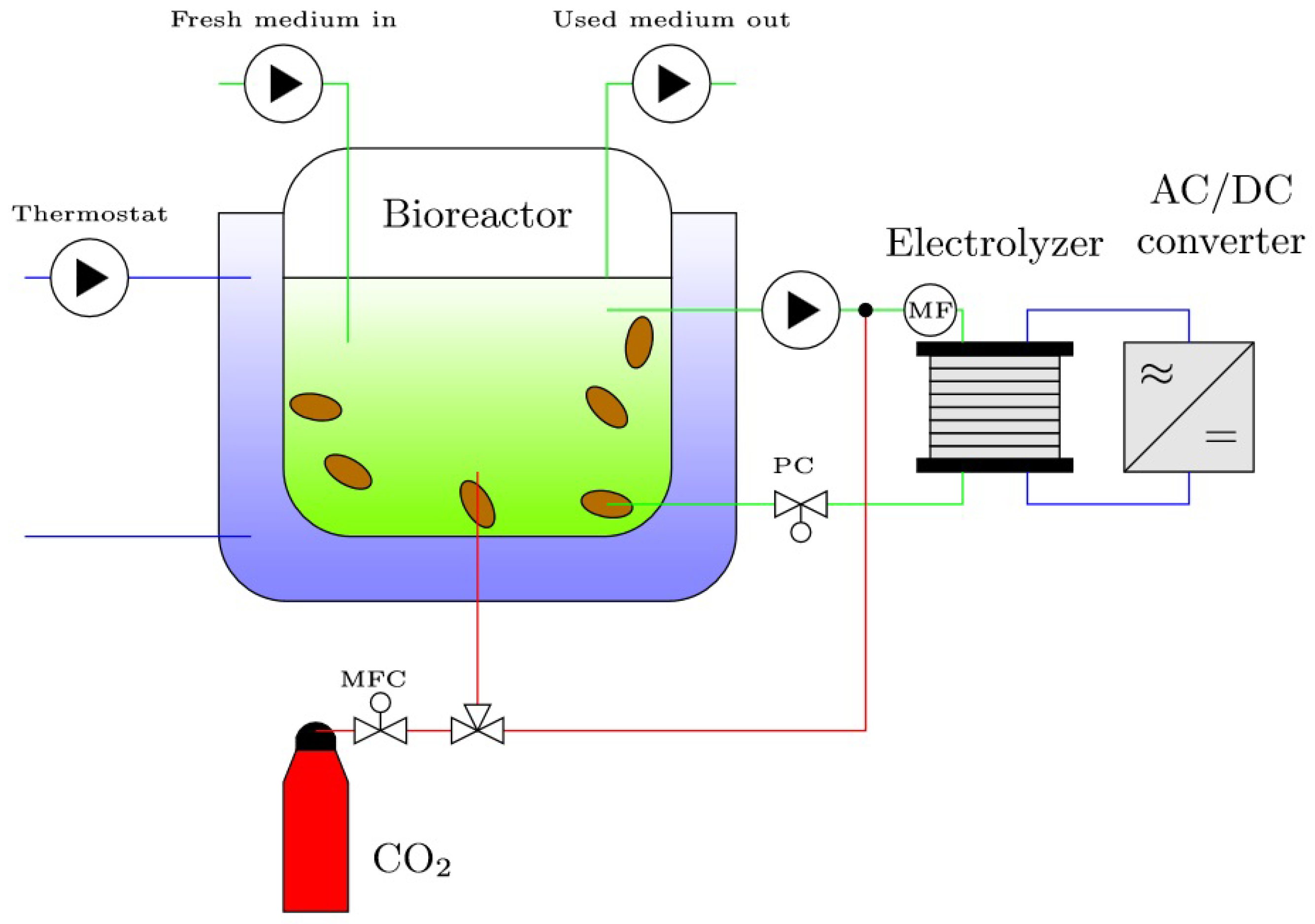
2.3. Conceptual Design of the Electrobioreactor System
- A high electrode area per volume;
- A constant distance between electrodes, leading to constant current densities;
- Simple series connection of electrolytic cells;
- Low leakage currents, resulting in enhanced power densities;
- Stack voltage levels suitable for industrial AC/DC converters;
- A high energy efficiency.
3. Materials and Methods
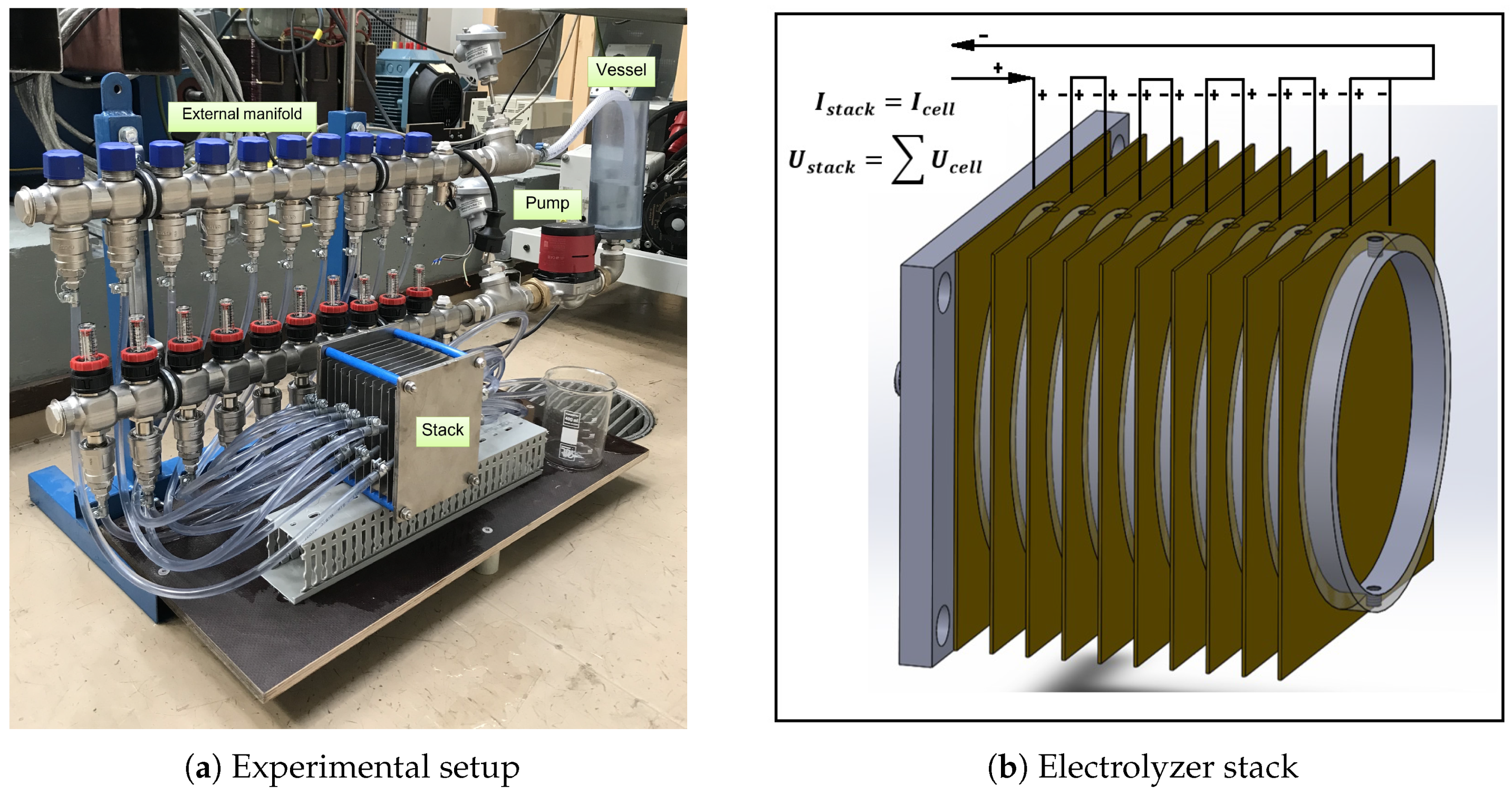
3.1. Experimental Setup
3.2. Electrolyte
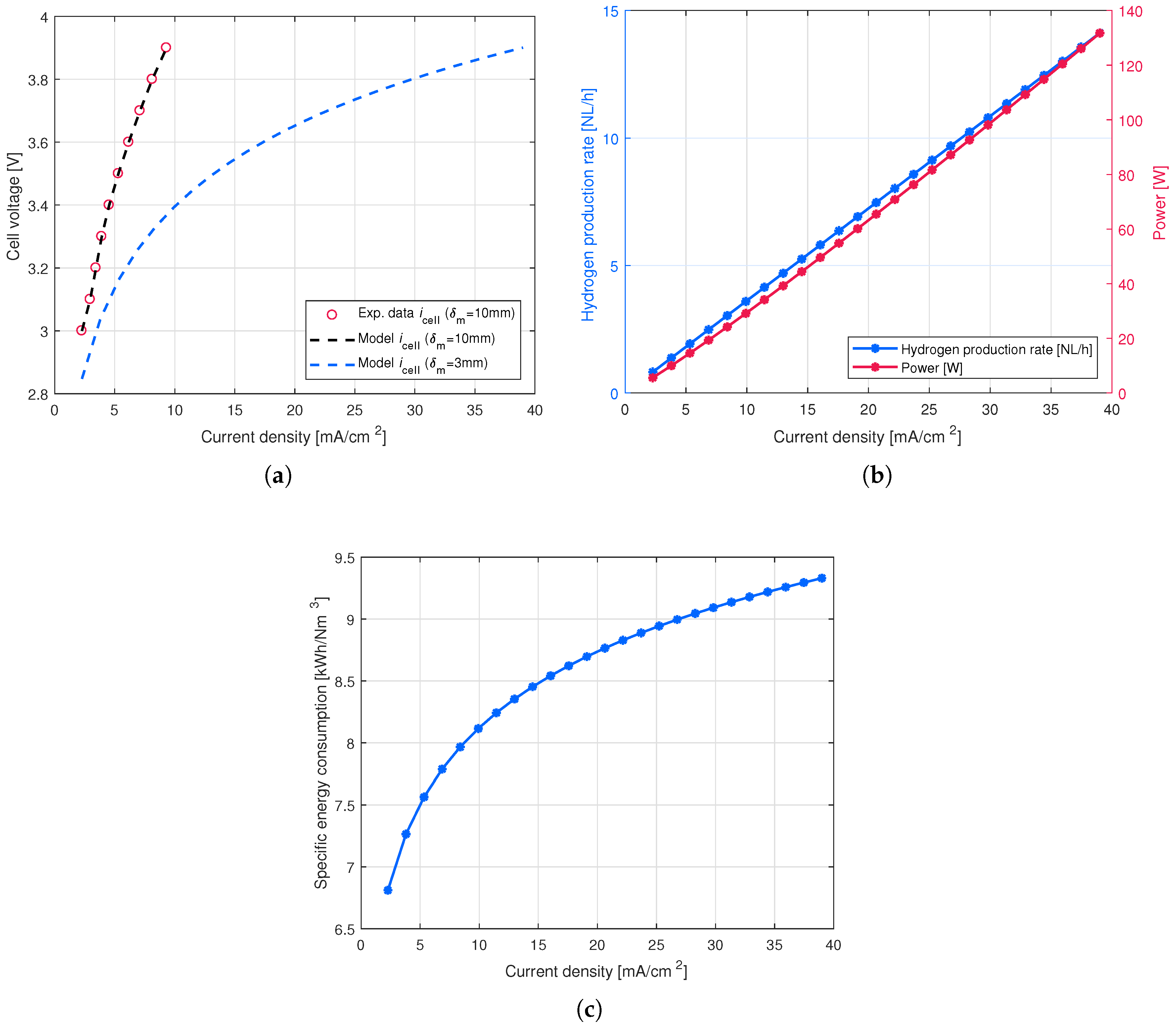
4. Results and Discussion
4.1. Effect of Electrode Distance on the Cell Performance
4.2. Volumetric Productivity
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Materials and Methods
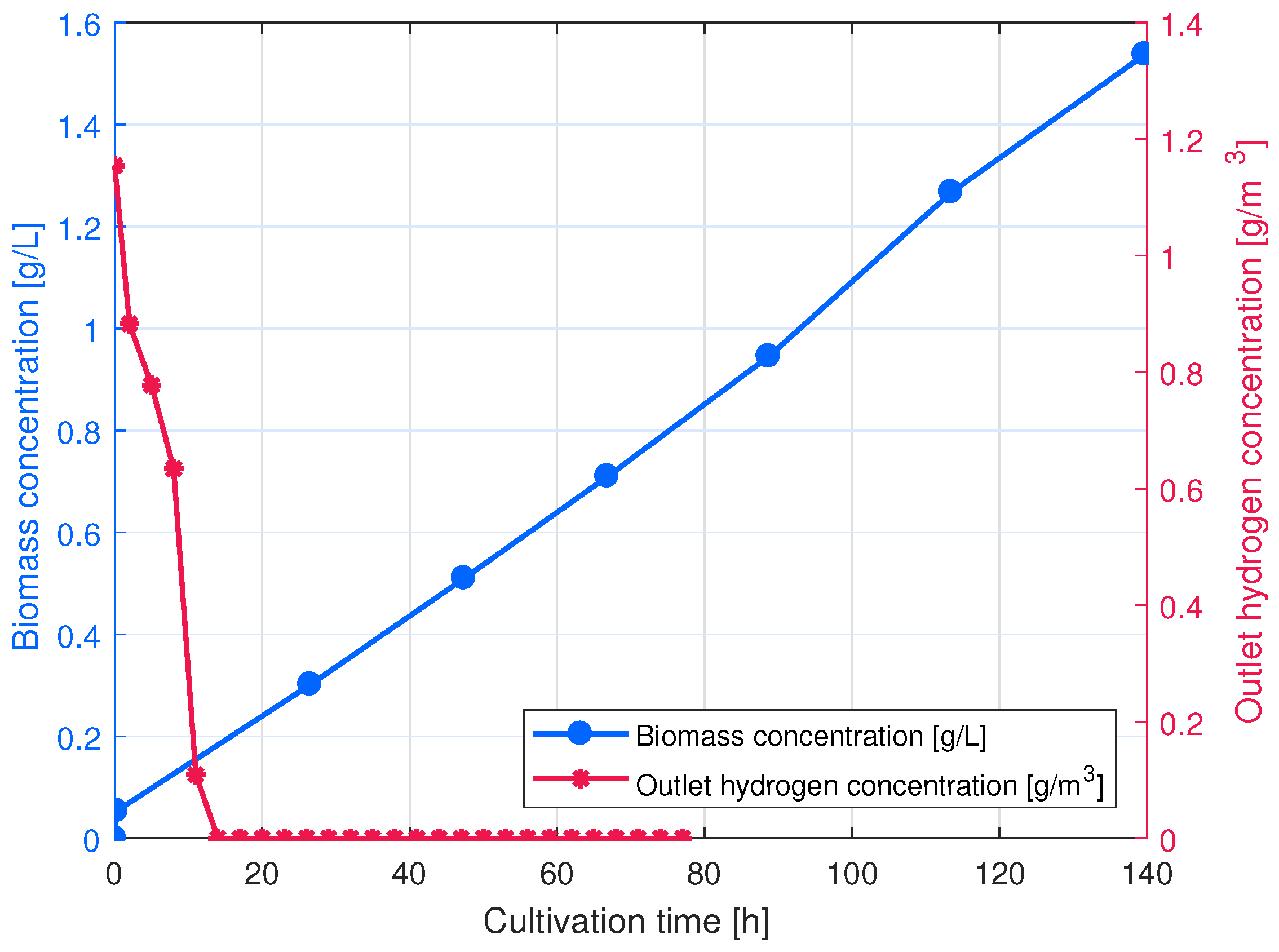
Appendix A.1. BES Cultivation
Appendix A.2. HOB Growth Rate Calculation from the Outlet Hydrogen Concentration Data
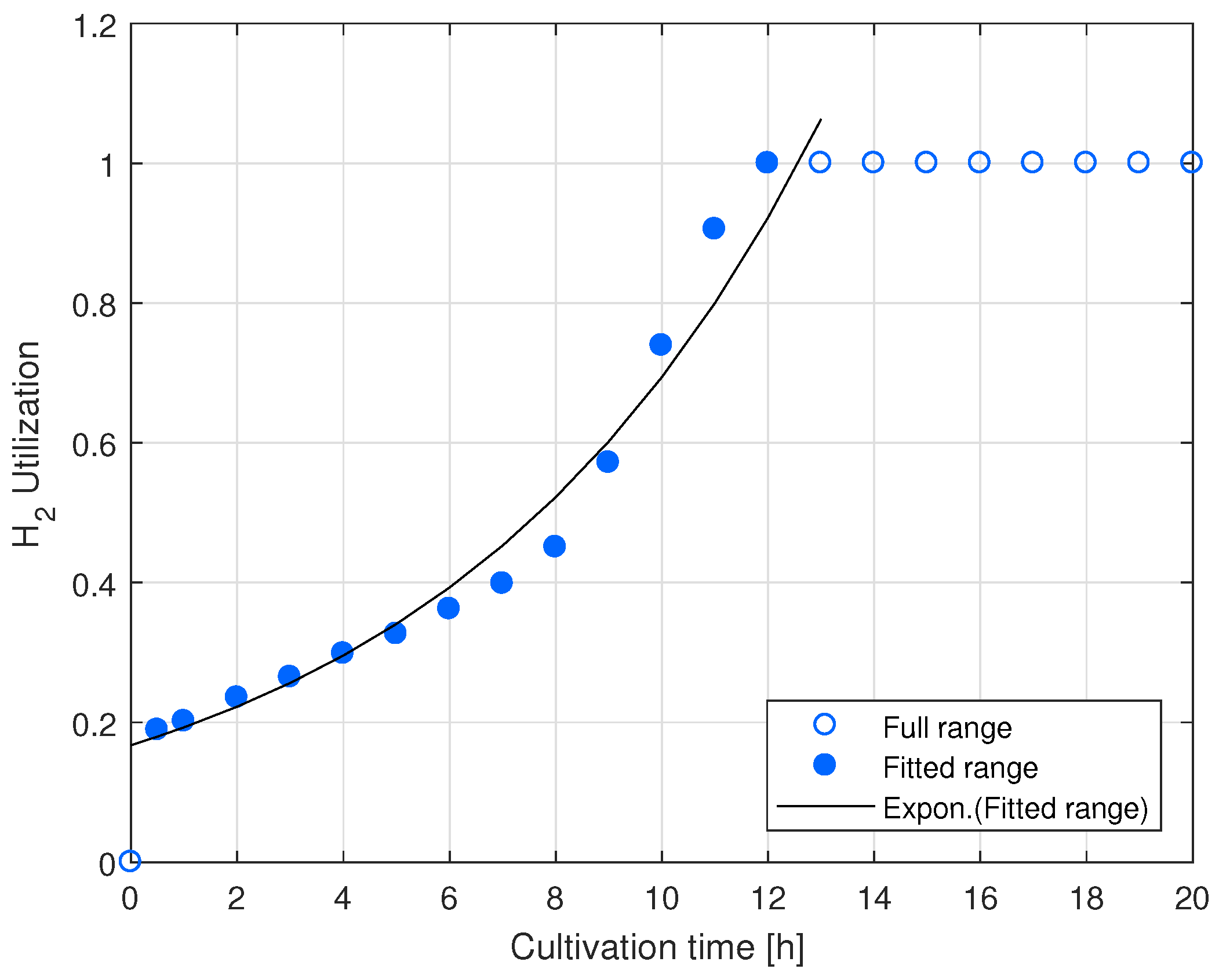
References
- Brown, T.W.; Bischof-niemz, T.; Blok, K.; Breyer, C.; Lund, H.; Mathiesen, B.V. Response to ‘Burden of proof: A comprehensive review of the feasibility of 100% renewable-electricity systems. Renew. Sustain. Energy Rev. 2018, 92, 834–847. [Google Scholar] [CrossRef]
- Kousksou, T.; Bruel, P.; Jamil, A.; El Rhafiki, T.; Zeraouli, Y. Energy storage: Applications and challenges. Sol. Energy Mater. Sol. Cells 2014, 120, 59–80. [Google Scholar] [CrossRef]
- Vidal, F.; Koponen, J.; Ruuskanen, V.; Bajamundi, C.; Kosonen, A.; Simell, P.; Ahola, J.; Frilund, C.; Elfving, J.; Reinikainen, M.; et al. Power-to-X technology using renewable electricity and carbon dioxide from ambient air: SOLETAIR proof-of-concept and improved process concept. J. CO2 Util. 2018, 28, 235–246. [Google Scholar]
- Pikaar, I.; Matassa, S.; Rabaey, K.; Bodirsky, B.L.; Popp, A.; Herrero, M.; Verstraete, W. Microbes and the Next Nitrogen Revolution. Environ. Sci. Technol. 2017, 51, 7297–7303. [Google Scholar] [CrossRef]
- Kracke, F.; Wong, A.B.; Maegaard, K.; Deutzmann, J.S.; Hubert, M.A.; Hahn, C.; Jaramillo, T.F.; Spormann, A.M. Robust and biocompatible catalysts for efficient hydrogen-driven microbial electrosynthesis. Commun. Chem. 2019, 2, 45. [Google Scholar] [CrossRef]
- Matassa, S.; Boon, N.; Verstraete, W. Resource recovery from used water: The manufacturing abilities of hydrogen-oxidizing bacteria. Water Res. 2015, 68, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Linder, T. Making the case for edible microorganisms as an integral part of a more sustainable and resilient food production system. Food Secur. 2019. [Google Scholar] [CrossRef]
- Pikaar, I.; Vrieze, J.D.; Rabaey, K.; Herrero, M.; Smith, P.; Verstraete, W. Science of the Total Environment Carbon emission avoidance and capture by producing in-reactor microbial biomass based food, feed and slow release fertilizer: Potentials and limitations. Sci. Total. Environ. 2018, 644, 1525–1530. [Google Scholar] [CrossRef] [PubMed]
- Volova, T.G.; Barashkov, V.A. Characteristics of proteins synthesized by hydrogen-oxidizing microorganisms. Appl. Biochem. Microbiol. 2010, 46, 574–579. [Google Scholar] [CrossRef]
- Oesterholt, F.; Matassa, S.; Palmen, L.; Roest, K.; Verstraete, W. Pilot scale production of single cell proteins using the power-to-protein concept Future global challenges. In Proceedings of the 2nd International Resource Recovery Conference, New York, NY, USA, 5–9 August 2018; pp. 1–16. [Google Scholar]
- Matassa, S.; Verstraete, W.; Pikaar, I.; Boon, N. Autotrophic nitrogen assimilation and carbon capture for microbial protein production by a novel enrichment of hydrogen-oxidizing bacteria. Water Res. 2016, 101, 137–146. [Google Scholar] [CrossRef]
- Yu, J. Bio-based products from solar energy and carbon dioxide. Trends Biotechnol. 2014, 32, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Torella, J.P.; Gagliardi, C.J.; Chen, J.S.; Bediako, D.K.; Colón, B.; Way, J.C.; Silver, P.A.; Nocera, D.G. Efficient solar-to-fuels production from a hybrid microbial–water-splitting catalyst system. Proc. Natl. Acad. Sci. USA 2015, 112, 2337–2342. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Ziesack, M.; Silver, P.A. Water splitting–biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis. Science 2016, 352, 1210–1213. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Sakimoto, K.K.; Colón, B.C.; Silver, P.A.; Nocera, D.G. Ambient nitrogen reduction cycle using a hybrid inorganic–biological system. Proc. Natl. Acad. Sci. USA 2017, 114, 6450–6455. [Google Scholar] [CrossRef]
- Liu, C.; Colón, B.E.; Silver, P.A.; Nocera, D.G. Solar-powered CO2 reduction by a hybrid biological∣inorganic system. J. Photochem. Photobiol. A Chem. 2018, 358, 411–415. [Google Scholar] [CrossRef]
- Nangle, S.N.; Sakimoto, K.K.; Silver, P.A.; Nocera, D.G. Biological-inorganic hybrid systems as a generalized platform for chemical production. Curr. Opin. Chem. Biol. 2017, 41, 107–113. [Google Scholar] [CrossRef]
- Krieg, T.; Sydow, A.; Schro, U.; Schrader, J.; Holtmann, D. Reactor concepts for bioelectrochemical syntheses and energy conversion. Trends Biotechnol. 2014, 32, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Decourt, B.; Lajoie, B.; Debarre, R.; Soupa, O. The Hydrogen-Based Energy Conversion FactBook; The SBC Energy Institute: Melbourne, Australia, 2014. [Google Scholar]
- Ursúa, A.; Gandía, L.; Sanchis, P. Hydrogen Production From Water Electrolysis: Current Status and Future Trends. Proc. IEEE 2012, 100, 410–426. [Google Scholar] [CrossRef]
- Yu, J.; Munasinghe, P. Gas Fermentation Enhancement for Chemolithotrophic Growth of Cupriavidus necator on Carbon Dioxide. Fermentation 2018, 4, 63. [Google Scholar] [CrossRef]
- Reilly, C.O.; Farrell, M.; Harvey, D.; Cassidy, J. Operation of an inexpensive bipolar alkaline electrolyser producing a mix of H2/O2 fuel. Int. J. Hydrogen Energy 2015, 41, 2197–2201. [Google Scholar] [CrossRef]
- DSMZ GmbH. 81. Mineral Medium For Chemolithotrophic Growth (H-3); DSMZ: Braunschweig, Germany, 2011. [Google Scholar]
- Pfennig, N. Rhodopseudomonas globiformis, sp. n., a new species of the Rhodospirillaceae. Arch. Microbiol. 1974, 100, 197–206. [Google Scholar] [CrossRef]
- Roeßler, M.; Sewald, X.; Müller, V. Chloride dependence of growth in bacteria. FEMS Microbiol. Lett. 2006, 225, 161–165. [Google Scholar]
- Givirovskiy, G.; Ruuskanen, V.; Ojala, L.; Lienemann, M.; Kokkonen, P.; Ahola, J. Electrode material studies and cell voltage characteristics of the in situ water electrolysis performed in a pH-neutral electrolyte in bioelectrochemical systems. Heliyon 2019, 5, e01690. [Google Scholar] [CrossRef]
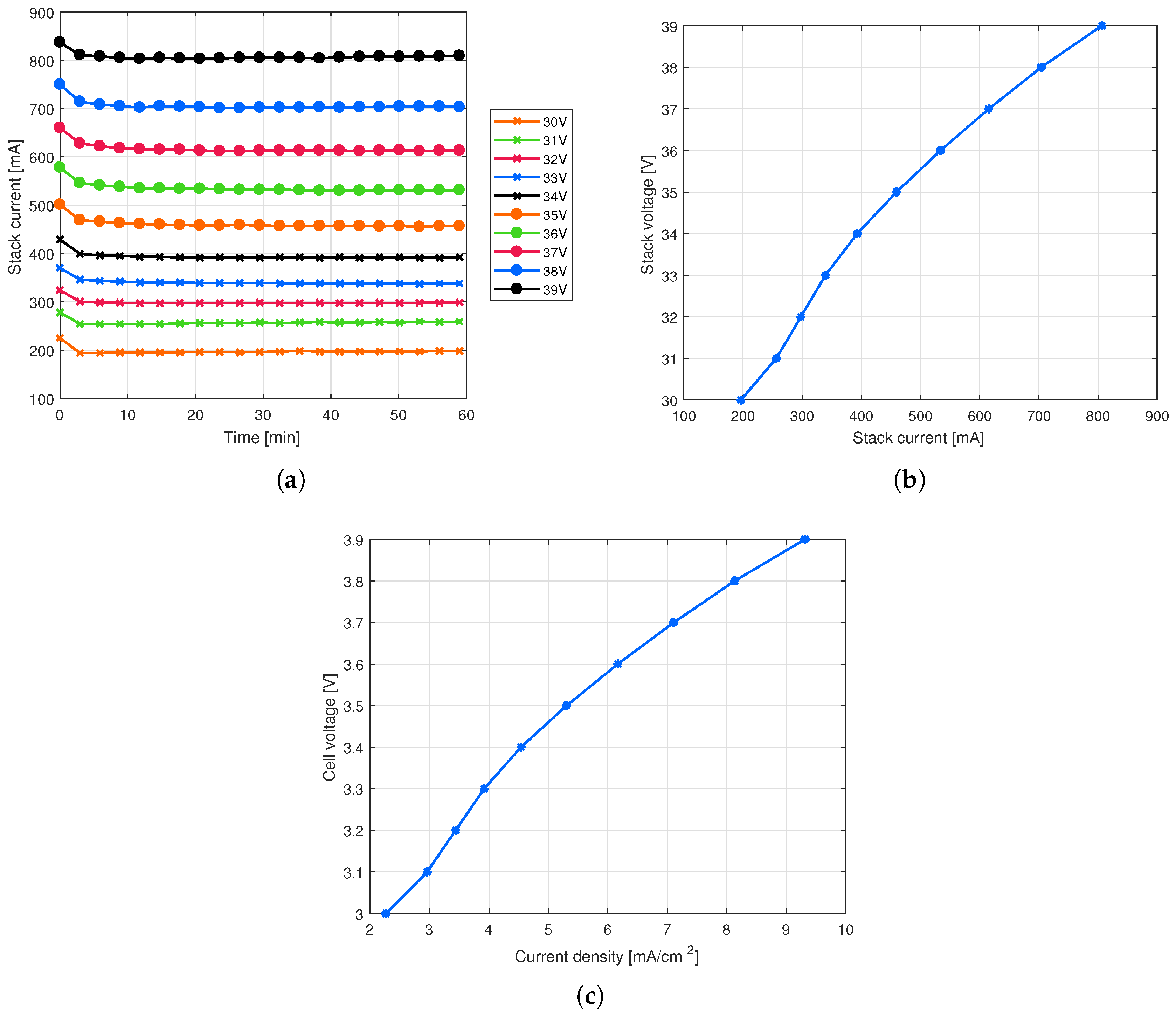
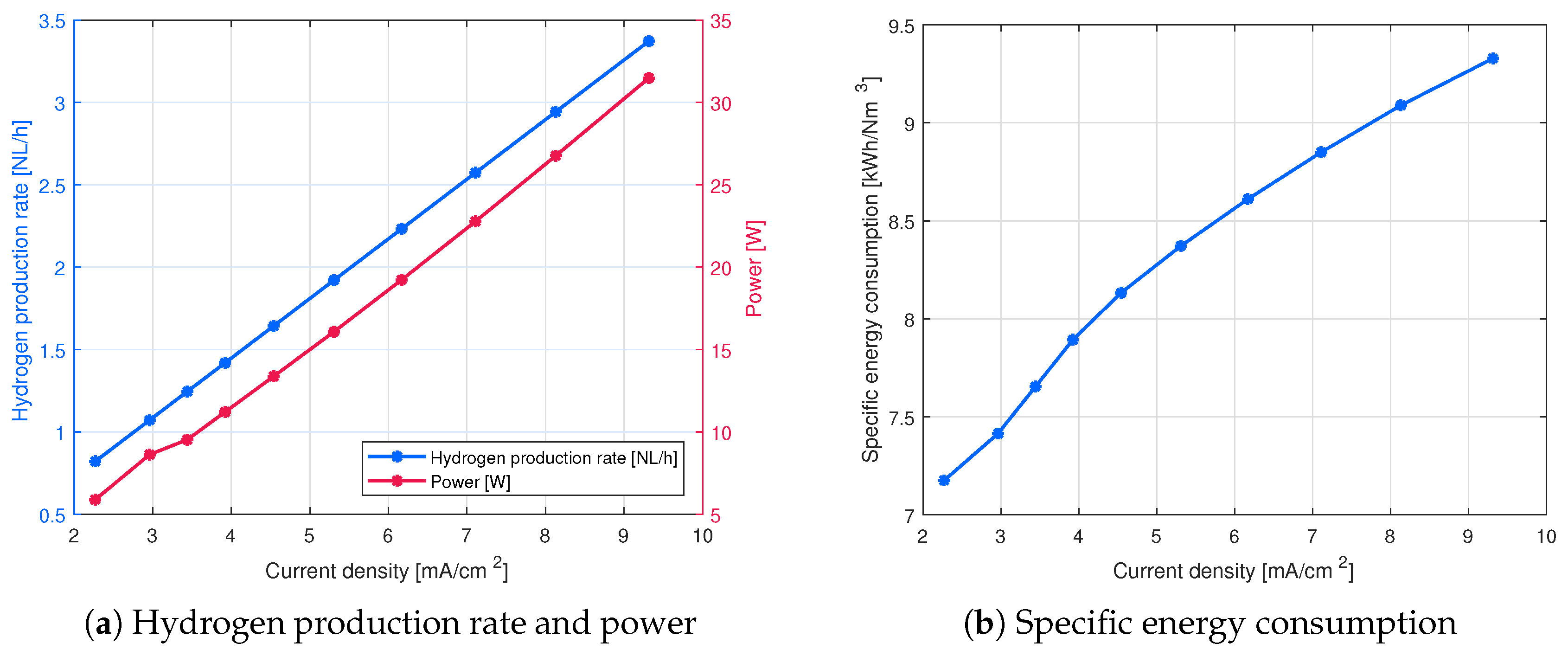
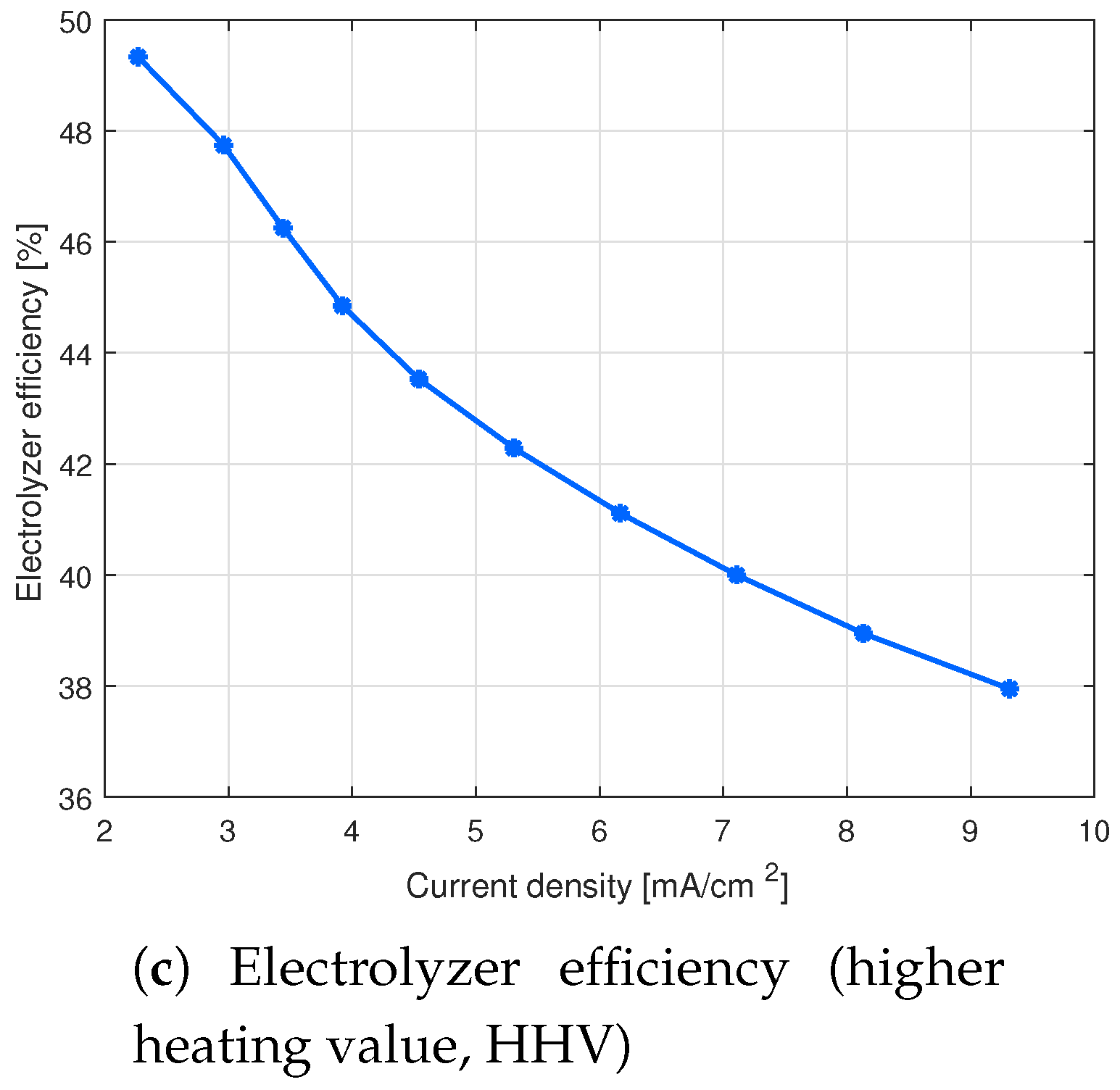
[V] | [A] | [NL/h] | [kWh/(Nm)] | [%] |
|---|---|---|---|---|
| 30 | 0.20 | 0.82 | 7.18 | 49.33 |
| 31 | 0.26 | 1.07 | 7.42 | 47.74 |
| 32 | 0.30 | 1.25 | 7.65 | 46.25 |
| 33 | 0.34 | 1.42 | 7.89 | 44.85 |
| 34 | 0.39 | 1.64 | 8.13 | 43.53 |
| 35 | 0.46 | 1.92 | 8.37 | 42.29 |
| 36 | 0.53 | 2.23 | 8.61 | 41.11 |
| 37 | 0.62 | 2.57 | 8.85 | 40.00 |
| 38 | 0.70 | 2.94 | 9.09 | 38.95 |
| 39 | 0.81 | 3.37 | 9.33 | 37.95 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Givirovskiy, G.; Ruuskanen, V.; Ojala, L.S.; Kokkonen, P.; Ahola, J. In Situ Water Electrolyzer Stack for an Electrobioreactor. Energies 2019, 12, 1904. https://doi.org/10.3390/en12101904
Givirovskiy G, Ruuskanen V, Ojala LS, Kokkonen P, Ahola J. In Situ Water Electrolyzer Stack for an Electrobioreactor. Energies. 2019; 12(10):1904. https://doi.org/10.3390/en12101904
Chicago/Turabian StyleGivirovskiy, Georgy, Vesa Ruuskanen, Leo S. Ojala, Petteri Kokkonen, and Jero Ahola. 2019. "In Situ Water Electrolyzer Stack for an Electrobioreactor" Energies 12, no. 10: 1904. https://doi.org/10.3390/en12101904
APA StyleGivirovskiy, G., Ruuskanen, V., Ojala, L. S., Kokkonen, P., & Ahola, J. (2019). In Situ Water Electrolyzer Stack for an Electrobioreactor. Energies, 12(10), 1904. https://doi.org/10.3390/en12101904






