Abstract
The pretreatment of seaweed by washing in freshwater is often used in seaweed biofuel research studies. However, the effect of washing seaweed prior to anaerobic digestion (AD) does not appear to have been greatly studied. This study examines washing Sargassum muticum with freshwater and its effect on ultimate and proximate analyses, salt content, methane production from anaerobic digestion, and leachate loss from ensiling. Washing with freshwater significantly (p < 0.01) increased moisture content (unwashed 85.6%, washed 89.1%) but significantly (p < 0.05) reduced ash (unwashed 32.7% dry weight dw, washed 30.6% dw) and salt content (unwashed ash containing 51.5%, washed 42.5%). The dry biomass higher heating value was significantly (p < 0.05) increased by washing due to the lower ash content (11.5 to 12.6 kJ g−1 dw). There was no significant change in the protein or lipid content, although washing increased the nitrogen content (3.85–4.77% dw). Washing significantly (p < 0.05) increased leachate losses during ensiling, with total leachate losses increasing after washing (12.7–25.2%). The methane yield from anaerobic digestion (28 days) was not statistically significantly different (p > 0.05) between unwashed (0.225 L CH4 g−1 VS) and washed samples (0.177 L CH4 g−1 VS). However, washing delayed biomethane production.
Keywords:
Sargassum muticum; washing; anaerobic digestion; seaweed; pretreatment; ensiling; macroalgae 1. Introduction
Seaweeds grow in brackish or salt water and, unlike terrestrial crops, do not require agricultural land for cultivation, thus avoiding competition for freshwater and land needed for food production [1]. The potential biomass yield can be higher for seaweed than for terrestrial plants per unit area. Brown seaweeds which are grown “under cultured conditions” can have yields of ~13.1 kg dry weight (dw) m−2 year−1 compared to the yield from sugarcane of only ~10 kg dw m−2 year−1 [2]. Growth systems that do not compete for land or freshwater, which are needed for crops, and high potential biomass yields have led to considerable research interest in the use of both micro- and macroalgae as sources of biofuel. Sargassum muticum, a brown seaweed which is an invasive species to Europe, is attracting research interest as a potential feedstock for biofuels and biorefineries [3,4,5,6].
Anaerobic digestion (AD) is generally the process of choice for energy production from high water content biomass, and many groups have reported that macroalgae are a suitable feedstock for AD [7]. Nevertheless, practical yields of biogas from the AD of seaweed are considerably below the theoretical maximum, and increasing CH4 yield is, therefore, the most critical factor in improving process energy balance and reducing greenhouse gas emissions [8]. A variety of biomass pretreatment methods, such as thermal, mechanical, thermochemical, and enzymatic treatments, have been shown to improve biomethane production by 19–68% [9]. Chisti [10], in a review of the constraints to the commercialisation of algal fuels, suggested saline algal biomass should be washed in freshwater to reduce the salt content, and washing in freshwater is a pretreatment step which is often used in a wide a variety of seaweed biofuel research studies [6,11,12,13,14,15,16,17,18,19]. However, the effect of washing seaweed prior to AD does not appear to have been greatly studied.
The washing of Ulva in freshwater does not change methane yield [20,21], despite a 2.5% decrease in volatile solids (VS) concentration and a 23% decrease in total solids (TS) content of the green alga, due to the removal of gravel and sand [21]. However, Adams et al. [22] reported that washing Laminaria digitata increased methane yield despite a reduction in soluble carbohydrate content, possibly because of the influence of the decreased salt content.
In four recent studies of ensiling seaweeds, three different washing treatments were used prior to ensiling: Redden et al. [23] washed with seawater; Herrmann et al. [24] and Cabrita et al. [25] washed with cold tap water; and Milledge and Harvey [26] did not wash the seaweed before ensiling. These differences in pretreatment could be a potential factor in the disparity in the loss of material (leaching) during ensiling between the four studies [23]. However, the species and environmental growth conditions may also have substantial effects. In three recent studies of the AD of S. muticum (Sargassum muticum), two studies did not wash the seaweed prior to digestion, and one did with seawater [5,26,27]. Despite these difference in washing pretreatment before ensilage and AD, there appears to have been little research on the washing pretreatment of seaweed in general and S. muticum in particular. This research, thus, examines the effect of freshwater washing on ensilage and anaerobic digestion.
2. Methods
2.1. Sample Collection
S. muticum was collected in June 2017 from Minis Bay, Kent, England (Ordnance Survey National Grid Map reference TR287697).
2.2. Sample Preparation
Unwashed seaweed: holdfasts together with chalk and other natural contaminants, such as mud, sand, chalk, small animals, and other seaweeds attached to S. muticum, were removed by hand without washing. The samples were bagged, frozen, and stored at −20 °C.
Washed seaweed: holdfasts together with chalk and other natural contaminants, such as mud, sand, chalk, small animals, and other seaweeds attached to S. muticum, were removed by hand. The remaining S. muticum was then washed in running tap water for 30 s and allowed to drain for 5 min. The samples were bagged, frozen, and stored at −20 °C.
2.3. Dry Weight Determination
The British Standards simplified oven drying method for the determination of moisture content in solid biofuels was used to establish moisture content [28]. All measurements were repeated in triplicate, and a mean value and standard deviation (SD) are reported. Samples after drying were stored in sealed containers at 4 °C for further experimentation.
2.4. Ash Determination
The British Standards method for determination of ash content in solid biofuels was used to establish the ash content of oven dried samples [29]. All measurements were carried out in triplicate, and a mean value is reported. Ash was also examined by X-ray diffraction (XRD) analysis after grinding in a pestle and mortar to a fine powder <10 μm.
2.5. Salt Content Determination
The Mohr silver nitrate and potassium chromate titration method was used to determine the salt (sodium chloride) content of the ashed samples [30,31]. A mean value is reported from two determinations per sample.
2.6. Elemental Analysis
Flash dynamic combustion (Flash EA1112 CHNS Elemental Analyser, Thermo Scientific, Waltham, MA, USA) was used to determine the carbon, hydrogen, nitrogen, and sulphur content of the dried seaweed biomass. The oxygen content was calculated by difference. A mean is reported from a minimum of two determinations per sample.
2.7. Protein Content
The protein content was determined in duplicate on 0.1 g freeze-dried samples of washed and unwashed seaweeds, crushed into a fine powder with liquid nitrogen, by the Lowry method using ovalbumin as the protein standard [32]. The blue colour of the final solution, the result of the presence of amino acids (mainly tyrosine and tryptophan), was quantified using absorbance at 750 nm with a UV-visible spectrophotometer (Jenway 6305, Bibby Scientific, Dunmow, Essex, UK).
2.8. Lipid Content
The lipid content was determined in triplicate for freeze-dried samples of washed and unwashed seaweed using a modified Bligh and Dyer method [33].
2.9. Calorific or High Heating Value Determination
Higher heating values (HHV) or gross calorific values (CV) were measured using the UKAS (United Kingdom Accreditation Service) method for determination of calorific value with a Parr Model 1341 Bomb Calorimeter [34]. Combustion in an adiabatic bomb containing oxygen under pressure oxidised the samples. The HHV was determined by measuring the temperature change in a known mass of water. The dissolved sulphate and nitrate were calculated from titration to adjust for their contribution. A mean is reported from a minimum of two determinations per sample.
2.10. Ensiling
Approximately 200 g of either washed or unwashed seaweed were placed in preweighed 300 mm × 200 mm food-grade composite (polyamide and polyethylene) bags (Andrew James Worldwide, Ferryhill, Co Durham. DL17 8JH), vacuum sealed (Andrew James Worldwide VS517), and weighed. The bags were then stored in the dark for 60 days in a temperature controlled cabinet at 20 °C ± 1 °C. At the end of the 60-day storage period, the bag was suspended by one corner with a 5-mm diameter hole in the upper corner of the bag releasing the vacuum. The lowest corner of the bag was cut to create a 30-mm opening. Leachate was allowed to drain from the bag for 10 min into a preweighed measuring cylinder via a funnel and 1-mm sieve. Then, the bag and its contents were weighed to calculate the mass of ensiled material. The funnel, sieve, and measuring cylinder were also weighed to calculate the mass of the leachate. The pH of the leachate was also measured (Hauna Instruments HI2210, Woonsocket, RI, USA).
2.11. Methane Potential Determination
The biomethane potential (BMP) of the washed and unwashed seaweed (excluding leachate) was analysed using a biomethane potential test system (CJC Labs Ltd., Nether Wasdale, Seascale, UK), shown in Figure 1. The equipment consists of a controlled temperature water bath with 8 × 1 L plastic digestion containers, with each digester connected via a CO2 fixing bottle to a tipping cup volumetric gas measuring device.

Figure 1.
CJC biomethane potential system (courtesy of CJC labs).
The 1 L digestion vessels were filled with inoculum and substrate, and made-up to a volume of 400 mL with deionised water. The inoculum was collected from an internal recirculation granular sludge anaerobic digester treating papermaking liquid waste at Smurfit Kappa Townsend Hook Paper Makers (Mill Street, Snodland, Kent, UK). Table 1 gives the analysis of the inoculum solids. Three experimental replicates using 10 g wet weight of each variant, at an inoculum-to-substrate ratio of 9:1 on a volatile solids basis, were carried out, together with a control containing no substrate but containing inoculum.

Table 1.
Ash and CHONS content of inoculum [26].
After filling the digesters, the headspace was flushed with nitrogen, and the digestion bottles were sealed. The digesters were incubated for 28 days at a mesophilic temperature of 37 °C in a water bath. The contents of each digester were continuously mixed throughout the test by a slowly rotating paddle (~40 rpm). Biogas from each digester was passed through a fixing bottle containing 80 mL of 3 M NaOH solution (containing thymolphthalein indicator) for fixation of carbon dioxide. The resultant methane produced was subsequently measured in a tipping cup volumetric gas measuring device submerged in deionised water. Methane volume, pressure, and temperature data were recorded continuously, and gas volumes were normalised (100 kPa, 0 °C, dry gas).
The pH was measured (Hauna Instruments HI221) for each sample at the beginning and end of the BMP test.
2.12. Statistical Analysis
IBM SPSS Statistics (version 23) was used for two-way analysis of variance (ANOVA) with data tests for skewness (0.5 to −0.5), kurtosis (1 to −1), normality (Kolmogorov–Smirnov (>0.05), and Shapiro–Wilks (>0.05). A two-way ANOVA was performed to examine the effect washing and time and their interaction on daily cumulative methane production from the BMP test.
Microsoft Excel 2013was used for one-way ANOVA and all other statistical analyses. One-way ANOVAs were conducted to compare the effect of washing on water, ash, salt, lipid content, protein content, and calorific value.
3. Results
3.1. Dry Weight Determination
The moisture content of the unwashed 85.6% (SD 0.32%) and washed samples 89.1% (SD 0.82%) were statistically different (p < 0.01), with the washed containing 4% more moisture than the unwashed.
3.2. Ash and Salt Determination
The ash content of the washed sample (30.6%; SD 1.05%) was significantly lower (p < 0.05) than the unwashed samples (32.7%; SD 0.58%).
The results of the XRD analyses are shown in Figure 2 and Figure 3. Washing reduced chloride salts, both sodium and potassium, and inorganic sulphates (sodium carbonate sulphate and magnesium sulphate hydrate), but water hardness salts were increased (calcite). The Mohr salt analysis (NaCl) also showed that the washing significantly (p < 0.05) reduced salt, with the unwashed ash containing 51.5% (SD 1.4%) and the washed ash containing 42.5% (SD 3.5%).
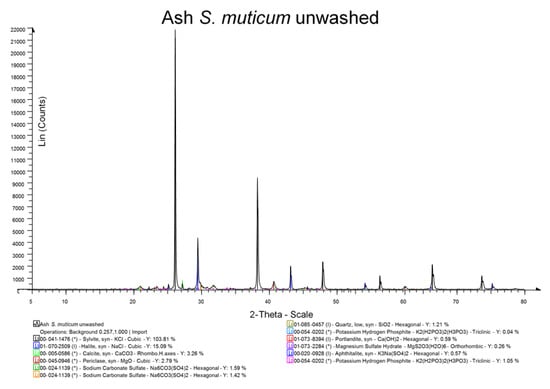
Figure 2.
XRD analysis of unwashed Sargassum muticum.
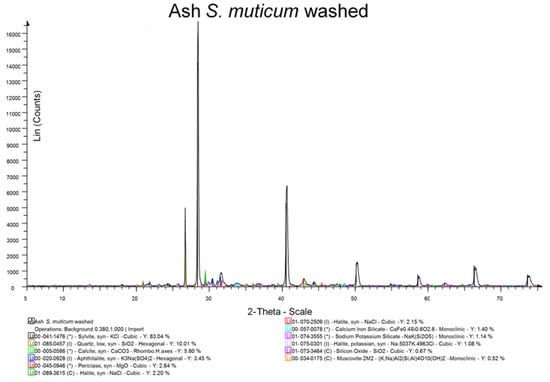
Figure 3.
XRD analysis of unwashed S. muticum.
3.3. Elemental Analysis
The results of the average values of the elemental analysis are shown in Table 2.

Table 2.
Elemental composition of S. muticum before and after freshwater washing.
3.4. Lipid and Protein
There is no statistical difference (p > 0.05) in the lipid or protein contents of the washed and unwashed samples (Table 3). On an ash-free dry weight (afdw) basis, the average lipid content is 7.7% for the unwashed and 9.5% for the washed, and the average protein content is 23.9% for the unwashed and 20.9% for the washed. Carbohydrate content (including fibre) is often calculated by difference (total less ash, protein, and lipid). The overall carbohydrate figures are thus 46.1% dw (68.4% afdw) for the unwashed and 48.3% dw (69.6% afdw) for the washed.

Table 3.
Lipid and protein contents of S. muticum before and after freshwater washing.
3.5. High Heating Value
There is a statistically significant (p < 0.05) difference between the HHV values (Table 4) of unwashed and washed S. muticum.

Table 4.
The higher heating value of S. muticum before and after freshwater washing.
3.6. Ensiling
The pH of the seaweed after 60 days ensiling is shown in Table 5. There was little gas production in any of the ensiling bags except for one bag of unwashed material where 300 mL of gas was removed by a gas syringe prior to opening the bag. Bubbling this gas through 3 M NaOH removed 40% of the volume of the gas. Visually, there appeared to be more liquid in the bags containing washed seaweed compared to the unwashed samples, and the washed materials were greener in colour than the brown unwashed ensiled seaweed.

Table 5.
pH of washed and unwashed S. muticum after ensiling for 60 days (n = 3).
The ultimate and proximate analysis of ensiled, both unwashed and washed, S. muticum is shown in Table 6.

Table 6.
Ultimate and proximate analysis for unensiled and ensiled S. muticum and the leachate produced from ensiling with and without washing pretreatment.
Leachate total losses (wet weight) as a percentage of the initial wet weights were 12.7% (SD 1.66%) for the unwashed, which were significantly lower (p < 0.05) than that for the washed sample 25.2% (SD 4.63%)
3.7. Methane Production
There was no significant difference (p > 0.05) in the final pH after 28 days in the digestion vessels between the unwashed samples (7.35; SD 0.02) and the washed sample (7.34; SD 0.01).
The total methane yield after 28 days from the unwashed sample was 0.225 L CH4 g−1 VS (SD 0.046) and 0.177 L CH4 g−1 VS (SD 0.085) for the washed. Although this is a reduction of 21% in methane yield, the difference in methane yield between the washed and unwashed samples was not statistically significant (p > 0.05). Figure 4 shows the net biomethane production of gas production over 28 days. A two-way ANOVA to examine the effect of washing and time and their interaction on daily cumulative methane production from the BMP test found that washing, time, and the interaction between washing and time all had a statistically significant effect on cumulative methane yield. It would appear that washing modifies the time course of biomethane production by significantly delaying the initial rate of microbial digestion to produce biomethane, but washing does not statistically significantly change the final total (ultimate) methane yield.
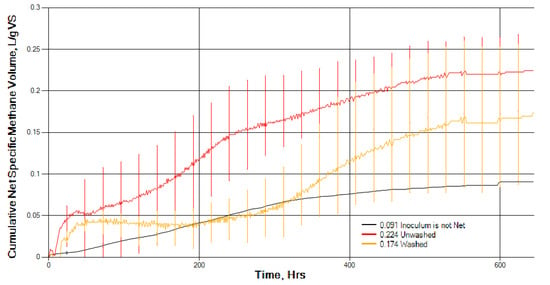
Figure 4.
Net cumulative biomethane yield for unwashed and washed S. muticum (error bars = maximum and minimum readings n = 3).
4. Discussion
4.1. Algal Composition
Table 7 shows a comparison of the current, proximate, and ultimate analyses and higher heating values (HHV) with previously published data for unwashed S. muticum collected from the Kent coast. The current samples are higher in salt and lower in organic sulphur than previously collected seaweed and have a lower HHV. However, the HHV is similar to that reported for beach-cast Sargassum spp. 11.4 kJ g−1 dw [35].

Table 7.
Ultimate and proximate analysis of unwashed S. muticum collected from the Kent coast [26,36] and the current study.
There appears to be an increase in ash and a reduction in nitrogen (protein) during the growing season from March to July. In a study of the composition S. muticum from Galicia, northwest Spain, the values for ash were lowest from March till May and highest from June till October [37]. Wernberg et al. [38] found that, although the carbon content of S. muticum was found to be relatively constant at ~30%, the nitrogen content varies throughout the growing season, being highest in the spring (3–4%) and lowest in the autumn (1–2%). Jard et al. (2013) found the nitrogen content of S. muticum harvested in Brittany, France during July at between 1% and 1.7% dw.
The protein content of a biomass can be estimated from the nitrogen content using an N factor which is based on the types of proteins present in the biomass. A great many commonly occurring proteins contain ~16% nitrogen, and a factor of 6.25 has been most frequently used [39]. The N factor of 6.25 has been widely used in the algal literature [40,41]. A proportion of nitrogen that is not associated with proteins, but with compounds such as DNA, pigments, and free amino acids, may be present in the biomass [42]. Thus, the commonly used multiplier of 6.25 can cause the protein content to be overestimated [42,43] Factors used for common foods vary between 5.2 and 6.3 [39] and for algae between 3.75 and 6.4 [42,43]. Angell et al. [41] have suggested a general factor of 4.56 for brown seaweed. The protein content for unwashed S. muticum measured by the Lowry method was 16.1% and the N content was 3.9%, indicating an N factor of 4.1. Gonzalez-Lopez et al. [6] found the typical average protein content of S. muticum to be 6.9% of the total dry weight, but Hardouin et al. [44] reported protein levels of 29%. The protein content of S. muticum growing in UK waters was found to be highest in April and May [45], and Balboa et al. [37] found the protein content was lowest from May to June (7%) and highest from November to March (11%) (Total protein content was estimated by Kjeldahl total nitrogen using a nitrogen factor of 5.38).
The lipid content of the unwashed seaweed of 5.2% is towards the upper-end of that typical of brown seaweed (0.3–6%) [46,47,48] and above that previously reported for S. muticum of <1.3% [44,49] and 1.6–3.2% [37].
Effect of Washing on Algal Composition
Washing in freshwater significantly (p < 0.05) reduced the ash and NaCl content (dw) of S. muticum but increased moisture (p < 0.01). The XRD analysis confirmed there was a loss of inorganic chlorides, but there was an increase in water hardness salts (calcite). Washing reduced inorganic sulphates but had little effect on the organic sulphur content (1.0% unwashed and 1.1% washed afdw basis). Adams et al. [22] also found that freshwater washing reduced biomass ash content with the ash content of Laminaria digitate collected in winter, reducing from 30.3% to 25.5% of total solids. Díaz-Vázquez et al. [35] also found that washing reduced the ash content of Sargassum spp. from 27.5% to 12.5% dw. These findings confirm the view of Chisti [10] that freshwater washing reduces the salt content and may be useful for fuel production methods where salt is a potential inhibitor. Although the changes in carbon content due to washing on a dry weight basis are relatively small (<1%) (Table 2), the increase in moisture content after freshwater washing results in a 24% reduction in TS in the wet feedstock. As a result, the carbon content of the washed biomass reduced by 25% on a wet weight basis, from 3.89% for the unwashed material to 2.93% for the washed. This 25% reduction of carbon content in the wet seaweed feedstock following washing may make freshwater washing impractical on an industrial scale, despite the reduction in salt content. Not only will the overall costs increase due to the cost of the washing process itself but also because of the increased cost of downstream processing as a result of the larger mass of wet material that has to be handled for the same carbon content (methane potential).
Nitrogen content increased on washing from 3.9% to 4.8% on a dry weight basis and from 5.7% to 6.9% on an afdw basis, but protein content decreased from 16.1% to 14.5% on a dry weight basis and from 23.9% to 20.9% on an afdw basis. However, none of these changes were statistically different (p > 0.05). Nevertheless, the results may indicate that there is a change in the relative proportions of amino acids and other organic nitrogenous compounds. Díaz-Vázquez et al. [35] found a reduction in protein from 11.3% to 10.9% on a dry weight basis using a bicinchoninic acid (BCA) assay. The sensitivity of BCA and Lowry colorimetric methods is similar [50]. However, both rely on the use of a protein calibration standards [42], and they required a protein standard for calibration that should have a similar level of the amino acids causing the change (primarily tyrosine and tryptophan for Lowry, and arginine, lysine, and histidine for Bradford) to that found in seaweed biomass. The selection of an appropriate protein standard representative of the sample protein to be measured is, therefore, crucial and further research on the amino acid composition of S. muticum is required together with studies on the effect of washing on the fixed and free amino acids of seaweed.
The lipid content increased on washing from 5.2% to 6.6%, although it was not statically significant. Díaz-Vázquez et al. [35] also found the lipid content of Sargassum spp. biomass increased on freshwater washing from 0.3% ± 0.1% to 0.7% ± 0.3%. This could be due to the greater loss of polar compounds during washing relative to nonpolar compounds. The HHV increased significantly (p < 0.05; n = 3) after washing, from 11.5 kJ g−1 (SD 0.26) to 12.6 kJ g−1 (SD 0.65), and reflects the increased proportion of lipid and reduced ash content.
4.2. Ensiling
The final pH of the unwashed sample was 6.41 (SD 0.80), which is considerably higher than that previously found for ensilage of S. muticum (5.05; SD 0.49) [26]. However, one of the triplicates appeared to have undergone ‘full’ anaerobic digestion rather than ensilage, with a gas production of ~300 mL and a CO2 content of 40%. If this sample is removed, the revised average pH is 5.96 (SD 0.21), which is still higher than the previous study. The final pH values in this study were higher than those found in other studies on ensiling a variety of different species of seaweed: pH 3.2–4.0 [23], 4.7 [51], 4–5.7 [24], and 4.48–5.15 [25]. There are considerable differences in the final pH achieved after ensiling between different species of seaweed used in the various studies. In addition to the effect of species, this current study and the previous ensiling study on S. muticum may also indicate that there are seasonal variations, which could be due to differences in the biochemical composition of the seaweed and/or composition of the natural bacteria present. However, considerably more research is required.
A pH < 4.3 has been recommended for successful grass silage [52], and this study did not achieve that. Seaweed silage has a high water content relative to typical terrestrial forage crops, and the required pH may be lower than that recommended for grass; clostridial fermentation and the production of butyric acid is likely to occur. The buffering capacities of the seaweeds are higher than terrestrial forages, probably due to the high anion content [25], and thus higher lactic acid production may be required to achieve a sufficiently low pH in seaweed to ensure the necessary low pH to prevent clostridial fermentation.
The silage-making process in terrestrial crops can be enhanced by the addition of Lactobacillus, such as L. plantarum, resulting in a more rapid pH reduction and greater product stability [53,54]. Mixtures of Lactobacillus and proprietary bacterial strains are routinely used commercially in silage making from land-based forage crops. Redden et al. [23] found the pH of L. plantarum after 30 and 365 days ensiling was less for the samples treated by spraying with a culture of Lactobacillus plantar. The addition of a Lactobacillus culture may be beneficial in further reducing pH and inconsistencies between batches because of variations in natural bacteria. Conversely, Cabrita et al. [25] found the use of a lactic acid bacteria inoculant before the ensiling of three seaweed species (Gracilaria vermiculophylla, Ulva rigida, and Saccharina latissima) had only minor effects on fermentation. However, these two studies used different washing pretreatments; Redden et al. [23] used seawater, and Cabrita et al. [25] used freshwater.
Leachate losses for the unwashed samples (12.7%) during ensilage were higher than those previously found for S. muticum from the Kent coast (8.1%) [26]. When pH is not quickly lowered, clostridial fermentation of lactic acid and water-soluble carbohydrates to butyric acid and breakdown of proteins by proteolytic clostridia can occur [25]. Hence, the higher leachate loss in this study may be the result of bacterial degradation as a result of the higher pH. One sample of unwashed seaweed produced 300 mL of gas, of which 40% of the volume of the gas was dissolved in NaOH, potentially indicating anaerobic digestion, and this may be due to higher pH and the further breakdown by acetogenic and methanogenic bacteria.
Unlike the previous study of ensiling S. muticum [26], there was no loss of organic sulphur. The ensilage in both of these studies relied only on the naturally present bacteria on the seaweed, and a reduction in naturally present sulphur-reducing bacteria may be one potential reason for the low loss of organic sulphur in the current trial. Considerably more research is required on the seasonal and environmental changes in the bacteria associated with seaweed.
Effect of Washing on Ensilage
Leachate losses from ensiling were statistically significantly higher (p < 0.05) after freshwater washing pretreatment (25.2%; SD 4.63%) relative to unwashed S. muticum (12.7%; SD 1.7%). However, the ash and salt contents of the unwashed leachate were significantly higher (p < 0.001) than the washed (Table 6), probably as a result of ash and salt being previously removed in the freshwater washing. Despite the greater loss of leachate in the washed sample, the moisture content of the washed biomass remained statistically significantly higher (p < 0.001) than the unwashed biomass after ensiling. The ash content of the unwashed biomass remained statistically higher (p < 0.001) than washed biomass after ensiling even though the ash content of the unwashed leachate was higher than that of the washed material. The higher heating value of the washed sample remained statistically higher (P < 0.005) than that of the unwashed material after ensiling. In summary, the biomass of the washed sample remained wetter, with its biomass have a higher heating value and lower ash content than unwashed material.
4.3. Anaerobic Digestion
The average BMP of unwashed S. muticum was 0.225 L CH4 g−1 VS, which was higher than that found in other recent studies (0.10–0.13 L CH4 g−1 VS) [5,26,27]. Jard et al. [27] pretreated seaweed by washing in seawater and Soto et al. [5] oven dried their seaweed, and these differences in pretreatment may be a factor in the lower methane yields. There were also differences in the degree of mixing during the BMP test. This study used continuous paddle mixing, whereas, in the previous study by Milledge and Harvey [26], the contents were mixed by a slowly rotating rod agitator at 30 rpm, operating for 60 s at a time interval of 60 s. Jard et al. [27] shook the reactors regularly throughout the incubation period, and Soto et al. [5] did not describe any mixing. Mixing method and mixing intensity have direct effects on the biogas yield, with mixing tending to increase biogas yield [55,56,57,58].
Effect of Washing on Anaerobic Digestion
Washing does not have a statistically significant effect on the final methane yield, but it does delay the initial rate of biomethane production, resulting in a significant delay in peak methane production. This may be due to a reduction in easily metabolised organic compounds (soluble carbohydrates). Adams et al. [22] reported that freshwater washing L. digitata reduced its soluble carbohydrate content, and Bruhn et al. [21] found a 2.5% decrease in volatile solids concentration after washing Ulva with freshwater.
Like other eukaryotic organisms, macroalgae harbour a rich diversity of associated nonparasitic microorganisms with functions related to host health and defence [59,60,61], and the removal of potential natural hydrolytic bacteria could be another potential reason for delayed gas production following freshwater washing. However, further research is required. Additional work is being considered to extend the current study to determine which natural bacteria are present initially on seaweed and how preprocessing affects them, using techniques such as 16S RNA analysis.
The ash content of seaweed biomass was reduced by washing. Trace elements such as cobalt and selenium have a considerable effect on AD [62], and thus the change in the mineral content of the seaweed after washing may be a further contributory factor in the delay in methane production, but again, considerable further study is required.
5. Conclusions
Washing in freshwater seems to have an effect on the composition of seaweed and must be taken into account when comparing research studies. Although washing seaweeds in freshwater prior to anaerobic digestion can reduce salt, an inhibitor of AD, it will add to the overall processing costs. Washing S. muticum did not affect overall BMP gas production during AD, but it did significantly slow down the initial rate of biomethane production. This could be due to the removal of readily digested soluble molecules in washing and/or the removal of naturally present hydrolytic bacteria from the surface of the seaweed. However, further research work appears to be warranted on the effect of washing on seaweed composition, particularly amino acids and carbohydrates, and natural bacterial biota and its effect on ensilage and anaerobic digestion.
Author Contributions
Conceptualisation, J.J.M. and B.V.N.; Methodology, J.J.M. and B.V.N.; Statistical Analysis, J.J.M. and B.V.N.; Experimental Investigation, J.J.M., B.V.N., and M.S.S.; Resources, B.V.N. and P.J.H.; Data Analysis, J.J.M.; Writing—Original Draft Preparation, J.J.M.; Writing—Review & Editing, J.J.M., B.V.N., and P.J.H.
Funding
This research received no external funding.
Acknowledgments
The author would like to acknowledge the financial support of the University of Greenwich, Andrew Hurt for assistance with the XRD and XRF analyses, and Debbie Bartlett for the collection of the seaweed.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Milledge, J.J.; Harvey, P.J. Potential process ‘hurdles’ in the use of macroalgae as feedstock for biofuel production in the British Isles. J. Chem. Technol. Biotechnol. 2016, 91, 2221–2234. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, R.; Yaakob, Z.; Takriff, M.S. Potential of the micro and macro algae for biofuel production: A brief review. BioResources 2014, 9, 1606–1633. [Google Scholar] [CrossRef]
- Milledge, J.J.; Nielsen, B.V.; Bailey, D. High-value products from macroalgae: the potential uses of the invasive brown seaweed, Sargassum muticum. Rev. Environ. Sci. Biotechnol. 2015, 15, 67–88. [Google Scholar] [CrossRef]
- Balboa, E.; Moure, A.; Domínguez, H. Valorization of Sargassum muticum biomass according to the biorefinery concept. Mar. Drugs 2015, 13, 3745–3760. [Google Scholar] [CrossRef] [PubMed]
- Soto, M.; Vazquez, M.A.; de Vega, A.; Vilarino, J.M.; Fernandez, G.; de Vicente, M.E. Methane potential and anaerobic treatment feasibility of Sargassum muticum. Bioresour. Technol. 2015, 189, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Lopez, N.; Moure, A.; Dominguez, H. Hydrothermal fractionation of Sargassum muticum biomass. J. Appl. Phycol. 2012, 24, 1569–1578. [Google Scholar] [CrossRef]
- Sutherland, A.; Varela, J. Comparison of various microbial inocula for the efficient anaerobic digestion of Laminaria hyperborea. BMC Biotechnol. 2014, 14, 7. [Google Scholar] [CrossRef] [PubMed]
- Mayfield, S.P. Consortium for Algal Biofuel Commercialization (CAB-COMM) Final Report. Available online: https://www.osti.gov/servlets/purl/1234700 (accessed on 12 May 2016).
- Barbot, Y.; Thomsen, C.; Thomsen, L.; Benz, R. Anaerobic digestion of Laminaria japonica waste from industrial production residues in laboratory- and pilot-scale. Mar. Drugs 2015, 13, 5947–5975. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y. Constraints to commercialization of algal fuels. J. Biotechnol. 2013, 167, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, M.; Nakamura, K.; Ariga, O.; Nakasaki, K. Production of high concentrations of bioethanol from seaweeds that contain easily hydrolyzable polysaccharides. Process Biochem. 2011, 46, 2111–2116. [Google Scholar] [CrossRef]
- Wang, X.; Liu, X.; Wang, G. Two-stage hydrolysis of invasive algal feedstock for ethanol fermentation. J. Integr. Plant Biol. 2011, 53, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yoon, J.J.; Park, H.D.; Lim, D.J.; Kim, S.H. Anaerobic digestibility of algal bioethanol residue. Bioresour. Technol. 2012, 113, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Jones, J.M.; Kubacki, M.L.; Bridgeman, T. Classification of macroalgae as fuel and its thermochemical behaviour. Bioresour. Technol. 2008, 99, 6494–6504. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Gupta, R.; Kumar, G.; Sahoo, D.; Kuhad, R.C. Bioethanol production from Gracilaria verrucosa, a red alga, in a biorefinery approach. Bioresour. Technol. 2013, 135, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Choi, J.W.; Suh, D.J.; Ha, J.M.; Hwang, J.W.; Jung, H.W.; Lee, K.Y.; Woo, H.C. Production of brown algae pyrolysis oils for liquid biofuels depending on the chemical pretreatment methods. Energy Convers. Manag. 2014, 86, 371–378. [Google Scholar] [CrossRef]
- McKennedy, J.; Sherlock, O. Anaerobic digestion of marine macroalgae: A review. Renew. Sustain. Energy Rev. 2015, 52, 1781–1790. [Google Scholar] [CrossRef]
- Suutari, M.; Leskinen, E.; Fagerstedt, K.; Kuparinen, J.; Kuuppo, P.; Blomster, J. Macroalgae in biofuel production. Phycol. Res. 2015, 63, 1–18. [Google Scholar] [CrossRef]
- Tabassum, M.R.; Xia, A.; Murphy, J.D. The effect of seasonal variation on biomethane production from seaweed and on application as a gaseous transport biofuel. Bioresour. Technol. 2016, 209, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Nikolaison, L.; Dahl, J.; Bech, K.S.; Bruhn, A.; Rasmussen, M.B.; Bjerre, A.B.; Nielsen, H.B.; Ambus, P.; Rost, K.A.; Kadar, Z.; et al. Energy Production from Macroalgae. In Proceedings of the 20th European Biomass Conference and Exhibition, Milan, Italy, 18–22 June 2012; pp. 91–93. [Google Scholar]
- Bruhn, A.; Dahl, J.; Nielsen, H.B.; Nikolaisen, L.; Rasmussen, M.B.; Markager, S.; Olesen, B.; Arias, C.; Jensen, P.D. Bioenergy potential of Ulva lactuca: Biomass yield, methane production and combustion. Bioresour. Technol. 2011, 102, 2595–2604. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.M.M.; Schmidt, A.; Gallagher, J.A. The impact of sample preparation of the macroalgae Laminaria digitata on the production of the biofuels bioethanol and biomethane. J. Appl. Phycol. 2015, 27, 985–991. [Google Scholar] [CrossRef]
- Redden, H.; Milledge, J.J.; Greenwell, H.C.; Dyer, P.W.; Harvey, P.J. Changes in higher heating value and ash content of seaweed during ensiling. J. Appl. Phycol. 2016, 29, 1037–1046. [Google Scholar] [CrossRef]
- Herrmann, C.; FitzGerald, J.; O'Shea, R.; Xia, A.; O'Kiely, P.; Murphy, J.D. Ensiling of seaweed for a seaweed biofuel industry. Bioresour. Technol. 2015, 196, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, A.R.J.; Maia, M.R.G.; Sousa-Pinto, I.; Fonseca, A.J.M. Ensilage of seaweeds from an integrated multi-trophic aquaculture system. Algal Res. 2017, 24, 290–298. [Google Scholar] [CrossRef]
- Milledge, J.J.; Harvey, P.J. Ensilage and anaerobic digestion of Sargassum muticum. J. Appl. Phycol. 2016, 28, 3021–3030. [Google Scholar] [CrossRef]
- Jard, G.; Marfaing, H.; Carrere, H.; Delgenes, J.P.; Steyer, J.P.; Dumas, C. French Brittany macroalgae screening: Composition and methane potential for potential alternative sources of energy and products. Bioresour. Technol. 2013, 144, 492–498. [Google Scholar] [CrossRef] [PubMed]
- British Standards Institution (BSI). Solid biofuels. Determination of moisture content. Oven dry method. Total moisture. Simplified method. In BS EN 14774-2: 2009; BSI: London, UK, 2009. [Google Scholar]
- BSI. Solid biofuels -determination of ash content. In BS EN 14775: 2009; BSI: London, UK, 2009. [Google Scholar]
- Pearson, D. Laboratory Techniques in Food Analysis; Butterworth: London, UK, 1973. [Google Scholar]
- Skoog, D.A. Fundamentals of Analytical Chemistry 8th; International Student, Ed.; Thomson/Brooks/Cole: London, UK, 2004. [Google Scholar]
- Redmile-Gordon, M.A.; Armenise, E.; White, R.P.; Hirsch, P.R.; Goulding, K.W.T. A comparison of two colorimetric assays, based upon Lowry and Bradford techniques, to estimate total protein in soil extracts. Soil. Biol. Biochem. 2013, 67, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Matyash, V.; Liebisch, G.; Kurzchalia, T.V.; Shevchenko, A.; Schwudke, D. Lipid extraction by methyl-tert-butyl ether for high-throughput lipidomics. J. Lipid Res. 2008, 49, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- British Standards Institution (BSI). Determination of the gross heat of combustion (calorific value). In BS EN ISO 1716: 2010; BSI: London, UK, 2010. [Google Scholar]
- Díaz-Vázquez, L.M.; Rojas-Pérez, A.; Fuentes-Caraballo, M.; Robles-Ramos, I.V.; Jena, U.; Das, K.C. Demineralization of Sargassum spp. macroalgae biomass: selective hydrothermal liquefaction process for bio-oil production. Front. Energy Res. 2015, 3, 1–11. [Google Scholar]
- Milledge, J.J.; Staple, A.; Harvey, P. Slow Pyrolysis as a Method for the Destruction of Japanese Wireweed, Sargassum muticum. Environ. Nat. Resour. Res. 2015, 5, 28–36. [Google Scholar] [CrossRef]
- Balboa, E.M.; Gallego-Fábrega, C.; Moure, A.; Domínguez, H. Study of the seasonal variation on proximate composition of oven-dried Sargassum muticum biomass collected in Vigo Ria, Spain. J. Appl. Phycol. 2016, 28, 1943–1953. [Google Scholar] [CrossRef]
- Wernberg, T.; Thomsen, M.S.; Staehr, P.A.; Pedersen, M.F. Comparative phenology of Sargassum muticum and Halidrys siliquosa (Phaeophyceae: Fucales) in Limfjorden, Denmark. Bot. Mar. 2001, 44, 31–39. [Google Scholar] [CrossRef]
- Merrill, A.L.; Watts, B.K. Energy Values of Foods: Basis & Duration; Slight Revised February 1973; US Department of Agriculture: Washington, DC, USA, 1955.
- Lourenco, S.O.; Barbarino, E.; Lavin, P.L.; Marque, U.M.L.; Aidar, E. Distribution of intracellular nitrogen in marine microalgae: Calculation of new nitrogen-to-protein conversion factors. Eur. J. Phycol. 2004, 39, 17–32. [Google Scholar] [CrossRef]
- Angell, A.R.; Mata, L.; Nys, R.; Paul, N.A. The protein content of seaweeds: a universal nitrogen-to-protein conversion factor of five. J. Appl. Phycol. 2015, 28, 511–524. [Google Scholar] [CrossRef]
- González López, C.V.; Garcia, M.D.C.; Fernandez, F.G.A.; Bustos, C.S.; Chisti, Y.; Sevilla, J.M.F. Protein measurements of microalgal and cyanobacterial biomass. Bioresour. Technol. 2010, 101, 7587–7591. [Google Scholar] [CrossRef] [PubMed]
- Safi, C.; Charton, M.; Pignolet, O.; Silvestre, F.; Vaca-Garcia, C.; Pontalier, P.-Y. Influence of microalgae cell wall characteristics on protein extractability and determination of nitrogen-to-protein conversion factors. J. Appl. Phycol. 2013, 25, 523–529. [Google Scholar] [CrossRef]
- Hardouin, K.; Burlot, A.S.; Umami, A.; Tanniou, A.; Stiger-Pouvreau, V.; Widowati, I.; Bedoux, G.; Bourgougnon, N. Biochemical and antiviral activities of enzymatic hydrolysates from different invasive French seaweeds. J. Appl. Phycol. 2014, 26, 1029–1042. [Google Scholar] [CrossRef]
- Gorham, J.; Lewey, S.A. Seasonal changes in the chemical composition of Sargassum muticum. Mar Biol. 1984, 80, 103–107. [Google Scholar] [CrossRef]
- Tiwari, B.; Troy, D. Seaweed Sustainability: Food and Non-Food Applications, 1st ed.; Academic Press: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Lenstra, W.J.; Hal, J.W.V.; Reith, J.H. Economic aspects of open ocean seaweed cultivation. In Proceedings of the Energy Research Center of the Netherlands, Montpellier, France, 7–10 November 2011. [Google Scholar]
- Streefland, M. Algae and Aquatic Biomass for a Sustainable Production of 2nd Generation Biofuels; Deliverable 1.5-Report on Biofuel Production Processes from Micro, Macroalgae and other Aquatic; AquaFUELs: Brussels, Belgium, 2010. [Google Scholar]
- Shekhar, S.H.S.; Lyons, G.; McRoberts, C.; McCall, D.; Carmichael, E.; Andrews, F.; McCormack, R. Brown seaweed species from Strangford Lough: Compositional analyses of seaweed species and biostimulant formulations by rapid instrumental methods. J. Appl. Phycol. 2012, 24, 1141–1157. [Google Scholar] [CrossRef]
- Vrsanska, M.; Kumbar, V. A comparison of Biuret, Lowry and Bradford methods for measuring the egg's proteins. Mendel Net 2015, 22, 394–398. [Google Scholar]
- Black, W.A.P. The preservation of seaweed by ensiling and bactericides. J. Sci. Food Agric. 1955, 6, 14–23. [Google Scholar] [CrossRef]
- Genever, L. Making Grass Silage for Better Returns; Agriculture and Horticulture Development Board (AHDB): Huntingdon, UK, 2011. [Google Scholar]
- Wang, M.S.; Yang, C.H.; Jia, L.J.; Yu, K.F. Effect of Lactobacillus buchneri and Lactobacillus plantarum on the fermentation characteristics and aerobic stability of whipgrass silage in laboratory silos. Grassl. Sci. 2014, 60, 233–239. [Google Scholar] [CrossRef]
- Davies, D.R.; Merry, R.J.; Williams, A.P.; Bakewell, E.L.; Leemans, D.K.; Tweed, J.K. Proteolysis during ensilage of forages varying in soluble sugar content. J. Dairy Sci. 1998, 81, 444–453. [Google Scholar] [CrossRef]
- Lindmark, J.; Thorin, E.; Bel Fdhila, R.; Dahlquist, E. Effects of mixing on the result of anaerobic digestion: Review. Renew. Sustain. Energy Rev. 2014, 40, 1030–1047. [Google Scholar] [CrossRef]
- Wu, B.X. Integration of mixing, heat transfer, and biochemical reaction kinetics in anaerobic methane fermentation. Biotechnol. Bioeng. 2012, 109, 2864–2874. [Google Scholar] [CrossRef] [PubMed]
- US Environmental Protection Agency. Process Design Manual for Sludge Treatment and Disposal. Available online: https://cfpub.epa.gov/si/si_public_record_report.cfm?dirEntryId=132184 (accessed on 13 December 2012).
- Jingura, R.M.; Kamusoko, R. Methods for determination of biomethane potential of feedstocks: A review. Biofuel Res. J. 2017, 4, 573–586. [Google Scholar] [CrossRef]
- Egan, S.; Thomas, T.; Kjelleberg, S. Unlocking the diversity and biotechnological potential of marine surface associated microbial communities. Curr. Opin. Microbiol. 2008, 11, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Dubilier, N.; Bergin, C.; Lott, C. Symbiotic diversity in marine animals: the art of harnessing chemosynthesis. Nat. Rev. Microbiol. 2008, 6, 725–740. [Google Scholar] [CrossRef] [PubMed]
- Wahl, M.; Goecke, F.; Labes, A.; Dobretsov, S.; Weinberger, F. The second skin: ecological role of epibiotic biofilms on marine organisms. Front. Microbiol. 2012, 3, 292. [Google Scholar] [CrossRef] [PubMed]
- Banks, C.J.; Zhang, Y.; Jiang, Y.; Heaven, S. Trace element requirements for stable food waste digestion at elevated ammonia concentrations. Bioresour. Technol. 2012, 104, 127–135. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).