Laboratory Study on Changes in the Pore Structures and Gas Desorption Properties of Intact and Tectonic Coals after Supercritical CO2 Treatment: Implications for Coalbed Methane Recovery
Abstract
:1. Introduction
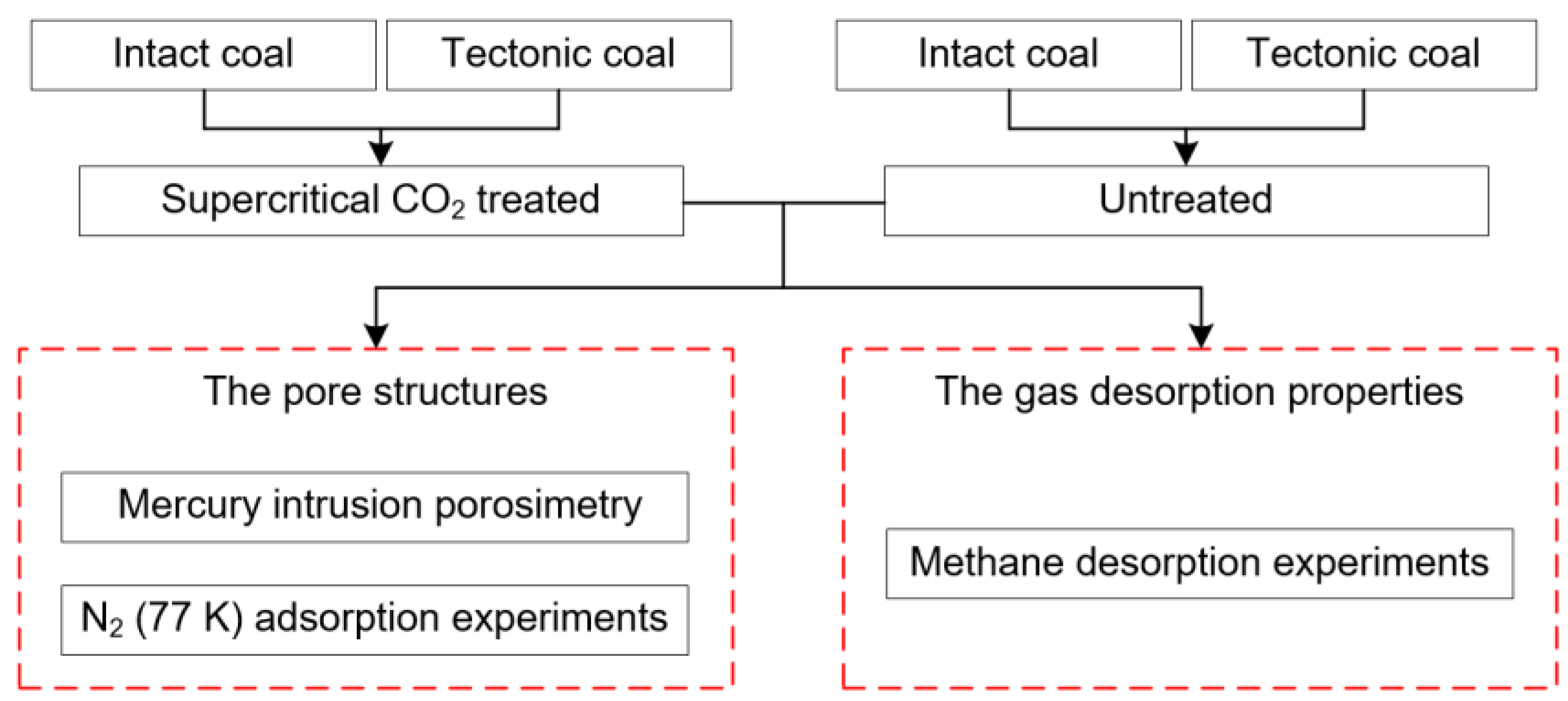
2. Materials and Methods
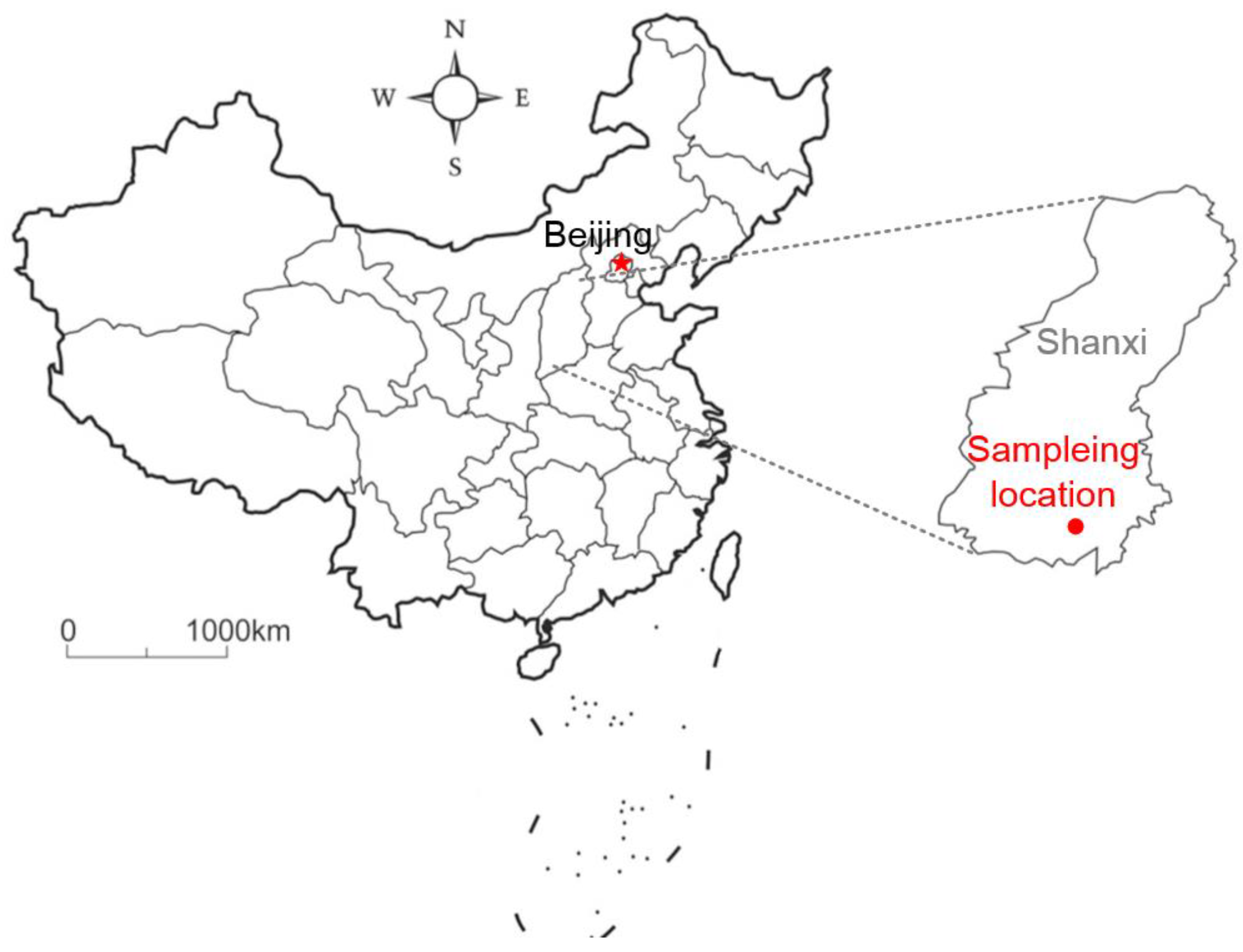
2.1. Coal Samples
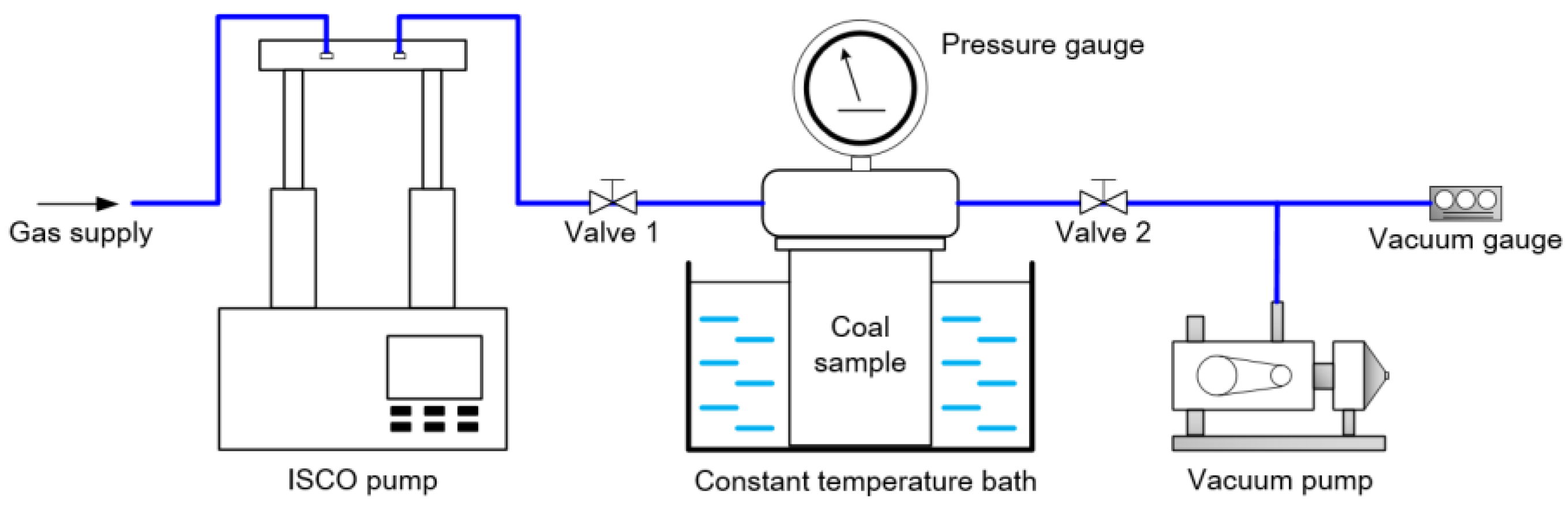
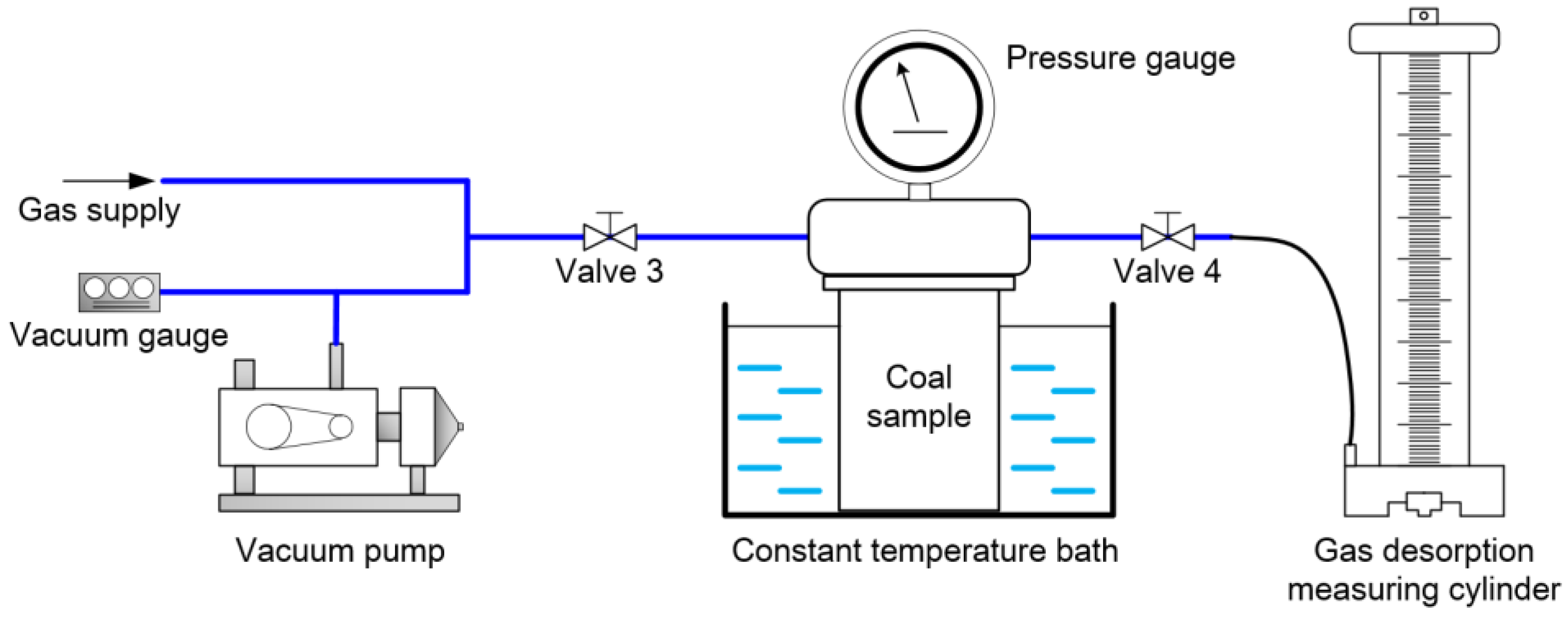
2.2. Experimental Methods
3. Results and Discussion
3.1. Pore Structure Analysis
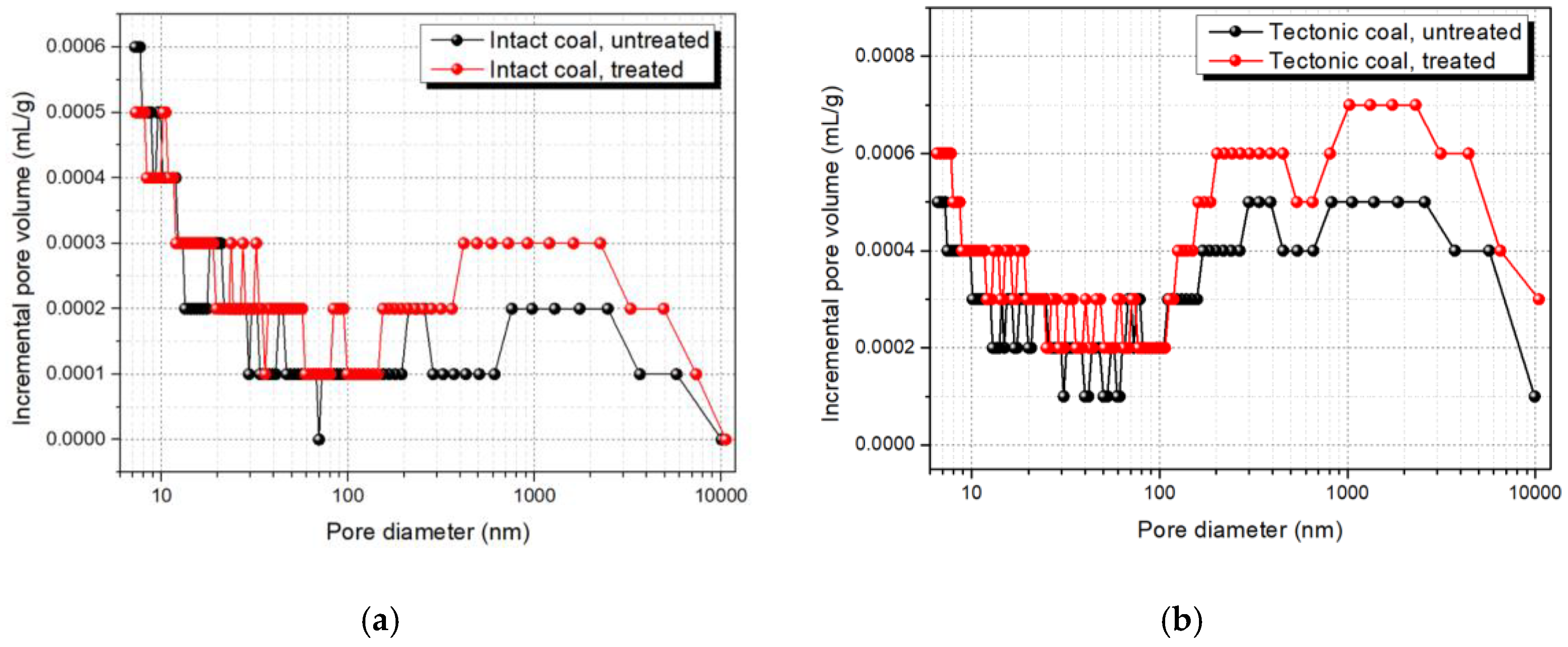
3.1.1. Pore Size Distribution
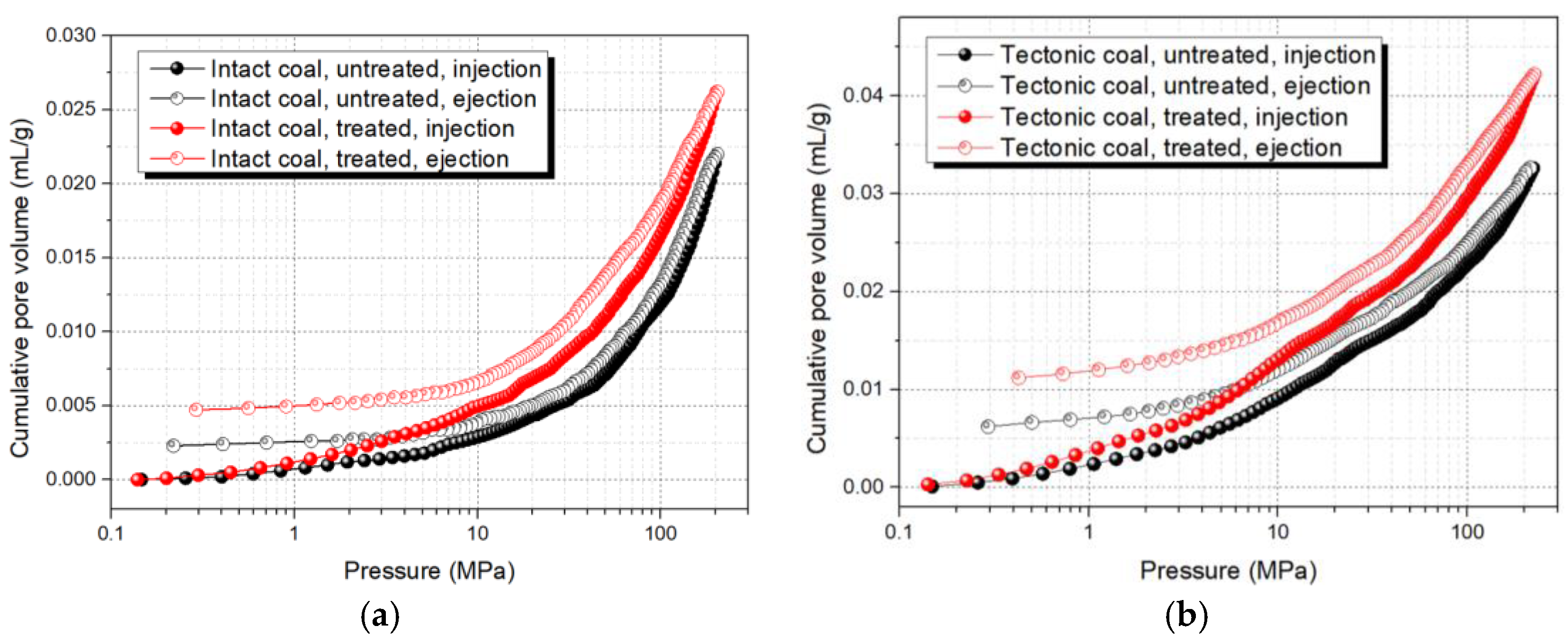
3.1.2. Pore Volume and Surface Area
3.1.3. Pore Connectivity
3.2. Desorption Analysis
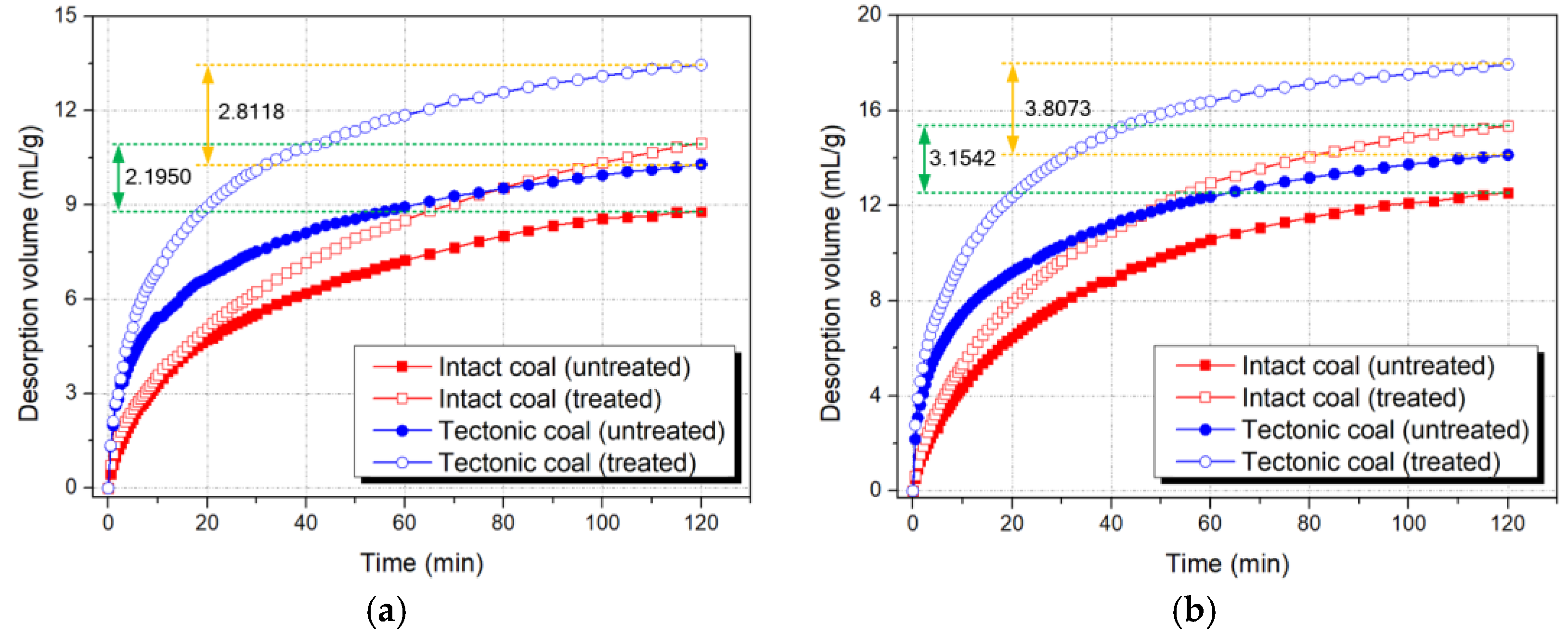
3.2.1. Gas Desorption Curves
3.2.2. Gas Desorption Diffusion Coefficients
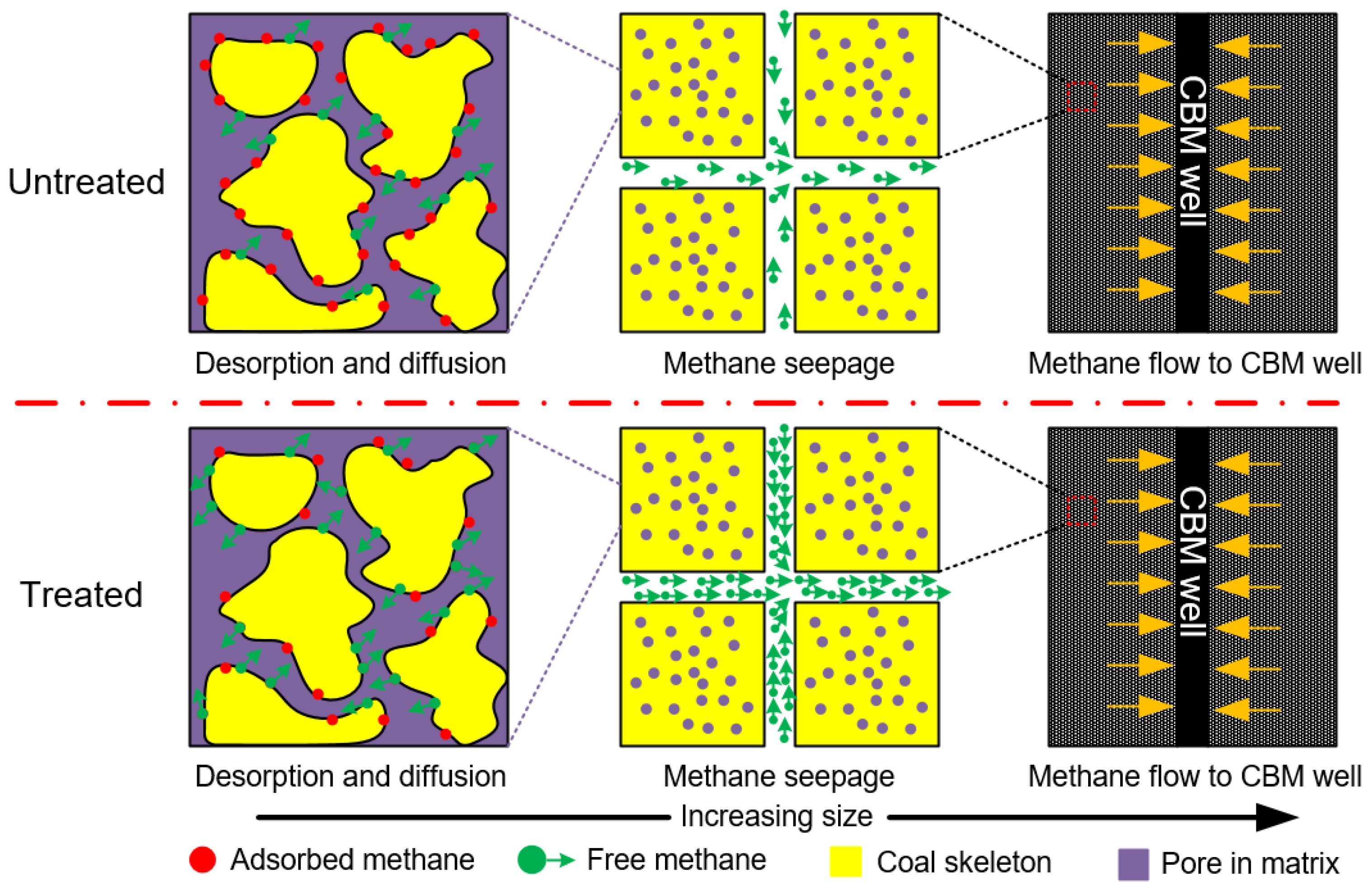
3.2.3. Implication for CO2-ECBM in Tectonic Coal Reservoirs
4. Conclusions
- (1)
- Compared with intact coal, the macropores and mesopores in tectonic coal were obviously larger after ScCO2 treatment. Additional, the TPV, SSA, and pore connectivity of treated tectonic coal were significantly improved. Pore structure analysis showed that tectonic coal was significantly affected by ScCO2 treatment. This was because tectonic coal contained more minerals and the mobilizing effect in tectonic coal was more pronounced.
- (2)
- The results of the methane desorption experiment showed that the desorption capacity of intact coal and tectonic coal was improved to a certain extent by ScCO2 treatment; however, the diffusion coefficient of the treated tectonic coal increased twice as much as that of intact coal. This change was consistent with the pore structure experimental results. The enhancement of the tectonic coal’s diffusion capacity after ScCO2 treatment can partially overcome the limitation imposed on tectonic coal reservoir CBM development by the coal’s inherent low permeability. The results of this study may provide new insights into CO2-ECBM in tectonic coal reservoirs.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zou, Q.; Lin, B.; Zheng, C.; Hao, Z.; Zhai, C.; Liu, T.; Liang, J.; Yan, F.; Yang, W.; Zhu, C. Novel integrated techniques of drilling-slotting-separation-sealing for enhanced coal bed methane recovery in underground coal mines. J. Nat. Gas Sci. Eng. 2015, 26, 960–973. [Google Scholar] [CrossRef]
- Gale, W. A review of energy associated with coal bursts. Int. J. Min. Sci. Technol. 2018, 28, 755–761. [Google Scholar] [CrossRef]
- Yu, G.; Zhai, C.; Qin, L.; Tang, Z.; Wu, S.; Xu, J. Changes to coal pores by ultrasonic wave excitation of different powers. J. China Univ. Min. Technol. 2018, 47, 264–270. [Google Scholar]
- Liu, C.; Li, S.; Yang, S. Gas emission quantity prediction and drainage technology of steeply inclined and extremely thick coal seams. Int. J. Min. Sci. Technol. 2018, 28, 415–422. [Google Scholar]
- Liang, B.; Jia, L.; Sun, W.; Jiang, Y. Experimental on the law of coal deformation and permeability under desorption and seepage. J. China Univ. Min. Technol. 2018, 47, 935–941. [Google Scholar]
- Chang, K.; Tian, H. Technical scheme and application of pressure-relief gas extraction in multi-coal seam mining region. Int. J. Min. Sci. Technol. 2018, 28, 483–489. [Google Scholar]
- Zou, Q.; Lin, B. Fluid−solid coupling characteristics of gas-bearing coal subjected to hydraulic slotting: An experimental investigation. Energy Fuels 2018, 32, 1047–1060. [Google Scholar] [CrossRef]
- White, C.M.; Smith, D.H.; Jones, K.L.; Goodman, A.L.; Jikich, S.A.; LaCount, R.B.; DuBose, S.B.; Ozdemir, E.; Morsi, B.I.; Schroeder, K.T. Sequestration of carbon dioxide in coal with enhanced coalbed methane recovery: A review. Energy Fuels 2005, 19, 659–724. [Google Scholar] [CrossRef]
- Haszeldine, R.S. Carbon capture and storage: How green can black be? Science 2009, 325, 1647–1652. [Google Scholar] [CrossRef]
- Zhang, X.; Ranjith, P.G.; Perera, M.S.A. Gas transportation and enhanced coalbed methane recovery processes in deep coal seams: A review. Energy Fuels 2016, 30, 8832–8849. [Google Scholar] [CrossRef]
- Gale, J. Geological storage of CO2: What do we know, where are the gaps and what more needs to be done? Energy 2004, 29, 1329–1338. [Google Scholar] [CrossRef]
- Perera, M. A Comprehensive Overview of CO2 Flow Behaviour in Deep Coal Seams. Energies 2018, 11, 906. [Google Scholar] [CrossRef]
- Fulton, P.F.; Parente, C.A.; Rogers, B.A.; Shah, N.; Reznik, A. A laboratory investigation of enhanced recovery of methane from coal by carbon dioxide injection. In SPE Unconventional Gas Recovery Symposium; Society of Petroleum Engineers: Pittsburgh, PA, USA, 1980. [Google Scholar]
- Reznik, A.A.; Singh, P.K.; Foley, W.L. An analysis of the effect of CO2 injection on the recovery of in-situ methane from bituminous coal: An experimental simulation. Soc. Petrol. Eng. J. 1984, 24, 521–528. [Google Scholar] [CrossRef]
- Jessen, K.; Tang, G.-Q.; Kovscek, A.R. Laboratory and simulation investigation of enhanced coalbed methane recovery by gas injection. Transp. Porous Media 2008, 73, 141–159. [Google Scholar] [CrossRef]
- Dutka, B.; Kudasik, M.; Pokryszka, Z.; Skoczylas, N.; Topolnicki, J.; Wierzbicki, M. Balance of CO2/CH4 exchange sorption in a coal briquette. Fuel Process. Technol. 2013, 106, 95–101. [Google Scholar] [CrossRef]
- Stevens, S.H.; Kuuskraa, V.A.; Spector, D.; Riemer, P. CO2 sequestration in deep coal seams: Pilot results and worldwide potential. In Proceedings of the Greenhouse Gas Control Technology, Saskatoon, SK, Canada, 4–6 October 1999; pp. 175–180. [Google Scholar]
- Span, R.; Wagner, W. A new equation of state for carbon dioxide covering the fluid region from the triple-point temperature to 1100 K at pressures up to 800 MPa. J. Phys. Chem. Ref. Data 1996, 25, 1509–1596. [Google Scholar] [CrossRef]
- Larsen, J.W. The effects of dissolved CO2 on coal structure and properties. Int. J. Coal Geol. 2004, 57, 63–70. [Google Scholar] [CrossRef]
- Zhang, K.; Cheng, Y.; Jin, K.; Guo, H.; Liu, Q.; Dong, J.; Li, W. Effects of Supercritical CO2 Fluids on Pore Morphology of Coal: Implications for CO2 Geological Sequestration. Energy Fuels 2017, 31, 4731–4741. [Google Scholar] [CrossRef]
- Chen, R.; Qin, Y.; Wei, C.; Wang, L.; Wang, Y.; Zhang, P. Changes in pore structure of coal associated with Sc-CO2 extraction during CO2-ECBM. Appl. Sci. 2017, 7, 931. [Google Scholar] [CrossRef]
- Liu, C.J.; Wang, G.X.; Sang, S.X.; Rudolph, V. Changes in pore structure of anthracite coal associated with CO2 sequestration process. Fuel 2010, 89, 2665–2672. [Google Scholar] [CrossRef]
- Kolak, J.J.; Hackley, P.C.; Ruppert, L.F.; Warwick, P.D.; Burruss, R.C. Using ground and intact coal samples to evaluate hydrocarbon fate during supercritical CO2 injection into coal beds: Effects of particle size and coal moisture. Energy Fuels 2015, 29, 5187–5203. [Google Scholar] [CrossRef]
- Zhang, D.; Gu, L.; Li, S.; Lian, P.; Tao, J. Interactions of supercritical CO2 with coal. Energy Fuels 2013, 27, 387–393. [Google Scholar] [CrossRef]
- Busch, A.; Gensterblum, Y. CBM and CO2-ECBM related sorption processes in coal: A review. Int. J. Coal Geol. 2011, 87, 49–71. [Google Scholar] [CrossRef]
- Cai, Y.; Liu, D.; Pan, Z.; Yao, Y.; Li, J.; Qiu, Y. Pore structure and its impact on CH4 adsorption capacity and flow capability of bituminous and subbituminous coals from Northeast China. Fuel 2013, 103, 258–268. [Google Scholar] [CrossRef]
- Lu, S.Q.; Cheng, Y.P.; Li, W.; Wang, L. Pore structure and its impact on CH4 adsorption capability and diffusion characteristics of normal and deformed coals from Qinshui Basin. Int. J. Oil Gas Coal Technol. 2015, 10, 76–78. [Google Scholar] [CrossRef]
- Ju, Y.W.; Jiang, B.; Hou, Q.L.; Wang, G.L. The new structure-genetic classification system in tectonically deformed coals and its geological significance. J. China Coal Soc. 2004, 29, 513–517. [Google Scholar]
- Shi, J.Q.; Durucan, S. A bidisperse pore diffusion model for methane displacement desorption in coal by CO injection. Fuel 2003, 82, 1219–1229. [Google Scholar] [CrossRef]
- An, F.H.; Cheng, Y.P. An explanation of large-scale coal and gas outbursts in underground coal mines: The effect of low-permeability zones on abnormally abundant gas. Nat. Hazard Earth Syst. 2014, 14, 4751–4775. [Google Scholar] [CrossRef]
- Clarkson, C.R.; Bustin, R.M. The effect of pore structure and gas pressure upon the transport properties of coal: A laboratory and modeling study. 1. Isotherms and pore volume distributions. Fuel 1999, 78, 1333–1344. [Google Scholar] [CrossRef]
- Pan, J.; Zhao, Y.; Hou, Q.; Jin, Y. Nanoscale pores in coal related to coal rank and deformation structures. Transp. Porous Media 2015, 107, 543–554. [Google Scholar] [CrossRef]
- Wong, S.; Law, D.; Deng, X.; Robinson, J.; Kadatz, B.; Gunter, W.D.; Ye, J.; Feng, S.; Fan, Z. Enhanced coalbed methane and CO2 storage in anthracitic coals—Micro-pilot test at South Qinshui, Shanxi, China. Int. J. Greenhouse Gas Control 2007, 1, 215–2228. [Google Scholar] [CrossRef]
- Washburn, E.W. The dynamics of capillary flow. Phys. Rev. Ser. 1921, 17, 273–283. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Landers, J.; Gor, G.Y.; Neimark, A.V. Density functional theory methods for characterization of porous materials. Colloids Surf. A 2013, 437, 3–32. [Google Scholar] [CrossRef]
- Hodot, B.B. Outburst of Coal and Coalbed Gas; China Industry Press: Beijing, China, 1966. [Google Scholar]
- Hou, Q.; Li, H.; Fan, J.; Ju, Y.; Wang, T.; Li, X.; Wu, Y. Structure and coalbed methane occurrence in tectonically deformed coals. Sci. China Earth Sci. 2012, 55, 1755–1763. [Google Scholar] [CrossRef]
- Li, M.; Jiang, B.; Lin, S.; Lan, F.; Wang, J. Structural controls on coalbed methane reservoirs in faer coal mine, southwest china. J. Earth Sci.-China 2013, 24, 437–448. [Google Scholar] [CrossRef]
- Perera, M.S.A. Influences of CO2 injection into deep coal seams: A review. Energy Fuels 2017, 31, 10324–10334. [Google Scholar] [CrossRef]
- Wellman, T.P.; Grigg, R.B.; Mcpherson, B.J.; Svec, R.K.; Lichtner, P.C. Evaluation of CO2–brine–reservoir rock interaction with laboratory flow tests and reactive transport modeling. In International Symposium on Oilfield Chemistry; Society of Petroleum Engineers: Pittsburgh, PA, USA, 2003. [Google Scholar]
- Cao, Y.; Davis, A.; Liu, R.; Liu, X.; Zhang, Y. The influence of tectonic deformation on some geochemical properties of coals—A possible indicator of outburst potential. Int. J. Coal Geol. 2003, 53, 69–79. [Google Scholar] [CrossRef]
- Qu, Z.; Wang, G.G.X.; Jiang, B.; Rudolph, V.; Dou, X.; Li, M. Experimental study on the porous structure and compressibility of tectonized coals. Energy Fuels 2010, 24, 2964–2973. [Google Scholar] [CrossRef]
- Rouquerol, F.; Rouquerol, J.; Sing, K. Adsorption by Powders and Porous Solids; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Wang, H.; Fu, X.; Jian, K.; Li, T.; Luo, P. Changes in coal pore structure and permeability during N2 injection. J. Nat. Gas Sci. Eng. 2015, 27, 1234–1241. [Google Scholar] [CrossRef]
- Guo, H.; Cheng, Y.; Yuan, L.; Wang, L.; Zhou, H. Unsteady-state diffusion of gas in coals and its relationship with coal pore structure. Energy Fuels 2016, 30, 7014–7024. [Google Scholar] [CrossRef]
- Pan, Z.; Connell, L.D.; Camilleri, M.; Connelly, L. Effects of matrix moisture on gas diffusion and flow in coal. Fuel 2010, 89, 3207–3217. [Google Scholar] [CrossRef]
- Liu, Z.; Cheng, Y.; Dong, J.; Jiang, J.; Wang, L.; Li, W. Master role conversion between diffusion and seepage on coalbed methane production: Implications for adjusting suction pressure on extraction borehole. Fuel 2018, 223, 373–384. [Google Scholar] [CrossRef]
- Jiang, H.; Cheng, Y.; Yuan, L. A Langmuir-like desorption model for reflecting the inhomogeneous pore structure of coal and its experimental verification. RSC. Adv. 2014, 5, 2434–2440. [Google Scholar] [CrossRef]
- Crank, J. Mathematics of Diffusion. In Handbook of Chemistry Physic; Oxford University Press: Oxford, UK, 1975. [Google Scholar]
- Yang, Q.; Wang, Y. Theory of methane diffusion from coal cuttings and its application. J. China Coal Soc. 1986, 11, 87–94. [Google Scholar]
- Shouqing, L.; Yuanping, C.; Hongxing, Z.; Haijun, G. Gas desorption characteristics of the high-rank intact coal and fractured coal. Int. J. Min. Sci. Technol. 2015, 25, 819–825. [Google Scholar]
- Liu, C.; Zhou, F.; Yang, K.; Xiao, X.; Liu, Y. Failure analysis of borehole liners in soft coal seam for gas drainage. Eng. Failure Anal. 2014, 42, 274–283. [Google Scholar] [CrossRef]

| Samples | Type | R0, max (%) | Langmuir Volume | Langmuir Pressure | Proximate Analysis (wt.%) | Macerals (vol.%) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mad (%) | Aad (%) | Vdaf (%) | V (%) | I (%) | M (%) | |||||
| Intact coal | Anthracite | 3.13 | 45.83 | 0.81 | 1.35 | 14.26 | 8.46 | 79.70 | 17.22 | 3.08 |
| Tectonic coal | 3.26 | 48.77 | 0.89 | 1.58 | 17.09 | 8.24 | 76.42 | 16.72 | 6.86 | |
| Sample | Porosity (%) | Pore Volume Distribution (%) | |||
|---|---|---|---|---|---|
| V1/Vt | V2/Vt | V3/Vt | V4/Vt | ||
| Intact coal, untreated | 4.18 | 27.73 | 56.36 | 12.27 | 3.64 |
| Intact coal, treated | 4.97 | 20.23 | 58.40 | 16.03 | 5.34 |
| Tectonic coal, untreated | 5.23 | 19.63 | 46.63 | 24.85 | 8.90 |
| Tectonic coal, treated | 6.44 | 19.43 | 44.55 | 24.88 | 11.14 |
| Sample | BJH-TPV (mL/g) | BET-SSA (m2/g) | MIP-TPV (mL/g) | MIP-SSA (m2/g) |
|---|---|---|---|---|
| Intact coal, untreated | 0.015 | 3.421 | 0.0220 | 0.165 |
| Intact coal, treated | 0.016 | 3.862 | 0.0262 | 0.190 |
| Tectonic coal, untreated | 0.019 | 3.964 | 0.0326 | 0.214 |
| Tectonic coal, treated | 0.021 | 4.402 | 0.0422 | 0.256 |
| Samples | Intact Coal | Tectonic Coal | Pressure | ||||
|---|---|---|---|---|---|---|---|
| Untreated | Treated | Change | Untreated | Treated | Change | ||
| D (×10−12 m2/s) | 2.6360 | 3.8480 | 45.98% | 3.9991 | 7.5020 | 87.59% | 2 MPa |
| 4.2155 | 6.0091 | 42.55% | 5.2912 | 9.8848 | 86.82% | 4 MPa | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, E.; Liang, Y.; Li, L.; Zou, Q.; Niu, F. Laboratory Study on Changes in the Pore Structures and Gas Desorption Properties of Intact and Tectonic Coals after Supercritical CO2 Treatment: Implications for Coalbed Methane Recovery. Energies 2018, 11, 3419. https://doi.org/10.3390/en11123419
Su E, Liang Y, Li L, Zou Q, Niu F. Laboratory Study on Changes in the Pore Structures and Gas Desorption Properties of Intact and Tectonic Coals after Supercritical CO2 Treatment: Implications for Coalbed Methane Recovery. Energies. 2018; 11(12):3419. https://doi.org/10.3390/en11123419
Chicago/Turabian StyleSu, Erlei, Yunpei Liang, Lei Li, Quanle Zou, and Fanfan Niu. 2018. "Laboratory Study on Changes in the Pore Structures and Gas Desorption Properties of Intact and Tectonic Coals after Supercritical CO2 Treatment: Implications for Coalbed Methane Recovery" Energies 11, no. 12: 3419. https://doi.org/10.3390/en11123419
APA StyleSu, E., Liang, Y., Li, L., Zou, Q., & Niu, F. (2018). Laboratory Study on Changes in the Pore Structures and Gas Desorption Properties of Intact and Tectonic Coals after Supercritical CO2 Treatment: Implications for Coalbed Methane Recovery. Energies, 11(12), 3419. https://doi.org/10.3390/en11123419





