Abstract
Biochar (BC) is increasingly applied in agriculture; however, due to its adsorption and degradation properties, biochar may also affect the efficacy of fumigant in amended soil. Our research is intended to study the effects of two types of biochars (BC-1 and BC-2) on the efficacy and emission of methyl isothiocyanate (MITC) in biochar amendment soil. Both types of biochars can significantly reduce MITC emission losses, but, at the same time, decrease the concentration of MITC in the soil. The efficacy of MITC for controlling soil-borne pests (Meloidogyne spp., Fusarium spp. Phytophthora spp., Abutilon theophrasti and Digitaria sanguinalis) was reduced when the biochar (BC-1 and BC-2) was applied at a rate of higher than 1% and 0.5% (on a weight basis) (on a weight basis), respectively. However, increased doses of dazomet (DZ) were able to offset decreases in the efficacy of MITC in soils amended with biochars. Biochars with strong adsorption capacity (such as BC-1) substantially reduced MITC degradation rate by 6.2 times, and increased by 4.1 times following amendment with biochar with high degradability (e.g., BC-2), compared to soil without biochar amendment. This is due to the adsorption and degradation of biochar that reduces MITC emission losses and pest control.
1. Introduction
Soil fumigants are commonly used worldwide to control soil-borne fungal pathogens, nematodes and weeds in high-value crops such as cut flowers and vegetables. Following the phase-out of methyl bromide (MBr), dazomet (DZ) provided a widespread approach for improving the effectiveness of soil disinfestations [1,2,3]. In moist soil, DZ rapidly decomposes to its active ingredient, methyl isothiocyanate (MITC), which is toxic to soil-borne pests, especially fungi, some soil arthropods, and ectoparasitic nematodes [4,5]. However, the high application rates of DZ (294–450 kg·ha−1) and high vapor pressure of MITC (20.7 mmHg at 20 °C) result in the significant volatilization of MITC following fumigation, and this may cause environmental and health problems because of the irritant, lachrymatory and toxic properties of the gas [4,6]. Hence, it is imperative to develop strategies to minimize the emissions of MITC while ensuring high activity against pathogens. Applying biochar (BC) to the soil surface has been demonstrated to reduce the losses of the fumigant caused by volatilization. For example, Wang et al. reported that a small amount of biochar added to the soil surface can reduce 1,3-dichloropropene emissions by more than 92% and reduce chloropicrin losses by 85.7%–97.7% [7,8]. Biochar soil amendments, therefore, offer a possible approach for reducing the emission of MITC [9].
Biochar is a carbon-rich, porous, intentionally produced charcoal made in low oxygen conditions from natural organic materials [10]. Because of its specific properties—high carbon content, rich pore structure, and stable physical and chemical properties—biochar has a potential role in carbon sequestration and the suppression of emissions of greenhouse gases from soil, as well as the improvement of crop productivity and soil health. Consequently, biochar soil amendments are attracting increasing attention from policy makers in China and other countries. However, types of biochar that have an exceptionally high sorption [10] or degradation [7,11] capacity for organic chemicals, especially pesticides, may have both positive and negative impacts on pest management. On the one hand, the high retentive qualities of biochar may prevent or reduce the leaching of soil-applied herbicides and insecticides [12,13] and may decrease the rate at which they are degraded by soil microorganisms. On the other hand, types of biochar that have strong sorption or degradation affinities for pesticides may result in low effective concentrations for pest management, meaning that greater amounts of pesticide must be applied to achieve the same level of pest protection. For example, Kookana et al. demonstrated a decreased efficacy of herbicides and lower bioavailability of pesticides in biochar-amended soils [14,15]. Depending on the sorption strength of the particular biochar, amending the soil with biochar may adversely impact pest control [10].
Obviously, biochar has great potential in terms of reducing emissions of the MITC fumigant, but it also has the potential to adversely impact pest control. Therefore, it is necessary to find a balance between reducing emissions and ensuring adequate agricultural pest control when using biochar and MITC. Previously, our experiments have shown that some biochars could drastically accelerate and some biochars could decelerate the degradation of MITC [16], while both types of biochar accelerated degradation and adsorption potential to reduce MITC emission. However, the impact of biochar on the efficacy and fate of MITC remains poorly understood. Furthermore, it is not clear how MITC fumigation would be impacted by types of biochar that have different physical and chemical characteristics as a result of different feedstock and production parameters. Here, we select two type of biochars: one suppresses MITC degradation and the second accelerates its degradation, in order to investigate the effects of biochar on the efficacy, emission, degradation and adsorption of MITC in biochar-amended soil and to determine the appropriate balance between reducing MITC emissions and ensuring its availability for pest control. The information obtained from this study will be useful for evaluating the effect of biochar on the bioavailability and efficacy of MITC and for evaluating the use of biochar to reduce MITC emissions, as well as identifying the necessary conditions for optimal degradation.
2. Results and Discussion
2.1. Effects of Biochar Amendment on Methyl Isothiocyanate (MITC) Emissions
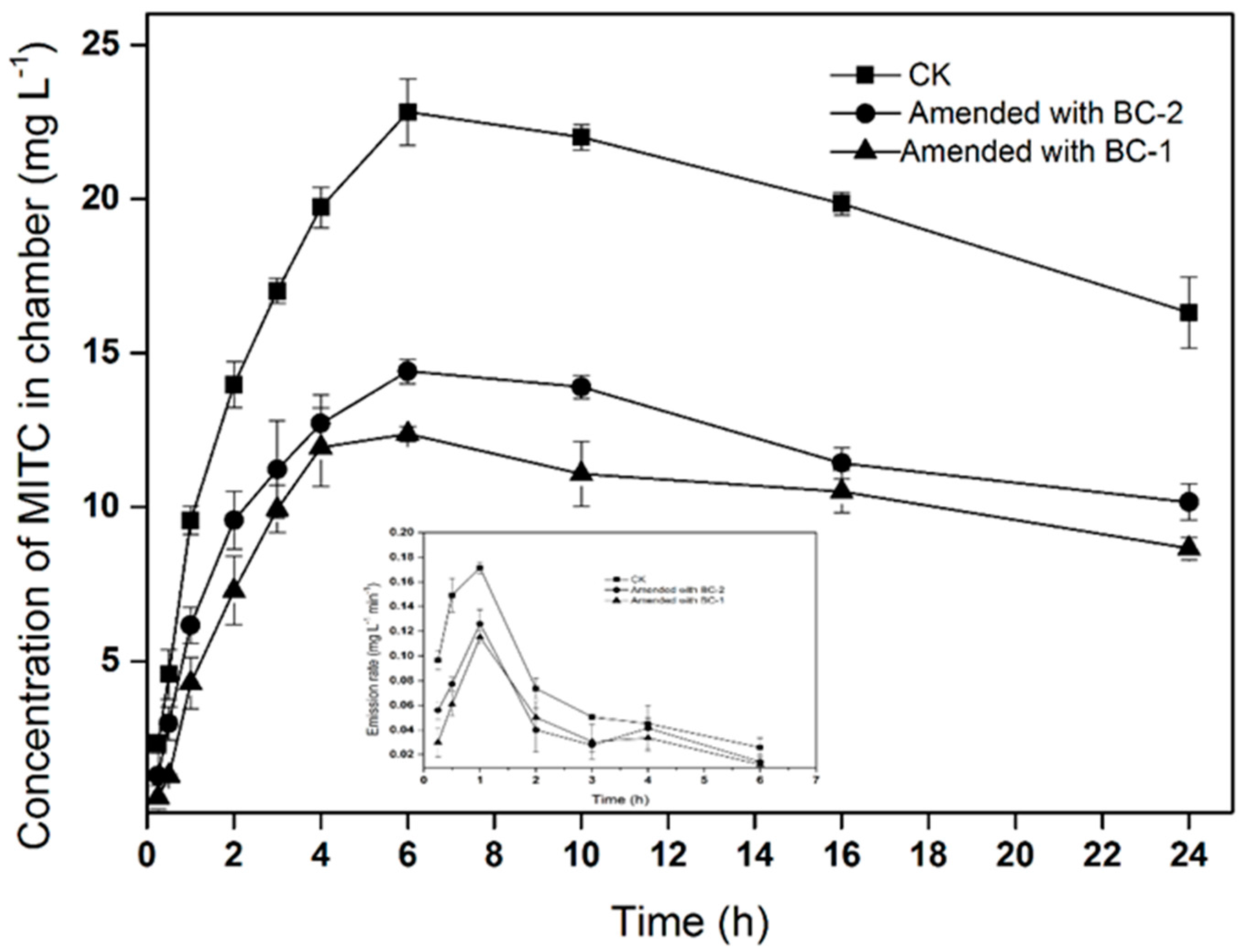
MITC emission losses were markedly reduced in soil amended with biochar (Figure 1). The maximum air concentrations of the MITC in the chambers were 22.8, 12.3, and 14.4 mg·L−1 in the CK (soil without biochar), BC-1, and BC-2 treatments, respectively. Compared with fumigation without biochar, BC-1 and BC-2 reduced the total fumigant emission losses by 46.1% and 36.8%, respectively. Correspondingly, the emission rates of MITC decreased in soil treated with BC-1 and BC-2. In the three treatments, the emissions flux initially increased with time, exhibiting a peak flux at 1 h after injection, before subsequently declining with time. The maximum emissions fluxes of MITC were 0.17, 0.11, and 0.12 mg·L−1·min−1 in the CK, BC-1 and BC-2 treatments, respectively. The results indicated that BC-1 or BC-2 amendments in the soil surface could significantly reduce MITC emissions in the air.
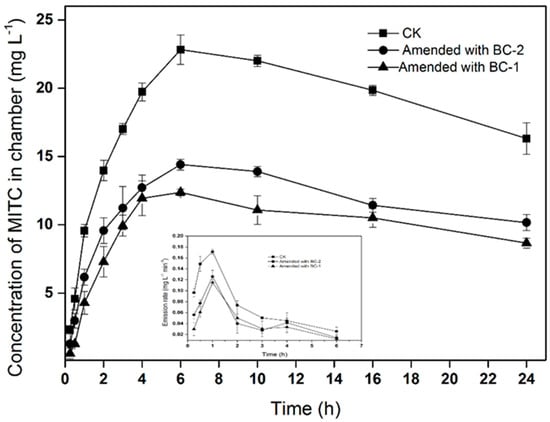
Figure 1.
Concentration of methyl isothiocyanate (MITC) in chamber treatments and emission rates of MITC. Data points indicate means of three replicate cells, and error bars indicate standard errors of the mean.
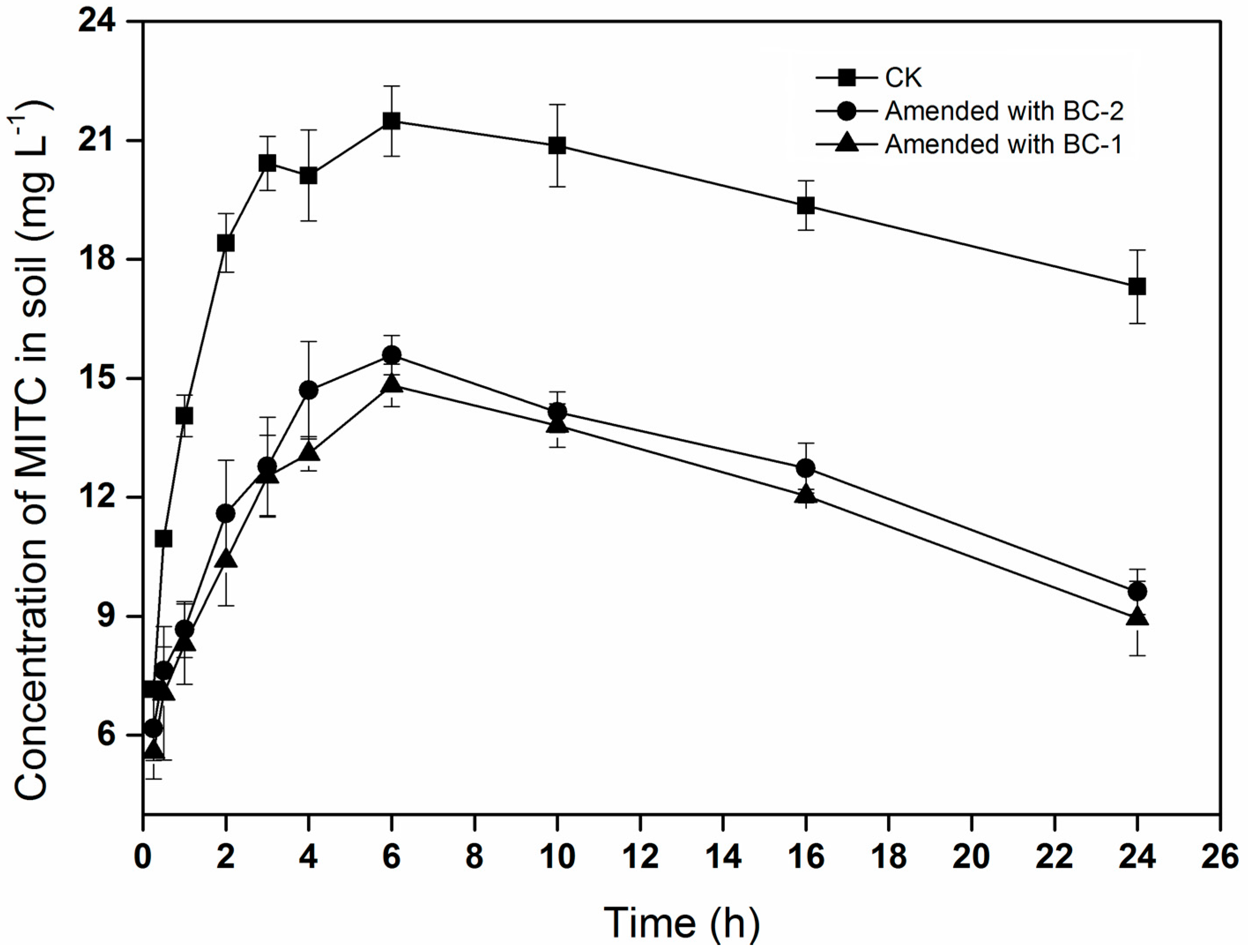
The trend of MITC concentrations in the soil was similar to that in the chamber air, increasing initially and then declining with time (Figure 2). The concentrations of MITC in the soil treated with biochar (BC-1 or BC-2) were significantly lower than in CK. The maximum concentrations of MITC were 21.5, 14.8, and 15.6 mg·L−1 in the CK, BC-1, and BC-2 treatments, respectively. These results showed that biochar amendments could reduce the concentrations of MITC in the soil, potentially reducing its efficacy for controlling soil-borne pests.
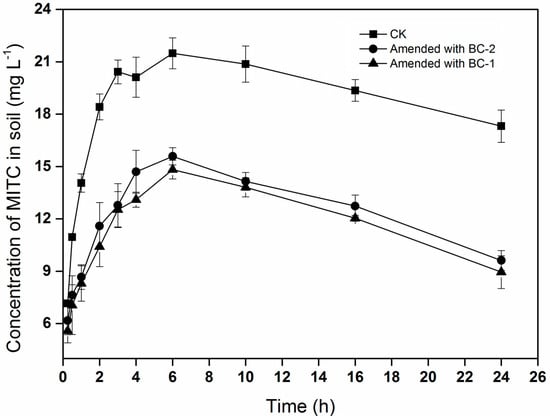
Figure 2.
Concentration of MITC in soil. Data points indicate means of three replicate cells, and error bars indicate standard error of the mean.
2.2. Effects of Biochar Amendment on the Efficacy of MITC against Soil-Borne Pests
The efficacies of MITC with respect to root-knot nematodes (Meloidogyne spp.), weed seeds (Abutilon theophrasti and Digitaria sanguinalis) and key soil-borne fungi (Fusarium spp. and Phytophthora spp.) in soil amended with BC-1 or BC-2 are listed in Table 1 and Table 2, respectively. As shown in Table 1, the corrected mortality of nematodes following fumigation of unamended soil did not differ from that in soil amended with BC-1 at rates of 0.1% to 5%; however, the corrected mortality of nematodes was significantly lower in soil amended with 10% BC-1. When the BC-1 amendment rates were less than 2% and 1%, there was no significant difference in the efficacies of MITC on control of Digitaria sanguinalis and Abutilon theophrasti, respectively. Compared with samples without biochar, the efficacy of MITC against Phytophthora spp. and Fusarium spp. was reduced significantly when the BC-1 amendment rates were greater than 2% and 1%, respectively. The above results indicated that BC-1 amendment rates less than or equal to 1% did not have negative effects on MITC’s control of soil-borne pests.

Table 1.
Pest control efficacy of methyl isothiocyanate (MITC) in soil amended with biochar BC-1.

Table 2.
Pest control efficacy of MITC in soil amended with biochar BC-2.
The effects of BC-2 on MITC’s efficacy against soil-borne pests were similar to BC-1 (Table 2). The efficacy of MITC against root-knot nematode (Meloidogyne spp.) and weed seeds (Abutilon theophrasti and Digitaria sanguinalis) was inhibited significantly after BC-2 amendment at rates above 1% in soil. However, the efficacy against Fusarium spp. and Phytophthora spp. was not reduced by BC-2 amendment rates at or below 1% compared with unamended soil. An amendment rate of less than or equal to 0.5% in BC-2 did not have negative effects on MITC’s control of soil-borne pests.
To obtain a sufficient control effect, a larger amount of DZ is required in soil amended with biochar. The efficacy of MITC against soil-borne pests was investigated in soil amended with 1% BC-1 or 0.5% BC-2, respectively (Table 3 and Table 4). The results indicated that the tested biochars (BC-1 or BC-2) have a negative impact on the biological activity of MITC on most soil-borne pests at a lower application rate of DZ (for example, 25 or 50 mg·kg−1). For BC-1, to achieve the comprehensive prevention of all pests in amended soil, an amount of DZ greater than or equal to 125 mg·kg−1 was required. For BC-2, a DZ application rate greater than or equal to 100 mg·kg−1 did not have negative effects on MITC’s control of soil-borne pests. Consequently, increased doses of DZ were able to offset decreases in the efficacy of MITC in soils amended with biochar. The pest control efficacy of MITC in Daxing soil is higher than that in Tongzhou soil (for example, Table 3 and Table 4). This may be because Tongzhou soil has a history of previous applications of Metham sodium (MS) or DZ (history), while Daxing soil does not (nonhistory). Many studies show that accelerated MITC degradation in history soils resulted in a significant reduction in Verticillium dahliae, Sclerotium rolfsii and Fusarium spp. mortality compared to exposure in nonhistory soils [17,18].

Table 3.
Results of MITC against nematodes and fungal pathogens and weeds in soil amended with biochar BC-1.

Table 4.
Results of MITC against nematodes and fungal pathogens and weeds in soil amended with biochar BC-2.
2.3. Effects of Biochar Amendment on MITC Degradation and Adsorption
The degradation parameters of MITC in soil, biochar alone, and soil amended with biochar (BC-1 or BC-2) are listed in Table 5. The results show that MITC degradation is slower in BC-1 alone and soil amended with BC-1 (at 1%) than in soil alone. Compared with the control, the degradation rate of MITC was 6.2 times slower in soil amended with 1% BC-1, and the degradation rate was as much as 14 times slower in pure BC-1. It is clear that BC-1 can significantly inhibit the degradation of MITC. In contrast, BC-2 is able to drastically accelerate the degradation of MITC. The MITC degradation rates (k) in soil amended with 1% BC-2 or BC-2 alone increased 4.1 times and 11.3 times over those in soil alone, respectively. We also observed that the MITC degradation was significantly accelerated after amending the soil with biochar that possessed a high degradation capacity. The above results indicated that biochar has the ability to degrade MITC and that the degradation capacity differs with the type of biochar.

Table 5.
MITC degradation in pure biochar, biochar-amended soil, and unamended soil.
Previous studies have shown that the major degradation pathway for MITC is reaction with hydroxyl (˙OH) radicals [19]. In addition, there are many organic radicals existing in biochar [20,21], and these free radicals could induce ˙OH generation. Fang et al. [22] reported that the proposed mechanism of ˙OH generation was for free radicals in biochar to transfer electrons to O2 to produce the superoxide radical anion and hydrogen peroxide, which reacts further with free radicals to produce ˙OH. This indicates that the ˙OH generated on biochar may contribute to MITC degradation. Moreover, Chen et al. suggested that the H/C (hydrogen atom/carbon atom ratio) atomic ratio of organic components in biochar can be used to characterize their aromaticity and polarity [23]: the higher the H/C value, the lower the aromaticity. Therefore, higher H/C values mean that more functional groups are able to generate ˙OH and more MITC could be degraded. For example, BC-2 biochar had a higher H/C value (0.25), and the MITC degradation rate in BC-2 was greater.
Soil amended with BC-1 inhibited MITC degradation, possibly due to BC-1’s high adsorption capacity for MITC. To clarify the mechanism by which biochar inhibits MITC degradation, we investigated the biochar adsorption kinetics for MITC at different temperatures. The parameters of MITC adsorption by biochar are listed in Table 6. The r2 values of all treatments were at least 0.82, and the calculated amounts of MITC adsorption at the equilibrium time (qe) approached the values measured in the experiment, indicating that the observed adsorption of MITC to biochar provides a good fit to Bangham models [24]. At 30 °C, the amounts of MITC adsorbed onto BC-1 and BC-2 at the equilibrium time (qe) were 57.87 mg·g−1 and 11.97 mg·g−1, respectively, indicating that BC-1 has a higher adsorption capacity (for MITC) than BC-2 but much lower than AC (activated charcoal, 211.91 mg·g−1), possibly because MITC may be bound to BC-1 and is not available to degrade. In addition, BC-1 has a low H/C value, and chemical degradation was weak, so the MITC degradation rate in BC-1 or soil amended with BC-1 was lower than in unamended soil. As the temperature increased from 15 °C to 45 °C, the adsorption rates of biochar increased; however, the amount of adsorption at the equilibrium time (qe) fell as the temperature increased. Kołohynska et al. note that the sorption capacity of biochar depends mainly on the polarity, aromaticity, surface area, and pore size distribution, etc. [25]. The tested biochars have different surfaces and different pore structures, providing different adsorption rates and adsorption capacities. In general, the adsorption capacity of biochar for MITC increased with the SSA (specific surface area) of the biochar [25]. For example, BC-1 had a larger SSA (382.81 m2·g−1) with a higher amount of adsorption (57.87 mg·g−1), while BC-2 (SSA 36.14 m2·g−1) had a smaller SSA and lower amount of adsorption (11.97 mg·g−1).

Table 6.
The kinetic parameters of biochar (BC-1 and BC-2) adsorption of MITC at 15, 30, and 45 °C.
As noted above, the degradation rates of MITC were much lower in biochar (BC-1) and biochar-amended soil than in unamended soil. This occurred because MITC was adsorbed onto the biochar and thus degraded much more slowly, decreasing as the adsorption capacity increased. In contrast, BC-2 has a weaker absorbability of MITC but possesses a higher degradability; thus, the degradation rates of MITC were much faster in the biochar (BC-2) and biochar-amended soil than in unamended soil. Therefore, biochar’s good absorbability or degradability of MITC were speculated to play an important role in reducing or accelerating MITC’s degradation rate in soil amended with these types of biochar. Studies have shown that the degradation of MITC comprises both biological and chemical degradation and that biodegradation accounted for 51%–97% of the total degradation [26,27]. The slow degradation rate of MITC in biochar-amended soil is likely due to reduced microbial degradation. In addition, the surface of biochar contains a large number of chemical functional groups [23], and a vast amount of free radicals may potentially accelerate the degradation of MITC via radical reaction. In summary, the dissipation of MITC in soil amended with biochar depends on the balance between the amount of adsorption and degradation and is positively correlated with the SSA and H/C values, respectively.
However, the effects of biochar on fumigant emissions are known to be complex. Studies have shown that the emission of 1,3-D was reduced after biochar was applied to the surface, due to the enhanced adsorption of 1,3-D onto the biochar [8,28], while the emission of chloropicrin (CP) was reduced by biochar due to the accelerated degradation of CP in soil amended with biochar [7]. We found that biochar used in this experiment can significantly reduce the volatilization of MITC by degradation or adsorption. However, at the same time, biochar amendments also decrease the concentration of MITC in the soil, which potentially reduces its efficacy for controlling soil-borne pests. Through adsorption or degradation, biochar can minimize the concentration of MITC in the soil at different levels. In addition, the amount of reduction varies with different types of biochar. BC-1 has a high SSA (382.2 m2·g−1) and a small H/C value (0.01), with a k value of 0.08 d−1 and qe value of 57.87 mg·g−1 at 30 °C, meaning that it has a greater absorbability of MITC, resulting in a reduced concentration of MITC in the air and soil. On the contrary, BC-2, with a larger H/C value (0.25) and a smaller SSA (36.1 m2·g−1), (the k value was 15.19 d−1, qe value was 11.97 mg·g−1 at 30 °C), has a greater degradative effect on MITC and leads to a reduction of MITC both in air and soil. However, the reduction of MITC in the air and soil with the amendment of BC-1 was greater than that in BC-2 (Figure 1 and Figure 2), which may be due to the greater adsorption of MITC by BC-1 rather than the degradation of MITC by BC-2. It is precisely because the concentration of MITC is reduced in soil amended by biochar that its efficacy in controlling soil-borne pests is reduced. For example, 5% BC-1 or 2% BC-2 amendments significantly reduce the efficiency of Phytophthora spp. and Fusarium spp. Increased doses of DZ were able to offset decreases in the efficacy of MITC in soils amended with biochar. When the DZ rate was only 25 mg·kg−1, the efficacy of MITC against Abutilon theophrasti was reduced in soil amended with 1% BC-1, compared with unamended soil. However, the above reduction in efficacy was alleviated by increasing the application dose of DZ to 100 mg·kg−1 (corresponding to a field rate of 360 kg·ha−1).
Wang et al. have indicated that biochar amendment at less than 1% in soil did not have negative effects on the levels of pathogens and nematode control achieved by chloropicrin fumigation [7], and the efficacy of Dimethyl Disulfide (DMDS) for controlling root-knot nematodes and Fusarium spp. was not reduced when biochar was applied at a rate less than 2% and 0.5%, respectively [29]. Another study noted that, while nematode control was adequate in the specific system studied, biochar amendment could adversely impact pest control depending on the sorption strength of the particular type of biochar [10]. The actual impact on the efficacy will be a function of the interplay between the application rates of the pesticide and the biochar and the sorption capacity of the specific biochar for the specific pesticide. This issue should be considered when determining the desirable physical and chemical characteristics of biochar for agronomic systems. In the specific experimental system studied here, adequate pest control was achieved at a standard biochar application rate (13 Mg·ha−1). However, different source materials and different production processes create types of biochar with different physical and chemical properties, as well as different adsorption properties or degradability. For example, biochars produced at higher temperatures have been shown to have substantially higher sorption capacities than those produced at lower temperatures. It is apparent that the potential detrimental impact of biochar amendments on pest control must be taken into account when considering the use of biochar in agriculture. In summary, the impact of biochar soil amendments on the efficacy of MITC against soil-borne pests depends on the biochar type, amendment rate, and the application dose of DZ. Because biochar can play a significant role in reducing fumigant emissions [7,8], it is important to select an appropriate biochar amendment that does not affect the efficacy of fumigants such as MITC.
3. Materials and Methods
3.1. Soil, Biochars and Chemicals
Soil samples were collected from the top 20 cm of cucumber greenhouses in Tongzhou and Daxing, Beijing, where the occurrences of nematodes and soil-borne pathogens were severe. The samples were taken at the end of the cropping season. The soil from Tongzhou was composed of 73.24% sand, 5.83% silt, and 20.93% clay, with an organic matter content of 9.12 g·kg−1 soil and a pH of 7.1; the soil from Daxing was composed of 81.44% sand, 10.34% silt, 8.22% clay, with an organic matter content of 14.50 g·kg−1 soil and a pH of 8.2. The soil was sieved through a 2 mm mesh and then mixed thoroughly before use. The soil moisture was adjusted to 15% (w/w).
The two types of biochar (BC-1 and BC-2) were made at 500 °C from Crofton weed and wood pellets, respectively. BC-1 was composed of 86.48% C, 11.70% O, 1.10% H, and 0.72% N, with an SSA of 382.21 m−2·g−1 and a pH of 10.5. BC-2 was composed of 82.94% C, 4.13% O, 1.70% H, and 0.42% N, with an SSA of 36.14 m−2·g−1 and a pH of 10.2. Particularly, BC-1 has a very low H/C (hydrogen atom/carbon atom ratio) value (0.01), while BC-2 with a high H/C value (0.25). The specific surface area (SSA) was determined by a V-Sorb 2800P surface area (Gold APP Instruments Corporation, China), and elemental composition was measured using a CHN element analyzer (vario PYRO cube, Elementar Analysensysteme Gmbh, Germany) [30]. The biochar was sieved through a 2 mm screen before use. An analytical standard of MITC (98.0% purity,) was provided by Damas-beta (ShenZhen, China). DZ 98 MG was obtained from Nantong Shizhuang Chemical Co., Ltd., JiangShu, China. Sodium sulfate anhydrous and ethyl acetate (both analytical grade) were obtained from Beijing Chemical Works, Beijing, China.
3.2. Emission Determination
The columns were constructed of two open stainless steel columns (each 6.0 cm high with 15 cm internal diameter) [31]; one was used as a soil column, while the other was used as a chamber to collect the emitted gas. MITC emissions were measured after MITC was injected into soil columns at 6.0 cm depth and the application of BC-1 or BC-2 to the surface soil (1.5 cm depth). Soil (water content 14% w/w) was packed into each soil column to a bulk density of 1.3 g·cm−3. Pre-weighed (5.4 g, corresponded to biochar rates of 1%) BC-1 or BC-2 were uniformly mixed into the top 1.5 cm layer of the soil column before packing. Soil without biochar was used as a blank control (CK). After MITC (100 mg·kg−1 soil) was injected, the soil column and the chamber were sealed together using aluminum foil tape. Each treatment was repeated three times. Then, the soil columns were placed in incubators at 28 °C.
After 0.25, 0.5, 1, 2, 3, 4, 8, 12, 16 and 24 h, 0.5 mL of gas was taken from the chamber and from the soil (at 6.0 cm depth) by a gastight syringe to determine the MITC concentrations. Previous gas-sealed tests have indicated that gas contentions in chambers remain unchanged more than 5 days, longer than our testing time of 24 h. Each gas sample was put into a 20 mL clear headspace vial containing 0.2 g of sodium sulfate that was immediately crimp-sealed with an aluminum cap and Teflon-faced butyl-rubber septum. The headspaces of the vials containing soil gas samples were analyzed using an Agilent 7890A gas chromatograph (GC) coupled with an Agilent 7694E headspace sampler and a micro electron capture detector (Agilent Technologies Inc., Palo Alto, CA, USA). The gas chromatography conditions were the same as those in the degradation experiments (Section 3.4), and autosampler headspace conditions were as follows: 1.0 mL sample loop; 125, 130, and 135 °C for sample equilibration, loop, and transfer line temperatures, respectively.
3.3. Dose-Response Experiment
The soil samples were amended with BC-1 and BC-2 (Tongzhou soil amended with BC-1, Daxing soil amended with BC-2, respectively) at rates of 0.1%, 0.25%, 0.5%, 1%, 2%, 5%, and 10% (w/w), in order to determine the effects of various amendment rates of biochar on the efficacy of DZ fumigation. Six hundred grams of soil amended with or without biochar was weighed into 2.5-L desiccators. DZ was uniformly mixed into the soil of each desiccator at the rate of 100 mg·kg−1 soil (corresponding to a field rate of 360 kg·ha−1). Ten Abutilon theophrasti seeds and fifteen Digitaria sanguinalis seeds were buried at 2 cm below the soil surface in each desiccator [32] after the application of DZ, and the desiccator was immediately sealed with a cover. The desiccators were placed in incubators at 28 °C. The desiccators were opened after 7 days of incubation, and the residual fumigant was released for a day. The weed height was measured with a caliper. Fusarium spp. and Phytophthora spp. were isolated from the soil and assessed quantitatively using the methods described by Komada [33] and Masago et al. [34], respectively. Root-knot nematodes (Meloidogyne spp.) were extracted from 100 g subsamples using the methods described by Liu [35].
To determine the effects of MTTC in controlling soil-borne pests in soil amended with biochar, DZ was applied at rates of 25, 50, 75, 100, 125, 150, and 175 mg·kg−1 soil (corresponding to rates of 90, 180, 270, 360, 450, 540, 630 kg·ha−1 in the field; its recommended dosage is 294–450 kg·ha−1) amended with or without 1% BC-1 or 0.5% BC-2 (Tongzhou soil amended with BC-1, Daxing soil amended with BC-2). Other operations were similar to the experiment described above.
The nematode mortality was calculated according to Equation (1):
where X is nematode mortality (%), N1 is the number of dead nematodes, and N2 is the number of live nematodes. The numbers of dead and live nematodes were counted under a dissecting microscope.
The corrected nematode mortality was calculated according to Equation (2):
where Y is the corrected nematode mortality (%), X1 is the nematode mortality in treatments (%), and X2 is the nematode mortality in the control (%).
The efficacy of controlling fungi or weeds was calculated according to Equation (3):
where Y is the efficacy in controlling fungi or weeds, X1 is the fungal population or weed height in the control, and X2 is the fungal population or weed height in treated plots.
3.4. Degradation Experiment
A laboratory incubation experiment was conducted to determine the effect of biochar amendments on the degradation of MITC, using a method similar to that reported in Qin et al. [36]. Soil samples (Tongzhou soil) at 10% water content (w/w) were amended with biochar BC-1 or BC-2 at the rate of 1% (w/w). Eight grams of amended or unamended soil was placed in a 20-mL clear headspace vial, and 5 μL of ethyl acetate containing 48 mg·mL−1 of MITC was added to each vial. The vials were sealed with an aluminum cover and Teflon-faced septa immediately after fumigant application. The treated vials were then inverted and placed in incubators at 30 °C. After incubation for 0.5, 1, 3, 4, 7, 10, 15, 30, 40, 60 and 90 days, three replicate samples from each treatment were removed from the incubator and immediately stored at −80 °C until the analysis of the MITC concentration. In the same experiment, we also determined the degradation rate of MITC in pure biochar using the following method. A total of 0.2 g biochar was weighed into a 20 mL clear headspace vial, and the vials were crimp-sealed with an aluminum cap and Teflon-faced butyl-rubber septum immediately after adding 5 μL standard solution of MITC-ethyl acetate. Other operations were similar to those described in the experiment above.
The extraction procedure for soil samples was similar to the methodology described by Hadiri [37]. Eight grams of anhydrous sodium sulfate and 8 mL ethyl acetate were added to each frozen vial, and the vials were recapped immediately. The vials were vortexed for 30 s, placed in a table concentrator and shaken for 60 min, and then subsequently allowed to settle for 1 h. After settling, the supernatants were filtered (using a 0.22-µm nylon syringe filter) into 2 mL vials for the fumigant analysis by Agilent 7890 A gas chromatography. A gas chromatograph with a micro-ECD (GC-μECD) and an HP-5 capillary column (30 m length × 320 μm × 0.25 μm film thickness) (Agilent Technologies, Inc.) was used to analyze the MITC. The detector and inlet temperatures were 300 °C and 250 °C, respectively. The oven temperature was held at 60 °C for 10 min. Under these conditions, the retention time of MITC was 5.4 min. Preliminary experiments indicated that the MITC recovery efficiency in the soil ranged from 89% to 103% using the above procedures. The LOD (lowest detectable limit) and LOQ (limit of quantitation) for MITC in the soil samples were 0.004 and 0.0170 mg·kg−1, respectively. First-order kinetics were used to fit the MITC degradation behavior, using the following equation:
where C (mg·kg−1) and C0 (mg·kg−1) are the concentrations in soil at time t (day) and time t0 (day), respectively; k (day−1) is the first-order rate constant; and t is the incubation time (day):
When using the first-order kinetics model, the half-life of degradation was calculated using Equation (5). The first-order kinetics model was fitted using Origin Pro 8.0 software (version 8.0, OriginLab Corporation, Wellesley Hills, MA, USA).
3.5. Adsorption Experiment
For the adsorption experiment, 0.1 g biochar was weighed into a number of 20 mL clear headspace vials. The two types of biochar (BC-1 and BC-2,) and one activated charcoal (AC) were tested at three temperature settings (15, 30 and 45 °C). All of the vials for each sampling time were placed in a container. The container was constructed of two open stainless steel cylindrical boxes of 12 cm ID, each 4 cm high, similar to the permeability cell described by Papiernik et al. [31]. Before assembling the container, approximately 0.2 g of pure solid MITC was added to a 2 mL vial, which was placed in the bottom of the container. This amount of fumigant was sufficient to generate a saturated vapor phase inside the container. The container was sealed with aluminum tape and carefully placed in incubators with the required temperature setting. After incubation for 3, 6, 12, 24, 48, 72 and 96 h, a container was removed from the incubator. The aluminum tape was removed and the container was opened and ventilated for half an hour in a fume hood. The biochar was then extracted and analyzed for adsorbed MITC using the same method described in the degradation experiments.
The Bangham model [24] was tested against the adsorption kinetic data, and the rate constant k of adsorption was calculated using the Bangham Equation (6), as follows:
where qt (mg·g−1) is the amount of MITC adsorption at time t (h); qe (mg·g−1) is the calculated amount of adsorption at equilibrium; t (h) is the time in hours; k (h−z) is the rate constant of adsorption; and z is a constant in relation to the adsorbent.
4. Conclusions
Both of the biochars used in this study (BC-1 and BC-2) can significantly affect the degradation rate of MITC (reduce or accelerate). The dose-response experiments indicated that there were no negative effects on the control of pathogens, nematodes and weeds when the biochar amendment was less than 1% or 0.5% for BC-1 and BC-2, respectively, in soil at conventional dosages of the fumigant dazomet. The information obtained in this study will be useful for evaluating the effects of biochar on the bioavailability and efficacy of MITC.
Acknowledgments
This study was supported by the National Natural Science Foundation Project of China (31572035). The authors would like to express their thanks to Melanie Miller for help with the English language.
Author Contributions
Wensheng Fang and Qiuxia Wang designed the study and wrote the protocol; Dawei Han and Dongdong Yan carried out determination of the biochar characteristics; Wensheng Fang, Bin Huang, Dawei Han and Xiaoman Liu performed most of the experiments; Wensheng Fang, Jun Li and Meixia Guo managed the literature search and analyses; Wensheng Fang, Qiuxia Wang and Dawei Han analyzed the data; Wensheng Fang, Qiuxia Wang and Aocheng Cao were responsible for the overall design and wrote the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ruzo, L.O. Physical, chemical and environmental properties of selected chemical alternatives for the pre-plant use of methyl bromide as soil fumigant. Pest Manag. Sci. 2006, 62, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.N. Development of alternative strategies for management of soilborne pathogens currently controlled with methyl bromide. Annu. Rev. Phytopathol. 2003, 41, 325–350. [Google Scholar] [CrossRef] [PubMed]
- Ajwa, H.A.; Klose, S.; Nelson, S.D.; Minuto, A.; Gullino, M.L.; Lamberti, F.; Lopez-Aranda, J.M. Alternatives to methyl bromide in strawberry production in the United States of America and the Mediterranean region. Phytopathol. Mediterr. 2003, 42, 220–244. [Google Scholar]
- Saeed, I.A.M.; Rouse, D.I.; Harkin, J.M.; Smith, K.P. Effects of soil water content and soil temperature on efficacy of metham-sodium against verticillium dahliae. Plant Dis. 1997, 81, 773–776. [Google Scholar] [CrossRef]
- Frick, A.; Zebarth, B.; Szeto, S. Behavior of the soil fumigant methyl isothiocyanate in repacked soil columns. J. Environ. Qual. 1998, 27, 1158–1169. [Google Scholar] [CrossRef]
- Kiely, T.; Donaldson, D.; Grube, A. Pesticides Industry Sales and Usage; Office of Prevention, Pesticides and Toxic Substances, United States Environment Protection Agency: Washington, DC, USA, 2004; p. 16.
- Wang, Q.; Yan, D.; Liu, P.; Mao, L.; Wang, D.; Fang, W.; Li, Y.; Ouyang, C.; Guo, M.; Cao, A. Chloropicrin emission reduction by soil amendment with biochar. PLoS ONE 2015, 10, e0129448. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Mao, L.; Wang, D.; Yan, D.; Ma, T.; Liu, P.; Zhang, C.; Wang, R.; Guo, M.; Cao, A. Emission reduction of 1,3-dichloropropene by soil amendment with biochar. J. Environ. Qual. 2014, 43, 1656–1662. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Ashworth, D.J.; Gan, J.; Yates, S.R. Biochar amendment to the soil surface reduces fumigant emissions and enhances soil microorganism recovery. Environ. Sci. Technol. 2016, 50, 1182–1189. [Google Scholar] [CrossRef] [PubMed]
- Graber, E.R.; Tsechansky, L.; Khanukov, J.; Oka, Y. Sorption, volatilization, and efficacy of the fumigant 1,3-dichloropropene in a biochar-amended soil. Soil Sci. Soc. Am. J. 2011, 75, 1365. [Google Scholar] [CrossRef]
- Jablonowski, N.D.; Krutz, J.L.; Martinazzo, R.; Zajkoska, P.; Hamacher, G.; Borchard, N.; Burauel, P. Transfer of atrazine degradation capability to mineralize aged 14C-labeled atrazine residues in soils. J. Agric. Food Chem. 2013, 61, 6161–6166. [Google Scholar] [CrossRef] [PubMed]
- Spokas, K.A.; Koskinen, W.C.; Baker, J.M.; Reicosky, D.C. Impacts of woodchip biochar additions on greenhouse gas production and sorption/degradation of two herbicides in a Minnesota soil. Chemosphere 2009, 77, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Pan, L.; Ying, G.; Kookana, R.S. Enhanced and irreversible sorption of pesticide pyrimethanil by soil amended with biochars. J. Environ. Sci. 2010, 22, 615–620. [Google Scholar] [CrossRef]
- Kookana, R.S. The role of biochar in modifying the environmental fate, bioavailability, and efficacy of pesticides in soils: A review. Aust. J. Soil Res. 2010, 48, 627–637. [Google Scholar] [CrossRef]
- Nag, S.K.; Kookana, R.; Smith, L.; Krull, E.; Macdonald, L.M.; Gill, G. Poor efficacy of herbicides in biochar-amended soils as affected by their chemistry and mode of action. Chemosphere 2011, 84, 1572–1577. [Google Scholar] [PubMed]
- Fang, W.; Wang, Q.; Han, D.; Liu, P.; Huang, B.; Yan, D.; Ouyang, C.; Li, Y.; Cao, A. The effects and mode of action of biochar on the degradation of methyl isothiocyanate in soil. Sci. Total Environ. 2016, 565, 339–345. [Google Scholar] [PubMed]
- Primo, P.D.; Gamliel, A.; Austerweil, M.; Steiner, B.; Beniches, M.; Peretz-Alon, I.; Katan, J. Accelerated degradation of metam-sodium and dazomet in soil: Characterization and consequences for pathogen control. Crop Prot. 2003, 22, 635–646. [Google Scholar] [CrossRef]
- Warton, B.; Matthiessen, J.N. The crucial role of calcium interacting with soil pH in enhanced biodegradation of metam-sodium. Pest Manag. Sci. 2005, 61, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Hebert, V.R.; Miller, G.C. Gas-phase reaction of methyl isothiocyanate and methyl isocyanate with hydroxyl radicals under static relative rate conditions. J. Agric. Food Chem. 2014, 62, 1792–1795. [Google Scholar]
- Bourke, J.; Manleyharris, M.; Fushimi, C.; Dowaki, K. Do all carbonized charcoals have the same chemical structure? 2. A model of the chemical structure of carbonized charcoal. Ind. Eng. Chem. Res. 2007, 46, 5954–5967. [Google Scholar] [CrossRef]
- Lehmann, J.D.; Joseph, S. Biochar for Environmental Management: Science and Technology; Earthscan: London, UK, 2009; Volume 25, pp. 15801–15811. [Google Scholar]
- Fang, G.; Zhu, C.; Dionysiou, D.D.; Gao, J.; Zhou, D. Mechanism of hydroxyl radical generation from biochar suspensions: Implications to diethyl phthalate degradation. Bioresour. Technol. 2015, 176, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Johnson, E.J.; Chefetz, B.; Zhu, L.; Xing, B. Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: Role of polarity and accessibility. Environ. Sci. Technol. 2005, 39, 6138–6146. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cao, B.; Wang, S.; Yuan, Q. H2S catalytic oxidation on impregnated activated carbon: Experiment and modelling. Chem. Eng. J. 2006, 118, 133–139. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Wnętrzak, R.; Leahy, J.J.; Hayes, M.H.B.; Kwapiński, W.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Dungan, R.S.; Gan, J.; Yates, S.R. Accelerated degradation of methyl isothiocyanate in soil. Water Air Soil Pollut. 2003, 142, 299–310. [Google Scholar] [CrossRef]
- Gan, J.; Papiernik, S.K.; Yates, S.R.; Jury, W.A. Temperature and moisture effects on fumigant degradation in soil. J. Environ. Qual. 1999, 28, 1436–1441. [Google Scholar] [CrossRef]
- Wang, Q.; Gao, S.; Wang, D.; Spokas, K.; Cao, A.; Yan, D. Mechanisms for 1,3-dichloropropene dissipation in biochar-amended soils. J. Agric. Food Chem. 2016, 64, 2531–2540. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Fang, W.; Yan, D.; Han, D.; Li, Y.; Ouyang, C.; Guo, M.; Cao, A. The effects of biochar amendment on dimethyl disulfide emission and efficacy against soil-borne pests. Water Air Soil Pollut. 2016, 227, 1–9. [Google Scholar] [CrossRef]
- Silber, A.; Levkovitch, I.; Graber, E.R. pH-dependent mineral release and surface properties of cornstraw biochar: Agronomic implications. Environ. Sci. Technol. 2010, 44, 9318–9323. [Google Scholar] [CrossRef] [PubMed]
- Papiernik, S.K.; Yates, S.; Gan, J. An approach for estimating the permeability of agricultural films. Environ. Sci. Technol. 2001, 35, 1240–1246. [Google Scholar] [CrossRef] [PubMed]
- Klose, S.; Acosta, V. Microbial community composition and enzyme activities in a sandy loam soil after fumigation with methyl bromide or alternative biocides. Soil Biol. Biochem. 2006, 38, 1243–1254. [Google Scholar] [CrossRef]
- Komada, H. Development of a selective medium for quantitative isolation of Fusarium oxysporum from natural soil. Rev. Plant Prot. Res. 1975, 8, 114–124. [Google Scholar]
- Masago, H.; Yoshikawa, M.; Fukada, M. Selective inhibition of Pythium spp. on a medium for direct isolation of Phytophthora spp. from soils and plants. Phytopathology 1977, 67, 425–428. [Google Scholar] [CrossRef]
- Liu, W. Plant Pathogenic Nematodes; China Agriculture Press: Beijing, China, 2004; Volume 2000, p. 373. [Google Scholar]
- Qin, R.; Gao, S.; Ajwa, H.; Hanson, B.D.; Trout, T.J.; Wang, D.; Guo, M. Interactive effect of organic amendment and environmental factors on degradation of 1,3-dichloropropene and chloropicrin in soil. J. Agric. Food Chem. 2009, 57, 9063–9070. [Google Scholar] [CrossRef] [PubMed]
- Hadiri, N.E.; Ammati, M.; Chgoura, M.; Mounir, K. Behavior of 1,3-dichloropropene and methyl isothiocyanate in undisturbed soil columns. Chemosphere 2003, 52, 893–899. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).