Abstract
The production of biodiesel from low-cost raw materials which generally contain high amounts of free fatty acids (FFAs) is a valuable alternative that would make their production costs more competitive than petroleum-derived fuel. Currently, the production of biodiesel from this kind of raw materials comprises a two-stage process, which requires an initial acid-catalyzed esterification of the FFA, followed by a base-catalyzed transesterification of the triglycerides. Commonly, the acid H2SO4 is the catalyst on the first step of this process. It must be said, however, that major drawbacks such as substantial reactor corrosion and the great generation of wastes, including the salts formed due to neutralization of the mineral acid, are negative and virtually unsurmountable aspects of this protocol. In this paper, tin(II) chloride dihydrate (SnCl2·2H2O), an inexpensive Lewis acid, was evaluated as catalyst on the ethanolysis of oleic acid, which is the major component of several fat and vegetable oils feedstocks. Tin chloride efficiently promoted the conversion of oleic acid into ethyl oleate in ethanol solution and in soybean oil samples, under mild reaction conditions. The SnCl2 catalyst was shown to be as active as the mineral acid H2SO4. Its use has relevant advantages in comparison to mineral acids catalysts, such as less corrosion of the reactors and as well as avoiding the unnecessary neutralization of products. Herein, the effect of the principal parameters of reaction on the yield and rate of ethyl oleate production has been investigated. Kinetic measurements revealed that the esterification of oleic acid catalyzed by SnCl2·2H2O is first-order in relation to both FFAs and catalyst concentration. Experimentally, it was verified that the energy of activation of the esterification reaction of oleic acid catalyzed by SnCl2 was very close those reported for H2SO4.
1. Introduction
Biodiesel is a suitable substitute for petroleum-derived diesel. It is biodegradable, almost sulfurless and a renewable fuel, though still not produced by environmentally friendly routes. This alternative fuel consists of methyl or ethyl esters, a result of either transesterification of triacylglycerides (TG) or esterification of free fatty acids (FFAs) [1]. Biodiesel fuel has become more attractive because of its environmental benefits, due to the fact that plants and vegetable oils and animal fats are renewable biomass sources [2]. Currently, most of the biodiesel comes up from transesterification of edible resources such as animal fats, vegetable oils, and even waste cooking oils, under alkaline catalysis conditions [3,4,5]. However, the high consumption of catalysts, the formation of soaps, and the low yields, make biodiesel currently more expensive than petroleum-derived fuel [6].
The common processes of biodiesel production from low-cost raw materials use mineral acids as catalysts, owing to the high amounts of FFAs that those resources contain, which make the manufacture of biodiesel from these feedstocks incompatible with alkaline catalysts [7]. Thus, two alternative approaches are normally used. The first is a two-step process, which requires an initial acid-catalyzed esterification of the FFA, followed by a base-catalyzed transesterification of the TG. Second, a one-step process that only uses an acid catalyst that simultaneously promotes both esterification and transesterification reactions [8]. It must be said, however, that major drawbacks such as reactor corrosion and substantial generation of by-products and wastes, including the salts formed as a result of the mineral acid neutralization, which must be disposed off into the environment, are negative and virtually unsurmountable aspects for the mineral acid-catalyzed process [9].
1.1. Homogeneous catalysts based on Brønsted acids for FFAs esterification reactions.
The alkaline catalysts show high performance for obtaining vegetable oils with high quality, but a question often arises; that is, the oils contain significant amounts of FFA which cannot be converted into biodiesel but rather give a lot of soap. An alternative way of processing these vegetable oils is to use a Brønsted acid catalyst. Currently, the catalysts more used in biodiesel production are the organic acids, such as the derivates of toluenesulfonic acid and, more often, mineral acids such as H2SO4. However, this latter process gives rise to problems linked to the corrosive action of the liquid acid catalyst and to the high quantity of by-products formed. In addition, the fact that homogeneous acids such as sulfuric acid need long reaction times, more than the alkaline catalysts, becomes a serious problem. These problems can be minimized by use of high catalyst concentrations. A further complication of working with high amounts of acid catalyst becomes apparent during the catalyst neutralization process, which precedes product separation. Since CaO addition during neutralization is proportional to the concentration of acid needed in the reactor, high acid concentrations lead to increased CaO costs, greater waste formation and higher production costs. Thus, we can be conclude that the main drawback of the pre-esterification method consists in the necessity to remove and to neutralize the homogeneous acid catalyst from the oil after pre-esterification [8, 9]. Actually, a 10,000 ton/year biodiesel plant co-produces 2,000 ton/year of CaSO4 [8], with the sulfuric acid being neutralized with CaO. A solid acid catalyst could to eliminate these problems, but nowadays the proposed catalysts have not yielded satisfactory results, and much greater research efforts are necessary [8,9,10]. Moreover, the necessity to eliminate the water formed in FFA esterification still remains because the presence of water may to contribute to the leaching of the catalyst [8,9,10]. Another drawback of heterogeneous catalysis is the necessity for drastic reaction conditions. Thus, the environmental aspects of biodiesel production have been assuming significant importance. The effluent generated in the catalyst neutralization processes and the biodiesel co-products must be discarded in the environment causing great environmental impact. For these reasons, the alternative use of Lewis acids as catalysts for that process has been the goal of several discussions and will be addressed on next section.
1.2. Environmental benefits of use of Lewis acid catalysts for Biodiesel production from FFA esterification reactions.
A great-advantage with acid catalysts is that they can produce biodiesel directly from low-cost lipid feedstocks, generally associated with high FFA concentrations (low-cost feedstocks, such as used cooking oil and greases, commonly have FFAs levels > 6%). However, the acid-catalyzed conversion of low-cost feedstocks leads to the formation of significant quantities of water, which has a negative effect on biodiesel production since water can hydrolyze the ester products, producing (again) FFAs. Though ester hydrolysis can occur under either acid or base catalysis, acid catalysis is generally believed to be more tolerant of moisture and high FFA levels in the starting feedstock and, hence, more suitable for low-grade fats and greases.
The biodiesel manufacturing process has to accomplish the reduction of environmentally undesirable products, provoked by conventional catalytic processes [10]. The extensive demand for cleaner methodologies has forced the chemical industry to search for environmentally-friend acidic catalysts. A notable example are the solid acids [11]. These solid catalysts, which normally present Lewis acidity, are easily separated from the reaction media and are potentially less corrosive to the reactors [12,13,14,15]. However, their use usually require drastic reaction conditions, i.e. high temperatures and elevated pressure. Thus, the development of alternative catalysts for the esterification of FFAs, based on the Lewis acids, which operate under mild conditions of reaction and are less corrosive, rather than traditional Brønsted acids, is one of the main challenges to be overcome. Moreover, this technology could thus allow the production of biodiesel at more competitive costs, in processes of lower environmental impact [16].
The use of Lewis acids in organic synthesis, especially in catalysis, is one of the most rapidly developing fields in synthetic organic chemistry. As result, catalysts based on metal salts such as aluminium, titanium and tin halides are widely used in several synthetic methods [17]. Conversely, the use of catalysts based on metal complexes as such rare earth metal triflates and metallocene compounds has been also developed [18]. Recently, the use of both types of catalysts, i.e. metal salts and metal complexes in esterification and transesterification reactions for biodiesel production has been described. For instance, Di-Serio and co-workers have reported the use of carboxylic salts of metals such as Cd, Mn, Pb and Zn for the production of biodiesel from vegetable oil containing high amounts of FFA [19]. However, among the metals Lewis acids with more applicability on esterification reactions, those containing tin metal have been more extensively explored [20,21,22]. For instance, Otera and co-workers have reported the successful use of distannoxanes as catalysts in esterification reactions of several organic substrates [23]. In addition, other organotin compounds such as BuSn(O)OH have shown high catalytic activity [24]. Conversely, although the catalytic activity of some tributyltin alkoxides is recognized, the final yield of the process proved to be disappointing [24]. Recently, the use of metal complexes with the pyrone ligand (3-hydroxy-2-methyl-4-pyrone) as catalysts in soybean oil methanolysis under homogeneous conditions was described by Ziani and co-workers, and resulted in a high yield of fatty acid methyl esters (FAME) [25, 26]. Despite the successful use of these catalysts, their application to biodiesel manufacturing is restricted due to their cost and delicate conditions of synthesis. Moreover, these catalysts are active only for methanolysis reactions. In view of that, replacing the corrosive liquid catalysts by alternative Lewis acid catalysts might be the lead for the development of an innovative technology for biodiesel production [26].
In this work, the use of SnCl2·2H2O as catalyst for the ethanolysis of oleic acid (pure and added to soybean oil) was evaluated. The investigation described herein involves the correlation between key parameters of reaction such as catalyst and fatty acid concentration, as well as the influence of the molar ratio FFA/ethanol and the temperature upon the ethyl oleate production.
2. Experimental Section
2.1. Chemicals
All the chemicals were purchased from known chemical companies. They were used without prior purification, unless otherwise stated. Ethanol was purified twice by distillation over phosphorous pentoxide. The SnCl2·2H2O was purchased from Sigma-Aldrich (Milwaukee, WI), and sulfuric acid (H2SO4, 98 %wt) from Química Moderna (São Paulo, SP, Brazil). Hexane and chloroform were purchased from Vetec (São Paulo, SP, Brazil). The oleic acid and their ethyl ester were purchased from Química Moderna (São Paulo, SP, Brazil).
2.2. General reaction procedure
The reactions were carried out in a 50 mL three-necked glass flask equipped with a reflux condenser and a thermometer. All catalytic tests were performed in triplicate. In the catalytic runs, excess of ethanol was added to the reactor containing the FFA in order to drive the equilibrium towards the formation of ester and for becomes the reaction rate not dependent of the concentration of this substrate. In a typical run, ethanol solution (10 mL, 120 mmol) and oleic acid (1 mmol) was heated to the reflux temperature followed by addition of SnCl2·2H2O (0.01 to 0.4 mmol).
2.2.1. The SnCl2 catalyst versus H2SO4
Catalytic runs with sulfuric acid were also carried out for comparison purposes with the latter since this is a common homogeneous catalyst used in a number of related investigations. The sulfuric acid and tin chloride concentrations used were 0.1 and 0.4 mmol respectively. The reaction was continuously monitored by GC analysis of aliquots taken at regular intervals of 30 minutes. The yields for those catalytic processes were obtained by means of the GC peak areas of the ethyl ester in comparison with the corresponding calibration curve. Alternatively, aliquots from the reactions were titrated against an alcoholic solution of KOH, aiming a monitoring at short intervals of time (ca. 10 min). The amount of KOH to neutralize the residual fatty acid and the acid catalyst (SnCl2 or H2SO4) was taking in account to calculate the concentration of the ethyl oleate in solution. Although negligible hydrolysis of FAEE may take place in these reactions, the results were satisfactorily reproducible and were in accordance with those obtained by CG.
2.2.2. Esterification of soybean oil with high amounts of oleic acid
Soybean oil (3.26 g) containing 5 and 10% w/w oleic acid was prepared manually by mixing oil and oleic acid. Ethanol (120 mmol, 10 mL) containing SnCl2 was then added to the reactor and process started and maintained at reflux temperature. The molar ratio soybean oil:catalyst was maintained at 100:1. The reactions were carried out in a 50-mL three-necked glass flask equipped with a reflux condenser, a conventional thermometer and a magnetic stirrer. Samples regular time intervals were taken and the residual oleic acid was determined via titration with an alcoholic solution of KOH (similarly to item 3.2.1.). The experiments were performed in triplicate.
2.3. Kinetic measurements
Two procedures have been used herein to evaluate the effect of the catalyst and the oleic acid concentrations on the reaction rate. For the fatty acid, the molar ratio between ethanol, oleic acid and SnCl2 has been preserved to 120:1:0.1. For the catalyst, only the molar ratio ethanol: oleic acid was maintained constant, i.e. 120:1. The catalyst concentration was range of 0.01-0.4 mmol. In both procedures, the residual acid was titrated against an alcoholic solution of KOH 0.01 mol·L-1 to follow the progress of the reaction.
2.4. Identification of the reaction products
The reaction products were isolated by a column chromatography (silica) using a 1:1 CHCl3/hexane mixture as eluent. The analysis by GC-MS were carried out in a Shimadzu GC 17A gas chromatograph coupled with a MS-QP 5050A mass spectrometer Shimadzu (Tokyo, Japan) with a DB5 capillary column (30 m length, 0.25 mm i.d., 0.25 mm film thickness) and helium as the carrier gas at 2 mL/min.
The temperature program was as follows: 80 °C for 1 min, 10 °C/min up to 280 °C and holding time of 5 min. The GC injector and MS ion source temperatures have been in the range of 250 to 260 °C. The MS detector has operated in the EI mode at 70 eV, with a scanning range of 50 to 500 m/z.
3. Results and Discussion
3.1. General aspects
The ethanolysis of FFA is a typical reversible acid-catalyzed reaction that produces ester and water as by-product:
In this work, first the esterification of oleic acid with ethanol in the absence of the acidic catalysts (SnCl2·2H2O and H2SO4) was conducted. This produced no significant yields of ethyl oleate, in spite of the high molar ratio of ethanol/fatty acid used, i.e. 120:1, (see Experimental for full details). Very low conversions of oleic acid into ethyl oleate (ca. 12%, see Figure 1) occurred, even after more than 12 hours of reaction.
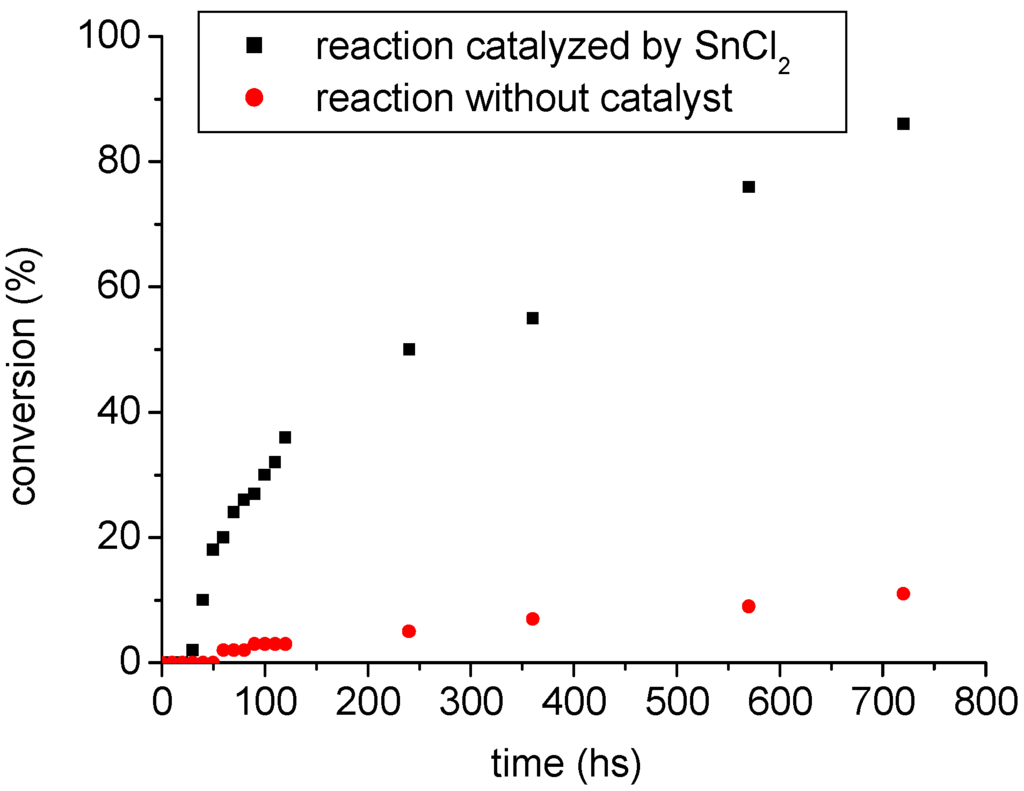
Figure 1.
Trend of conversion of oleic acid into ethyl oleate.
Figure 1.
Trend of conversion of oleic acid into ethyl oleate.
Conversely, in the presence of SnCl2, much greater yield (>90%) with a high selectivity (>93%, see Experimental) were obtained after 12 hours of reaction. Thus, these results are evidence that the excess of ethanol has no significant effect on the yield or the reaction rate.
In all catalytic runs the ethyl oleate yield increased as the reaction time increased. However, when the reaction time goes beyond 12 hours, the ester concentration remains almost invariable. The catalytic tests were performed in triplicate, with a molar ratio fatty acid: catalyst (100:1). It is worth mentioning that these conditions were not optimized to achieve higher yields at shorter reaction times. However, they provide an adequate methodology to evaluate the catalytic activity of SnCl2·2H2O on these reactions.
3.2. The SnCl2 catalyst versus H2SO4: A comparative study
The performance of both acids catalysts was outstanding, as shown in Figure 2, where the kinetic curves are presented. Note that the reaction yields increase steadily, reach the maximum values (ca. 90 %) after a reaction time of ca. 120 minutes and thus stay almost invariable afterwards.
The two acidic catalysts have different structures and acid character and certainly, different mechanisms of action. In spite of that they displayed quite similar activities, as can be confirmed by the attainment of comparable ethyl oleate yields at a given reaction time (Figure 2). Note that reaction yields have increased steadily to a maximum value of ca. 90 %, in approximately 120 minutes after setting up the reaction. The monitoring of reaction for periods higher than 120 minutes reveals that the yields of both remained invariable after this time, as can be observed by data of Table 1.
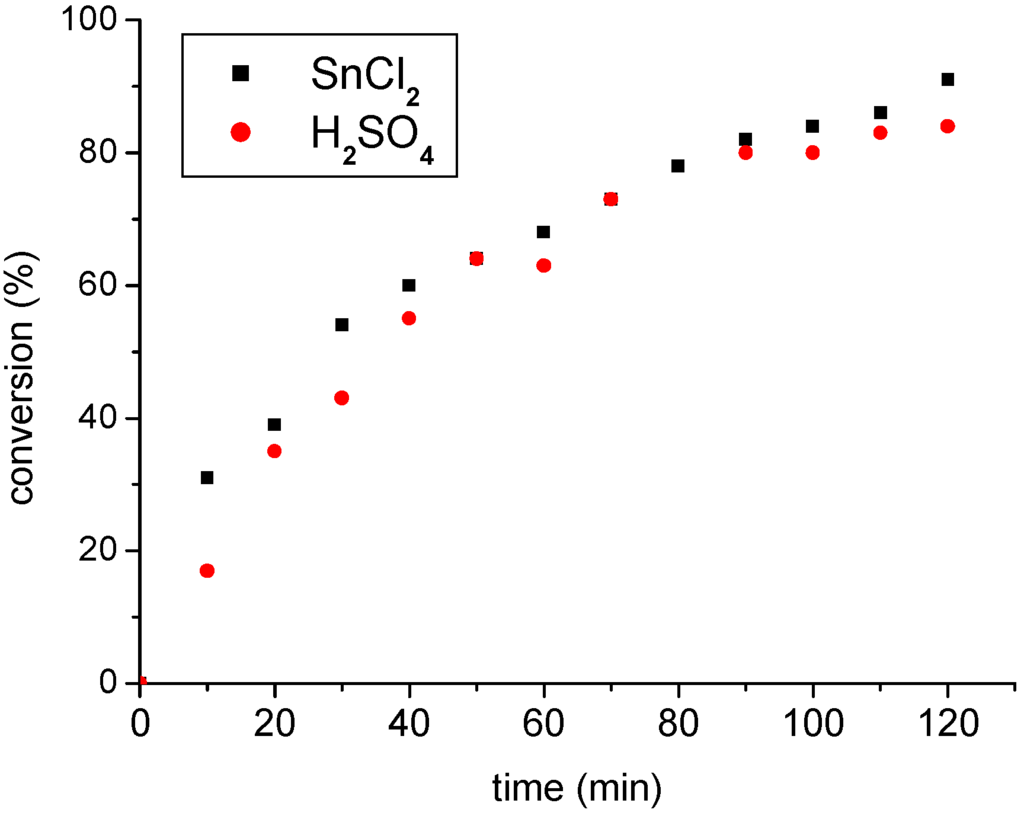
Figure 2.
Ethanolysis of oleic catalyzed by Brønsted (H2SO4) and Lewis acids (SnCl2).
Figure 2.
Ethanolysis of oleic catalyzed by Brønsted (H2SO4) and Lewis acids (SnCl2).

Table 1.
Conversion and selectivity of ethanolysis of oleic acid catalyzed by SnCl2 and H2SO4 1,2
| Time (h) | SnCl2 | H2SO4 | ||
|---|---|---|---|---|
| Conversion (%) | Selectivity (%) | Conversion (%) | Selectivity (%) | |
| 0 | 0 | 0 | 0 | 0 |
| 1 | 62 | 92 | 65 | 90 |
| 2 | 93 | 94 | 92 | 89 |
| 4 | 90 | 95 | 94 | 87 |
| 6 | 90 | 93 | 95 | 89 |
| 8 | 91 | 93 | 94 | 88 |
1 Conversion and selectivity were determined by GC analyses;2 Reaction conditions as described in section 2.2.1.
3.3. Ethanolysis of oleic acid catalyzed by SnCl2·2H2O: kinetic studies
3.3.1. The effect of oleic acid concentration
Literature data have attributed a first order dependence on fatty acid concentration in the majority of esterification reactions [27]. From this standpoint, in this present case we can assume that equation 1 could be used to describe the substrate concentration variation with relation to time:
Ln [oleic acid]t = -kt + ln [oleic acid]0
Thus, to obtain kinetic information, i.e. the reaction order in relation to the oleic acid concentration, plots of ln[oleic acid] versus time were thus built for each acid-catalyzed process (Figure 3 presents only the data of ethanolysis of the oleic acid tin-catalyzed).
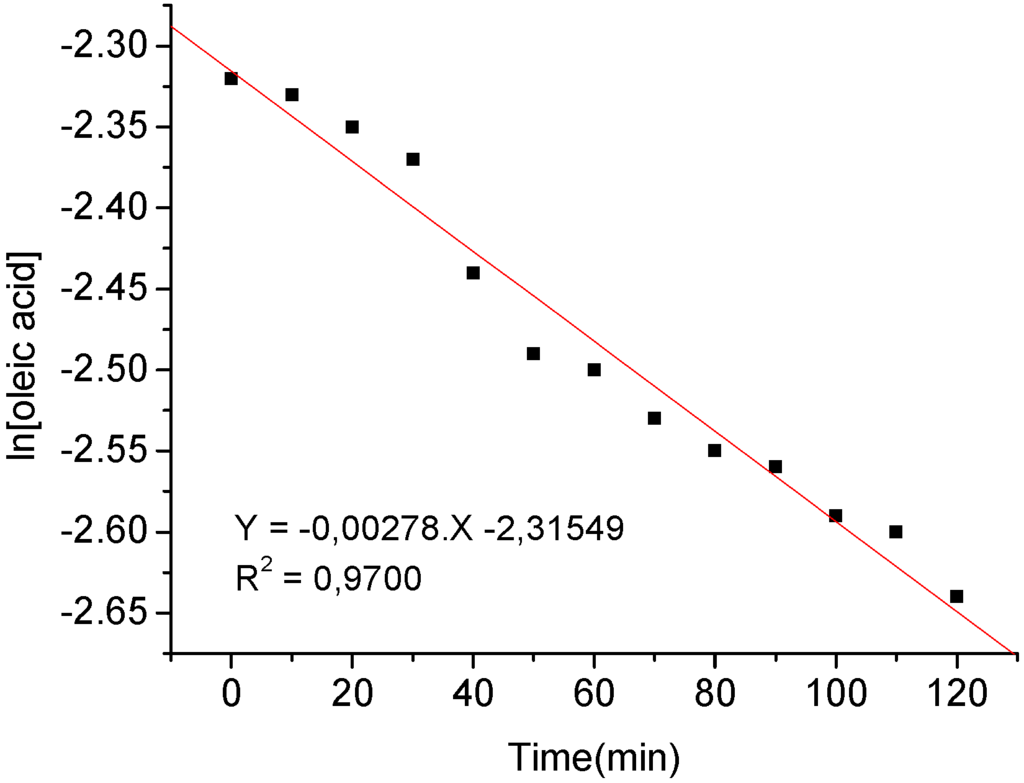
Figure 3.
Arrenhius plot of oleic acid concentration as function of time for ethanolysis catalyzed by SnCl2.
Figure 3.
Arrenhius plot of oleic acid concentration as function of time for ethanolysis catalyzed by SnCl2.
The resulting data fit a first order kinetic behavior. It is important to mention that a high molar excess of ethanol in relation to oleic acid (120:1) was used to assure that the ethanol concentration would remain essentially constant during the reaction course (see Experimental). As displayed in Table 2, the high correlation coefficients of the resulting linear equations obtained are indicative of the fact that there is a first order dependenence for both esterification reaction catalyzed by sulfuric acid and tin chloride. From the angular coefficients of such equations, the rate constants (k) and then the half-life times (t1/2 = k/0.693) for each process was thus obtained.

Table 2.
The effect of substrate concentration on the ethanolysis of oleic acid catalyzed by SnCl2.
| Catalyst | Linear Equation | T1/2 (min) | R2 |
|---|---|---|---|
| H2SO4 | ln[oleic acid] = -0.00359 *t-2.4414 | 206 | 0.9850 |
| SnCl2 | ln[oleic acid] = -0.00278 *t-2.3155 | 249 | 0.9700 |
3.3.2. The effect of SnCl2·2H2O concentration
The effectiveness of the catalyst SnCl2·2H2O has been investigated over a broad range of concentrations but only the significant results are shown in Figure 4. To control the accuracy of the kinetic data related to the residual concentration of oleic acid, it has been essential to maintain the acid-catalyzed process distant from the equilibrium position. In view of that, the kinetic measurements have been carried out within two hours of reaction. Furthermore, since the GC column used herein (DB5 capillary) is not appropriate to analyze fatty acids, the concentration of the residual oleic acid was determined directly from the reaction medium via titration with an alcoholic solution of KOH (more details are described in Experimental Section) [28].
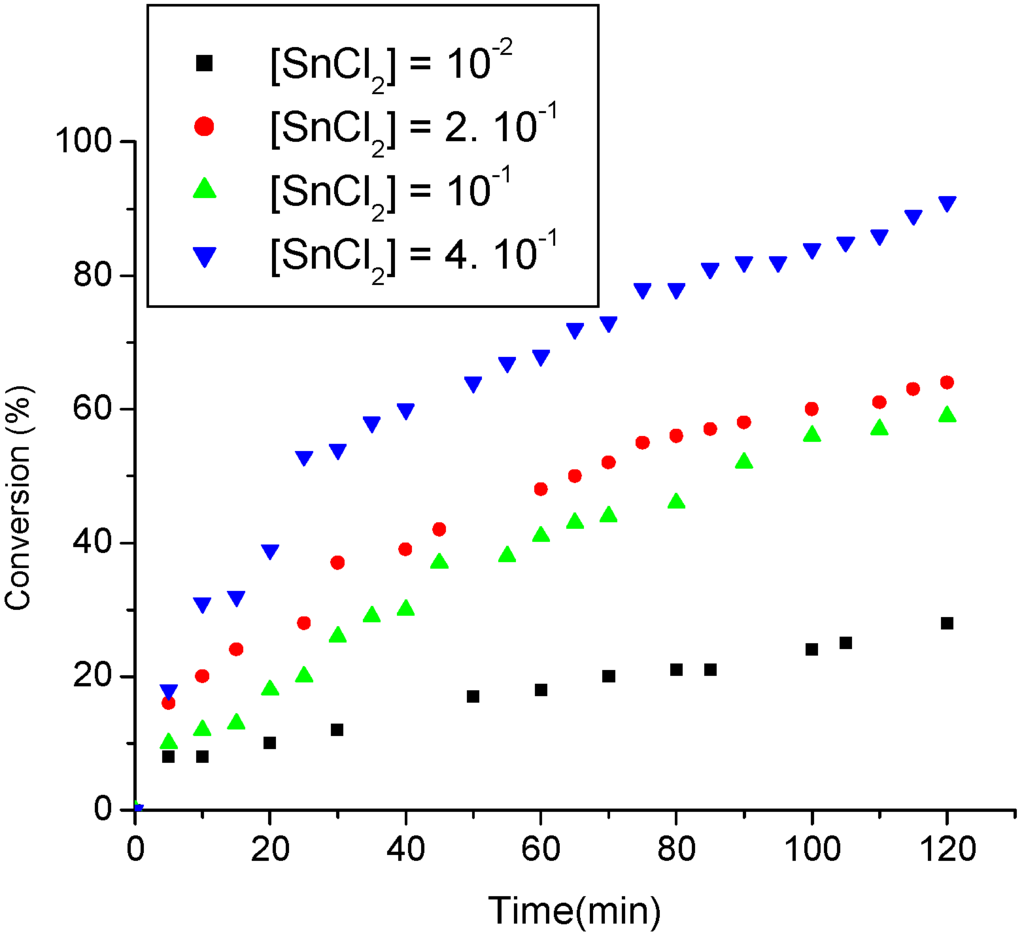
Figure 4.
Effect of the concentration of SnCl2·2H2O catalyst on the ethanolysis of oleic acid.
Figure 4.
Effect of the concentration of SnCl2·2H2O catalyst on the ethanolysis of oleic acid.
For to calculate the initial rate, only the first interval of thirty minutes was taken into account, because in this period the dependence in relation to oleic acid concentration is of pseudo-zero order. This statement is not completely true, because in this time interval the substrate oleic acid is partially consumed. However, this effect is minimized because the initial concentration of oleic acid used was higher in relation to the other experiments. Thus, with a relatively acceptable error, we assume that only the tin chloride concentration is ranging in each run. Another important aspect is that chromatography analyses showed that the oleic acid is mostly converted into the ethyl ester (ca.>90%) in all catalytic runs. The initial SnCl2 concentration in each reaction varied from 0.01-0.40 mmol. The oleic acid load employed was 1 mmol and a large excess of ethanol was used. As a general tendency, an increase in the catalyst concentration caused an improvement in the ethyl oleate yields at any reaction time. Low molar ratios between the oleic acid and SnCl2 (1:0.01) afforded approximately 24% of ethyl oleate after two hours of reaction. However, the use of high molar ratio (1:0.4) for the same reaction interval has revealed a significant yield increase (up to ca. 90%). The results shown in Figure 4 are concomitant with the linear correlation shown in Figure 5.
The increases of the catalyst concentration has a noticeable effect on the conversion rate of the oleic acid into ethyl oleate. This fact can be attributed to higher number of molecules of substrate activated by polarization of the carbonyl, in presence of Sn+2 catalyst. Thus, the nucleophilic attack by ethanol becomes more favorable and consequently, a increases on the formation of ester was observed. The angular coefficient of the curve obtained is suggestive of a first-order dependence (ca. 0.87), in relation to catalyst concentration (see equation 2 and Figure 5):
ln [SnCl2] = - 5,58 – 0.87 ln (initial rate) R2 = 0.9784
Kulkarni and Sawant have found a second-order kinetic rate in the esterification of lauric acid with castor oil relative to the same catalyst, SnCl2 [29]. However, the reaction conditions in their study, including temperature and the substrate, were different from those described in this work.
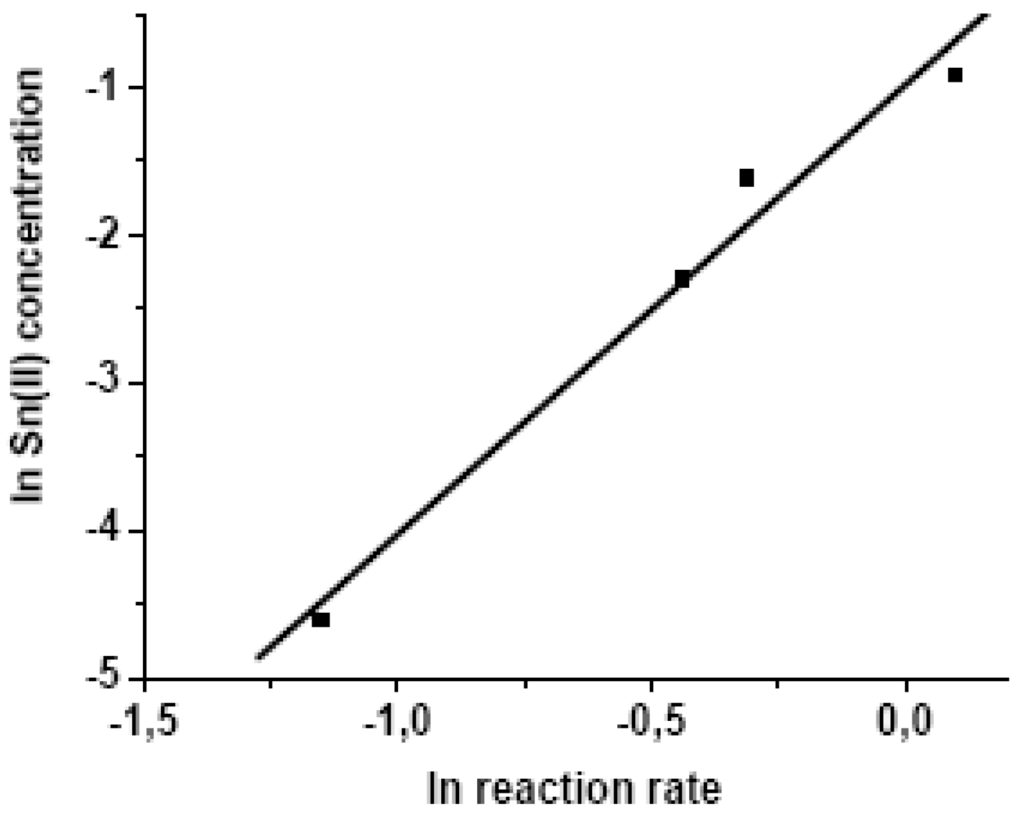
Figure 5.
Plot linear of ln [SnCl2] (effect of the concentration of SnCl2·2H2O catalyst on the ethanolysis of oleic acid).
Figure 5.
Plot linear of ln [SnCl2] (effect of the concentration of SnCl2·2H2O catalyst on the ethanolysis of oleic acid).
3.3.3. The effect of temperature on the ethanolysis of oleic acid SnCl2-catalyzed
It was verified that the temperature noticeably affects both reaction rate and conversion of oleic acid into ethyl oleate. For instance, at room temperature, even for longer time reactions, only low conversions was obtained. However, at temperatures up of 45 °C, occurs a significance increase of the initial rate of reaction, and consequently, highers conversions were reached.
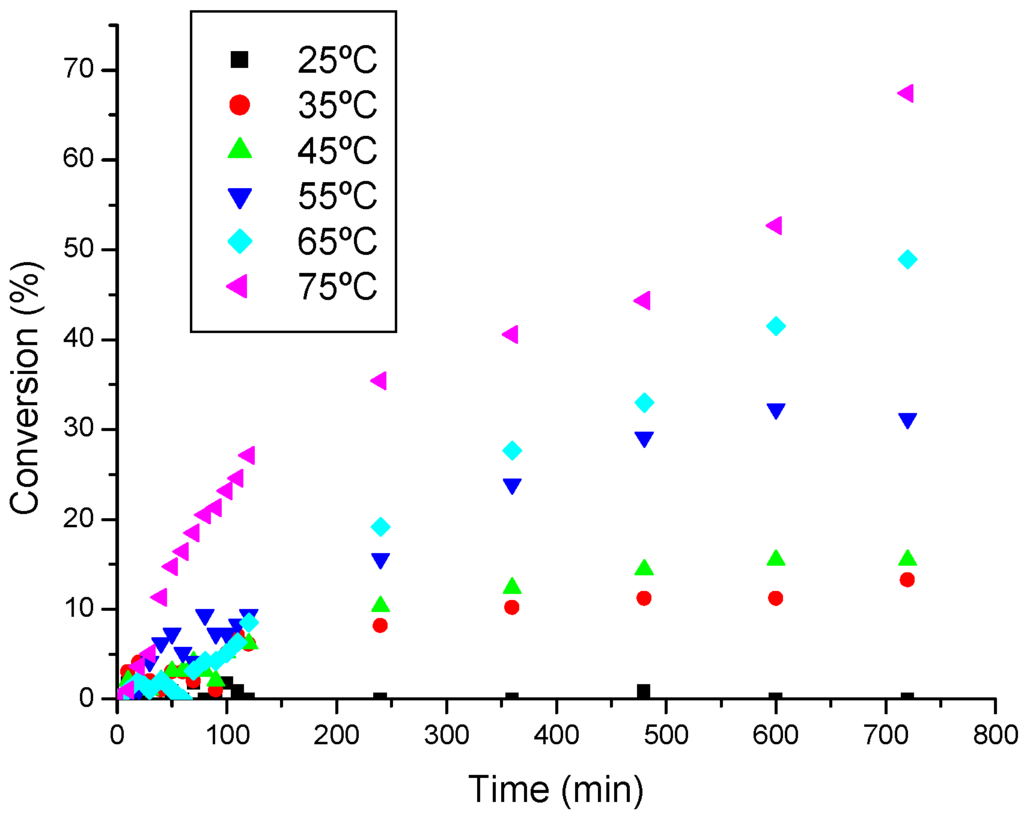
Figure 6.
Effect of the temperature on the ethanolysis of oleic acid catalyzed by SnCl2.
Figure 6.
Effect of the temperature on the ethanolysis of oleic acid catalyzed by SnCl2.
Table 3 shows the values for the rate constant (k) obtained at each temperature (see Figure 6). Expectedly, the increase in the reaction temperature has caused a corresponding improvement on the reaction rate, especially at range of 45-75 °C. From of the resulting data shown in Table 3, the curve of the Figure 7 was constructed and employing a linear regression method, the angular coefficient (-E/ R) of the curve obtained allow us to calculate the activation energy of this process.

Table 3.
Linear equations for the decrease in oleic acid concentration as a function of time and the respective values of R2 and K.
| Temperature (ºC) | ln([oleic acid]/[oleic acid]o) = -kt | Linearity Coeffcient R2 |
|---|---|---|
| 35 | –3.0892 x10-6.t | -0.9090 |
| 45 | –4.2939 x 10-6.t | -0.9547 |
| 55 | –9.7396 x 10-6.t | -0.9762 |
| 65 | –1.6522 x 10-5.t | -0.9938 |
| 75 | –2.1535 x 10-5.t | -0.9701 |
a Rate constant for ethanolysis of oleic acid catalyzed by SnCl2 at each reaction temperature.
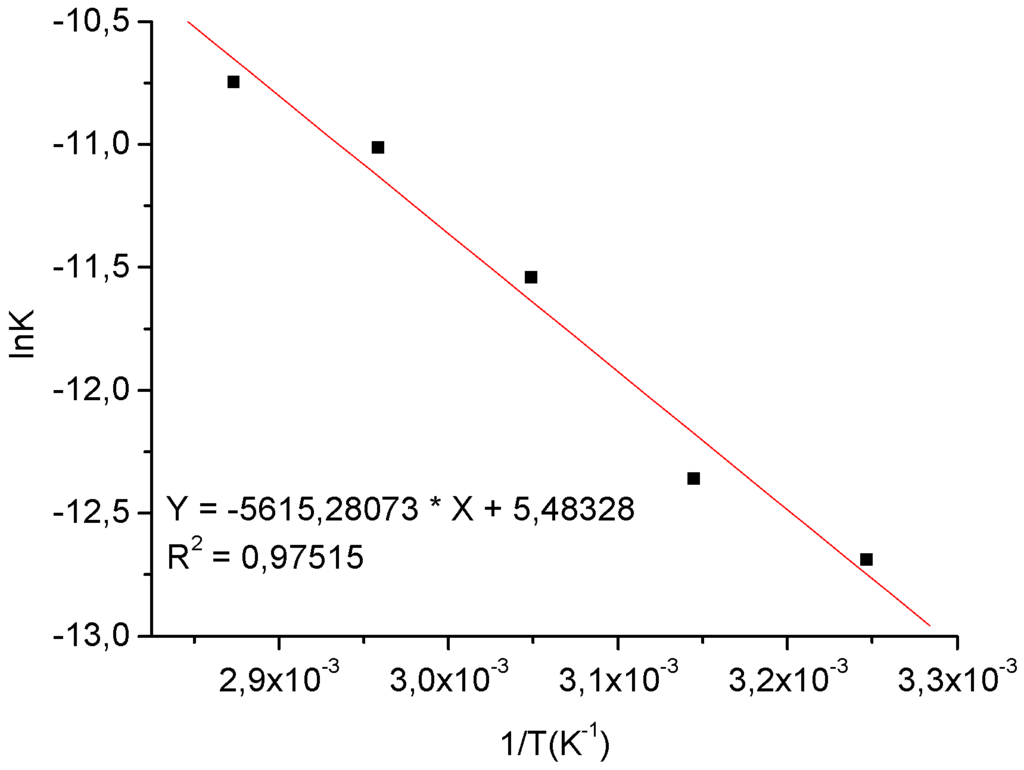
Figure 7.
Linear plot of ln K versus 1/T resulting from esterification of oleic acid catalyzed by tin chloride.
Figure 7.
Linear plot of ln K versus 1/T resulting from esterification of oleic acid catalyzed by tin chloride.
An activation energy of 46.69 kJ.mol-1 was determined This value is approximately similar to those found by Berrios and co-workers in the kinetic study of the H2SO4-catalyzed esterification of free fatty acids in sunflower oil [28]. These researchers found values of 50.74 Kj.mol-1 and 42.76 kJ.mol-1 in reactions where the catalyst concentration was of 5% and 10% w/w respectively. Note that, in spite of these relatively similar results, the ratio between the oleic acid and catalyst in our experiments was different those described by them (10:1), even for the two acidic catalysts with such different structures, i.e. H2SO4 and SnCl2.
3.3.4. Esterification of soybean oil with high amounts of FFA catalyzed by SnCl2.
Esterification reactions were carried out at reflux temperature, with the soybean oil having approximately 5 and 10 %w/w of additional oleic acid and with the molar ratio soybean oil:catalyst being maintained at 100:1 (more details in the Experimental).
It should be noted that the SnCl2 catalyst also showed a high activity on the ethanolysis of soybean oil with high amounts of oleic acid (Figure 8). Tin chloride efficiently lowered the acid value in a short time interval (ca. 120 min) under mild reaction conditions. Previously, we have found that no transesterification product was detected under these conditions (data no shown). However, an important result is that although the medium was of different viscosity and polarity compared to previous experiments described here, the catalytic activity of tin chloride remained unchanged and was not affected by the presence of soybean oil. This fact is indicative of the potential activity of tin chloride in the esterification reactions of raw materials with high amount of fatty acids.
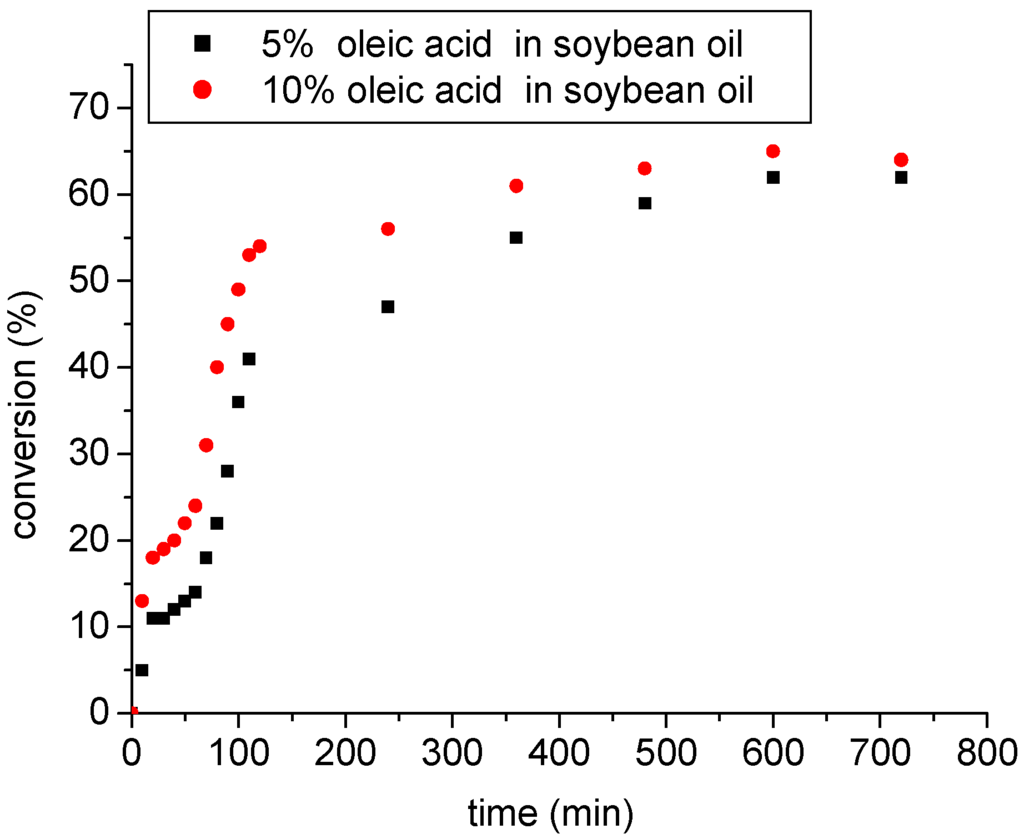
Figure 8.
Esterification of soybean oil with high amounts of oleic acid catalyzed by SnCl2.
Figure 8.
Esterification of soybean oil with high amounts of oleic acid catalyzed by SnCl2.
4. Conclusions
The authors think that even if biodiesel is not the total solution to any energy crisis, it certainly is an important component of a combined strategic approach to decrease our current dependence on fossil fuels.In these sense, the development of alternative catalysts for making biodiesel production more efficient and more environmentally benign assumes a strategic importance. In this paper, we have evaluated the catalytic activity of the SnCl2·2H2O in homogeneous phase in the esterification of FFA for biodiesel production, as an alternative to sulfuric acid. Tin chloride efficiently promotes the esterification of oleic acid in ethanol solutions and in the presence of soybean oil. The high yields achievable under mild reaction conditions are comparable to those obtained with a common acid catalysts such as sulfuric acid (H2SO4). Therefore, SnCl2 is a potential catalyst for the production biodiesel from low-cost raw materials, which currently have higher amounts of FFAs. Kinetic measurements revealed that the acid-catalyzed esterification is first-order relative to the concentration of both oleic acid and the SnCl2. The advantages of this protocol are the use of an available low-cost catalyst, which is easy to manipulate and potentially less corrosive. In addition, studies of the recovered and reutilization of the SnCl2 in consecutive catalytic processes are being developed in our labs. The results of this work suggest SnCl2 as a promising acid-catalyst for the production of biodiesel, in lower environmental impact processes. As a final remark, we believe that the application of SnCl2 can potentially promote a reduction of the costs related to biodiesel production.
Acknowledgements
The authors are grateful to the Brazilian research agencies, FUNARBE, FAPEMIG and CNPq for the financial support.
References
- Maa, F.; Hannab, M.A. Biodiesel Production-A Review. Bioresour. Technol. 1999, 70, 1–15. [Google Scholar]
- Zheng, S.; Kates, M.; Dubé, M.A.; Mclean, D.D. Acid-catalyzed production of biodiesel from waste frying oil. Biomass Bioener. 2006, 30, 267–272. [Google Scholar] [CrossRef]
- Chhetri, A.B.; Watts, K.C.; Islam, M.R. Waste Cooking Oil as an Alternate Feedstok for Biodiesel Prouction. Energies 2008, 1, 3–18. [Google Scholar] [CrossRef]
- Kima, H.J.; Kang, B.S.; Kim, M.J.; Park, Y.M.; Kimb, D.K.; Lee, J.S.; Lee, K.Y. Transesterification of vegetable oil to biodiesel using heterogeneous base catalyst. Catal. Today 2004, 93–95, 315–320. [Google Scholar] [CrossRef]
- Haas, M.J.; Karen, M.S.; Marmer, W.N.; Foglia, T.A. In situ alkaline transesterification: an effective method for the production of fatty acid esters from vegetable oils. J. Am. Oil Chem. Soc. 2004, 81, 83–89. [Google Scholar] [CrossRef]
- Haas, M.J. Improving the economics of biodiesel production through the use of low value lipids as feedstocks: vegetable oil soapstock. Fuel Process Technol. 2005, 86, 1087–1096. [Google Scholar] [CrossRef]
- Lotero, E.; Liu, Y.; Lopez, D.E.; Suwannakarn, K.; Bruce, D.A.; Goodwin, J.G. Synthesis of Biodiesel via Acid Catalysis. Ind. Eng. Chem. Res. 2005, 44, 25353–25363. [Google Scholar] [CrossRef]
- Zullaikah, S.; Lai, C.C.; Vali, S.R.; Ju, Y.H. A Two-step acid-catalyzed process for the production of biodiesel from rice bran oil. Bioresour. Technol. 2005, 96, 1889–1896. [Google Scholar] [CrossRef] [PubMed]
- Corma, A.; Garcıa, H. Lewis acids: From conventional homogeneous to green homogeneous and heterogeneous catalysis. Chem. Ver. 2003, 103, 4307–4365. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, J.M.; Miguel, V.U.; Errazu, A.F. Heterogeneous esterification of oil with high amount of free fatty acids. Fuel 2007, 86, 906–910. [Google Scholar] [CrossRef]
- Timofeeva, M.N. Acid Catalysis by Heteropoly Acids. Appl. Catal. 2003, 256, 19–35. [Google Scholar]
- Reis, S.C.M.; Lachter, E.R.R.; Nascimento, R.S.V.; Rodrigues Jr., J.A.; Reid, M.G. Transesterification of Brazilian vegetable oils with methanol over ion-exchange resins. J. Am. Oil Chem. Soc. 2005, 82, 661–665. [Google Scholar] [CrossRef]
- Ferreira, D.A.C.; Meneghetti, M.R.; Meneghetti, S.M.P.; Wolf, C.R. Methanolysis of soybean oil in the presence of tin(IV) complexes. Appl. Catal. 2007, 317, 58–61. [Google Scholar] [CrossRef]
- Alizadeh, M.H.; Kermanil, T.; Tayebee, R. A method for the acetylation of alcohols catalyzed by heteropolyoxometallates. Monatsh Chem. 2007, 138, 165–170. [Google Scholar] [CrossRef]
- Narasimharao, K.; Brown, D.R.; Lee, A.F.; Newman, A.D.; Siril, P.F.; Tavener, S.J.; Wilson, K. Structure–activity relations in Cs-doped heteropolyacid catalysts for biodiesel production. J. Catal. 2007, 248, 226–234. [Google Scholar] [CrossRef]
- Einloft, S.; Magalhães, T.O.; Donato, A.; Dullius, J.; Ligabue, R. Biodiesel from Rice Bran Oil: Transesterification by Tin Compounds. Ener. Fuels 2008, 22, 671–675. [Google Scholar] [CrossRef]
- Saito, S.; Yamamoto, H. Designer Lewis acid catalysts—bulky aluminium reagents for selective organic synthesis. Chem. Commun. 1997, 1585–1592. [Google Scholar] [CrossRef]
- Macedo, C.C.S.; Abreu, F.R.; Tavares, A.P.; Alves, M.B.; Zara, L.F.; Rubin, J.C.; Suares, P.A.Z. New Heteregoneous Metal-Oxides Based Catalyst for Biodiesel oil Transesterification. J. Braz. Chem. Soc. 2006, 17, 1291–1296. [Google Scholar] [CrossRef]
- Di Serio, M.; Tesser, R.; Dimiccoli, M.; Cammarota, F.; Nastasi, M.; Santacesaria, E. Synthesis of biodiesel via homogeneous Lewis acid catalyst. J. Mol. Catal. A 2005, 239, 111–117. [Google Scholar] [CrossRef]
- Jousseaume, B.; Laporte, C.; Rascle, M.-C.; Toupance, T. Investigations in the catalytic species of the distannoxane-catalyzed transcarbamoylation. Chem. Commun. 2003, 1428–1429. [Google Scholar] [CrossRef]
- Baumhof, P.; Mazitschek, R.; Giannis, A. A Mild Effective Method for the Transesterification of Carboxylic Acid Esters. Angew. Chem. Int. Ed. 2001, 40, 3672–3678. [Google Scholar] [CrossRef]
- Mascaretti, O.A.; Furlán, L.E. Esterifications, Transesterifications, and Deesterifications Mediated by Organotin Oxydes, Hydroxydes and Alkoxydes. Aldrichim. Acta 1997, 30, 55–68. [Google Scholar]
- Otera, J.; Dan-oh, N.; Nozaki, H. Novel Template Effects of Distannoxane Catalysts in Highly Efficient Transesterification and Esterification. J. Org. Chem. 1991, 56, 5307–5311. [Google Scholar] [CrossRef]
- Hoydonckx, H. E.; De Vos, D.E.; Chavan, S.A.; Jacobs, P.A. Esterification and transesterification of renewables chemicals. Topics Catal. 2004, 27, 83–88. [Google Scholar] [CrossRef]
- Abreu, F.R.; Lima, D.G.; Hamú, E.H.; Einloft, S.; Rubim, J.; Suarez, P.A.Z. New Metals Catalysts for Soybean Oil Transesterification. J. Am. Oil Chem. Soc. 2003, 80, 601–604. [Google Scholar] [CrossRef]
- Abreu, F.R.; Lima, D.G.; Hamú, E.H.; Wolf, C.R.; Suarez, P.A.Z. New multi-phase catalytic systems based on tin compounds active for vegetable oil transesterification reaction. J. Mol. Catal. A 2005, 227, 263–268. [Google Scholar] [CrossRef]
- Silva, M.J.; Augusti, R.; Cardoso, A.L. Investigations on the esterification of fatty acids catalyzed by heteropolyacids H3PW12O40. J. Am. Oil Chem. Soc. 2008, 85, 555–560. [Google Scholar]
- Berrios, M.; Siles, J.; Martın, M.A. A kinetic Study of the Esterification of Free fatty Acids (FFA) in Sunflower Oil. Fuel 2007, 86, 2383–2388. [Google Scholar] [CrossRef]
- Kulkarni, M.G.; Sawant, S.B. Kinetics of the Catalytic Esterification of Castor Oil with Lauric Acid Using n-Butyl Benzene as a Water Entrainer. J. Am. Oil. Chem. Soc. 2003, 80, 1033–1038. [Google Scholar] [CrossRef]
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
