Children’s Exposures to Pyrethroid Insecticides at Home: A Review of Data Collected in Published Exposure Measurement Studies Conducted in the United States
Abstract
:1. Introduction
2. Methods
2.1. Environmental Measurements Data
2.2. Urinary Biomonitoring Data
| Reference | Study Name a | Location | Year | N b | Age (years) | Media Collected | Sampling Frequency | Analytical Method e | Pyrethroids Measured |
|---|---|---|---|---|---|---|---|---|---|
| Bradman et al. [6] | CHAMACOS-QEA | Salinas Valley of Monterey California | 2002 | 20 | 0–2 | Outdoor air | 24-h integrated sample; at 2.5 L/min | Gas chromatograph/mass spectrometer in the selected ion monitoring mode | cis-and trans-Allethrin, bifenthrin, cyfluthrin, λ-cyhalothrin, cypermethrin, deltamethrin, esfenvalerate, cis- and trans-permethrin, phenothrin, resmethrin, tetramethrin |
| Indoor air | 24-h integrated sample; at 2.5 L/min | ||||||||
| Carpet dust | High volume small surface sampler (HVS3); 1 m2 area of carpet of main living area | ||||||||
| Hard floor surface wipe | SOF-WICK pad wetted with 10 mL 2-propanol; sample collected in a 900 cm2 area in kitchen or dining room | ||||||||
| Solid food | 24-h duplicate diet sample | ||||||||
| Liquid food | 24-h duplicate diet sample | ||||||||
| Surface Press | 3M Empore disks pressed on a hard floor for 5-min | ||||||||
| Clothing | Clothing worn for 3–4 h | ||||||||
| Chuang and Wilson [20] c | PEPCOT | Raleigh-Durham-Chapel Hill area of North Carolina | 2003–2005 | 50 | 3 | Solid food | 24-h duplicate diet sample; one sample collected annually for three consecutive years | Gas chromatograph/mass spectrometer in the selected ion monitoring mode | Bifenthrin, cyfluthrin, λ-cyhalothrin, cypermethrin, deltamethrin, esfenvalerate, fenpropathrin, fenvalerate, cis- and trans-permethrin, phenothrin, resmethrin, tetramethrin |
| 51 | <3 | ||||||||
| Julien et al. [15] | HPHI | Boston, Massa-chusetts | 2002–2003 | 42 | 4–17 | Dust (carpets & sofas combined), | 9 A Eureka Mighty-mite vacuum cleaner sampled ‘all accessible surfaces’ in living rooms. | Gas chromatograph/mass spectrometer in the multiple ion monitoring mode | Bifenthrin, cyfluthrin, λ-cyhalothrin, cypermethrin, deltamethrin, esfenvalerate, permethrin, phenothrin, resmethrin, tetramethrin |
| Hard floor surface wipes | SOF-WICK pad wetted with 5 mL of 2-propanol; sample collected each in a 929 cm2 area in kitchen and living room | ||||||||
| Lu et al. [9] d | CPES | Seattle, Washington | 2003–2004 | 23 | 3–11 | Solid and liquid foods | 24-h duplicate food samples consisting of nonorganic fruits, fruit juices, and vegetables. One 24-h food sample collected in the summer and fall of 2003. | Gas chromatograph with a halogen specific detector and/or a mass spectrometer | Bifenthrin, cypermethrin, esfenvalerate, permethrin |
| Atlanta, Georgia | 2006–2007 | 23 | 3–12 | 24-h duplicate food samples consisting of nonorganic fruits, fruit juices, and vegetables. One 24-h food sample collected in the summer 2005 and winter 2006. | |||||
| Morgan et al. [7,12] d | CTEPP (main study) | North Carolina & Ohio (6 counties in each state) | 2000–2001 | 257 | 2–5 | Soil | Top 0.5 cm of 0.1 m2 area of soil where child played most often | Gas chromatograph/mass spectrometer in the selected ion monitoring mode | Cyfluthrin, cis- and trans-permethrin |
| Outdoor air | 48-h integrated sample; at 4.0 L/min | ||||||||
| Indoor air | 48-h integrated sample; at 4.0 L/min | ||||||||
| Carpet dust | HVS3; 0.76 m2 area of carpet in most used room | ||||||||
| Solid food | 48-h duplicate diet sample | ||||||||
| Liquid food | 48-h duplicate diet sample | ||||||||
| Hard floor surface wipe | SOF-WICK pad wetted with 2 mL of 2-propanol on a 0.14 m2 area of floor child spent most time. | ||||||||
| Transferable residues | Polyurethane foam roller method; 1 roller to sample 3 different locations where child spent most time indoors | ||||||||
| Quandt et al. [13] | CFW | Western North Carolina and Virginia | 2001 | 41 | 1–7 | Wipes (hand, hard floor and toy) | SOF-WICK pad wetted with 10 mL of 2-propanol for 3 different wipes (all surfaces of hands, 0.42–0.84 m2 area of floor, mainly kitchens and hallways; samples collected from 2–3 non-plush toys | Gas chromatograph/ mass spectrometer | Esfenvalerate and cis- and trans-permethrin |
| Quiros-Alcala et al. [21] d | PDLHS | Salinas and Oakland, California | 2006 | 15 | 3–6 | Carpet dust | HVS3; 1–2 m2 area of carpet where child spent time playing | Electron impact gas chromatograph/mass spectrometer in multiple ion detection mode | Allethrin, bifenthrin, cypermethrin, deltamethrin, esfenvalerate, imiprothrin, cis-permethrin, trans-permethrin, phenothrin, prallethrin |
| 13 | |||||||||
| Starr et al. [16] | CTEPP-VAC | North Carolina & Ohio (6 counties in each state) | 2000–2001 | 85 | 2–5 | Vacuum dust bags | Existing bag from homeowners vacuum cleaner | Gas chromatograph/mass spectrometer in the selected ion monitoring mode | Allethrin, cyfluthrin, λ-cyhalothrin, cypermethrin, deltamethrin, esfenvalerate, fenpropathrin, imiprothrin, cis-permethrin, trans-permethrin, phenothrin, prallethrin, resmethrin, tetramethrin |
| Tulve et al. [17] | JAX-EXP | Jacksonville, Florida | 2001 | 9 | 4–6 | Outdoor air | 24-h integrated sample; at 3.8 L/min | Gas chromatograph/mass spectrometer in the multiple ion monitoring mode | cis-and trans-Allethrin, bifenthrin, cyfluthrin, λ-cyhalothrin, cypermethrin, deltamethrin, esfenvalerate, cis- and trans-permethrin, phenothrin, tetramethrin |
| Indoor air | 24-h integrated sample; at 3.8 L/min | ||||||||
| Hard floor wipe | SOF-WICK pad wetted with 10 mL of 2-propanol; sample collected each in a 929 cm2 in child’s play area and recent pesticide application area | ||||||||
| Solid food | 24-h duplicate diet sample | ||||||||
| Clothing (socks) | Socks worn for 1 or more hours |
| References | Study Name a | Location | Year of study | N | Age (years) | Collection Method | Frequency of Collection | Analytical Method | Measured Urinary Metabolites b |
|---|---|---|---|---|---|---|---|---|---|
| Barr et al. [8] | NHANES | National | 1999–2000 | 483 | 6–11 | Spot sample | One convenience sample | High performance liquid chromatograph- electrospray chemical ionization/tandem mass spectrometer | 3-PBA, 4-F-3-PBA, cis-DCCA, trans-DCCA, cis-DBCA |
| 2001–2002 | 580 | 6–11 | |||||||
| Lu et al. [5,18] c | CPES (summer) | Seattle, Washington | 2003 | 23 | 3–11 | Spot samples | One morning void and one bedtime void each day for 15 consecutive days; each sample analyzed separately | Gas chromatograph with a halogen specific detector and/or mass spectrometer | 3-PBA, 4-F-3-PBA, cis-DCCA, trans-DCCA, cis-DBCA |
| CPES (year) | Seattle, Washington | 2003–2004 | 23 | ||||||
| Morgan et al. [7] | CTEPP-OH | Ohio (six counties) | 2001 | 127 | 2–5 | Spot samples | Up to 6 spot urine samples (morning void, after lunch, and after dinner/before bedtime); samples pooled over a 48-h period | Gas chromatograph/mass spectrometer in the selected ion monitoring mode | 3-PBA |
| Naeher et al. [19] d | JAX-BIO | Jacksonville, Florida | 2001 | 203 | 4–6 | Spot sample | One convenience void | High performance liquid chromatograph-atmospheric pressure chemical ionization/ tandem mass spectrometer | 3-PBA, 4-F-3-PBA, cis-DCCA, trans-DCCA, cis- |
| Tulve et al. [17] d | JAX-EXP | Jacksonville, Florida | 2001 | 9 | 4–6 | Spot sample | One morning void | Gas chromatograph/mass spectrometer in the multiple ion monitoring mode | 3-PBA, 4F-3-PBA, cis-DCCA, trans-DCCA |
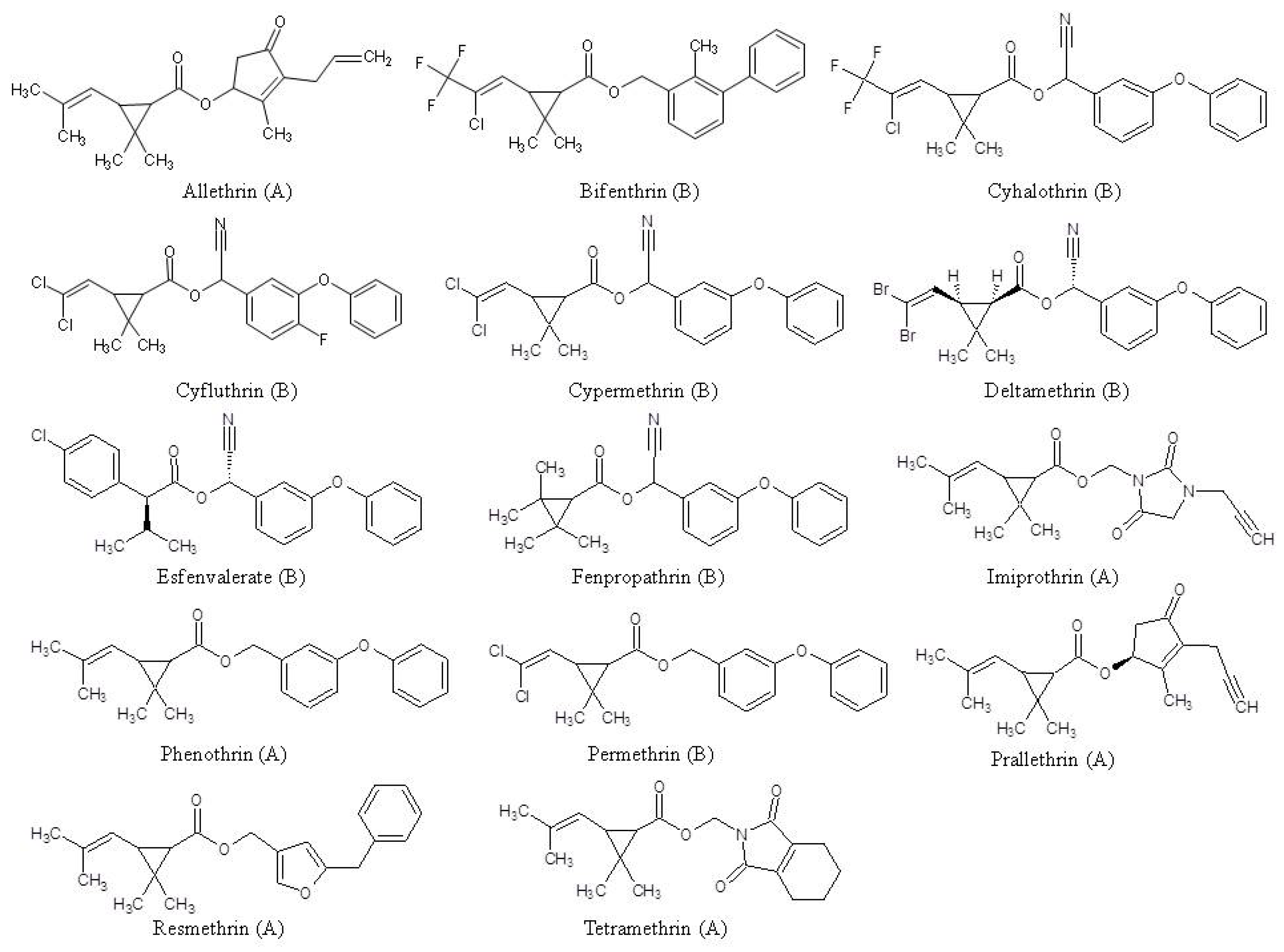
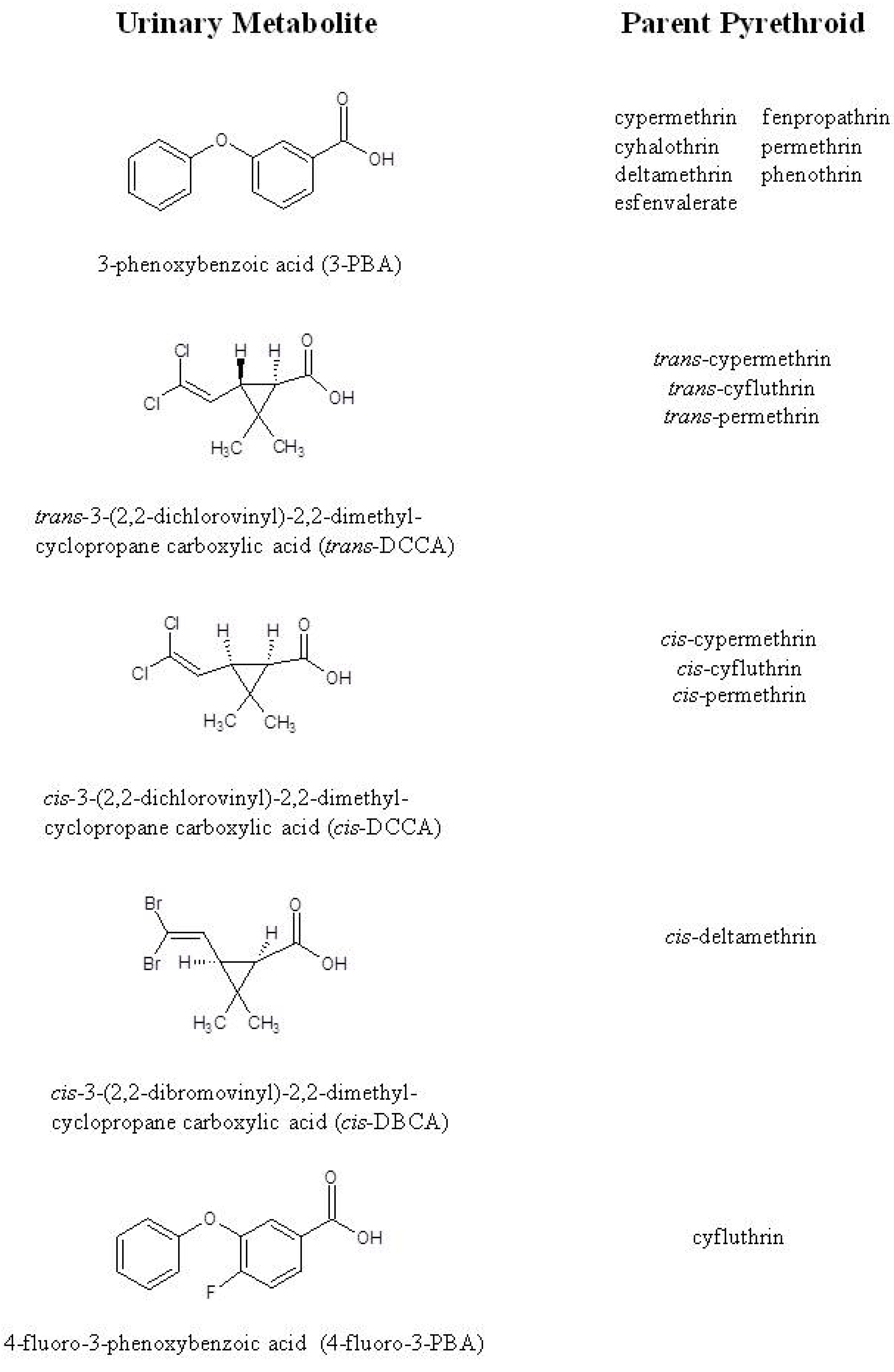
3. Results and Discussion
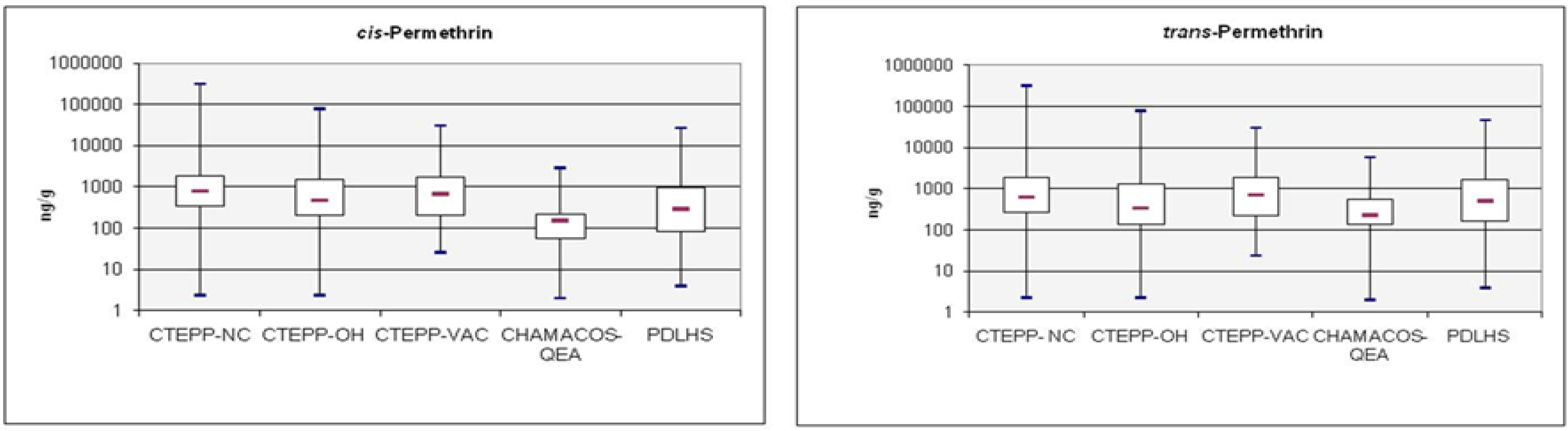
3.1. Levels of Pyrethroids in Environmental Media
3.2. Urinary Concentrations of Pyrethroid Metabolites
| Metabolite | Study a | Year | Age (years) | N b | LOD c(ng/mL) | % Detect | Mean ± SD | Minimum | 25th d | 50th | 75th | 95th | Maximum |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| cis-DCCA | NHANES | 1999–2000 | 6–11 | 468 | 0.1 | 47 | NR h | NR | NR | < i | 0.33 | 1.7 | NR |
| 2001–2002 | 6–11 | 580 | 0.1 | 26 | NR | NR | NR | < | 0.11 | 0.73 | NR | ||
| JAX-BIO e | 2001 | 4–6 | 201 | 0.2 | 87 | 2.2 ± 7.5 | NR | NR | 0.60 | 1.5 | 7.2 | 94.4 | |
| JAX-EXP e | 2001 | 4–6 | 9 | 0.2 | 100 | NR | NR | NR | 0.62 | 3.9 | NR | 43 | |
| CPES (summer) f | 2003 | 3–11 | 724 | 0.2 | 35 | 0.33 ± 1 | < | < | < | 0.33 | NR | 15 | |
| CPES (year) f | 2003–2004 | 3–11 | 706 | 0.2 | 44 | 0.5 ± 2.0 | < | < | < | 0.70 | 1.1 | 38.8 | |
| trans-DCCA | NHANES | 1999–2000 | 6–11 | 478 | 0.4 | 42 | NR | NR | NR | < | 0.97 | 4.2 | NR |
| 2001–2002 | 6–11 | 576 | 0.4 | 29 | NR | NR | NR | < | 0.47 | 2.5 | NR | ||
| JAX-BIO | 2001 | 4–6 | 201 | 0.1 | 93 | 3.6 ± 8.4 | NR | NR | 1.1 | 2.8 | 12.7 | 85.7 | |
| JAX-EXP | 2001 | 4–6 | 9 | 0.2 | 100 | NR | NR | NR | 1.1 | 6.9 | NR | 61 | |
| CPES (summer) | 2003 | 3–11 | 724 | 0.4 | 71 | 1.24 ± 2.6 | < | 0.14 | 0.38 | 0.99 | NR | 25 | |
| CPES (year) | 2003–2004 | 3–11 | 706 | 0.4 | 83 | 1.4 ± 4.1 | < | 0.3 | 1.0 | 1.5 | 3.7 | 81.6 | |
| 4-F-3-PBA g | JAX-BIO | 2001 | 4–6 | 201 | 0.1 | 70 | 0.2 ± 0.2 | NR | NR | 0.1 | 0.2 | 0.4 | 3.0 |
| JAX-EXP | 2001 | 4–6 | 9 | 0.1 | 56 | NR | NR | NR | 0.09 | 0.26 | NR | 1.7 | |
| CPES (summer) | 2003 | 3–11 | 724 | 0.2 | 2 | 0.02 ± 0.2 | < | < | < | < | NR | 3.5 | |
| CPES (year) | 2003–2004 | 3–11 | 706 | 0.2 | 19 | 0.2 ± 0.4 | < | < | < | < | 1.2 | 3.5 | |
| DBCA g | CPES (summer) | 2003 | 3–11 | 724 | 0.1 | 2 | 0.004 ± 0.02 | < | < | < | < | NR | 0.1 |
| CPES (year) | 2003–2004 | 3–11 | 706 | 0.1 | 6 | 0.007 ± 0.03 | < | < | < | < | 0.04 | 0.2 | |
| 3-PBA | NHANES | 1999–2000 | 6–11 | 483 | 0.1 | 72 | NR | NR | NR | 0.32 | 1.1 | 8.6 | NR |
| 2001–2002 | 6–11 | 580 | 0.1 | 75 | NR | NR | NR | 0.30 | 0.76 | 3.4 | NR | ||
| CTEPP-OH | 2001 | 2–5 | 127 | 0.2 | 67 | 0.9 ± 3.2 | < | 0.2 | 0.3 | 0.7 | 1.9 | 33.8 | |
| JAX-BIO | 2001 | 4–6 | 201 | 0.2 | 100 | 5.0 ± 10.2 | NR | NR | 1.9 | 4.2 | 20.6 | 89.3 | |
| JAX-EXP | 2001 | 4–6 | 9 | 0.5 | 100 | NR | NR | NR | 2.2 | 29 | NR | 99 | |
| CPES (summer) | 2003 | 3–11 | 724 | 0.1 | 82 | 1.22 ± 2.4 | < | 0.22 | 0.45 | 0.97 | NR | 25 | |
| CPES (year) | 2003–2004 | 3–11 | 706 | 0.1 | 94 | 1.5 ± 3.1 | < | 0.5 | 1.2 | 2.6 | 4.1 | 51.4 |
3.3. Contributions of Pyrethroid Exposure by Route
3.4. Limitations
4. Conclusions
- • Levels of pyrethroids and their environmentally-occurring metabolites in individual food items for several consecutive days or a week including seasonally as certain types of foods (i.e., fruits and vegetables) likely contribute to the intermittent dietary exposures of children to current-use pyrethroids,
- • Spatiotemporal variability of pyrethroids and their environmentally-degraded metabolites in media (i.e., dust and on surfaces) at residences, since pyrethroid levels can vary substantially within and between rooms, particularly before and after pesticide use,
- • Temporal variability of urinary pesticide metabolites over a day and for several consecutive days as there can be substantial intra-individual and inter-individual variability of pyrethroid metabolites in children’s urine samples even over a short period of time (e.g., 48-h period). The volume of the urine void and time of the current and previous urine void should be recorded as this information can be used to calculate the metabolite excretion rate (ng/h) for a pyrethroid which is independent of urine volume,
- • Toxicokinetics of environmentally-degraded metabolites in humans (e.g., in vitro assays) to determine whether these metabolites are unchanged or are further metabolized to other products in the body before urinary elimination.
- • Accurate and more descriptive pesticide product use information including active ingredients as these products can contain single or multiple pyrethroids (including one or more isomers per pesticide), and
- • Accurate and more descriptive daily activity patterns of children as their exposures to pesticides are highly dependent on their day-to-day activities such as their individual eating patterns, activity levels, locations they spend their time, hand-to-mouth activity, and object-to-mouth activity.
Disclaimer
Conflict of Interest
References
- U.S. Environmental Protection Agency (EPA). Agrichemicals Food Quality Protection Act—Children and Consumers. Available online: http://www.epa.gov/agriculture/factsheets/epa-305-f-00-005ag.html (accessed on 15 November 2011).
- Weston, D.P.; You, J.; Lydy, M.J. Distribution and toxicity of sediment-associated pesticides in agricultural-dominated water bodies of California’s Central Valley. Environ. Sci. Technol. 2004, 38, 2752–2759. [Google Scholar] [CrossRef]
- Gan, J.; Spurlock, F.; Hendley, P.; Weston, D. Chapter 1: Synthetic Pyrethroid Use Patterns, Properties, and Environmental Effects. In Synthetic Pyrethroids, Occurrence and Behavior in Aquatic Environments; American Chemical Society: Washington, DC, USA, 2008; Volume 991, pp. 3–25. [Google Scholar]
- Kaneko, H. Pyrethroids: Mammalian metabolism and toxicity. J. Agric. Food Chem. 2011, 59, 2786–2791. [Google Scholar] [CrossRef]
- Lu, C.; Barr, D.B.; Pearson, M.; Bartell, S.; Bravo, R. A longitudinal approach to assessing urban and suburban children’s exposure to pyrethroid pesticides. Environ. Health Perspect. 2006, 114, 1419–1423. [Google Scholar] [CrossRef]
- Bradman, A.; Whitaker, D.; Quiros, L.; Castorina, R.; Henn, B.C.; Nishiko, M.; Morgan, J.; Barr, D.B.; Harnley, M.; Brisbin, J.A.; et al. Pesticides and their metabolites in the homes and urine of farmworker children living in the Salinas Valley, CA. J. Expo. Sci. Environ. Epidemiol. 2007, 17, 331–349. [Google Scholar] [CrossRef]
- Morgan, M.K.; Sheldon, L.S.; Croghan, C.W.; Jones, P.A.; Chuang, J.C.; Wilson, N.K. An observational study of 127 preschool children at their homes and daycare centers in Ohio: Environmental pathways to cis- and trans-permethrin exposure. Environ. Res. 2007, 104, 266–274. [Google Scholar] [CrossRef]
- Barr, D.B.; Olsson, A.O.; Wong, L.Y.; Udunka, S.; Baker, S.E.; Whitehead, R.D.; Magsumboi, M.S.; Williams, B.L.; Needham, L.L. Urinary concentrations of metabolites of pyrethroid insecticides in the general U.S. population: National health and nutrition examination survey 1999–2002. Environ. Health Perspect. 2010, 118, 742–748. [Google Scholar]
- Lu, C.; Schenck, F.J.; Pearson, M.A.; Wong, J.W. Assessing children’s dietary pesticide exposure: Direct measurement of pesticide residues in 24-hr duplicate food samples. Environ. Health Perspect. 2010, 118, 1625–1630. [Google Scholar] [CrossRef]
- Leng, G.; Kuhn, K.H.; Idel, H. Biological monitoring of pyrethroids in blood and pyrethroid metabolites in urine: Applications and limitations. Sci. Total Environ. 1997, 199, 173–181. [Google Scholar] [CrossRef]
- Leng, G.; Ranft., U.; Sugiri, D.; Hadnagy, W.; Berger-Preib, E.; Idel, H. Pyrethroids used indoors—Biological monitoring of exposure to pyrethroids following an indoor pest control operation. Int. J. Hyg. Environ. Health 2003, 206, 85–92. [Google Scholar] [CrossRef]
- Morgan, M.K.; Sheldon, L.S.; Croghan, C.W.; Chuang, J.C.; Lordo, R.; Wilson, N.K.; Lyu, C.; Brinkman, M.; Morse, N.; Chou, Y.; et al. A Pilot Study of Children’a Total Exposure to Persistent Pesticides and Other Persistent Organic Pollutants (CTEPP); EPA/600/R-041/193; US Environmental Protection Agency: Durham, NC, USA, 2004. [Google Scholar]
- Quandt, S.A.; Arcury, T.A.; Rao, P.; Snively, B.M.; Camann, D.E.; Doran, A.M.; Yau, A.Y.; Hoppin, J.A.; Jackson, D.S. Agricultural and residential pesticides in wipe samples from farm worker family residences in North Carolina and Virginia. Environ. Health Perspect. 2004, 118, 1625–1630. [Google Scholar]
- Bouvier, G., Seta; Vigouroux-Villard, A.; Blanchard, O.; Momas, I. Insecticide urinary metabolites in non-occupationally exposed populations. J. Toxicol. Environ. Health B 2005, 8, 485–512. [Google Scholar]
- Julien, R.; Adamkiewicz, G.; Levy, J.; Bennett, D.B.; Nishioka, M.; Spengler, J.D. Pesticide loadings of select organophosphate and pyrethroid pesticides in urban public housing. J. Expo. Sci. Environ. Epidemiol. 2008, 18, 167–174. [Google Scholar] [CrossRef]
- Starr, J.; Graham, S.; Stout, D.; Andrews, K.; Nishioka, M. Pyrethroid pesticides and their metabolites in vacuum cleaner dust collected from homes and day-care centers. Environ. Res. 2008, 108, 271–279. [Google Scholar] [CrossRef]
- 17. Tulve, N.S., Egeghy; Fortmann, R.C.; Whitaker, D.A.; Nishioka, M.G.; Naeher, L.P.; Hilliard, A. Multimedia measurements and activity patterns in an observational pilot study of nine young children. J. Expo. Sci. Environ. Epidemiol. 2008, 18, 31–44. [Google Scholar] [CrossRef]
- Lu, C.; Barr, D.B.; Pearson, M.; Walker, L.A.; Bravo, R. The attribution of urban and suburban children’s exposure to synthetic pyrethroid insecticides: A longitudinal assessment. J. Expo. Sci. Environ. Epidemiol. 2009, 19, 69–78. [Google Scholar] [CrossRef]
- Naeher, L.P.; Tulve, N.S.; Egeghy, P.P.; Barr, D.B.; Adetona, O.; Fortmann, R.C.; Needham, L.L.; Bozeman, E.; Hilliard, A.; Sheldon, L.S. Organophosphorus and pyrethroid insecticide urinary metabolite concentrations in young children living in a southeastern United States city. Sci. Total Environ. 2010, 408, 1145–1153. [Google Scholar]
- Chuang, J.C.; Wilson, N.K. Multiresidue analysis of organophosphate and pyrethroid pesticides in duplicate-diet solid food by pressurized liquid extraction. J. Environ. Sci. Health B 2011, 46, 41–50. [Google Scholar] [CrossRef]
- Quiros-Alcala, L.; Bradman, A.; Nishioka, M.; Harnly, M.; Hubbard, A.; McKone, T.; Ferber, J.; Eskenazi, B. Pesticides in house dust from urban and farmworker households in California: An observational measurement study. Environ. Health 2011, 10, 3–15. [Google Scholar] [CrossRef] [Green Version]
- O’Rourke, M.K.; Lizardi, P.S.; Rogan, S.P.; Freeman, N.C.; Aguirre, A.; Saint, C.G. Pesticide exposure and creatinine variation among young children. J. Expo. Anal. Environ. Epidemiol. 2000, 10, 672–681. [Google Scholar] [CrossRef]
- Barr, D.B.; Wilder, L.C.; Caudill, S.P.; Gonzalez, A.J.; Needham, L.L.; Pirkle, J.L. Urinary creatinine concentrations in the U.S. population: Implications for urinary biologic monitoring measurements. Environ. Health Perspect. 2005, 113, 192–200. [Google Scholar]
- Fortin, M.C.; Bouchard, M.; Carrier, G.; Dumas, P. Biological monitoring of exposure to pyrethrins and pyrethroids in a metropolitan population of the Province of Quebec, Canada. Environ. Res. 2008, 107, 343–350. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Fourth National Report on Human Exposure to Environmental Chemicals. Available online: http://www.cdc.gov/exposurereport/ (accessed on 2 November 2011).
- Standard Practice for Collection of Floor Dust for Chemical Analysis D5438-94. In Annual Book of ASTM Standards; American Society of Testing Materials: West Conshohoken, PA, USA, 1994.
- Mercier, F.; Glorennec, P.; Thomas, O.; Le Bot, B. Organic contamination of settled house dust, a review for exposure assessment purposes. Environ. Sci. Technol. 2011, 45, 6716–6727. [Google Scholar]
- Egeghy, P.P.; Sheldon, L.S.; Fortmann, R.C.; Stout, D.M.; Tulve, N.S.; Cohen Hubal, E.; Melnyk, L.J.; Morgan, M.K.; Jones, P.A.; Whitaker, D.; et al. Important Exposure Factors for Children: An Analysis of Laboratory and Observational Field Data Characterizing Cumulative Exposure to Pesticides; EPA 600/R-07/013; Environmental Protection Agency: Washington, DC, USA, 2007. [Google Scholar]
- Riederer, A.M.; Hunter, R.E.; Hayden, S.W.; Ryan, P.B. Pyrethroid and organophosphorus pesticides in composite diet samples from Atlanta, USA adults. Environ. Sci. Technol. 2010, 44, 483–490. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA). Total Diet Study Analytical Results. Available online: http://www.fda.gov/Food/FoodSafety/FoodContaminantsAdulteration/TotalDietStudy/ucm184293.htm (accessed on 2 November 2011).
- Soderland, D.M.; Clark, J.M.; Sheets, L.P.; Mullin, L.S.; Piccirillo, V.J.; Sargent, D.; Stevens, J.T.; Weiner, M.L. Mechanisms of pyrethroid neurotoxicity: Implications for cumulative risk assessment. Toxicology 2002, 171, 3–59. [Google Scholar] [CrossRef]
- Shafer, T.J.; Meyer, D.A.; Crofton, K.M. Developmental neurotoxicity of pyrethroid insecticides: Critical review and future research needs. Environ. Health Perspect. 2005, 113, 123–136. [Google Scholar]
- Couture, C.; Fortin, M.C.; Carrier, G.; Dumas, P.; Tremblay, C.; Bouchard, M. Assessment of exposure to pyrethroids and pyrethrins in a rural population of the Monteregie Area, Quebec, Canada. J. Occup. Environ. Hyg. 2009, 6, 341–352. [Google Scholar] [CrossRef]
- Liu, W.; Gan, J.J. Determination of enantiomers of synthetic pyrethroids in water by solid phase microextraction—Enantioselective gas chromatography. J. Agric. Food Chem. 2004, 52, 736–741. [Google Scholar] [CrossRef]
- Allan, I.J.; House, W.A., Parker; Carter, J.E. Diffusion of the synthetic pyrethroid permethrin into bed-sediments. Environ. Sci. Technol. 2005, 39, 523–530. [Google Scholar] [CrossRef]
- Lu, C.; Bravo, R.; Caltabiano, L.M.; Irish, R.M.; Weerasekera, G.; Barr, D.B. The presence of dialkylphosphates in fresh fruitjuices: Implication for organophosphorus pesticide exposure and risk assessments. J. Toxicol. Environ. Health A 2005, 68, 209–227. [Google Scholar] [CrossRef]
- Morgan, M.K.; Sheldon, L.S.; Croghan, C.W.; Jones, P.A.; Robertson, G.; Chuang, J.C.; Wilson, N.K.; Lyu, C. Exposures of preschool children to chlorpyrifos and its degradation product 3,5,6-trichloro-2-pyridinol in their everyday environments. J. Expo. Sci. Environ. Epidemiol. 2005, 15, 297–309. [Google Scholar]
- Wilson, N.K.; Chuang, J.C.; Hand, K.M.; Brinkman, M.; Tefft, M.E.; Finegold, J.K.; Aysucue, L.C.; Humphreys, N.L.; Chapman, M.M.; Wilson, L.K.; et al. Organophosphate and Pyrethroid Pesticides in Environmental and Children’s Personal Samples Collected at 50 Homes of Infants and Toddlers. In Proceedings of the 2004 Annual Meeting of the International Society of Exposure Analysis, Philadelphia, PA, USA, 17–21 October 2004.
- Eadsforth, C.V.; Baldwin, M.K. Human dose-excretion studies with the pyrethroid insecticide, cypermethrin. Xenobiotica 1983, 13, 67–72. [Google Scholar] [CrossRef]
- Eadsforth, C.V.; Bragt, P.C.; van Sittert, N.J. Human dose-excretion studies with pyrethroid insecticide cypermethrin and alphacypermethrin: Relevance for biological monitoring. Xenobiotica 1988, 18, 603–614. [Google Scholar] [CrossRef]
- Egeghy, P.P.; Cohen Hubal, E.A.; Tulve, N.S.; Melnyk, L.J.; Morgan, M.K.; Fortmann, R.C.; Sheldon, L.S. Review of pesticide urinary biomarker measurements from selected children’s obsevational exposure studies. Int. J. Environ. Public Health 2011, 8, 1727–1754. [Google Scholar] [CrossRef]
- Riederer, A.M.; Bartell, S.M.; Barr, D.B.; Ryan, P.B. Diet and nondiet predictors of urinary 3-phenoxybenzoic acid in NHANES 1999–2002. Environ. Health Perspect. 2008, 116, 1015–1022. [Google Scholar] [CrossRef]
- Integrated Risk Information System. Available online: http://www.epa.gov/IRIS (accessed on 15 November 2011).
- Huckle, K.R.; Hutson, D.H.; Millburn, P. Species differences in the metabolism of 3-phenoxybenzoic acid. Drug Metab. Dispos. 1981, 9, 352–359. [Google Scholar]
- Tulve, N.S.; Egeghy, P.P.; Fortmann, R.C.; Xue, J.; Evans, J.; Whitaker, D.A.; Croghan, C.W. Methodologies for estimating cumulative human exposures to current-use pyrethroid pesticides. J. Expo. Sci. Environ. Epidemiol. 2011, 21, 317–327. [Google Scholar] [CrossRef]
- Kuivila, K.M.; Hladik, M.L.; Ingersoll, C.G.; Kemble, N.E.; Moran, P.W.; Calhoun, D.L.; Nowell, L.H.; Gilliiom, R.J. Occurrence and potential sources of pyrethroid insecticides in stream sediments from seven U.S. metropolitan areas. Environ. Sci. Technol. 2012, 46, 4297–4303. [Google Scholar]
Supplementary Files
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Morgan, M.K. Children’s Exposures to Pyrethroid Insecticides at Home: A Review of Data Collected in Published Exposure Measurement Studies Conducted in the United States. Int. J. Environ. Res. Public Health 2012, 9, 2964-2985. https://doi.org/10.3390/ijerph9082964
Morgan MK. Children’s Exposures to Pyrethroid Insecticides at Home: A Review of Data Collected in Published Exposure Measurement Studies Conducted in the United States. International Journal of Environmental Research and Public Health. 2012; 9(8):2964-2985. https://doi.org/10.3390/ijerph9082964
Chicago/Turabian StyleMorgan, Marsha K. 2012. "Children’s Exposures to Pyrethroid Insecticides at Home: A Review of Data Collected in Published Exposure Measurement Studies Conducted in the United States" International Journal of Environmental Research and Public Health 9, no. 8: 2964-2985. https://doi.org/10.3390/ijerph9082964
APA StyleMorgan, M. K. (2012). Children’s Exposures to Pyrethroid Insecticides at Home: A Review of Data Collected in Published Exposure Measurement Studies Conducted in the United States. International Journal of Environmental Research and Public Health, 9(8), 2964-2985. https://doi.org/10.3390/ijerph9082964




