Seasonal Variation of Water Quality and Phytoplankton Response Patterns in Daya Bay, China
Abstract
:1. Introduction
2. Materials and Methods
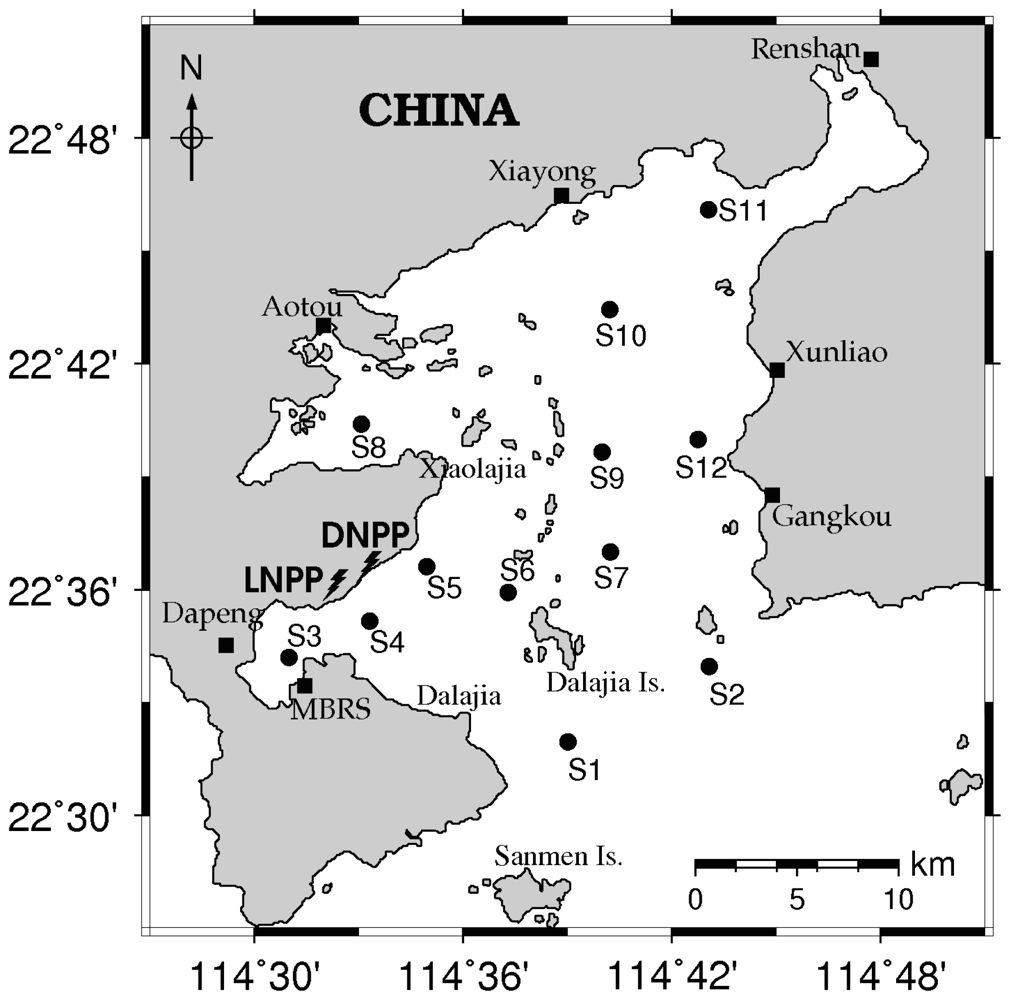
2.1. Sampling Design and Analysis of Samples
2.2. Data Analysis
3. Results
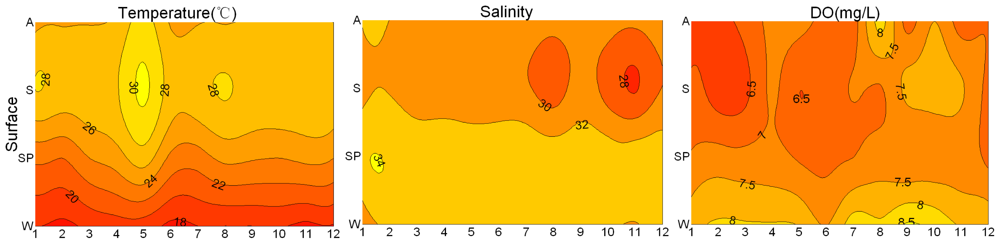
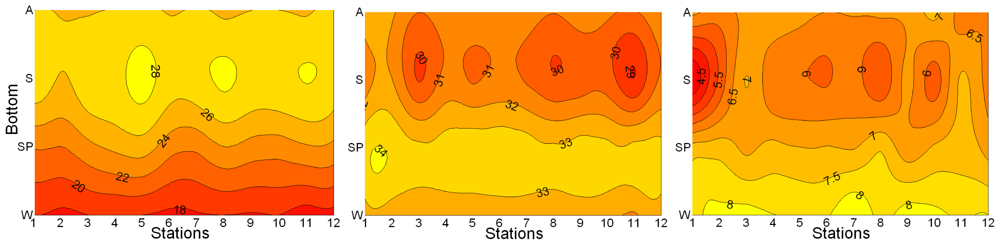
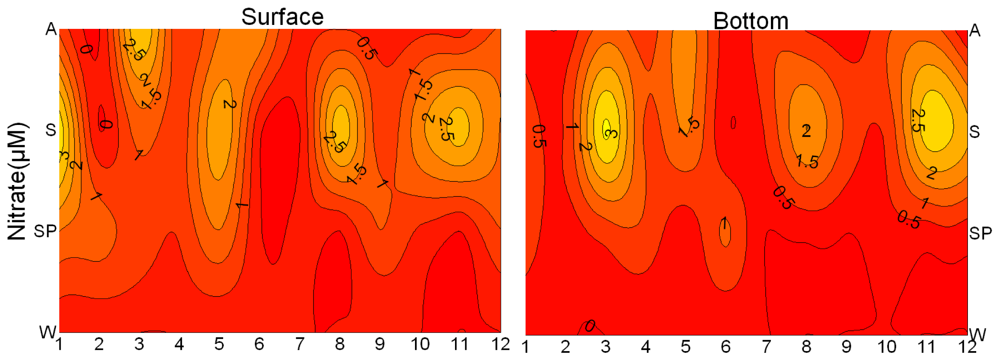
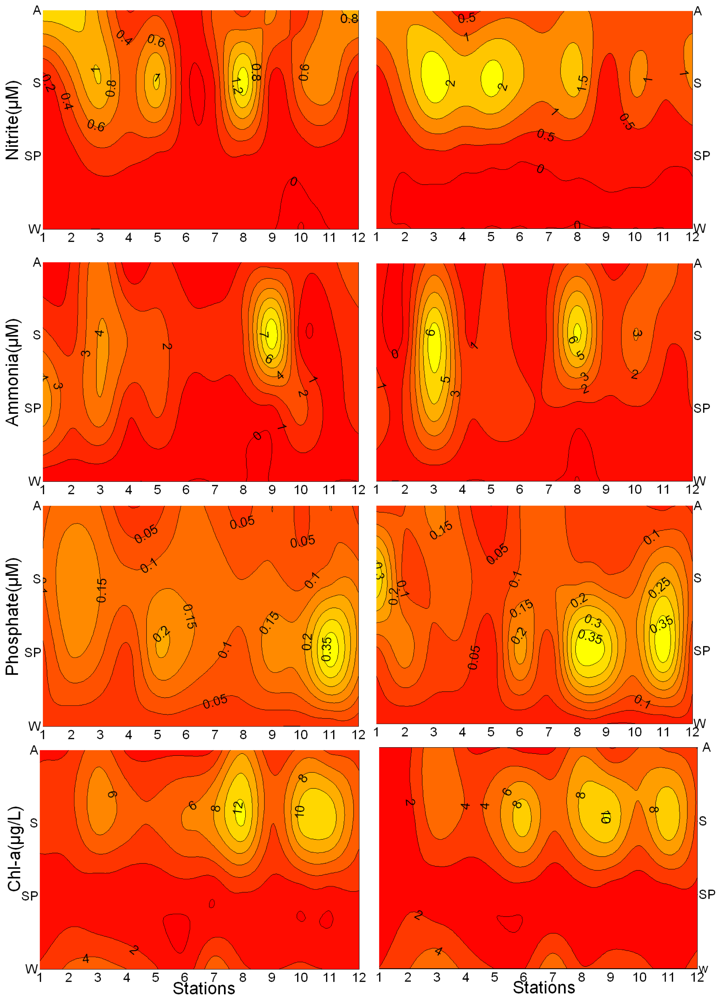
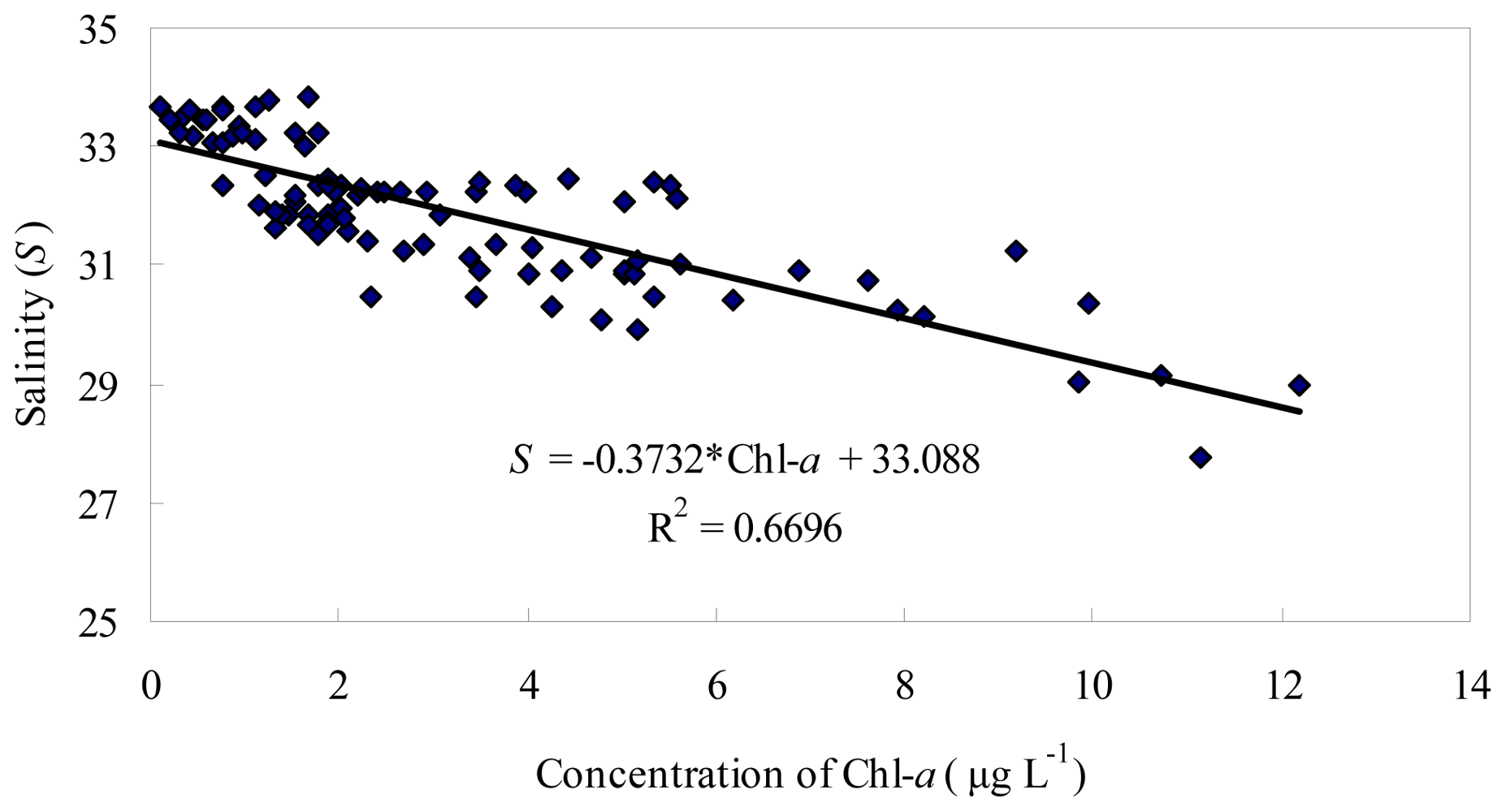
3.1. Seasonal and Spatial Variation of Environmental Parameters
3.2. Principal Component Analysis
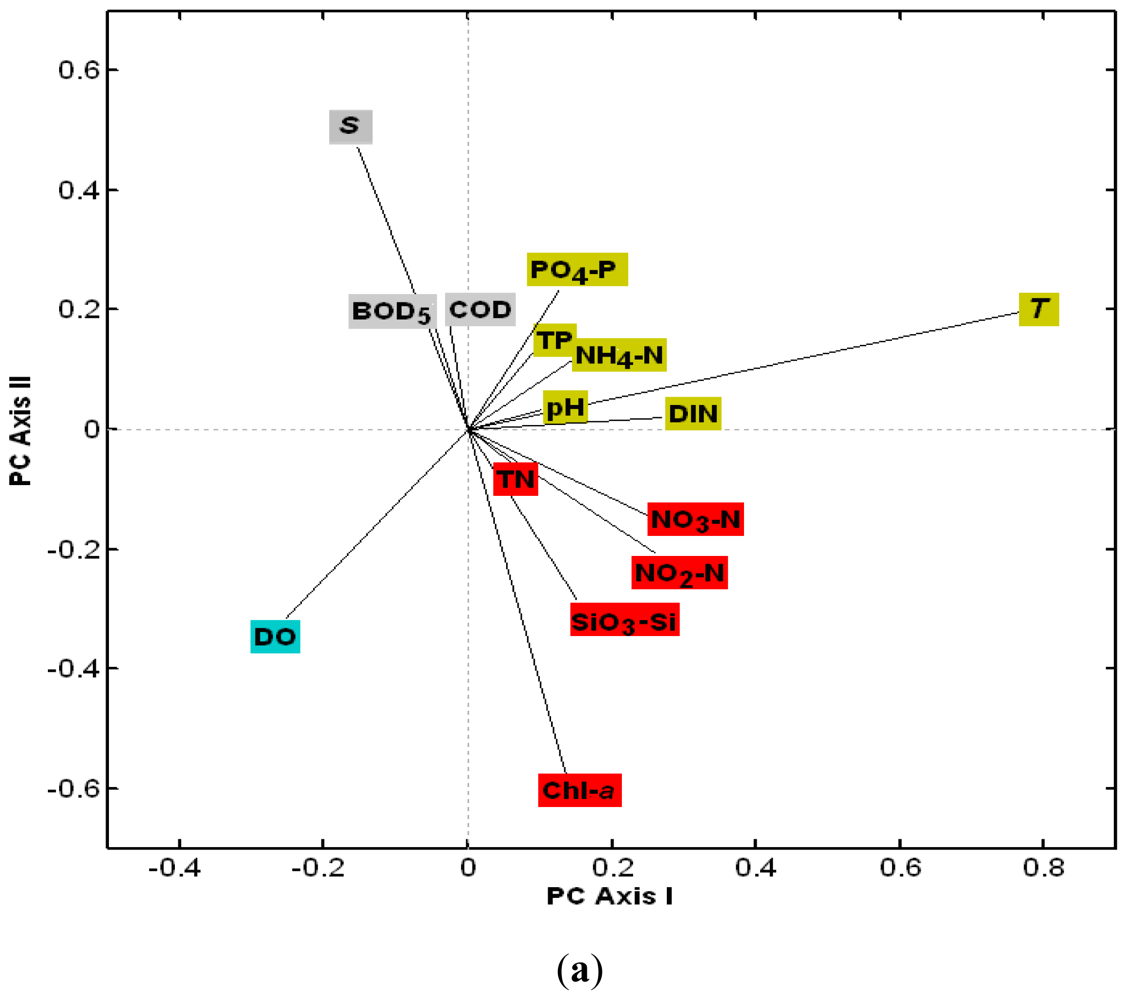
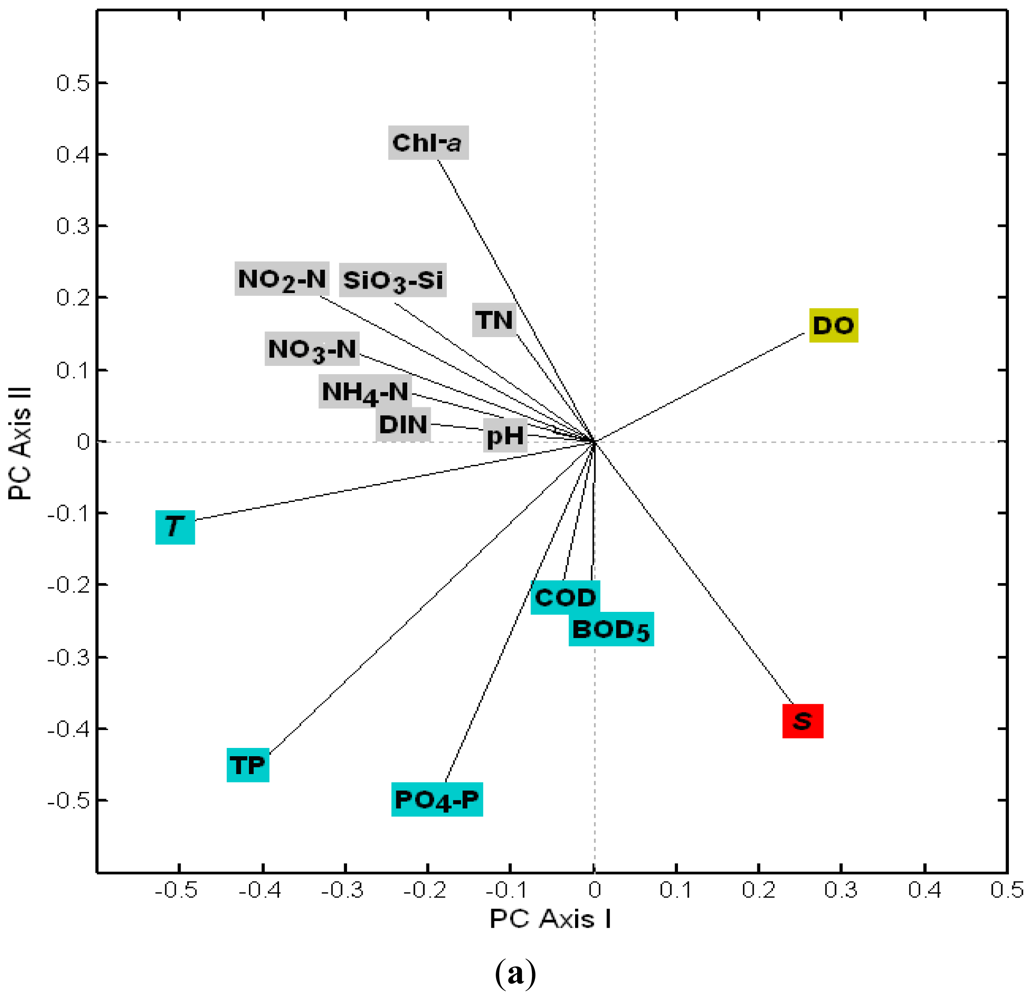
3.2.1. The Loadings of Water Quality Parameters on the First Four PCs
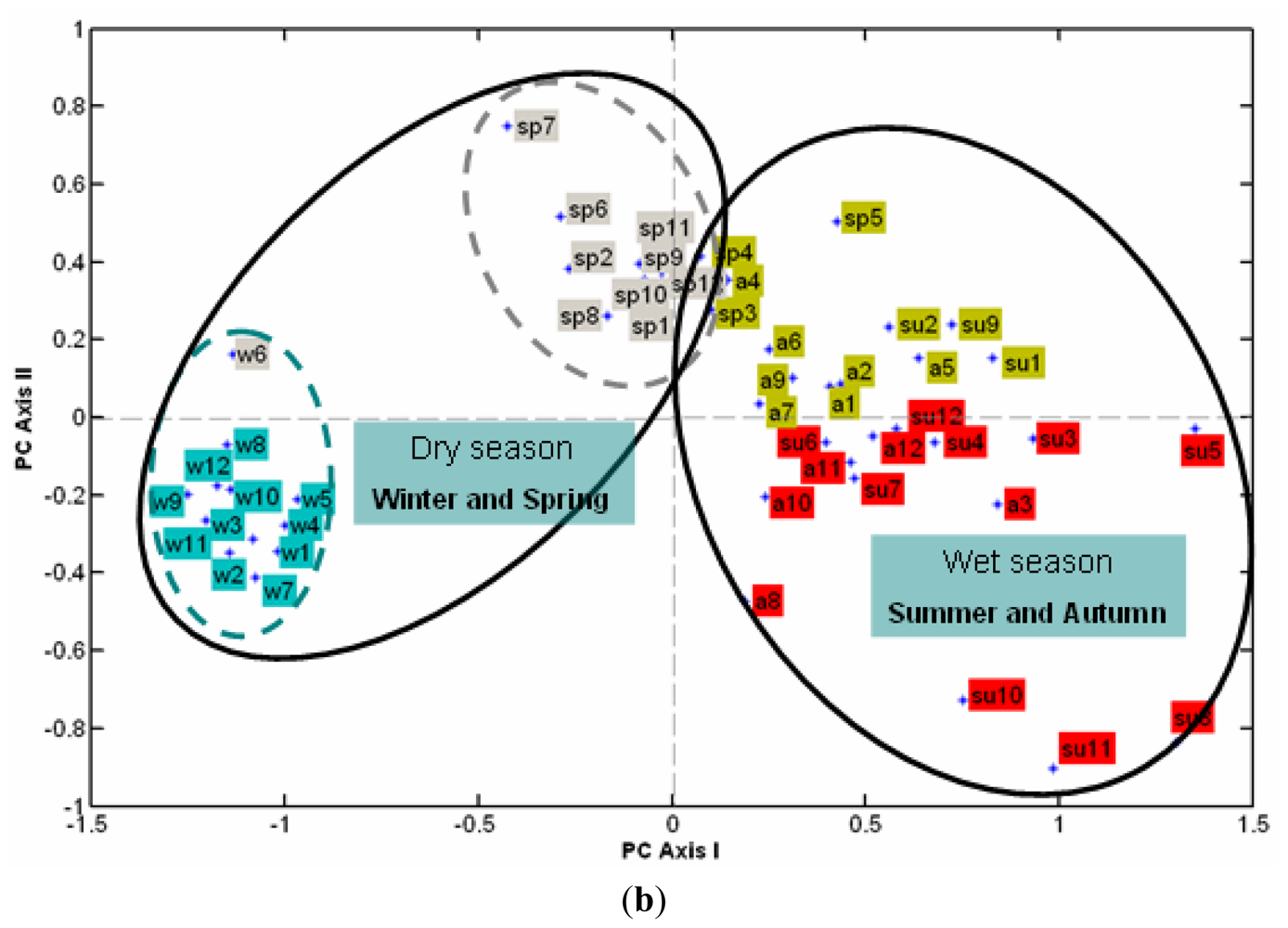
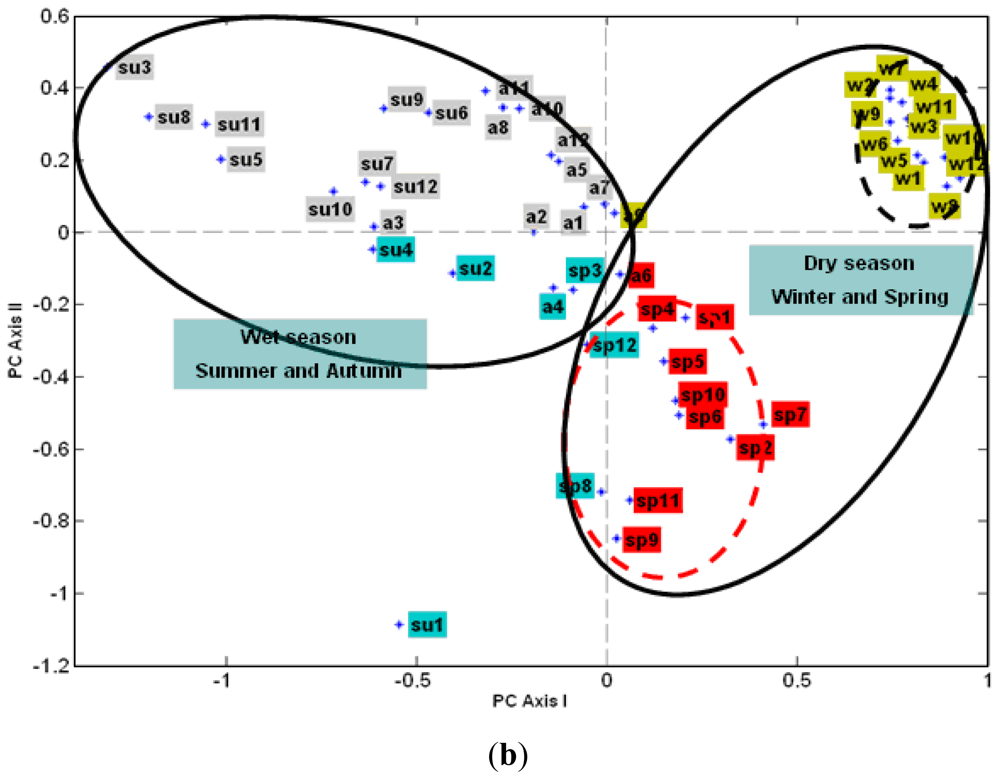
3.2.2. The Effect of Station Score on the First Two PCs
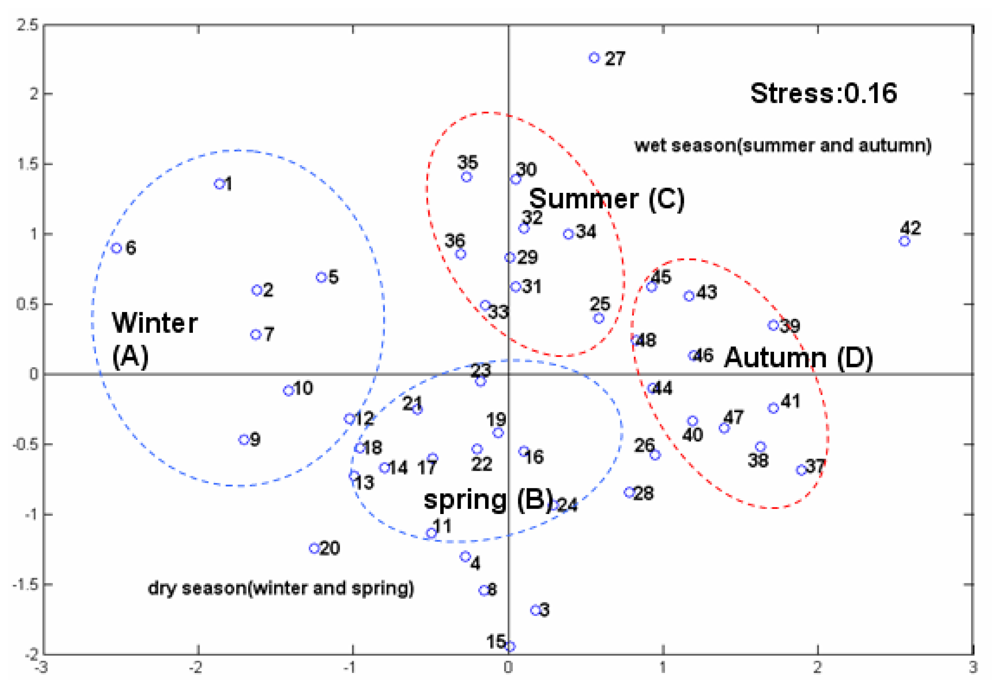
3.3. Phytoplankton Community Structure
4. Discussion
5. Conclusions
Acknowledgements
References
- Wu, ML; Wang, YS. Using chemometrics to evaluate anthropogenic effects in Daya Bay, China. Estuarine Coastal Shelf Sci 2007, 72, 732–742. [Google Scholar]
- Jordan, TE; Correll, DL; Weller, DE. Effects of agriculture on discharges of nutrients from Coastal Plain watersheds of Chesapeake Bay. J Environ Q 1997, 26, 836–848. [Google Scholar]
- Tsujimoto, A; Nomura, R; Yasuhara, M; Yamazaki, H; Yoshikawa, S. Impact of eutrophication on shallow marine benthic foraminifers over the last 150 years in Osaka Bay, Japan. Mar. Micropaleontol 2006, 60, 258–268. [Google Scholar]
- MÖLlmann, C; Diekmann, R; MÜLler-Karulis, B; Kornilovs, G; Plikshs, M; Axe, P. Reorganization of a large marine ecosystem due to atmospheric and anthropogenic pressure: A discontinuous regime shift in the Central Baltic Sea. Glob. Change Biol 2009, 15, 1377–1393. [Google Scholar]
- Lapointe, BE; Clark, MW. Nutrient inputs from the watershed and coastal eutrophication in the Florida Keys. Estuaries 1992, 15, 465–476. [Google Scholar]
- Howarth, R; Sharpley, A; Walker, D. Sources of nutrient pollution to coastal waters in the United States: Implications for achieving coastal water quality goals. Estuaries Coasts 2002, 25, 656–676. [Google Scholar]
- Moncheva, S; Gotsis-Skretas, O; Pagou, K; Krastev, A. Phytoplankton blooms in black sea and mediterranean coastal ecosystems subjected to anthropogenic eutrophication: Similarities and differences. Estuarine Coastal Shelf Sci 2001, 53, 281–295. [Google Scholar]
- Cloern, JE. Our evolving conceptual model of the coastal eutrophication problem. Mar. Ecol.-Prog. Ser 2001, 210, 223–253. [Google Scholar]
- Abdullah, MI; Danielsen, M. Chemical-criteria for marine eutrophication with special reference to the Oslofjord, Norway. Hydrobiologia 1992, 235, 711–722. [Google Scholar]
- Aguilera, PA; Castro, H; Rescia, A; Schmitz, MF. Methodological development of an index of coastal water quality: Application in a tourist area. Environ. Manag 2001, 27, 295–301. [Google Scholar]
- Dettmann, EH. Effect of water residence time on annual export and denitrification of nitrogen in estuaries: A model analysis. Estuaries 2001, 24, 481–490. [Google Scholar]
- Ronnberg, C; Bonsdorff, E. Baltic Sea eutrophication: Area-specific ecological consequences. Hydrobiologia 2004, 514, 227–241. [Google Scholar]
- Sagert, S; Rieling, T; Eggert, A; Schubert, H. Development of a phytoplankton indicator system for the ecological assessment of brackish coastal waters (German Baltic Sea coast). Hydrobiologia 2008, 611, 91–103. [Google Scholar]
- Dziock, F; Henle, K; Foeckler, F; Follner, K; Scholz, M. Biological indicator systems in floodplains—A review. Int. Rev. Hydrobiol 2006, 91, 271–291. [Google Scholar]
- Testa, J; Kemp, W; Boynton, W; Hagy, J. Long-term changes in water quality and productivity in the Patuxent River Estuary: 1985 to 2003. Estuaries Coasts 2008, 31, 1021–1037. [Google Scholar]
- Cabecinha, E; Cortes, R; Cabral, J; Ferreira, T; Lourenco, M; Pardal, M. Multi-scale approach using phytoplankton as a first step towards the definition of the ecological status of reservoirs. Ecol. Indic 2009, 9, 240–255. [Google Scholar]
- Wang, YS; Lou, ZP; Sun, CS; Wu, ML; Han, SH. Multivariate statistical analysis of water quality and phytoplankton characteristics in Daya Bay, China, from 1999 to 2002. Oceanologia 2006, 48, 193–211. [Google Scholar]
- Aßmus, J; Melle, W; Tjøstheim, D; Edwards, M. Seasonal cycles and long-term trends of plankton in shelf and oceanic habitats of the Norwegian Sea in relation to environmental variables. Deep Sea Res. Part II 2009, 56, 1895–1909. [Google Scholar]
- Claquin, P; Longphuirt, SN; Fouillaron, P; Huonnic, P; Ragueneau, O; Klein, C; Leynaert, A. Effects of simulated benthic fluxes on phytoplankton dynamic and photosynthetic parameters in a mesocosm experiment (Bay of Brest, France). Estuarine Coastal Shelf Sci 2010, 86, 93–101. [Google Scholar]
- Clarke, KR. Non-parametric multivariate analysis of changes in community structure. Aust. J. Ecol 1993, 18, 117–143. [Google Scholar]
- McCollin, T; Shanks, AM; Dunn, J. Changes in zooplankton abundance and diversity after ballast water exchange in regional seas. Mar. Pollut. Bull 2008, 56, 834–844. [Google Scholar]
- Tew, K; Chou, WR; Chuang, SF; He, CC; Liao, JH; Fang, LS. Comparison of the decapod communities in an anthropogenic impacted lagoon and its adjacent coastal waters. Hydrobiologia 2008, 607, 1–10. [Google Scholar]
- Sun, CC; Wang, YS; Sun, S; Zhang, FZ. Dynamic analysis of phytoplankton community characteristics in Daya Bay, China. Acta Ecol. Sinica 2006, 26, 3948–3958. [Google Scholar]
- Wang, YS; Lou, ZP; Sun, CC; Sun, S. Ecological environment changes in Daya Bay, China, from 1982 to 2004. Mar. Pollut. Bull 2008, 56, 1871–1879. [Google Scholar]
- Song, X; Huang, L; Zhang, J; Huang, H; Li, T; Su, Q. Harmful algal blooms (HABs) in Daya Bay, China: An in situ study of primary production and environmental impacts. Mar. Pollut. Bull 2009, 58, 1310–1318. [Google Scholar]
- Xu, GZ. Environments and Resources of Daya Bay; Hefei Science Press: Hefei, China, 1998. [Google Scholar]
- Song, X; Huang, L; Zhang, J; Huang, X; Zhang, J; Yin, J; Tan, Y; Liu, S. Variation of phytoplankton biomass and primary production in Daya Bay during spring and summer. Mar. Pollut. Bull 2004, 49, 1036–1044. [Google Scholar]
- Tang, DL; Kester, DR; Wang, ZD; Lian, JS; Hiroshi, K. AVHRR satellite remote sensing and shipboard measurements of the thermal plume from the Daya Bay, nuclear power station, China. Remote Sens. Environ 2003, 84, 506–515. [Google Scholar]
- Liu, S; Huang, LM; Huang, H; Liang, JS; Min, LA; Tao, L. Ecological response of phytoplankton to the operation of Daya Bay nuclear power station. Mar. Environ. Sci 2006, 25, 9–13. [Google Scholar]
- San Diego-McGlone, ML; Villanoy, CL; Aliño, PM. Nutrient mediated stress on the marine communities of a coastal lagoon (Puerto Galera, Philippines). Mar. Pollut. Bull 1995, 31, 355–366. [Google Scholar]
- Malone, T; Conley, D; Fisher, T; Glibert, P; Harding, L; Sellner, K. Scales of nutrient-limited phytoplankton productivity in Chesapeake Bay. Estuaries Coasts 1996, 19, 371–385. [Google Scholar]
- Hidalgo-GonzÁLez, RM; Alvarez-Borrego, S. Chlorophyll profiles and the water column structure in the Gulf of California. Oceanol. Acta 2001, 24, 19–28. [Google Scholar]
- Taylor, MH; Tam, J; Blaskovic, V; Espinoza, P; Ballon, RM; Wosnitza-Mendo, C; Arguelles, J; Diaz, E; Purca, S; Ochoa, N; et al. Trophic modeling of the Northern Humboldt Current Ecosystem, Part II: Elucidating ecosystem dynamics from 1995 to 2004 with a focus on the impact of ENSO. Prog. Oceanogr 2008, 79, 366–378. [Google Scholar] [Green Version]
- Riegman, R; Kuipers, BR; Noordeloos, AAM; Witte, HJ. Size-differential control of phytoplankton and the structure of plankton communities. Neth. J. Sea Res 1993, 31, 255–265. [Google Scholar]
- Wei, GF; Wang, ZD; Lian, JS. Succession of dominant phytoplank–ton species in spring 2002 at Dapeng’Ao Cove, Daya Bay. Acta Ecol. Sinica 2003, 23, 2285–2292. [Google Scholar]
| Parameter | Surface | Bottom | ||||||
|---|---|---|---|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC4 | PC1 | PC2 | PC3 | PC4 | |
| T | 0.7754 | −0.1987 | 0.0125 | 0.3023 | 0.502 | −0.1127 | 0.2514 | −0.0756 |
| pH | 0.102 | −0.0329 | 0.1233 | 0.1385 | 0.0497 | 0.0132 | 0.0119 | −0.0163 |
| S | −0.1539 | −0.4744 | −0.1255 | −0.1125 | −0.2428 | −0.3668 | 0.0177 | −0.3448 |
| DO | −0.2535 | 0.316 | 0.1568 | −0.1276 | −0.2547 | 0.1529 | −0.2819 | −0.0906 |
| BOD5 | −0.0469 | −0.1745 | 0.6619 | 0.0597 | 0.0028 | −0.2294 | 0.0098 | 0.4245 |
| COD | −0.0257 | −0.1801 | 0.6116 | 0.1161 | 0.0409 | −0.2141 | 0.1092 | 0.425 |
| Chl-a | 0.1359 | 0.5779 | 0.1367 | 0.031 | 0.1927 | 0.4013 | −0.3423 | 0.4434 |
| TN | 0.0608 | 0.0569 | 0.2448 | −0.2493 | 0.1071 | 0.1702 | −0.3496 | −0.2304 |
| NO3-N | 0.2517 | 0.1459 | 0.0284 | −0.4487 | 0.3024 | 0.1301 | −0.2491 | −0.0113 |
| NO2-N | 0.2622 | 0.2086 | −0.1022 | 0.0624 | 0.3559 | 0.2176 | 0.2283 | −0.2461 |
| NH4-N | 0.1475 | −0.1194 | 0.0692 | −0.4392 | 0.2372 | 0.0732 | −0.2411 | −0.2637 |
| TP | 0.0933 | −0.1334 | 0.0551 | −0.1064 | 0.4013 | −0.4433 | −0.3708 | −0.1655 |
| PO4-P | 0.1275 | −0.2349 | −0.1349 | 0.0523 | 0.1842 | −0.4857 | −0.1892 | 0.2221 |
| SiO3-Si | 0.1525 | 0.2863 | 0.1133 | 0.2308 | 0.2428 | 0.1948 | 0.1607 | 0.2049 |
| DIN | 0.2709 | −0.0199 | 0.0579 | −0.5611 | 0.2077 | 0.0269 | 0.4838 | −0.087 |
| Variance (%) | 56.19 | 12.39 | 9.26 | 7.81 | 36.21 | 14.09 | 6.87 | 6.09 |
| Cumulative (%) | 56.20 | 68.58 | 77.84 | 85.65 | 36.21 | 50.30 | 57.17 | 63.26 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, C.-C.; Wang, Y.-S.; Wu, M.-L.; Dong, J.-D.; Wang, Y.-T.; Sun, F.-L.; Zhang, Y.-Y. Seasonal Variation of Water Quality and Phytoplankton Response Patterns in Daya Bay, China. Int. J. Environ. Res. Public Health 2011, 8, 2951-2966. https://doi.org/10.3390/ijerph8072951
Sun C-C, Wang Y-S, Wu M-L, Dong J-D, Wang Y-T, Sun F-L, Zhang Y-Y. Seasonal Variation of Water Quality and Phytoplankton Response Patterns in Daya Bay, China. International Journal of Environmental Research and Public Health. 2011; 8(7):2951-2966. https://doi.org/10.3390/ijerph8072951
Chicago/Turabian StyleSun, Cui-Ci, You-Shao Wang, Mei-Lin Wu, Jun-De Dong, Yu-Tu Wang, Fu-Lin Sun, and Yan-Ying Zhang. 2011. "Seasonal Variation of Water Quality and Phytoplankton Response Patterns in Daya Bay, China" International Journal of Environmental Research and Public Health 8, no. 7: 2951-2966. https://doi.org/10.3390/ijerph8072951
APA StyleSun, C.-C., Wang, Y.-S., Wu, M.-L., Dong, J.-D., Wang, Y.-T., Sun, F.-L., & Zhang, Y.-Y. (2011). Seasonal Variation of Water Quality and Phytoplankton Response Patterns in Daya Bay, China. International Journal of Environmental Research and Public Health, 8(7), 2951-2966. https://doi.org/10.3390/ijerph8072951