Biomarkers of Immunotoxicity for Environmental and Public Health Research
Abstract
:1. Introduction
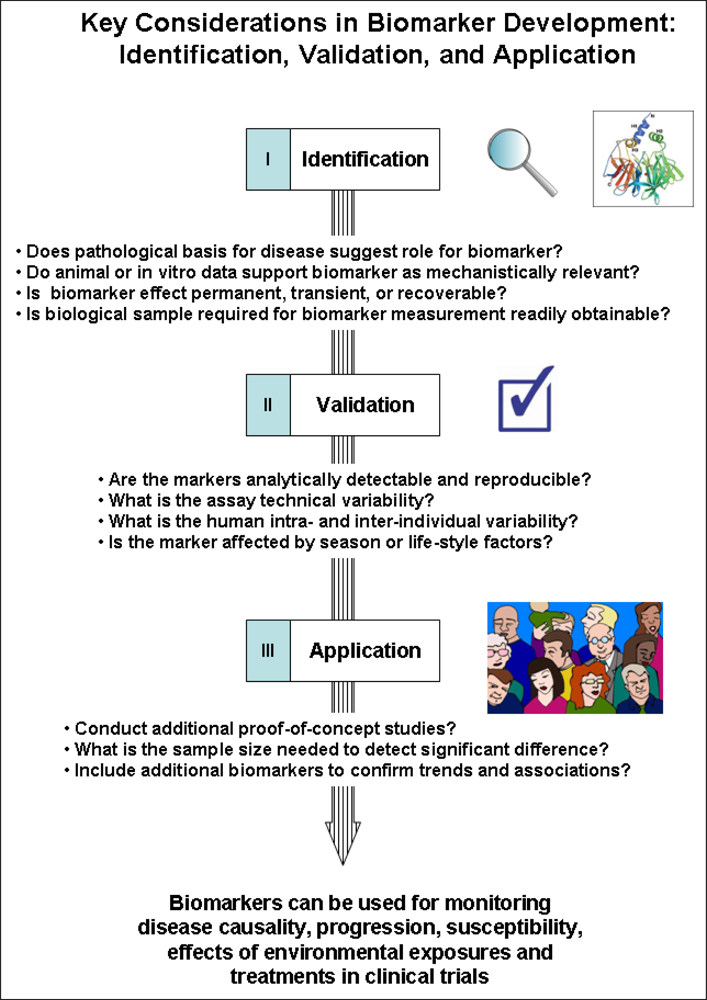
2. Key Considerations for Developing Biomarkers of Immunotoxicity
3. Sample Collection and Analytical Methods
4. Case Studies: Asthma and Chronic Obstructive Pulmonary Disease (COPD)
5. Emerging Methods
6. Discussion and Conclusions
References
- Finn, OJ. Cancer immunology. N. Engl. J. Med 2008, 358, 2704–2715. [Google Scholar]
- Hansson, GK; Nilsson, J. Introduction: Atherosclerosis as inflammation: A controversial concept becomes accepted. J. Intern. Med 2008, 263, 462–463. [Google Scholar]
- Libby, P. The molecular mechanisms of the thrombotic complications of atherosclerosis. J. Intern. Med 2008, 263, 517–527. [Google Scholar]
- Chou, MY; Hartvigsen, K; Hansen, LF; Fogelstrand, L; Shaw, PX; Boullier, A; Binder, CJ; Witztum, JL. Oxidation-specific epitopes are important targets of innate immunity. J. Intern. Med 2008, 263, 479–488. [Google Scholar]
- Biomarkers Definitions Working Group; Atkinson, AJ; Colburn, WA; DeGruttola, VG; DeMets, DL; Downing, GJ; Hoth, DF; Oates, JA; Peck, CC; Schooley, RT; Spilker, BA; Woodcock, J; Zeger, SL. Biomarkers and surrogate endpoints: Preferred definitions and conceptual framework. Clin. Pharm. Ther 2001, 69, 89–95. [Google Scholar]
- Altar, CA. The biomarkers consortium: On the critical path of drug discovery. Clin. Pharm. Ther 2008, 83, 361–364. [Google Scholar]
- Swenberg, JA; Fryar-Tita, E; Jeong, YC; Boysen, G; Starr, T; Walker, VE; Albertini, RJ. Biomarkers in toxicology and risk assessment: Informing critical dose-response relationships. Chem. Res. Toxicol 2008, 21, 253–265. [Google Scholar]
- Baker, M. In biomarkers we trust? Nat. Biotechnol 2005, 23, 297–304. [Google Scholar]
- Dieterle, F; Marrer, E. New technologies around biomarkers and their interplay with drug development. Anal. Bioanal. Chem 2008, 390, 141–154. [Google Scholar]
- Banerji, U; de Bono, J; Judson, I; Kaye, S. Biomarkers in early clinical trials: The committed and the skeptics. Clin Cancer Res 2008, 14, 2512:3–2512:4. [Google Scholar]
- Bohler, T; Nolting, J; Kamar, N; Gurragchaa, P; Reisener, K; Glander, P; Neumayer, H-H; Budde, K; Klupp, J. Validation of immunological biomarkers for the pharmacodynamic monitoring of immunosuppressive drugs in humans. Ther. Drug Monit 2007, 29, 77–86. [Google Scholar]
- Metcalf, SW; Orloff, KG. Biomarkers of exposure in community settings. J. Toxicol. Environ. Health 2004, 67, 715–726. [Google Scholar]
- Lesko, LJ; Atkinson, AJ, Jr. Use of biomarkers and surrogate endpoints in drug development and regulatory decision making: Criteria, validation, strategies. Annu. Rev. Pharmacol. Toxicol 2001, 41, 347–366. [Google Scholar]
- Buyse, M; Molenberghs, G; Burzykowski, T; Renard, D; Geys, H. The validation of surrogate endpoints in meta-analyses of randomized experiments. Biostatistics 2000, 1, 49–67. [Google Scholar]
- Immunotoxicology and Immunopharmacology, 3rd ed; Luebke, RW; House, R; Kimber, I (Eds.) CRC Press: Boca Raton, FL, USA, 2007.
- De Jong, WH; Van Loveren, H. Screening of xenobiotics for direct immunotoxicity in an animal study. Methods 2007, 41, 3–8. [Google Scholar]
- Descotes, J. Methods of evaluating immunotoxicity. Expert Opin. Drug Metab. Toxicol 2006, 2, 249–259. [Google Scholar]
- Descotes, J; Testud, F. Toxicovigilance: A new approach for the hazard identification and risk assessment of toxicants in human beings. Toxicol. Appl. Pharmacol 2005, 207, 599–603. [Google Scholar]
- Winkler, O; Hadnagy, W; Idel, H. Cytokines detectable in saliva of children as appropriate markers of local immunity of the oral cavity—An approach for the use in air pollution studies. Int. J. Hyg. Environ. Health 2001, 204, 181–184. [Google Scholar]
- Soo-Quee Koh, D; Choon-Huat Koh, G. The use of salivary biomarkers in occupational and environmental medicine. Occup. Environ. Med 2007, 64, 202–210. [Google Scholar]
- Simpson, JL; Wood, LG; Gibson, PG. Inflammatory mediators in exhaled breath, induced sputum and saliva. Clin. Exp. Allergy 2005, 35, 1180–1185. [Google Scholar]
- Robroeks, CM; Jobsis, Q; Damoiseaux, JG; Heijmans, P; Rosias, P; Hendriks, H; Dompeling, E. Cytokines in exhaled breath condensate of children with asthma and cystic fibrosis. Ann. Allergy Asthma Immunol 2006, 96, 349–355. [Google Scholar]
- Holland, NT; Pfleger, L; Berger, E; Ho, A; Bastaki, M. Molecular epidemiology biomarkers—Sample collection and processing considerations. Toxicol. Appl. Pharmacol 2005, 206, 261–268. [Google Scholar]
- Hsieh, SY; Chen, RK; Pan, YH; Lee, HL. Systematical evaluation of the effects of sample collection procedures on low-molecular-weight serum/plasma proteome profiling. Proteomics 2006, 6, 3189–3198. [Google Scholar]
- Baggerly, KA; Morris, JS; Coombes, KR. Reproducibility of SELDI-TOF protein patterns in serum: Comparing datasets from different experiments. Bioinformatics 2004, 20, 777–785. [Google Scholar]
- Diamandis, EP. Point: Proteomic patterns in biological fluids: Do they represent the future of cancer diagnostics? Clin. Chem 2003, 49, 1272–1275. [Google Scholar]
- Diamandis, EP. Analysis of serum proteomic patterns for early cancer diagnosis: Drawing attention to potential problems. J. Nat. Cancer Inst 2004, 96, 353–356. [Google Scholar]
- Jason, J; Larned, J. Single-cell cytokine profiles in normal humans: Comparison of flow cytometric reagents and stimulation protocols. J. Immunol. Method 1997, 207, 13–22. [Google Scholar]
- Duramad, P; McMahon, CW; Hubbard, A; Eskenazi, B; Holland, NT. Flow cytometric detection of intracellular TH1/TH2 cytokines using whole blood: Validation of immunologic biomarker for use in epidemiologic studies. Cancer Epidemiol. Biomarkers Prev 2004, 13, 1452–1458. [Google Scholar]
- Luque-Garcia, JL; Neubert, TA. Sample preparation for serum/plasma profiling and biomarker identification by mass spectrometry. J. Chromatogr 2007, 1153, 259–276. [Google Scholar]
- Richens, JL; Urbanowicz, RA; Metcalf, R; Corne, J; O’Shea, P; Fairclough, L. Quantitative validation and comparison of multiplex cytokine kits. J. Biomol. Screen 2010, 15, 562–568. [Google Scholar]
- Lash, GE; Pinto, LA. Multiplex cytokine analysis technologies. Expert Rev. Vaccines 2010, 9, 1231–1237. [Google Scholar]
- Maecker, HT; McCoy, JP, Jr; Amos, M; Elliott, J; Gaigalas, A; Wang, L; Aranda, R; Banchereau, J; Boshoff, C; Braun, J; et al. A model for harmonizing flow cytometry in clinical trials. Nat. Immunol 2010, 11, 975–978. [Google Scholar]
- Cosio, MG; Majo, J; Cosio, MG. Inflammation of the airways and lung parenchyma in COPD: Role of T cells. Chest 2002, 121, 160S–165S. [Google Scholar]
- Hogg, JC. Infection and COPD. Exp. Lung Res 2005, 31, 72–73. [Google Scholar]
- Baraldo, S; Lokar, OK; Turato, G; Zuin, R; Saetta, M. The role of lymphocytes in the pathogenesis of asthma and COPD. Curr. Med. Chem 2007, 14, 2250–2256. [Google Scholar]
- Bottema, RW; Reijmerink, NE; Kerkhof, M; Koppelman, GH; Stelma, FF; Gerritsen, J; Thijs, C; Brunekreef, B; van Schayck, CP; Postma, DS. IL13, CD14, pet and tobacco smoke influence atopy in 3 Dutch cohorts: The allergenic study. Eur. Resp. J 2008, 32, 593–602. [Google Scholar]
- Heaton, T; Rowe, J; Turner, S; Aalberse, RC; de Klerk, N; Suriyaarachchi, D; Serralha, M; Holt, BJ; Hollams, E; Yerkovich, S; et al. An immunoepidemiological approach to asthma: Identification of in vitro T-cell response patterns associated with different wheezing phenotypes in children. Lancet 2005, 365, 142–149. [Google Scholar]
- Feleszko, W; Zawadzka-Krajewska, A; Matysiak, K; Lewandowska, D; Peradzyńska, J; Dinh, QT; Hamelmann, E; Groneberg, DA; Kulus, M. Parental tobacco smoking is associated with augmented IL-13 secretion in children with allergic asthma. J. Allerg. Clin. Immunol 2006, 117, 97–102. [Google Scholar]
- Duramad, P; Harley, K; Lipsett, M; Bradman, A; Eskenazi, B; Holland, NT; Tager, IB. |Early environmental exposures and intracellular Th1/Th2 cytokine profiles in 24-month-old children living in an agricultural area. Environ. Health Perspect 2006, 114, 1916–1922. [Google Scholar]
- Friedrich, N; Husemoen, LL; Petersmann, A; Nauck, M; Völzke, H; Linneberg, A. The association between alcohol consumption and biomarkers of alcohol exposure with total serum immunoglobulin E levels. Alcohol. Clin. Exp. Res 2008, 32, 983–990. [Google Scholar]
- Costa, C; Rufino, R; Traves, SL; Silva, JRL; Barnes, PJ; Donnelly, LE. CXCR3 and CCR5 chemokines in induced sputum from patients with COPD. Chest 2008, 133, 26–33. [Google Scholar]
- Smyth, LJ; Starkey, C; Vestbo, J; Singh, D. CD4-regulatory cells in COPD patients. Chest 2007, 132, 156–163. [Google Scholar]
- Hacievliyagil, SS; Gunen, H; Mutlu, LC; Karabulut, AB; Temel, İ. Association between cytokines in induced sputum and severity of chronic obstructive pulmonary disease. Res. Med 2006, 100, 846–854. [Google Scholar]
- Niemela, O. Biomarkers in alcoholism. Clin. Chim. Acta 2007, 377, 39–49. [Google Scholar]
- Barnes, PJ. Immunology of asthma and chronic obstructive pulmonary disease. Nat. Rev 2008, 8, 183–192. [Google Scholar]
- Saetta, M; Mariani, M; Panina-Bordignon, P; Turato, G; Buonsanti, C; Baraldo, S; Bellettato, CM; Papi, A; Corbetta, L; Zuin, R; et al. Increased expression of the chemokine receptor CXCR3 and its ligand CXCL10 in peripheral airways of smokers with chronic obstructive pulmonary disease. Amer. J. Respir. Crit. Care Med 2002, 165, 1404–1409. [Google Scholar]
- Snell, N; Newbold, P. The clinical utility of biomarkers in asthma and COPD. Curr. Opin. Pharmacol 2008, 8, 222–235. [Google Scholar]
- Chung, KF; Barnes, PJ. Cytokines in asthma. Thorax 1999, 54, 825–857. [Google Scholar]
- Ngoc, PL; Gold, DR; Tzianabos, AO; Weiss, ST; Celedón, JC. Cytokines, allergy, and asthma. Curr. Opin. Allergy Clin. Immunol 2005, 5, 161–166. [Google Scholar]
- Macfarlane, AJ; Kon, OM; Smith, SJ; Zeibecoglou, K; Khan, LN; Barata, LT; McEuen, AR; Buckley, MG; Walls, AF; Meng, Q; et al. Basophils, eosinophils, and mast cells in atopic and nonatopic asthma and in late-phase allergic reactions in the lung and skin. J. Allerg. Clin. Immunol 2000, 105, 99–107. [Google Scholar]
- Marone, G; Triggiani, M; de Paulis, A. Mast cells and basophils: Friends as well as foes in bronchial asthma? Trends Immunol 2005, 26, 25–31. [Google Scholar]
- Yssel, H; Abbal, C; Pene, J; Bousquet, J. The role of IgE in asthma. Clin Exp Allergy 1998, 28(Suppl. 5), 104–109. [Google Scholar]
- Brown, V; Warke, TJ; Shields, MD; Ennis, M. T cell cytokine profiles in childhood asthma. Thorax 2003, 58, 311–316. [Google Scholar]
- Yeatts, K; Sly, P; Shore, S; Weiss, S; Martinez, F; Geller, A; Bromberg, P; Enright, P; Koren, H; Weissman, D; Selgrade, MJ. A brief targeted review of susceptibility factors, environmental exposures, asthma incidence, and recommendations for future asthma incidence research. Environ. Health Perspect 2006, 114, 634–640. [Google Scholar]
- Baldini, M; Lohman, IC; Halonen, M; Erickson, RP; HoltM, PG; Martinez, FD. A Polymorphism* in the 5’ flanking region of the CD14 gene is associated with circulating soluble CD14 levels and with total serum immunoglobulin E. Am. J. Respir. Cell Mol. Biol 1999, 20, 976–983. [Google Scholar]
- Kaneko, H; Matsui, E; Asano, T; Kato, Z; Teramoto, T; Aoki, M; Kawamoto, N; Lian, LA; Kasahara, K; Kondo, N. Suppression of IFN-gamma production in atopic group at the acute phase of RSV infection. Pediatr. Allergy Immunol 2006, 17, 370–375. [Google Scholar]
- Machura, E; Mazur, B; Kwiecien, J; Karczewska, K. Intracellular production of IL-2, IL-4, IFN-gamma, and TNF-alpha by peripheral blood CD3+ and CD4+ T cells in children with atopic dermatitis. Eur. J. Pediatr 2007, 166, 789–795. [Google Scholar]
- Hollams, EM; Deverell, M; Serralha, M; Suriyaarachchi, D; Parsons, F; Zhang, G; de Klerk, N; Holt, BJ; Ladyman, C; Sadowska, A; et al. Elucidation of asthma phenotypes in atopic teenagers through parallel immunophenotypic and clinical profiling. J. Allerg. Clin. Immunol 2009, 124, 463–470. [Google Scholar]
- Benninghoff, AD. Toxicoproteomics—The next step in the evolution of environmental biomarkers. Toxicol. Sci 2007, 95, 1–4. [Google Scholar]
- Baken, KA; Vandebriel, RJ; Pennings, JL; Kleinjansa, JC; van Loveren, H. Toxicogenomics in the assessment of immunotoxicity. Methods 2007, 41, 132–141. [Google Scholar]
- Salam, MT; Gauderman, WJ; McConnell, R; Lin, P-C; Gilliland, FD. Transforming growth factor-β1 C-509T polymorphism, oxidant stress, and early-onset childhood asthma. Amer. J. Respir. Crit. Care Med 2007, 176, 1192–1199. [Google Scholar]
- Pruett, SB; Holladay, SD; Prater, MR; Yucesoy, B; Luster, MI. The promise of genomics and proteomics in immunotoxicology and immunopharmacology. In Immunotoxicology and Immunopharmacology, 3rd ed; Luebke, RW, House, R, Kimber, I, Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 79–95. [Google Scholar]
- Burns-Naas, LA; Kerkvliet, NI; Laskin, DL; Bortner, CD; Burchiel, SW. The use of multiparameter flow cytometry in immunotoxicology and immunopharmacology. In Immunotoxicology and Immunopharmacology, 3rd ed; Luebke, RW, House, R, Kimber, I, Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 97–122. [Google Scholar]
- Diaz-Ramos, MC; Engel, P; Bastos, R. Towards a comprehensive human cell-surface immunome database. Immunol. Lett 2010, 134, 183–187. [Google Scholar]
- Tomar, N; De, RK. Immunoinformatics: An integrated scenario. Immunology 2010, 131, 153–168. [Google Scholar]
- Korber, B; LaBute, M; Yusim, K. Immunoinformatics comes of age. PLoS Comput Biol 2006, 2, e71:0484–e71:0494. [Google Scholar]
- Tong, JC; Ren, EC. Immunoinformatics: Current trends and future directions. Drug Discov. Today 2009, 14, 684–689. [Google Scholar]
- Yan, Q. Immunoinformatics and systems biology methods for personalized medicine. Meth. Mol. B 2010, 662, 203–220. [Google Scholar]
- Lassere, MN. The biomarker-surrogacy evaluation schema: A review of the biomarker-surrogate literature and a proposal for a criterion-based, quantitative, multidimensional hierarchical levels of evidence schema for evaluating the status of biomarkers as surrogate endpoints. Stat. Methods Med. Res 2008, 17, 303–340. [Google Scholar]
- Maksymowych, WP; Landewe, R; Boers, M; Garnero, P; Geusens, P; El-Gabalawy, H; Heinegard, D; Kraus, VB; Krause, V; Lohmander, S; et al. Development of draft validation criteria for a soluble biomarker to be regarded as a valid biomarker reflecting structural damage endpoints in rheumatoid arthritis and spondyloarthritis clinical trials. J. Rheumatol 2007, 34, 634–640. [Google Scholar]
- Lassere, MN; Johnson, KR; Boers, M; Tugwell, P; Brooks, P; Simon, L; Strand, V; Conaghan, PG; Ostergaard, M; Maksymowych, WP; et al. Definitions and validation criteria for biomarkers and surrogate endpoints: Development and testing of a quantitative hierarchical levels of evidence schema. J. Rheumatol 2007, 34, 607–615. [Google Scholar]
- Kant, AK; Graubard, BI. Ethnic and socioeconomic differences in variability in nutritional biomarkers. Amer. J. Clin. Nutr 2008, 87, 1464–1471. [Google Scholar]
- Duramad, P; Tager, IB; Holland, NT. Cytokines and other immunological biomarkers in children’s environmental health studies. Toxicol. lett 2007, 172, 48–59. [Google Scholar]
- Colosio, C; Birindelli, S; Corsini, E; Galli, CL; Maroni, M. Low level exposure to chemicals and immune system. Toxicol. Appl. Pharmacol 2005, 207, 320–328. [Google Scholar]
- Shearer, WT; Rosenblatt, HM; Gelman, RS; Oyomopito, R; Plaeger, S; Stiehm, ER; Wara, DW; Douglas, SD; Luzuriaga, K; McFarland, EJ; et al. Lymphocyte subsets in healthy children from birth through 18 years of age: The pediatric AIDS clinical trials group P1009 study. J. Allerg. Clin. Immunol 2003, 112, 973–980. [Google Scholar]
- Wolf, JM; Nicholls, E; Chen, E. Chronic stress, salivary cortisol, and alpha-amylase in children with asthma and healthy children. Biol. Psychol 2008, 78, 20–28. [Google Scholar]
- Chen, E; Fisher, EB; Bacharier, LB; Strunk, RC. Socioeconomic status, stress, and immune markers in adolescents with asthma. Psychosom. Med 2003, 65, 984–992. [Google Scholar]
| Immune Markers | Examples of Endpoints | Biological Samples |
|---|---|---|
| Cellular phenotype; activation markers | CD3, CD4, CD8, CD11c, CD19, CD25, CD56, CD14, basophils, neutrophils; Activation markers: CD69, CD45RO, CD45RA | Whole blood, Urine |
| Antibodies | IgM, IgD, IgG, IgA, IgE, | Plasma, Breastmilk |
| Cytokines | IL-2, IL-4, IL-5, IL-10, IL-13, IFN-γ, TNF-α, GM-CSF | Serum/plasma, peripheral blood, urine, saliva |
| Chemokines | RANTES, IP-10, MIP-1α, MIP-1β, MDC, TARC | Serum/plasma |
| Proliferation Tests | Mitogenic stimulation (PHA, Concavalin A, specific antigen) | Peripheral blood |
| Description of Epidemiology Study Design & Subjects | Method of exposure assessment | Biological sample and (immune biomarkers employed) | Key findings and evaluation of concordance | |
|---|---|---|---|---|
| Atopy | Longitudinal/Prospective; (n = 3,062), combined birth cohorts (ages 1–8 years) | Questionnaire; indoor environment, pet exposure | Peripheral blood (total and specific IgE and CD14/IL13 genotypes) | Atopy influenced by IL13 in <8 years and CD14 with pet interaction in ages 4 and 8 (Bottema et al. 2008) [37] |
| Longitudinal; birth cohort (n = 172) | Peripheral blood (differential cell counts and IFN-γ, TNF-α, IL-4, IL-5, IL-9, Il-10, IL-13 by ELISA) | Atopy associated with increased Th2; bronchial hyperresponsiveness associated with Th1 (Heaton et al. 2005) [38] | ||
| Cross-sectional; children ages 6–16 (n = 24) vs. reference group | Questionnaire; parental tobacco smoke | Nasopharangeal aspirate (analyzed for IL-13 cytokine levels) | ETS augments secretion of IL-13 (Feleszko et al. 2006) [39] | |
| Asthma | Longitudinal/Prospective; birth cohort (n = 239) | Questionnaire; pesticide and allergen exposures | Peripheral blood (intracellular IFN-γ and IL-4 in T-helper cells) | Th2 cells associated with asthma and wheeze; Th1 associated with breastfeeding and parental occupation in agriculture (Duramad et al. 2006) [40] |
| Asthma | Cross-sectional; children with asthma (n = 33) vs. health controls | Questionnaire | Exhaled breath condensate (IFN-γ, TNF-α, IL-2, IL-4, IL-5, IL-10) | Cytokine levels low but detectable; processing method needs improvement (Robroeks et al. 2006) [22] |
| Case-control retrospective; adults ages 20–79 (n = 3,443) | Questionnaire and blood evaluation: ethanol levels, CDT1, GGT, ASAT, ALAT | Peripheral Blood; (serum IgE) | Positive associations between alcohol consumption and total IgE serum levels in atopic subjects (Friedrich et al. 2008) [41] | |
| Chronic Obstructive Pulmonary Disease (COPD) | Cross-sectional; patients with COPD (n = 35), non-smokers (n = 18), and smokers (n = 20) | Questionnaire; criteria for non-smokers was normal spirometry results | Induced sputum (differential cell counts; CXCL9, CXCL10, CXCL11, and CCL5 by ELISA) | CXCR3 and CCL5 increased in COPD patients compared with non smokers (Costa, et al. 2008) [42] |
| Cross-sectional; patients with COPD (n = 26), smokers (n = 19), healthy non-smokers (n = 5) | Questionnaire; history of smoking | Bronchial Alveolar Lavage (BAL) and peripheral blood (CD3, CD4, CD8, CD45RA, CD25, CD69) | Increased CD8 and CD4+CD25+ in COPD BAL samples (Smyth et al. 2007) [43] | |
| Cross-sectional; COPD (n = 30), divided into two categories: Forced-expiratory volume in 1 second (FEV1) <50% and >50% | Questionnaire; smoking status | Induced sputum (IL-6, IL-8 and TNF-α) | Mean levels of three cytokines elevated in severe vs. moderate COPD (Hacievliyagil et al. 2005) [44] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Duramad, P.; Holland, N.T. Biomarkers of Immunotoxicity for Environmental and Public Health Research. Int. J. Environ. Res. Public Health 2011, 8, 1388-1401. https://doi.org/10.3390/ijerph8051388
Duramad P, Holland NT. Biomarkers of Immunotoxicity for Environmental and Public Health Research. International Journal of Environmental Research and Public Health. 2011; 8(5):1388-1401. https://doi.org/10.3390/ijerph8051388
Chicago/Turabian StyleDuramad, Paurene, and Nina T. Holland. 2011. "Biomarkers of Immunotoxicity for Environmental and Public Health Research" International Journal of Environmental Research and Public Health 8, no. 5: 1388-1401. https://doi.org/10.3390/ijerph8051388
APA StyleDuramad, P., & Holland, N. T. (2011). Biomarkers of Immunotoxicity for Environmental and Public Health Research. International Journal of Environmental Research and Public Health, 8(5), 1388-1401. https://doi.org/10.3390/ijerph8051388




