Effects of Soil Oxygen Conditions and Soil pH on Remediation of DDT-contaminated Soil by Laccase from White Rot Fungi
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Laccase and Enzymatic Activity Measurement
2.3. Preparation of DDT-contaminated Soils
2.4. Enzymatic Remediation of DDT-contaminated Soil
2.5. Sample Pre-Treatment
2.6. Gas Chromatography Analysis of DDT
2.7. Statistical Analysis
3. Results and Discussion
3.1. Effects of Different Atmospheres on Remediation of DDT-contaminated Soil by Laccase
3.2. Effects of Different Flooding Conditions on Remediation of DDT-contaminated Soil by Laccase
3.3. Effects of Different Soil pH on Remediation of DDT-contaminated Soil by Laccase
4. Conclusions
Acknowledgments
References and Notes
- John, FQ, III; Sherry, AM; Mahendra, KJ; James, MT. Reductive Dechlorination of DDE to DDMU in Marine Sediment Microcosms. Science 1998, 280, 722–724. [Google Scholar]
- Qiu, L; Zhang, J; Luo, Y. Residues of HCH and DDT in agricultural soils of north of Zhejiang and its risk evaluation. J Agro-Environ Sci 2005, 24, 1161–1165. (in Chinese).. [Google Scholar]
- Gong, Z; Wang, X; Li, B; Cao, J; Xu, F; Cui, Y; Zhang, W; Shen, W; Qin, B; Sun, Y; Tao, S. The residues distribution of DDT and its metabolites in soils from Tianjin Region, China. Acta Sci Circumstantiae 2003, 23, 447–451. (in Chinese).. [Google Scholar]
- Gao, H; Jiang, X. Bioaccumulation of organochlorine pesticides and quality safety in vegetables from Nanjing suburb. Acta Sci Circumstantiae 2005, 25, 91–93. (in Chinese).. [Google Scholar]
- Silcox, GD; Larsen, FS; Owens, WD; Choroszy-Marshall, M. Kinetics of hydrocarbon and pesticide removal from clay soils during thermal treatment in a pilot-scale rotary kiln. Waste Manage 1995, 15, 339–349. [Google Scholar]
- Parfitt, RL; Whitton, JS; Susarla, S. Removal of DDT residues from soil by leaching with surfactants. Commun. Soil Sci. Plant Anal 1995, 26, 2231–2241. [Google Scholar]
- Villa, RD; Pupo Nogueira, RFP. Oxidation of p,p′-DDT and p,p′-DDE in highly and long-term contaminated soil using Fenton reaction in a slurry system. Sci. Total Environ 2006, 371, 11–18. [Google Scholar]
- Yao, F; Jiang, X; Yu, G; Wang, F; Bian, Y. Evaluation of accelerated dechlorination of p,p′- DDT in acidic paddy soil. Chemosphere 2006, 64, 628–633. [Google Scholar]
- Gautam, SK; Sumathi, S. Dechlorination of DDT, DDD and DDE in soil (slurry) phase using magnesium/palladium system. J. Colloid Interface Sci 2006, 304, 144–151. [Google Scholar]
- Lin, C; Chang, TC. Photosensitized reduction of DDT using visible light: The intermediates and pathways of dechlorination. Chemosphere 2007, 66, 1003–1011. [Google Scholar]
- Megharaj, M; Boul, H; Thiele, J. Effects of DDT and its metabolites on soil algae and enzymatic activity. Biol. Fertil. Soils 1999, 29, 130–134. [Google Scholar]
- Gonzalez, M; Miglioranza, K; Moreno, De, J; Moreno, V. Organochlorine pesticide residues in Leek (Allium porrum) crops grown on untreated soils from an agricultural environment. J. Agric. Food Chem 2003, 51, 5024–5029. [Google Scholar]
- Lunney, A; Zeer, B; Reimer, K. Uptake of weathered DDT in vascular plants: potential for phytoremediation. Environ. Sci. Technol 2004, 38, 6147–6154. [Google Scholar]
- An, Q; Dong, Y; Wang, H; Wei, F. Uptake of aged residual and current-applied DDT and its metabolites, DDD, DDMS and DDMU by paddy rice (Oryza Sativa). Acta Pedo Sin 2008, 45, 686–692. (in Chinese).. [Google Scholar]
- Gaw, S; Palmer, G; Kim, N; Wilkins, A. Preliminary evidence that copper inhibits the degradation of DDT to DDE in pip and stonefruit orchard soils in Auckland region, New Zealand. Environ. Pollut 2003, 122, 1–5. [Google Scholar]
- Kantachote, D; Singleton, J; Naidu, R; McClure, N; Megharaj, M. Sodium application enhances DDT transformation in a long-term contaminated soil. Water Air Soil Pollut 2004, 154, 115–124. [Google Scholar]
- Purnomo, AS; Kamei, I; Londo, R. Degradation of 1,1,1-trchloro-2,2-bis (4-chlorophenyl) ethane (DDT) by Brown-Rot Fungi. J. Biosci. Bioeng 2008, 105, 614–621. [Google Scholar]
- Sutherland, TD; Horne, I; Weir, KM; Coppin, CW; Williams, MR; Selleck, M; Russell, RJ; Oakeshott, JG. Enzymatic bioremediation from enzyme discovery to applications. Clin. Exp. Pharmacol. Physiol 2004, 31, 817–821. [Google Scholar]
- Baldrian, P. Fungal laccase-occurrence and properties. FEMS Microbiol. Rev 2006, 30, 215–242. [Google Scholar]
- Ahn, MY; Dec, J; Kim, JE; Bollag, JM. Treatment of 2,4-dichlorophenol polluted soil with free and immobilized laccase. J. Environ. Qual 2002, 31, 1509–1515. [Google Scholar]
- Wu, Y; Teng, Y; Li, Z; Liao, X; Luo, Y. Potential role of polycyclic aromatic hydrocarbons (PAHs) oxidation by fungal laccase in the remediation of an aged contaminated soil. Soil Biol. Biochem 2008, 40, 789–796. [Google Scholar]
- Khlifi, R; Sayadi, S; Belbahri, L; Woodward, S; Mechichi, T. Effect of HBT on the stability of laccase during the decolourization of textile wastewaters. J. Chem. Technol. Biotechnol 2009, 84, 1828–1833. [Google Scholar]
- Ruggiero, P; Dec, J; Bollag, JM. Soil as a catalytic system. In Soil biochemistry; Stotzky, G, Bollag, JM, Eds.; Marcel Dekker Inc: New York, NY, USA, 1996; Volume 9, pp. 79–122. [Google Scholar]
- Davies, GJ; Ducros, V. Laccase. In Handbook of Metalloproteins; Messerschmidt, A, Huber, R, Poulos, T, Wieghardt, K, Eds.; Wiley Ltd.: New York, NY, USA, 2001; Volume 2, pp. 1359–1368. [Google Scholar]
- Wong, DWS. Structure and Action Mechanism of Ligninolytic Enzymes. Appl. Biochem. Biotechnol 2009, 157, 174–209. [Google Scholar]
- Gianfreda, L; Xu, F; Bollag, JM. Laccases: A Useful Group of Oxidoreductive Enzymes. Biorem. J 1999, 3, 1–26. [Google Scholar]
- Huang, H; Zhang, X. Regulation of environment pH by white rot fungi and its reciprocity in laccase production. J Microbiol 2006, 26, 37–40. (in Chinese).. [Google Scholar]
- Wang, Y; Zhou, C. Condition and partial characterization of laccase from polyporus W38. J Microbiol 2002, 22, 28–29. (in Chinese).. [Google Scholar]
- Leonowicz, A; Edgehill, RU; Bollag, JM. The effect of pH on the transformation of syringic and vanillic acids by the laccases of Rhizoctonia praticola and Trametes versicolor. Arch. Microbiol 1984, 137, 89–96. [Google Scholar]
- Chu, WK; Wong, MH; Zhang, J. Accumulation, distribution and transformation of DDT and PCBs by Phragmites australis and Oryza sativa L.: II. Enzyme study. Environ. Geochem. Health 2006, 28, 169–181. [Google Scholar]
- You, G; Sayles, G; Kupferle, M; Kim, I; Bishop, P. Anaerobic DDT biotransformation: enhancement by application of surfactants and low oxidation reduction potential. Chemosphere 1996, 32, 2269–2284. [Google Scholar]
- Leonowicz, A; Sarkar, JM; Bollag, JM. Improvement in stability of an immobilized fungal laccase. Appl. Microbiol. Biotechnol 1988, 29, 129–135. [Google Scholar]
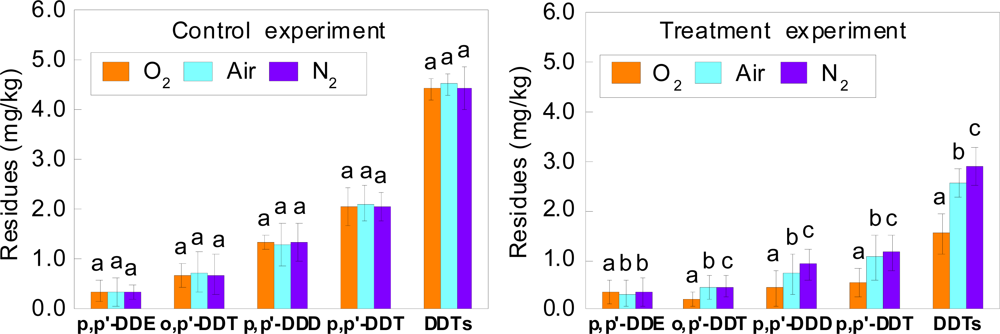
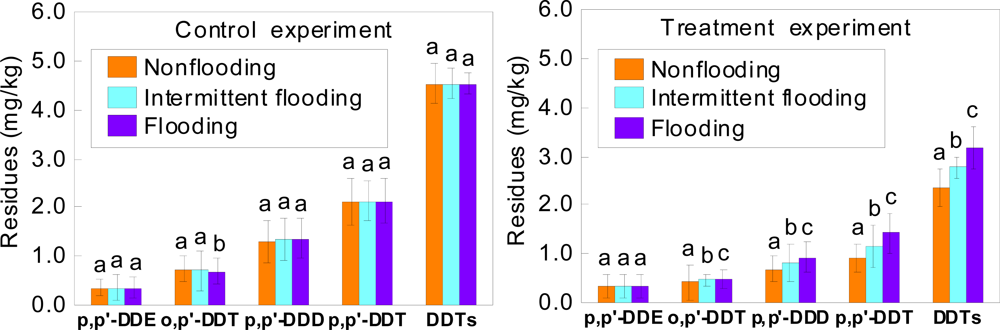
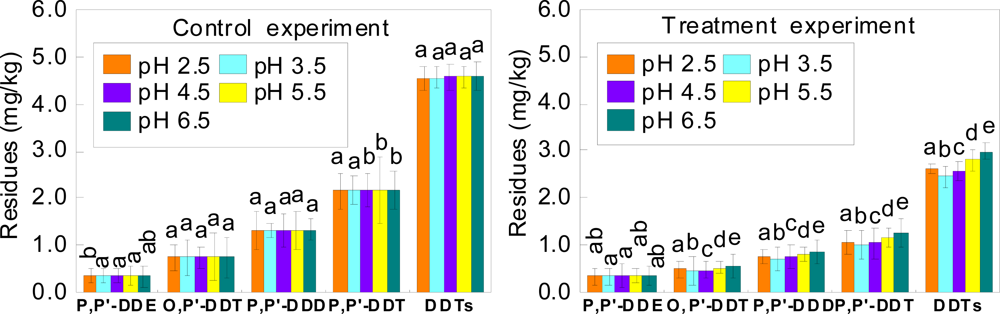
| Atmosphere | p,p′- DDE | o,p′-DDT | p,p′-DDD | p,p′-DDT | DDTs |
|---|---|---|---|---|---|
| N2 | 1.5% | 38.8% | 34.7% | 50.5% | 40.5% |
| Air | 5.4% | 41.5% | 48.9% | 55.0% | 48.5% |
| O2 | 1.9% | 74.1% | 68.9% | 76.5% | 68.6% |
| Flooding condition | p,p′- DDE | o,p′-DDT | p,p′-DDD | p,p′-DDT | DDTs |
|---|---|---|---|---|---|
| Flooding | 3.5% | 38.0% | 34.1% | 39.0% | 34.8% |
| Intermittent flooding | 3.9% | 39.4% | 42.3% | 50.0% | 42.8% |
| Nonflooding | 5.7% | 45.4% | 50.9% | 60.8% | 51.5% |
| Soil pH | p,p′- DDE | o,p′-DDT | p,p′-DDD | p,p′-DDT | DDTs |
|---|---|---|---|---|---|
| 2.5 | 5.5% | 38.6% | 47.0% | 54.2% | 46.1% |
| 3.5 | 5.8% | 42.9% | 50.7% | 57.9% | 49.7% |
| 4.5 | 5.7% | 40.3% | 48.3% | 55.6% | 47.4% |
| 5.5 | 5.1% | 33.4% | 42.6% | 50.5% | 42.2% |
| 6.5 | 4.9% | 30.4% | 39.5% | 46.1% | 38.7% |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, Y.; Yi, X. Effects of Soil Oxygen Conditions and Soil pH on Remediation of DDT-contaminated Soil by Laccase from White Rot Fungi. Int. J. Environ. Res. Public Health 2010, 7, 1612-1621. https://doi.org/10.3390/ijerph7041612
Zhao Y, Yi X. Effects of Soil Oxygen Conditions and Soil pH on Remediation of DDT-contaminated Soil by Laccase from White Rot Fungi. International Journal of Environmental Research and Public Health. 2010; 7(4):1612-1621. https://doi.org/10.3390/ijerph7041612
Chicago/Turabian StyleZhao, Yuechun, and Xiaoyun Yi. 2010. "Effects of Soil Oxygen Conditions and Soil pH on Remediation of DDT-contaminated Soil by Laccase from White Rot Fungi" International Journal of Environmental Research and Public Health 7, no. 4: 1612-1621. https://doi.org/10.3390/ijerph7041612
APA StyleZhao, Y., & Yi, X. (2010). Effects of Soil Oxygen Conditions and Soil pH on Remediation of DDT-contaminated Soil by Laccase from White Rot Fungi. International Journal of Environmental Research and Public Health, 7(4), 1612-1621. https://doi.org/10.3390/ijerph7041612




