Parasite Zoonoses and Wildlife: Emerging Issues
Abstract
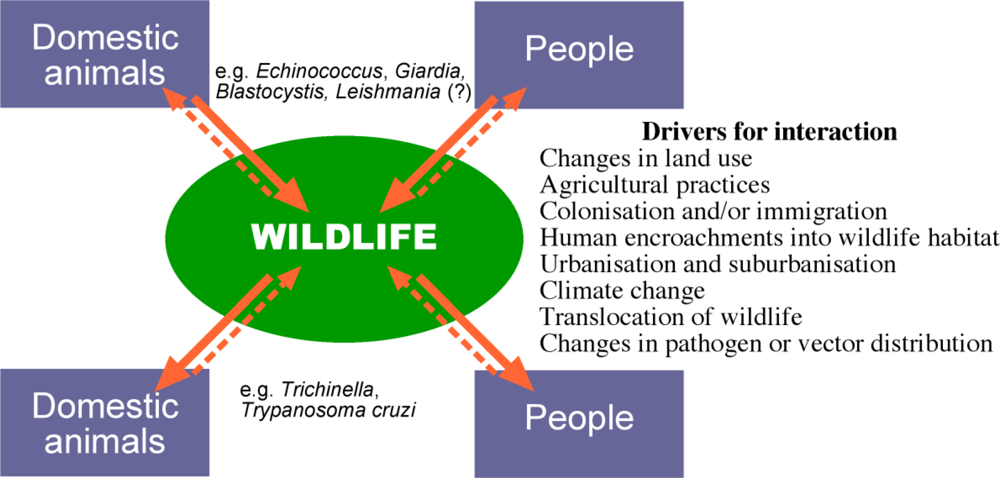
:1. Pathogens in Wildlife
2. Spill-Over vs Spill-Back
3. Transmission of Parasites from Humans to Wildlife
3.1. Sarcoptes/Sarcoptic Mange
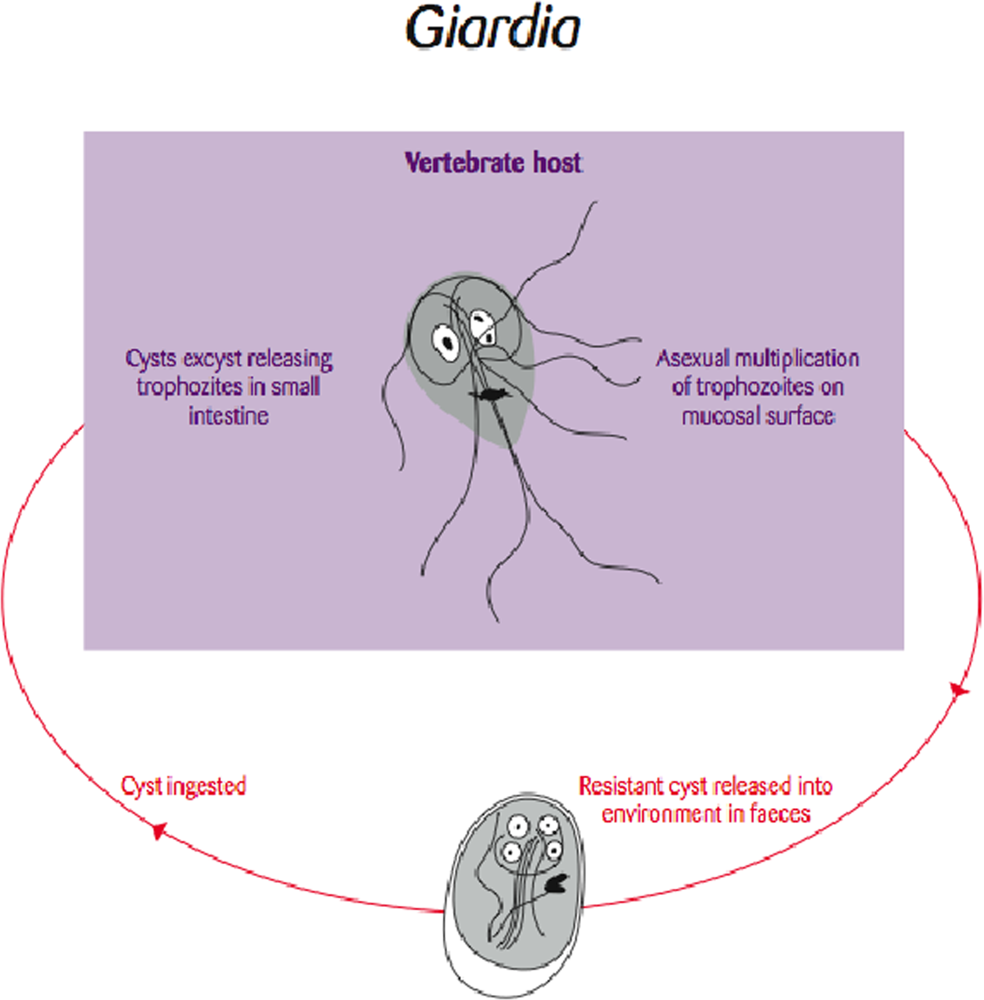
3.2. Giardia/Beaver Fever
4. Transmission of Parasites between Wildlife and Domestic Animals
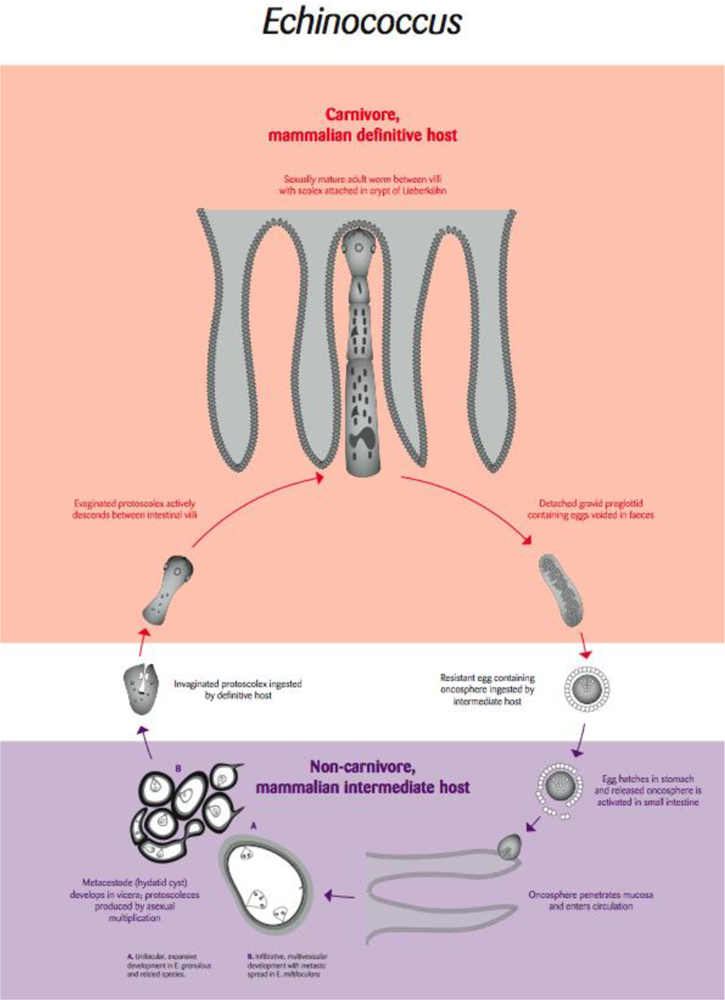
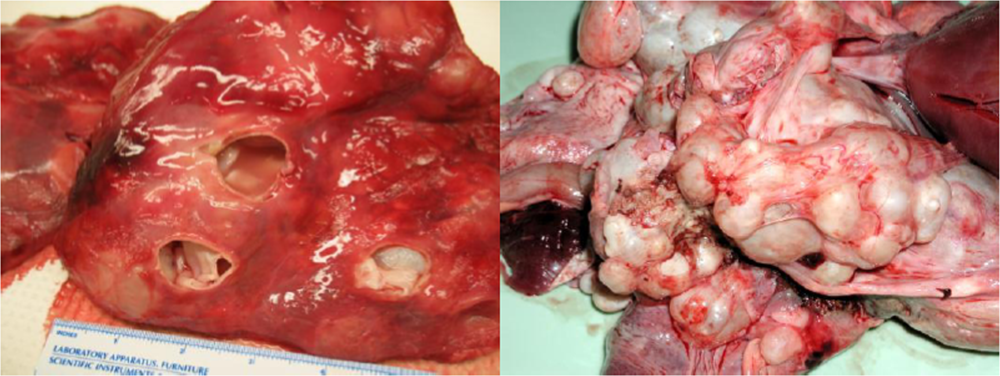
4.1. Echinococcus: Hydatid Disease
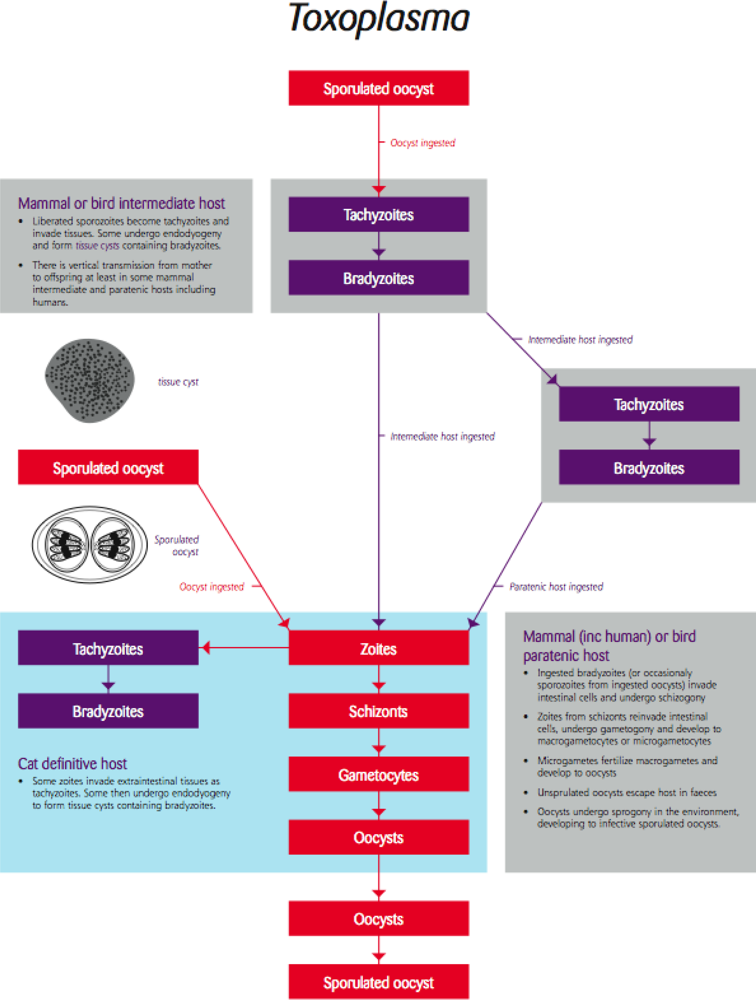
4.2. Toxoplasma/Toxoplasmosis
5. The Potential for Disease Emergence in Wildlife
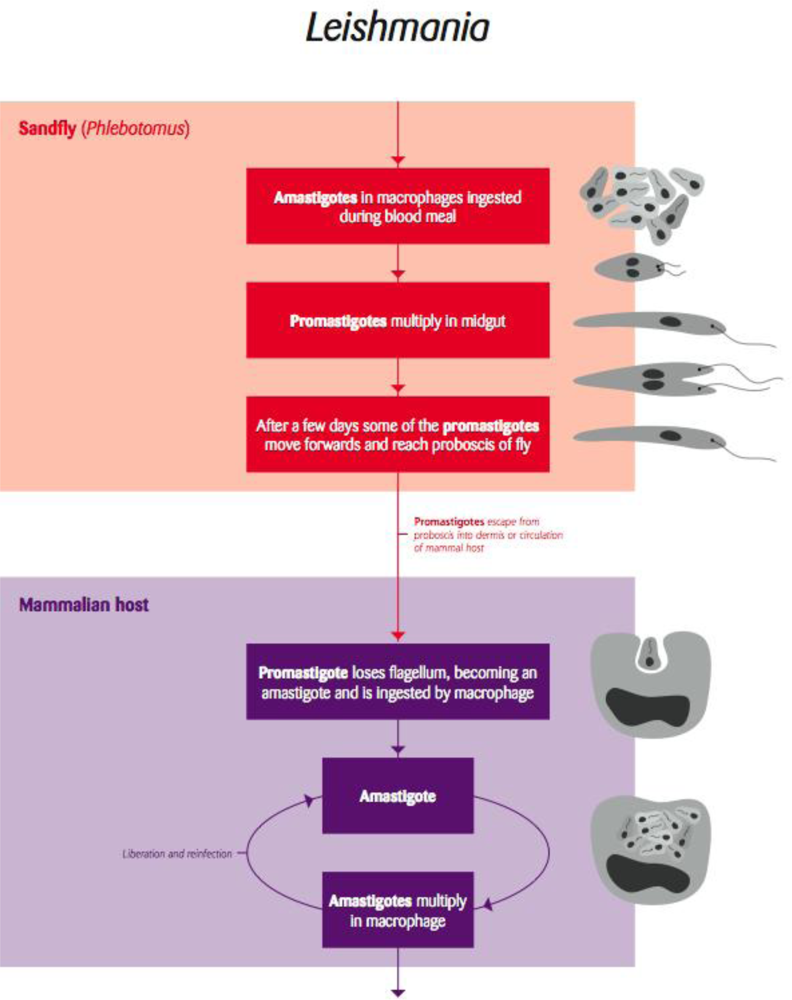
5.1. Leishmania and Trypanosoma
6. Concluding Remarks and Perspectives for the Future
References
- Daszak, P; Cunningham, AA; Hyatt, AD. Emerging infectious diseases of wildlife – threats to biodiversity and human health. Science 2000, 287, 443–449. [Google Scholar]
- Holmes, JC. Parasites as threats to biodiversity in shrinking ecosystems. Biodivers. Conservn 1996, 5, 975–983. [Google Scholar]
- McCallum, H; Dobson, A. Detecting disease and parasite threats to endangered species and ecosystems. Trends Ecol. Evol 1995, 10, 190–194. [Google Scholar]
- LoGiudice, K; Ostfeld, RS; Schmidt, KA; Keesing, F. The ecology of infectious disease: Effects of host diversity and community composition on Lyme disease risk. Proc. Natl. Acad. Sci. U.S.A 2003, 100, 567–571. [Google Scholar]
- Maillard, J-C; Gonzalez, J-P. Biodiversity and emerging disease. Ann. N.Y. Acad. Sci 2006, 1081, 1–16. [Google Scholar]
- Woodroffe, R. Managing disease threats to wild mammals. Animal Conserv 1999, 2, 185–190. [Google Scholar]
- Kruse, H; Kirkemo, AM; Handeland, K. Wildlife as source of zoonotic infections. Emerg. Inf. Dis 2004, 10, 2067–2072. [Google Scholar]
- Thompson, RCA; Murrell, KD. Parasitic Zoonoses – Emerging Issues, Thematic Issue. Int. J. Parasitol 2005, 35, 1153–1332. [Google Scholar]
- Pence, DB; Ueckermann, E. Sarcoptic manage in wildlife. Rev. Sci. Tech 2002, 21, 385–398. [Google Scholar]
- Martin, RW; Handasyde, KA; Skerratt, LF. Current distribution of sarcoptic mange in wombats. Aust. Vet. J 1998, 76, 411–414. [Google Scholar]
- Skerratt, LF; Campbell, NJ; Murrell, A; Walton, S; Kemp, D; Barker, SC. The mitochondrial 12S gene is a suitable marker of populations of Sarcoptes scabei from wombats, dogs and humans in Australia. Parasitol. Res 2002, 88, 376–379. [Google Scholar]
- Kettlewell, JS; Bettiol, SS; Davies, N; Milstein, T; Goldsmid, JM. Epidemiology of giardiasis in Tasmania: a potential risk to residents and visitors. J. Travel Med 1998, 5, 127–130. [Google Scholar]
- Thompson, RCA. The zoonotic significance and molecular epidemiology of Giardia and giardiasis. Vet. Parasitol 2004, 126, 15–35. [Google Scholar]
- Grazyk, TK; DaSilva, AJ; Cranfield, MR; Nizeyi, JB; Kalema, GR; Pieniazek, NJ. Cryptopsoridium parvum Genotype 2 infections in free ranging mountain gorillas (Gorilla gorilla beringei) of the Bwindi Impenetrable National Park, Uganda. Parasitol. Res 2001, 87, 368–370. [Google Scholar]
- Graczyk, TK; Bosco-Nizeyi, J; Ssebide, B; Thompson, RCA; Read, C; Cranfield, MR. Anthropozoonotic Giardia duodenalis genotype (assemblage) A infections in habitats of freeranging human-habituated gorillas, Uganda. J. Parasitol 2002, 88, 905–909. [Google Scholar]
- Kutz, SJ; Thompson, RCA; Polley, L; Kandola, K; Nagy, J; Wielinga, CM; Elkin, BT. Giardia assemblage a: human genotype in muskoxen in the Canadian Arctic. Parasit. Vectors 2008, 1, 32. [Google Scholar]
- Aloisio, F; Filippini, G; Antenucci, P; Lepri, E; Pezzotti, G; Cacciò, SM; Pozio, E. Severe weight loss in lambs infected with Giardia duodenalis assemblage B. Vet. Parasitol 2006, 142, 154–158. [Google Scholar]
- Olson, ME; McAllister, TA; Deselliers, L; Morck, DW; Cheng, KJ; Buret, AG; Ceri, H. Effects of giardiasis on production in a domestic ruminant (lamb) model. Am. J. Vet. Res 1995, 56, 1470–1474. [Google Scholar]
- Thompson, RCA; Palmer, CS; O’Handley, R. The public health and clinical significance of Giardia and Cryptosporidium in domestic animals. Vet. J 2008, 177, 18–25. [Google Scholar]
- Adams, PJ; Monis, PT; Elliot, AD; Thompson, RCA. Cyst morphology and sequence analysis of the small subunit rDNA and ef1 identifies a novel Giardia genotype in a quenda (Isoodon obesulus) from Western Australia. Inf. Gen. Evol 2004, 4, 365–370. [Google Scholar]
- Thompson, RCA; Lymbery, AJ; Pearce, DA; Finn, KC; Reynoldson, JA; Meloni, BP. Giardia duodenalis: exposure to metronidazole inhibits competitive interactions between isolates of the parasite in vitro. J. Parasitol 1996, 82, 679–683. [Google Scholar]
- Schantz, PM; Chai, J; Craig, PS; Eckert, J; Jenkins, DJ; Macpherson, CNL; Thakur, A. 1995; Epidemiology and control of hydatid disease. In Echinococcus and Hydatid Disease; CAB International: Wallingford, U.K, 1995.
- Storandt, ST; Virchow, DR; Dryden, MW; Hygnstrom, SE; Kazacos, KR. Distribution and prevalence of Echinococcus multilocularis in wild predators in Nebraska, Kansas, and Wyoming. J. Parasitol 2002, 88, 420–422. [Google Scholar]
- Schweiger, A; Ammann, RW; Candina, D; Clavien, PA; Eckert, J; Gottstein, B; Halkic, N; Muellhaupt, B; Prinz, BM; Reichen, J; Tarr, PE; Torgerson, PR; Deplazes, P. Human alveolar echinococcosis after fox population increase, Switzerland. Emerg. Inf. Dis 2007, 13, 878–882. [Google Scholar]
- Romig, T; Thoma, D; Weible, AK. Echinococcus multilocularis – a zoonosis of anthropogenic environments? J. Helminthol 2006, 80, 207–212. [Google Scholar]
- Vervaeke, M; Vervaeke, M; Davis, S; Leirs, H; Verhagen, R. Implications of increased susceptibility to predation for managing the sylvatic cycle of Echinococcus multilocularis. Parasitol 2006, 132, 893–901. [Google Scholar]
- Thompson, RCA. The taxonomy, phylogeny and transmission of Echinococcus. Exp. Parasitol 2008, 119, 439–446. [Google Scholar]
- Joly, DO; Messier, F. The distribution of Echinococcus granulosus in moose: evidence for parasite-induced vulnerability to predation by wolves? Oecologia 2004, 140, 586–590. [Google Scholar]
- Thompson, RCA; Boxell, AC; Ralston, BJ; Constantine, CC; Hobbs, RP; Shury, T; Olson, ME. Molecular and morphological characterisation of Echinococcus in cervids from North America. Parasitol 2006, 132, 439–447. [Google Scholar]
- Mech, LD. The Wolves of Isle Royale; University Press of the Pacific: Honolulu, Hawaii, U.S.A, 2002. [Google Scholar]
- Petersen, RO; Hadler, KP; Freeman, HI. Predator-prey populations with parasitic infection. J. Math. Biol 1989, 27, 609–631. [Google Scholar]
- Corbett, L. The Dingo in Australia and Asia; J.B. Books: Marleston, South Australia, 2001. [Google Scholar]
- Durie, PH; Riek, RF. The role of the dingo and wallaby in the infestation of cattle with hydatids (Echinococcus granulosus (Batsch, 1786) Rudolphi, 1805) in Queensland. Aust. Vet. J 1952, 28, 249–254. [Google Scholar]
- Barnes, TS; Morton, JM; Coleman, GT. Clustering of hydatid infection in macropodids. Int. J. Parasitol 2007, 37, 943–952. [Google Scholar]
- Eckert, J. Echinococcosis in animals: clinical aspects, diagnosis and treatment. In WHO/OIE Manual on Echinococcosis in Humans and Animals: a Public Health Problem of Global Concern; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Schwabe, CW. Current status of hydatid disease: a zoonosis of increasing importance. In The Biology of Echinococcus and Hydatid Disease; George Allen & Unwin: London, UK, 1986. [Google Scholar]
- Jenkins, DJ; Romig, T; Thompson, RCA. Emergence/re-emergence of Echinococcus spp. – a global update. Int. J. Parasitol 2005, 35, 1205–1219. [Google Scholar]
- Nelson, GS. The parasite and the host. In Parasites and Western Man; MTP Press: Lancaster, UK, 1979. [Google Scholar]
- Dubey, JP. Toxoplasmosis. In Microbiology and Microbial Infections Vol 5; Arnold: London, UK, 1998. [Google Scholar]
- Thompson, RCA; Traub, RJ; Parameswaran, N. Molecular epidemiology of foodborne parasitic zoonoses. In Food-Borne Parasitic Zoonoses Zoonoses; Spinger: New York, USA, 2007. [Google Scholar]
- Berdoy, M; Webster, JP; Macdonald, DW. Fatal attraction in rats infected with Toxoplasma gondii. Proc. Biol. Sci 2000, 267, 1591–1594. [Google Scholar]
- Obendorf, DL; Statham, P; Driessen, M. Detection of agglutinating antibodies to Toxoplasma gondii in sera from free-ranging eastern barred bandicoots (Perameles gunnii). J. Wildlife Dis 1996, 32, 623–626. [Google Scholar]
- Conrad, PA; Miller, MA; Kreuder, C; James, ER; Mazet, J; Dabritz, H; Jessup, DA; Gulland, F; Grigg, ME. Transmission of Toxoplasma: clues from the study of sea otters as sentinels of Toxoplasma gondii flow into the marine environment. Int. J. Parasitol 2005, 35, 1155–1168. [Google Scholar]
- Miller, MA; Grigg, ME; Kreuder, C; James, ER; Melli, AC; Crosbie, PR; Jessup, DA; Boothroyd, JC; Brownstein, D; Conrad, PA. An unusual genotype of Toxoplasma gondii is common in California sea otters (Enhydra lutris nereis) and is a cause of mortality. Int. J. Parasitol 2004, 34, 275–284. [Google Scholar]
- Abbott, I. Mammalian faunal collapse in Western Australia, 1875–1925: the hypothesised role of epizootic disease and a conceptual model of its origin, introduction, transmission, and spread. Aust. Zool 2006, 33, 530–561. [Google Scholar]
- Thompson, RCA. The future impact of societal and cultural factors on parasitic disease – some emerging issues. Int. J. Parasitol 2001, 31, 949–959. [Google Scholar]
- Gonzalez, LE. Toxoplasma gondii infection lower anxiety as measured in the plus-maze and social interaction tests in rats A behavioural analysis. Behav. Brain Res 2007, 177, 70–79. [Google Scholar]
- Rose, K; Curtis, J; Baldwin, T; Mathis, A; Kumar, B; Sakthianandeswaren, A; Spurck, T; Low Choy, J; Handman, E. Cutaneous leishmaniasis in red kangaroos: isolation andcharacterisation of the causative organisms. Int. J. Parasitol 2004, 34, 655–664. [Google Scholar]
- Stark, D; Pett, S; Marriott, D; Harkness, J. Post-Kala-Azar dermal leishmaniasis due to Leishmania infantum in a human immunodeficiency virus type 1-infected patient. J. Clin. Microbiol 2006, 44, 1178–1180. [Google Scholar]
- Konecny, P; Stark, DJ. An Australian case of New World cutaneous leishmaniasis. Med. J. Aust 2007, 19, 315–317. [Google Scholar]
- Thompson, RCA; Owen, IL; Puana, I; Banks, D; Davis, TME; Reid, SA. Parasites and biosecurity – the example of Australia. Trends Parasitol 2003, 19, 410–416. [Google Scholar]
- Smith, A; Clark, P; Averis, S; Lymbery, AJ; Wayne, AF; Morris, KD; Thompson, RCA. Trypanosomes in a declining species of threatened Australian marsupial, the brush-tailed bettong Bettongia penicillata (Marsupialia: Potoroidae). Parasitol 2008, 135, 1329–1335. [Google Scholar]
- Wyatt, KB; Campos, PF; Gilbert, MTP; Kolokotronis, S-O; Hynes, WH; DeSalle, R; Daszak, P; MacPhee, RDE; Greenwood, AD. Historical mammal extinction on Christmas Island (Indian Ocean) correlates with introduced infectious disease. PLoS One 2008, 3, e3602. [Google Scholar]
Share and Cite
Thompson, R.C.A.; Kutz, S.J.; Smith, A. Parasite Zoonoses and Wildlife: Emerging Issues. Int. J. Environ. Res. Public Health 2009, 6, 678-693. https://doi.org/10.3390/ijerph6020678
Thompson RCA, Kutz SJ, Smith A. Parasite Zoonoses and Wildlife: Emerging Issues. International Journal of Environmental Research and Public Health. 2009; 6(2):678-693. https://doi.org/10.3390/ijerph6020678
Chicago/Turabian StyleThompson, R.C. Andrew, Susan J. Kutz, and Andrew Smith. 2009. "Parasite Zoonoses and Wildlife: Emerging Issues" International Journal of Environmental Research and Public Health 6, no. 2: 678-693. https://doi.org/10.3390/ijerph6020678
APA StyleThompson, R. C. A., Kutz, S. J., & Smith, A. (2009). Parasite Zoonoses and Wildlife: Emerging Issues. International Journal of Environmental Research and Public Health, 6(2), 678-693. https://doi.org/10.3390/ijerph6020678




