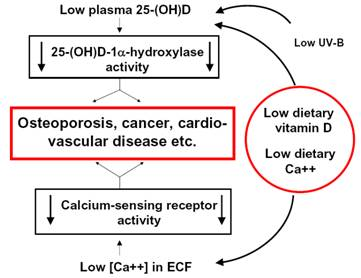
Vitamin D and Calcium Insufficiency-Related Chronic Diseases: an Emerging World-Wide Public Health Problem
Abstract
:1. Introduction
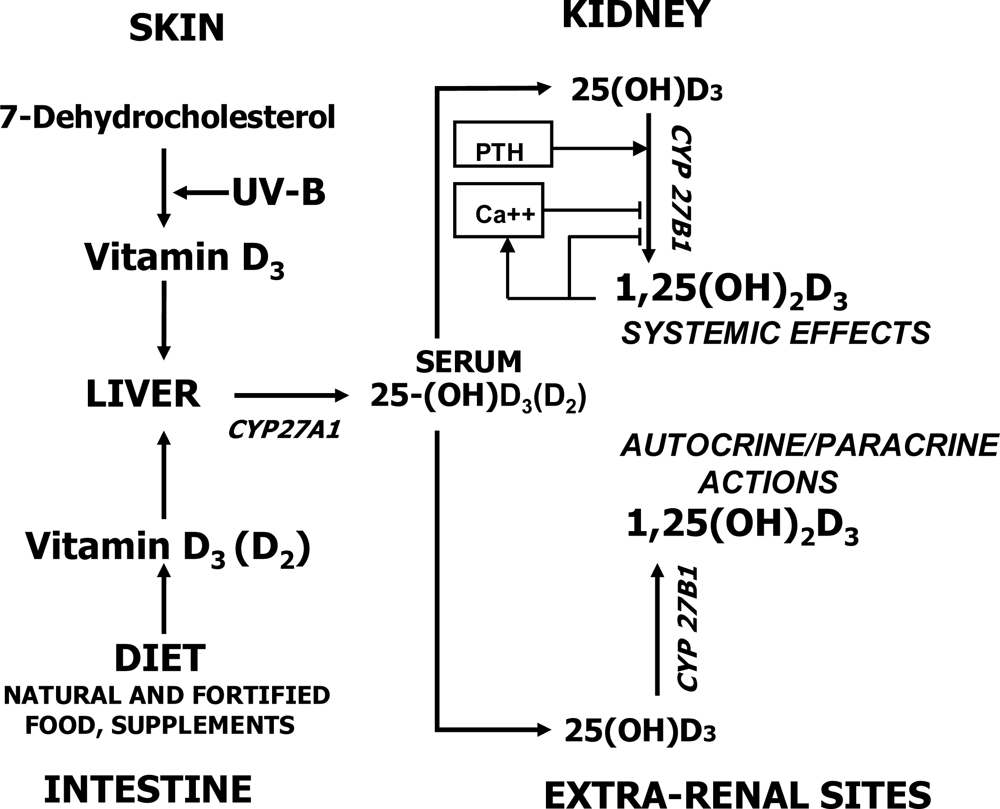
2. Why a Low Vitamin D Status and a Nutritional Calcium Deficit are Risk Factors for many Chronic Diseases
3. Vitamin D Insufficiency: A World-Wide Phenomenon
3.1. Definition of Vitamin D Insufficiency
3.2. Epidemiology of Vitamin D Insufficiency
3.3. Vitamin D Insufficiency in Different Population Segments
4. Inadequate Calcium Intake: A World-Wide Problem
4.1. Recommended Calcium Intake Levels
4.2. Epidemiology of Calcium Intake
4.3. Population Segments with Low Habitual Calcium Intake
5. Strategies for disease prevention
5.1. Need to Increase Combined Intakes to Daily 800 IU Vitamin D and 1,200 mg Calcium
5.2. Rationale for Advocating Combined Intake of Vitamin D and Calcium
6. What can be Done?
6.1. Supplementation by Fixed Vitamin D/Calcium Combination Tablets
6.2. Vitamin D and Calcium Enrichment in Single Foodstuffs
6.3. Vitamin D and Calcium Addition to Cereal Grain Products
Acknowledgments
References
- Peterlik, M; Cross, HS. Vitamin D and calcium deficits predispose for multiple chronic diseases. Eur. J. Clin. Invest 2005, 35, 290–304. [Google Scholar]
- Peterlik, M; Grant, WB; Cross, HS. Calcium, vitamin D and cancer. Anticancer Res 2009, 29, 3687–3698. [Google Scholar]
- Peterlik, M; Cross, HS. Vitamin D and calcium insufficiency-related chronic diseases: molecular and cellular pathophysiology. Eur J Clin Nutr, 2009; doi:10.1038/ejcn.2009.105. [Google Scholar]
- Zehnder, D; Bland, R; Williams, MC; McNinch, RW; Howie, AJ; Stewart, PM; Hewison, M. Extrarenal expression of 25-hydroxyvitamin D3-1 alpha-hydroxylase. J. Clin. Endocrinol. Metab 2001, 86, 888–894. [Google Scholar]
- Pillai, S; Bikle, DD; Elias, PM. 1,25-Dihydroxyvitamin D production and receptor binding in human keratinocytes varies with differentiation. J. Biol. Chem 1988, 263, 5390–5395. [Google Scholar]
- Cross, HS; Peterlik, M; Reddy, GS; Schuster, I. Vitamin D metabolism in human colon adenocarcinoma-derived Caco-2 cells: expression of 25-hydroxyvitamin D3-1alpha-hydroxylase activity and regulation of side-chain metabolism. J. Steroid Biochem. Mol. Biol 1997, 62, 21–28. [Google Scholar]
- Schwartz, GG; Eads, D; Rao, A; Cramer, SD; Willingham, MC; Chen, TC; Jamieson, DP; Wang, L; Burnstein, KL; Holick, MF; Koumenis, C. Pancreatic cancer cells express 25-hydroxyvitamin D-1 alpha-hydroxylase and their proliferation is inhibited by the prohormone 25-hydroxyvitamin D3. Carcinogenesis 2004, 25, 1015–1026. [Google Scholar]
- Becker, S; Cordes, T; Diesing, D; Diedrich, K; Friedrich, M. Expression of 25-hydroxyvitamin D3-1alpha-hydroxylase in human endometrial tissue. J. Steroid Biochem. Mol. Biol 2007, 103, 771–775. [Google Scholar]
- Schwartz, GG; Whitlatch, LW; Chen, TC; Lokeshwar, BL; Holick, MF. Human prostate cells synthesize 1,25-dihydroxyvitamin D3 from 25-hydroxyvitamin D3. Cancer Epidemiol. Biomarkers Prev 1998, 7, 391–395. [Google Scholar]
- Friedrich, M; Rafi, L; Mitschele, T; Tilgen, W; Schmidt, W; Reichrath, J. Analysis of the vitamin D system in cervical carcinomas, breast cancer and ovarian cancer. Recent Results Cancer Res 2003, 164, 239–246. [Google Scholar]
- van Driel, M; Koedam, M; Buurman, CJ; Hewison, M; Chiba, H; Uitterlinden, AG; Pols, HA; van Leeuwen, JP. Evidence for auto/paracrine actions of vitamin D in bone: 1alphahydroxylase expression and activity in human bone cells. FASEB J 2006, 20, 2417–2419. [Google Scholar]
- Atkins, GJ; Anderson, PH; Findlay, DM; Welldon, KJ; Vincent, C; Zannettino, AC; O’Loughlin, PD; Morris, HA. Metabolism of vitamin D3 in human osteoblasts: evidence for autocrine and paracrine activities of 1alpha,25-dihydroxyvitamin D3. Bone 2007, 40, 1517–1528. [Google Scholar]
- Zehnder, D; Bland, R; Chana, RS; Wheeler, DC; Howie, AJ; Williams, MC; Stewart, PM; Hewison, M. Synthesis of 1,25-dihydroxyvitamin D3 by human endothelial cells is regulated by inflammatory cytokines: a novel autocrine determinant of vascular cell adhesion. J. Am. Soc. Nephrol 2002, 13, 621–629. [Google Scholar]
- Eyles, DW; Smith, S; Kinobe, R; Hewison, M; McGrath, JJ. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat 2005, 29, 21–30. [Google Scholar]
- Hewison, M; Freeman, L; Hughes, SV; Evans, KN; Bland, R; Eliopoulos, AG; Kilby, MD; Moss, PA; Chakraverty, R. Differential regulation of vitamin D receptor and its ligand in human monocyte-derived dendritic cells. J. Immunol 2003, 170, 5382–5390. [Google Scholar]
- Sigmundsdottir, H; Pan, J; Debes, GF; Alt, C; Habtezion, A; Soler, D; Butcher, EC. DCs metabolize sunlight-induced vitamin D3 to ‘program’ T cell attraction to the epidermal chemokine CCL27. Nat. Immunol 2007, 8, 285–293. [Google Scholar]
- Anderson, PH; O’Loughlin, PD; May, BK; Morris, HA. Modulation of CYP27B1 and CYP24 mRNA expression in bone is independent of circulating 1,25(OH)2D3 levels. Bone 2005, 36, 654–662. [Google Scholar]
- Cross, HS; Kallay, E. Regulation of the colonic vitamin D system for prevention of tumor progression: an update. Future Oncol 2009, 5, 493–507. [Google Scholar]
- Dobnig, H; Pilz, S; Scharnagl, H; Renner, W; Seelhorst, U; Wellnitz, B; Kinkeldei, J; Boehm, BO; Weihrauch, G; Maerz, W. Independent association of low serum 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels with all-cause and cardiovascular mortality. Arch. Intern. Med 2008, 168, 1340–1349. [Google Scholar]
- Tfelt-Hansen, J; Brown, EM. The calcium-sensing receptor in normal physiology and pathophysiology: a review. Crit. Rev. Clin. Lab. Sci 2005, 42, 35–70. [Google Scholar]
- Dvorak, MM; Siddiqua, A; Ward, DT; Carter, DH; Dallas, SL; Nemeth, EF; Riccardi, D. Physiological changes in extracellular calcium concentration directly control osteoblast function in the absence of calciotropic hormones. Proc. Natl. Acad. Sci. U S A 2004, 101, 5140–5145. [Google Scholar]
- Chang, W; Tu, C; Chen, TH; Bikle, D; Shoback, D. The extracellular calcium-sensing receptor (CaSR) is a critical modulator of skeletal development. Sci Signal 2008, 1, ra1. [Google Scholar]
- Brown, EM; Lian, JB. New insights in bone biology: unmasking skeletal effects of the extracellular calcium-sensing receptor. Sci Signal 2008, 1, pe40. [Google Scholar]
- Rodland, KD. The role of the calcium-sensing receptor in cancer. Cell Calcium 2004, 35, 291–295. [Google Scholar]
- McCarron, DA; Reusser, ME. Finding consensus in the dietary calcium-blood pressure debate. J. Am. Coll. Nutr 1999, 18, 398S–405S. [Google Scholar]
- Hintzpeter, B; Mensink, GB; Thierfelder, W; Muller, MJ; Scheidt-Nave, C. Vitamin D status and health correlates among German adults. Eur. J. Clin. Nutr 2008, 62, 1079–1089. [Google Scholar]
- Judd, SE; Nanes, MS; Ziegler, TR; Wilson, PW; Tangpricha, V. Optimal vitamin D status attenuates the age-associated increase in systolic blood pressure in white Americans: results from the third National Health and Nutrition Examination Survey. Am. J. Clin. Nutr 2008, 87, 136–141. [Google Scholar]
- Davies, KM; Heaney, RP; Recker, RR; Lappe, JM; Barger-Lux, MJ; Rafferty, K; Hinders, S. Calcium intake and body weight. J. Clin. Endocrinol. Metab 2000, 85, 4635–4638. [Google Scholar]
- Arunabh, S; Pollack, S; Yeh, J; Aloia, JF. Body fat content and 25-hydroxyvitamin D levels in healthy women. J. Clin. Endocrinol. Metab 2003, 88, 157–161. [Google Scholar]
- Alemzadeh, R; Kichler, J; Babar, G; Calhoun, M. Hypovitaminosis D in obese children and adolescents: relationship with adiposity, insulin sensitivity, ethnicity, and season. Metabolism 2008, 57, 183–191. [Google Scholar]
- Pittas, AG; Lau, J; Hu, FB; Dawson-Hughes, B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J. Clin. Endocrinol. Metab 2007, 92, 2017–2029. [Google Scholar]
- Reis, JP; von Muhlen, D; Kritz-Silverstein, D; Wingard, DL; Barrett-Connor, E. Vitamin D, parathyroid hormone levels, and the prevalence of metabolic syndrome in community-dwelling older adults. Diabetes Care 2007, 30, 1549–1555. [Google Scholar]
- Wang, TJ; Pencina, MJ; Booth, SL; Jacques, PF; Ingelsson, E; Lanier, K; Benjamin, EJ; D’Agostino, RB; Wolf, M; Vasan, RS. Vitamin D deficiency and risk of cardiovascular disease. Circulation 2008, 117, 503–511. [Google Scholar]
- Bostick, RM; Kushi, LH; Wu, Y; Meyer, KA; Sellers, TA; Folsom, AR. Relation of calcium, vitamin D, and dairy food intake to ischemic heart disease mortality among postmenopausal women. Am. J. Epidemiol 1999, 149, 151–161. [Google Scholar]
- Hagstrom, E; Hellman, P; Larsson, TE; Ingelsson, E; Berglund, L; Sundstrom, J; Melhus, H; Held, C; Lind, L; Michaelsson, K; Arnlov, J. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation 2009, 119, 2765–2771. [Google Scholar]
- Need, AG; O’Loughlin, PD; Morris, HA; Coates, PS; Horowitz, M; Nordin, BE. Vitamin D metabolites and calcium absorption in severe vitamin D deficiency. J. Bone Miner. Res 2008, 23, 1859–1863. [Google Scholar]
- Hansen, KE; Jones, AN; Lindstrom, MJ; Davis, LA; Engelke, JA; Shafer, MM. Vitamin D insufficiency: disease or no disease? J. Bone Miner. Res 2008, 23, 1052–1060. [Google Scholar]
- Chapuy, MC; Preziosi, P; Maamer, M; Arnaud, S; Galan, P; Hercberg, S; Meunier, PJ. Prevalence of vitamin D insufficiency in an adult normal population. Osteoporos. Int 1997, 7, 439–443. [Google Scholar]
- Whiting, SJ; Calvo, MS. Dietary recommendations for vitamin D: a critical need for functional end points to establish an estimated average requirement. J. Nutr 2005, 135, 304–309. [Google Scholar]
- Bischoff-Ferrari, HA; Giovannucci, E; Willett, WC; Dietrich, T; Dawson-Hughes, B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am. J. Clin. Nutr 2006, 84, 18–28. [Google Scholar]
- Holick, MF. Vitamin D: A millenium perspective. J. Cell. Biochem 2003, 88, 296–307. [Google Scholar]
- Ovesen, L; Andersen, R; Jakobsen, J. Geographical differences in vitamin D status, with particular reference to European countries. Proc. Nutr. Soc 2003, 62, 813–821. [Google Scholar]
- Kuchuk, NO; van Schoor, NM; Pluijm, SM; Chines, A; Lips, P. Vitamin D status, parathyroid function, bone turnover, and BMD in postmenopausal women with osteoporosis: global perspective. J. Bone Miner. Res 2009, 24, 693–701. [Google Scholar]
- Kudlacek, S; Schneider, B; Peterlik, M; Leb, G; Klaushofer, K; Weber, K; Woloszczuk, W; Willvonseder, R. Assessment of vitamin D and calcium status in healthy adult Austrians. Eur. J. Clin. Invest 2003, 33, 323–331. [Google Scholar]
- Brot, C; Vestergaard, P; Kolthoff, N; Gram, J; Hermann, AP; Sorensen, OH. Vitamin D status and its adequacy in healthy Danish perimenopausal women: relationships to dietary intake, sun exposure and serum parathyroid hormone. Br. J. Nutr 2001, 86, S97–103. [Google Scholar]
- Hypponen, E; Power, C. Hypovitaminosis D in British adults at age 45 y: nationwide cohort study of dietary and lifestyle predictors. Am. J. Clin. Nutr 2007, 85, 860–868. [Google Scholar]
- Lamberg-Allardt, CJ; Outila, TA; Karkkainen, MU; Rita, HJ; Valsta, LM. Vitamin D deficiency and bone health in healthy adults in Finland: could this be a concern in other parts of Europe? J. Bone Miner. Res 2001, 16, 2066–2073. [Google Scholar]
- Islam, MZ; Lamberg-Allardt, C; Karkkainen, M; Outila, T; Salamatullah, Q; Shamim, AA. Vitamin D deficiency: a concern in premenopausal Bangladeshi women of two socio-economic groups in rural and urban region. Eur. J. Clin. Nutr 2002, 56, 51–56. [Google Scholar]
- McGrath, JJ; Kimlin, MG; Saha, S; Eyles, DW; Parisi, AV. Vitamin D insufficiency in south-east Queensland. Med. J. Aust 2001, 174, 150–151. [Google Scholar]
- Nakamura, K; Nashimoto, M; Matsuyama, S; Yamamoto, M. Low serum concentrations of 25-hydroxyvitamin D in young adult Japanese women: a cross sectional study. Nutrition 2001, 17, 921–925. [Google Scholar]
- Looker, AC; Pfeiffer, CM; Lacher, DA; Schleicher, RL; Picciano, MF; Yetley, EA. Serum 25-hydroxyvitamin D status of the US population: 1988–1994 compared with 2000–2004. Am. J. Clin. Nutr 2008, 88, 1519–1527. [Google Scholar]
- Rucker, D; Allan, JA; Fick, GH; Hanley, DA. Vitamin D insufficiency in a population of healthy western Canadians. CMAJ 2002, 166, 1517–1524. [Google Scholar]
- Pasco, JA; Henry, MJ; Nicholson, GC; Sanders, KM; Kotowicz, MA. Vitamin D status of women in the Geelong Osteoporosis Study: association with diet and casual exposure to sunlight. Med. J. Aust 2001, 175, 401–405. [Google Scholar]
- van der Mei, IA; Ponsonby, AL; Engelsen, O; Pasco, JA; McGrath, JJ; Eyles, DW; Blizzard, L; Dwyer, T; Lucas, R; Jones, G. The high prevalence of vitamin D insufficiency across Australian populations is only partly explained by season and latitude. Environ. Health Perspect 2007, 115, 1132–1139. [Google Scholar]
- Rockell, JE; Skeaff, CM; Williams, SM; Green, TJ. Serum 25-hydroxyvitamin D concentrations of New Zealanders aged 15 years and older. Osteoporos. Int 2006, 17, 1382–1389. [Google Scholar]
- Nelson, ML; Blum, JM; Hollis, BW; Rosen, C; Sullivan, SS. Supplements of 20 microg/d cholecalciferol optimized serum 25-hydroxyvitamin D concentrations in 80% of premenopausal women in winter. J. Nutr 2009, 139, 540–546. [Google Scholar]
- van der Wielen, RP; Lowik, MR; van den Berg, H; de Groot, LC; Haller, J; Moreiras, O; van Staveren, WA. Serum vitamin D concentrations among elderly people in Europe. Lancet 1995, 346, 207–210. [Google Scholar]
- Wicherts, IS; van Schoor, NM; Boeke, AJ; Visser, M; Deeg, DJ; Smit, J; Knol, DL; Lips, P. Vitamin D status predicts physical performance and its decline in older persons. J. Clin. Endocrinol. Metab 2007, 92, 2058–2065. [Google Scholar]
- Neuprez, A; Bruyere, O; Collette, J; Reginster, JY. Vitamin D inadequacy in Belgian postmenopausal osteoporotic women. BMC Public Health 2007, 7, 1–9. [Google Scholar]
- Andersen, R; Molgaard, C; Skovgaard, LT; Brot, C; Cashman, KD; Chabros, E; Charzewska, J; Flynn, A; Jakobsen, J; Karkkainen, M; Kiely, M; Lamberg-Allardt, C; Moreiras, O; Natri, AM; O’Brien, M; Rogalska-Niedzwiedz, M; Ovesen, L. Teenage girls and elderly women living in northern Europe have low winter vitamin D status. Eur. J. Clin. Nutr 2005, 59, 533–541. [Google Scholar]
- Lappe, JM; Davies, KM; Travers-Gustafson, D; Heaney, RP. Vitamin D status in a rural postmenopausal female population. J. Am. Coll. Nutr 2006, 25, 395–402. [Google Scholar]
- Flicker, L; Mead, K; MacInnis, RJ; Nowson, C; Scherer, S; Stein, MS; Thomasx, J; Hopper, JL; Wark, JD. Serum vitamin D and falls in older women in residential care in Australia. J. Am. Geriatr. Soc 2003, 51, 1533–1538. [Google Scholar]
- Nakamura, K; Nashimoto, M; Hori, Y; Yamamoto, M. Serum 25-hydroxyvitamin D concentrations and related dietary factors in peri- and postmenopausal Japanese women. Am. J. Clin. Nutr 2000, 71, 1161–1165. [Google Scholar]
- Hintzpeter, B; Scheidt-Nave, C; Muller, MJ; Schenk, L; Mensink, GB. Higher prevalence of vitamin D deficiency is associated with immigrant background among children and adolescents in Germany. J. Nutr 2008, 138, 1482–1490. [Google Scholar]
- Guillemant, J; Le, HT; Maria, A; Allemandou, A; Peres, G; Guillemant, S. Wintertime vitamin D deficiency in male adolescents: effect on parathyroid function and response to vitamin D3 supplements. Osteoporos. Int 2001, 12, 875–879. [Google Scholar]
- Vieth, R; Cole, DE; Hawker, GA; Trang, HM; Rubin, LA. Wintertime vitamin D insufficiency is common in young Canadian women, and their vitamin D intake does not prevent it. Eur. J. Clin. Nutr 2001, 55, 1091–1097. [Google Scholar]
- Green, TJ; Skeaff, CM; Rockell, JE; Venn, BJ; Lambert, A; Todd, J; Khor, GL; Loh, SP; Muslimatun, S; Agustina, R; Whiting, SJ. Vitamin D status and its association with parathyroid hormone concentrations in women of child-bearing age living in Jakarta and Kuala Lumpur. Eur. J. Clin. Nutr 2008, 62, 373–378. [Google Scholar]
- Puri, S; Marwaha, RK; Agarwal, N; Tandon, N; Agarwal, R; Grewal, K; Reddy, DH; Singh, S. Vitamin D status of apparently healthy schoolgirls from two different socioeconomic strata in Delhi: relation to nutrition and lifestyle. Br. J. Nutr 2008, 99, 876–882. [Google Scholar]
- Foo, LH; Zhang, Q; Zhu, K; Ma, G; Trube, A; Greenfield, H; Fraser, DR. Relationship between vitamin D status, body composition and physical exercise of adolescent girls in Beijing. Osteoporos. Int 2009, 20, 417–425. [Google Scholar]
- Woo, J; Lam, CW; Leung, J; Lau, WY; Lau, E; Ling, X; Xing, X; Zhao, XH; Skeaff, CM; Bacon, CJ; Rockell, JE; Lambert, A; Whiting, SJ; Green, TJ. Very high rates of vitamin D insufficiency in women of child-bearing age living in Beijing and Hong Kong. Br. J. Nutr 2008, 99, 1330–1334. [Google Scholar]
- Hollis, BW; Wagner, CL. Vitamin D deficiency during pregnancy: an ongoing epidemic. Am. J. Clin. Nutr 2006, 84, 273. [Google Scholar]
- Hypponen, E; Laara, E; Reunanen, A; Jarvelin, MR; Virtanen, SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet 2001, 358, 1500–1503. [Google Scholar]
- Hypponen, E; Power, C. Vitamin D status and glucose homeostasis in the 1958 British birth cohort: the role of obesity. Diabetes Care 2006, 29, 2244–2246. [Google Scholar]
- Wortsman, J; Matsuoka, LY; Chen, TC; Lu, Z; Holick, MF. Decreased bioavailability of vitamin D in obesity. Am. J. Clin. Nutr 2000, 72, 690–693. [Google Scholar]
- Andersen, R; Molgaard, C; Skovgaard, LT; Brot, C; Cashman, KD; Jakobsen, J; Lamberg-Allardt, C; Ovesen, L. Pakistani immigrant children and adults in Denmark have severely low vitamin D status. Eur. J. Clin. Nutr 2008, 62, 625–634. [Google Scholar]
- Meyer, HE; Falch, JA; Sogaard, AJ; Haug, E. Vitamin D deficiency and secondary hyperparathyroidism and the association with bone mineral density in persons with Pakistani and Norwegian background living in Oslo, Norway, The Oslo Health Study. Bone 2004, 35, 412–417. [Google Scholar]
- Roy, DK; Berry, JL; Pye, SR; Adams, JE; Swarbrick, CM; King, Y; Silman, AJ; O’Neill, TW. Vitamin D status and bone mass in UK South Asian women. Bone 2007, 40, 200–204. [Google Scholar]
- Nowson, CA; Margerison, C. Vitamin D intake and vitamin D status of Australians. Med. J. Aust 2002, 177, 149–152. [Google Scholar]
- Anonymous. Human vitamin and mineral requirements Report of a Joint FAO/WHO Expert Consultation. Food and Agriculture Organization, Rome, Italy, 2002.
- Anke, M. Lebensnotwendigkeit, Toxizität, Versorgung, scheinbare Absorption und Bilanz der Alkali- und Erdalkalimetalle beim Menschen. Sitzungsberichte der Mathematisch-Naturwissenschaftlichen Klasse der Akademie gemeinnütziger Wissenschaften 2006, 13, 9–58. [Google Scholar]
- Schulze, MB; Linseisen, J; Kroke, A; Boeing, H. Macronutrient, vitamin, and mineral intakes in the EPIC-Germany cohorts. Ann. Nutr. Metab 2001, 45, 181–189. [Google Scholar]
- Vyas, A; Greenhalgh, A; Cade, J; Sanghera, B; Riste, L; Sharma, S; Cruickshank, K. Nutrient intakes of an adult Pakistani, European and African-Caribbean community in inner city Britain. J. Hum. Nutr. Diet 2003, 16, 327–337. [Google Scholar]
- Ma, J; Johns, RA; Stafford, RS. Americans are not meeting current calcium recommendations. Am. J. Clin. Nutr 2007, 85, 1361–1366. [Google Scholar]
- Pasco, JA; Henry, MJ; Nicholson, GC; Brennan, SL; Kotowicz, MA. Behavioural and physical characteristics associated with vitamin D status in women. Bone 2009, 44, 1085–1091. [Google Scholar]
- Metcalf, PA; Scragg, RK; Tukuitonga, CF; Dryson, EW. Dietary intakes of middle-aged European, Maori and Pacific Islands people living in New Zealand. N. Z. Med. J 1998, 111, 310–313. [Google Scholar]
- Islam, MZ; Lamberg-Allardt, C; Karkkainen, M; Ali, SM. Dietary calcium intake in premenopausal Bangladeshi women: do socio-economic or physiological factors play a role? Eur. J. Clin. Nutr 2003, 57, 674–680. [Google Scholar]
- Whedon, GD. Recent advances in management of osteoporosis. In Phosphate and Mineral in Health and Disease; Massry, SG, Ritz, E, Jahn, H, Eds.; Plenum Press: New York, USA, 1980; pp. 600–613. [Google Scholar]
- Amorim Cruz, JA; Moreiras, O; Brzozowska, A. Longitudinal changes in the intake of vitamins and minerals of elderly Europeans. SENECA Investigators. Eur. J. Clin. Nutr 1996, 50, S77–85. [Google Scholar]
- Nakamura, K; Saito, T; Yoshihara, A; Ishikawa, M; Tsuchiya, Y; Oshiki, R; Kobayashi, R; Maruyama, K; Hyodo, K; Nashimoto, M; Tsugawa, N; Okano, T; Oyama, M; Yamamoto, M. Low calcium intake is associated with increased bone resorption in postmenopausal Japanese women: Yokogoshi Study. Public Health Nutr, 2009; Epub ahead of print, doi:10.1017/S1368980009005084. [Google Scholar]
- Abrams, SA; Griffin, IJ; Hawthorne, KM; Gunn, SK; Gundberg, CM; Carpenter, TO. Relationships among vitamin D levels, parathyroid hormone, and calcium absorption in young adolescents. J. Clin. Endocrinol. Metab 2005, 90, 5576–5581. [Google Scholar]
- Rubin, LA; Hawker, GA; Peltekova, VD; Fielding, LJ; Ridout, R; Cole, DE. Determinants of peak bone mass: clinical and genetic analyses in a young female Canadian cohort. J. Bone Miner. Res 1999, 14, 633–643. [Google Scholar]
- Pettifor, JM. Nutritional rickets: deficiency of vitamin D, calcium, or both? Am. J. Clin. Nutr 2004, 80, 1725S–1729S. [Google Scholar]
- Thacher, TD; Fischer, PR; Pettifor, JM; Lawson, JO; Isichei, CO; Reading, JC; Chan, GM. A comparison of calcium, vitamin D, or both for nutritional rickets in Nigerian children. N. Engl. J. Med 1999, 341, 563–568. [Google Scholar]
- Graff, M; Thacher, TD; Fischer, PR; Stadler, D; Pam, SD; Pettifor, JM; Isichei, CO; Abrams, SA. Calcium absorption in Nigerian children with rickets. Am. J. Clin. Nutr 2004, 80, 1415–1421. [Google Scholar]
- Balasubramanian, K; Rajeswari, J; Govil, YC; Agarwal, AK; Kumar, A; Bhatia, V. Varying role of vitamin D deficiency in the etiology of rickets in young children vs. adolescents in northern India. J. Trop. Pediatr 2003, 49, 201–206. [Google Scholar]
- Calvo, MS; Whiting, SJ; Barton, CN. Vitamin D intake: a global perspective of current status. J. Nutr 2005, 135, 310–316. [Google Scholar]
- Yetley, EA; Brule, D; Cheney, MC; Davis, CD; Esslinger, KA; Fischer, PW; Friedl, KE; Greene-Finestone, LS; Guenther, PM; Klurfeld, DM; L’Abbe, MR; McMurry, KY; Starke-Reed, PE; Trumbo, PR. Dietary reference intakes for vitamin D: justification for a review of the 1997 values. Am. J. Clin. Nutr 2009, 89, 719–727. [Google Scholar]
- Cashman, KD; Hill, TR; Lucey, AJ; Taylor, N; Seamans, KM; Muldowney, S; Fitzgerald, AP; Flynn, A; Barnes, MS; Horigan, G; Bonham, MP; Duffy, EM; Strain, JJ; Wallace, JM; Kiely, M. Estimation of the dietary requirement for vitamin D in healthy adults. Am. J. Clin. Nutr 2008, 88, 1535–1542. [Google Scholar]
- Cashman, KD; Wallace, JM; Horigan, G; Hill, TR; Barnes, MS; Lucey, AJ; Bonham, MP; Taylor, N; Duffy, EM; Seamans, K; Muldowney, S; Fitzgerald, AP; Flynn, A; Strain, JJ; Kiely, M. Estimation of the dietary requirement for vitamin D in free-living adults ≥64 y of age. Am. J. Clin. Nutr 2009, 89, 1366–1374. [Google Scholar]
- Viljakainen, HT; Vaisanen, M; Kemi, V; Rikkonen, T; Kroger, H; Laitinen, E; Rita, H; Lamberg-Allardt, C. Wintertime vitamin D supplementation inhibits seasonal variation of calcitropic hormones and maintains bone turnover in healthy men. J. Bone Miner. Res 2009, 24, 346–352. [Google Scholar]
- Gorham, ED; Garland, CF; Garland, FC; Grant, WB; Mohr, SB; Lipkin, M; Newmark, HL; Giovannucci, E; Wei, M; Holick, MF. Vitamin D and prevention of colorectal cancer. J. Steroid Biochem. Mol. Biol 2005, 97, 179–194. [Google Scholar]
- Garland, CF; Gorham, ED; Mohr, SB; Grant, WB; Giovannucci, EL; Lipkin, M; Newmark, H; Holick, MF; Garland, FC. Vitamin D and prevention of breast cancer: Pooled analysis. J. Steroid Biochem. Mol. Biol 2007, 103, 708–711. [Google Scholar]
- Weaver, CM. Age related calcium requirements due to changes in absorption and utilization. J. Nutr 1994, 124, 1418S–1425S. [Google Scholar]
- Tang, BM; Eslick, GD; Nowson, C; Smith, C; Bensoussan, A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet 2007, 370, 657–666. [Google Scholar]
- Shin, MH; Holmes, MD; Hankinson, SE; Wu, K; Colditz, GA; Willett, WC. Intake of dairy products, calcium, and vitamin D and risk of breast cancer. J. Natl. Cancer Inst 2002, 94, 1301–1311. [Google Scholar]
- Slattery, ML; Sorenson, AW; Ford, MH. Dietary calcium intake as a mitigating factor in colon cancer. Am. J. Epidemiol 1988, 128, 504–514. [Google Scholar]
- Berube, S; Diorio, C; Verhoek-Oftedahl, W; Brisson, J. Vitamin D, calcium, and mammographic breast densities. Cancer Epidemiol. Biomarkers Prev 2004, 13, 1466–1472. [Google Scholar]
- Boonen, S; Lips, P; Bouillon, R; Bischoff-Ferrari, HA; Vanderschueren, D; Haentjens, P. Need for additional calcium to reduce the risk of hip fracture with vitamin D supplementation: evidence from a comparative metaanalysis of randomized controlled trials. J. Clin. Endocrinol. Metab 2007, 92, 1415–1423. [Google Scholar]
- Lappe, JM; Travers-Gustafson, D; Davies, KM; Recker, RR; Heaney, RP. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am. J. Clin. Nutr 2007, 85, 1586–1591. [Google Scholar]
- Cho, E; Smith-Warner, SA; Spiegelman, D; Beeson, WL; van den Brandt, PA; Colditz, GA; Folsom, AR; Fraser, GE; Freudenheim, JL; Giovannucci, E; Goldbohm, RA; Graham, S; Miller, AB; Pietinen, P; Potter, JD; Rohan, TE; Terry, P; Toniolo, P; Virtanen, MJ; Willett, WC; Wolk, A; Wu, K; Yaun, SS; Zeleniuch-Jacquotte, A; Hunter, DJ. Dairy foods, calcium, and colorectal cancer: a pooled analysis of 10 cohort studies. J. Natl. Cancer Inst 2004, 96, 1015–1022. [Google Scholar]
- Ishihara, J; Inoue, M; Iwasaki, M; Sasazuki, S; Tsugane, S. Dietary calcium, vitamin D, and the risk of colorectal cancer. Am. J. Clin. Nutr 2008, 88, 1576–1583. [Google Scholar]
- Berube, S; Diorio, C; Masse, B; Hebert-Croteau, N; Byrne, C; Cote, G; Pollak, M; Yaffe, M; Brisson, J. Vitamin D and calcium intakes from food or supplements and mammographic breast density. Cancer Epidemiol. Biomarkers Prev 2005, 14, 1653–1659. [Google Scholar]
- Lamberg-Allardt, C. Vitamin D in foods and as supplements. Prog. Biophys. Mol. Biol 2006, 92, 33–38. [Google Scholar]
- O’Donnell, S; Cranney, A; Horsley, T; Weiler, HA; Atkinson, SA; Hanley, DA; Ooi, DS; Ward, L; Barrowman, N; Fang, M; Sampson, M; Tsertsvadze, A; Yazdi, F. Efficacy of food fortification on serum 25-hydroxyvitamin D concentrations: systematic review. Am. J. Clin. Nutr 2008, 88, 1528–1534. [Google Scholar]
- Natri, AM; Salo, P; Vikstedt, T; Palssa, A; Huttunen, M; Karkkainen, MU; Salovaara, H; Piironen, V; Jakobsen, J; Lamberg-Allardt, CJ. Bread fortified with cholecalciferol increases the serum 25-hydroxyvitamin D concentration in women as effectively as a cholecalciferol supplement. J. Nutr 2006, 136, 123–127. [Google Scholar]
- Mocanu, V; Stitt, PA; Costan, AR; Voroniuc, O; Zbranca, E; Luca, V; Vieth, R. Long-term effects of giving nursing home residents bread fortified with 125 microg (5000 IU) vitamin D3 per daily serving. Am. J. Clin. Nutr 2009, 89, 1132–1137. [Google Scholar]
- Newmark, HL; Heaney, RP; Lachance, PA. Should calcium and vitamin D be added to the current enrichment program for cereal-grain products? Am. J. Clin. Nutr 2004, 80, 264–270. [Google Scholar]
- Daly, RM; Bass, S; Nowson, C. Long-term effects of calcium-vitamin-D3-fortified milk on bone geometry and strength in older men. Bone 2006, 39, 946–953. [Google Scholar]
- Daly, RM; Brown, M; Bass, S; Kukuljan, S; Nowson, C. Calcium- and vitamin D3-fortified milk reduces bone loss at clinically relevant skeletal sites in older men: a 2-year randomized controlled trial. J. Bone Miner. Res 2006, 21, 397–405. [Google Scholar]

| Vitamin D insufficiency | Nutritional Calcium deficit |
|---|---|
| A. Convincing evidence from multiple epidemiological (prospective, cross-sectional, retrospective) large cohort studies, interventional trials and experimental studies | |
| Osteoporosis | Osteoporosis |
| Cancer (colorectal, breast) | Cancer (colorectal, breast) |
| B. Good evidence from >3 observational studies and/or interventional trials | |
| Cancer (renal, prostate, endometrial, ovarian, esophageal, gastric, pancreatic, bladder) Hodgkin’s and non-Hodgkin’s lymphoma | Cancer (renal) |
| Cardiovascular disease | Cardiovascular disease |
| Hypertension | |
| Neuromuscular dysfunctions | Neuromuscular dysfunctions |
| Diabetes mellitus Type I | |
| Tuberculosis | |
| Gingivitis | |
| Periodontal disease, tooth loss | Periodontal disease, tooth loss |
| C. Emerging evidence from observational studies | |
| Hypertension | |
| Metabolic Syndrome | Metabolic Syndrome |
| Diabetes mellitus Type II | Diabetes mellitus Type II |
| D. Evidence mainly from studies with animal models of the respective human disease | |
| Inflammatory bowel disease | Inflammatory bowel disease |
| Multiple Sclerosis | Multiple Sclerosis |
| Country | Population segment | % Vitamin D insufficiency | Study | ||
|---|---|---|---|---|---|
| with upper reference limit at | |||||
| Age (yr) | Gender | 25–30 nM | 50 nM | ||
| Europe | |||||
| Austria | 19–79 | M + F | 26 | 60 | Kudlacek et al. [44] |
| Denmark | 45–58 | F | 7 | 40 | Brot et al. [45] |
| Finland | 31–43 | M + F | 27 | Lamberg–A. et al.[47] | |
| France | 35–65 | M + F | 14 | Chapuy et al. [38] | |
| Germany | 18–79 | M + F | 58 | Hintzpeter et al. [26] | |
| UK | 45 | M + F | 16 | 47 | Hypponen & Power [46] |
| North America | |||||
| Canada (Alberta) | 27–89 | M + F | 61 | Rucker et al. [52] | |
| USA | 20–49 | M + F | 5 | 32 | Looker et al. [51] |
| South–East Asia | |||||
| Bangladesh | 16–40 | F | 12 – 17 | Islam et al. [48] | |
| Japan | 30–66 | F | 10 | Nakamura et al. [50] | |
| South Pacific | |||||
| Australia (Queensland) | 17–65 | M + F | 8 a) | 23 | McGrath et al. [49] |
| Australia (Queensland) | <60 | M + F | 40 | Van der Mei et al. [54] | |
| Australia (Victoria) | 20–92 | F | 11 | 43 | Pasco et al. [53] |
| Australia (Geelong) | <60 | F | 37 | Van der Mei et al. [54] | |
| Australia (Tasmania) | <60 | M + F | 67 | Van der Mei et al. [54] | |
| New Zealand | 15–65+ | M + F | 48 | Rockell et al. [55] | |
| Country | Population segment | % Vitamin D insufficiency | Study | ||
|---|---|---|---|---|---|
| with upper reference limit of serum 25-(OH)D at | |||||
| Age (yr) | Gender | 25–30 nM | 50 nM | ||
| Europe | |||||
| Belgium | 76.5 ± 7.5 | F | 16 | 43 | Neuprez et al. [59] |
| Denmark | 70–75 | F | 17 | 55 | Andersen et al. [60] |
| Finland | 70–75 | F | 10 | 57 | Andersen et al. [60] |
| Ireland | 70–75 | F | 14 | 60 | Andersen et al. [60] |
| Italy | 75–80 | F | 92 | v. d. Wielen et al. [57] | |
| Netherlands | 65+ | M + F | 13 | 52 | Wicherts et al. [58] |
| Poland | 70–75 | F | 25 | 92 | Andersen et al. [60] |
| Switzerland | 75–80 | M | 12 | v. d. Wielen et al. [57] | |
| North America | |||||
| USA | 55+ | F | 4 | 14 | Lappe et al. [61] |
| USA | 50–70+ | M | 10 | 27 | Looker et al. [51] |
| East Asia | |||||
| Japan | 46–82 | F | 5 | 28 | Nakamura et al. [63] |
| South Pacific | |||||
| Australia | 75+ | F | 22 | Flicker et al. [62] | |
| New Zealand | 65+ | M | 41 | Rockell et al. [55] | |
| Country | Population segment | % Vitamin D insufficiency | Study | ||
|---|---|---|---|---|---|
| with upper reference limit of serum 25–(OH)D at | |||||
| Age (yrs) | Gender | 25–30 nM | 50 nM | ||
| Europe | |||||
| Germany | 3–17 | M | 18 | 62 | Hintzpeter et al.[64] |
| Germany | 3–17 | F | 18 | 65 | Hintzpeter et al. [64] |
| France | 13–16 | M | 78 | Guillemant et al. [65] | |
| Denmark | 12–13 | F | 51 | 93 | Andersen et al. [60] |
| Finland | 12–13 | F | 37 | 97 | Andersen et al. [60] |
| Ireland | 12–13 | F | 26 | 89 | Andersen et al. [60] |
| Poland | 12–13 | F | 33 | 87 | Andersen et al. [60] |
| North America | |||||
| USA | 12–19 | M | 10 | 24 | Looker et al. [51] |
| USA | 12–19 | F | 16 | 31 | Looker et al. [51] |
| Canada | 18–35 | F | >15 | Vieth et al. [66] | |
| South–East Asia | |||||
| India (Delhi) | 6 – 18 | F | 91 | Puri et al. [68] | |
| China | 20–35 | F | 18–40 | >90 | Woo et al. [70] |
| China | 15 | F | 31 | 89 | Foo et al. [69] |
| Indonesia | 18–40 | F | 63 | Green et al. [67] | |
| Malaysia | 18–40 | F | 60 | Green et al. [67] | |
| Japan | 19–30 | F | 42 | Nakamura et al. [50] | |
| South Pacific | |||||
| New Zealand | 15–18 | M | 55 | Rockell et al. [55] | |
| New Zealand | 19–24 | F | 52 | Rockell et al. [55] | |
| Country | Age (yrs) | DRI a) (mg/day) | Calcium intake (mg/day) | Study | |
|---|---|---|---|---|---|
| Gender | |||||
| M | F | ||||
| Europe | |||||
| Austria | 19–79 | >1,000 | 561 (±290) b) | 576 (±309) b) | Kudlacek et al. [44] |
| <40 | 1,000 | 604 (±345) b) | 560 (±299) b) | Kudlacek et al. [44] | |
| 40–60 | >1,000 | 590 (±318) b) | 561 (±287) b) | Kudlacek et al. [44] | |
| Germany | 18–79 | >1,000 | 1,181 (902–1,535) | 1,082 (849–1,379) | Hintzpeter et al. [26] |
| Adults | 1,000 | 619 (213–1,025) | 705 (313–1,094) | Anke [80] | |
| 40–64 | >1,000 | 774 (334–1,330) c) | 707 (287–1,225) c) | Schulze et al. [81] | |
| UK f) | 45–55 | 1,000 | 1,133 (950–1,316) | 1,063 (931–1,195) | Vyas et al. [82] |
| North America | |||||
| USA | 19–50 | 1,000 | 812 (788–837) | 626 (596–659) | Ma et al. [83] |
| South–East Asia | |||||
| Bangladesh | 16 – 40 | 1,000 | 180 e) | Islam et al. [86] | |
| Indonesia | 18–40 | 1,000 | 270 (239–302) d) | Green et al. [67] | |
| Malaysia | 18–40 | 1,000 | 386 (353–420) d) | Green et al. [67] | |
| South Pacific | |||||
| Australia | 20–94 | >1,000 | 643 (±340) b) | Pasco et al. [84] | |
| M + F | |||||
| New Zealand | 40–64 | >1,000 | 794 (8–1,580) e) | Metcalf et al. [85] | |
| Country | Calcium intake (mg/day) | Study | |||
|---|---|---|---|---|---|
| Age (yrs) | DRI (mg/d)a) | M | F | ||
| Europe | |||||
| Austria | >60 | 1,300 | 503 (±221) b) | 569 (±287) b) | Kudlacek et al. [44] |
| Belgium | 75–80 | 1,300 | 748 (324–1,166) c) | 676 (287–1,101) c) | Amorim Cruz et al. [88] |
| Denmark | 70–75 | 1,300 | 544 (127–1,812)d) | Andersen et al. [60] | |
| Finland | 70–75 | 1,300 | 975 (404–2,313) d) | Andersen et al. [60] | |
| France | 75–80 | 1,300 | 620 (402–1,010) c) | 635 (428–944) c) | Amorim Cruz et al. [88] |
| Ireland | 70–75 | 1,300 | 824 (339–1,669) d) | Andersen et al. [60] | |
| Netherlands | 75–80 | 1,300 | 1,036 (725–1,447) c) | 1,010(612–1,616) c) | Amorim Cruz et al. [88] |
| Poland | 70–75 | 1,300 | 325 (86–851) d) | Andersen et al. [60] | |
| North America | |||||
| USA | >55 | 1,300 | 611 (381–892) c) | Lappe et al. [61] | |
| South–East Asia | |||||
| Japan | 65–75 | 1,300 | 527 (±195) b) | Nakamura et al. [89] | |
| Country | Calcium intake (mg/day) | Study | |||
|---|---|---|---|---|---|
| Gender | |||||
| Age (yr) | DRI a) (mg/day) | M + F | F | ||
| Europe | |||||
| Denmark | 12.6 | 1,300 | 831 (260–2,475) b) | Andersen et al. [60] | |
| Finland | 12.6 | 1,300 | 1,092 (546–2,452) b) | Andersen et al. [60] | |
| Ireland | 12.6 | 1,300 | 728 (54–2,259) b) | Andersen et al. [60] | |
| Poland | 12.6 | 1,300 | 524 (117–1,580) b) | Andersen et al. [60] | |
| North America | |||||
| USA | 12.7 | 1,300 | 906 (417–1,616) c) | Abrams et al. [90] | |
| Canada | 18–35 | 1,000 | 562 (0–2,630) c) | Rubin et al. [91] | |
| South-East Asia | |||||
| India (Delhi) | 6–18 | 700–1,300 | 575 (±219) d) | Puri et al. [68] | |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Peterlik, M.; Boonen, S.; Cross, H.S.; Lamberg-Allardt, C. Vitamin D and Calcium Insufficiency-Related Chronic Diseases: an Emerging World-Wide Public Health Problem. Int. J. Environ. Res. Public Health 2009, 6, 2585-2607. https://doi.org/10.3390/ijerph6102585
Peterlik M, Boonen S, Cross HS, Lamberg-Allardt C. Vitamin D and Calcium Insufficiency-Related Chronic Diseases: an Emerging World-Wide Public Health Problem. International Journal of Environmental Research and Public Health. 2009; 6(10):2585-2607. https://doi.org/10.3390/ijerph6102585
Chicago/Turabian StylePeterlik, Meinrad, Steven Boonen, Heide S. Cross, and Christel Lamberg-Allardt. 2009. "Vitamin D and Calcium Insufficiency-Related Chronic Diseases: an Emerging World-Wide Public Health Problem" International Journal of Environmental Research and Public Health 6, no. 10: 2585-2607. https://doi.org/10.3390/ijerph6102585
APA StylePeterlik, M., Boonen, S., Cross, H. S., & Lamberg-Allardt, C. (2009). Vitamin D and Calcium Insufficiency-Related Chronic Diseases: an Emerging World-Wide Public Health Problem. International Journal of Environmental Research and Public Health, 6(10), 2585-2607. https://doi.org/10.3390/ijerph6102585