Introduction
Water quality analysis is one of the most important aspects in groundwater studies. The hydro chemical study reveals quality of water that is suitable for drinking, agriculture and industrial purposes. Further, it is possible to understand the change in [
1,
2] quality due to rock-water interaction or any type of anthropogenic influence. Groundwater often consists of seven major chemical elements- Ca
+2, Mg
+2, Cl
−1, HCO
3−1 Na
+1, K
+1, and SO4
−2. The chemical parameters of groundwater play a significant role in classifying and assessing water quality. Considering the individual and paired ionic concentration, certain indices are proposed to find out the alkali hazards. Residual sodium carbonate (RSC) can be used as a criterion for finding the suitability of irrigation waters. It was observed that the criteria used in the classification of waters for a particular purpose considering the individual concentration may not find its suitability for other purposes and better results can be obtained only by considering the combined chemistry of all the ions rather than individual or paired ionic characters [
3–
5]. Chemical classification also throws light on the concentration of various predominant cations, anions and their interrelationships. A number of techniques and methods have been developed to interpret the chemical data. Zaporozee [
6] has summarized the various modes of data representation and has discussed their possible uses.
Presentation of chemical analysis in graphical form makes understanding of complex groundwater system simpler and quicker. Methods of representing the chemistry of water like Collin’s bar diagram [
5], radiating vectors of Maucha [
7], and parallel and horizontal axes of Stiff [
8], have been used in many parts of the world to show the proportion of ionic concentration in individual samples. Subramanian [
9] followed a series of methods to interpret and classify the chemistry of groundwater in hard rock, including coastal zones in the southern parts of India.
The objective of the present work is to discuss the major ion chemistry of groundwater of Tumkur Taluk. In this case the methods proposed by piper, Back and Hanshaw, Wilcox, Eaton, Todd [
10] and USSL (US Salinity Laboratory) classification have been used to study critically the hydrochemical characteristics of groundwater of Tumkur Taluk.
Results and Discussion
Maximum and minimum concentration of major ions present in the groundwater from the study area is presented in
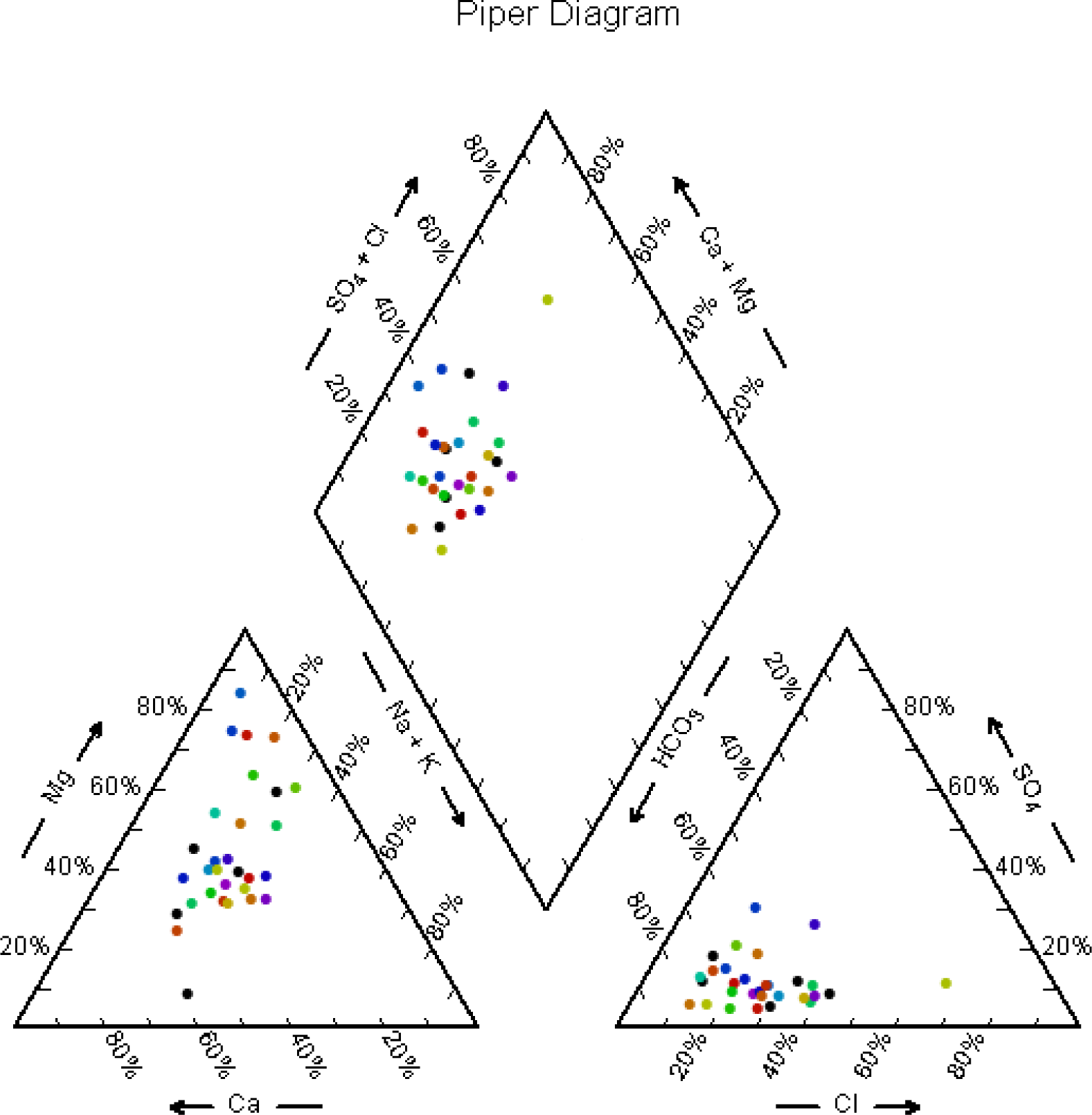
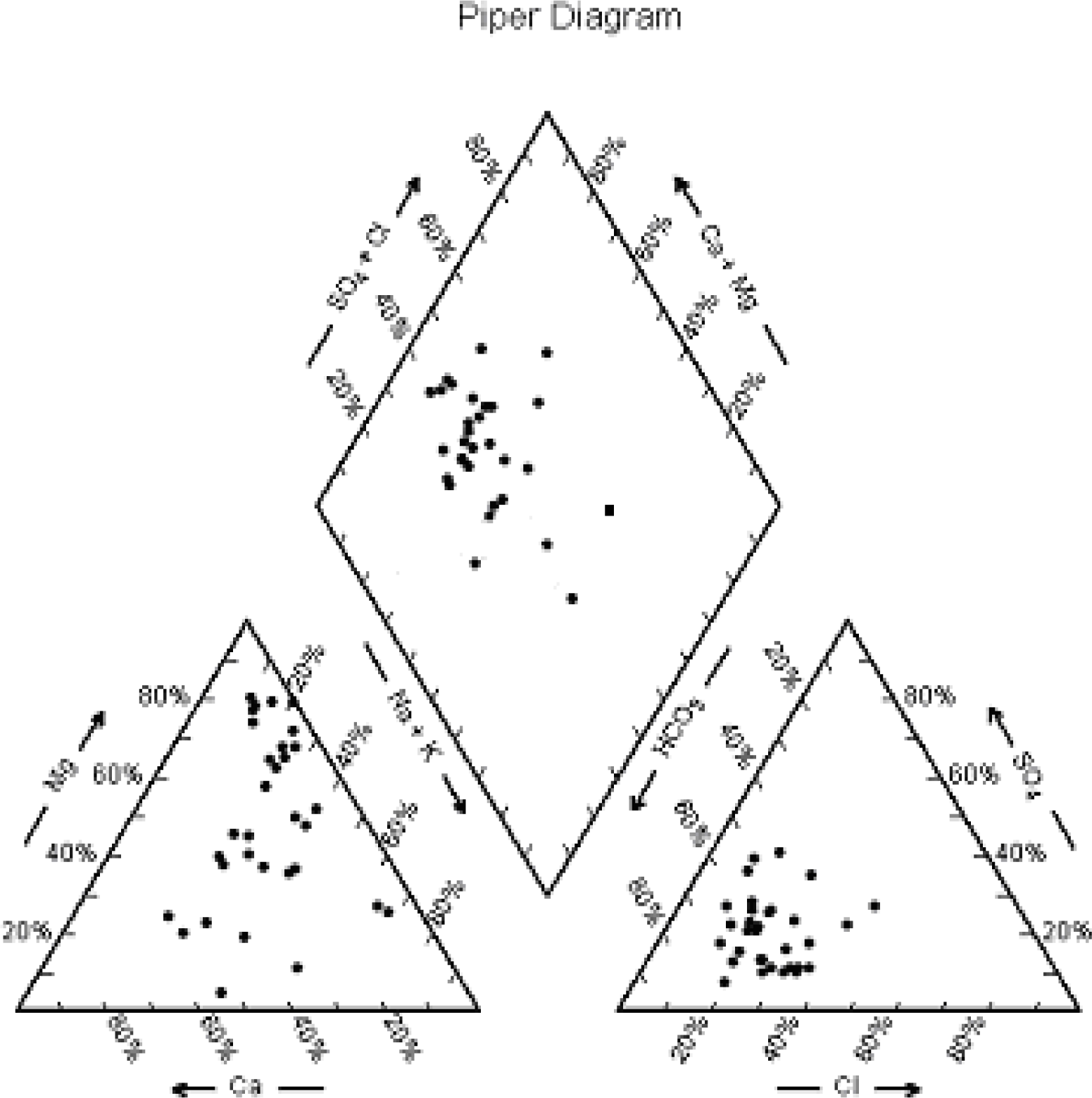
Table 1. The Piper-Hill diagram [
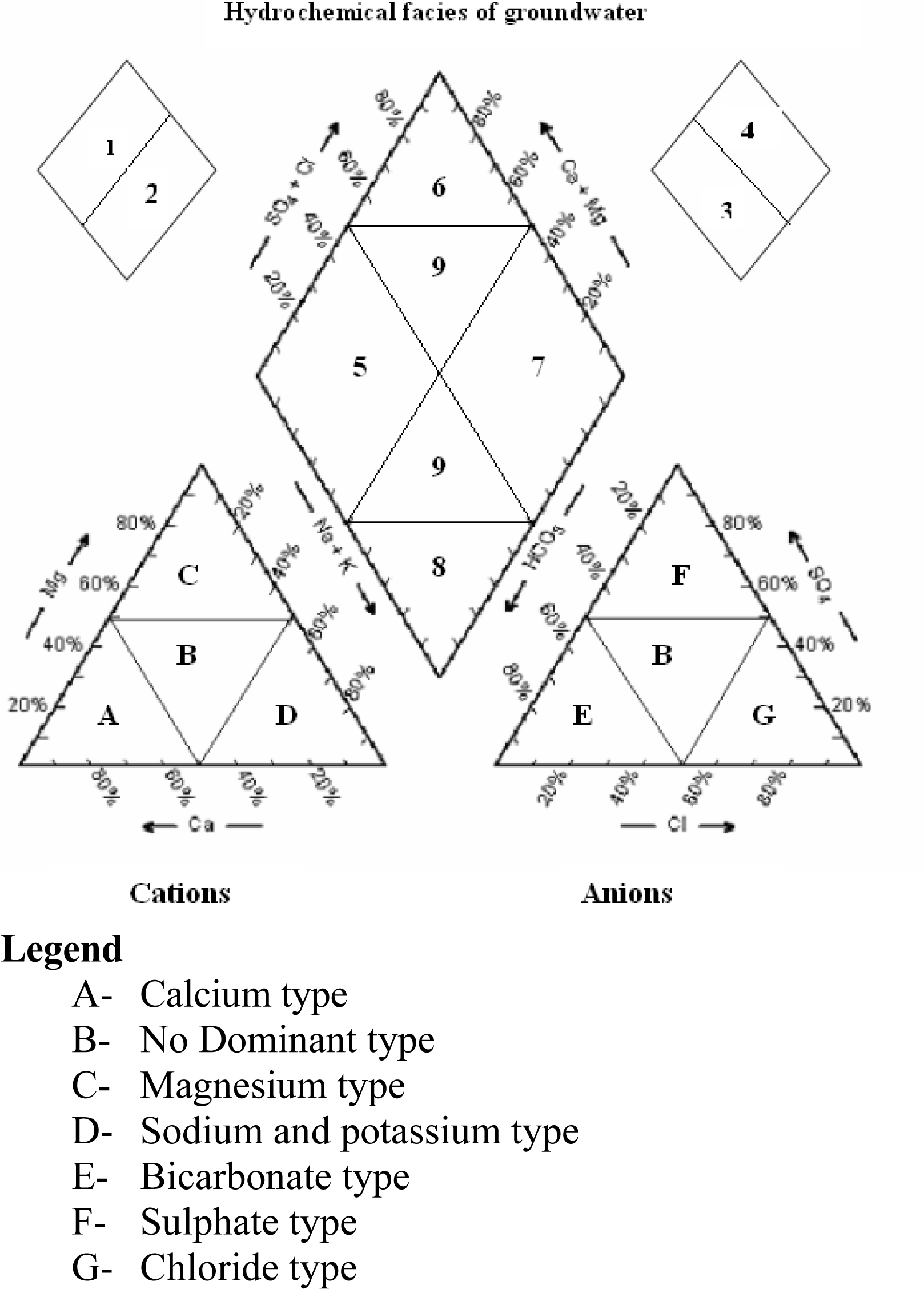
13] is used to infer hydro-geochemical facies. These plots include two triangles, one for plotting cations and the other for plotting anions. The cations and anion fields are combined to show a single point in a diamond-shaped field, from which inference is drawn on the basis of hydro-geochemical facies concept. These tri-linear diagrams are useful in bringing out chemical relationships among groundwater samples in more definite terms rather than with other possible plotting methods.
Chemical data of representative samples from the study area presented by plotting them on a Piper-tri-linear diagram for pre-and post-monsoon (
figures 3 and
4). These diagrams reveal the analogies, dissimilarities and different types of waters in the study area, which are identified and listed in
Table 2. The concept of hydrochemical facies was developed in order to understand and identify the water composition in different classes.
Facies are recognizable parts of different characters belonging to any genetically related system. Hydrochemical facies are distinct zones that possess action and anion concentration categories. To define composition class, Back and co-workers [
14] suggested subdivisions of the tri-linear diagram (
figure 5). The interpretation of distinct facies from the 0 to 10% and 90 to 100% domains on the diamond-shaped cation to anion graph is more helpful than using equal 25% increments. It clearly explains the variations or domination of cation and anion concentrations during pre-monsoon and post-monsoon. Ca-Mg-type of water predominated during pre-monsoon. The percentage of samples falling under Ca-Mg-type was 90 during pre-monsoon season. Similar type of water is predominated during post-monsoon also with 100 % water samples. For anion concentration, HCO
3-type of water predominated during pre-monsoon with 87.5% samples and during post-monsoon with 91% samples. There is no significant change in the hydro-chemical facies noticed during the study period (pre- and post-monsoon), which indicates that most of the major ions are natural in origin. The reason is groundwater passing through igneous rocks dissolves only small quantities of mineral matters because of the relative insolubility of the rock composition.
Water hardness is caused primarily by the presence of cations such as calcium and magnesium and anions such as carbonate, bicarbonate, chloride and sulfate in water. Water hardness has no known adverse effects; however, some evidence indicates its role in heart disease [
15]. Hard water is unsuitable for domestic use. In Tumkur region, the total hardness varies between 70 to 1060 ppm for the pre-monsoon (Jan–Feb 2006) period. For the post-monsoon period (Nov–Dec 2006), the value varies from 55 to 824 ppm. According to Sawyer and McCarty’s[
16] classification for hardness, 26 samples fall under moderately hard class and 242 samples fall under hard and very hard class for pre-monsoon water samples. The hardness classification is given in
Table 3. The suitability of groundwater for irrigation purposes depends upon its mineral constituents. The general criteria for judging the quality are: (i) Total salt concentration as measured by electrical conductivity (EC) (ii) Relative proportion of sodium to other principal cations as expressed by SAR, (iii) Bicarbonate (HCO
3−) and (iv) Boron.
Wilcox [
17] classified groundwater for irrigation purposes based on per cent sodium and Electrical conductivity. Eaton [
18] recommended the concentration of residual sodium carbonate to determine the suitability of water for irrigation purposes. The US Salinity Laboratory of the Department of Agriculture adopted certain techniques based on which the suitability of water for agriculture is explained.
The sodium in irrigation waters is usually denoted as per cent sodium and can be determined using the following formula [
17].
where the quantities of Ca
2+, Mg
2+ Na
+ and K
+ are expressed in milliequivalents per litre (epm).
The classification of groundwater samples with respect to per cent sodium is shown in
Table 4. It is observed that about 250 samples are excellent to good during pre-monsoon and 254 samples are excellent to good during post- monsoon. In waters having high concentration of bicarbonate, there is tendency for calcium and magnesium to precipitate as the water in the soil becomes more concentrated. As a result, the relative proportion of sodium in the water is increased in the form of sodium carbonate. RSC is calculated using the following equation.
where all ionic concentrations are expressed in epm.
According to the US Department of Agriculture, water having more than 2.5 epm of RSC is not suitable for irrigation purposes. Groundwater of the study area is classified on the basis of RSC and the results are presented in
Table 5 for both pre- and post-monsoon seasons. Based on RSC values, over 267 samples have values less than 1.25 and are safe for irrigation during pre-monsoon. During post-monsoon 272 samples were safe for irrigation. Only 2 samples in the pre-monsoon and 7 samples in the post-monsoon are fair.
The most important characteristics of irrigation water in determining its quality are: (i) Total concentration of soluble salts; ii) Relative proportion of sodium to other principal cations; (iii) Concentration of boron or other element that may be toxic, and (iv) Under some condition, bicarbonate concentration as related to the concentration of calcium plus magnesium. These have been termed as the salinity hazard [
17], sodium hazard, boron hazard and bicarbonate hazard. In the past, the sodium hazard has been expressed as per cent sodium of total cations. A better measure of the sodium hazard for irrigation is the SAR which is used to express reactions with the soil. SAR is computed as
where all ionic concentrations are expressed in epm
The classification of groundwater samples from the study area with respect to SAR is represented in
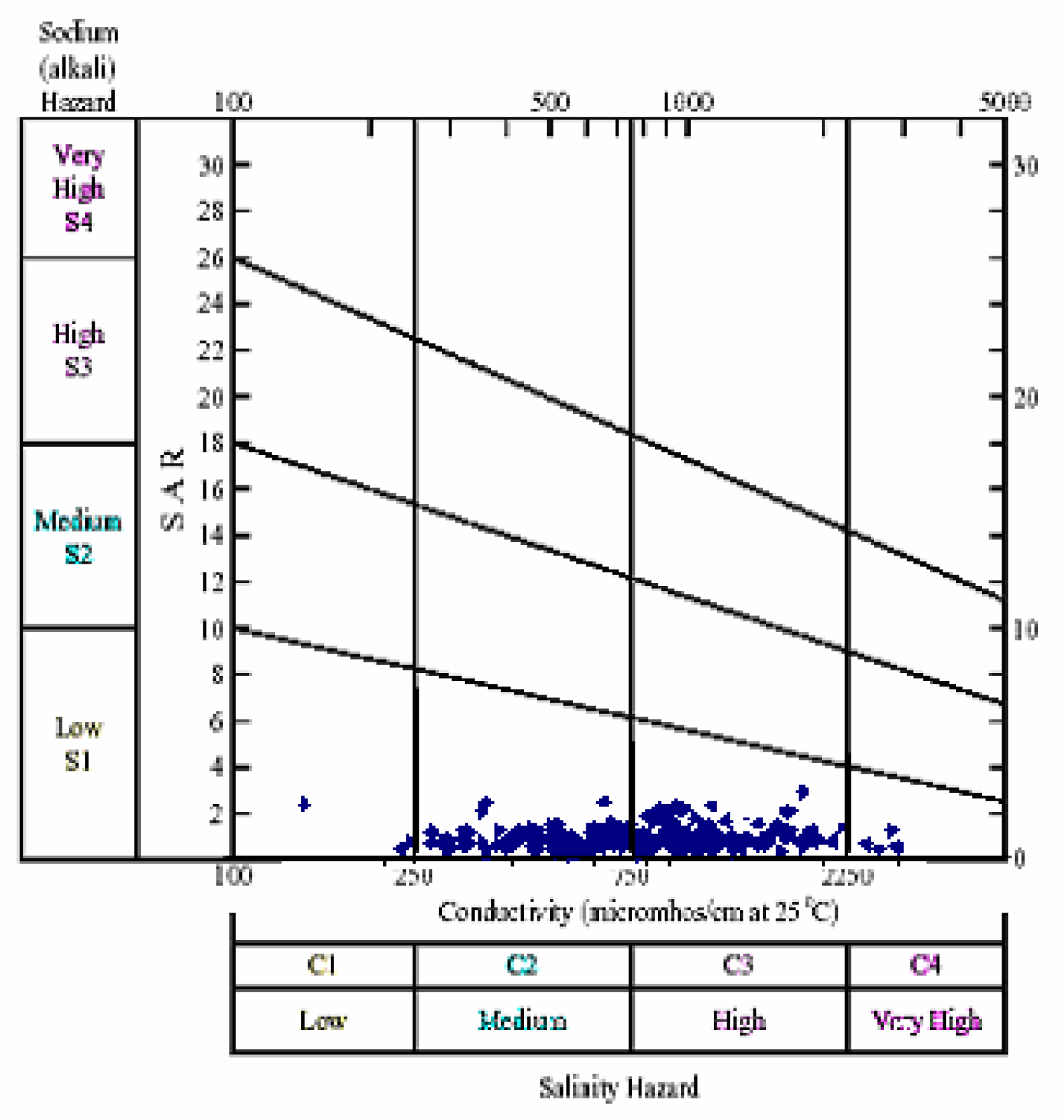
Table 6. During Pre- and post-monsoon, the SAR value of all the samples are found to be less than 10, and are classified as excellent for irrigation When the SAR and specific conductance of water are known, the classification of water for irrigation can be determined by graphically plotting these values on the US salinity (USSL) diagram (
figure 6 &
7). The groundwater of Tumkur taluk is in general Ca-Mg-HCO
3 type during both pre- and post monsoon seasons of the year 2006. About 98% of the samples are grouped within C2S1 and C3S1 classes in both pre- and post-monsoon (
figure 6 &
7).
For the purpose of diagnosis and classification, the total concentration of soluble salts (salinity hazard) in irrigation water can be expressed in terms of specific conductance. Classification of groundwater based on salinity hazard is presented in
table 7. It is found from the EC value, only 4 samples during pre-monsoon and 3 samples during post-monsoon were found to be unsuitable for irrigation purposes.
In Tumkur, Taluk, the groundwater is generally Ca-Mg-HCO3 type, which is mainly due to the geology of the area which comprises igneous rocks of crystalline nature, in which the major units are gneisses and granites. Ground water in the study area occurs under water table conditions in the weathered and fractured granite, Gneisses.