Effect of Arsenic and Chromium on the Serum Amino-Transferases Activity in Indian Major Carp, Labeo rohita
Abstract
:Introduction
Materials and Methods
Fish Collection and Maintenance
Determination of Median Lethal Concentration (96-h LC50)
Results and Discussion
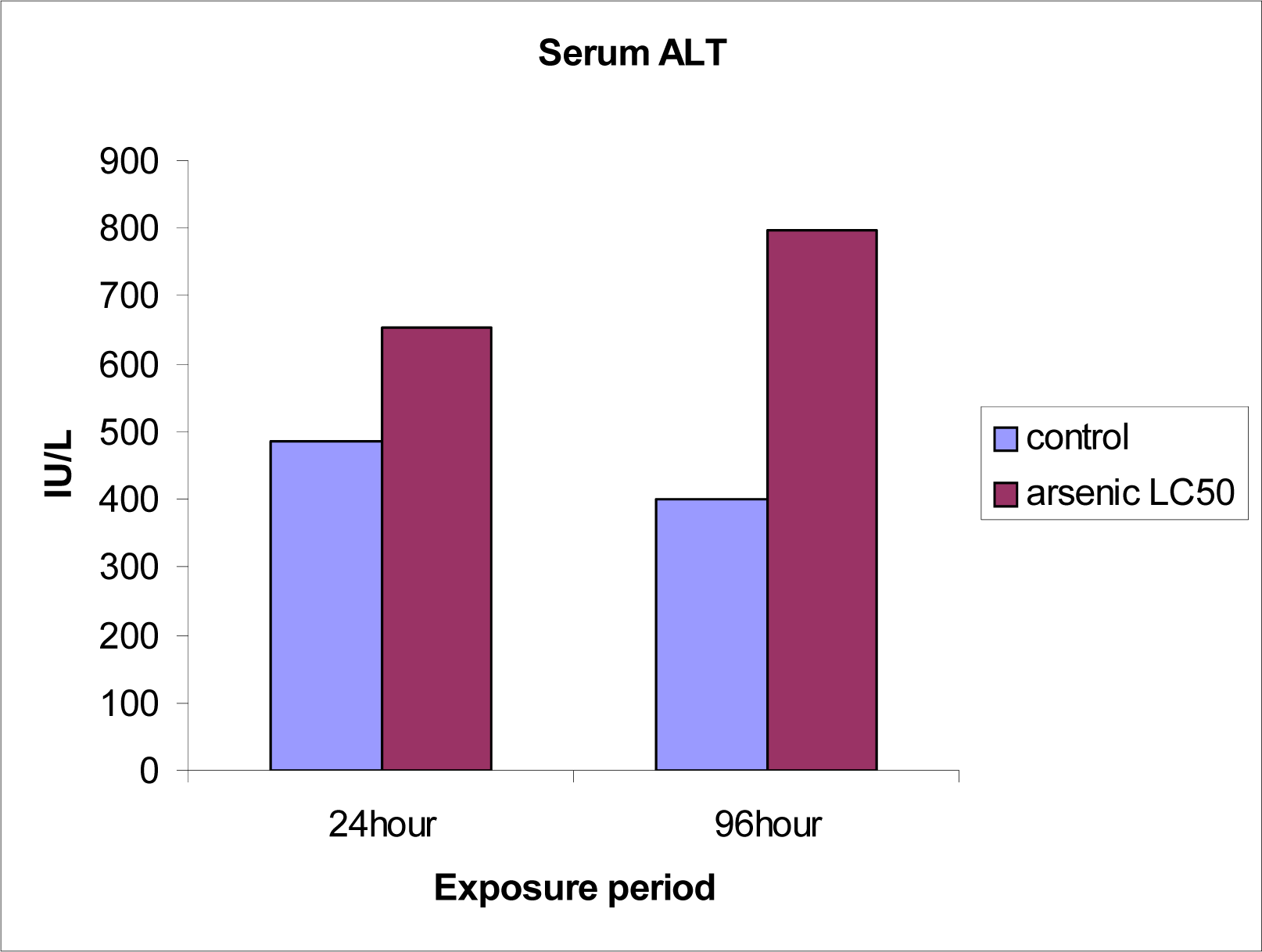
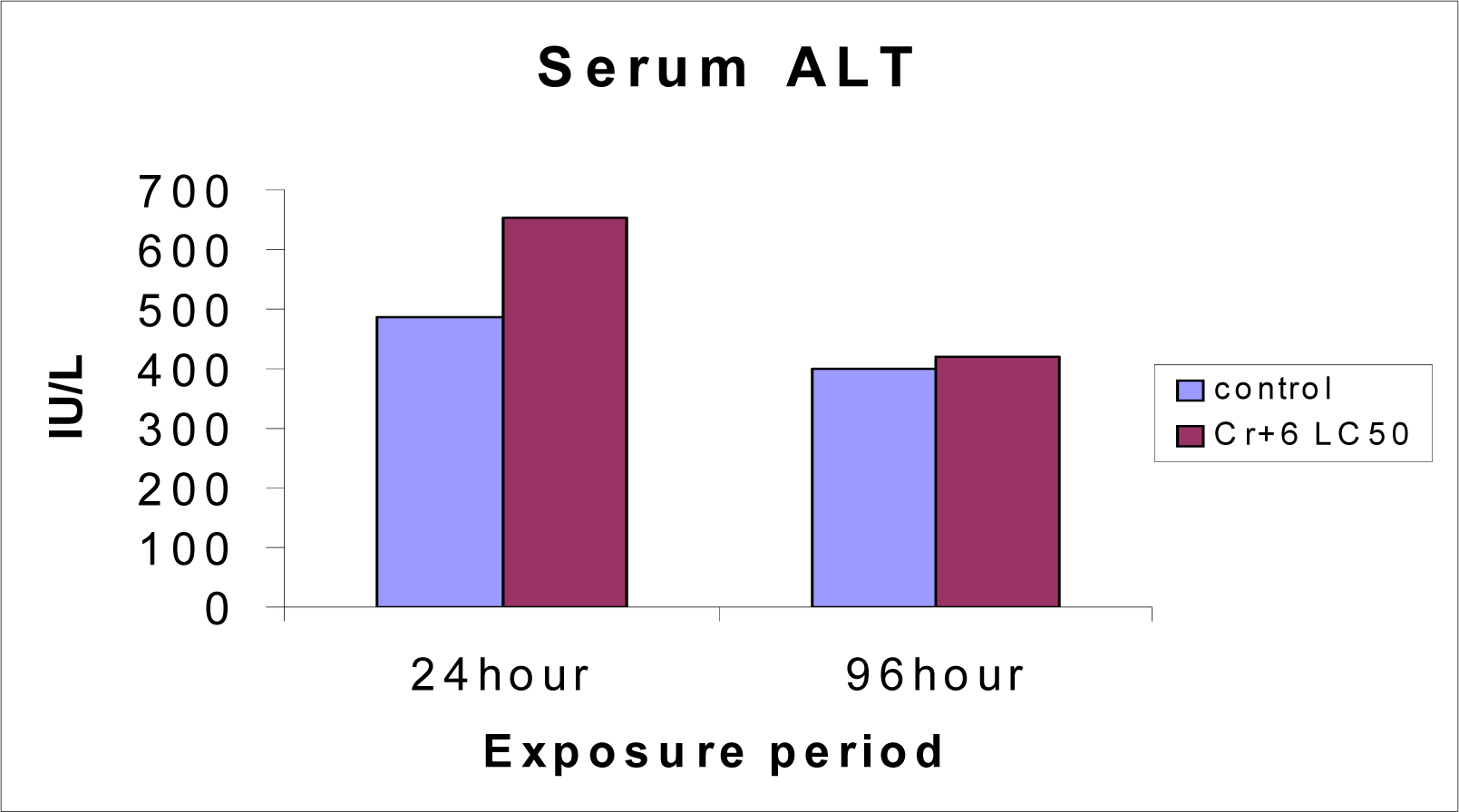
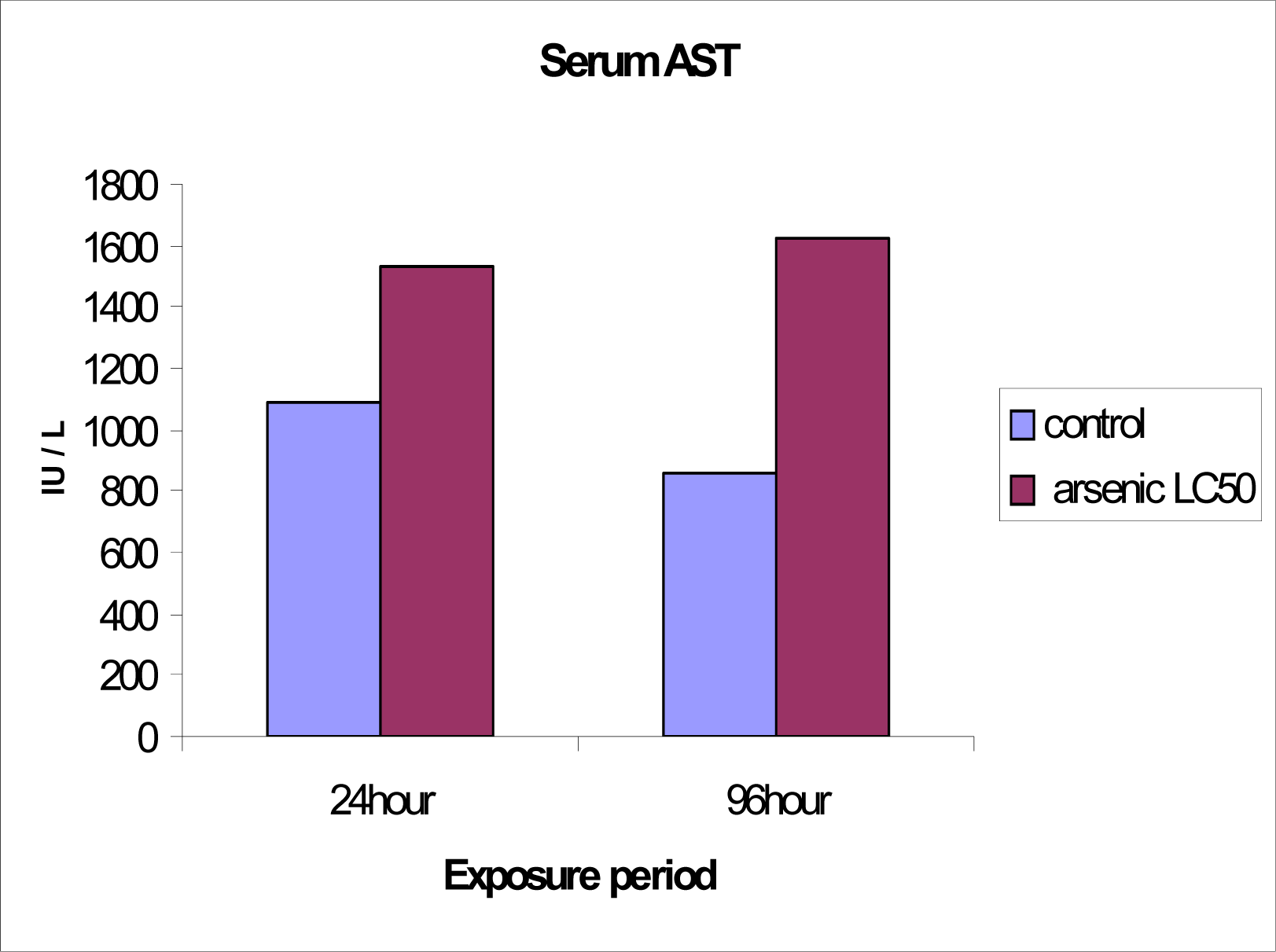
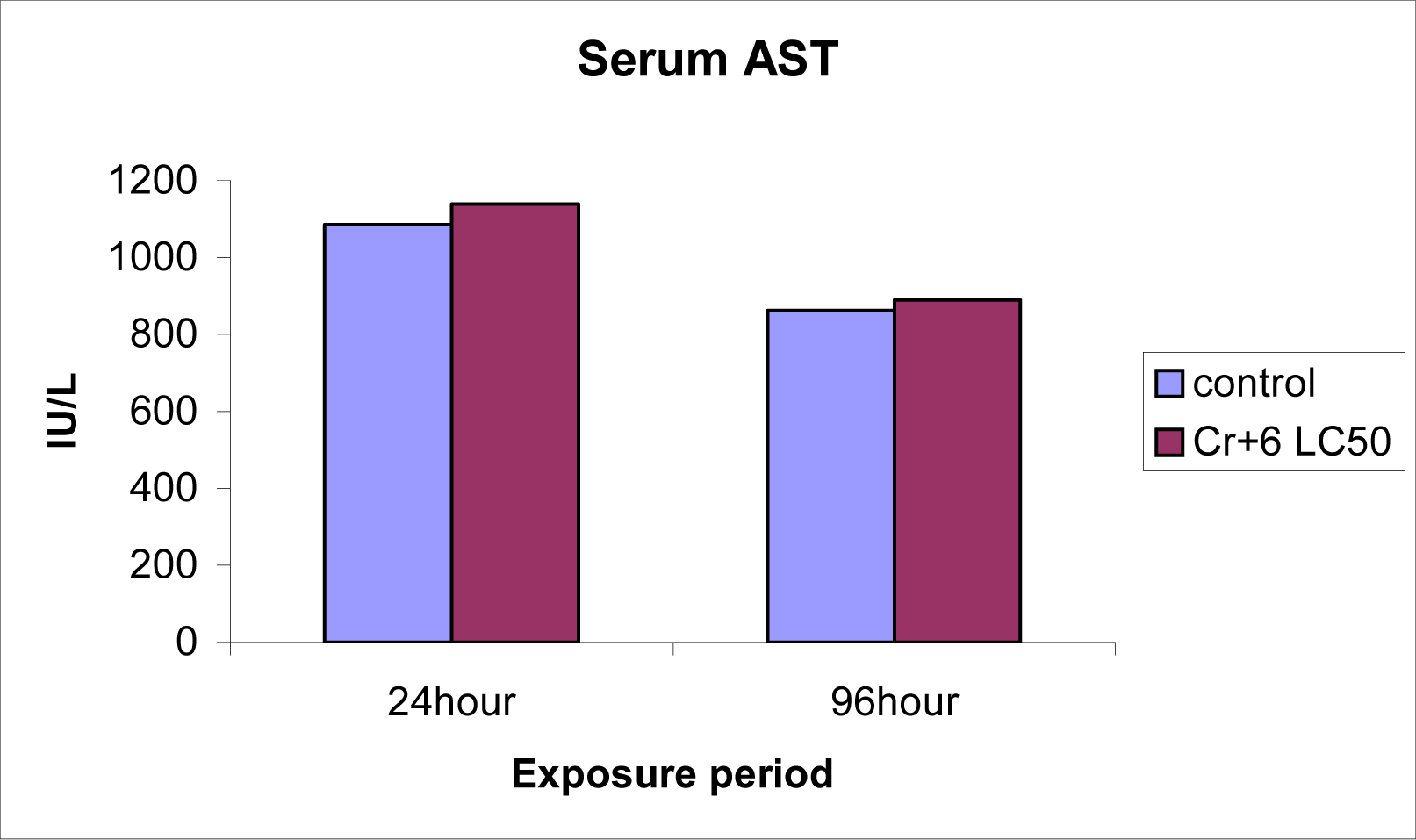
Serum ALT and AST levels
References
- Patlolla, Anita K; Tchounwou, Paul B. Serum Acetyl Cholinesterase as a Biomarker of Arsenic Induced Neurotoxicity in Sprague-Dawley Rats. Int. J. Environ. Res. Public Health 2005, 2(1), 80–83. [Google Scholar]
- Vutukuru, SS. Acute Effects of Hexavalent Chromium on Survival, Oxygen Consumption, Haematological Parameters and Some Biochemical Profiles of the Indian Major Carp. Labeo rohita Int. J. Environ. Res. Public Health 2005, 2(3), 456–462. [Google Scholar]
- Casillas, E; Meyers, M; Ames, W. Relationship of serum chemistry values to liver and kidney histopathology in English sole (Parophrys vetulus) after acute exposure to carbon tetrachloride. Aquatic Toxicology 1983, 3, 61–78. [Google Scholar]
- Escher, M; Wahli, T; Buttner, S; Meier, W; Burkhardt Holm, P. The effect of sewage plant effluent on brown trout (Salmo trutta Fario): a cage experiment. Aquatic Science 1999, 61, 93–110. [Google Scholar]
- Jacobson-Kram, D; Keller, KA. ‘Toxicology Testing Handbook’; Marcel Decker: New York, 2001. [Google Scholar]
- U.S.E.P.A. E.R.S Methods for acute toxicity tests with fish, Macro invertebrates and amphibians’. U.S.E.P.A., E.R.S, EPA 660/3-75-009 1975, 61. [Google Scholar]
- Finney, DJ. ‘Probit analysis’, 2nd edition; Cambridge University Press: England, 1953. [Google Scholar]
- Braunbeck, T. Sublethal and chronic effects of pollutants on freshwater fish; Oxford, UK; Blackwell, 1994. [Google Scholar]
- Burtis, CA; Ashwood, ER. The fundamentals of clinical chemistry; Saunders: Philadelphia, PA, 1996. [Google Scholar]
- Yang, Jen-Lee; Chen, Hon-Cheng. Serum Metabolic Enzyme Activities and Hepatocyte Ultrastructure of Common Carp after Gallium Exposure. Zoological Studies 2003, 42(3), 455–461. [Google Scholar]
- Markovich, D; James, KM. Heavy metals mercury, cadmium and chromium inhibit the activity of the mammalian liver and kidney sulphate transporter sat-1. Toxicol. Appl. Pharmacol 1999, 154, 181–187. [Google Scholar]
- Nimmi, Authur J. Review of Biochemical methods and other indicators to assess Fish health in aquatic ecosystem containing toxic chemicals. J. Great Lakes Res 1990, 16(4), 529–541. [Google Scholar]
| S.No | Toxicant | Regression Equation (Log +2) (Y = (ȳ–b x̄)+b x) | 96h LC50 (mg/L) | 95% Fiducial Limits (mg/L) |
|---|---|---|---|---|
| 1. | As2O3 | −20.12 +7.27x | 28.30 ± 0.23 | 26.1– 30.40 |
| Arsenic as | - | 21.10 | 19.6 – 22.6 | |
| 2. | K2Cr2O7 | −5.9091+1.77x | 61 ± 0.18 | 60.99 – 61.00 |
| Chromium as | - | 21.56 | 21.56–21.56 | |
© 2007 MDPI All rights reserved.
Share and Cite
Vutukuru, S.S.; Prabhath, N.A.; Raghavender, M.; Yerramilli, A. Effect of Arsenic and Chromium on the Serum Amino-Transferases Activity in Indian Major Carp, Labeo rohita. Int. J. Environ. Res. Public Health 2007, 4, 224-227. https://doi.org/10.3390/ijerph2007030005
Vutukuru SS, Prabhath NA, Raghavender M, Yerramilli A. Effect of Arsenic and Chromium on the Serum Amino-Transferases Activity in Indian Major Carp, Labeo rohita. International Journal of Environmental Research and Public Health. 2007; 4(3):224-227. https://doi.org/10.3390/ijerph2007030005
Chicago/Turabian StyleVutukuru, Sesha Srinivas, N. Arun Prabhath, M. Raghavender, and Anjaneyulu Yerramilli. 2007. "Effect of Arsenic and Chromium on the Serum Amino-Transferases Activity in Indian Major Carp, Labeo rohita" International Journal of Environmental Research and Public Health 4, no. 3: 224-227. https://doi.org/10.3390/ijerph2007030005
APA StyleVutukuru, S. S., Prabhath, N. A., Raghavender, M., & Yerramilli, A. (2007). Effect of Arsenic and Chromium on the Serum Amino-Transferases Activity in Indian Major Carp, Labeo rohita. International Journal of Environmental Research and Public Health, 4(3), 224-227. https://doi.org/10.3390/ijerph2007030005




